The Origin of Capsid-Derived Immune Complexes and Their Impact on HBV-Induced Liver Diseases
Abstract
1. Introduction
2. New Surprises in HBV Study
3. Immune Complexes in Viral Infections
4. Capsid-Derived Immune Complex: Revival of an Old Concept
5. Passive or Active Release of HBV Capsid?
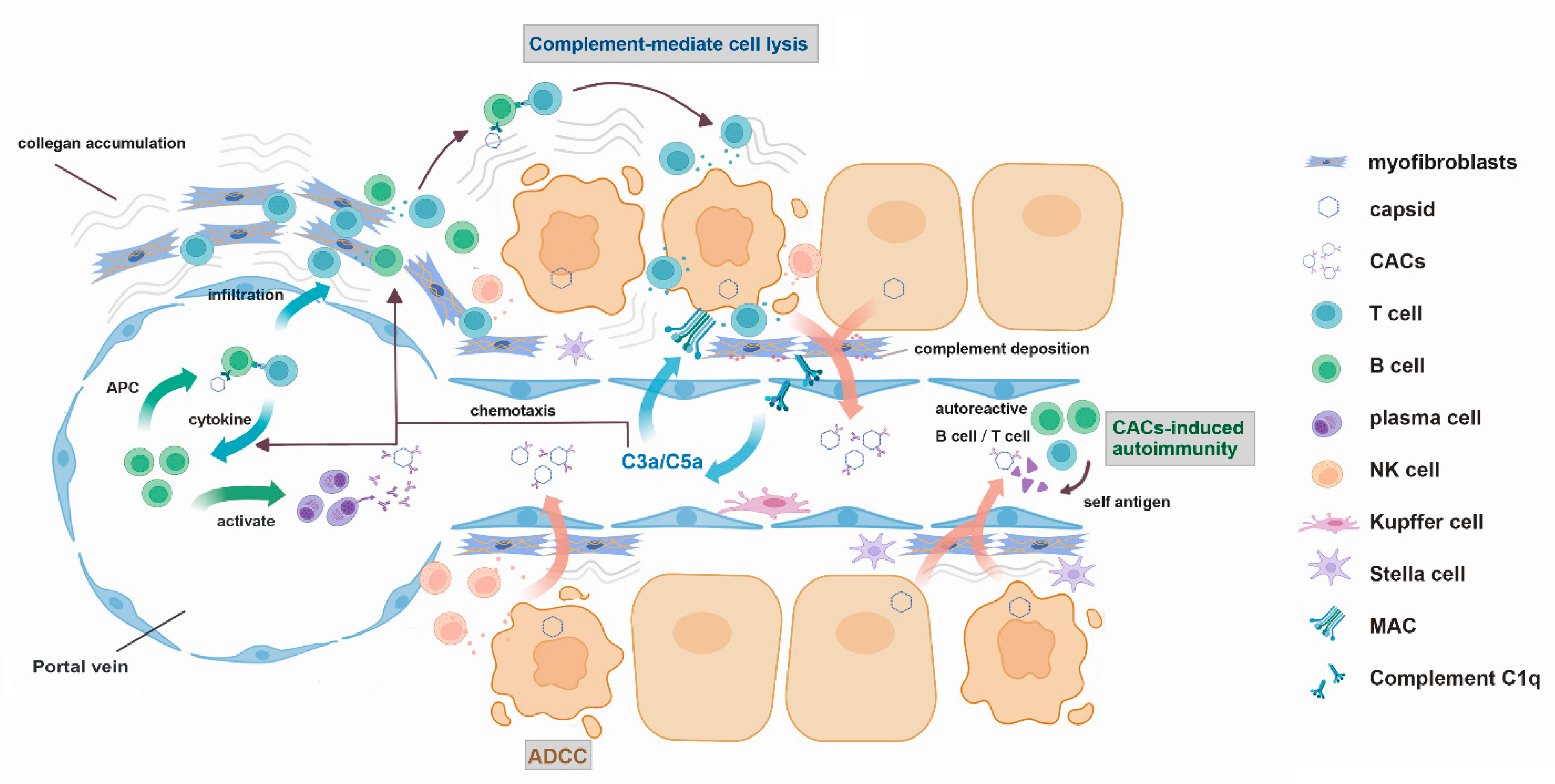
6. Possible Mechanism for CACs-Elicited Liver Pathology
6.1. Complement-Mediated Direct Lysis
6.2. Antibody-Dependent Cellular Cytotoxicity (ADCC)
6.3. Complement-Mediated Chemotaxis and Leukocyte Infiltration
6.4. Antigen Presentation Mediated by Core-Specific B Cells
6.5. Epitope Spreading and the Nurturing of Autoimmunity
7. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Sarin, S.; Kumar, M.; Lau, G.; Abbas, Z.; Chan, H.; Chen, C.; Chen, D.; Chen, H.; Chen, P.; Chien, R. Asian-Pacific clinical practice guidelines on the management of hepatitis B: A 2015 update. Hepatol. Int. 2016, 10, 1–98. [Google Scholar] [PubMed]
- Ott, J.J.; Stevens, G.A.; Groeger, J.; Wiersma, S.T. Global epidemiology of hepatitis B virus infection: New estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine 2012, 30, 2212–2219. [Google Scholar] [CrossRef]
- Nayagam, S.; Thursz, M.; Sicuri, E.; Conteh, L.; Wiktor, S.; Low-Beer, D.; Hallett, T.B. Requirements for global elimination of hepatitis B: A modelling study. Lancet Infect. Dis. 2016, 16, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Ott, J.J.; Horn, J.; Krause, G.; Mikolajczyk, R.T. Time trends of chronic HBV infection over prior decades—A global analysis. J. Hepatol. 2017, 66, 48–54. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, X.; Wu, M.; Ghildyal, R.; Yuan, Z. Animal Models for the Study of Hepatitis B Virus Pathobiology and Immunity: Past, Present, and Future. Front. Microbiol. 2021, 12, 715450. [Google Scholar] [CrossRef]
- Summers, J.; O’Connell, A.; Millman, I. Genome of hepatitis B virus: Restriction enzyme cleavage and structure of DNA extracted from Dane particles. Proc. Natl. Acad. Sci. USA 1975, 72, 4597–4601. [Google Scholar] [CrossRef]
- Glebe, D.; Bremer, C.M. The molecular virology of hepatitis B virus. Semin. Liver Dis. 2013, 33, 103–112. [Google Scholar] [CrossRef]
- Ghany, M.G.; King, W.C.; Lisker-Melman, M.; Lok, A.S.F.; Terrault, N.; Janssen, H.L.A.; Khalili, M.; Chung, R.T.; Lee, W.M.; Lau, D.T.Y.; et al. Comparison of HBV RNA and Hepatitis B Core Related Antigen With Conventional HBV Markers Among Untreated Adults With Chronic Hepatitis B in North America. Hepatology 2021, 74, 2395–2409. [Google Scholar] [CrossRef]
- Guidotti, L.G.; Ando, K.; Hobbs, M.V.; Ishikawa, T.; Runkel, L.; Schreiber, R.D.; Chisari, F.V. Cytotoxic T lymphocytes inhibit hepatitis B virus gene expression by a noncytolytic mechanism in transgenic mice. Proc. Natl. Acad. Sci. USA 1994, 91, 3764–3768. [Google Scholar] [CrossRef]
- Asabe, S.; Wieland, S.F.; Chattopadhyay, P.K.; Roederer, M.; Engle, R.E.; Purcell, R.H.; Chisari, F.V. The size of the viral inoculum contributes to the outcome of hepatitis B virus infection. J. Virol. 2009, 83, 9652–9662. [Google Scholar] [CrossRef]
- Maini, M.K.; Burton, A.R. Restoring, releasing or replacing adaptive immunity in chronic hepatitis B. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 662–675. [Google Scholar] [CrossRef]
- Song, L.W.; Liu, P.G.; Liu, C.J.; Zhang, T.Y.; Cheng, X.D.; Wu, H.L.; Yang, H.C.; Hao, X.K.; Yuan, Q.; Zhang, J.; et al. Quantitative hepatitis B core antibody levels in the natural history of hepatitis B virus infection. Clin. Microbiol. Infect. 2015, 21, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Liu, K. Complete and incomplete hepatitis B virus particles: Formation, function, and application. Viruses 2017, 9, 56. [Google Scholar] [CrossRef]
- Kock, J.; Theilmann, L.; Galle, P.; Schlicht, H.J. Hepatitis B virus nucleic acids associated with human peripheral blood mononuclear cells do not originate from replicating virus. Hepatology 1996, 23, 405–413. [Google Scholar] [CrossRef] [PubMed]
- van Campenhout, M.J.H.; van Bommel, F.; Pfefferkorn, M.; Fischer, J.; Deichsel, D.; Boonstra, A.; van Vuuren, A.J.; Berg, T.; Hansen, B.E.; Janssen, H.L.A. Host and viral factors associated with serum hepatitis B virus RNA levels among patients in need for treatment. Hepatology 2018, 68, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shen, T.; Huang, X.; Kumar, G.R.; Chen, X.; Zeng, Z.; Zhang, R.; Chen, R.; Li, T.; Zhang, T. Serum hepatitis B virus RNA is encapsidated pregenome RNA that may be associated with persistence of viral infection and rebound. J. Hepatol. 2016, 65, 700–710. [Google Scholar] [CrossRef]
- Liu, S.; Zhou, B.; Valdes, J.D.; Sun, J.; Guo, H. Serum Hepatitis B Virus RNA: A New Potential Biomarker for Chronic Hepatitis B Virus Infection. Hepatology 2019, 69, 1816–1827. [Google Scholar] [CrossRef]
- Deng, R.; Liu, S.; Shen, S.; Guo, H.; Sun, J. Circulating hepatitis B virus RNA: From biology to clinical applications. Hepatology 2022, 76, 1520–1530. [Google Scholar] [CrossRef]
- Lam, A.M.; Ren, S.; Espiritu, C.; Kelly, M.; Lau, V.; Zheng, L.; Hartman, G.D.; Flores, O.A.; Klumpp, K. Hepatitis B Virus Capsid Assembly Modulators, but Not Nucleoside Analogs, Inhibit the Production of Extracellular Pregenomic RNA and Spliced RNA Variants. Antimicrob. Agents Chemother. 2017, 61, e00680-17. [Google Scholar] [CrossRef]
- Bai, L.; Zhang, X.; Kozlowski, M.; Li, W.; Wu, M.; Liu, J.; Chen, L.; Zhang, J.; Huang, Y.; Yuan, Z. Extracellular hepatitis B virus RNAs are heterogeneous in length and circulate as capsid-antibody complexes in addition to virions in chronic hepatitis B patients. J. Virol. 2018, 92, e00798-18. [Google Scholar] [CrossRef]
- Ning, X.; Nguyen, D.; Mentzer, L.; Adams, C.; Lee, H.; Ashley, R.; Hafenstein, S.; Hu, J. Secretion of genome-free hepatitis B virus–single strand blocking model for virion morphogenesis of para-retrovirus. PLoS Pathog. 2011, 7, e1002255. [Google Scholar] [CrossRef]
- Ning, X.; Luckenbaugh, L.; Liu, K.; Bruss, V.; Sureau, C.; Hu, J. Common and Distinct Capsid and Surface Protein Requirements for Secretion of Complete and Genome-Free Hepatitis B Virions. J. Virol. 2018, 92, e00272-18. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Li, C.; Ding, J.; Wu, M.; Tang, Y.; Yuan, Z.; Zhang, X. The role of hepatitis B virus surface proteins in regulating the maturation and secretion of complete and incomplete virions. J. Gen. Virol. 2022, 103, 001733. [Google Scholar] [CrossRef] [PubMed]
- Guzman, M.G.; Vazquez, S. The complexity of antibody-dependent enhancement of dengue virus infection. Viruses 2010, 2, 2649–2662. [Google Scholar] [CrossRef]
- Dejnirattisai, W.; Jumnainsong, A.; Onsirisakul, N.; Fitton, P.; Vasanawathana, S.; Limpitikul, W.; Puttikhunt, C.; Edwards, C.; Duangchinda, T.; Supasa, S.; et al. Cross-reacting antibodies enhance dengue virus infection in humans. Science 2010, 328, 745–748. [Google Scholar] [CrossRef]
- Dammacco, F.; Sansonno, D. Therapy for hepatitis C virus-related cryoglobulinemic vasculitis. N. Engl. J. Med. 2013, 369, 1035–1045. [Google Scholar] [CrossRef]
- Shulman, N.R.; Barker, L.F. Virus-like antigen, antibody, and antigen-antibody complexes in hepatitis measured by complement fixation. Science 1969, 165, 304–306. [Google Scholar] [CrossRef]
- Almeida, J.; Waterson, A. Immune complexes in hepatitis. Lancet 1969, 294, 983–986. [Google Scholar] [CrossRef]
- Gocke, D.J.; Hsu, K.; Morgan, C.; Bombardieri, S.; Lockshin, M.; Christian, C.L. Association between polyarteritis and Australia antigen. Lancet 1970, 2, 1149–1153. [Google Scholar] [CrossRef]
- Combes, B.; Shorey, J.; Barrera, A.; Stastny, P.; Eigenbrodt, E.; Hull, A.; Carter, N. Glomerulonephritis with deposition of Australia antigen-antibody complexes in glomerular basement membrane. Lancet 1971, 298, 234–237. [Google Scholar] [CrossRef]
- Trepo, C.G.; Zucherman, A.J.; Bird, R.C.; Prince, A.M. The role of circulating hepatitis B antigen/antibody immune complexes in the pathogenesis of vascular and hepatic manifestations in polyarteritis nodosa. J. Clin. Pathol. 1974, 27, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Janssen, H.L.; van Zonneveld, M.; van Nunen, A.B.; Niesters, H.G.; Schalm, S.W.; de Man, R.A. Polyarteritis nodosa associated with hepatitis B virus infection. The role of antiviral treatment and mutations in the hepatitis B virus genome. Eur. J. Gastroenterol. Hepatol. 2004, 16, 801–807. [Google Scholar] [CrossRef] [PubMed]
- De Vos, R.; Ray, M.; Desmet, V. Electron microscopy of hepatitis B virus components in chronic active liver disease. J. Clin. Pathol. 1979, 32, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Yamada, G.; Sakamoto, Y.; Mizuno, M.; Nishihara, T.; Kobayashi, T.; Takahashi, T.; Nagashima, H. Electron and immunoelectron microscopic study of Dane particle formation in chronic hepatitis B virus infection. Gastroenterology 1982, 83, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Trevisan, A.; Realdi, G.; Alberti, A.; Ongaro, G.; Pornaro, E.; Meliconi, R. Core antigen-specific immunoglobulin G bound to the liver cell membrane in chronic hepatitis B. Gastroenterology 1982, 82, 218–222. [Google Scholar] [CrossRef]
- Michalak, T.I.; Lau, J.Y.; McFarlane, B.M.; Alexander, G.J.; Eddleston, A.L.; Williams, R. Antibody-directed complement-mediated cytotoxicity to hepatocytes from patients with chronic hepatitis B. Clin. Exp. Immunol. 1995, 100, 227–232. [Google Scholar] [CrossRef]
- Moller, B.; Hopf, U.; Poschke, S.; Alexander, M.; Friedrich-Jahnicke, B.; Mostertz, P.; L’Age, M. Serological assessment of HBcAg and HBV DNA: Its prognostic relevance in acute hepatitis B. Liver 1987, 7, 298–305. [Google Scholar] [CrossRef]
- Möller, B.; Hopf, U.; Stemerowicz, R.; Henze, G.; Gelderblom, H. HBcAg expressed on the surface of circulating Dane particles in patients with hepatitis B virus infection without evidence of anti-HBc formation. Hepatology 1989, 10, 179–185. [Google Scholar] [CrossRef]
- Possehl, C.; Repp, R.; Heermann, K.H.; Korec, E.; Uy, A.; Gerlich, W.H. Absence of free core antigen in anti-HBc negative viremic hepatitis B carriers. Arch. Virol. Suppl. 1992, 4, 39–41. [Google Scholar] [CrossRef]
- Hong, X.; Luckenbaugh, L.; Mendenhall, M.; Walsh, R.; Cabuang, L.; Soppe, S.; Revill, P.A.; Burdette, D.; Feierbach, B.; Delaney, W.; et al. Characterization of Hepatitis B Precore/Core-Related Antigens. J. Virol. 2021, 95, e01695-20. [Google Scholar] [CrossRef]
- Lauber, C.; Seitz, S.; Mattei, S.; Suh, A.; Beck, J.; Herstein, J.; Borold, J.; Salzburger, W.; Kaderali, L.; Briggs, J.A.G.; et al. Deciphering the Origin and Evolution of Hepatitis B Viruses by Means of a Family of Non-enveloped Fish Viruses. Cell Host Microbe 2017, 22, 387–399.e6. [Google Scholar] [CrossRef] [PubMed]
- K Murphy, C.W. Janeway’s Immunobiology, 9th ed.; W. W. Norton & Company: New York, NY, USA, 2016. [Google Scholar]
- Chen, Z.; Diaz, G.; Pollicino, T.; Zhao, H.; Engle, R.E.; Schuck, P.; Shen, C.-H.; Zamboni, F.; Long, Z.; Kabat, J. Role of humoral immunity against hepatitis B virus core antigen in the pathogenesis of acute liver failure. Proc. Natl. Acad. Sci. USA 2018, 115, E11369–E11378. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, S.; Zou, Z.; Shi, J.; Zhao, J.; Fan, R.; Qin, E.; Li, B.; Li, Z.; Xu, X.; et al. Hypercytolytic activity of hepatic natural killer cells correlates with liver injury in chronic hepatitis B patients. Hepatology 2011, 53, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Wells, R.G. The portal fibroblast: Not just a poor man’s stellate cell. Gastroenterology 2014, 147, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Castellano, G.; Franzin, R.; Stasi, A.; Divella, C.; Sallustio, F.; Pontrelli, P.; Lucarelli, G.; Battaglia, M.; Staffieri, F.; Crovace, A.; et al. Complement Activation During Ischemia/Reperfusion Injury Induces Pericyte-to-Myofibroblast Transdifferentiation Regulating Peritubular Capillary Lumen Reduction Through pERK Signaling. Front. Immunol. 2018, 9, 1002. [Google Scholar] [CrossRef] [PubMed]
- Holt, A.P.; Haughton, E.L.; Lalor, P.F.; Filer, A.; Buckley, C.D.; Adams, D.H. Liver myofibroblasts regulate infiltration and positioning of lymphocytes in human liver. Gastroenterology 2009, 136, 705–714. [Google Scholar] [CrossRef]
- Farci, P.; Diaz, G.; Chen, Z.; Govindarajan, S.; Tice, A.; Agulto, L.; Pittaluga, S.; Boon, D.; Yu, C.; Engle, R.E. B cell gene signature with massive intrahepatic production of antibodies to hepatitis B core antigen in hepatitis B virus–associated acute liver failure. Proc. Natl. Acad. Sci. USA 2010, 107, 8766–8771. [Google Scholar] [CrossRef]
- Milich, D.R.; Chen, M.; Schödel, F.; Peterson, D.L.; Jones, J.E.; Hughes, J.L. Role of B cells in antigen presentation of the hepatitis B core. Proc. Natl. Acad. Sci. USA 1997, 94, 14648–14653. [Google Scholar] [CrossRef]
- McFarlane, B.M.; McSorley, C.G.; Vergani, D.; McFarlane, I.G.; Williams, R. Serum autoantibodies reacting with the hepatic asialoglycoprotein receptor protein (hepatic lectin) in acute and chronic liver disorders. J. Hepatol. 1986, 3, 196–205. [Google Scholar] [CrossRef]
- McFarlane, B.M.; Bridger, C.B.; Smith, H.M.; Antonov, K.A.; Naoumov, N.; Williams, R.; McFarlane, I.G. Autoimmune mechanisms in chronic hepatitis B and delta virus infections. Eur. J. Gastroenterol. Hepatol. 1995, 7, 615–621. [Google Scholar]
- Treichel, U.; McFarlane, B.M.; Seki, T.; Krawitt, E.L.; Alessi, N.; Stickel, F.; McFarlane, I.G.; Kiyosawa, K.; Furuta, S.; Freni, M.A.; et al. Demographics of anti-asialoglycoprotein receptor autoantibodies in autoimmune hepatitis. Gastroenterology 1994, 107, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Diao, J.; Michalak, T.I. Virus-induced anti-asialoglycoprotein receptor autoimmunity in experimental hepadnaviral hepatitis. Hepatology 1997, 25, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Diao, J.; Churchill, N.D.; Michalak, T.I. Complement-mediated cytotoxicity and inhibition of ligand binding to hepatocytes by woodchuck hepatitis virus-induced autoantibodies to asialoglycoprotein receptor. Hepatology 1998, 27, 1623–1631. [Google Scholar] [CrossRef]
- Cooper, M.D.; Alder, M.N. The evolution of adaptive immune systems. Cell 2006, 124, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Flajnik, M.F. Comparative analyses of immunoglobulin genes: Surprises and portents. Nat. Rev. Immunol. 2002, 2, 688–698. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Tang, Y.; Wu, M.; Wang, C.; Hu, L.; Zhang, Z. The Origin of Capsid-Derived Immune Complexes and Their Impact on HBV-Induced Liver Diseases. Viruses 2022, 14, 2766. https://doi.org/10.3390/v14122766
Zhang X, Tang Y, Wu M, Wang C, Hu L, Zhang Z. The Origin of Capsid-Derived Immune Complexes and Their Impact on HBV-Induced Liver Diseases. Viruses. 2022; 14(12):2766. https://doi.org/10.3390/v14122766
Chicago/Turabian StyleZhang, Xiaonan, Yijie Tang, Min Wu, Cong Wang, Lyuyin Hu, and Zhanqing Zhang. 2022. "The Origin of Capsid-Derived Immune Complexes and Their Impact on HBV-Induced Liver Diseases" Viruses 14, no. 12: 2766. https://doi.org/10.3390/v14122766
APA StyleZhang, X., Tang, Y., Wu, M., Wang, C., Hu, L., & Zhang, Z. (2022). The Origin of Capsid-Derived Immune Complexes and Their Impact on HBV-Induced Liver Diseases. Viruses, 14(12), 2766. https://doi.org/10.3390/v14122766






