Determinants of Anti-S Immune Response at 9 Months after COVID-19 Vaccination in a Multicentric European Cohort of Healthcare Workers—ORCHESTRA Project
Abstract
1. Introduction
2. Methods
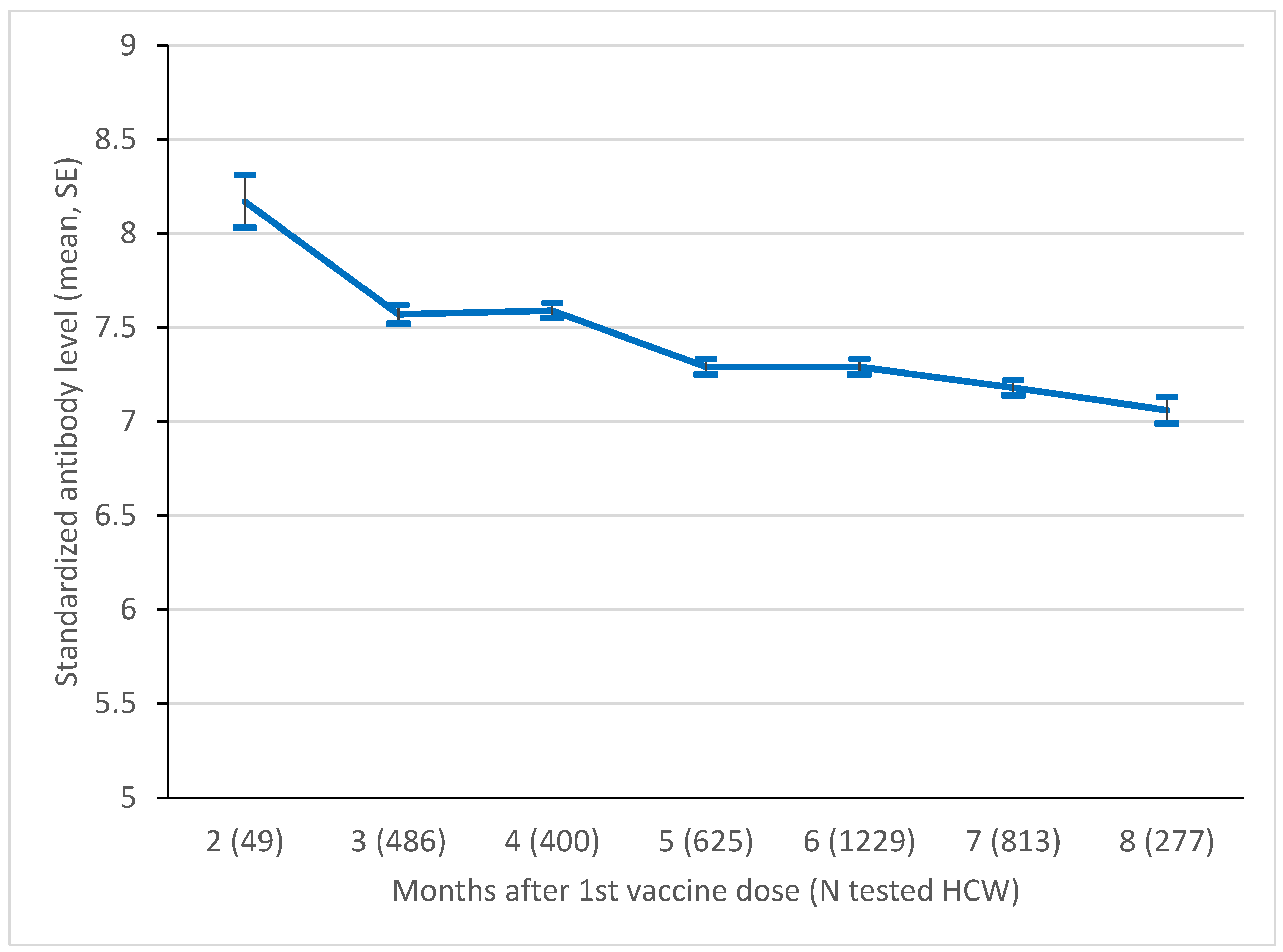
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Venugopal, U.; Jilani, N.; Rabah, S.; Shariff, M.A.; Jawed, M.; Mendez Batres, A.; Abubacker, M.; Menon, S.; Pillai, A.; Shabarek, N.; et al. SARS-CoV-2 seroprevalence among health care workers in a New York City hospital: A cross-sectional analysis during the COVID-19 pandemic. Int. J. Infect. Dis. 2021, 102, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Iversen, K.; Bundgaard, H.; Hasselbalch, R.B.; Kristensen, J.H.; Nielsen, P.B.; Pries-Heje, M.; Knudsen, A.D.; Christensen, C.E.; Fogh, K.; Norsk, J.B.; et al. Risk of COVID-19 in health-care workers in Denmark: An observational cohort study. Lancet Infect. Dis. 2020, 20, 1401–1408. [Google Scholar] [CrossRef] [PubMed]
- Joint Committee on Vaccination and Immunisation. Advice on Priority Groups for COVID-19 Vaccination, 30 December 2020. London, Department of Health and Social Care. 2020. Available online: https://www.gov.uk/government/publications/priority-groups-for-coronavirus-covid-19-vaccination-advice-from-the-jcvi-30-december-2020/joint-committee-on-vaccination-and-immunisation-advice-on-priority-groups-for-covid-19-vaccination-30-december-2020 (accessed on 1 September 2022).
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Creech, C.B.; Gettigan, J.M.; Khetan, S.; Segall, N.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.L.; Andrews, N.; Gower, C.; Robertson, C.; Stowe, J.; Tessier, E.; Simmons, R.; Cottrell, S.; Roberts, R.; O’Doherty, M.; et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on COVID-19 related symptoms, hospital admissions, and mortality in older adults in England: Test negative case-control study. BMJ 2021, 373, n1088. [Google Scholar] [CrossRef] [PubMed]
- Fiolet, T.; Kherabi, Y.; MacDonald, C.J.; Ghosn, J.; Peiffer-Smadja, N. Comparing COVID-19 vaccines for their characteristics, efficacy and effectiveness against SARS-CoV-2 and variants of concern: A narrative review. Clin. Microbiol. Infect. 2022, 28, 202–221. [Google Scholar] [CrossRef] [PubMed]
- Vallée, A.; Vasse, M.; Mazaux, L.; Bonan, B.; Amiel, C.; Zia-Chahabi, S.; Chan-Hew-Wai, A.; Farfour, E.; Camps, E.; Touche, P.; et al. An Immunogenicity Report for the Comparison between Heterologous and Homologous Prime-Boost Schedules with ChAdOx1-S and BNT162b2 Vaccines. J. Clin. Med. 2021, 10, 3817. [Google Scholar] [CrossRef]
- Cabezas, C.; Coma, E.; Mora-Fernandez, N.; Li, X.; Martinez-Marcos, M.; Fina, F.; Fabregas, M.; Hermosilla, E.; Jover, A.; Contel, J.C.; et al. Associations of BNT162b2 vaccination with SARS-CoV-2 infection and hospital admission and death with COVID-19 in nursing homes and healthcare workers in Catalonia: Prospective cohort study. BMJ 2021, 374, n1868. [Google Scholar] [CrossRef]
- Gilbert, P.B.; Montefiori, D.C.; McDermott, A.B.; Fong, Y.; Benkeser, D.; Deng, W.; Zhou, H.; Houchens, C.R.; Martins, K.; Jayashankar, L.; et al. Immune correlates analysis of the mRNA-1273 COVID-19 vaccine efficacy clinical trial. Science 2022, 375, 43–50. [Google Scholar] [CrossRef]
- Cho, A.; Wrammert, J. Implications of broadly neutralizing antibodies in the development of a universal influenza vaccine. Curr. Opin. Virol. 2016, 17, 110–115. [Google Scholar] [CrossRef]
- Turkkan, A.; Saglik, I.; Turan, C.; Sahin, A.; Akalin, H.; Ener, B.; Kara, A.; Celebi, S.; Sahin, E.; Hacimustafaoglu, M. Nine-month course of SARS-CoV-2 antibodies in individuals with COVID-19 infection. Ir. J. Med. Sci. 2022, 191, 2803–2811. [Google Scholar] [CrossRef] [PubMed]
- Lumley, S.F.; O’Donnell, D.; Stoesser, N.E.; Matthews, P.C.; Howarth, A.; Hatch, S.B.; Marsden, B.D.; Cox, S.; James, T.; Warren, F.; et al. Antibody Status and Incidence of SARS-CoV-2 Infection in Health Care Workers. N. Engl. J. Med. 2021, 384, 533–540. [Google Scholar] [CrossRef]
- Available online: https://orchestra-cohort.eu (accessed on 1 September 2022).
- Visci, G.; Zunarelli, C.; Mansour, I.; Porru, S.; De Palma, G.; Duval, X.; Monaco, M.G.L. Serological response after SARS-CoV2 vaccination in healthcare workers: A multicenter study. Med. Lav. 2022, 113, e2022022. [Google Scholar]
- Collatuzzo, G.; Visci, G.; Violante, F.S.; Porru, S.; Spiteri, G.; Monaco, M.G.L.; Fillon, F.L.; Negro, C.; Janke, C.; Castelletti, N.; et al. Determinants of anti-S immune response at 6 months after COVID-19 vaccination in a multicentric European cohort of healthcare workers—ORCHESTRA project. Front. Immunol. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Zhu, J.; Liu, Z.; Li, C.; Guo, Y.; Wang, Y.; Chen, K. Kinetics of severe acute respiratory syndrome coronavirus 2 infection antibody responses. Front. Immunol. 2022, 13, 864278. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xiang, T.; Liang, B.; Deng, H.; Wang, H.; Feng, X.; Monaco, M.G.L.; Spiteri, G.; Carta, A.; Lippi, G.; et al. Characterization of SARS-CoV-2-Specific Humoral and Cellular Immune Responses Induced by Inactivated COVID-19 Vaccines in a Real-World Setting. Front. Immunol. 2021, 12, 802858. [Google Scholar] [CrossRef]
- Yalçın, T.Y.; Topçu, D.I.; Doğan, Ö.; Aydın, S.; Sarı, N.; Erol, Ç.; Kuloğlu, Z.E.; Azap, K.; Can, F.; Arslan, H.; et al. Immunogenicity after two doses of inactivated virus vaccine in healthcare workers with and without previous COVID-19 infection: Prospective observational study. J. Med. Virol. 2022, 94, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Luevano, K.; Espino-Vazquez, A.N.; Flores-Acosta, G.; Bernaldez-Sarabia, J.; Cabanillas-Bernal, O.; Gasperin-Bulbarela, J.; Gonzalez-Sanchez, R.; Comas-Garcia, A.; Licea-Navarro, A.F. Neutralizing antibodies levels are increased in individuals with heterologous vaccination and hybrid immunity with Ad5-nCoV in the north of Mexico. PLoS ONE 2022, 17, e0269032. [Google Scholar] [CrossRef]
- Violán, C.; Torán-Monserrat, P.; Quirant, B.; Lamonja-Vicente, N.; Carrasco-Ribelles, L.A.; Chacón, C.; Manresa-Dominguez, J.M.; Ramos-Roure, F.; Dacosta-Aguayo, R.; Palacios-Fernández, C.; et al. Kinetics of humoral immune response over 17 months of COVID-19 pandemic in a large cohort of healthcare workers in Spain: The ProHEpiC-19 study. BMC Infect. Dis. 2022, 22, 721. [Google Scholar] [CrossRef]
- Wolszczak-Biedrzycka, B.; Bieńkowska, A.; Dorf, J. Assessment of Post-Vaccination Antibody Response Eight Months after the Administration of BNT1622b2 Vaccine to Healthcare Workers with Particular Emphasis on the Impact of Previous COVID-19 Infection. Vaccines 2021, 9, 1508. [Google Scholar] [CrossRef]
- Wolszczak-Biedrzycka, B.; Bieńkowska, A.; Zaborowska, J.E.; Smolińska-Fijołek, E.; Biedrzycki, G.; Dorf, J. Anti-SARS-CoV-2S Antibody Levels in Healthcare Workers 10 Months after the Administration of Two BNT162b2 Vaccine Doses in View of Demographic Characteristic and Previous COVID-19 Infection. Vaccines 2022, 10, 741. [Google Scholar] [CrossRef]
- Dimeglio, C.; Herin, F.; Martin-Blondel, G.; Miedougé, M.; Izopet, J. Antibody Titers and Protection against a SARS-CoV-2 Infection. J. Infect. 2021, 2, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, P.; Curtis, N. Factors That Influence the Immune Response to Vaccination. Clin. Microbiol. Rev. 2019, 32, e00084-18. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Shaw, R.H.; Stuart, A.S.V.; Greenland, M.; Aley, P.K.; Andrews, N.J.; Cameron, J.C.; Charlton, S.; Clutterbuck, E.A.; Collins, A.M.; et al. Safety and immunogenicity of heterologous versus homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine (Com-COV): A single-blind, randomised, non-inferiority trial. Lancet 2021, 398, 856–869. [Google Scholar] [CrossRef] [PubMed]
- Lu, S. Heterologous prime-boost vaccination. Curr. Opin. Immunol. 2009, 21, 346–351. [Google Scholar] [CrossRef]
- Torres, I.; Poujois, S.; Albert, E.; Colomina, J.; Navarro, D. Evaluation of a rapid antigen test (Panbio™ COVID-19 Ag rapid test device) for SARS-CoV-2 detection in asymptomatic close contacts of COVID-19 patients. Clin. Microbiol. Infect. 2021, 27, e1–e636. [Google Scholar] [CrossRef]
- Dinnes, J.; Deeks, J.J.; Berhane, S.; Taylor, M.; Adriano, A.; Davenport, C.; Dittrich, S.; Emperador, D.; Takwoingi, Y.; Cunningham, J.; et al. Cochrane COVID-19 Diagnostic Test Accuracy Group. Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARS-CoV-2 infection. Cochrane Database Syst Rev. 2021, 3, CD013705. [Google Scholar]
- Böger, B.; Fachi, M.M.; Vilhena, R.O.; Cobre, A.F.; Tonin, F.S.; Pontarolo, R. Systematic review with meta-analysis of the accuracy of diagnostic tests for COVID-19. Am. J. Infect. Control. 2021, 49, 21–29. [Google Scholar] [CrossRef]
- Parry, H.M.; Bruton, R.; Stephens, C.; Brown, K.; Amirthalingam, G.; Hallis, B.; Otter, A.; Zuo, J.; Moss, P. Extended interval BNT162b2 vaccination enhances peak antibody generation in older people. medRxiv 2021, 5, 1–20. [Google Scholar]
| Variables | Observed | Percent | Mean | Std. Error |
|---|---|---|---|---|
| Cohort | ||||
| Italy—Bologna | 4.402 | 21.5 | 6.46 | 0.02 |
| Italy—Brescia | 6.250 | 30.5 | 5.79 | 0.01 |
| Italy—Trieste | 1995 | 9.7 | 5.83 | 0.03 |
| Italy—Verona | 3250 | 15.9 | 6.23 | 0.02 |
| Germany—Munich | 3473 | 17.0 | 2.48 | 0.01 |
| Slovakia—Multicenter | 567 | 2.8 | 5.75 | 0.05 |
| Spain—Northern Barcelona region | 412 | 2.0 | 5.61 | 0.06 |
| Spain—Oviedo | 127 | 0.6 | 5.20 | 0.16 |
| Gender | 3473 | |||
| Male | 5515 | 27.2 | 5.41 | 0.02 |
| Female | 14,750 | 72.8 | 5.35 | 0.02 |
| Age group | ||||
| ≤29 | 2667 | 13.0 | 5.53 | 0.03 |
| 30–39 | 4555 | 22.2 | 5.42 | 0.02 |
| 40–49 | 4825 | 23.6 | 5.36 | 0.02 |
| ≥50 | 8429 | 41.2 | 5.30 | 0.02 |
| Job title | ||||
| Administration | 1364 | 8.1 | 5.75 | 0.03 |
| Technician | 1349 | 8.0 | 5.97 | 0.03 |
| Nurses | 6314 | 37.7 | 6.02 | 0.01 |
| Physician (including residents) | 4363 | 26.0 | 6.04 | 0.01 |
| Other HCW (including auxiliary workers) | 3376 | 20.1 | 5.93 | 0.02 |
| Previous COVID-19 infection (PCR) | ||||
| Never infected | 17,889 | 87.4 | 5.21 | 0.01 |
| Infected before vaccination | 2069 | 10.1 | 6.55 | 0.03 |
| Infected after 1st dose | 462 | 2.3 | 6.31 | 0.07 |
| Infected at both times | 48 | 0.2 | 6.07 | 0.11 |
| Previous COVID-19 infection (anti-N ser. test) * | ||||
| Never infected | 8313 | 82.1 | 4.17 | 0.02 |
| Infected at least once | 1808 | 17.9 | 6.43 | 0.03 |
| Type of vaccine | ||||
| Comirnaty | 19,824 | 97.1 | 5.39 | 0.01 |
| Spikevax | 446 | 2.2 | 4.91 | 0.10 |
| Vaxzevria | 47 | 0.2 | 3.21 | 0.31 |
| Mixed vaccines | 108 | 0.5 | 3.50 | 0.15 |
| Number of doses | ||||
| One dose received | 260 | 1.3 | 5.82 | 0.12 |
| Two doses received | 20,216 | 98.7 | 5.36 | 0.01 |
| Variables | Obs | Mean | Std. dev | Minimum | Maximum |
|---|---|---|---|---|---|
| Days since previous serology test to 9-month serology test | 14,541 | 141 | 43.48 | 1 | 277 |
| Days since 1st vaccine dose to 9-month serology test | 20,476 | 250 | 25.60 | 210 | 330 |
| Days since last vaccine dose to 9-month serology test | 20,476 | 227 | 28.14 | 0 | 331 |
| RR | 95% CI | |
|---|---|---|
| Cohorts | ||
| Italy—Bologna | ref | |
| Italy—Brescia | 0.42 | 0.41–0.44 |
| Italy—Trieste | 0.59 | 0.57–0.62 |
| Italy—Verona | 0.72 | 0.69–0.75 |
| Germany—Munich | 0.02 | 0.02–0.02 |
| Slovakia—Multicentre | 0.40 | 0.37–0.43 |
| Spain—Northern Barcelona region | 0.21 | 0.19–0.23 |
| Spain—Oviedo | 0.31 | 0.27–0.36 |
| Sex | ||
| Male | ref | |
| Female | 1.05 | 1.03–1.08 |
| Age group | ||
| 10 years increase | 0.87 | 0.86–0.88 |
| <29 | - | |
| 30–39 | - | |
| 40–49 | - | |
| ≥50 | - | |
| Days since last vaccine dose to 9-month serology | ||
| 10 days increase | 0.97 | 0.97–0.98 |
| Previous COVID-19 infection (detection: PCR/antiN serology test) | ||
| Never infected | ref | |
| Infected at least once | 3.03 | 2.92–3.13 |
| Number of doses | ||
| 1 dose received | ref | |
| 2 doses received | 1.22 | 1.09–1.36 |
| Type of vaccine received | ||
| Pfizer | ref | |
| Moderna | 1.51 | 1.39–1.64 |
| AstraZeneca | 0.57 | 0.44–0.73 |
| Mixed vaccines | 1.33 | 1.12–1.57 |
| Previous COVID-19 infection (detection: PCR) | ||
| Never infected | ref | |
| Infected before vaccination | 2.64 | 2.53–2.76 |
| Infected after 1st dose | 2.68 | 2.47–2.92 |
| Infected at both times | 2.87 | 2.19–3.77 |
| Previous COVID-19 infection (detection: antiN) * | ||
| Never infected | ref | |
| Infected at least once | 4.02 | 3.86–4.19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Collatuzzo, G.; Lodi, V.; Feola, D.; De Palma, G.; Sansone, E.; Sala, E.; Janke, C.; Castelletti, N.; Porru, S.; Spiteri, G.; et al. Determinants of Anti-S Immune Response at 9 Months after COVID-19 Vaccination in a Multicentric European Cohort of Healthcare Workers—ORCHESTRA Project. Viruses 2022, 14, 2657. https://doi.org/10.3390/v14122657
Collatuzzo G, Lodi V, Feola D, De Palma G, Sansone E, Sala E, Janke C, Castelletti N, Porru S, Spiteri G, et al. Determinants of Anti-S Immune Response at 9 Months after COVID-19 Vaccination in a Multicentric European Cohort of Healthcare Workers—ORCHESTRA Project. Viruses. 2022; 14(12):2657. https://doi.org/10.3390/v14122657
Chicago/Turabian StyleCollatuzzo, Giulia, Vittorio Lodi, Daniela Feola, Giuseppe De Palma, Emanuele Sansone, Emma Sala, Christian Janke, Noemi Castelletti, Stefano Porru, Gianluca Spiteri, and et al. 2022. "Determinants of Anti-S Immune Response at 9 Months after COVID-19 Vaccination in a Multicentric European Cohort of Healthcare Workers—ORCHESTRA Project" Viruses 14, no. 12: 2657. https://doi.org/10.3390/v14122657
APA StyleCollatuzzo, G., Lodi, V., Feola, D., De Palma, G., Sansone, E., Sala, E., Janke, C., Castelletti, N., Porru, S., Spiteri, G., Monaco, M. G. L., Larese Filon, F., Negro, C., Cegolon, L., Beresova, J., Fabianova, E., Carrasco-Ribelles, L. A., Toràn-Monserrat, P., Rodriguez-Suarez, M. M., ... Boffetta, P. (2022). Determinants of Anti-S Immune Response at 9 Months after COVID-19 Vaccination in a Multicentric European Cohort of Healthcare Workers—ORCHESTRA Project. Viruses, 14(12), 2657. https://doi.org/10.3390/v14122657











