Wild Red Deer (Cervus elaphus) Do Not Play a Role as Vectors or Reservoirs of SARS-CoV-2 in North-Eastern Poland
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Collection and RNA Isolation
2.2. SARS-CoV-2 Case Definition
2.3. Real-Time RT-PCR
2.4. Immunofluorescent Assay (IFA) for Antibodies against SARS-CoV-2 Detection
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shan, C.; Yao, Y.F.; Yang, X.L.; Zhou, Y.W.; Gao, G.; Peng, Y.; Yang, L.; Hu, X.; Xiong, J.; Jiang, R.D.; et al. Infection with novel coronavirus (SARS-CoV-2) causes pneumonia in Rhesus macaques. Cell Res. 2020, 30, 670. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z.; et al. Susceptibility of ferrets, cats, dogs and other domesticated animals to SARS-coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [CrossRef]
- Rabalski, L.; Kosinski, M.; Smura, T.; Aaltonen, K.; Kant, R.; Sironen, T.; Szewczyk, B.; Grzybek, M. Severe Acute Respiratory Syndrome Coronavirus 2 in Farmed Mink (Neovison vison), Poland. Emerg. Infect. Dis. 2021, 27, 2333–2339. [Google Scholar] [CrossRef]
- Oreshkova, N.; Molenaar, R.J.; Vreman, S.; Harders, F.; Oude Munnink, B.B.; Hakze-van der Honing, R.W.; Gerhards, N.; Tolsma, P.; Bouwstra, R.; Sikkema, R.S.; et al. SARS-CoV-2 infection in farmed minks, the Netherlands, April and May 2020. Eurosurveillance 2020, 25, 2001005. [Google Scholar] [CrossRef]
- Mahdy, M.A.A.; Younis, W.; Ewaida, Z. An Overview of SARS-CoV-2 and Animal Infection. Front. Vet. Sci. 2020, 7, 596391. [Google Scholar] [CrossRef]
- Rodriguez-Morales, A.J.; Dhama, K.; Sharun, K.; Tiwari, R.; Bonilla-Aldana, D.K. Susceptibility of felids to coronaviruses. Vet. Rec. 2020, 186, e21. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Mossman, K.; Baker, M.L. Zooanthroponotic potential of SARS-CoV-2 and implications of reintroduction into human populations. Cell Host Microbe 2021, 29, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Delahay, R.J.; de la Fuente, J.; Smith, G.C.; Sharun, K.; Snary, E.L.; Flores Girón, L.; Nziza, J.; Fooks, A.R.; Brookes, S.M.; Lean, F.Z.X.; et al. Assessing the risks of SARS-CoV-2 in wildlife. One Heal. Outlook 2021, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, M. SARS-CoV-2 and the human-animal interface: Outbreaks on mink farms. Lancet Infect. Dis. 2020, 21, 18–19. [Google Scholar] [CrossRef]
- Farag, E.A.; Islam, M.M.; Enan, K.; El-Hussein, A.-R.M.; Bansal, D.; Haroun, M. SARS-CoV-2 at the human-animal interface: A review. Heliyon 2021, 7, e08496. [Google Scholar] [CrossRef]
- Oude Munnink, B.B.; Sikkema, R.S.; Nieuwenhuijse, D.F.; Molenaar, R.J.; Munger, E.; Molenkamp, R.; van der Spek, A.; Tolsma, P.; Rietveld, A.; Brouwer, M.; et al. Transmission of SARS-CoV-2 on mink farms between humans and mink and back to humans. Science 2020, 371, eabe5901. [Google Scholar] [CrossRef]
- Boklund, A.; Hammer, A.S.; Quaade, M.L.; Rasmussen, T.B.; Lohse, L.; Strandbygaard, B.; Jørgensen, C.S.; Olesen, A.S.; Hjerpe, F.B.; Petersen, H.H.; et al. SARS-CoV-2 in Danish Mink Farms: Course of the Epidemic and a Descriptive Analysis of the Outbreaks in 2020. Animals 2021, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Rabalski, L.; Kosinski, M.; Mazur-Panasiuk, N.; Szewczyk, B.; Bienkowska-Szewczyk, K.; Kant, R.; Sironen, T.; Pyrc, K.; Grzybek, M. Zoonotic spill-over of SARS-CoV-2: Mink-Adapted virus in humans. Clin. Microbiol. Infect. 2022, 28, 451.e1–451.e4. [Google Scholar] [CrossRef]
- Hammer, A.S.; Quaade, M.L.; Rasmussen, T.B.; Fonager, J.; Rasmussen, M.; Mundbjerg, K.; Lohse, L.; Strandbygaard, B.; Jørgensen, C.S.; Alfaro-Núñez, A.; et al. SARS-CoV-2 Transmission between Mink (Neovison vison) and Humans, Denmark. Emerg. Infect. Dis. 2021, 27, 547. [Google Scholar] [CrossRef]
- Chandler, J.C.; Bevins, S.N.; Ellis, J.W.; Linder, T.J.; Tell, R.M.; Jenkins-Moore, M.; Root, J.J.; Lenoch, J.B.; Robbe-Austerman, S.; DeLiberto, T.J.; et al. SARS-CoV-2 exposure in wild white-tailed deer (Odocoileus virginianus). Proc. Natl. Acad. Sci. USA 2021, 118, e2114828118. [Google Scholar] [CrossRef]
- Palmer, M.V.; Martins, M.; Falkenberg, S.; Buckley, A.; Caserta, L.C.; Mitchell, P.K.; Cassmann, E.D.; Rollins, A.; Zylich, N.C.; Renshaw, R.W.; et al. Susceptibility of White-Tailed Deer (Odocoileus virginianus) to SARS-CoV-2. J. Virol. 2021, 95, e00083-21. [Google Scholar] [CrossRef] [PubMed]
- Cool, K.; Gaudreault, N.N.; Morozov, I.; Trujillo, J.D.; Meekins, D.A.; McDowell, C.; Carossino, M.; Bold, D.; Mitzel, D.; Kwon, T.; et al. Infection and transmission of ancestral SARS-CoV-2 and its alpha variant in pregnant white-Tailed deer. Emerg. Microbes Infect. 2022, 11, 95–112. [Google Scholar] [CrossRef]
- Hale, V.L.; Dennis, P.M.; McBride, D.S.; Nolting, J.M.; Madden, C.; Huey, D.; Ehrlich, M.; Grieser, J.; Winston, J.; Lombardi, D.; et al. SARS-CoV-2 infection in free-ranging white-Tailed deer. Nature 2022, 602, 481–486. [Google Scholar] [CrossRef]
- Vandegrift, K.J.; Yon, M.; Surendran-Nair, M.; Gontu, A.; Amirthalingam, S.; Nissly, R.H.; Levine, N.; Stuber, T.; DeNicola, A.J.; Boulanger, J.R.; et al. Detection of SARS-CoV-2 Omicron variant (B.1.1.529) infection of white-tailed deer. bioRxiv Prepr. Serv. Biol. 2022. [Google Scholar]
- Roundy, C.M.; Nunez, C.M.; Thomas, L.F.; Auckland, L.D.; Tang, W.; Richison, J.J.; Green, B.R.; Hilton, C.D.; Cherry, M.J.; Pauvolid-Corrêa, A.; et al. High Seroprevalence of SARS-CoV-2 in White-Tailed Deer (Odocoileus virginianus) at One of Three Captive Cervid Facilities in Texas. Microbiol. Spectr. 2022, 10, e00576-22. [Google Scholar] [CrossRef]
- Kuchipudi, S.V.; Surendran-Nair, M.; Ruden, R.M.; Yon, M.; Nissly, R.H.; Vandegrift, K.J.; Nelli, R.K.; Li, L.; Jayarao, B.M.; Maranas, C.D.; et al. Multiple spillovers from humans and onward transmission of SARS-CoV-2 in white-tailed deer. Proc. Natl. Acad. Sci. USA 2022, 119, e2121644119. [Google Scholar] [CrossRef]
- Kotwa, J.D.; Massé, A.; Gagnier, M.; Aftanas, P.; Blais-Savoie, J.; Bowman, J.; Buchanan, T.; Chee, H.-Y.; Dibernardo, A.; Kruczkiewicz, P.; et al. First detection of SARS-CoV-2 infection in Canadian wildlife identified in free-ranging white-tailed deer (Odocoileus virginianus) from southern Québec, Canada. bioRxiv 2022, 476458. [Google Scholar]
- Palermo, P.M.; Orbegozo, J.; Watts, D.M.; Morrill, J.C. SARS-CoV-2 Neutralizing Antibodies in White-Tailed Deer from Texas. Vector-Borne Zoonotic Dis. 2021, 22, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Pickering, B.; Lung, O.; Maguire, F.; Kruczkiewicz, P.; Kotwa, J.D.; Buchanan, T.; Gagnier, M.; Guthrie, J.L.; Jardine, C.M.; Marchand-Austin, A.; et al. Highly divergent white-tailed deer SARS-CoV-2 with potential deer-to-human transmission. bioRxiv 2022, 481551. [Google Scholar]
- World Organisation for Animal Health OIE Statement on Monitoring White-Tailed Deer for SARS-CoV-2; World Organisation for Animal Health: Paris, France, 2022.
- Holding, M.; Otter, A.D.; Dowall, S.; Takumi, K.; Hicks, B.; Coleman, T.; Hemingway, G.; Royds, M.; Findlay-Wilson, S.; Curran-French, M.; et al. Screening of wild deer populations for exposure to SARS-CoV-2 in the United Kingdom, 2020–2021. Transbound. Emerg. Dis. 2022, 69, e3244–e3249. [Google Scholar] [CrossRef] [PubMed]
- Wernike, K.; Fischer, L.; Holsteg, M.; Aebischer, A.; Petrov, A.; Marquart, K.; Schotte, U.; Schön, J.; Hoffmann, D.; Hechinger, S.; et al. Serological screening in wild ruminants in Germany, 2021/2022: No evidence of SARS-CoV-2, bluetongue virus or pestivirus spread but high seroprevalences against Schmallenberg virus. Transbound. Emerg. Dis. 2022, 69, e3289–e3296. [Google Scholar] [CrossRef]
- Moreira-Soto, A.; Walzer, C.; Czirják, G.Á.; Richter, M.H.; Marino, S.F.; Posautz, A.; De Yebra Rodo, P.; McEwen, G.K.; Drexler, J.F.; Greenwood, A.D. Serological Evidence That SARS-CoV-2 Has Not Emerged in Deer in Germany or Austria during the COVID-19 Pandemic. Microorganisms 2022, 10, 748. [Google Scholar] [CrossRef] [PubMed]
- Zachos, F.E.; Hartl, G.B. Phylogeography, population genetics and conservation of the European red deer Cervus elaphus. Mamm. Rev. 2011, 41, 138–150. [Google Scholar] [CrossRef]
- Głόwny Urząd Statystyczny. Obszary Tematyczne/Roczniki Statystyczne/Roczniki Statystyczne/Rocznik Demograficzny. Głόwny Urząd Statystyczny: Warsaw, Poland, 2013. [Google Scholar]
- World Organisation for Animal Health Infection with SARS-COV-2 in Animals; World Organisation for Animal Health: Paris, France, 2022.
- Grzybek, M.; Sironen, T.; Mäki, S.; Tołkacz, K.; Alsarraf, M.; Strachecka, A.; Paleolog, J.; Biernat, B.; Szczepaniak, K.; Behnke-Borowczyk, J.; et al. Zoonotic Virus Seroprevalence among Bank Voles, Poland, 2002–2010. Emerg. Infect. Dis. 2019, 25, 1607–1609. [Google Scholar] [CrossRef] [PubMed]
- Bourret, V.; Dutra, L.; Alburkat, H.; Mäki, S.; Lintunen, E.; Wasniewski, M.; Kant, R.; Grzybek, M.; Venkat, V.; Asad, H.; et al. Serological surveillance for wild rodent infection with SARS-CoV-2 in Europe. bioRxiv 2022, 502439. [Google Scholar]
- Grzybek, M.; Tołkacz, K.; Sironen, T.; Mäki, S.; Alsarraf, M.; Behnke-Borowczyk, J.; Biernat, B.; Nowicka, J.; Vaheri, A.; Henttonen, H.; et al. Zoonotic Viruses in Three Species of Voles from Poland. Animals 2020, 10, 1820. [Google Scholar] [CrossRef] [PubMed]
- Damas, J.; Hughes, G.M.; Keough, K.C.; Painter, C.A.; Persky, N.S.; Corbo, M.; Hiller, M.; Koepfli, K.-P.; Pfenning, A.R.; Zhao, H.; et al. Broad host range of SARS-CoV-2 predicted by comparative and structural analysis of ACE2 in vertebrates. Proc. Natl. Acad. Sci. USA 2020, 117, 22311–22322. [Google Scholar] [CrossRef] [PubMed]
- Grund, M.D.; McAninch, J.B.; Wiggers, E.P. Seasonal Movements and Habitat Use of Female White-Tailed Deer Associated with an Urban Park. J. Wildl. Manage. 2002, 66, 123. [Google Scholar] [CrossRef]
- Blanchong, J.A.; Sorin, A.B.; Scribner, K.T. Genetic diversity and population structure in urban white-tailed deer. J. Wildl. Manage. 2013, 77, 855–862. [Google Scholar] [CrossRef]
- Etter, D.R.; Hollis, K.M.; Van Deelen, T.R.; Ludwig, D.R.; Chelsvig, J.E.; Anchor, C.L.; Warner, R.E. Survival and Movements of White-Tailed Deer in Suburban Chicago, Illinois. J. Wildl. Manage. 2002, 66, 500. [Google Scholar] [CrossRef]
- Coppes, J.; Burghardt, F.; Hagen, R.; Suchant, R.; Braunisch, V. Human recreation affects spatio-temporal habitat use patterns in red deer (Cervus elaphus). PLoS ONE 2017, 12, e0175134. [Google Scholar] [CrossRef]
- Grear, D.A.; Samuel, M.D.; Scribner, K.T.; Weckworth, B.V.; Langenberg, J.A. Influence of genetic relatedness and spatial proximity on chronic wasting disease infection among female white-tailed deer. J. Appl. Ecol. 2010, 47, 532–540. [Google Scholar] [CrossRef]
- Walton, L.; Marion, G.; Davidson, R.S.; White, P.C.L.; Smith, L.A.; Gavier-Widen, D.; Yon, L.; Hannant, D.; Hutchings, M.R. The ecology of wildlife disease surveillance: Demographic and prevalence fluctuations undermine surveillance. J. Appl. Ecol. 2016, 53, 1460–1469. [Google Scholar] [CrossRef]
- Grzybek, M.; Alsarraf, M.; Tołkacz, K.; Behnke-Borowczyk, J.; Biernat, B.; Stańczak, J.; Strachecka, A.; Guz, L.; Szczepaniak, K.; Paleolog, J.; et al. Seroprevalence of TBEV in bank voles from Poland-a long-term approach. Emerg. Microbes Infect. 2018, 7, 145. [Google Scholar] [CrossRef]
- Grzybek, M.; Tołkacz, K.; Alsarraf, M.; Dwużnik, D.; Szczepaniak, K.; Tomczuk, K.; Biernat, B.; Behnke, J.M.; Bajer, A. Seroprevalence of Tick-Borne Encephalitis Virus in Three Species of Voles (Microtus spp.) in Poland. J. Wildl. Dis. 2020, 56, 492. [Google Scholar] [CrossRef]
- Tołkacz, K.; Alsarraf, M.; Kowalec, M.; Dwużnik, D.; Grzybek, M.; Behnke, J.M.; Bajer, A. Bartonella infections in three species of Microtus: Prevalence and genetic diversity, vertical transmission and the effect of concurrent Babesia microti infection on its success. Parasit. Vectors 2018, 11, 491. [Google Scholar] [CrossRef]
- Grzybek, M.; Cybulska, A.; Tołkacz, K.; Alsarraf, M.; Behnke-Borowczyk, J.; Szczepaniak, K.; Strachecka, A.; Paleolog, J.; Moskwa, B.; Behnke, J.M.; et al. Seroprevalence of Trichinella spp. infection in bank voles (Myodes glareolus)-A long term study. Int. J. Parasitol. Parasites Wildl. 2019, 9, 144–148. [Google Scholar] [CrossRef]
- Food and Agriculture Organization, World Organisation for Animal Health, and World Health Organization. Joint Statement on the Prioritisation of Monitoring SARS-CoV-2 Infection in Wildlife and Preventing the Formation of Animal Reservoirs; World Organisation for Animal Health: Paris, France, 2022. [Google Scholar]
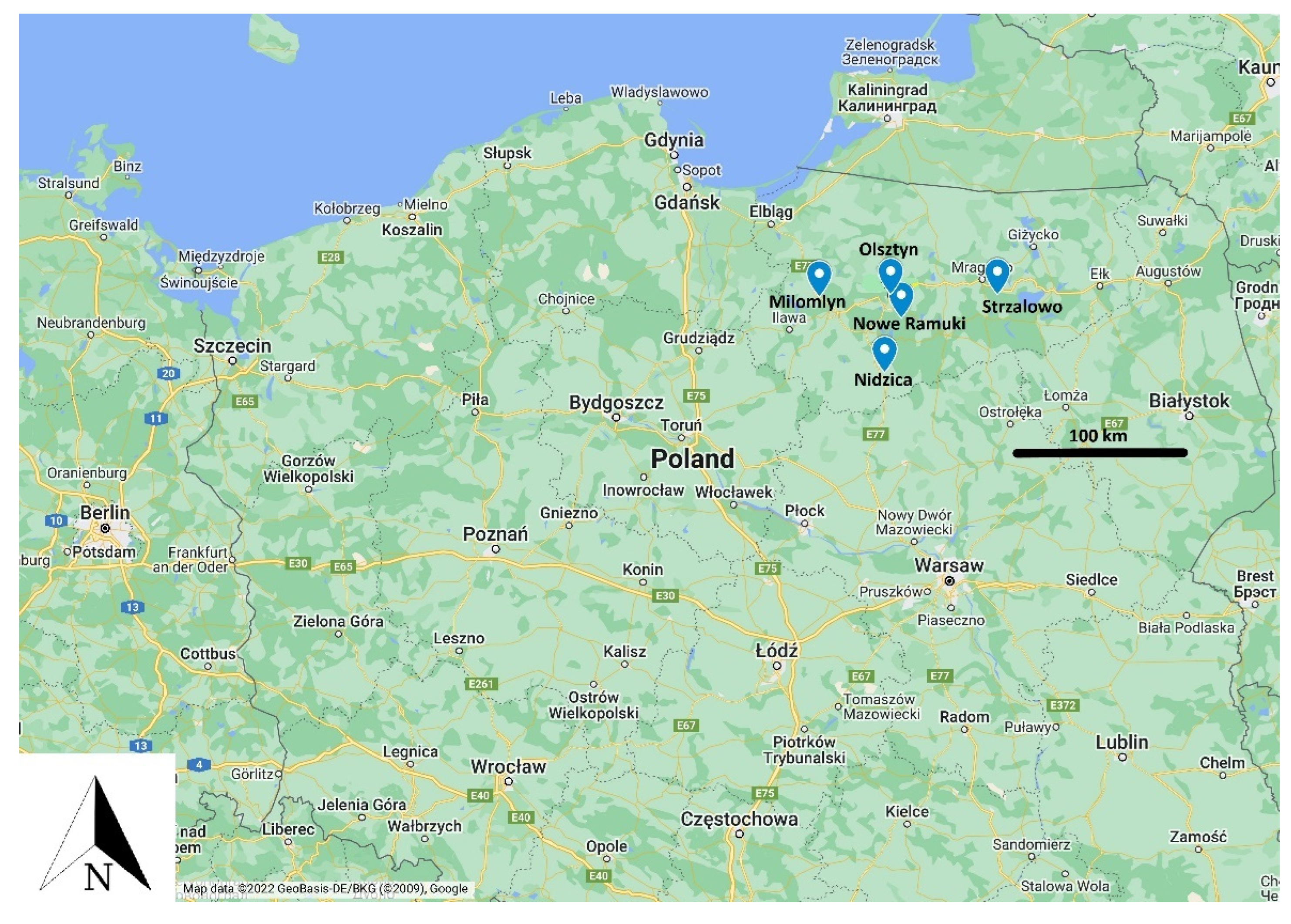
| Hunting District/Deer | Doe | Bull | Fawn | Total |
|---|---|---|---|---|
| Strzalowo | 16 | 2 | 0 | 18 |
| Olsztyn | 12 | 4 | 3 | 19 |
| Milomlyn | 4 | 0 | 1 | 5 |
| Nidzica | 8 | 4 | 4 | 16 |
| Nowe Ramuki | 20 | 7 | 5 | 32 |
| Total | 60 | 17 | 13 | 90 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krupińska, M.; Borkowski, J.; Goll, A.; Nowicka, J.; Baranowicz, K.; Bourret, V.; Strandin, T.; Mäki, S.; Kant, R.; Sironen, T.; et al. Wild Red Deer (Cervus elaphus) Do Not Play a Role as Vectors or Reservoirs of SARS-CoV-2 in North-Eastern Poland. Viruses 2022, 14, 2290. https://doi.org/10.3390/v14102290
Krupińska M, Borkowski J, Goll A, Nowicka J, Baranowicz K, Bourret V, Strandin T, Mäki S, Kant R, Sironen T, et al. Wild Red Deer (Cervus elaphus) Do Not Play a Role as Vectors or Reservoirs of SARS-CoV-2 in North-Eastern Poland. Viruses. 2022; 14(10):2290. https://doi.org/10.3390/v14102290
Chicago/Turabian StyleKrupińska, Martyna, Jakub Borkowski, Aleksander Goll, Joanna Nowicka, Karolina Baranowicz, Vincent Bourret, Tomas Strandin, Sanna Mäki, Ravi Kant, Tarja Sironen, and et al. 2022. "Wild Red Deer (Cervus elaphus) Do Not Play a Role as Vectors or Reservoirs of SARS-CoV-2 in North-Eastern Poland" Viruses 14, no. 10: 2290. https://doi.org/10.3390/v14102290
APA StyleKrupińska, M., Borkowski, J., Goll, A., Nowicka, J., Baranowicz, K., Bourret, V., Strandin, T., Mäki, S., Kant, R., Sironen, T., & Grzybek, M. (2022). Wild Red Deer (Cervus elaphus) Do Not Play a Role as Vectors or Reservoirs of SARS-CoV-2 in North-Eastern Poland. Viruses, 14(10), 2290. https://doi.org/10.3390/v14102290









