The Role of Male Reproductive Organs in the Transmission of African Swine Fever—Implications for Transmission
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.1.1. Trial A
2.1.2. Trial B
2.2. Viruses
2.3. Cells
2.4. Processing of Samples
2.5. Pathogen Detection
2.5.1. Quantitative Polymerase Chain Reaction (qPCR)
2.5.2. Reverse Transcription qPCR (RT-qPCR)
2.5.3. Detection of Infectious Virus (Virus Isolation)
2.6. Histopathology
2.6.1. Immunohistochemistry (IHC) and Semi-Quantitative Scoring of Viral Antigens
2.6.2. RNAScope In Situ Hybridization (ISH)
2.6.3. Dual RNAScope ISH and Immunofluorescence
2.6.4. Pathomorphology and Semi-Quantitative Scoring of Lesions
2.7. Transmission Electron Microscopy
3. Results
3.1. Clinial Signs and Gross Pathology
3.1.1. Trial A
3.1.2. Trial B
3.2. Pathogen Detection
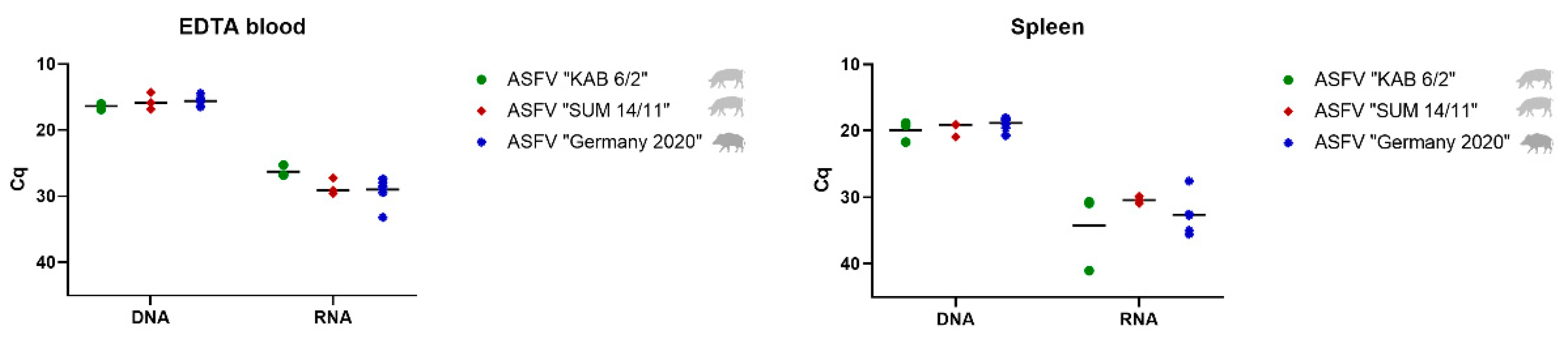
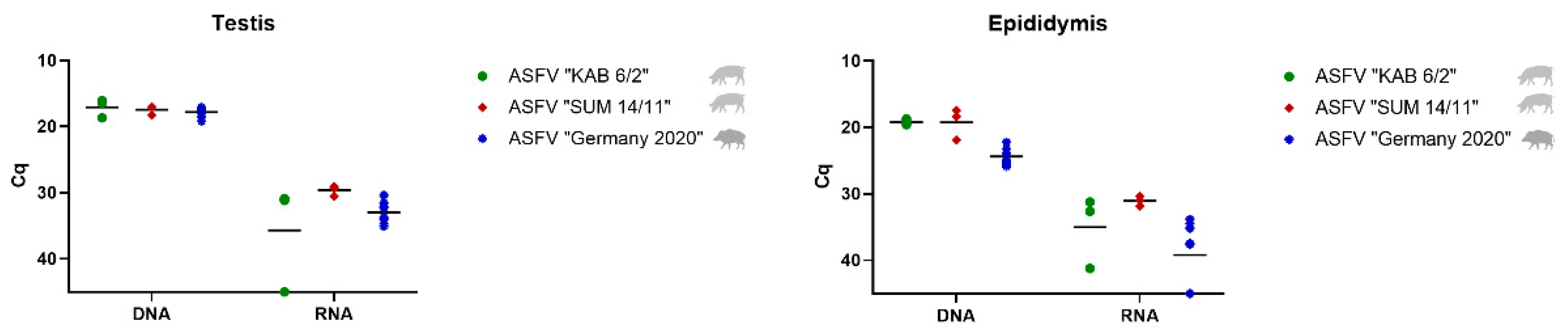
3.2.1. Detection and Quantification of Viral DNA and mRNA
Blood and Spleen
Testis and Epididymis
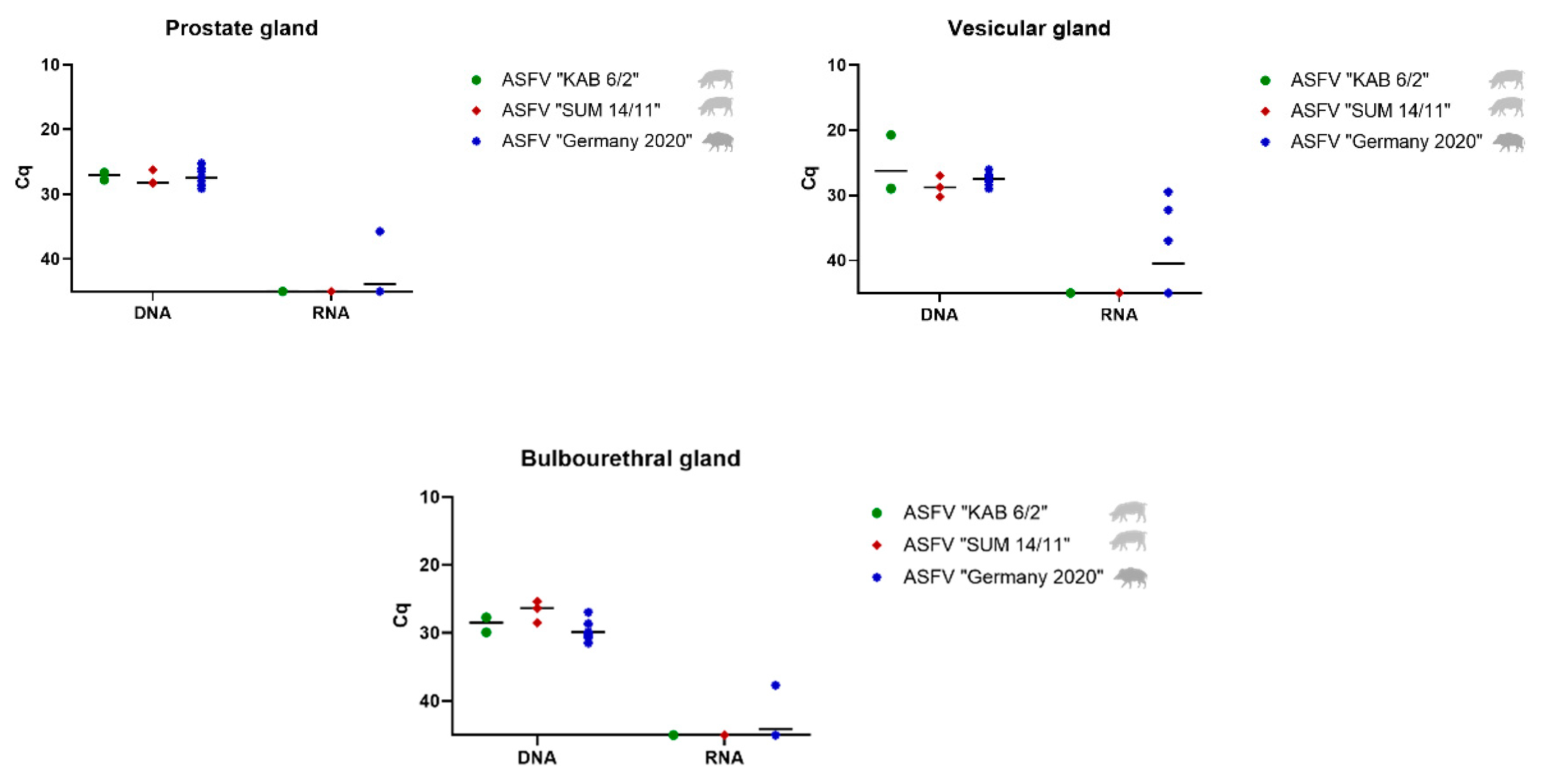
Prostate, Vesicular and Bulbourethral Gland
3.2.2. Detection of Infectious Virus
3.3. Detection and Quantification of Viral DNA, mRNA and Infectious Virus in Epididymal Sperm
3.4. Histopathology
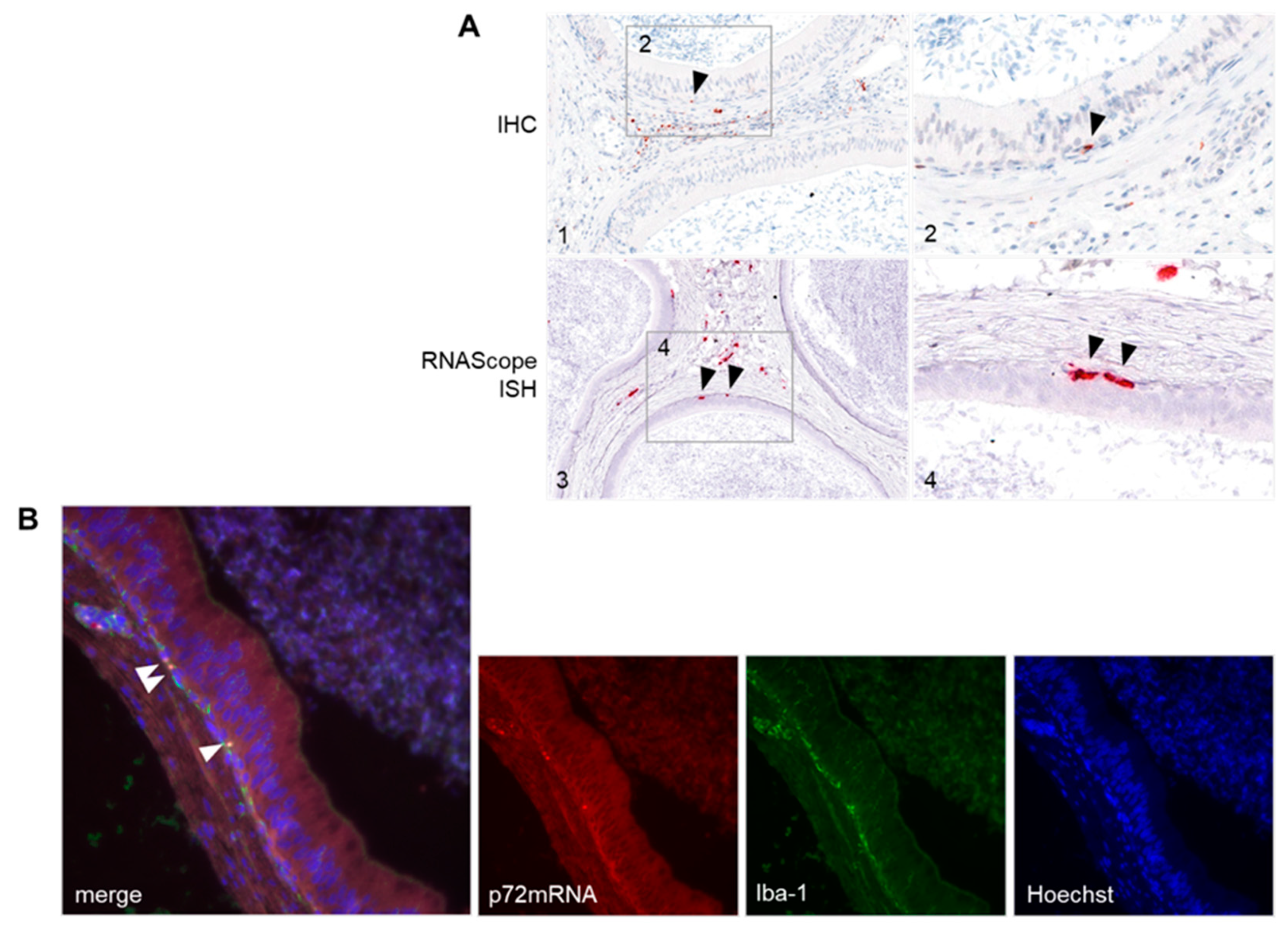
3.4.1. Distribution of Viral Antigen and mRNA in Male Reproductive Organs
3.4.2. Detection of ASFV-Infected Target Cells in the Male Reproductive Tract
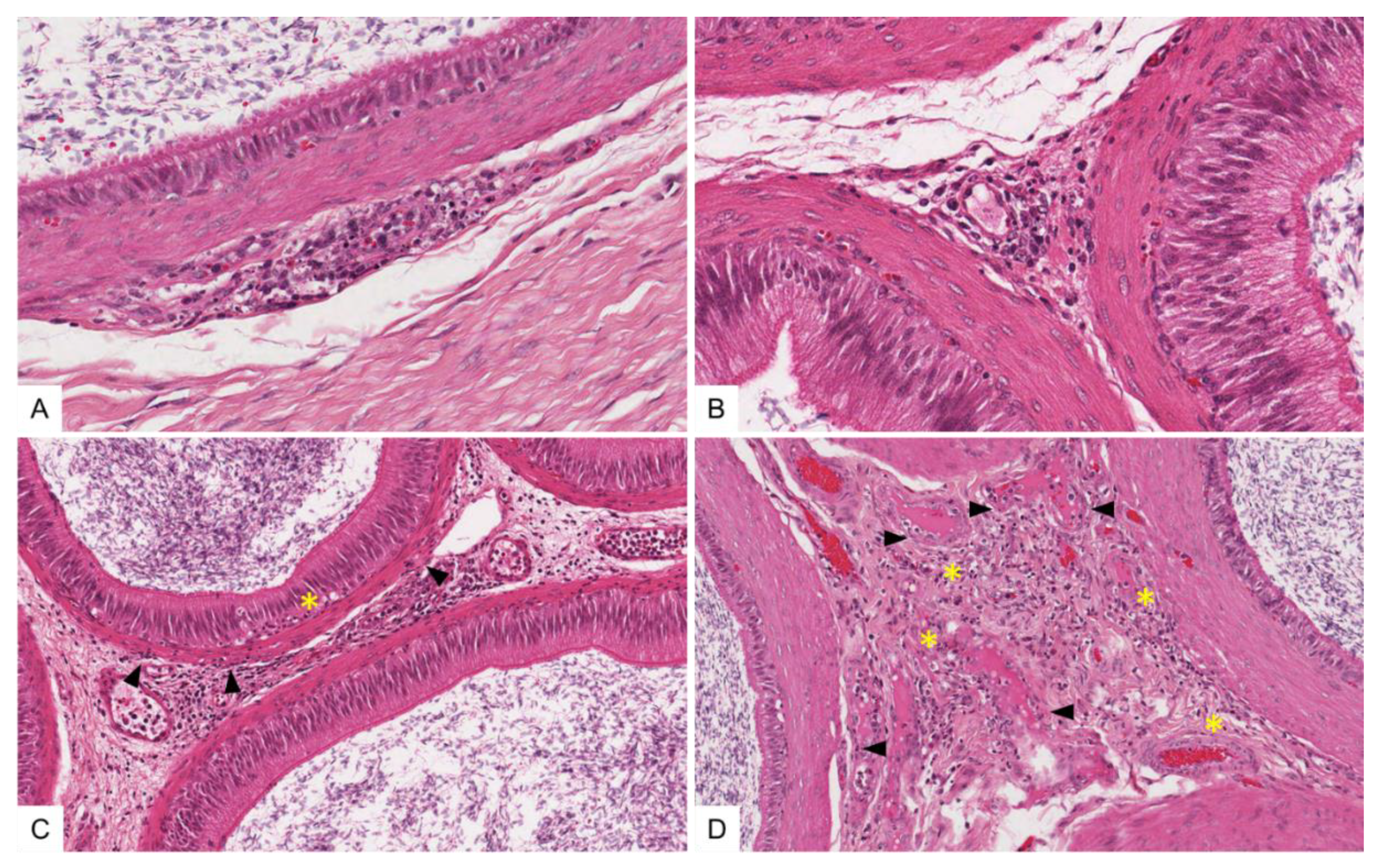
3.4.3. Histopathological Lesions in the Male Reproductive Tract
3.5. Electron Microscopy of In Vitro Inoculated Sperm
4. Discussion
4.1. African Swine Fever Virus Is Found in All Male Reproductive Tissues and Epididymal Sperm
4.2. Replication Is Linked to Mononuclear Cells of the Respective Tissues
4.3. ASFV Does Not Directly Infect Spermatozoa
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gallardo, M.C.; Reoyo, A.T.; Fernandez-Pinero, J.; Iglesias, I.; Munoz, M.J.; Arias, M.L. African swine fever: A global view of the current challenge. Porc. Health Manag. 2015, 1, 21. [Google Scholar] [CrossRef]
- Penrith, M.L.; Vosloo, W.; Jori, F.; Bastos, A.D. African swine fever virus eradication in Africa. Virus Res. 2013, 173, 228–246. [Google Scholar] [CrossRef] [PubMed]
- Dixon, L.K.; Stahl, K.; Jori, F.; Vial, L.; Pfeiffer, D.U. African Swine Fever Epidemiology and Control. Annu. Rev. Anim. Biosci. 2020, 8, 221–246. [Google Scholar] [CrossRef] [PubMed]
- Maes, D.; Van Soom, A.; Appeltant, R.; Arsenakis, I.; Nauwynck, H. Porcine semen as a vector for transmission of viral pathogens. Theriogenology 2016, 85, 27–38. [Google Scholar] [CrossRef]
- Thacker, B.J.; Larsen, R.E.; Joo, H.S.; Leman, A.D. Swine diseases transmissible with artificial insemination. J. Am. Vet. Med. Assoc. 1984, 185, 511–516. [Google Scholar] [PubMed]
- Pikalo, J.; Schoder, M.E.; Sehl, J.; Breithaupt, A.; Tignon, M.; Cay, A.B.; Gager, A.M.; Fischer, M.; Beer, M.; Blome, S. The African swine fever virus isolate Belgium 2018/1 shows high virulence in European wild boar. Transbound. Emerg. Dis. 2020, 67, 1654–1659. [Google Scholar] [CrossRef]
- Mittelholzer, C.; Moser, C.; Tratschin, J.D.; Hofmann, M.A. Analysis of classical swine fever virus replication kinetics allows differentiation of highly virulent from avirulent strains. Vet. Microbiol. 2000, 74, 293–308. [Google Scholar] [CrossRef]
- Galindo-Cardiel, I.; Ballester, M.; Solanes, D.; Nofrarías, M.; López-Soria, S.; Argilaguet, J.M.; Lacasta, A.; Accensi, F.; Rodríguez, F.; Segalés, J. Standardization of pathological investigations in the framework of experimental ASFV infections. Virus Res. 2013, 173, 180–190. [Google Scholar] [CrossRef]
- Sehl, J.; Pikalo, J.; Schäfer, A.; Franzke, K.; Pannhorst, K.; Elnagar, A.; Blohm, U.; Blome, S.; Breithaupt, A. Comparative Pathology of Domestic Pigs and Wild Boar Infected with the Moderately Virulent African Swine Fever Virus Strain “Estonia 2014”. Pathogens 2020, 9, 662. [Google Scholar] [CrossRef]
- Ehling, C.; Rath, D.; Struckmann, C.; Frenzel, A.; Schindler, L.; Niemann, H. Utilization of frozen-thawed epididymal ram semen to preserve genetic diversity in Scrapie susceptible sheep breeds. Theriogenology 2006, 66, 2160–2164. [Google Scholar] [CrossRef]
- Lubisi, B.A.; Bastos, A.D.; Dwarka, R.M.; Vosloo, W. Molecular epidemiology of African swine fever in East Africa. Arch. Virol. 2005, 150, 2439–2452. [Google Scholar] [CrossRef] [PubMed]
- Bastos, A.D.; Penrith, M.L.; Cruciere, C.; Edrich, J.L.; Hutchings, G.; Roger, F.; Couacy-Hymann, E.; Thomson, G.R. Genotyping field strains of African swine fever virus by partial p72 gene characterisation. Arch. Virol. 2003, 148, 693–706. [Google Scholar] [CrossRef] [PubMed]
- Zani, L.; Forth, J.H.; Forth, L.; Nurmoja, I.; Leidenberger, S.; Henke, J.; Carlson, J.; Breidenstein, C.; Viltrop, A.; Hoper, D.; et al. Deletion at the 5’-end of Estonian ASFV strains associated with an attenuated phenotype. Sci. Rep. 2018, 8, 6510. [Google Scholar] [CrossRef] [PubMed]
- Hübner, A.; Keßler, C.; Pannhorst, K.; Forth, J.H.; Kabuuka, T.; Karger, A.; Mettenleiter, T.C.; Fuchs, W. Identification and characterization of the 285L and K145R proteins of African swine fever virus. J. Gen. Virol. 2019, 100, 1303–1314. [Google Scholar] [CrossRef]
- Hoffmann, B.; Depner, K.; Schirrmeier, H.; Beer, M. A universal heterologous internal control system for duplex real-time RT-PCR assays used in a detection system for pestiviruses. J. Virol. Methods 2006, 136, 200–209. [Google Scholar] [CrossRef]
- King, D.P.; Reid, S.M.; Hutchings, G.H.; Grierson, S.S.; Wilkinson, P.J.; Dixon, L.K.; Bastos, A.D.; Drew, T.W. Development of a TaqMan PCR assay with internal amplification control for the detection of African swine fever virus. J. Virol. Methods 2003, 107, 53–61. [Google Scholar] [CrossRef]
- Pikalo, J.; Deutschmann, P.; Fischer, M.; Roszyk, H.; Beer, M.; Blome, S. African Swine Fever Laboratory Diagnosis-Lessons Learned from Recent Animal Trials. Pathogens 2021, 10, 177. [Google Scholar] [CrossRef]
- Hoffmann, B.; Schulz, C.; Beer, M. First detection of Schmallenberg virus RNA in bovine semen, Germany, 2012. Vet. Microbiol. 2013, 167, 289–295. [Google Scholar] [CrossRef]
- Tignon, M.; Gallardo, C.; Iscaro, C.; Hutet, E.; Van der Stede, Y.; Kolbasov, D.; De Mia, G.M.; Le Potier, M.F.; Bishop, R.P.; Arias, M.; et al. Development and inter-laboratory validation study of an improved new real-time PCR assay with internal control for detection and laboratory diagnosis of African swine fever virus. J. Virol. Methods 2011, 178, 161–170. [Google Scholar] [CrossRef]
- Carrascosa, A.L.; Bustos, M.J.; de Leon, P. Methods for growing and titrating African swine fever virus: Field and laboratory samples. Curr. Protoc. Cell Biol. 2011, 53, 26.14.1–26.14.25. [Google Scholar] [CrossRef]
- Fischer, M.; Mohnke, M.; Probst, C.; Pikalo, J.; Conraths, F.J.; Beer, M.; Blome, S. Stability of African swine fever virus on heat-treated field crops. Transbound. Emerg. Dis. 2020, 67, 2318–2323. [Google Scholar] [CrossRef]
- De Pauw, I.M.; Van Soom, A.; Laevens, H.; Verberckmoes, S.; de Kruif, A. Sperm binding to epithelial oviduct explants in bulls with different nonreturn rates investigated with a new in vitro model. Biol. Reprod. 2002, 67, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Knox, R.V. Artificial insemination in pigs today. Theriogenology 2016, 85, 83–93. [Google Scholar] [CrossRef]
- Nathues, C.; Perler, L.; Bruhn, S.; Suter, D.; Eichhorn, L.; Hofmann, M.; Nathues, H.; Baechlein, C.; Ritzmann, M.; Palzer, A.; et al. An Outbreak of Porcine Reproductive and Respiratory Syndrome Virus in Switzerland Following Import of Boar Semen. Transbound. Emerg. Dis. 2016, 63, e251–e261. [Google Scholar] [CrossRef] [PubMed]
- Hennecken, M.; Stegeman, J.A.; Elbers, A.R.; van Nes, A.; Smak, J.A.; Verheijden, J.H. Transmission of classical swine fever virus by artificial insemination during the 1997-1998 epidemic in The Netherlands: A descriptive epidemiological study. Vet. Q. 2000, 22, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vizcaino, J.M.; Mur, L.; Gomez-Villamandos, J.C.; Carrasco, L. An Update on the Epidemiology and Pathology of African Swine Fever. J. Comp. Pathol. 2014, 152, 9–21. [Google Scholar] [CrossRef]
- Kurscheidt, F.A.; Mesquita, C.S.S.; Damke, G.; Damke, E.; Carvalho, A.; Suehiro, T.T.; Teixeira, J.J.V.; da Silva, V.R.S.; Souza, R.P.; Consolaro, M.E.L. Persistence and clinical relevance of Zika virus in the male genital tract. Nat. Rev. Urol. 2019, 16, 211–230. [Google Scholar] [CrossRef]
- Schindell, B.G.; Webb, A.L.; Kindrachuk, J. Persistence and Sexual Transmission of Filoviruses. Viruses 2018, 10, 683. [Google Scholar] [CrossRef]
- Zhao, S.; Zhu, W.; Xue, S.; Han, D. Testicular defense systems: Immune privilege and innate immunity. Cell Mol. Immunol. 2014, 11, 428–437. [Google Scholar] [CrossRef]
- De Rose, R.; Fernandez, C.S.; Hedger, M.P.; Kent, S.J.; Winnall, W.R. Characterisation of macaque testicular leucocyte populations and T-lymphocyte immunity. J. Reprod. Immunol. 2013, 100, 146–156. [Google Scholar] [CrossRef]
- Nashan, D.; Malorny, U.; Sorg, C.; Cooper, T.; Nieschlag, E. Immuno-competent cells in the murine epididymis. Int. J. 1989, 12, 85–94. [Google Scholar] [CrossRef]
- Gomez-Villamandos, J.C.; Carrasco, L.; Bautista, M.J.; Sierra, M.A.; Quezada, M.; Hervas, J.; Chacon Mde, L.; Ruiz-Villamor, E.; Salguero, F.J.; Sonchez-Cordon, P.J.; et al. African swine fever and classical swine fever: A review of the pathogenesis. Dtsch. Tierarztl. Wochenschr. 2003, 110, 165–169. [Google Scholar]
- Leir, S.-H.; Yin, S.; Kerschner, J.L.; Cosme, W.; Harris, A. An atlas of human proximal epididymis reveals cell-specific functions and distinct roles for CFTR. Life Sci. Alliance 2020, 3, e202000744. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, H.; Roca, J.; Álvarez-Rodríguez, M.; Martinez-Serrano, C.A. How does the boar epididymis regulate the emission of fertile spermatozoa? Anim. Reprod. Sci. 2021, 106829. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Yu, C.; He, C.; Mei, C.; Liao, A.; Huang, D. The Immune Characteristics of the Epididymis and the Immune Pathway of the Epididymitis Caused by Different Pathogens. Front. Immunol. 2020, 11, 2115. [Google Scholar] [CrossRef]
- Serre, V.; Robaire, B. Distribution of Immune Cells in the Epididymis of the Aging Brown Norway Rat Is Segment-Specific and Related to the Luminal Content1. Biol. Reprod. 1999, 61, 705–714. [Google Scholar] [CrossRef]
- Wang, Y.F.; Holstein, A.F. Intraepithelial lymphocytes and macrophages in the human epididymis. Cell Tissue Res. 1983, 233, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, R.; Légaré, C.; Lamontagne-Proulx, J.; Breton, S.; Soulet, D. Revisiting structure/functions of the human epididymis. Andrology 2019, 7, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Flickinger, C.J.; Bush, L.A.; Howards, S.S.; Herr, J.C. Distribution of leukocytes in the epithelium and interstitium of four regions of the Lewis rat epididymis. Anat. Rec. 1997, 248, 380–390. [Google Scholar] [CrossRef]
- Salam, A.P.; Horby, P.W. The Breadth of Viruses in Human Semen. Emerg. Infect. Dis. 2017, 23, 1922–1924. [Google Scholar] [CrossRef]
- Petrunkina, A.M.; Waberski, D.; Bollwein, H.; Sieme, H. Identifying non-sperm particles during flow cytometric physiological assessment: A simple approach. Theriogenology 2010, 73, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, F.; Nagano, T. Regional differentiation of cell junctions in the excurrent duct epithelium of the rat testis as revealed by freeze-fracture. Anat. Rec. 1978, 191, 503–519. [Google Scholar] [CrossRef] [PubMed]
- Wallach, E.E.; Wolff, H. The biologic significance of white blood cells in semen. Fertil. Steril. 1995, 63, 1143–1157. [Google Scholar] [CrossRef]
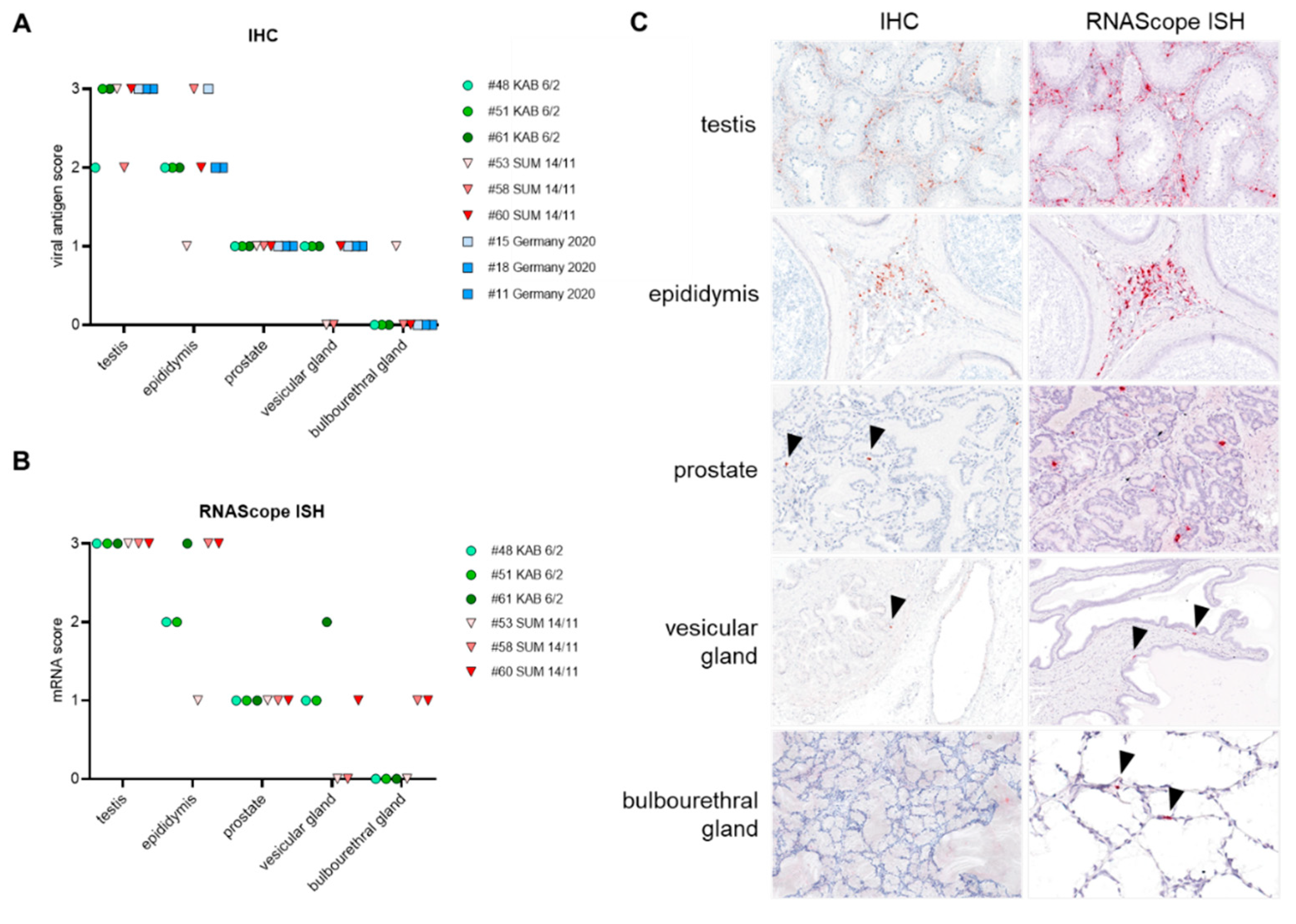
| Testis | Epididymis | Prostate | Vesicular Gland | Bulbourethral Gland | |
|---|---|---|---|---|---|
| Vasculitis/vasculopathy | X | X | X | X | X |
| Inflammatory infiltration of fibromuscular stroma | X | X | X | X | X |
| Interstitial single cell apoptosis/necrosis | X | X | X | X | X |
| Destruction of (tubuloalveolar) glands | - | - | X | X | X |
| Destruction of seminiferous tubules/epididymal ducts | X | X | - | - | - |
| Presence/absence of luminal spermatozoa * | X | X | - | - | - |
| Inoculum | ASFV “KAB 6/2” | ASFV “SUM 14/11” | |||||
|---|---|---|---|---|---|---|---|
| Animal ID | #48 | #51 | #61 | #53 | #58 | #60 | |
| Age (months) | 7 | 7 | |||||
| Necropsy (days post inoculation) | 7 | 8 | |||||
| Final clinical score points | 19 | 16.5 | 7.5 | 6 | 6 | 6 | |
| Blood | DNA Cq value | 16.86 | 16.01 | 16.13 | 16.80 | 15.86 | 14.28 |
| mRNA Cq value | 25.26 | 26.75 | 26.79 | 29.57 | 29.08 | 27.23 | |
| Infectivity | +++ | +++ | +++ | +++ | +++ | +++ | |
| Spleen | DNA Cq value | 21.69 | 18.8 | 19.18 | 20.88 | 19.07 | 19.08 |
| mRNA Cq value | 41.01 | 30.91 | 30.66 | 30.89 | 30.39 | 29.86 | |
| Infectivity | +++ | +++ | +++ | +++ | +++ | +++ | |
| Testis | DNA Cq value | 18.65 | 16.41 | 16.05 | 18.25 | 16.98 | 17.03 |
| mRNA Cq value | - | 31.14 | 30.86 | 30.52 | 29.02 | 29.40 | |
| Infectivity | - | - | +++ | +++ | +++ | +++ | |
| Epididymis | DNA Cq value | 19.55 | 18.74 | 19.29 | 21.90 | 18.25 | 17.43 |
| mRNA Cq value | 41.18 | 31.22 | 32.62 | 31.84 | 30.95 | 30.34 | |
| Infectivity | - | +++ | +++ | ++ | +++ | +++ | |
| Epididymal sperm | DNA Cq value | 25.64 | 25.49 | 25.32 | 22.21 | 27.12 | 24.74 |
| mRNA Cq value | 31.59 | 30.32 | 30.44 | 31.10 | 41.40 | 35.01 | |
| Infectivity | +++ | +++ | +++ | +++ | +++ | +++ | |
| Prostate gland | DNA Cq value | 26.70 | 27.77 | 26.6 | 28.30 | 28.19 | 26.21 |
| mRNA Cq value | - | - | - | - | - | - | |
| Infectivity | - | + | +++ | +++ | + | - | |
| Vesicular gland | DNA Cq value | 28.90 | 28.99 | 28.71 | 30.19 | 28.74 | 26.97 |
| mRNA Cq value | - | - | - | - | - | - | |
| Infectivity | +++ | +++ | +++ | +++ | +++ | + | |
| Bulbourethral gland | DNA Cq value | 27.70 | 29.97 | 27.74 | 28.47 | 26.36 | 25.35 |
| mRNA Cq value | - | - | - | - | - | - | |
| Infectivity | +++ | +++ | ++ | ++ | +++ | - | |
| Inoculum | ASFV “Germany 2020” | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Animal ID | #10 | #11 | #13 | #14 | #15 | #17 | #18 | #19 | |
| Age (months) | 6 | ||||||||
| Necropsy (days post inoculation) | 7 | ||||||||
| Final clinical score points | 5 | 4 | 5 | 6 | 5 | 5 | 5.5 | 5 | |
| EDTA Blood | DNA Cq value | 14.43 | 15.15 | 16.41 | 16.46 | 15.60 | 15.50 | 15.72 | 15.42 |
| mRNA Cq value | 33.16 | 28.93 | 29.43 | 28.52 | 27.38 | 28.93 | 27.95 | 27.41 | |
| Infectivity | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | |
| Spleen | DNA Cq value | 18.10 | 18.40 | 18.00 | 18.38 | 19.59 | 18.29 | 20.70 | 18.96 |
| mRNA Cq value | 27.54 | 34.94 | 32.71 | 32.50 | 35.53 | 32.66 | 32.43 | 32.59 | |
| Infectivity | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | |
| Testis | DNA Cq value | 17.12 | 17.49 | 17.07 | 19.16 | 17.36 | 18.50 | 17.88 | 17.58 |
| mRNA Cq value | 35.06 | 34.66 | 32.13 | 32.23 | 30.33 | 33.71 | 34.01 | 31.52 | |
| Infectivity | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | |
| Epididymis | DNA Cq value | 23.86 | 23.22 | 25.53 | 23.97 | 22.21 | 25.27 | 25.86 | 24.94 |
| mRNA Cq value | 34.45 | 37.43 | - | 35.17 | - | 37.57 | - | 33.82 | |
| Infectivity | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | |
| Prostate gland | DNA Cq value | 26.48 | 25.17 | 27.29 | 27.91 | 25.96 | 28.63 | 29.11 | 29.18 |
| mRNA Cq value | - | 35.70 | - | - | - | - | - | - | |
| Infectivity | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | |
| Vesicular gland | DNA Cq value | 27.13 | 25.99 | 27.64 | 28.98 | 26.82 | 27.88 | 26.86 | 28.37 |
| mRNA Cq value | 36.97 | - | 29.47 | 32.23 | - | - | - | - | |
| Infectivity | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | |
| Bulbourethral gland | DNA Cq value | 30.65 | 26.92 | 30.19 | 31.45 | 30.27 | 29.81 | 30.55 | 28.63 |
| mRNA Cq value | 37.66 | - | - | - | - | - | - | - | |
| Infectivity | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roszyk, H.; Franzke, K.; Breithaupt, A.; Deutschmann, P.; Pikalo, J.; Carrau, T.; Blome, S.; Sehl-Ewert, J. The Role of Male Reproductive Organs in the Transmission of African Swine Fever—Implications for Transmission. Viruses 2022, 14, 31. https://doi.org/10.3390/v14010031
Roszyk H, Franzke K, Breithaupt A, Deutschmann P, Pikalo J, Carrau T, Blome S, Sehl-Ewert J. The Role of Male Reproductive Organs in the Transmission of African Swine Fever—Implications for Transmission. Viruses. 2022; 14(1):31. https://doi.org/10.3390/v14010031
Chicago/Turabian StyleRoszyk, Hanna, Kati Franzke, Angele Breithaupt, Paul Deutschmann, Jutta Pikalo, Tessa Carrau, Sandra Blome, and Julia Sehl-Ewert. 2022. "The Role of Male Reproductive Organs in the Transmission of African Swine Fever—Implications for Transmission" Viruses 14, no. 1: 31. https://doi.org/10.3390/v14010031
APA StyleRoszyk, H., Franzke, K., Breithaupt, A., Deutschmann, P., Pikalo, J., Carrau, T., Blome, S., & Sehl-Ewert, J. (2022). The Role of Male Reproductive Organs in the Transmission of African Swine Fever—Implications for Transmission. Viruses, 14(1), 31. https://doi.org/10.3390/v14010031








