The Role of γδ T Cells as a Line of Defense in Viral Infections after Allogeneic Stem Cell Transplantation: Opportunities and Challenges
Abstract
1. Allogeneic Stem Cell Transplantation Platforms and Viral Reactivations
2. A Common Immunological Nominator for All Transplantation Platforms
3. γδ T Cells, the Frequently Forgotten Child, within the Context of Allo-HSCT
4. CMV Infections Alter the γδ T Cell Repertoire after Allo-HSCT
5. γδ T Cells Immune Reconstitution after Allo-HSCT and Interplay with EBV Infections
6. The Role of γδ T Cells in Other Viruses after Allo-HSCT: An Unexplored Field
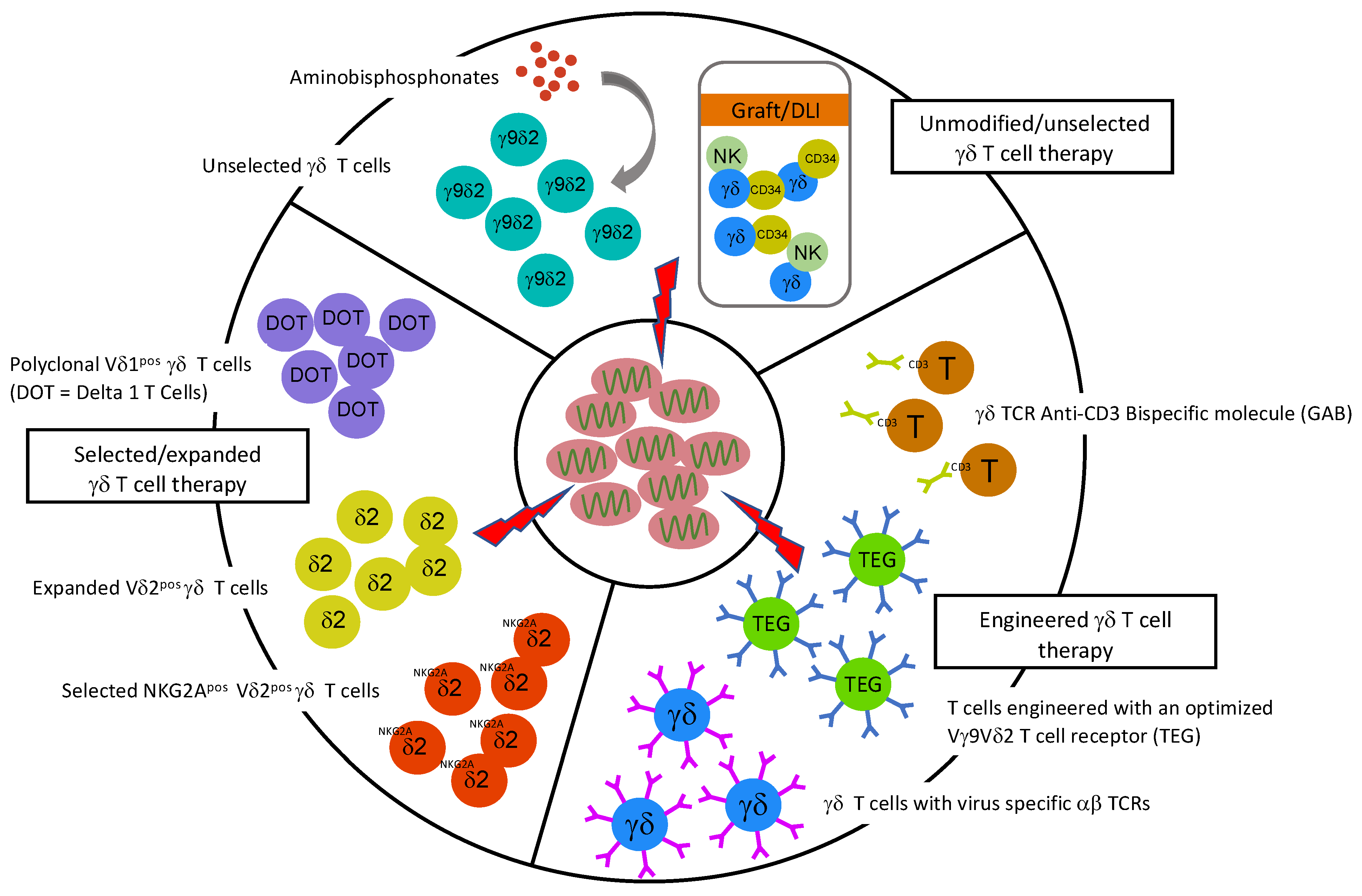
7. Unmodified γδ T Cells for Treatment of Viruses after Allo-SCT
8. Picking and Engineering Winners from γδ T Cells and Their Receptors for Future Anti-Viral Therapies
9. Conclusion within the Context of Other Anti-Viral Compounds and Re-Imbursement Dilemmas
Author Contributions
Funding
Conflicts of Interest
References
- de Witte, M.; Daenen, L.G.M.; van der Wagen, L.; van Rhenen, A.; Raymakers, R.; Westinga, K.; Kuball, J. Allogeneic Stem Cell Transplantation Platforms With Ex Vivo and In Vivo Immune Manipulations: Count and Adjust. Hemasphere 2021, 5, e580. [Google Scholar] [CrossRef]
- Chang, Y.J.; Wu, D.P.; Lai, Y.R.; Liu, Q.F.; Sun, Y.Q.; Hu, J.; Hu, Y.; Zhou, J.F.; Li, J.; Wang, S.Q.; et al. Antithymocyte Globulin for Matched Sibling Donor Transplantation in Patients With Hematologic Malignancies: A Multicenter, Open-Label, Randomized Controlled Study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2020, 38, 3367–3376. [Google Scholar] [CrossRef] [PubMed]
- Walker, I.; Panzarella, T.; Couban, S.; Couture, F.; Devins, G.; Elemary, M.; Gallagher, G.; Kerr, H.; Kuruvilla, J.; Lee, S.J.; et al. Addition of anti-thymocyte globulin to standard graft-versus-host disease prophylaxis versus standard treatment alone in patients with haematological malignancies undergoing transplantation from unrelated donors: Final analysis of a randomised, open-label, multicentre, phase 3 trial. Lancet Haematol. 2020, 7, e100–e111. [Google Scholar]
- Finke, J.; Bethge, W.A.; Schmoor, C.; Ottinger, H.D.; Stelljes, M.; Zander, A.R.; Volin, L.; Ruutu, T.; Heim, D.A.; Schwerdtfeger, R.; et al. Standard graft-versus-host disease prophylaxis with or without anti-T-cell globulin in haematopoietic cell transplantation from matched unrelated donors: A randomised, open-label, multicentre phase 3 trial. Lancet. Oncol. 2009, 10, 855–864. [Google Scholar] [CrossRef]
- Soiffer, R.J.; Kim, H.T.; McGuirk, J.; Horwitz, M.E.; Johnston, L.; Patnaik, M.M.; Rybka, W.; Artz, A.; Porter, D.L.; Shea, T.C.; et al. Prospective, Randomized, Double-Blind, Phase III Clinical Trial of Anti-T-Lymphocyte Globulin to Assess Impact on Chronic Graft-Versus-Host Disease-Free Survival in Patients Undergoing HLA-Matched Unrelated Myeloablative Hematopoietic Cell Transplantation. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2017, 35, 4003–4011. [Google Scholar] [CrossRef] [PubMed]
- Green, K.; Pearce, K.; Sellar, R.S.; Jardine, L.; Nicolson, P.L.R.; Nagra, S.; Bigley, V.; Jackson, G.; Dickinson, A.M.; Thomson, K.; et al. Impact of Alemtuzumab Scheduling on Graft-versus-Host Disease after Unrelated Donor Fludarabine and Melphalan Allografts. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2017, 23, 805–812. [Google Scholar] [CrossRef]
- Carpenter, B.; Haque, T.; Dimopoulou, M.; Atkinson, C.; Roughton, M.; Grace, S.; Denovan, S.; Fielding, A.; Kottaridis, P.D.; Griffiths, P.; et al. Incidence and dynamics of Epstein-Barr virus reactivation after alemtuzumab-based conditioning for allogeneic hematopoietic stem-cell transplantation. Transplantation 2010, 90, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Cieri, N.; Greco, R.; Crucitti, L.; Morelli, M.; Giglio, F.; Levati, G.; Assanelli, A.; Carrabba, M.G.; Bellio, L.; Milani, R.; et al. Post-transplantation Cyclophosphamide and Sirolimus after Haploidentical Hematopoietic Stem Cell Transplantation Using a Treosulfan-based Myeloablative Conditioning and Peripheral Blood Stem Cells. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2015, 21, 1506–1514. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.; Lanino, E.; Cesaro, S.; Zecca, M.; Vassallo, E.; Faraci, M.; De Bortoli, M.; Barat, V.; Prete, A.; Fagioli, F. Feasibility and Outcome of Haploidentical Hematopoietic Stem Cell Transplantation with Post-Transplant High-Dose Cyclophosphamide for Children and Adolescents with Hematologic Malignancies: An AIEOP-GITMO Retrospective Multicenter Study. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2016, 22, 902–909. [Google Scholar] [CrossRef]
- Retière, C.; Willem, C.; Guillaume, T.; Vié, H.; Gautreau-Rolland, L.; Scotet, E.; Saulquin, X.; Gagne, K.; Béné, M.C.; Imbert, B.M.; et al. Impact on early outcomes and immune reconstitution of high-dose post-transplant cyclophosphamide vs anti-thymocyte globulin after reduced intensity conditioning peripheral blood stem cell allogeneic transplantation. Oncotarget 2018, 9, 11451–11464. [Google Scholar] [CrossRef] [PubMed]
- Pasquini, M.C.; Devine, S.; Mendizabal, A.; Baden, L.R.; Wingard, J.R.; Lazarus, H.M.; Appelbaum, F.R.; Keever-Taylor, C.A.; Horowitz, M.M.; Carter, S.; et al. Comparative outcomes of donor graft CD34+ selection and immune suppressive therapy as graft-versus-host disease prophylaxis for patients with acute myeloid leukemia in complete remission undergoing HLA-matched sibling allogeneic hematopoietic cell transplantation. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2012, 30, 3194–3201. [Google Scholar]
- Barba, P.; Hilden, P.; Devlin, S.M.; Maloy, M.; Dierov, D.; Nieves, J.; Garrett, M.D.; Sogani, J.; Cho, C.; Barker, J.N.; et al. Ex Vivo CD34(+)-Selected T Cell-Depleted Peripheral Blood Stem Cell Grafts for Allogeneic Hematopoietic Stem Cell Transplantation in Acute Leukemia and Myelodysplastic Syndrome Is Associated with Low Incidence of Acute and Chronic Graft-versus-Host Disease and High Treatment Response. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2017, 23, 452–458. [Google Scholar]
- de Witte, M.A.; Janssen, A.; Nijssen, K.; Karaiskaki, F.; Swanenberg, L.; van Rhenen, A.; Admiraal, R.; van der Wagen, L.; Minnema, M.C.; Petersen, E.; et al. αβ T-cell graft depletion for allogeneic HSCT in adults with hematological malignancies. Blood Adv. 2021, 5, 240–249. [Google Scholar] [CrossRef]
- Laberko, A.; Sultanova, E.; Gutovskaya, E.; Shipitsina, I.; Shelikhova, L.; Kurnikova, E.; Muzalevskii, Y.; Kazachenok, A.; Pershin, D.; Voronin, K.; et al. Mismatched related vs matched unrelated donors in TCRαβ/CD19-depleted HSCT for primary immunodeficiencies. Blood 2019, 134, 1755–1763. [Google Scholar] [CrossRef] [PubMed]
- Maschan, M.; Shelikhova, L.; Ilushina, M.; Kurnikova, E.; Boyakova, E.; Balashov, D.; Persiantseva, M.; Skvortsova, Y.; Laberko, A.; Muzalevskii, Y.; et al. TCR-alpha/beta and CD19 depletion and treosulfan-based conditioning regimen in unrelated and haploidentical transplantation in children with acute myeloid leukemia. Bone Marrow Transplant. 2016, 51, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Bertaina, A.; Merli, P.; Rutella, S.; Pagliara, D.; Bernardo, M.E.; Masetti, R.; Pende, D.; Falco, M.; Handgretinger, R.; Moretta, F. HLA-haploidentical stem cell transplantation after removal of αβ+ T and B cells in children with nonmalignant disorders. Blood 2014, 124, 822–826. [Google Scholar] [CrossRef]
- Kierkels, G.J.J.; van Diest, E.; Hernández-López, P.; Scheper, W.; de Bruin, A.C.M.; Frijlink, E.; Aarts-Riemens, T.; van Dooremalen, S.F.J.; Beringer, D.X.; Oostvogels, R.; et al. Characterization and modulation of anti-αβTCR antibodies and their respective binding sites at the βTCR chain to enrich engineered T cells. Mol. Ther. Methods Clin. Dev. 2021, 22, 388–400. [Google Scholar] [CrossRef] [PubMed]
- Annaloro, C.; Serpenti, F.; Saporiti, G.; Galassi, G.; Cavallaro, F.; Grifoni, F.; Goldaniga, M.; Baldini, L.; Onida, F. Viral Infections in HSCT: Detection, Monitoring, Clinical Management, and Immunologic Implications. Front. Immunol. 2020, 11, 569381. [Google Scholar] [CrossRef]
- Hill, J.A.; Mayer, B.T.; Xie, H.; Leisenring, W.M.; Huang, M.L.; Stevens-Ayers, T.; Milano, F.; Delaney, C.; Sorror, M.L.; Sandmaier, B.M.; et al. The cumulative burden of double-stranded DNA virus detection after allogeneic HCT is associated with increased mortality. Blood 2017, 129, 2316–2325. [Google Scholar] [CrossRef] [PubMed]
- Rustia, E.; Violago, L.; Jin, Z.; Foca, M.D.; Kahn, J.M.; Arnold, S.; Sosna, J.; Bhatia, M.; Kung, A.L.; George, D.; et al. Risk Factors and Utility of a Risk-Based Algorithm for Monitoring Cytomegalovirus, Epstein-Barr Virus, and Adenovirus Infections in Pediatric Recipients after Allogeneic Hematopoietic Cell Transplantation. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2016, 22, 1646–1653. [Google Scholar] [CrossRef]
- Admiraal, R.; de Koning, C.C.H.; Lindemans, C.A.; Bierings, M.B.; Wensing, A.M.J.; Versluys, A.B.; Wolfs, T.F.W.; Nierkens, S.; Boelens, J.J. Viral reactivations and associated outcomes in the context of immune reconstitution after pediatric hematopoietic cell transplantation. J. Allergy Clin. Immunol. 2017, 140, 1643–1650.e9. [Google Scholar] [CrossRef] [PubMed]
- Illiaquer, M.; Imbert-Marcille, B.M.; Guillaume, T.; Planche, L.; Rimbert, M.; Bressollette-Bodin, C.; Le Bourgeois, A.; Peterlin, P.; Garnier, A.; Le Houerou, C.; et al. Impact of stem cell graft on early viral infections and immune reconstitution after allogeneic transplantation in adults. J. Clin. Virol. 2017, 93, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Langenhorst, J.B.; Boss, J.; van Kesteren, C.; Lalmohamed, A.; Kuball, J.; Egberts, A.C.G.; Boelens, J.J.; Huitema, A.D.R.; van Maarseveen, E.M. A semi-mechanistic model based on glutathione depletion to describe intra-individual reduction in busulfan clearance. Br. J. Clin. Pharm. 2020, 86, 1499–1509. [Google Scholar] [CrossRef]
- Bartelink, I.H.; Lalmohamed, A.; van Reij, E.M.; Dvorak, C.C.; Savic, R.M.; Zwaveling, J.; Bredius, R.G.; Egberts, A.C.; Bierings, M.; Kletzel, M.; et al. Association of busulfan exposure with survival and toxicity after haemopoietic cell transplantation in children and young adults: A multicentre, retrospective cohort analysis. Lancet Haematol. 2016, 3, e526–e536. [Google Scholar] [CrossRef]
- Admiraal, R.; Lindemans, C.A.; van Kesteren, C.; Bierings, M.B.; Versluijs, A.B.; Nierkens, S.; Boelens, J.J. Excellent T-cell reconstitution and survival depend on low ATG exposure after pediatric cord blood transplantation. Blood 2016, 128, 2734–2741. [Google Scholar] [CrossRef] [PubMed]
- Admiraal, R.; van Kesteren, C.; Jol-van der Zijde, C.M.; Lankester, A.C.; Bierings, M.B.; Egberts, T.C.; van Tol, M.J.; Knibbe, C.A.; Bredius, R.G.; Boelens, J.J. Association between anti-thymocyte globulin exposure and CD4+ immune reconstitution in paediatric haemopoietic cell transplantation: A multicentre, retrospective pharmacodynamic cohort analysis. Lancet Haematol. 2015, 2, e194–e203. [Google Scholar] [CrossRef]
- de Koning, C.; Prockop, S.; van Roessel, I.; Kernan, N.; Klein, E.; Langenhorst, J.; Szanto, C.; Belderbos, M.; Bierings, M.; Boulad, F.; et al. CD4+ T-cell reconstitution predicts survival outcomes after acute graft-versus-host-disease: A dual-center validation. Blood 2021, 137, 848–855. [Google Scholar] [CrossRef]
- van Roessel, I.; Prockop, S.; Klein, E.; Boulad, F.; Scaradavou, A.; Spitzer, B.; Kung, A.; Curran, K.; O’Reilly, R.J.; Kernan, N.A.; et al. Early CD4+ T cell reconstitution as predictor of outcomes after allogeneic hematopoietic cell transplantation. Cytotherapy 2020, 22, 503–510. [Google Scholar] [CrossRef]
- de Koning, C.; Langenhorst, J.; van Kesteren, C.; Lindemans, C.A.; Huitema, A.D.R.; Nierkens, S.; Boelens, J.J. Innate Immune Recovery Predicts CD4(+) T Cell Reconstitution after Hematopoietic Cell Transplantation. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2019, 25, 819–826. [Google Scholar] [CrossRef]
- de Koning, C.; Admiraal, R.; Nierkens, S.; Boelens, J.J. Human herpesvirus 6 viremia affects T-cell reconstitution after allogeneic hematopoietic stem cell transplantation. Blood Adv. 2018, 2, 428–432. [Google Scholar] [CrossRef]
- Schmid, C.; Kuball, J.; Bug, G. Defining the Role of Donor Lymphocyte Infusion in High-Risk Hematologic Malignancies. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2021, 39, 397–418. [Google Scholar] [CrossRef] [PubMed]
- Frederik Falkenburg, J.H.; Schmid, C.; Kolb, H.J.; Locatelli, F.; Kuball, J. Delayed Transfer of Immune Cells or the Art of Donor Lymphocyte Infusion. In The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapies; Carreras, E., Dufour, C., Mohty, M., Kröger, N., Eds.; Springer: Cham, Switzerland, 2019; pp. 443–448. [Google Scholar]
- Kuball, J.; Boelens, J.J. Clinical and Biological Concepts for Mastering Immune Reconstitution After HSCT: Toward Practical Guidelines and Greater Harmonization. In The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapies; Carreras, E., Dufour, C., Mohty, M., Kröger, N., Eds.; Springer: Cham, Switzerland, 2019; pp. 69–74. [Google Scholar]
- Locatelli, F.; Merli, P.; Pagliara, D.; Li Pira, G.; Falco, M.; Pende, D.; Rondelli, R.; Lucarelli, B.; Brescia, L.P.; Masetti, R.; et al. Outcome of children with acute leukemia given HLA-haploidentical HSCT after αβ T-cell and B-cell depletion. Blood 2017, 130, 677–685. [Google Scholar] [CrossRef]
- Admiraal, R.; Nierkens, S.; de Witte, M.A.; Petersen, E.J.; Fleurke, G.J.; Verrest, L.; Belitser, S.V.; Bredius, R.G.M.; Raymakers, R.A.P.; Knibbe, C.A.J.; et al. Association between anti-thymocyte globulin exposure and survival outcomes in adult unrelated haemopoietic cell transplantation: A multicentre, retrospective, pharmacodynamic cohort analysis. Lancet Haematol. 2017, 4, e183–e191. [Google Scholar] [CrossRef]
- Boelens, J.J.; Admiraal, R.; Kuball, J.; Nierkens, S. Fine-Tuning Antithymocyte Globulin Dosing and Harmonizing Clinical Trial Design. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 1175–1176. [Google Scholar] [CrossRef] [PubMed]
- Storek, J. Immunological reconstitution after hematopoietic cell transplantation—Its relation to the contents of the graft. Expert Opin. Biol. Ther. 2008, 8, 583–597. [Google Scholar] [CrossRef]
- Dunbar, A.; Schauwvlieghe, A.; Algoe, S.; van Hellemond, J.J.; Reynders, M.; Vandecasteele, S.; Boelens, J.; Depuydt, P.; Rijnders, B. Epidemiology of Pneumocystis jirovecii Pneumonia and (Non-)use of Prophylaxis. Front. Cell Infect. Microbiol. 2020, 10, 224. [Google Scholar] [CrossRef] [PubMed]
- Hirokawa, M.; Horiuchi, T.; Kawabata, Y.; Kitabayashi, A.; Miura, A.B. Reconstitution of gammadelta T cell repertoire diversity after human allogeneic hematopoietic cell transplantation and the role of peripheral expansion of mature T cell population in the graft. Bone Marrow Transplant. 2000, 26, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Ravens, S.; Schultze-Florey, C.; Raha, S.; Sandrock, I.; Drenker, M.; Oberdorfer, L.; Reinhardt, A.; Ravens, I.; Beck, M.; Geffers, R.; et al. Human gammadelta T cells are quickly reconstituted after stem-cell transplantation and show adaptive clonal expansion in response to viral infection. Nat. Immunol. 2017, 18, 393–401. [Google Scholar] [CrossRef]
- Sebestyen, Z.; Prinz, I.; Dechanet-Merville, J.; Silva-Santos, B.; Kuball, J. Translating gammadelta (gammadelta) T cells and their receptors into cancer cell therapies. Nat. Rev. Drug Discov. 2020, 19, 169–184. [Google Scholar] [CrossRef]
- Kabelitz, D.; Serrano, R.; Kouakanou, L.; Peters, C.; Kalyan, S. Cancer immunotherapy with gammadelta T cells: Many paths ahead of us. Cell Mol. Immunol. 2020, 17, 925–939. [Google Scholar] [CrossRef]
- Caron, J.; Ridgley, L.A.; Bodman-Smith, M. How to Train Your Dragon: Harnessing Gamma Delta T Cells Antiviral Functions and Trained Immunity in a Pandemic Era. Front. Immunol. 2021, 12, 666983. [Google Scholar] [CrossRef] [PubMed]
- Wesch, D.; Peters, C.; Oberg, H.H.; Pietschmann, K.; Kabelitz, D. Modulation of γδ T cell responses by TLR ligands. Cell Mol. Life Sci. 2011, 68, 2357–2370. [Google Scholar] [CrossRef]
- Couzi, L.; Pitard, V.; Moreau, J.F.; Merville, P.; Déchanet-Merville, J. Direct and Indirect Effects of Cytomegalovirus-Induced γδ T Cells after Kidney Transplantation. Front. Immunol. 2015, 6, 3. [Google Scholar] [CrossRef]
- Lamb, L.S., Jr.; Henslee-Downey, P.J.; Parrish, R.S.; Godder, K.; Thompson, J.; Lee, C.; Gee, A.P. Increased frequency of TCR gamma delta + T cells in disease-free survivors following T cell-depleted, partially mismatched, related donor bone marrow transplantation for leukemia. J. Hematother. 1996, 5, 503–509. [Google Scholar] [CrossRef]
- Godder, K.T.; Henslee-Downey, P.J.; Mehta, J.; Park, B.S.; Chiang, K.Y.; Abhyankar, S.; Lamb, L.S. Long term disease-free survival in acute leukemia patients recovering with increased gammadelta T cells after partially mismatched related donor bone marrow transplantation. Bone Marrow Transplant. 2007, 39, 751–757. [Google Scholar] [CrossRef]
- Perko, R.; Kang, G.; Sunkara, A.; Leung, W.; Thomas, P.G.; Dallas, M.H. Gamma delta T cell reconstitution is associated with fewer infections and improved event-free survival after hematopoietic stem cell transplantation for pediatric leukemia. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2015, 21, 130–136. [Google Scholar] [CrossRef]
- Minculescu, L.; Marquart, H.V.; Ryder, L.P.; Andersen, N.S.; Schjoedt, I.; Friis, L.S.; Kornblit, B.T.; Petersen, S.L.; Haastrup, E.; Fischer-Nielsen, A.; et al. Improved Overall Survival, Relapse-Free-Survival, and Less Graft-vs.-Host-Disease in Patients With High Immune Reconstitution of TCR Gamma Delta Cells 2 Months After Allogeneic Stem Cell Transplantation. Front. Immunol. 2019, 10, 1997. [Google Scholar] [CrossRef] [PubMed]
- Gaballa, A.; Stikvoort, A.; Önfelt, B.; Mattsson, J.; Sundin, M.; Watz, E.; Uhlin, M. T-cell frequencies of CD8(+) γδ and CD27(+) γδ cells in the stem cell graft predict the outcome after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2019, 54, 1562–1574. [Google Scholar] [CrossRef]
- Arruda, L.C.M.; Gaballa, A.; Uhlin, M. Impact of γδ T cells on clinical outcome of hematopoietic stem cell transplantation: Systematic review and meta-analysis. Blood Adv. 2019, 3, 3436–3448. [Google Scholar] [CrossRef] [PubMed]
- Vantourout, P.; Hayday, A. Six-of-the-best: Unique contributions of gammadelta T cells to immunology. Nat. Rev. Immunol. 2013, 13, 88–100. [Google Scholar] [CrossRef]
- Cano, C.E.; Pasero, C.; De Gassart, A.; Kerneur, C.; Gabriac, M.; Fullana, M.; Granarolo, E.; Hoet, R.; Scotet, E.; Rafia, C.; et al. BTN2A1, an immune checkpoint targeting Vγ9Vδ2 T cell cytotoxicity against malignant cells. Cell Rep. 2021, 36, 109359. [Google Scholar] [CrossRef] [PubMed]
- Rigau, M.; Ostrouska, S.; Fulford, T.S.; Johnson, D.N.; Woods, K.; Ruan, Z.; McWilliam, H.E.G.; Hudson, C.; Tutuka, C.; Wheatley, A.K.; et al. Butyrophilin 2A1 is essential for phosphoantigen reactivity by γδ T cells. Science 2020, 367, 6478. [Google Scholar] [CrossRef] [PubMed]
- Vyborova, A.; Beringer, D.X.; Fasci, D.; Karaiskaki, F.; van Diest, E.; Kramer, L.; de Haas, A.; Sanders, J.; Janssen, A.; Straetemans, T.; et al. γ9δ2T cell diversity and the receptor interface with tumor cells. J. Clin. Investig. 2020, 130, 4637–4651. [Google Scholar] [CrossRef] [PubMed]
- Willcox, C.R.; Vantourout, P.; Salim, M.; Zlatareva, I.; Melandri, D.; Zanardo, L.; George, R.; Kjaer, S.; Jeeves, M.; Mohammed, F.; et al. Butyrophilin-like 3 Directly Binds a Human Vγ4(+) T Cell Receptor Using a Modality Distinct from Clonally-Restricted Antigen. Immunity 2019, 51, 813–825.e4. [Google Scholar] [CrossRef]
- Starick, L.; Riano, F.; Karunakaran, M.M.; Kunzmann, V.; Li, J.; Kreiss, M.; Amslinger, S.; Scotet, E.; Olive, D.; De Libero, G.; et al. Butyrophilin 3A (BTN3A, CD277)-specific antibody 20.1 differentially activates Vγ9Vδ2 TCR clonotypes and interferes with phosphoantigen activation. Eur. J. Immunol. 2017, 47, 982–992. [Google Scholar] [CrossRef]
- Sebestyen, Z.; Scheper, W.; Vyborova, A.; Gu, S.; Rychnavska, Z.; Schiffler, M.; Cleven, A.; Cheneau, C.; van Noorden, M.; Peigne, C.M.; et al. RhoB Mediates Phosphoantigen Recognition by Vgamma9Vdelta2 T Cell Receptor. Cell Rep. 2016, 15, 1973–1985. [Google Scholar] [CrossRef]
- Harly, C.; Peigné, C.M.; Scotet, E. Molecules and Mechanisms Implicated in the Peculiar Antigenic Activation Process of Human Vγ9Vδ2 T Cells. Front. Immunol. 2014, 5, 657. [Google Scholar] [PubMed]
- Janssen, A.; Villacorta Hidalgo, J.; Beringer, D.X.; van Dooremalen, S.; Fernando, F.; van Diest, E.; Terrizi, A.R.; Bronsert, P.; Kock, S.; Schmitt-Graff, A.; et al. gammadelta T-cell Receptors Derived from Breast Cancer-Infiltrating T Lymphocytes Mediate Antitumor Reactivity. Cancer Immunol. Res. 2020, 8, 530–543. [Google Scholar] [CrossRef]
- Kierkels, G.J.J.; Scheper, W.; Meringa, A.D.; Johanna, I.; Beringer, D.X.; Janssen, A.; Schiffler, M.; Aarts-Riemens, T.; Kramer, L.; Straetemans, T.; et al. Identification of a tumor-specific allo-HLA-restricted γδTCR. Blood Adv. 2019, 3, 2870–2882. [Google Scholar] [CrossRef]
- Johanna, I.; Hernández-López, P.; Heijhuurs, S.; Bongiovanni, L.; de Bruin, A.; Beringer, D.; van Dooremalen, S.; Shultz, L.D.; Ishikawa, F.; Sebestyen, Z.; et al. TEG011 persistence averts extramedullary tumor growth without exerting off-target toxicity against healthy tissues in a humanized HLA-A*24:02 transgenic mice. J. Leukoc. Biol. 2020, 107, 1069–1079. [Google Scholar] [CrossRef]
- Johanna, I.; Straetemans, T.; Heijhuurs, S.; Aarts-Riemens, T.; Norell, H.; Bongiovanni, L.; de Bruin, A.; Sebestyen, Z.; Kuball, J. Evaluating in vivo efficacy—Toxicity profile of TEG001 in humanized mice xenografts against primary human AML disease and healthy hematopoietic cells. J. Immunother. Cancer 2019, 7, 69. [Google Scholar] [CrossRef]
- Johanna, I.; Hernández-López, P.; Heijhuurs, S.; Scheper, W.; Bongiovanni, L.; de Bruin, A.; Beringer, D.X.; Oostvogels, R.; Straetemans, T.; Sebestyen, Z.; et al. Adding Help to an HLA-A*24:02 Tumor-Reactive γδTCR Increases Tumor Control. Front. Immunol. 2021, 12, 4436. [Google Scholar] [CrossRef]
- Ciccone, E.; Viale, O.; Pende, D.; Malnati, M.; Battista Ferrara, G.; Barocci, S.; Moretta, A.; Moretta, L. Specificity of human T lymphocytes expressing a gamma/delta T cell antigen receptor. Recognition of a polymorphic determinant of HLA class I molecules by a gamma/delta clone. Eur. J. Immunol. 1989, 19, 1267–1271. [Google Scholar] [CrossRef] [PubMed]
- Spits, H.; Paliard, X.; Engelhard, V.H.; de Vries, J.E. Cytotoxic activity and lymphokine production of T cell receptor (TCR)-alpha beta+ and TCR-gamma delta+ cytotoxic T lymphocyte (CTL) clones recognizing HLA-A2 and HLA-A2 mutants. Recognition of TCR-gamma delta+ CTL clones is affected by mutations at positions 152 and 156. J. Immunol. 1990, 144, 4156–4162. [Google Scholar] [PubMed]
- Del Porto, P.; D’Amato, M.; Fiorillo, M.T.; Tuosto, L.; Piccolella, E.; Sorrentino, R. Identification of a novel HLA-B27 subtype by restriction analysis of a cytotoxic gamma delta T cell clone. J. Immunol. 1994, 153, 3093–3100. [Google Scholar]
- Fujishima, N.; Hirokawa, M.; Fujishima, M.; Yamashita, J.; Saitoh, H.; Ichikawa, Y.; Horiuchi, T.; Kawabata, Y.; Sawada, K.I. Skewed T cell receptor repertoire of Vdelta1(+) gammadelta T lymphocytes after human allogeneic haematopoietic stem cell transplantation and the potential role for Epstein-Barr virus-infected B cells in clonal restriction. Clin. Exp. Immunol. 2007, 149, 70–79. [Google Scholar] [CrossRef]
- Knight, A.; Madrigal, A.J.; Grace, S.; Sivakumaran, J.; Kottaridis, P.; Mackinnon, S.; Travers, P.J.; Lowdell, M.W. The role of Vdelta2-negative gammadelta T cells during cytomegalovirus reactivation in recipients of allogeneic stem cell transplantation. Blood 2010, 116, 2164–2172. [Google Scholar] [CrossRef] [PubMed]
- Khairallah, C.; Dechanet-Merville, J.; Capone, M. gammadelta T Cell-Mediated Immunity to Cytomegalovirus Infection. Front. Immunol. 2017, 8, 105. [Google Scholar] [CrossRef]
- de Witte, M.A.; Sarhan, D.; Davis, Z.; Felices, M.; Vallera, D.A.; Hinderlie, P.; Curtsinger, J.; Cooley, S.; Wagner, J.; Kuball, J.; et al. Early Reconstitution of NK and gammadelta T Cells and Its Implication for the Design of Post-Transplant Immunotherapy. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2018. [Google Scholar] [CrossRef] [PubMed]
- Airoldi, I.; Bertaina, A.; Prigione, I.; Zorzoli, A.; Pagliara, D.; Cocco, C.; Meazza, R.; Loiacono, F.; Lucarelli, B.; Bernardo, M.E.; et al. gammadelta T-cell reconstitution after HLA-haploidentical hematopoietic transplantation depleted of TCR-alphabeta+/CD19+ lymphocytes. Blood 2015, 125, 2349–2358. [Google Scholar] [CrossRef]
- Bertaina, A.; Zorzoli, A.; Petretto, A.; Barbarito, G.; Inglese, E.; Merli, P.; Lavarello, C.; Brescia, L.P.; De Angelis, B.; Tripodi, G.; et al. Zoledronic acid boosts gammadelta T-cell activity in children receiving alphabeta(+) T and CD19(+) cell-depleted grafts from an HLA-haplo-identical donor. Oncoimmunology 2017, 6, e1216291. [Google Scholar] [CrossRef]
- Scheper, W.; van Dorp, S.; Kersting, S.; Pietersma, F.; Lindemans, C.; Hol, S.; Heijhuurs, S.; Sebestyen, Z.; Grunder, C.; Marcu-Malina, V.; et al. gammadeltaT cells elicited by CMV reactivation after allo-SCT cross-recognize CMV and leukemia. Leukemia 2013, 27, 1328–1338. [Google Scholar] [CrossRef]
- Arruda, L.C.M.; Gaballa, A.; Uhlin, M. Graft gammadelta TCR Sequencing Identifies Public Clonotypes Associated with Hematopoietic Stem Cell Transplantation Efficacy in Acute Myeloid Leukemia Patients and Unravels Cytomegalovirus Impact on Repertoire Distribution. J. Immunol. 2019, 202, 1859–1870. [Google Scholar] [CrossRef]
- Prinz, I.; Thamm, K.; Port, M.; Weissinger, E.M.; Stadler, M.; Gabaev, I.; Jacobs, R.; Ganser, A.; Koenecke, C. Donor Vδ1+ γδ T cells expand after allogeneic hematopoietic stem cell transplantation and show reactivity against CMV-infected cells but not against progressing B-CLL. Exp. Hematol. Oncol. 2013, 2, 14. [Google Scholar] [CrossRef]
- Scheper, W.; Sebestyen, Z.; Kuball, J. Cancer Immunotherapy Using gammadeltaT Cells: Dealing with Diversity. Front. Immunol. 2014, 5, 601. [Google Scholar] [CrossRef] [PubMed]
- Scheper, W.; Gründer, C.; Kuball, J. Multifunctional γδ T cells and their receptors for targeted anticancer immunotherapy. Oncoimmunology 2013, 2, e23974. [Google Scholar] [CrossRef][Green Version]
- Elmaagacli, A.H.; Steckel, N.K.; Koldehoff, M.; Hegerfeldt, Y.; Trenschel, R.; Ditschkowski, M.; Christoph, S.; Gromke, T.; Kordelas, L.; Ottinger, H.D.; et al. Early human cytomegalovirus replication after transplantation is associated with a decreased relapse risk: Evidence for a putative virus-versus-leukemia effect in acute myeloid leukemia patients. Blood 2011, 118, 1402–1412. [Google Scholar] [CrossRef]
- Green, M.L.; Leisenring, W.M.; Xie, H.; Walter, R.B.; Mielcarek, M.; Sandmaier, B.M.; Riddell, S.R.; Boeckh, M. CMV reactivation after allogeneic HCT and relapse risk: Evidence for early protection in acute myeloid leukemia. Blood 2013, 122, 1316–1324. [Google Scholar] [CrossRef] [PubMed]
- Peric, Z.; Wilson, J.; Durakovic, N.; Ostojic, A.; Desnica, L.; Vranjes, V.R.; Marekovic, I.; Serventi-Seiwerth, R.; Vrhovac, R. Early human cytomegalovirus reactivation is associated with lower incidence of relapse of myeloproliferative disorders after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2018, 53, 1450–1456. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Hieber, M.; Labopin, M.; Beelen, D.; Volin, L.; Ehninger, G.; Finke, J.; Socié, G.; Schwerdtfeger, R.; Kröger, N.; Ganser, A.; et al. CMV serostatus still has an important prognostic impact in de novo acute leukemia patients after allogeneic stem cell transplantation: A report from the Acute Leukemia Working Party of EBMT. Blood 2013, 122, 3359–3364. [Google Scholar] [CrossRef] [PubMed]
- Broers, A.E.; van Der Holt, R.; van Esser, J.W.; Gratama, J.W.; Henzen-Logmans, S.; Kuenen-Boumeester, V.; Löwenberg, B.; Cornelissen, J.J. Increased transplant-related morbidity and mortality in CMV-seropositive patients despite highly effective prevention of CMV disease after allogeneic T-cell-depleted stem cell transplantation. Blood 2000, 95, 2240–2245. [Google Scholar] [CrossRef]
- Teira, P.; Battiwalla, M.; Ramanathan, M.; Barrett, A.J.; Ahn, K.W.; Chen, M.; Green, J.S.; Saad, A.; Antin, J.H.; Savani, B.N.; et al. Early cytomegalovirus reactivation remains associated with increased transplant-related mortality in the current era: A CIBMTR analysis. Blood 2016, 127, 2427–2438. [Google Scholar] [CrossRef] [PubMed]
- Litjens, N.H.R.; van der Wagen, L.; Kuball, J.; Kwekkeboom, J. Potential Beneficial Effects of Cytomegalovirus Infection after Transplantation. Front. Immunol. 2018, 9, 389. [Google Scholar] [CrossRef] [PubMed]
- Déchanet, J.; Merville, P.; Bergé, F.; Bone-Mane, G.; Taupin, J.L.; Michel, P.; Joly, P.; Bonneville, M.; Potaux, L.; Moreau, J.F. Major expansion of gammadelta T lymphocytes following cytomegalovirus infection in kidney allograft recipients. J. Infect. Dis. 1999, 179, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dechanet, J.; Merville, P.; Lim, A.; Retiere, C.; Pitard, V.; Lafarge, X.; Michelson, S.; Meric, C.; Hallet, M.M.; Kourilsky, P.; et al. Implication of gammadelta T cells in the human immune response to cytomegalovirus. J. Clin. Investig. 1999, 103, 1437–1449. [Google Scholar] [CrossRef]
- Pitard, V.; Roumanes, D.; Lafarge, X.; Couzi, L.; Garrigue, I.; Lafon, M.E.; Merville, P.; Moreau, J.F.; Dechanet-Merville, J. Long-term expansion of effector/memory Vdelta2-gammadelta T cells is a specific blood signature of CMV infection. Blood 2008, 112, 1317–1324. [Google Scholar] [CrossRef]
- Kaminski, H.; Ménard, C.; El Hayani, B.; Adjibabi, A.N.; Marsères, G.; Courant, M.; Zouine, A.; Pitard, V.; Garrigue, I.; Burrel, S.; et al. Characterization of a Unique γδ T-Cell Subset as a Specific Marker of Cytomegalovirus Infection Severity. J. Infect. Dis. 2021, 223, 655–666. [Google Scholar] [CrossRef]
- Styczynski, J.; van der Velden, W.; Fox, C.P.; Engelhard, D.; de la Camara, R.; Cordonnier, C.; Ljungman, P. Management of Epstein-Barr Virus infections and post-transplant lymphoproliferative disorders in patients after allogeneic hematopoietic stem cell transplantation: Sixth European Conference on Infections in Leukemia (ECIL-6) guidelines. Haematologica 2016, 101, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Bian, Z.; Wang, X.; Xu, L.P.; Fu, Q.; Wang, C.; Chang, Y.J.; Wang, Y.; Zhang, X.H.; Jiang, Z.; et al. Inverse correlation of Vdelta2(+) T-cell recovery with EBV reactivation after haematopoietic stem cell transplantation. Br. J. Haematol. 2018, 180, 276–285. [Google Scholar] [CrossRef]
- Xiang, Z.; Liu, Y.; Zheng, J.; Liu, M.; Lv, A.; Gao, Y.; Hu, H.; Lam, K.T.; Chan, G.C.; Yang, Y.; et al. Targeted activation of human Vgamma9Vdelta2-T cells controls epstein-barr virus-induced B cell lymphoproliferative disease. Cancer Cell 2014, 26, 565–576. [Google Scholar] [CrossRef]
- Djaoud, Z.; Guethlein, L.A.; Horowitz, A.; Azzi, T.; Nemat-Gorgani, N.; Olive, D.; Nadal, D.; Norman, P.J.; Munz, C.; Parham, P. Two alternate strategies for innate immunity to Epstein-Barr virus: One using NK cells and the other NK cells and gammadelta T cells. J. Exp. Med. 2017, 214, 1827–1841. [Google Scholar] [CrossRef]
- De Paoli, P.; Gennari, D.; Martelli, P.; Cavarzerani, V.; Comoretto, R.; Santini, G. Gamma delta T cell receptor-bearing lymphocytes during Epstein-Barr virus infection. J. Infect. Dis. 1990, 161, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Farnault, L.; Gertner-Dardenne, J.; Gondois-Rey, F.; Michel, G.; Chambost, H.; Hirsch, I.; Olive, D. Clinical evidence implicating gamma-delta T cells in EBV control following cord blood transplantation. Bone Marrow Transplant. 2013, 48, 1478–1479. [Google Scholar] [CrossRef] [PubMed]
- Barcy, S.; De Rosa, S.C.; Vieira, J.; Diem, K.; Ikoma, M.; Casper, C.; Corey, L. Gamma delta+ T cells involvement in viral immune control of chronic human herpesvirus 8 infection. J. Immunol. 2008, 180, 3417–3425. [Google Scholar] [CrossRef] [PubMed]
- Bukowski, J.F.; Morita, C.T.; Brenner, M.B. Recognition and destruction of virus-infected cells by human gamma delta CTL. J. Immunol. 1994, 153, 5133–5140. [Google Scholar]
- Verjans, G.M.; Roest, R.W.; van der Kooi, A.; van Dijk, G.; van der Meijden, W.I.; Osterhaus, A. Isopentenyl pyrophosphate-reactive Vgamma9Vdelta 2 T helper 1-like cells are the major gammadelta T cell subset recovered from lesions of patients with genital herpes. J. Infect. Dis. 2004, 190, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Juno, J.A.; Eriksson, E.M. γδ T-cell responses during HIV infection and antiretroviral therapy. Clin. Transl. Immunol. 2019, 8, e01069. [Google Scholar] [CrossRef]
- Carissimo, G.; Xu, W.; Kwok, I.; Abdad, M.Y.; Chan, Y.H.; Fong, S.W.; Puan, K.J.; Lee, C.Y.; Yeo, N.K.; Amrun, S.N.; et al. Whole blood immunophenotyping uncovers immature neutrophil-to-VD2 T-cell ratio as an early marker for severe COVID-19. Nat. Commun. 2020, 11, 5243. [Google Scholar] [CrossRef] [PubMed]
- Laing, A.G.; Lorenc, A.; Del Molino Del Barrio, I.; Das, A.; Fish, M.; Monin, L.; Muñoz-Ruiz, M.; McKenzie, D.R.; Hayday, T.S.; Francos-Quijorna, I.; et al. A dynamic COVID-19 immune signature includes associations with poor prognosis. Nat. Med. 2020, 26, 1623–1635. [Google Scholar] [CrossRef]
- Li, H.; Xiang, Z.; Feng, T.; Li, J.; Liu, Y.; Fan, Y.; Lu, Q.; Yin, Z.; Yu, M.; Shen, C.; et al. Human Vγ9Vδ2-T cells efficiently kill influenza virus-infected lung alveolar epithelial cells. Cell. Mol. Immunol. 2013, 10, 159–164. [Google Scholar] [CrossRef]
- Babbe, H.; Chester, N.; Leder, P.; Reizis, B. The Bloom’s syndrome helicase is critical for development and function of the alphabeta T-cell lineage. Mol. Cell Biol. 2007, 27, 1947–1959. [Google Scholar] [CrossRef]
- Cazzetta, V.; Bruni, E.; Terzoli, S.; Carenza, C.; Franzese, S.; Piazza, R.; Marzano, P.; Donadon, M.; Torzilli, G.; Cimino, M.; et al. NKG2A expression identifies a subset of human Vδ2 T cells exerting the highest antitumor effector functions. Cell Rep. 2021, 37, 109871. [Google Scholar] [CrossRef]
- Di Lorenzo, B.; Simões, A.E.; Caiado, F.; Tieppo, P.; Correia, D.V.; Carvalho, T.; da Silva, M.G.; Déchanet-Merville, J.; Schumacher, T.N.; Prinz, I.; et al. Broad Cytotoxic Targeting of Acute Myeloid Leukemia by Polyclonal Delta One T Cells. Cancer Immunol. Res. 2019, 7, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.R.; Correia, D.V.; Fernandes-Platzgummer, A.; da Silva, C.L.; da Silva, M.G.; Anjos, D.R.; Silva-Santos, B. Delta One T Cells for Immunotherapy of Chronic Lymphocytic Leukemia: Clinical-Grade Expansion/Differentiation and Preclinical Proof of Concept. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2016, 22, 5795–5804. [Google Scholar] [CrossRef] [PubMed]
- Leen, A.M.; Bollard, C.M.; Mendizabal, A.M.; Shpall, E.J.; Szabolcs, P.; Antin, J.H.; Kapoor, N.; Pai, S.Y.; Rowley, S.D.; Kebriaei, P.; et al. Multicenter study of banked third-party virus-specific T cells to treat severe viral infections after hematopoietic stem cell transplantation. Blood 2013, 121, 5113–5123. [Google Scholar] [CrossRef]
- van der Veken, L.T.; Hagedoorn, R.S.; van Loenen, M.M.; Willemze, R.; Falkenburg, J.H.; Heemskerk, M.H. Alphabeta T-cell receptor engineered gammadelta T cells mediate effective antileukemic reactivity. Cancer Res. 2006, 66, 3331–3337. [Google Scholar] [CrossRef] [PubMed]
- Dorrie, J.; Krug, C.; Hofmann, C.; Muller, I.; Wellner, V.; Knippertz, I.; Schierer, S.; Thomas, S.; Zipperer, E.; Printz, D.; et al. Human adenovirus-specific gamma/delta and CD8+ T cells generated by T-cell receptor transfection to treat adenovirus infection after allogeneic stem cell transplantation. PLoS ONE 2014, 9, e109944. [Google Scholar] [CrossRef]
- Capsomidis, A.; Benthall, G.; Van Acker, H.H.; Fisher, J.; Kramer, A.M.; Abeln, Z.; Majani, Y.; Gileadi, T.; Wallace, R.; Gustafsson, K.; et al. Chimeric Antigen Receptor-Engineered Human Gamma Delta T Cells: Enhanced Cytotoxicity with Retention of Cross Presentation. Mol. Ther. 2018, 26, 354–365. [Google Scholar] [CrossRef]
- Braham, M.V.J.; Minnema, M.C.; Aarts, T.; Sebestyen, Z.; Straetemans, T.; Vyborova, A.; Kuball, J.; Öner, F.C.; Robin, C.; Alblas, J. Cellular immunotherapy on primary multiple myeloma expanded in a 3D bone marrow niche model. Oncoimmunology 2018, 7, e1434465. [Google Scholar] [CrossRef]
- Straetemans, T.; Kierkels, G.J.J.; Doorn, R.; Jansen, K.; Heijhuurs, S.; Dos Santos, J.M.; van Muyden, A.D.D.; Vie, H.; Clemenceau, B.; Raymakers, R.; et al. GMP-Grade Manufacturing of T Cells Engineered to Express a Defined γδTCR. Front. Immunol. 2018, 9, 1062. [Google Scholar] [CrossRef]
- Grunder, C.; van, D.S.; Hol, S.; Drent, E.; Straetemans, T.; Heijhuurs, S.; Scholten, K.; Scheper, W.; Sebestyen, Z.; Martens, A.; et al. gamma9 and delta2CDR3 domains regulate functional avidity of T cells harboring gamma9delta2TCRs. Blood 2012, 120, 5153–5162. [Google Scholar] [CrossRef] [PubMed]
- Marcu-Malina, V.; Heijhuurs, S.; van, B.M.; Hartkamp, L.; Strand, S.; Sebestyen, Z.; Scholten, K.; Martens, A.; Kuball, J. Redirecting alphabeta T cells against cancer cells by transfer of a broadly tumor-reactive gammadeltaT-cell receptor. Blood 2011, 118, 50–59. [Google Scholar] [CrossRef]
- van Diest, E.; Hernández López, P.; Meringa, A.D.; Vyborova, A.; Karaiskaki, F.; Heijhuurs, S.; Gumathi Bormin, J.; van Dooremalen, S.; Nicolasen, M.J.T.; Gatti, L.; et al. Gamma delta TCR anti-CD3 bispecific molecules (GABs) as novel immunotherapeutic compounds. J. Immunother. Cancer 2021, 9, e003850. [Google Scholar] [CrossRef] [PubMed]
- Marty, F.M.; Ljungman, P.; Chemaly, R.F.; Maertens, J.; Dadwal, S.S.; Duarte, R.F.; Haider, S.; Ullmann, A.J.; Katayama, Y.; Brown, J.; et al. Letermovir Prophylaxis for Cytomegalovirus in Hematopoietic-Cell Transplantation. N. Engl. J. Med. 2017, 377, 2433–2444. [Google Scholar] [CrossRef] [PubMed]
- Gratwohl, A.; Sureda, A.; Baldomero, H.; Gratwohl, M.; Dreger, P.; Kröger, N.; Ljungman, P.; McGrath, E.; Mohty, M.; Nagler, A.; et al. Economics and Outcome After Hematopoietic Stem Cell Transplantation: A Retrospective Cohort Study. eBioMedicine 2015, 2, 2101–2109. [Google Scholar] [CrossRef]
- Chabannon, C.; Kuball, J.; Bondanza, A.; Dazzi, F.; Pedrazzoli, P.; Toubert, A.; Ruggeri, A.; Fleischhauer, K.; Bonini, C. Hematopoietic stem cell transplantation in its 60s: A platform for cellular therapies. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef]
- Chabannon, C.; Kuball, J.; McGrath, E.; Bader, P.; Dufour, C.; Lankester, A.; Basak, G.W.; Montoto, S.; Nagler, A.; Snowden, J.A.; et al. CAR-T cells: The narrow path between hope and bankruptcy? Bone Marrow Transplant. 2017, 52, 1588–1589. [Google Scholar] [CrossRef] [PubMed]
- Traversari, C.; Marktel, S.; Magnani, Z.; Mangia, P.; Russo, V.; Ciceri, F.; Bonini, C.; Bordignon, C. The potential immunogenicity of the TK suicide gene does not prevent full clinical benefit associated with the use of TK-transduced donor lymphocytes in HSCT for hematologic malignancies. Blood 2007, 109, 4708–4715. [Google Scholar] [CrossRef]
| Study | Patients | Donor | Intervention | Numbers | CMV | EBV | BK | Adeno |
|---|---|---|---|---|---|---|---|---|
| ATG | ||||||||
| Chang et al. [2] | Adult hematological malignancies | MRD | ATG-T | 263 | Day 100: 22.7% | Day 180: 7.8% | n.a. | n.a. |
| Walker et al. [3] | Adult hematological malignancies | MUD MMUD | ATG-T | 101 | n.a. | 20% DNAemia requiring therapy | n.a. | n.a. |
| Finke et al. [4] | Adult hematological malignancies | MRD MUD | ATG-F | 103 | 53.8% DNAemia 5.7% CMV disease | 5% PTLD | n.a. | n.a. |
| Soiffer et al. [5] | Adult AML, MDS, and ALL | MUD | ATG-F | 126 | 62% (R+) DNAemie | 1.6% PTLD | n.a. | n.a. |
| Alemtuzumab | ||||||||
| Green et al. [6] | Adult hematological malignancies | Matched Mismatched | Alemtuzumab | 313 | >80% (R+) DNAemia | n.a. | n.a. | n.a. |
| Carpenter et al. [7] | Adult hematological malignancies | MRD MMRD MUD MMUD | Alemtuzumab | 111 | n.a. | 2Y 40.3% DNAemia 1% PTLD | n.a. | n.a. |
| PTCy | ||||||||
| Cieri et al. [8] | Adult high risk hematological malignancy | Haplo | PTCy | 40 | 63% DNAemia 17% CMV disease | 15% DNAemia (66% of these pts treated). No PTLD | 18% | n.a. |
| Berger et al. [9] | Pediatric; high risk hematological malignancy | Haplo | PTCy | 33 | 36% DNAemia No CMV disease | 3% DNAemia No PTLD | 17% | 3% DNAemia; Not symptomatic |
| Retiere et al. [10] | Adult hematological malignancies | MRD MUD MMUD haplo | PTCy vs. ATG-T | 45 | DNAemia PTCY 27% ATG 40% | DNAemia requiring treatment PTCY 0% ATG 33% | PTCY 3%ATG 0% | PTCY 15% ATG 20% |
| αβT cell depletion | ||||||||
| De Witte et al. [13] | Adult hematological malignancies | MRD MUD MMUD | αβT cell depletion | 35 | 64% (R+) DNAemia 6% CMV disease | 44% | n.a. | n.a. |
| Laberko et al. [14] | Pediatric malignant + non-malignant | MUD haplo | αβT cell/CD19 depletion | 182 | 51% | 33% | n.a. | n.a. |
| Maschan et al. [15] | Pediatric high-risk AML | MUD MMUD Haplo | αβT cell/CD19 depletion | 33 | 52% DNAemia 6% CMV disease | 50% DNAemia; 6% Rituximab | n.a. | n.a. |
| Bertaina et al. [16] | Pediatric non-malignant | Haplo | αβT cell/CD19 depletion | 23 | 38% DNAemia CMV/adeno | 50% DNAemia; 6% Rituximab | n.a. | 38% DNAemia CMV/adeno |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janssen, A.; van Diest, E.; Vyborova, A.; Schrier, L.; Bruns, A.; Sebestyen, Z.; Straetemans, T.; de Witte, M.; Kuball, J. The Role of γδ T Cells as a Line of Defense in Viral Infections after Allogeneic Stem Cell Transplantation: Opportunities and Challenges. Viruses 2022, 14, 117. https://doi.org/10.3390/v14010117
Janssen A, van Diest E, Vyborova A, Schrier L, Bruns A, Sebestyen Z, Straetemans T, de Witte M, Kuball J. The Role of γδ T Cells as a Line of Defense in Viral Infections after Allogeneic Stem Cell Transplantation: Opportunities and Challenges. Viruses. 2022; 14(1):117. https://doi.org/10.3390/v14010117
Chicago/Turabian StyleJanssen, Anke, Eline van Diest, Anna Vyborova, Lenneke Schrier, Anke Bruns, Zsolt Sebestyen, Trudy Straetemans, Moniek de Witte, and Jürgen Kuball. 2022. "The Role of γδ T Cells as a Line of Defense in Viral Infections after Allogeneic Stem Cell Transplantation: Opportunities and Challenges" Viruses 14, no. 1: 117. https://doi.org/10.3390/v14010117
APA StyleJanssen, A., van Diest, E., Vyborova, A., Schrier, L., Bruns, A., Sebestyen, Z., Straetemans, T., de Witte, M., & Kuball, J. (2022). The Role of γδ T Cells as a Line of Defense in Viral Infections after Allogeneic Stem Cell Transplantation: Opportunities and Challenges. Viruses, 14(1), 117. https://doi.org/10.3390/v14010117






