The Nature of Immune Responses to Influenza Vaccination in High-Risk Populations
Abstract
1. Introduction
2. Influenza Vaccine Overview
Standard-of-Care Vaccines
3. Responses to Influenza Vaccination
3.1. Humoral Responses
3.2. Cell-Mediated Responses
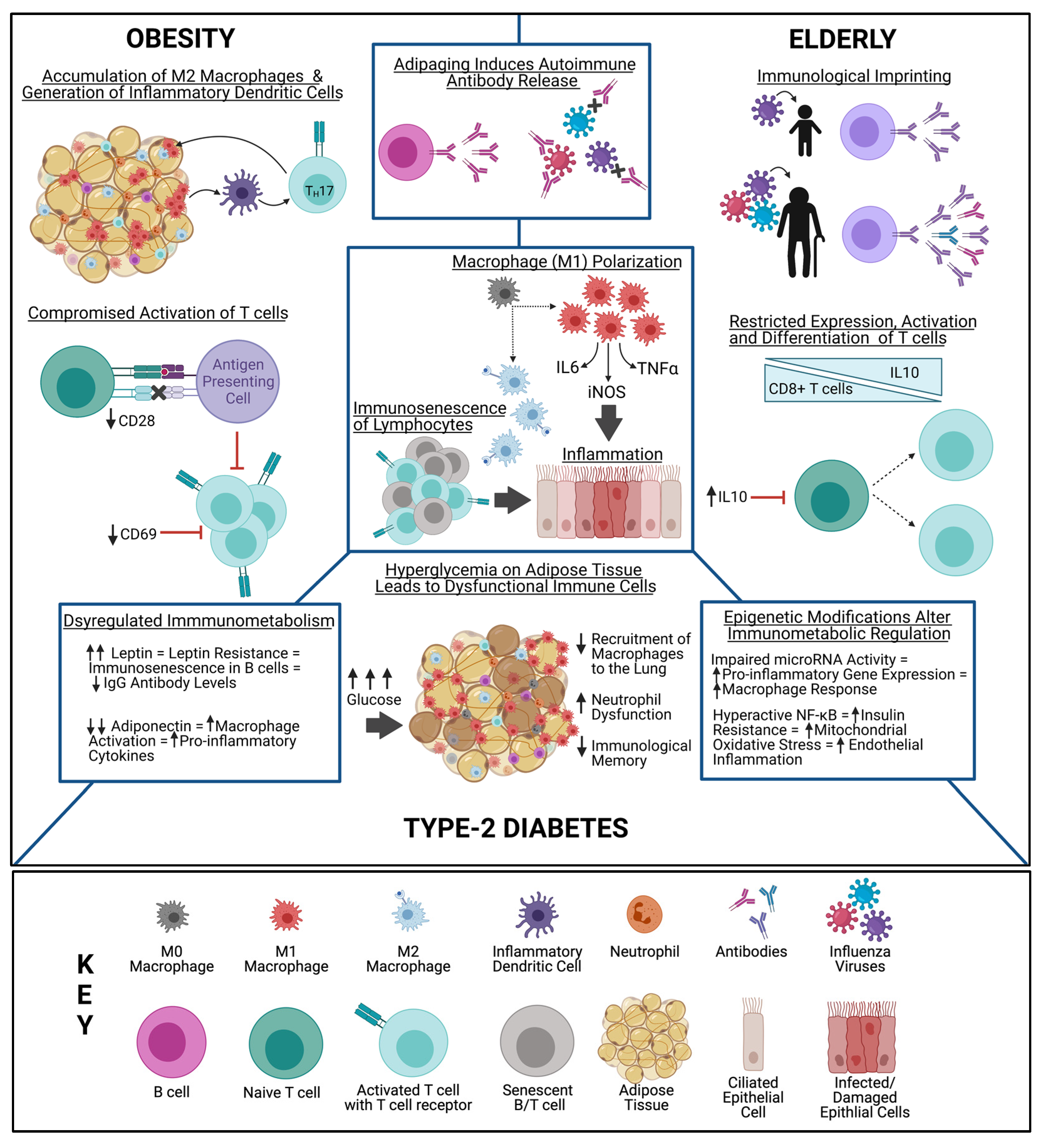
4. Unique Immune Environments in High-Risk Populations
4.1. Metabolic Syndrome
4.2. Adipose Tissue
4.3. B Cells
4.4. T Cells
4.5. Important Metabolic Markers
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Hodgson, S.H.; Mansatta, K.; Mallett, G.; Harris, V.; Emary, K.R.W.; Pollard, A.J. What defines an efficacious COVID-19 vaccine? A review of the challenges assessing the clinical efficacy of vaccines against SARS-CoV-2. Lancet Infect. Dis. 2021, 21, e26–e35. [Google Scholar] [CrossRef]
- Shervani, Z.; Khan, I.; Khan, T.; Qazi, U.Y. COVID-19 Vaccine. Adv. Infect. Dis. 2020, 10, 195–210. [Google Scholar] [CrossRef]
- Coustasse, A.; Kimble, C.; Maxik, K. COVID-19 and Vaccine Hesitancy: A Challenge the United States Must Overcome. J. Ambul. Care Manag. 2021, 44, 71–75. [Google Scholar] [CrossRef]
- Honce, R.; Schultz-Cherry, S. Impact of Obesity on Influenza a Virus Pathogenesis, Immune Response, and Evolution. Front. Immunol. 2019, 10, 1071. [Google Scholar] [CrossRef] [PubMed]
- Kass, D.A.; Duggal, P.; Cingolani, O. Obesity could shift severe COVID-19 disease to younger ages. Lancet 2020, 395, 1544–1545. [Google Scholar] [CrossRef]
- Gao, F.; Zheng, K.; Wang, X.-B.; Sun, Q.-F.; Pan, K.-H.; Wang, T.-Y.; Chen, Y.-P.; Targher, G.; Byrne, C.D.; George, J.; et al. Obesity Is a Risk Factor for Greater COVID-19 Severity. Diabetes Care 2020, 43, e72–e74. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.; Garg, S.; O’Halloran, A.; Whitaker, M.; Pham, H.; Anderson, E.J.; Armistead, I.; Bennett, N.M.; Billing, L.; Como-Sabetti, K.; et al. Risk Factors for Intensive Care Unit Admission and In-hospital Mortality Among Hospitalized Adults Identified through the US Coronavirus Disease 2019 (COVID-19)-Associated Hospitalization Surveillance Network (COVID-NET). Clin. Infect. Dis. 2021, 72, e206–e214. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Influenza (Seasonal). Available online: https://www.who.int/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 20 February 2021).
- Yan, S.; Weycker, D.; Sokolowski, S. US healthcare costs attributable to type A and type B influenza. Hum. Vaccines Immunother. 2017, 13, 2041–2047. Available online: https://pubmed.ncbi.nlm.nih.gov/28700268/ (accessed on 11 January 2021). [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Past Seasons Estimated Influenza Disease Burden. 2020. Available online: https://www.cdc.gov/flu/about/burden/past-seasons.html (accessed on 11 January 2021).
- Honce, R.; Karlsson, E.A.; Wohlgemuth, N.; Estrada, L.D.; Meliopoulos, V.A.; Yao, J.; Schultz-Cherry, S. Obesity-Related Microenvironment Promotes Emergence of Virulent Influenza Virus Strains. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Honce, R.; Wohlgemuth, N.; Meliopoulos, V.A.; Short, K.R.; Schultz-Cherry, S. Influenza in High-Risk Hosts—Lessons Learned from Animal Models. Cold Spring Harb. Perspect. Med. 2020, 10, 038604. [Google Scholar] [CrossRef]
- Neidich, S.D.; Green, W.D.; Rebeles, J.; Karlsson, E.A.; Schultz-Cherry, S.; Noah, T.L.; Chakladar, S.; Hudgens, M.G.; Weir, S.S.; Beck, M.A. Increased risk of influenza among vaccinated adults who are obese. Int. J. Obes. 2017, 41, 1324–1330. [Google Scholar] [CrossRef]
- Harris, J.A.; Moniz, M.H.; Iott, B.; Power, R.; Griggs, J.J. Obesity and the receipt of influenza and pneumococcal vaccination: A systematic review and meta-analysis. BMC Obes. 2016, 3, 24. [Google Scholar] [CrossRef]
- Karlsson, E.A.; Hertz, T.; Johnson, C.; Mehle, A.; Krammer, F.; Schultz-Cherry, S. Obesity Outweighs Protection Conferred by Adjuvanted Influenza Vaccination. mBio 2016, 7, e01144-16. [Google Scholar] [CrossRef]
- Park, H.-L.; Shim, S.-H.; Lee, E.-Y.; Cho, W.; Park, S.; Jeon, H.-J.; Ahn, S.-Y.; Kim, H.; Nam, J.-H. Obesity-induced chronic inflammation is associated with the reduced efficacy of influenza vaccine. Hum. Vaccines Immunother. 2014, 10, 1181–1186. [Google Scholar] [CrossRef]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Ward, Z.J.; Bleich, S.N.; Cradock, A.L.; Barrett, J.L.; Giles, C.M.; Flax, C.; Long, M.W.; Gortmaker, S.L. Projected U.S. State-Level Prevalence of Adult Obesity and Severe Obesity. N. Engl. J. Med. 2019, 381, 2440–2450. [Google Scholar] [CrossRef] [PubMed]
- Allard, R.; Leclerc, P.; Tremblay, C.; Tannenbaum, T.-N. Diabetes and the Severity of Pandemic Influenza A (H1N1) Infection. Diabetes Care 2010, 33, 1491–1493. [Google Scholar] [CrossRef] [PubMed]
- Marshall, R.J.; Armart, P.; Hulme, K.; Chew, K.Y.; Brown, A.C.; Hansbro, P.; Bloxham, C.J.; Flint, M.; Ronacher, K.; Bielefeldt-Ohmann, H.; et al. Glycemic Variability in Diabetes Increases the Severity of Influenza. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Verstraeten, T.; Fletcher, M.A.; Suaya, J.A.; Jackson, S.; Hall-Murray, C.K.; Scott, D.A.; Schmöle-Thoma, B.; Isturiz, R.E.; Gessner, B.D. Diabetes mellitus as a vaccine-effect modifier: A review. Expert Rev. Vaccines 2020, 19, 445–453. [Google Scholar] [CrossRef]
- Chen, L.; Magliano, D.; Zimmet, P.Z. The worldwide epidemiology of type 2 diabetes mellitus—Present and future perspectives. Nat. Rev. Endocrinol. 2011, 8, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.B.; Hashim, M.J.; King, J.K.; Govender, R.D.; Mustafa, H.; Al Kaabi, J. Epidemiology of Type 2 Diabetes—Global Burden of Disease and Forecasted Trends. J. Epidemiol. Glob. Health 2020, 10, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, K.; Wei, Y.; Szwajcer, A.; Rabbani, R.; Zarychanski, R.; Abou-Setta, A.M.; Mahmud, S.M. Efficacy and safety of high-dose influenza vaccine in elderly adults: A systematic review and meta-analysis. Vaccine 2017, 35, 2775–2780. [Google Scholar] [CrossRef] [PubMed]
- Domnich, A.; Arata, L.; Amicizia, D.; Puig-Barberà, J.; Gasparini, R.; Panatto, D. Effectiveness of MF59-adjuvanted seasonal influenza vaccine in the elderly: A systematic review and meta-analysis. Vaccine 2017, 35, 513–520. [Google Scholar] [CrossRef]
- Simonsen, L.; Taylor, R.J.; Viboud, C.; Miller, M.A.; Jackson, L.A. Mortality benefits of influenza vaccination in elderly people: An ongoing controversy. Lancet Infect. Dis. 2007, 7, 658–666. [Google Scholar] [CrossRef]
- Goodwin, K.; Viboud, C.; Simonsen, L. Antibody response to influenza vaccination in the elderly: A quantitative review. Vaccine 2006, 24, 1159–1169. [Google Scholar] [CrossRef] [PubMed]
- CDC. Seasonal Flu Shot. Available online: https://www.cdc.gov/flu/prevent/flushot.htm (accessed on 2 April 2021).
- Trombetta, C.M.; Gianchecchi, E.; Montomoli, E. Influenza vaccines: Evaluation of the safety profile. Hum. Vaccines Immunother. 2018, 14, 657–670. [Google Scholar] [CrossRef]
- Kumar, A.; Meldgaard, T.S.; Bertholet, S. Novel Platforms for the Development of a Universal Influenza Vaccine. Front. Immunol. 2018, 9, 600. [Google Scholar] [CrossRef]
- Krammer, F.; Smith, G.J.D.; Fouchier, R.A.M.; Peiris, M.; Kedzierska, K.; Doherty, P.C.; Palese, P.; Shaw, M.L.; Treanor, J.; Webster, R.G.; et al. Influenza. Nat. Rev. Dis. Prim. 2018, 4, 3. [Google Scholar] [CrossRef]
- Wong, S.-S.; Webby, R.J. Traditional and New Influenza Vaccines. Clin. Microbiol. Rev. 2013, 26, 476–492. [Google Scholar] [CrossRef] [PubMed]
- Erbelding, E.J.; Post, D.J.; Stemmy, E.J.; Roberts, P.C.; Augustine, A.D.; Ferguson, S.; Paules, C.I.; Graham, B.S.; Fauci, A.S. A Universal Influenza Vaccine: The Strategic Plan for the National Institute of Allergy and Infectious Diseases. J. Infect. Dis. 2018, 218, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Rothaeusler, K.; Baumgarth, N. B-cell fate decisions following influenza virus infection. Eur. J. Immunol. 2010, 40, 366–377. [Google Scholar] [CrossRef]
- Dugan, H.L.; Henry, C.; Wilson, P.C. Aging and influenza vaccine-induced immunity. Cell. Immunol. 2020, 348, 103998. [Google Scholar] [CrossRef] [PubMed]
- Janeway, J.C.A.; Travers, P.; Walport, M.; Shlomchik, M.J. Immunobiology: The Immune System in Health and Disease, 5th ed.; Garland Science: New York, NY, USA, 2001. [Google Scholar]
- Kosaraju, R.; Guesdon, W.; Crouch, M.J.; Teague, H.L.; Sullivan, E.M.; Karlsson, E.A.; Schultz-Cherry, S.; Gowdy, K.; Bridges, L.C.; Reese, L.R.; et al. B Cell Activity Is Impaired in Human and Mouse Obesity and Is Responsive to an Essential Fatty Acid upon Murine Influenza Infection. J. Immunol. 2017, 198, 4738–4752. [Google Scholar] [CrossRef] [PubMed]
- Frasca, D.; Diaz, A.; Romero, M.; Blomberg, B.B. Leptin induces immunosenescence in human B cells. Cell. Immunol. 2020, 348, 103994. [Google Scholar] [CrossRef]
- Wang, T.T.; Maamary, J.; Tan, G.S.; Bournazos, S.; Davis, C.W.; Krammer, F.; Schlesinger, S.J.; Palese, P.; Ahmed, R.; Ravetch, J.V. Anti-HA Glycoforms Drive B Cell Affinity Selection and Determine Influenza Vaccine Efficacy. Cell 2015, 162, 160–169. [Google Scholar] [CrossRef]
- Wang, T.T.; Bournazos, S.; Ravetch, J.V. Immunological responses to influenza vaccination: Lessons for improving vaccine efficacy. Curr. Opin. Immunol. 2018, 53, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Maamary, J.; Wang, T.T.; Tan, G.S.; Palese, P.; Ravetch, J.V. Increasing the breadth and potency of response to the seasonal influenza virus vaccine by immune complex immunization. Proc. Natl. Acad. Sci. USA 2017, 114, 10172–10177. [Google Scholar] [CrossRef] [PubMed]
- Lofano, G.; Gorman, M.J.; Yousif, A.S.; Yu, W.-H.; Fox, J.M.; Dugast, A.-S.; Ackerman, M.E.; Suscovich, T.J.; Weiner, J.; Barouch, D.; et al. Antigen-specific antibody Fc glycosylation enhances humoral immunity via the recruitment of complement. Sci. Immunol. 2018, 3, eaat7796. [Google Scholar] [CrossRef] [PubMed]
- Khurana, S.; Hahn, M.; Coyle, E.M.; King, L.R.; Lin, T.-L.; Treanor, J.; Sant, A.; Golding, H. Repeat vaccination reduces antibody affinity maturation across different influenza vaccine platforms in humans. Nat. Commun. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Sant, A.J. The Way Forward: Potentiating Protective Immunity to Novel and Pandemic Influenza Through Engagement of Memory CD4 T Cells. J. Infect. Dis. 2019, 219 (Suppl. 1), S30–S37. [Google Scholar] [CrossRef] [PubMed]
- Souquette, A.; Thomas, P.G. Past Life and Future Effects—How Heterologous Infections Alter Immunity to Influenza Viruses. Front. Immunol. 2018, 9, 1071. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.; Apovian, C. Macrophage functions in lean and obese adipose tissue. Metabolism 2017, 72, 120–143. [Google Scholar] [CrossRef]
- Lumeng, C.; Bodzin, J.L.; Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Investig. 2007, 117, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, C.; Liu, J.; Geletka, L.; Delaney, C.; DelProposto, J.; Desai, A.; Oatmen, K.; Martinez-Santibanez, G.; Julius, A.; Garg, S.; et al. Aging Is Associated with an Increase in T Cells and Inflammatory Macrophages in Visceral Adipose Tissue. J. Immunol. 2011, 187, 6208–6216. [Google Scholar] [CrossRef]
- Damsker, J.M.; Hansen, A.M.; Caspi, R.R. Th1 and Th17 cells. Ann. N. Y. Acad. Sci. 2010, 1183, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.W.; Zamarron, B.F.; Muir, L.A.; Singer, K.; Porsche, C.E.; DelProposto, J.B.; Geletka, L.; Meyer, K.A.; O’Rourke, R.W.; Lumeng, C.N. Adipose Tissue Dendritic Cells Are Independent Contributors to Obesity-Induced Inflammation and Insulin Resistance. J. Immunol. 2016, 197, 3650–3661. [Google Scholar] [CrossRef]
- Ma, W.-T.; Gao, F.; Gu, K.; Chen, D.-K. The Role of Monocytes and Macrophages in Autoimmune Diseases: A Comprehensive Review. Front. Immunol. 2019, 10, 1140. [Google Scholar] [CrossRef] [PubMed]
- Maroof, A.; Yorgensen, Y.M.; Li, Y.; Evans, J.T. Intranasal Vaccination Promotes Detrimental Th17-Mediated Immunity against Influenza Infection. PLoS Pathog. 2014, 10, e1003875. [Google Scholar] [CrossRef]
- Mráz, M.; Cinkajzlová, A.; Kloučková, J.; Lacinová, Z.; Kratochvílová, H.; Lipš, M.; Pořízka, M.; Kopecký, P.; Lindner, J.; Kotulák, T.; et al. Dendritic Cells in Subcutaneous and Epicardial Adipose Tissue of Subjects with Type 2 Diabetes, Obesity, and Coronary Artery Disease. Mediat. Inflamm. 2019, 2019, 1–7. [Google Scholar] [CrossRef]
- Gardner, J.K.; Cornwall, S.M.J.; Musk, A.W.; Alvarez, J.; Mamotte, C.D.S.; Jackaman, C.; Nowak, A.K.; Nelson, D.J. Elderly dendritic cells respond to LPS/IFN-γ and CD40L stimulation despite incomplete maturation. PLoS ONE 2018, 13, e0195313. [Google Scholar] [CrossRef] [PubMed]
- Paich, H.A.; Sheridan, P.A.; Handy, J.; Karlsson, E.A.; Schultz-Cherry, S.; Hudgens, M.G.; Noah, T.L.; Weir, S.S.; Beck, M.A. Overweight and obese adult humans have a defective cellular immune response to pandemic H1N1 Influenza a virus. Obesity 2013, 21, 2377–2386. [Google Scholar] [CrossRef] [PubMed]
- Frasca, D.; Blomberg, B.B. The Impact of Obesity and Metabolic Syndrome on Vaccination Success. In Interdisciplinary Topics in Gerontology and Geriatrics; S. Karger AG: Basel, Switzerland, 2020; Volume 43, pp. 86–97. [Google Scholar]
- Pérez, L.M.; Pareja-Galeano, H.; Sanchis-Gomar, F.; Emanuele, E.; Lucia, A.; Gálvez, B.G. ‘Adipaging’: Ageing and obesity share biological hallmarks related to a dysfunctional adipose tissue. J. Physiol. 2016, 594, 3187–3207. [Google Scholar] [CrossRef] [PubMed]
- Ongrádi, J.; Kövesdi, V. Factors that may impact on immunosenescence: An appraisal. Immun. Ageing 2010, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Taherkhani, A.; Kalantari, S.; Oskouie, A.A.; Nafar, M.; Taghizadeh, M.; Tabar, K. Network analysis of membranous glomerulonephritis based on metabolomics data. Mol. Med. Rep. 2018, 18, 4197–4212. [Google Scholar] [CrossRef] [PubMed]
- Frasca, D.; Ferracci, F.; Diaz, A.; Romero, M.; Lechner, S.; Blomberg, B.B. Obesity decreases B cell responses in young and elderly individuals. Obesity 2016, 24, 615–625. [Google Scholar] [CrossRef]
- Gostic, K.M.; Bridge, R.; Brady, S.; Viboud, C.; Worobey, M.; Lloyd-Smith, J.O. Childhood immune imprinting to influenza A shapes birth year-specific risk during seasonal H1N1 and H3N2 epidemics. PLoS Pathog. 2019, 15, e1008109. [Google Scholar] [CrossRef]
- Zhai, X.; Qian, G.; Wang, Y.; Chen, X.; Lu, J.; Zhang, Y.; Huang, Q.; Wang, Q. Elevated B Cell Activation is Associated with Type 2 Diabetes Development in Obese Subjects. Cell. Physiol. Biochem. 2016, 38, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, P.; Curtis, N. Factors That Influence the Immune Response to Vaccination. Clin. Microbiol. Rev. 2019, 32. [Google Scholar] [CrossRef] [PubMed]
- Bertola, A.; Ciucci, T.; Rousseau, D.; Bourlier, V.; Duffaut, C.; Bonnafous, S.; Blin-Wakkach, C.; Anty, R.; Iannelli, A.; Gugenheim, J.; et al. Identification of Adipose Tissue Dendritic Cells Correlated With Obesity-Associated Insulin-Resistance and Inducing Th17 Responses in Mice and Patients. Diabetes 2012, 61, 2238–2247. [Google Scholar] [CrossRef] [PubMed]
- De La Fuente, M.; De Castro, N.M. Obesity as a Model of Premature Immunosenescence. Curr. Immunol. Rev. 2012, 8, 63–75. [Google Scholar] [CrossRef]
- Lau, E.; Carroll, E.C.; Callender, L.A.; Hood, G.A.; Berryman, V.; Pattrick, M.; Finer, S.; Hitman, G.A.; Ackland, G.L.; Henson, S.M. Type 2 diabetes is associated with the accumulation of senescent T cells. Clin. Exp. Immunol. 2019, 197, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Osornio, S.A.; Cruz-Hernández, T.R.; Drago-Serrano, M.E.; Campos-Rodríguez, R. Immunity to influenza: Impact of obesity. Obes. Res. Clin. Pr. 2019, 13, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, C.N.; Saltiel, A.R. Inflammatory links between obesity and metabolic disease. J. Clin. Investig. 2011, 121, 2111–2117. [Google Scholar] [CrossRef] [PubMed]
- Frühbeck, G.; Catalán, V.; Rodríguez, A.; Ramírez, B.; Becerril, S.; Salvador, J.; Portincasa, P.; Colina, I.; Gómez-Ambrosi, J. Involvement of the leptin-adiponectin axis in inflammation and oxidative stress in the metabolic syndrome. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Ricci, R.; Bevilacqua, F. The potential role of leptin and adiponectin in obesity: A comparative review. Veter J. 2012, 191, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Chen, Y.; Heiman, M.; DiMarchi, R. Leptin: Structure, Function and Biology. In Vitamins and Hormones; Academic Press: Cambridge, MA, USA, 2005; Volume 71, pp. 345–372. [Google Scholar]
- Sánchez-Margalet, V.; Martín-Romero, C.; Santos-Alvarez, J.; Goberna, R.; Najib, S.; Gonzalez-Yanes, C. Role of leptin as an immunomodulator of blood mononuclear cells: Mechanisms of action. Clin. Exp. Immunol. 2003, 133, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, E.A.; Sheridan, P.A.; Beck, M.A. Diet-Induced Obesity Impairs the T Cell Memory Response to Influenza Virus Infection. J. Immunol. 2010, 184, 3127–3133. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-Y.; Van De Wall, E.; Laplante, M.; Azzara, A.; Trujillo, M.E.; Hofmann, S.M.; Schraw, T.; Durand, J.L.; Li, H.; Li, G.; et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J. Clin. Investig. 2007, 117, 2621–2637. [Google Scholar] [CrossRef]
- Kramer, C.K.; Zinman, B.; Retnakaran, R. Are Metabolically Healthy Overweight and Obesity Benign Conditions? Ann. Intern. Med. 2013, 159, 758–769. [Google Scholar] [CrossRef] [PubMed]
- Caballero, B. Humans against Obesity: Who Will Win? Adv. Nutr. 2019, 10 (Suppl. 1), S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Hebebrand, J.; Holm, J.-C.; Woodward, E.; Baker, J.L.; Blaak, E.; Schutz, D.D.; Farpour-Lambert, N.J.; Frühbeck, G.; Halford, J.G.; Lissner, L.; et al. A Proposal of the European Association for the Study of Obesity to Improve the ICD-11 Diagnostic Criteria for Obesity Based on the Three Dimensions Etiology, Degree of Adiposity and Health Risk. Obes. Facts 2017, 10, 284–307. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.; Honce, R.; Schultz-Cherry, S. Metabolic Syndrome and Viral Pathogenesis: Lessons from Influenza and Coronaviruses. J. Virol. 2020, 94. [Google Scholar] [CrossRef]
- Bijani, B.; Pahlevan, A.A.; Qasemi-Barqi, R.; Jahanihashemi, H. Metabolic syndrome as an independent risk factor of hypoxaemia in influenza A (H1N1) 2009 pandemic. Infez Med. 2016, 24, 123–130. [Google Scholar] [PubMed]
- Bandaru, P.; Rajkumar, H.; Nappanveettil, G. Altered or Impaired Immune Response to Hepatitis B Vaccine in WNIN/GR-Ob Rat: An Obese Rat Model with Impaired Glucose Tolerance. ISRN Endocrinol. 2011, 2011, 980105. [Google Scholar] [CrossRef] [PubMed]
- Ocak, S.; Eskiocak, A.F. The evaluation of immune responses to hepatitis B vaccination in diabetic and non-diabetic haemodialysis patients and the use of tetanus toxoid. Nephrology 2008, 13, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Eliakim, A.; Schwindt, C.; Zaldivar, F.; Casali, P.; Cooper, D.M. Reduced tetanus antibody titers in overweight children. Autoimmunity 2006, 39, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, P.A.; Paich, H.A.; Handy, J.; Karlsson, E.A.; Hudgens, M.G.; Sammon, A.B.; Holland, L.A.; Weir, S.; Noah, T.L.; Beck, M.A. Obesity is associated with impaired immune response to influenza vaccination in humans. Int. J. Obes. 2011, 36, 1072–1077. [Google Scholar] [CrossRef]
- Andersen, C.J.; Murphy, K.E.; Fernandez, M.L. Impact of Obesity and Metabolic Syndrome on Immunity. Adv. Nutr. 2016, 7, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Saely, C.H.; Geiger, K.; Drexel, H. Brown versus White Adipose Tissue: A Mini-Review. Gerontology 2012, 58, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Villarroya, F.; Cereijo, R.; Gavaldà-Navarro, A.; Villarroya, J.; Giralt, M. Inflammation of brown/beige adipose tissues in obesity and metabolic disease. J. Intern. Med. 2018, 284, 492–504. [Google Scholar] [CrossRef]
- Lee, B.-C.; Lee, J. Cellular and molecular players in adipose tissue inflammation in the development of obesity-induced insulin resistance. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2014, 1842, 446–462. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, P.; Liu, Y.; Liu, W.; Zha, L.; Chen, X.; Zheng, R.; Qi, K.; Zhang, Y. Maternal vitamin D deficiency increases the risk of obesity in male offspring mice by affecting the immune response. Nutrition 2021, 87–88, 111191. [Google Scholar] [CrossRef] [PubMed]
- Blaner, W.S. Vitamin A signaling and homeostasis in obesity, diabetes, and metabolic disorders. Pharmacol. Ther. 2019, 197, 153–178. [Google Scholar] [CrossRef]
- Penkert, R.R.; Cortez, V.; Karlsson, E.A.; Livingston, B.; Surman, S.L.; Li, Y.; Ross, A.C.; Schultz-Cherry, S.; Hurwitz, J.L. Vitamin A Corrects Tissue Deficits in Diet-Induced Obese Mice and Reduces Influenza Infection After Vaccination and Challenge. Obesity 2020, 28, 1631–1636. [Google Scholar] [CrossRef] [PubMed]
- Funcke, J.-B.; Scherer, P.E. Beyond adiponectin and leptin: Adipose tissue-derived mediators of inter-organ communication. J. Lipid Res. 2019, 60, 1648–1697. [Google Scholar] [CrossRef]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, S.; Manabe, I.; Nagasaki, M.; Hosoya, Y.; Yamashita, H.; Fujita, H.; Ohsugi, M.; Tobe, K.; Kadowaki, T.; Nagai, R.; et al. Adipogenesis in Obesity Requires Close Interplay Between Differentiating Adipocytes, Stromal Cells, and Blood Vessels. Diabetes 2007, 56, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Tsibane, T.; McGraw, P.A.; House, F.S.; Keefer, C.J.; Hicar, M.D.; Tumpey, T.M.; Pappas, C.; Perrone, L.A.; Martinez, O.; et al. Neutralizing antibodies derived from the B cells of 1918 influenza pandemic survivors. Nature 2008, 455, 532–536. [Google Scholar] [CrossRef]
- Meade, P.; Kuan, G.; Strohmeier, S.; Maier, H.E.; Amanat, F.; Balmaseda, A.; Ito, K.; Kirkpatrick, E.; Javier, A.; Gresh, L.; et al. Influenza Virus Infection Induces a Narrow Antibody Response in Children but a Broad Recall Response in Adults. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.; Ross, T.M. Preexisting influenza specific immunity and vaccine effectiveness. Expert Rev. Vaccines 2019, 18, 1043–1051. [Google Scholar] [CrossRef]
- Diaz, A.; Romero, M.; Vazquez, T.; Lechner, S.; Blomberg, B.B.; Frasca, D. Metformin improves in vivo and in vitro B cell function in individuals with obesity and Type-2 Diabetes. Vaccine 2017, 35, 2694–2700. [Google Scholar] [CrossRef]
- Ruderman, N.; Prentki, M. AMP kinase and malonyl-CoA: Targets for therapy of the metabolic syndrome. Nat. Rev. Drug Discov. 2004, 3, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Riejos, P.; Najib, S.; Santos-Alvarez, J.; Martín-Romero, C.; Pérez, A.P.; González-Yanes, C.; Sánchez-Margalet, V. Role of Leptin in the Activation of Immune Cells. Mediat. Inflamm. 2010, 2010, 568343. [Google Scholar] [CrossRef] [PubMed]
- La Cava, A.; Matarese, G. The weight of leptin in immunity. Nat. Rev. Immunol. 2004, 4, 371–379. [Google Scholar] [CrossRef]
- Ovsyannikova, I.G.; White, S.J.; Larrabee, B.R.; Grill, D.E.; Jacobson, R.M.; Poland, G.A. Leptin and leptin-related gene polymorphisms, obesity, and influenza A/H1N1 vaccine-induced immune responses in older individuals. Vaccine 2014, 32, 881–887. [Google Scholar] [CrossRef][Green Version]
- Guzik, T.J.; Cosentino, F. Epigenetics and Immunometabolism in Diabetes and Aging. Antioxidants Redox Signal. 2018, 29, 257–274. [Google Scholar] [CrossRef] [PubMed]
- Zacharioudaki, V.; Androulidaki, A.; Arranz, A.; Vrentzos, G.; Margioris, A.N.; Tsatsanis, C. Adiponectin Promotes Endotoxin Tolerance in Macrophages by Inducing IRAK-M Expression. J. Immunol. 2009, 182, 6444–6451. [Google Scholar] [CrossRef] [PubMed]
- Zavalza-Gómez, A.B.; Anaya-Prado, R.; Rincón-Sánchez, A.R.; Mora-Martínez, J.M. Adipokines and insulin resistance during pregnancy. Diabetes Res. Clin. Pr. 2008, 80, 8–15. [Google Scholar] [CrossRef]
- Pharmacovigilance, V. Definition and Application of Terms for Vaccine Pharmacovigilance; Council for International Organizations of Medical Sciences (CIOMS): Geneva, Switzerland, 2005. [Google Scholar]
- Kuo, H.; Shapiro, J.R.; Dhakal, S.; Morgan, R.; Fink, A.L.; Lui, H.; Westerbeck, J.W.; Sylvia, K.E.; Park, H.-S.; Ursin, R.L.; et al. Sex-specific effects of age and body mass index on antibody responses to seasonal influenza vaccines in healthcare workers. Vaccine 2021. [Google Scholar] [CrossRef] [PubMed]
- Ajana, F.; Sana, C.; Caulin, E. Existe-t-il des différences d’immunogénicité et de tolérance des vaccins en fonction du mode d’injection ? Médecine Mal. Infect. 2008, 38, 648–657. [Google Scholar] [CrossRef]
- Klein, S.L.; Pekosz, A. Sex-based Biology and the Rational Design of Influenza Vaccination Strategies. J. Infect. Dis. 2014, 209, S114–S119. [Google Scholar] [CrossRef] [PubMed]
- Roubidoux, E.K.; Carreño, J.M.; McMahon, M.; Jiang, K.; van Bakel, H.; Wilson, P.; Krammer, F. Mutations in the Hemagglutinin Stalk Domain Do Not Permit Escape from a Protective, Stalk-Based Vaccine-Induced Immune Response in the Mouse Model. mBio 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-Q.; Wohlbold, T.J.; Zheng, N.-Y.; Huang, M.; Huang, Y.; Neu, K.E.; Lee, J.; Wan, H.; Rojas, K.T.; Kirkpatrick, E.; et al. Influenza Infection in Humans Induces Broadly Cross-Reactive and Protective Neuraminidase-Reactive Antibodies. Cell 2018, 173, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Reneer, Z.B.; Jamieson, P.J.; Skarlupka, A.L.; Huang, Y.; Ross, T.M. Computationally Optimized Broadly Reactive H2 HA Influenza Vaccines Elicited Broadly Cross-Reactive Antibodies and Protected Mice from Viral Challenges. J. Virol. 2020, 95. [Google Scholar] [CrossRef] [PubMed]
| Healthy | Obesity | Type-2 Diabetes | Elderly | References | |
|---|---|---|---|---|---|
| Adipose Tissue Composition | Functional adipocytes; high rates of M2 macrophages; low rates of M1 macrophages | Dysfunctional hypertrophic and necrotic adipocytes; low rates of M2 macrophages; high rates of M1 macrophages | Dysfunctional necrotic adipocytes; increased rates of M1 macrophages; evidence of fibrosis | Dysfunctional necrotic adipocytes; prevalence of M1 macrophages | [46,47,48] |
| Dendritic Cells (DCs) | Functional DCs; balanced levels of anti-inflammatory Th1 cells to polarize M2 macrophages; balanced levels of pro-inflammatory Th17 cells to polarize M1 macrophages | High levels of inflammatory CD11c+ DCs that produce more pro-inflammatory Th17 cells to polarize M1 macrophages | Lower levels of inflammatory CD11c+ DCs with higher levels of inflammatory CD123+ plasmacytoid DCs | Healthy DCs but unreliable ability to induce an inflammatory response | [49,50,51,52,53,54,55] |
| B Cells | Regular levels of activation; regular cytokine release; regular class-switching; generation of broad neutralizing antibodies; active responses to novel antigen stimulation | High levels of activation; increased secretion of IL6 and TNFα that contributes to the pro-inflammatory environment; decreased class-switching; non-neutralizing antibodies; atypical late memory B cells accumulate at high levels in blood; impaired responses to novel antigen stimulation | High levels of activation; compromised regulatory B cell activity due to deficient IL-10 secretion; impaired responses to novel antigen stimulation | Decreased class switching; severely restricted B-cell receptor repertoires; highly specific, non-neutralizing antibodies; atypical late memory B cells accumulate at high levels in blood; impaired responses to novel antigen stimulation | [38,56,57,58,59,60,61,62,63] |
| T Cells | Healthy levels of CD4+ and CD8+ T cell activation; broad T cell receptor repertoires; proper costimulatory signaling; proper T cell expansion | Compromised activation of CD4+ and CD8+ T cells; lower levels of costimulatory signaling; decreased T cell expansion | Compromised activation of CD4+ and CD8+ T cells; T cell dysfunction that contributes to disease pathology | Compromised activation of CD4+ and CD8+ T cells; restricted T cell receptor repertoires; restricted T cell clones; poor activation; poor differentiation | [13,16,35,50,55,63,64,65,66] |
| Metabolic Markers | Balanced levels of adiponectin and leptin; proper insulin release | Low levels of adiponectin; high levels of leptin; high levels of insulin; insulin resistance | Low levels of adiponectin; high levels of leptin; high levels of insulin; insulin resistance | Metabolic markers are more dependent upon MetS status than age | [64,67,68,69,70,71,72,73,74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiggins, K.B.; Smith, M.A.; Schultz-Cherry, S. The Nature of Immune Responses to Influenza Vaccination in High-Risk Populations. Viruses 2021, 13, 1109. https://doi.org/10.3390/v13061109
Wiggins KB, Smith MA, Schultz-Cherry S. The Nature of Immune Responses to Influenza Vaccination in High-Risk Populations. Viruses. 2021; 13(6):1109. https://doi.org/10.3390/v13061109
Chicago/Turabian StyleWiggins, Kristin B., Maria A. Smith, and Stacey Schultz-Cherry. 2021. "The Nature of Immune Responses to Influenza Vaccination in High-Risk Populations" Viruses 13, no. 6: 1109. https://doi.org/10.3390/v13061109
APA StyleWiggins, K. B., Smith, M. A., & Schultz-Cherry, S. (2021). The Nature of Immune Responses to Influenza Vaccination in High-Risk Populations. Viruses, 13(6), 1109. https://doi.org/10.3390/v13061109






