Surveillance for Enteroviruses Associated with Hand, Foot, and Mouth Disease, and Other Mucocutaneous Symptoms in Spain, 2006–2020
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Clinical Samples
2.2. EV Genotyping and Phylogenetic Analyses
2.3. Statistical Analysis
3. Results
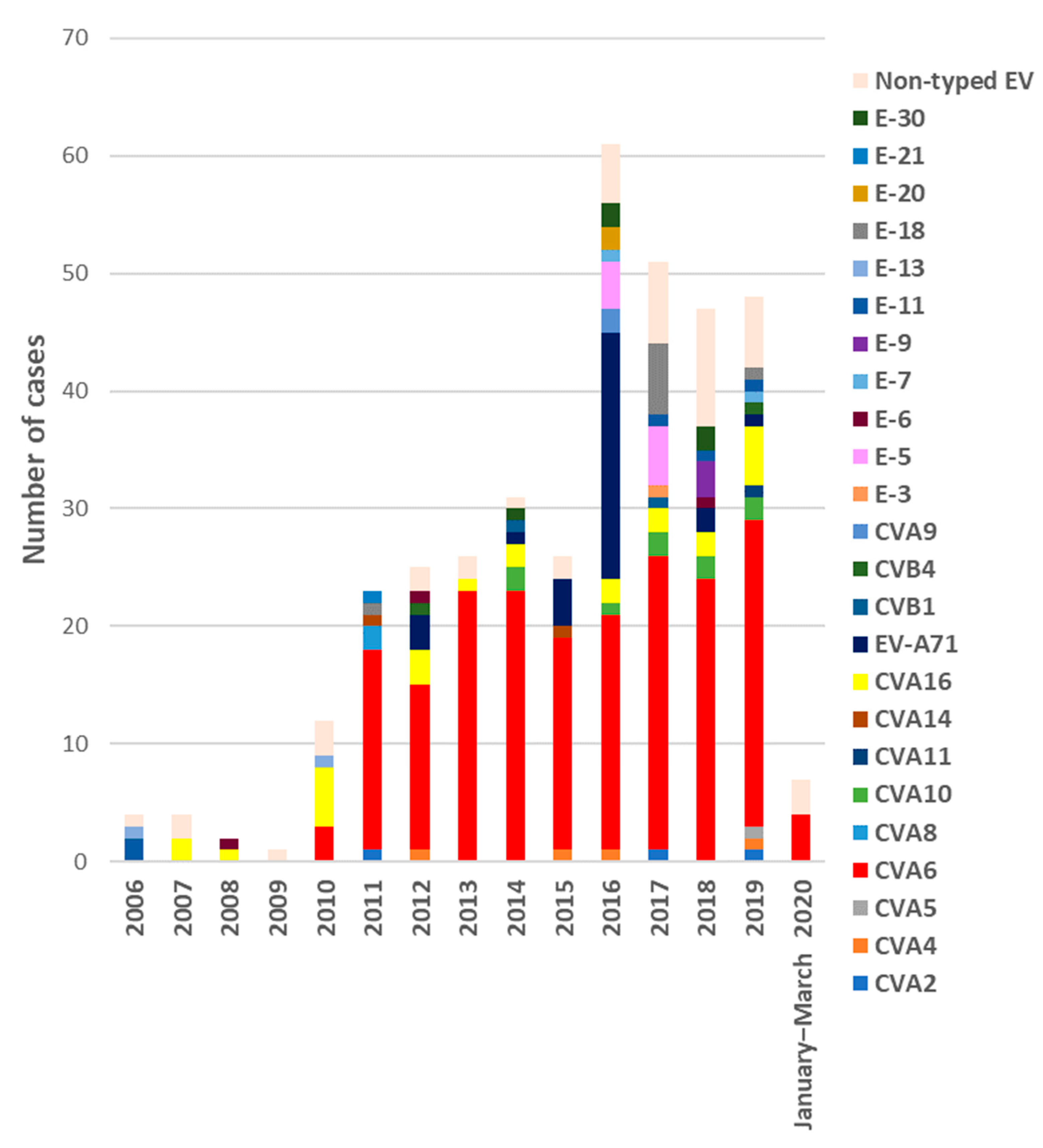
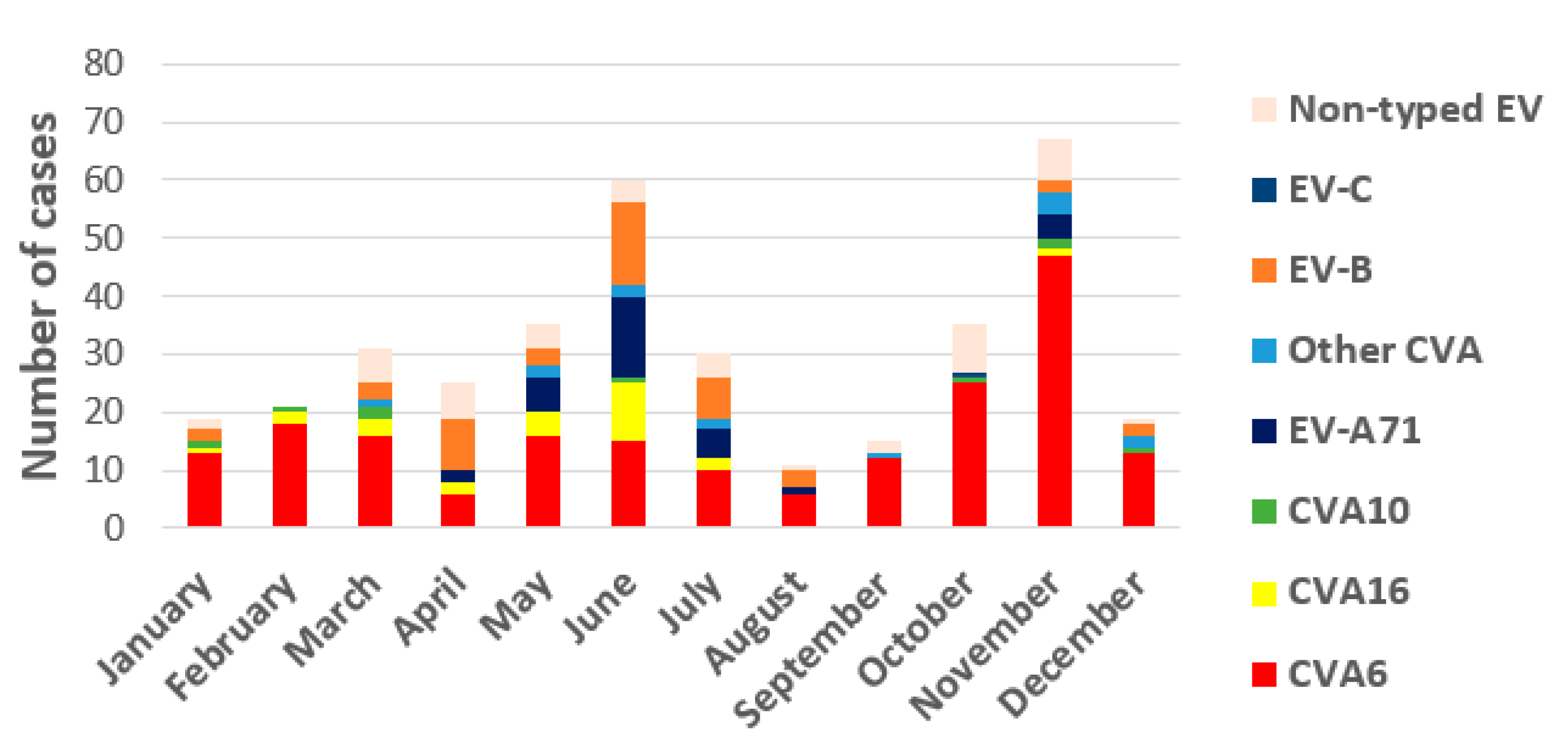
3.1. EV Type Characterization
3.2. Clinical and Epidemiological Characteristics of EV Infections
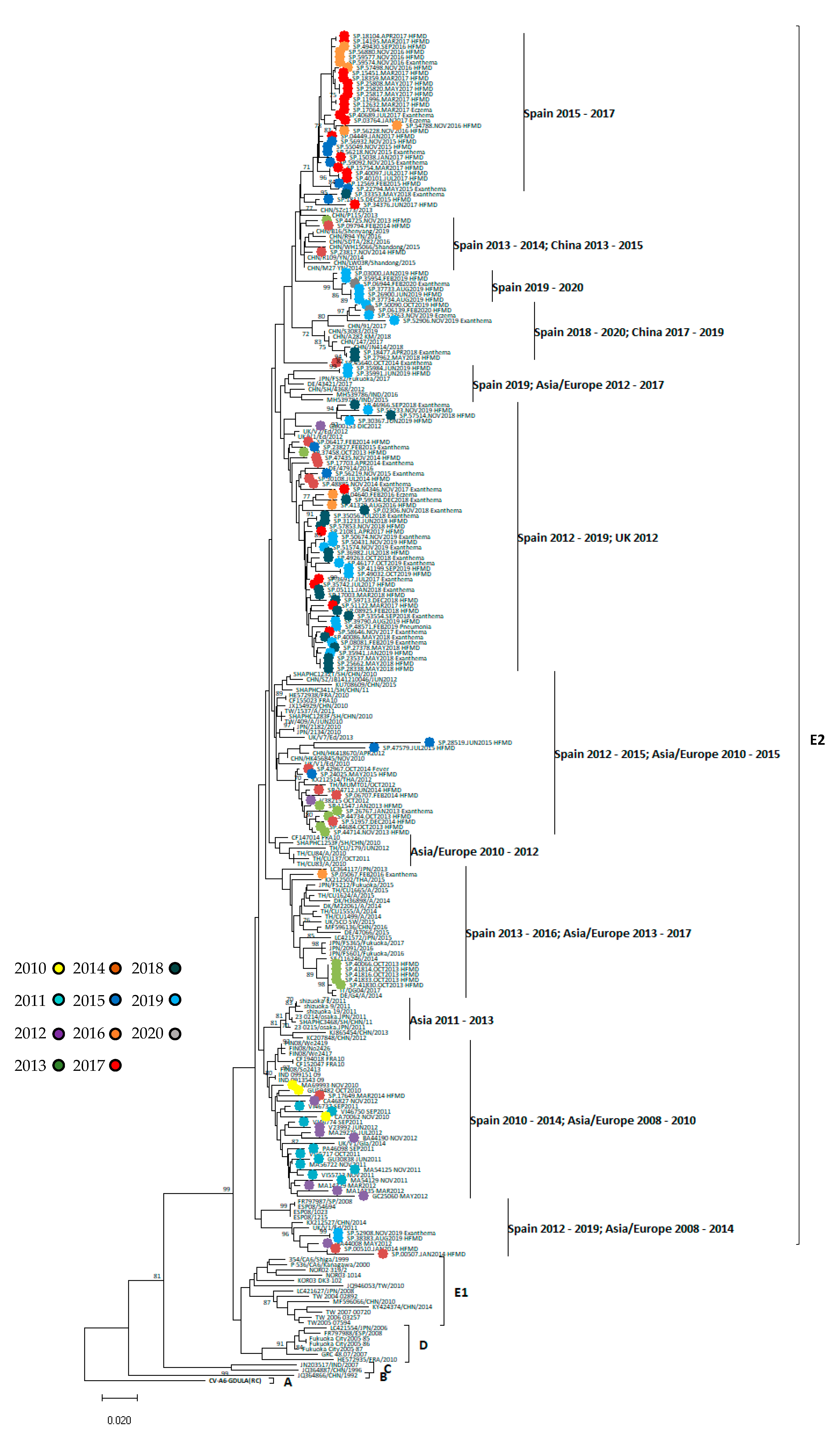
3.3. Phylogenetic Analysis of CVA6
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Palacios, G.; Casas, I.; Trallero, G. Clinical Microbiology: Human Enteroviruses. In Infectious Diseases, 3rd ed.; Cohen, J., Opal, S.M., Powderly, W.G., Calandra, T., Eds.; Mosby Elsevier: St. Louis, MO, USA, 2010; pp. 1528–1538. [Google Scholar]
- Zell, R.; Delwart, E.; Gorbalenya, A.E.; Hovi, T.; King, A.M.Q.; Knowles, N.J.; Lindberg, A.M.; Pallansch, M.A.; Palmenberg, A.C.; Reuter, G.; et al. ICTV Virus Taxonomy Profile: Picornaviridae. J. Gen. Virol. 2017, 98, 2421–2422. [Google Scholar] [CrossRef]
- Guerra, A.M.; Orille, E.; Waseem, M. Hand Foot and Mouth Disease. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Yang, F.; Zhang, T.; Hu, Y.; Wang, X.; Du, J.; Li, Y.; Sun, S.; Sun, X.; Li, Z.; Jin, Q. Survey of enterovirus infections from hand, foot and mouth disease outbreak in china, 2009. Virol. J. 2011, 8, 508. [Google Scholar] [CrossRef]
- Yip, C.C.Y.; Lau, S.K.P.; Woo, P.C.Y.; Yuen, K.-Y. Human enterovirus 71 epidemics: What’s next? Emerg. Health Threat. J. 2013, 6, 19780. [Google Scholar] [CrossRef]
- Pons-Salort, M.; Parker, E.P.K.; Grassly, N.C. The epidemiology of non-polio enteroviruses: Recent Advances and Outstanding Questions. Curr. Opin. Infect. Dis. 2015, 28, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Chiu, M.-L.; Luo, S.-T.; Chen, Y.-Y.; Chung, W.Y.; Duong, V.; Dussart, P.; Chan, Y.-F.; Perera, D.; Ooi, M.H.; Thao, N.T.T.; et al. Establishment of Asia-Pacific Network for Enterovirus Surveillance. Vaccine 2020, 38, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Dou, X.; Yan, C.; Wan, L.; Liu, H.; Li, M.; Wang, R.; Li, G.; Zhao, L.; Liu, Z.; et al. Epidemiological characteristics and trends of notifiable infectious diseases in China from 1986 to 2016. J. Glob. Health 2020, 10, 020803. [Google Scholar] [CrossRef]
- He, Y.-Q.; Chen, L.; Xu, W.-B.; Yang, H.; Wang, H.-Z.; Zong, W.-P.; Xian, H.-X.; Chen, H.-L.; Yao, X.-J.; Hu, Z.-L.; et al. Emergence, Circulation, and Spatiotemporal Phylogenetic Analysis of Coxsackievirus A6- and Coxsackievirus A10-Associated Hand, Foot, and Mouth Disease Infections from 2008 to 2012 in Shenzhen, China. J. Clin. Microbiol. 2013, 51, 3560–3566. [Google Scholar] [CrossRef] [PubMed]
- Bian, L.; Wang, Y.; Yao, X.; Mao, Q.; Xu, M.; Liang, Z. Coxsackievirus A6: A new emerging pathogen causing hand, foot and mouth disease outbreaks worldwide. Expert Rev. Anti Infect. Ther. 2015, 13, 1061–1071. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, C.; Gaunt, E.; Simmonds, P.; Broomfield, D.; Nwafor, N.; Wellington, L.; Templeton, K.; Willocks, L.; Schofield, O.; Harvala, H. Atypical hand, foot, and mouth disease associated with coxsackievirus A6 infection, Edinburgh, United Kingdom, January to February 2014. Eurosurveillance 2014, 19, 20745. [Google Scholar] [CrossRef] [PubMed]
- Neri, I.; Dondi, A.; Wollenberg, A.; Ricci, L.; Ricci, G.; Piccirilli, G.; Lazzarotto, T.; Patrizi, A. Atypical Forms of Hand, Foot, and Mouth Disease: A Prospective Study of 47 Italian Children. Pediatr. Dermatol. 2016, 33, 429–437. [Google Scholar] [CrossRef]
- Horsten, H.-H.; Kemp, M.; Fischer, T.K.; Lindahl, K.H.; Bygum, A. Atypical Hand, Foot, and Mouth Disease Caused by Coxsackievirus A6 in Denmark: A Diagnostic Mimicker. Acta Derm. Venereol. 2018, 98, 350–354. [Google Scholar] [CrossRef]
- Fujimoto, T.; Iizuka, S.; Enomoto, M.; Abe, K.; Yamashita, K.; Hanaoka, N.; Okabe, N.; Yoshida, H.; Yasui, Y.; Kobayashi, M.; et al. Hand, Foot, and Mouth Disease Caused by Coxsackievirus A6, Japan, 2011. Emerg. Infect. Dis. 2012, 18, 337–339. [Google Scholar] [CrossRef]
- Wei, S.-H.; Huang, Y.-P.; Liu, M.-C.; Tsou, T.-P.; Lin, H.-C.; Lin, T.-L.; Tsai, C.-Y.; Chao, Y.-N.; Chang, L.-Y.; Hsu, C.-M. An outbreak of coxsackievirus A6 hand, foot, and mouth disease associated with onychomadesis in Taiwan, 2010. BMC Infect. Dis. 2011, 11, 346. [Google Scholar] [CrossRef] [PubMed]
- Österback, R.; Vuorinen, T.; Linna, M.; Susi, P.; Hyypiä, T.; Waris, M. Coxsackievirus A6 and Hand, Foot, and Mouth Disease, Finland. Emerg. Infect. Dis. 2009, 15, 1485–1488. [Google Scholar] [CrossRef]
- Gonzalez, G.; Carr, M.J.; Kobayashi, M.; Hanaoka, N.; Fujimoto, T. Enterovirus-Associated Hand-Foot and Mouth Disease and Neurological Complications in Japan and the Rest of the World. Int. J. Mol. Sci. 2019, 20, 5201. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, H.; Ou, R.; Zhu, H.; Gan, L.; Zeng, Z.; Yuan, R.; Yu, H.; Ye, M. Epidemiological and clinical characteristics of severe hand-foot-and-mouth disease (HFMD) among children: A 6-year population-based study. BMC Public Health 2020, 20, 801. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, Y.; Zhang, C.; Zhan, W.; Xie, J.; Hu, S.; Chai, H.; Liu, P.; Zhao, H.; Tang, B.; et al. Clinical features and phylogenetic analysis of severe hand-foot-and-mouth disease caused by Coxsackievirus A6. Infect. Genet. Evol. 2020, 77, 104054. [Google Scholar] [CrossRef] [PubMed]
- Feder, H.M.; Bennett, N.; Modlin, J.F. Atypical hand, foot, and mouth disease: A vesiculobullous eruption caused by Coxsackie virus A6. Lancet Infect. Dis. 2014, 14, 83–86. [Google Scholar] [CrossRef]
- Mirand, A.; Le Sage, F.V.; Pereira, B.; Cohen, R.; Levy, C.; Archimbaud, C.; Peigue-Lafeuille, H.; Bailly, J.-L.; Henquell, C. Ambulatory Pediatric Surveillance of Hand, Foot and Mouth Disease as Signal of an Outbreak of Coxsackievirus A6 Infections, France, 2014–2015. Emerg. Infect. Dis. 2016, 22, 1884–1893. [Google Scholar] [CrossRef]
- Cabrerizo, M.; Díaz-Cerio, M.; Muñoz-Almagro, C.; Rabella, N.; Tarragó, D.; Romero, M.P.; Pena, M.J.; Calvo, C.; Rey-Cao, S.; Moreno-Docón, A.; et al. Molecular epidemiology of enterovirus and parechovirus infections according to patient age over a 4-year period in Spain. J. Med. Virol. 2017, 89, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Bubba, L.; Broberg, E.K.; Jasir, A.; Simmonds, P.; Harvala, H.; Enterovirus Study Collaborators. Circulation of Non-Polio Enteroviruses in 24 EU and EEA Countries between 2015 and 2017: A Retrospective Surveillance Study. Lancet Infect. Dis. 2020, 20, 350–361. [Google Scholar] [CrossRef]
- Bracho, M.A.; González-Candelas, F.; Valero, A.; Córdoba, J.; Salazar, A. Enterovirus Co-infections and Onychomadesis after Hand, Foot, and Mouth Disease, Spain, 2008. Emerg. Infect. Dis. 2011, 17, 2223–2231. [Google Scholar] [CrossRef]
- Montes, M.; Artieda, J.; Piñeiro, L.D.; Gastesi, M.; Diez-Nieves, I.; Cilla, G. Hand, Foot, and Mouth Disease Outbreak and Coxsackievirus A6, Northern Spain, 2011. Emerg. Infect. Dis. 2013, 19, 676–678. [Google Scholar] [CrossRef]
- Cabrerizo, M.; Tarragó, D.; Muñoz-Almagro, C.; Del Amo, E.; Domínguez-Gil, M.; Eiros, J.M.; López-Miragaya, I.; Pérez, C.; Reina, J.; Otero, A.; et al. Molecular epidemiology of enterovirus 71, coxsackievirus A16 and A6 associated with hand, foot and mouth disease in Spain. Clin. Microbiol. Infect. 2014, 20, O150–O156. [Google Scholar] [CrossRef]
- Andrés, C.; Guasch, E.; Piñana, M.; Fernandes, P.; Gimferrer, L.; Esso, D.V.; Codina, M.G.; Esperalba, J.; Vila, J.; Rodrigo, C.; et al. Recombinant CV-A6 strains related to hand-foot-mouth disease and herpangina at primary care centers (Barcelona, Spain). Future Microbiol. 2019, 14, 499–507. [Google Scholar] [CrossRef]
- Herrero, M.; Kutzner, H.; Fraga, J.; Llamas-Velasco, M. Immunohistochemical Study of 2 Cases of Coxsackie A6–Induced Atypical Hand-Foot-and-Mouth Disease. Am. J. Dermatopathol. 2019, 41, 741–743. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. A Guide to Clinical Management and Public Health Response for Hand, Foot and Mouth Disease; World Health Organization: Geneva, Switzerland, 2011; Available online: http://www.wpro.who.int/emerging_diseases/documents/HFMDGuidance/en (accessed on 2 April 2015).
- Cabrerizo, M.; Echevarría, J.E.; González, I.; De Miguel, T.; Trallero, G. Molecular epidemiological study of HEV-B enteroviruses involved in the increase in meningitis cases occurred in Spain during 2006. J. Med. Virol. 2008, 80, 1018–1024. [Google Scholar] [CrossRef]
- González-Sanz, R.; Taravillo, I.; Reina, J.; Navascués, A.; Moreno-Docón, A.; Aranzamendi, M.; Romero, M.P.; Del Cuerpo, M.; Pérez-González, C.; Pérez-Castro, S.; et al. Enterovirus D68-associated respiratory and neurological illness in Spain, 2014–2018. Emerg. Microbes Infect. 2019, 8, 1438–1444. [Google Scholar] [CrossRef]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; Mcgettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed]
- González-Sanz, R.; Casas-Alba, D.; Launes, C.; Muñoz-Almagro, C.; Ruiz-García, M.M.; Alonso, M.; González-Abad, M.J.; Megías, G.; Rabella, N.; Del Cuerpo, M.; et al. Molecular epidemiology of an enterovirus A71 outbreak associated with severe neurological disease, Spain, 2016. Eurosurveillance 2019, 24, 1800089. [Google Scholar] [CrossRef] [PubMed]
- Casas-Alba, D.; De Sevilla, M.F.; Valero-Rello, A.; Fortuny, C.; García-García, J.-J.; Ortez, C.; Muchart, J.; Armangué, T.; Jordan, I.; Luaces, C.; et al. Outbreak of brainstem encephalitis associated with enterovirus-A71 in Catalonia, Spain (2016): A clinical observational study in a children’s reference centre in Catalonia. Clin. Microbiol. Infect. 2017, 23, 874–881. [Google Scholar] [CrossRef]
- Mirand, A.; Schuffenecker, I.; Henquell, C.; Billaud, G.; Jugie, G.; Falcon, D.; Mahul, A.; Archimbaud, C.; Terletskaia-Ladwig, E.; Diedrich, S.; et al. Phylogenetic evidence for a recent spread of two populations of human enterovirus 71 in European countries. J. Gen. Virol. 2010, 91, 2263–2277. [Google Scholar] [CrossRef]
- McWilliam Leitch, E.C.; Cabrerizo, M.; Cardosa, J.; Harvala, H.; Ivanova, O.E.; Koike, S.; Kroes, A.C.M.; Lukashev, A.; Perera, D.; Roivainen, M.; et al. The Association of Recombination Events in the Founding and Emergence of Subgenogroup Evolutionary Lineages of Human Enterovirus 71. J. Virol. 2012, 86, 2676–2685. [Google Scholar] [CrossRef] [PubMed]
- Harvala, H.; Broberg, E.; Benschop, K.; Berginc, N.; Ladhani, S.; Susi, P.; Christiansen, C.; McKenna, J.; Allen, D.; Makiello, P.; et al. Recommendations for enterovirus diagnostics and characterisation within and beyond Europe. J. Clin. Virol. 2018, 101, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Rotulo, G.A.; Percivale, B.; Molteni, M.; Naim, A.; Brisca, G.; Piccotti, E.; Castagnola, E. The impact of COVID-19 lockdown on infectious diseases epidemiology: The experience of a tertiary Italian Pediatric Emergency Department. Am. J. Emerg. Med. 2021, 43, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Yang, X.-H.; Hu, S.-Q.; Zhan, W.-L.; Zhang, C.-B.; Liu, H.; Zhao, H.-Y.; Chai, H.-Y.; Chen, K.-Y.; Du, Q.-Y.; et al. Co-circulation of coxsackieviruses A-6, A-10, and A-16 causes hand, foot, and mouth disease in Guangzhou city, China. BMC Infect. Dis. 2020, 20, 271. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.N.A.; Wang, A.D.; Yin, M.; Lee, C.K.; Archuleta, S. Atypical hand, foot, and mouth disease: Eczema coxsackium can also occur in adults. Lancet Infect. Dis. 2014, 14, 1043. [Google Scholar] [CrossRef]
- Laga, A.C.; Shroba, S.M.; Hanna, J. Atypical Hand, Foot and Mouth Disease in Adults Associated with Coxsackievirus A6: A Clinico-Pathologic Study. J. Cutan. Pathol. 2016, 43, 940–945. [Google Scholar] [CrossRef]
- Broccolo, F.; Drago, F.; Ciccarese, G.; Genoni, A.; Puggioni, A.; Rosa, G.M.; Parodi, A.; Manukyan, H.; Laassri, M.; Chumakov, K.; et al. Severe atypical hand-foot-and-mouth disease in adults due to coxsackievirus A6: Clinical presentation and phylogenesis of CV-A6 strains. J. Clin. Virol. 2019, 110, 1–6. [Google Scholar] [CrossRef]
- Puenpa, J.; Vongpunsawad, S.; Österback, R.; Waris, M.; Eriksson, E.; Albert, J.; Midgley, S.; Fischer, T.K.; Eis-Hübinger, A.M.; Cabrerizo, M.; et al. Molecular epidemiology and the evolution of human coxsackievirus A6. J. Gen. Virol. 2016, 97, 3225–3231. [Google Scholar] [CrossRef]
- Gaunt, E.; Harvala, H.; Österback, R.; Sreenu, V.B.; Thomson, E.; Waris, M.; Simmonds, P. Genetic characterization of human coxsackievirus A6 variants associated with atypical hand, foot and mouth disease: A potential role of recombination in emergence and pathogenicity. J. Gen. Virol. 2015, 96, 1067–1079. [Google Scholar] [CrossRef] [PubMed]
| Epidemiological Features | |
| patients < 15 years-old | 317 (86.2%) |
| range | 4 days–14 years |
| mean age | 1.87 ± 1.73SD |
| patients > 15 years-old | 51 (13.8%) |
| range | 18-90 years |
| mean age | 36.52 ± 13.82SD |
| male/female | 225/143 |
| Clinical diagnosis | |
| HFMD | 215 (58.4%) |
| herpangina | 10 (2.7%) |
| exanthema/rash/petechiae | 127 (34.5%) |
| atypical HFMD | 14 (38%) |
| Side-symptoms | |
| onychomadesis | 16 (4.3%) |
| acute gastroenteritis | 2 (0.5%) |
| respiratory symptoms | 3 (0.8%) |
| NCS involvement (meningitis, encephalitis or paralysis) | 7 (1.9%) |
| EV Genotypes | |||||||
|---|---|---|---|---|---|---|---|
| Clinical Diagnosis | CVA6 (N = 197) | CVA16 (N = 25) | CVA10 (N = 9) | EV-A71 (N = 32) | Other CVA (N = 12) | EV-B (N = 47) | p Values |
| HFMD (N = 215) | 137 (63.7) | 13 (6) | 4 (1.9) | 28 (13.0) | 7 (3.2) | 10 (4.6) | <0.00001 |
| herpangina (N = 10) | 0 | 2 (20) | 3 (30) | 1 (10) | 1 (10) | 1 (10) | 0.5820 |
| exanthema/rash/pethechiae (N = 127) | 47 (37) | 9 (7.1) | 2 (1.6) | 3 (23.6) | 4 (3.1) | 36 (28.3) | 0.1812 |
| atypical HFMD (N = 14) | 13 (92.8) | 1 (7.1) | 0 | 0 | 0 | 0 | 0.00003 |
| Side-symptoms | |||||||
| onychomadesis (N = 16) | 6 (37.5) | 1 (6.2) | 0 | 1 (6.2) | 5 (31.2) | 2 (12.5) | 0.7289 |
| NCS involvement (N = 7) | 0 | 0 | 0 | 5 (71.4) | 0 | 0 | 0.0209 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-López, N.; Muñoz-Almagro, C.; Launes, C.; Navascués, A.; Imaz-Pérez, M.; Reina, J.; Romero, M.P.; Calvo, C.; Ruiz-García, M.; Megias, G.; et al. Surveillance for Enteroviruses Associated with Hand, Foot, and Mouth Disease, and Other Mucocutaneous Symptoms in Spain, 2006–2020. Viruses 2021, 13, 781. https://doi.org/10.3390/v13050781
Martínez-López N, Muñoz-Almagro C, Launes C, Navascués A, Imaz-Pérez M, Reina J, Romero MP, Calvo C, Ruiz-García M, Megias G, et al. Surveillance for Enteroviruses Associated with Hand, Foot, and Mouth Disease, and Other Mucocutaneous Symptoms in Spain, 2006–2020. Viruses. 2021; 13(5):781. https://doi.org/10.3390/v13050781
Chicago/Turabian StyleMartínez-López, Nieves, Carmen Muñoz-Almagro, Cristian Launes, Ana Navascués, Manuel Imaz-Pérez, Jordi Reina, María Pilar Romero, Cristina Calvo, Montserrat Ruiz-García, Gregoria Megias, and et al. 2021. "Surveillance for Enteroviruses Associated with Hand, Foot, and Mouth Disease, and Other Mucocutaneous Symptoms in Spain, 2006–2020" Viruses 13, no. 5: 781. https://doi.org/10.3390/v13050781
APA StyleMartínez-López, N., Muñoz-Almagro, C., Launes, C., Navascués, A., Imaz-Pérez, M., Reina, J., Romero, M. P., Calvo, C., Ruiz-García, M., Megias, G., Valencia-Ramos, J., Otero, A., & Cabrerizo, M. (2021). Surveillance for Enteroviruses Associated with Hand, Foot, and Mouth Disease, and Other Mucocutaneous Symptoms in Spain, 2006–2020. Viruses, 13(5), 781. https://doi.org/10.3390/v13050781







