Bioengineered Liver Cell Models of Hepatotropic Infections
Abstract
1. Introduction
1.1. Hepatitis Viruses
1.2. Plasmodium
2. Experimental Models of Liver Infection
2.1. Animal Models
2.2. D In Vitro Models (Cell Monolayers)
2.3. Bioengineered Liver Cell Models
2.3.1. 2D Cultures
2.3.2. Scaffold-Based 3D Cultures
2.3.3. Scaffold-Free 3D Cultures
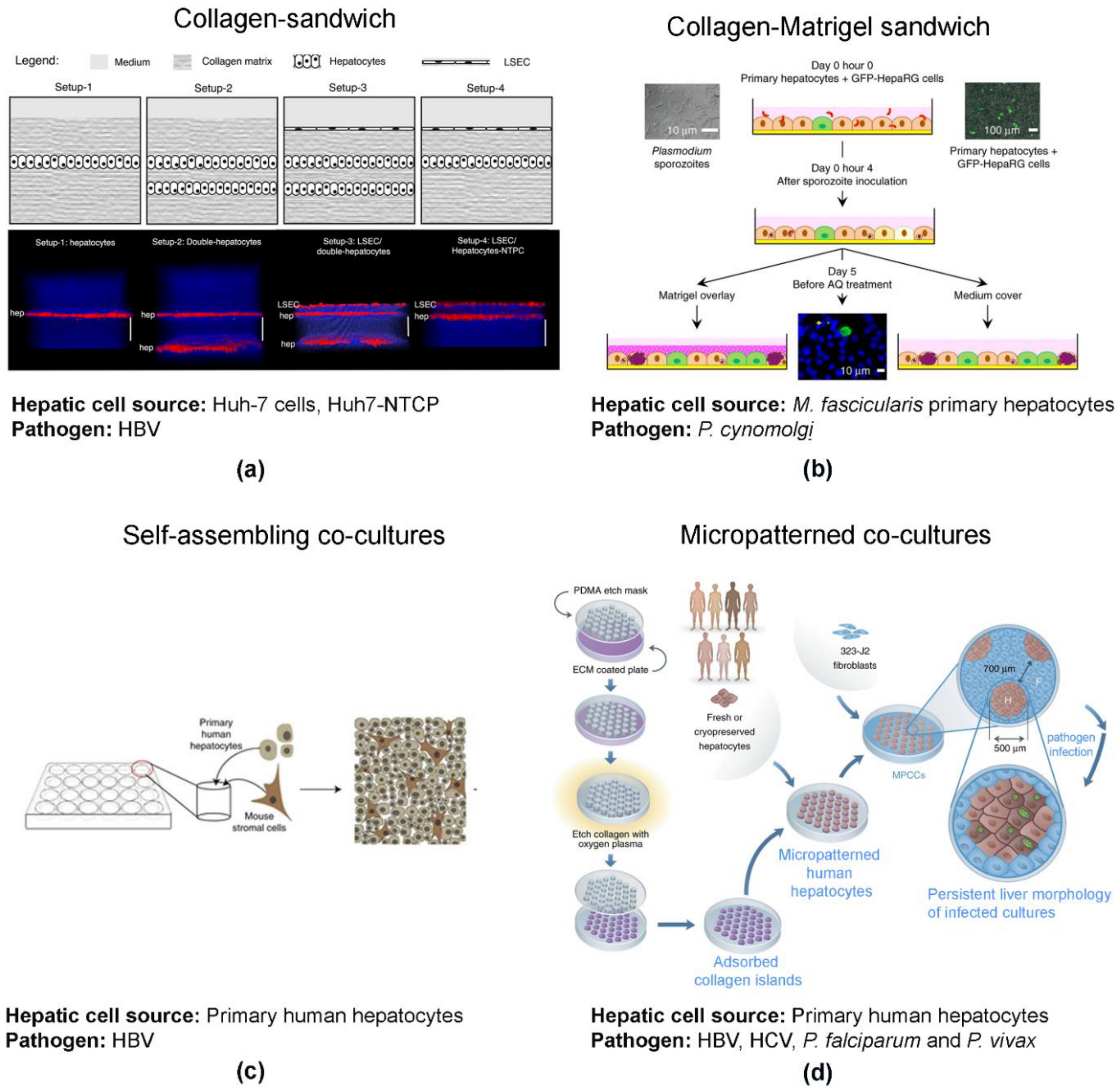
2.3.4. 3D Co-Cultures
3. Current Challenges, Trends and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Godoy, P.; Hewitt, N.J.; Albrecht, U.; Andersen, M.E.; Ansari, N.; Bhattacharya, S.; Bode, J.G.; Bolleyn, J.; Borner, C.; Böttger, J.; et al. Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells and their use in investigating mechanisms of hepatotoxicity, cell signaling and ADME. Arch. Toxicol. 2013, 87, 1315–1530. [Google Scholar] [CrossRef] [PubMed]
- Protzer, U.; Maini, M.K.; Knolle, P.A. Living in the liver: Hepatic infections. Nat. Rev. Immunol. 2012, 12, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Talwani, R.; Gilliam, B.L.; Howell, C. Infectious Diseases and the Liver. Clin. Liver Dis. 2011, 15, 111–130. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Hensley, L.; McKnight, K.L.; Hu, F.; Madden, V.; Ping, L.; Jeong, S.-H.; Walker, C.; Lanford, R.E.; Lemon, S.M. A pathogenic picornavirus acquires an envelope by hijacking cellular membranes. Nature 2013, 496, 367–371. [Google Scholar] [CrossRef]
- Shimotohno, K. HCV Assembly and Egress via Modifications in Host Lipid Metabolic Systems. Cold Spring Harb. Perspect. Med. 2021, 11, a036814. [Google Scholar] [CrossRef]
- Fu, R.M.; Decker, C.C.; Dao Thi, V.L. Cell Culture Models for Hepatitis E Virus. Viruses 2019, 11, 608. [Google Scholar] [CrossRef]
- Schlütter, J. Therapeutics: New drugs hit the target. Nature 2011, 474, S5–S7. [Google Scholar] [CrossRef]
- Sorbo, M.C.; Cento, V.; Di Maio, V.C.; Howe, A.Y.M.; Garcia, F.; Perno, C.F.; Ceccherini-Silberstein, F. Hepatitis C virus drug resistance associated substitutions and their clinical relevance: Update 2018. Drug Resist. Updates 2018, 37, 17–39. [Google Scholar] [CrossRef]
- Hill, A.; Cooke, G. Hepatitis C can be cured globally, but at what cost? Science 2014, 345, 141–142. [Google Scholar] [CrossRef][Green Version]
- Bailey, J.R.; Barnes, E.; Cox, A.L. Approaches, Progress, and Challenges to Hepatitis C Vaccine Development. Gastroenterology 2019, 156, 418–430. [Google Scholar] [CrossRef]
- WHO. World Malaria Report 2018; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Prudêncio, M.; Mota, M.M.; Mendes, A.M. A toolbox to study liver stage malaria. Trends Parasitol. 2011, 27, 565–574. [Google Scholar] [CrossRef]
- Mota, M.M.; Hafalla, J.C.R.; Rodriguez, A. Migration through host cells activates Plasmodium sporozoites for infection. Nat. Med. 2002, 8, 1318–1322. [Google Scholar] [CrossRef]
- Kaushansky, A.; Kappe, S.H.I. Selection and refinement: The malaria parasite’s infection and exploitation of host hepatocytes. Curr. Opin. Microbiol. 2015, 26, 71–78. [Google Scholar] [CrossRef]
- Vaughan, A.M.; Kappe, S.H.I. Malaria parasite liver infection and exoerythrocytic biology. Cold Spring Harb. Perspect. Med. 2017, 7. [Google Scholar] [CrossRef]
- Prudêncio, M.; Rodriguez, A.; Mota, M.M. The silent path to thousands of merozoites: The Plasmodium liver stage. Nat. Rev. Microbiol. 2006, 4, 849–856. [Google Scholar] [CrossRef]
- Duffy, P.E.; Patrick Gorres, J. Malaria vaccines since 2000: Progress, priorities, products. NPJ Vaccines 2020, 5, 48. [Google Scholar] [CrossRef]
- Antony, H.; Parija, S. Antimalarial drug resistance: An overview. Trop. Parasitol. 2016, 6, 30. [Google Scholar] [CrossRef]
- Wieland, S.F. The Chimpanzee Model for Hepatitis B Virus Infection. Cold Spring Harb. Perspect. Med. 2015, 5, a021469. [Google Scholar] [CrossRef]
- Berggren, K.A.; Suzuki, S.; Ploss, A. Animal Models Used in Hepatitis C Virus Research. Int. J. Mol. Sci. 2020, 21, 3869. [Google Scholar] [CrossRef]
- Killick-Kendrick, R.; Koontz, L.C.; Krotoski, W.A.; Garnham, P.C.C.; Bray, R.S.; Gwadz, R.W.; Wolf, R.; Stanfill, P.S.; Cogswell, F.B.; Sinden, R.; et al. Demonstration of Hypnozoites in Sporozoite-Transmitted Plasmodium vivax Infection *. Am. J. Trop. Med. Hyg. 1982, 31, 1291–1293. [Google Scholar] [CrossRef]
- Rodhain, J. Paradoxical behaviour of Plasmodium vivax in the chimpanzee. Trans. R. Soc. Trop. Med. Hyg. 1956, 50, 287–293. [Google Scholar] [CrossRef]
- Brown, R.J.P.; Tegtmeyer, B.; Sheldon, J.; Khera, T.; Todt, D.; Vieyres, G.; Weller, R.; Joecks, S.; Zhang, Y.; Pietschmann, T.; et al. Liver-expressed Cd302 and Cr1l limit hepatitis C virus cross-species transmission to mice. Sci. Adv. 2020, 6, 31. [Google Scholar] [CrossRef]
- Von Schaewen, M.; Hrebikova, G.; Ploss, A. Generation of Human Liver Chimeric Mice for the Study of Human Hepatotropic Pathogens. In Mouse Models for Drug Discovery; Humana Press: New York, NY, USA, 2016; pp. 79–101. [Google Scholar]
- Yong, K.S.M.; Her, Z.; Chen, Q. Humanized Mouse Models for the Study of Hepatitis C and Host Interactions. Cells 2019, 8, 604. [Google Scholar] [CrossRef]
- Sun, S.; Li, J. Humanized chimeric mouse models of hepatitis B virus infection. Int. J. Infect. Dis. 2017, 59, 131–136. [Google Scholar] [CrossRef]
- Carpentier, A.; Tesfaye, A.; Chu, V.; Nimgaonkar, I.; Zhang, F.; Lee, S.B.; Thorgeirsson, S.S.; Feinstone, S.M.; Liang, T.J. Engrafted human stem cell-derived hepatocytes establish an infectious HCV murine model. J. Clin. Invest. 2014, 124, 4953–4964. [Google Scholar] [CrossRef]
- Minkah, N.K.; Schafer, C.; Kappe, S.H.I. Humanized mouse models for the study of human malaria parasite biology, pathogenesis, and immunity. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef]
- Castell, J.V.; Jover, R.; Martínez-Jiménez, C.P.; Gómez-Lechón, M.J. Hepatocyte cell lines: Their use, scope and limitations in drug metabolism studies. Expert Opin. Drug Metab. Toxicol. 2006, 2, 183–212. [Google Scholar] [CrossRef]
- Meredith, L.W.; Hu, K.; Cheng, X.; Howard, C.R.; Baumert, T.F.; Balfe, P.; van de Graaf, K.F.; Protzer, U.; McKeating, J.A. Lentiviral hepatitis B pseudotype entry requires sodium taurocholate co-transporting polypeptide and additional hepatocyte-specific factors. J. Gen. Virol. 2016, 97, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Bartosch, B.; Dubuisson, J.; Cosset, F.L. Infectious hepatitis C virus pseudo-particles containing functional E1-E2 envelope protein complexes. J. Exp. Med. 2003, 197, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wang, Z.; Hu, F.; Su, L. Cell Culture Models and Animal Models for HBV Study. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2020; Volume 1179, pp. 109–135. [Google Scholar]
- Steinmann, E.; Pietschmann, T. Cell culture systems for hepatitis C virus. Curr. Top. Microbiol. Immunol. 2013, 369, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Pietschmann, T. In sero veritas: What serum markers teach us about HCV infection of primary human hepatocytes. Gut 2014, 63, 1375–1377. [Google Scholar] [CrossRef]
- Roth, A.; Maher, S.P.; Conway, A.J.; Ubalee, R.; Chaumeau, V.; Andolina, C.; Kaba, S.A.; Vantaux, A.; Bakowski, M.A.; Thomson-Luque, R.; et al. A comprehensive model for assessment of liver stage therapies targeting Plasmodium vivax and Plasmodium falciparum. Nat. Commun. 2018, 9, 1837. [Google Scholar] [CrossRef]
- Tweedell, R.E.; Tao, D.; Hamerly, T.; Robinson, T.M.; Larsen, S.; Grønning, A.G.B.; Norris, A.M.; King, J.G.; Law, H.C.H.; Baumbach, J.; et al. The Selection of a Hepatocyte Cell Line Susceptible to Plasmodium falciparum Sporozoite Invasion That Is Associated With Expression of Glypican-3. Front. Microbiol. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Chattopadhyay, R.; De La Vega, P.; Paik, S.H.; Murata, Y.; Ferguson, E.W.; Richie, T.L.; Ooi, G.T. Early transcriptional responses of HepG2-A16 liver cells to infection by Plasmodium falciparum sporozoites. J. Biol. Chem. 2011, 286, 26396–26405. [Google Scholar] [CrossRef]
- Gerold, G.; Meissner, F.; Bruening, J.; Welsch, K.; Perin, P.M.; Baumert, T.F.; Vondran, F.W.; Kaderali, L.; Marcotrigiano, J.; Khan, A.G.; et al. Quantitative Proteomics Identifies Serum Response Factor Binding Protein 1 as a Host Factor for Hepatitis C Virus Entry. Cell Rep. 2015, 12, 864–878. [Google Scholar] [CrossRef]
- Scarselli, E.; Ansuini, H.; Cerino, R.; Roccasecca, R.M.; Acali, S.; Filocamo, G.; Traboni, C.; Nicosia, A.; Cortese, R.; Vitelli, A. The human scavenger receptor class B type I is a novel candidate receptor for the hepatitis C virus. EMBO J. 2002, 21, 5017–5025. [Google Scholar] [CrossRef]
- Shi, Q.; Jiang, J.; Luo, G. Syndecan-1 Serves as the Major Receptor for Attachment of Hepatitis C Virus to the Surfaces of Hepatocytes. J. Virol. 2013, 87, 6866–6875. [Google Scholar] [CrossRef]
- Zhang, J.; Randall, G.; Higginbottom, A.; Monk, P.; Rice, C.M.; McKeating, J.A. CD81 Is Required for Hepatitis C Virus Glycoprotein-Mediated Viral Infection. J. Virol. 2004, 78, 1448–1455. [Google Scholar] [CrossRef]
- Verrier, E.R.; Colpitts, C.C.; Bach, C.; Heydmann, L.; Weiss, A.; Renaud, M.; Durand, S.C.; Habersetzer, F.; Durantel, D.; Abou-Jaoudé, G.; et al. A targeted functional RNA interference screen uncovers glypican 5 as an entry factor for hepatitis B and D viruses. Hepatology 2016, 63, 35–48. [Google Scholar] [CrossRef]
- Yan, H.; Zhong, G.; Xu, G.; He, W.; Jing, Z.; Gao, Z.; Huang, Y.; Qi, Y.; Peng, B.; Wang, H.; et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. eLife 2012, 1, 1–28. [Google Scholar] [CrossRef]
- Jiang, J.; Cun, W.; Wu, X.; Shi, Q.; Tang, H.; Luo, G. Hepatitis C Virus Attachment Mediated by Apolipoprotein E Binding to Cell Surface Heparan Sulfate. J. Virol. 2012, 86, 7256–7267. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.S.; Bradrick, S.; Qiang, G.; Mostafavi, A.; Chaturvedi, G.; Weinman, S.A.; Diehl, A.M.; Jhaveri, R. Up-regulation of Hedgehog pathway is associated with cellular permissiveness for hepatitis C virus replication. Hepatology 2011, 54, 1580–1590. [Google Scholar] [CrossRef] [PubMed]
- Sumpter, R.; Loo, Y.-M.; Foy, E.; Li, K.; Yoneyama, M.; Fujita, T.; Lemon, S.M.; Gale, M. Regulating Intracellular Antiviral Defense and Permissiveness to Hepatitis C Virus RNA Replication through a Cellular RNA Helicase, RIG-I. J. Virol. 2005, 79, 2689–2699. [Google Scholar] [CrossRef] [PubMed]
- Gripon, P.; Rumin, S.; Urban, S.; Le Seyec, J.; Glaise, D.; Cannie, I.; Guyomard, C.; Lucas, J.; Trepo, C.; Guguen-Guillouzo, C. Infection of a human hepatoma cell line by hepatitis B virus. Proc. Natl. Acad. Sci. USA 2002, 99, 15655–15660. [Google Scholar] [CrossRef] [PubMed]
- Luangsay, S.; Ait-Goughoulte, M.; Michelet, M.; Floriot, O.; Bonnin, M.; Gruffaz, M.; Rivoire, M.; Fletcher, S.; Javanbakht, H.; Lucifora, J.; et al. Expression and functionality of Toll- and RIG-like receptors in HepaRG cells. J. Hepatol. 2015, 63, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Lucifora, J.; Durantel, D.; Testoni, B.; Hantz, O.; Levrero, M.; Zoulim, F. Control of hepatitis B virus replication by innate response of HepaRG cells. Hepatology 2010, 51, 63–72. [Google Scholar] [CrossRef]
- Ni, Y.; Lempp, F.A.; Mehrle, S.; Nkongolo, S.; Kaufman, C.; Fälth, M.; Stindt, J.; Königer, C.; Nassal, M.; Kubitz, R.; et al. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes. Gastroenterology 2014, 146. [Google Scholar] [CrossRef]
- König, A.; Yang, J.; Jo, E.; Park, K.H.P.; Kim, H.; Than, T.T.; Song, X.; Qi, X.; Dai, X.; Park, S.; et al. Efficient long-term amplification of hepatitis B virus isolates after infection of slow proliferating HepG2-NTCP cells. J. Hepatol. 2019, 71, 289–300. [Google Scholar] [CrossRef]
- Ploss, A.; Evans, M.J.; Gaysinskaya, V.A.; Panis, M.; You, H.; de Jong, Y.P.; Rice, C.M. Human occludin is a hepatitis C virus entry factor required for infection of mouse cells. Nature 2009, 457, 882–886. [Google Scholar] [CrossRef]
- Evans, M.J.; von Hahn, T.; Tscherne, D.M.; Syder, A.J.; Panis, M.; Wölk, B.; Hatziioannou, T.; McKeating, J.A.; Bieniasz, P.D.; Rice, C.M. Claudin-1 is a hepatitis C virus co-receptor required for a late step in entry. Nature 2007, 446, 801–805. [Google Scholar] [CrossRef]
- Olsavsky, K.M.; Page, J.L.; Johnson, M.C.; Zarbl, H.; Strom, S.C.; Omiecinski, C.J. Gene expression profiling and differentiation assessment in primary human hepatocyte cultures, established hepatoma cell lines, and human liver tissues. Toxicol. Appl. Pharmacol. 2007, 222, 42–56. [Google Scholar] [CrossRef]
- Yang, W.; Qiu, C.; Biswas, N.; Jin, J.; Watkins, S.C.; Montelaro, R.C.; Coyne, C.B.; Wang, T. Correlation of the tight junction-like distribution of claudin-1 to the cellular tropism of hepatitis C virus. J. Biol. Chem. 2008, 283, 8643–8653. [Google Scholar] [CrossRef]
- Fukuhara, T.; Matsuura, Y. Role of miR-122 and lipid metabolism in HCV infection. J. Gastroenterol. 2013, 48, 169–176. [Google Scholar] [CrossRef]
- Jopling, C.L. Modulation of Hepatitis C Virus RNA Abundance by a Liver-Specific MicroRNA. Science 2005, 309, 1577–1581. [Google Scholar] [CrossRef]
- Narbus, C.M.; Israelow, B.; Sourisseau, M.; Michta, M.L.; Hopcraft, S.E.; Zeiner, G.M.; Evans, M.J. HepG2 Cells Expressing MicroRNA miR-122 Support the Entire Hepatitis C Virus Life Cycle. J. Virol. 2011, 85, 12087–12092. [Google Scholar] [CrossRef]
- Saeed, M.; Andreo, U.; Chung, H.Y.; Espiritu, C.; Branch, A.D.; Silva, J.M.; Rice, C.M. SEC14L2 enables pan-genotype HCV replication in cell culture. Nature 2015, 524, 471–475. [Google Scholar] [CrossRef]
- Risco-Castillo, V.; Topçu, S.; Marinach, C.; Manzoni, G.; Bigorgne, A.E.; Briquet, S.; Baudin, X.; Lebrun, M.; Dubremetz, J.-F.; Silvie, O. Malaria Sporozoites Traverse Host Cells within Transient Vacuoles. Cell Host Microbe 2015, 18, 593–603. [Google Scholar] [CrossRef]
- Bano, N.; Romano, J.D.; Jayabalasingham, B.; Coppens, I. Cellular interactions of Plasmodium liver stage with its host mammalian cell. Int. J. Parasitol. 2007, 37, 1329–1341. [Google Scholar] [CrossRef]
- Sattabongkot, J.; Yimamnuaychoke, N.; Leelaudomlipi, S.; Rasameesoraj, M.; Jenwithisuk, R.; Coleman, R.E.; Udomsangpetch, R.; Cui, L.; Brewer, T.G. Establishment of a human hepatocyte line that supports in vitro development of the exo-erythrocytic stages of the malaria parasites Plasmodium falciparum and P. vivax. Am. J. Trop. Med. Hyg. 2006, 74, 708–715. [Google Scholar] [CrossRef]
- Elaut, G.; Henkens, T.; Papeleu, P.; Snykers, S.; Vinken, M.; Vanhaecke, T.; Rogiers, V. Molecular Mechanisms Underlying the Dedifferentiation Process of Isolated Hepatocytes and Their Cultures. Curr. Drug Metab. 2006, 7, 629–660. [Google Scholar] [CrossRef]
- Lucifora, J.; Michelet, M.; Rivoire, M.; Protzer, U.; Durantel, D.; Zoulim, F. Two-dimensional-cultures of primary human hepatocytes allow efficient HBV infection: Old tricks still work! J. Hepatol. 2020, 73, 449–451. [Google Scholar] [CrossRef]
- Xiang, C.; Du, Y.; Meng, G.; Yi, L.S.; Sun, S.; Song, N.; Zhang, X.; Xiao, Y.; JieWan, J.; Yi, Z.; et al. Long-term functional maintenance of primary human hepatocytes in vitro. Science 2019, 364, 399–402. [Google Scholar] [CrossRef]
- Michailidis, E.; Vercauteren, K.; Mancio-Silva, L.; Andrus, L.; Jahan, C.; Ricardo-Lax, I.; Zou, C.; Kabbani, M.; Park, P.; Quirk, C.; et al. Expansion, in vivo-ex vivo cycling, and genetic manipulation of primary human hepatocytes. Proc. Natl. Acad. Sci. USA 2020, 117, 1678–1688. [Google Scholar] [CrossRef]
- Ishida, Y.; Yamasaki, C.; Yanagi, A.; Yoshizane, Y.; Fujikawa, K.; Watashi, K.; Abe, H.; Wakita, T.; Hayes, C.N.; Chayama, K.; et al. Novel robust in vitro hepatitis B virus infection model using fresh human hepatocytes isolated from humanized mice. Am. J. Pathol. 2015, 185, 1275–1285. [Google Scholar] [CrossRef]
- Roelandt, P.; Obeid, S.; Paeshuyse, J.; Vanhove, J.; Van Lommel, A.; Nahmias, Y.; Nevens, F.; Neyts, J.; Verfaillie, C.M. Human pluripotent stem cell-derived hepatocytes support complete replication of hepatitis C virus. J. Hepatol. 2012, 57, 246–251. [Google Scholar] [CrossRef]
- Ng, S.; Schwartz, R.E.; March, S.; Galstian, A.; Gural, N.; Shan, J.; Prabhu, M.; Mota, M.M.; Bhatia, S.N. Human iPSC-derived hepatocyte-like cells support plasmodium liver-stage infection in vitro. Stem Cell Rep. 2015, 4, 348–359. [Google Scholar] [CrossRef]
- Wu, X.; Robotham, J.M.; Lee, E.; Dalton, S.; Kneteman, N.M.; Gilbert, D.M.; Tang, H. Productive hepatitis C virus infection of stem cell-derived hepatocytes reveals a critical transition to viral permissiveness during differentiation. PLoS Pathog. 2012, 8. [Google Scholar] [CrossRef]
- Yan, F.; Wang, Y.; Zhang, W.; Chang, M.; He, Z.; Xu, J.; Shang, C.; Chen, T.; Liu, J.; Wang, X.; et al. Human embryonic stem cell–derived hepatoblasts are an optimal lineage stage for hepatitis C virus infection. Hepatology 2017, 66, 717–735. [Google Scholar] [CrossRef]
- Xia, Y.; Carpentier, A.; Cheng, X.; Block, P.D.; Zhao, Y.; Zhang, Z.; Protzer, U.; Liang, T.J. Human stem cell-derived hepatocytes as a model for hepatitis B virus infection, spreading and virus-host interactions. J. Hepatol. 2017, 66, 494–503. [Google Scholar] [CrossRef]
- Sakurai, F.; Mitani, S.; Yamamoto, T.; Takayama, K.; Tachibana, M.; Watashi, K.; Wakita, T.; Iijima, S.; Tanaka, Y.; Mizuguchi, H. Human induced-pluripotent stem cell-derived hepatocyte-like cells as an in vitro model of human hepatitis B virus infection. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Shlomai, A.; Schwartz, R.E.; Ramanan, V.; Bhatta, A.; De Jong, Y.P.; Bhatia, S.N.; Rice, C.M. Modeling host interactions with hepatitis B virus using primary and induced pluripotent stem cell-derived hepatocellular systems. Proc. Natl. Acad. Sci. USA 2014, 111, 12193–12198. [Google Scholar] [CrossRef] [PubMed]
- Cotovio, J.P.; Fernandes, T.G. Production of Human Pluripotent Stem Cell-Derived Hepatic Cell Lineages and Liver Organoids: Current Status and Potential Applications. Bioengineering 2020, 7, 36. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Matsuzaki, J.; Katsuda, T.; Saito, Y.; Saito, H.; Ochiya, T. Generation of functional human hepatocytes in vitro: Current status and future prospects. Inflamm. Regen. 2019, 39, 13. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Liu, X.; Zhang, L.; Li, X.; Zhang, Y.; Wu, K.; Chen, Y.; Cao, J.; Hou, W.; Zhang, J.; et al. A chimeric humanized mouse model by engrafting the human induced pluripotent stem cell-derived hepatocyte-like cell for the chronic hepatitis B virus infection. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, L.; Hui, L. Cell Plasticity in Liver Regeneration. Trends Cell Biol. 2020, 30, 329–338. [Google Scholar] [CrossRef]
- Petropolis, D.B.; Faust, D.M.; Tolle, M.; Rivière, L.; Valentin, T.; Neuveut, C.; Hernandez-Cuevas, N.; Dufour, A.; Olivo-Marin, J.-C.; Guillen, N. Human Liver Infection in a Dish: Easy-To-Build 3D Liver Models for Studying Microbial Infection. PLoS ONE 2016, 11, e0148667. [Google Scholar] [CrossRef]
- Khetani, S.R.; Bhatia, S.N. Microscale culture of human liver cells for drug development. Nat. Biotechnol. 2008, 26, 120–126. [Google Scholar] [CrossRef]
- Dembélé, L.; Franetich, J.-F.; Lorthiois, A.; Gego, A.; Zeeman, A.-M.; Kocken, C.H.M.; Le Grand, R.; Dereuddre-Bosquet, N.; van Gemert, G.-J.; Sauerwein, R.; et al. Persistence and activation of malaria hypnozoites in long-term primary hepatocyte cultures. Nat. Med. 2014, 20, 307–312. [Google Scholar] [CrossRef]
- Winer, B.Y.; Huang, T.S.; Pludwinski, E.; Heller, B.; Wojcik, F.; Lipkowitz, G.E.; Parekh, A.; Cho, C.; Shrirao, A.; Muir, T.W.; et al. Long-term hepatitis B infection in a scalable hepatic co-culture system. Nat. Commun. 2017, 8. [Google Scholar] [CrossRef]
- Winer, B.Y.; Gaska, J.M.; Lipkowitz, G.; Bram, Y.; Parekh, A.; Parsons, L.; Leach, R.; Jindal, R.; Cho, C.H.; Shrirao, A.; et al. Analysis of Host Responses to Hepatitis B and Delta Viral Infections in a Micro-scalable Hepatic Co-culture System. Hepatology 2020, 71, 14–30. [Google Scholar] [CrossRef]
- March, S.; Ramanan, V.; Trehan, K.; Ng, S.; Galstian, A.; Gural, N.; Scull, M.A.; Shlomai, A.; Mota, M.M.; Fleming, H.E.; et al. Micropatterned coculture of primary human hepatocytes and supportive cells for the study of hepatotropic pathogens. Nat. Protoc. 2015, 10, 2027–2053. [Google Scholar] [CrossRef]
- Ploss, A.; Khetani, S.R.; Jones, C.T.; Syder, A.J.; Trehan, K.; Gaysinskaya, V.A.; Mu, K.; Ritola, K.; Rice, C.M.; Bhatia, S.N. Persistent hepatitis C virus infection in microscale primary human hepatocyte cultures. Proc. Natl. Acad. Sci. USA 2010, 107, 3141–3145. [Google Scholar] [CrossRef]
- Gural, N.; Mancio-Silva, L.; He, J.; Bhatia, S.N. Engineered Livers for Infectious Diseases. Cell. Mol. Gastroenterol. Hepatol. 2018, 5, 131–144. [Google Scholar] [CrossRef]
- Kinge, C.N.W.; Bhoola, N.H.; Kramvis, A. In vitro systems for studying different genotypes/sub-genotypes of hepatitis B virus: Strengths and limitations. Viruses 2020, 12, 353. [Google Scholar]
- Witt-Kehati, D.; Alaluf, M.B.; Shlomai, A. Advances and challenges in studying hepatitis B virus In vitro. Viruses 2016, 8, 21. [Google Scholar] [CrossRef]
- Gong, Z.J.; De Meyer, S.; Roskams, T.; van Pelt, J.F.; Soumillion, A.; Crabbe, T.; Yap, S.H. Hepatitis B virus infection in microcarrier-attached immortalized human hepatocytes cultured in molecularporous membrane bags: A model for long-term episomal replication of HBV. J. Viral Hepat. 1998, 5, 377–387. [Google Scholar] [CrossRef]
- Sainz, B.; TenCate, V.; Uprichard, S.L. Three-dimensional Huh7 cell culture system for the study of Hepatitis C virus infection. Virol. J. 2009, 6, 103. [Google Scholar] [CrossRef]
- Serra, M.; Brito, C.; Correia, C.; Alves, P.M. Process engineering of human pluripotent stem cells for clinical application. Trends Biotechnol. 2012, 30, 350–359. [Google Scholar] [CrossRef]
- Silva, A.C.; Delgado, I.; Sousa, M.F.Q.; Carrondo, M.J.T.; Alves, P.M. Scalable culture systems using different cell lines for the production of Peste des Petits ruminants vaccine. Vaccine 2008, 26, 3305–3311. [Google Scholar] [CrossRef]
- Sousa, M.; Fenge, C.; Rupprecht, J.; Tappe, A.; Greller, G.; Alves, P.; Carrondo, M.; Roldão, A. Process intensification for Peste des Petites Ruminants Virus vaccine production. Vaccine 2019, 37, 7041–7051. [Google Scholar] [CrossRef]
- Cunha, B.; Aguiar, T.; Silva, M.M.; Silva, R.J.S.; Sousa, M.F.Q.; Pineda, E.; Peixoto, C.; Carrondo, M.J.T.; Serra, M.; Alves, P.M. Exploring continuous and integrated strategies for the up- and downstream processing of human mesenchymal stem cells. J. Biotechnol. 2015, 213, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Lupberger, J.; Mund, A.; Kock, J.; Hildt, E. Cultivation of HepG2.2.15 on Cytodex-3: Higher yield of hepatitis B virus and less subviral particles compared to conventional culture methods. J. Hepatol. 2006, 45, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Ishii, K.; Ishihara, Y.; Yoshizaki, S.; Tanaka, K.; Gotoh, Y.; Aizaki, H.; Kohara, M.; Yoshioka, H.; Mori, Y.; et al. Production of infectious hepatitis C virus particles in three-dimensional cultures of the cell line carrying the genome-length dicistronic viral RNA of genotype 1b. Virology 2006, 351, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Sousa, M.F.Q.; Silva, M.M.; Giroux, D.; Hashimura, Y.; Wesselschmidt, R.; Lee, B.; Roldão, A.; Carrondo, M.J.T.; Alves, P.M.; Serra, M. Production of oncolytic adenovirus and human mesenchymal stem cells in a single-use, Vertical-Wheel bioreactor system: Impact of bioreactor design on performance of microcarrier-based cell culture processes. Biotechnol. Prog. 2015, 31, 1600–1612. [Google Scholar] [CrossRef] [PubMed]
- Marcelino, I.; Sousa, M.; Verissimo, C.; Cunha, A.; Carrondo, M.; Alves, P. Process development for the mass production of Ehrlichia ruminantium. Vaccine 2006, 24, 1716–1725. [Google Scholar] [CrossRef] [PubMed]
- Widell, A.; Hansson, B.G.; Nordenfelt, E. A microcarrier cell culture system for large scale production of hepatitis a virus. J. Virol. Methods 1984, 8, 63–71. [Google Scholar] [CrossRef]
- Akahori, Y.; Kato, H.; Fujita, T.; Moriishi, K.; Tanaka, Y.; Watashi, K.; Imamura, M.; Chayama, K.; Wakita, T.; Hijikata, M. Establishment of a novel hepatitis B virus culture system using immortalized human hepatocytes. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Molina-Jimenez, F.; Benedicto, I.; Dao Thi, V.L.; Gondar, V.; Lavillette, D.; Marin, J.J.; Briz, O.; Moreno-Otero, R.; Aldabe, R.; Baumert, T.F.; et al. Matrigel-embedded 3D culture of Huh-7 cells as a hepatocyte-like polarized system to study hepatitis C virus cycle. Virology 2012, 425, 31–39. [Google Scholar] [CrossRef]
- Wang, W.; Soroka, C.J.; Mennone, A.; Rahner, C.; Harry, K.; Pypaert, M.; Boyer, J.L. Radixin Is Required to Maintain Apical Canalicular Membrane Structure and Function in Rat Hepatocytes. Gastroenterology 2006, 131, 878–884. [Google Scholar] [CrossRef]
- Clapéron, A.; Debray, D.; Redon, M.-J.; Mergey, M.; Nguyen Ho-Bouldoires, T.H.; Housset, C.; Fabre, M.; Fouassier, L. Immunohistochemical profile of ezrin and radixin in human liver epithelia during fetal development and pediatric cholestatic diseases. Clin. Res. Hepatol. Gastroenterol. 2013, 37, 142–151. [Google Scholar] [CrossRef]
- Baktash, Y.; Madhav, A.; Coller, K.E.; Randall, G. Single Particle Imaging of Polarized Hepatoma Organoids upon Hepatitis C Virus Infection Reveals an Ordered and Sequential Entry Process. Cell Host Microbe 2018, 23, 382–394.e5. [Google Scholar] [CrossRef]
- Rajalakshmy, A.; Malathi, J.; Madhavan, H.; Samuel, J. Mebiolgel, a thermoreversible polymer as a scaffold for three dimensional culture of Huh7 cell line with improved hepatocyte differentiation marker expression and HCV replication. Indian J. Med. Microbiol. 2015, 33, 554. [Google Scholar] [CrossRef]
- Jayal, P.; Behera, P.; Mullick, R.; Ramachandra, S.G.; Das, S.; Kumar, A.; Karande, A. Responsive polymer-assisted 3D cryogel supports Huh7.5 as in vitro hepatitis C virus model and ectopic human hepatic tissue in athymic mice. Biotechnol. Bioeng. 2021, 118, 1286–1304. [Google Scholar] [CrossRef]
- Nugraha, B.; Hong, X.; Mo, X.; Tan, L.; Zhang, W.; Chan, P.-M.; Kang, C.H.; Wang, Y.; Beng, L.T.; Sun, W.; et al. Galactosylated cellulosic sponge for multi-well drug safety testing. Biomaterials 2011, 32, 6982–6994. [Google Scholar] [CrossRef]
- Ananthanarayanan, A.; Nugraha, B.; Triyatni, M.; Hart, S.; Sankuratri, S.; Yu, H. Scalable Spheroid Model of Human Hepatocytes for Hepatitis C Infection and Replication. Mol. Pharm. 2014, 11, 2106–2114. [Google Scholar] [CrossRef]
- Wells, R.G. The role of matrix stiffness in regulating cell behavior. Hepatology 2008, 47, 1394–1400. [Google Scholar] [CrossRef]
- Zeisel, M.B.; Fofana, I.; Fafi-Kremer, S.; Baumert, T.F. Hepatitis C virus entry into hepatocytes: Molecular mechanisms and targets for antiviral therapies. J. Hepatol. 2011, 54, 566–576. [Google Scholar] [CrossRef]
- Chua, A.C.Y.; Ananthanarayanan, A.; Ong, J.J.Y.; Wong, J.Y.; Yip, A.; Singh, N.H.; Qu, Y.; Dembele, L.; McMillian, M.; Ubalee, R.; et al. Hepatic spheroids used as an in vitro model to study malaria relapse. Biomaterials 2019, 216, 119221. [Google Scholar] [CrossRef]
- Zeeman, A.-M.; Lakshminarayana, S.B.; van der Werff, N.; Klooster, E.J.; Voorberg-van der Wel, A.; Kondreddi, R.R.; Bodenreider, C.; Simon, O.; Sauerwein, R.; Yeung, B.K.S.; et al. PI4 Kinase Is a Prophylactic but Not Radical Curative Target in Plasmodium vivax-Type Malaria Parasites. Antimicrob. Agents Chemother. 2016, 60, 2858–2863. [Google Scholar] [CrossRef]
- McCrary, M.W.; Bousalis, D.; Mobini, S.; Song, Y.H.; Schmidt, C.E. Decellularized tissues as platforms for in vitro modeling of healthy and diseased tissues. Acta Biomater. 2020, 111, 1–19. [Google Scholar] [CrossRef]
- Zhang, Z.Z.; Xu, H.M.; Mazza, G.; Zhang, M.M.; Frenguelli, L.; Liu, Q.B.; Al-Akkad, W.; Ren, J.H.; Zhao, R.Q.; Ren, F.; et al. Decellularized human liver scaffold-based three-dimensional culture system facilitate hepatitis B virus infection. J. Biomed. Mater. Res. Part A 2019, 107, 1744–1753. [Google Scholar] [CrossRef]
- Bell, C.C.; Dankers, A.C.A.; Lauschke, V.M.; Sison-Young, R.; Jenkins, R.; Rowe, C.; Goldring, C.E.; Park, K.; Regan, S.L.; Walker, T.; et al. Comparison of Hepatic 2D Sandwich Cultures and 3D Spheroids for Long-term Toxicity Applications: A Multicenter Study. Toxicol. Sci. 2018, 162, 655–666. [Google Scholar] [CrossRef]
- Bell, C.C.; Hendriks, D.F.G.; Moro, S.M.L.; Ellis, E.; Walsh, J.; Renblom, A.; Fredriksson Puigvert, L.; Dankers, A.C.A.; Jacobs, F.; Snoeys, J.; et al. Characterization of primary human hepatocyte spheroids as a model system for drug-induced liver injury, liver function and disease. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef]
- Tostões, R.M.; Leite, S.B.; Serra, M.; Jensen, J.; Björquist, P.; Carrondo, M.J.T.; Brito, C.; Alves, P.M. Human liver cell spheroids in extended perfusion bioreactor culture for repeated-dose drug testing. Hepatology 2012, 55, 1227–1236. [Google Scholar] [CrossRef]
- Rebelo, S.P.; Costa, R.; Silva, M.M.; Marcelino, P.; Brito, C.; Alves, P.M. Three-dimensional co-culture of human hepatocytes and mesenchymal stem cells: Improved functionality in long-term bioreactor cultures. J. Tissue Eng. Regen. Med. 2017, 11, 2034–2045. [Google Scholar] [CrossRef]
- Leite, S.B.; Wilk-Zasadna, I.; Zaldivar, J.M.; Airola, E.; Reis-Fernandes, M.A.; Mennecozzi, M.; Guguen-Guillouzo, C.; Chesne, C.; Guillou, C.; Alves, P.M.; et al. Three-Dimensional HepaRG Model As An Attractive Tool for Toxicity Testing. Toxicol. Sci. 2012, 130, 106–116. [Google Scholar] [CrossRef]
- Rebelo, S.P.; Costa, R.; Estrada, M.; Shevchenko, V.; Brito, C.; Alves, P.M. HepaRG microencapsulated spheroids in DMSO-free culture: Novel culturing approaches for enhanced xenobiotic and biosynthetic metabolism. Arch. Toxicol. 2015, 89, 1347–1358. [Google Scholar] [CrossRef] [PubMed]
- Arez, F.; Rebelo, S.P.; Fontinha, D.; Simão, D.; Martins, T.R.; Machado, M.; Fischli, C.; Oeuvray, C.; Badolo, L.; Carrondo, M.J.T.; et al. Flexible 3D Cell-Based Platforms for the Discovery and Profiling of Novel Drugs Targeting Plasmodium Hepatic Infection. ACS Infect. Dis. 2019, 5, 1831–1842. [Google Scholar] [CrossRef] [PubMed]
- Dumoulin, P.C.; Trop, S.A.; Ma, J.; Zhang, H.; Sherman, M.A.; Levitskaya, J. Flow Cytometry Based Detection and Isolation of Plasmodium falciparum Liver Stages In Vitro. PLoS ONE 2015, 10, e0129623. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Prieto, A.M.; Skelton, J.K.; Wai, S.N.; Large, E.; Lussignol, M.; Vizcay-Barrena, G.; Hughes, D.; Fleck, R.A.; Thursz, M.; Catanese, M.T.; et al. 3D microfluidic liver cultures as a physiological preclinical tool for hepatitis B virus infection. Nat. Commun. 2018, 9, 682. [Google Scholar] [CrossRef]
- Hu, C.; Wu, Z.; Li, L. Mesenchymal stromal cells promote liver regeneration through regulation of immune cells. Int. J. Biol. Sci. 2020, 16, 893–903. [Google Scholar] [CrossRef]
- De Bartolo, L.; Salerno, S.; Morelli, S.; Giorno, L.; Rende, M.; Memoli, B.; Procino, A.; Andreucci, V.E.; Bader, A.; Drioli, E. Long-term maintenance of human hepatocytes in oxygen-permeable membrane bioreactor. Biomaterials 2006, 27, 4794–4803. [Google Scholar] [CrossRef]
- Bell, C.C.; Chouhan, B.; Andersson, L.C.; Andersson, H.; Dear, J.W.; Williams, D.P.; Söderberg, M. Functionality of primary hepatic non-parenchymal cells in a 3D spheroid model and contribution to acetaminophen hepatotoxicity. Arch. Toxicol. 2020, 94, 1251–1263. [Google Scholar] [CrossRef]
- Simian, M.; Bissell, M.J. Organoids: A historical perspective of thinking in three dimensions. J. Cell Biol. 2017, 216, 31–40. [Google Scholar] [CrossRef]
- Huch, M.; Koo, B.K. Modeling mouse and human development using organoid cultures. Development 2015, 142, 3113–3125. [Google Scholar] [CrossRef]
- Nuciforo, S.; Heim, M.H. Organoids to model liver disease. JHEP Rep. 2021, 3, 100198. [Google Scholar] [CrossRef]
- Nie, Y.-Z.; Zheng, Y.-W.; Miyakawa, K.; Murata, S.; Zhang, R.-R.; Sekine, K.; Ueno, Y.; Takebe, T.; Wakita, T.; Ryo, A.; et al. Recapitulation of hepatitis B virus–host interactions in liver organoids from human induced pluripotent stem cells. EBioMedicine 2018, 35, 114–123. [Google Scholar] [CrossRef]
- Camp, J.G.; Sekine, K.; Gerber, T.; Loeffler-Wirth, H.; Binder, H.; Gac, M.; Kanton, S.; Kageyama, J.; Damm, G.; Seehofer, D.; et al. Multilineage communication regulates human liver bud development from pluripotency. Nature 2017, 546, 533–538. [Google Scholar] [CrossRef]
- Zaret, K.S. Regulatory phases of early liver development: Paradigms of organogenesis. Nat. Rev. Genet. 2002, 3, 499–512. [Google Scholar] [CrossRef]
- Meuleman, P.; Libbrecht, L.; Wieland, S.; De Vos, R.; Habib, N.; Kramvis, A.; Roskams, T.; Leroux-Roels, G. Immune Suppression Uncovers Endogenous Cytopathic Effects of the Hepatitis B Virus. J. Virol. 2006, 80, 2797–2807. [Google Scholar] [CrossRef]
- Shin, E.-C.; Sung, P.S.; Park, S.-H. Immune responses and immunopathology in acute and chronic viral hepatitis. Nat. Rev. Immunol. 2016, 16, 509–523. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.-C.; Sun, T.; Ching, A.K.K.; He, M.; Li, J.-W.; Wong, A.M.; Co, N.N.; Chan, A.W.H.; Li, P.-S.; Lung, R.W.M.; et al. Viral-Human Chimeric Transcript Predisposes Risk to Liver Cancer Development and Progression. Cancer Cell 2014, 25, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Lau, D.T.-Y.; Negash, A.; Chen, J.; Crochet, N.; Sinha, M.; Zhang, Y.; Guedj, J.; Holder, S.; Saito, T.; Lemon, S.M.; et al. Innate Immune Tolerance and the Role of Kupffer Cells in Differential Responses to Interferon Therapy Among Patients With HCV Genotype 1 Infection. Gastroenterology 2013, 144, 402–413.e12. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carpentier, A.; Sheldon, J.; Vondran, F.W.R.; Brown, R.J.P.; Pietschmann, T. Efficient acute and chronic infection of stem cell-derived hepatocytes by hepatitis C virus. Gut 2020, 69, 1659–1666. [Google Scholar] [CrossRef]
- Cheng, X.; Xia, Y.; Serti, E.; Block, P.D.; Chung, M.; Chayama, K.; Rehermann, B.; Liang, T.J. Hepatitis B virus evades innate immunity of hepatocytes but activates cytokine production by macrophages. Hepatology 2017, 66, 1779–1793. [Google Scholar] [CrossRef]
- Cha, S.-J.; Park, K.; Srinivasan, P.; Schindler, C.W.; van Rooijen, N.; Stins, M.; Jacobs-Lorena, M. CD68 acts as a major gateway for malaria sporozoite liver infection. J. Exp. Med. 2015, 212, 1391–1403. [Google Scholar] [CrossRef]
- Nlinwe, O.N.; Kusi, K.A.; Adu, B.; Sedegah, M. T-cell responses against Malaria: Effect of parasite antigen diversity and relevance for vaccine development. Vaccine 2018, 36, 2237–2242. [Google Scholar] [CrossRef]
- Wahlicht, T.; Vièyres, G.; Bruns, S.A.; Meumann, N.; Büning, H.; Hauser, H.; Schmitz, I.; Pietschmann, T.; Wirth, D. Controlled Functional Zonation of Hepatocytes In Vitro by Engineering of Wnt Signaling. ACS Synth. Biol. 2020, 9, 1638–1649. [Google Scholar] [CrossRef]
- Ebrahimkhani, M.R.; Neiman, J.A.S.; Raredon, M.S.B.; Hughes, D.J.; Griffith, L.G. Bioreactor technologies to support liver function in vitro. Adv. Drug Deliv. Rev. 2014, 69–70, 132–157. [Google Scholar] [CrossRef]
- Simão, D.; Pinto, C.; Fernandes, P.; Peddie, C.J.; Piersanti, S.; Collinson, L.M.; Salinas, S.; Saggio, I.; Schiavo, G.; Kremer, E.J.; et al. Evaluation of helper-dependent canine adenovirus vectors in a 3D human CNS model. Gene Ther. 2016, 23, 86–94. [Google Scholar] [CrossRef]
- Esch, E.W.; Bahinski, A.; Huh, D. Organs-on-chips at the frontiers of drug discovery. Nat. Rev. Drug Discov. 2015, 14, 248–260. [Google Scholar] [CrossRef]
- Domansky, K.; Inman, W.; Serdy, J.; Dash, A.; Lim, M.H.M.; Griffith, L.G. Perfused multiwell plate for 3D liver tissue engineering. Lab Chip 2010, 10, 51–58. [Google Scholar] [CrossRef]
- Moradi, E.; Jalili-Firoozinezhad, S.; Solati-Hashjin, M. Microfluidic organ-on-a-chip models of human liver tissue. Acta Biomater. 2020, 116, 67–83. [Google Scholar] [CrossRef]
- Kietzmann, T. Metabolic zonation of the liver: The oxygen gradient revisited. Redox Biol. 2017, 11, 622–630. [Google Scholar] [CrossRef]
- Cox, C.R.; Lynch, S.; Goldring, C.; Sharma, P. Current Perspective: 3D Spheroid Models Utilizing Human-Based Cells for Investigating Metabolism-Dependent Drug-Induced Liver Injury. Front. Med. Technol. 2020, 2. [Google Scholar] [CrossRef]
- Kang, Y.B.; Eo, J.; Mert, S.; Yarmush, M.L.; Usta, O.B. Metabolic Patterning on a Chip: Towards in vitro Liver Zonation of Primary Rat and Human Hepatocytes. Sci. Rep. 2018, 8, 8951. [Google Scholar] [CrossRef]
- Du, Y.; Li, N.; Yang, H.; Luo, C.; Gong, Y.; Tong, C.; Gao, Y.; Lü, S.; Long, M. Mimicking liver sinusoidal structures and functions using a 3D-configured microfluidic chip. Lab Chip 2017, 17, 782–794. [Google Scholar] [CrossRef]
- Trapecar, M.; Communal, C.; Velazquez, J.; Maass, C.A.; Huang, Y.-J.; Schneider, K.; Wright, C.W.; Butty, V.; Eng, G.; Yilmaz, O.; et al. Gut-Liver Physiomimetics Reveal Paradoxical Modulation of IBD-Related Inflammation by Short-Chain Fatty Acids. Cell Syst. 2020, 10, 223–239.e9. [Google Scholar] [CrossRef]
- Irimia, D.; Wang, X. Inflammation-on-a-Chip: Probing the Immune System Ex Vivo. Trends Biotechnol. 2018, 36, 923–937. [Google Scholar] [CrossRef]
- Bhise, N.S.; Manoharan, V.; Massa, S.; Tamayol, A.; Ghaderi, M.; Miscuglio, M.; Lang, Q.; Shrike Zhang, Y.; Shin, S.R.; Calzone, G.; et al. A liver-on-a-chip platform with bioprinted hepatic spheroids. Biofabrication 2016, 8, 014101. [Google Scholar] [CrossRef]
- Cuvellier, M.; Ezan, F.; Oliveira, H.; Rose, S.; Fricain, J.-C.; Langouët, S.; Legagneux, V.; Baffet, G. 3D culture of HepaRG cells in GelMa and its application to bioprinting of a multicellular hepatic model. Biomaterials 2021, 269, 120611. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.G.; Funk, J.; Robbins, J.B.; Crogan-Grundy, C.; Presnell, S.C.; Singer, T.; Roth, A.B. Bioprinted 3D Primary Liver Tissues Allow Assessment of Organ-Level Response to Clinical Drug Induced Toxicity In Vitro. PLoS ONE 2016, 11, e0158674. [Google Scholar] [CrossRef] [PubMed]
- Abaci, A.; Guvendiren, M. Designing Decellularized Extracellular Matrix-Based Bioinks for 3D Bioprinting. Adv. Healthc. Mater. 2020, 9, 2000734. [Google Scholar] [CrossRef] [PubMed]

| Family, Genus | Attachment and Entry Factors/Receptors | Main Life Cycle Hallmarks | Associated Diseases | Prevalence and Mortality b | |
|---|---|---|---|---|---|
| Genome | |||||
| Genotypes | |||||
| HAV | Picornaviridae, Hepatovirus | Viral: • - Host: • HAVCR1 | • Acute infection • Exists in two envelopment forms: naked and quasi-enveloped [4] • Does not integrate into the host genome • Direct translation of viral genome (IRES-mediated) • Release by cell lysis | • Mild hepatitis (common) • Fulminant acute hepatitis (rarely) | Every year, 1.4 million people are infected, and in 2016 7134 deaths occurred, mainly due to fulminant hepatitis. |
| Linear ssRNA (+) | |||||
| 6 (I to VI) | |||||
| HBV | Hepadnaviridae, Orthohepadnavirus | Viral: • (Large) S Host: • NTCP • HS | • Acute infection, may evolve to chronic • Enveloped virus • Can integrate into the host genome • The genome is a mini-chromosome transcribed by host polymerases into 4 mRNAs • Release by budding (ESCRT-dependent) | • Hepatitis • HCC • Cirrhosis | In 2015, 257 million people were estimated to be chronically infected, and 887,000 deaths occurred, mostly from cirrhosis and HCC. |
| Circular dsDNA (c) | |||||
| 10 (A to J) | |||||
| HCV | Flaviridae, Hepacivirus | Viral: • E1/E2 Host: • CD81 • HS • LDLR • SRBI • CLDN1 • OCLN • NPC1L1 | • Chronic infection • Enveloped virus • Does not integrate into the host genome • Direct translation of the viral genome (IRES-mediated) • Release by exocytosis after budding into the ER via lipoprotein assembly and secretion (reviewed in [5]) | • Hepatitis • HCC | In 2016, 71 million people were estimated to be chronically infected, and 399,000 deaths occurred, mostly from cirrhosis and HCC. |
| Linear ssRNA (+) | |||||
| 6 (1 to 6) | |||||
| HDV | Unassigned, Deltavirus | Viral: • HBV’s (Large) S Host: • NTCP • HS | • Acute infection • Enveloped virus • Does not integrate into the host genome • Satellite virus: infection requires the host cell to be co-infected with HBV. • Replication occurs by rolling circle, single genome being cleaved/ligated by HDV ribozymes • Release by budding using HBV proteins | • Fulminant acute hepatitis • Severe chronic active hepatitis • HCC | Affects 5% of people with HBV; deaths included in the HBV mortality. |
| Circular ssRNA (-) | |||||
| 8 (1 and 8) | |||||
| HEV | Hepeviridae, Orthohepevirus | Viral: • - Host: • HS • Unknown receptor | • Acute infection, may evolve to chronic • Exists in two envelopment forms: naked (enteric route) and quasi-enveloped (bloodstream) • Does not integrate into the host genome • Direct translation of the viral genome (caped genome) • Released by budding (ESCRT-dependent) at both the basolateral and apical side of the hepatocyte. The latter leads to striping-off of the envelope by bile acids resulting in naked particles [6] | • Mild hepatitis • Associated with a high mortality rate during pregnancy (fulminant liver failure) | Every year, 30 million people are infected with 3.3 million symptomatic cases, and in 2015, 44,000 deaths occurred, mainly due to fulminant hepatitis during pregnancy. |
| linear ssRNA (+) | |||||
| 4 (1 to 4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arez, F.; Rodrigues, A.F.; Brito, C.; Alves, P.M. Bioengineered Liver Cell Models of Hepatotropic Infections. Viruses 2021, 13, 773. https://doi.org/10.3390/v13050773
Arez F, Rodrigues AF, Brito C, Alves PM. Bioengineered Liver Cell Models of Hepatotropic Infections. Viruses. 2021; 13(5):773. https://doi.org/10.3390/v13050773
Chicago/Turabian StyleArez, Francisca, Ana F. Rodrigues, Catarina Brito, and Paula M. Alves. 2021. "Bioengineered Liver Cell Models of Hepatotropic Infections" Viruses 13, no. 5: 773. https://doi.org/10.3390/v13050773
APA StyleArez, F., Rodrigues, A. F., Brito, C., & Alves, P. M. (2021). Bioengineered Liver Cell Models of Hepatotropic Infections. Viruses, 13(5), 773. https://doi.org/10.3390/v13050773







