Circulation of Fluorescently Labelled Phage in a Murine Model
Abstract
1. Introduction
2. Materials and Methods
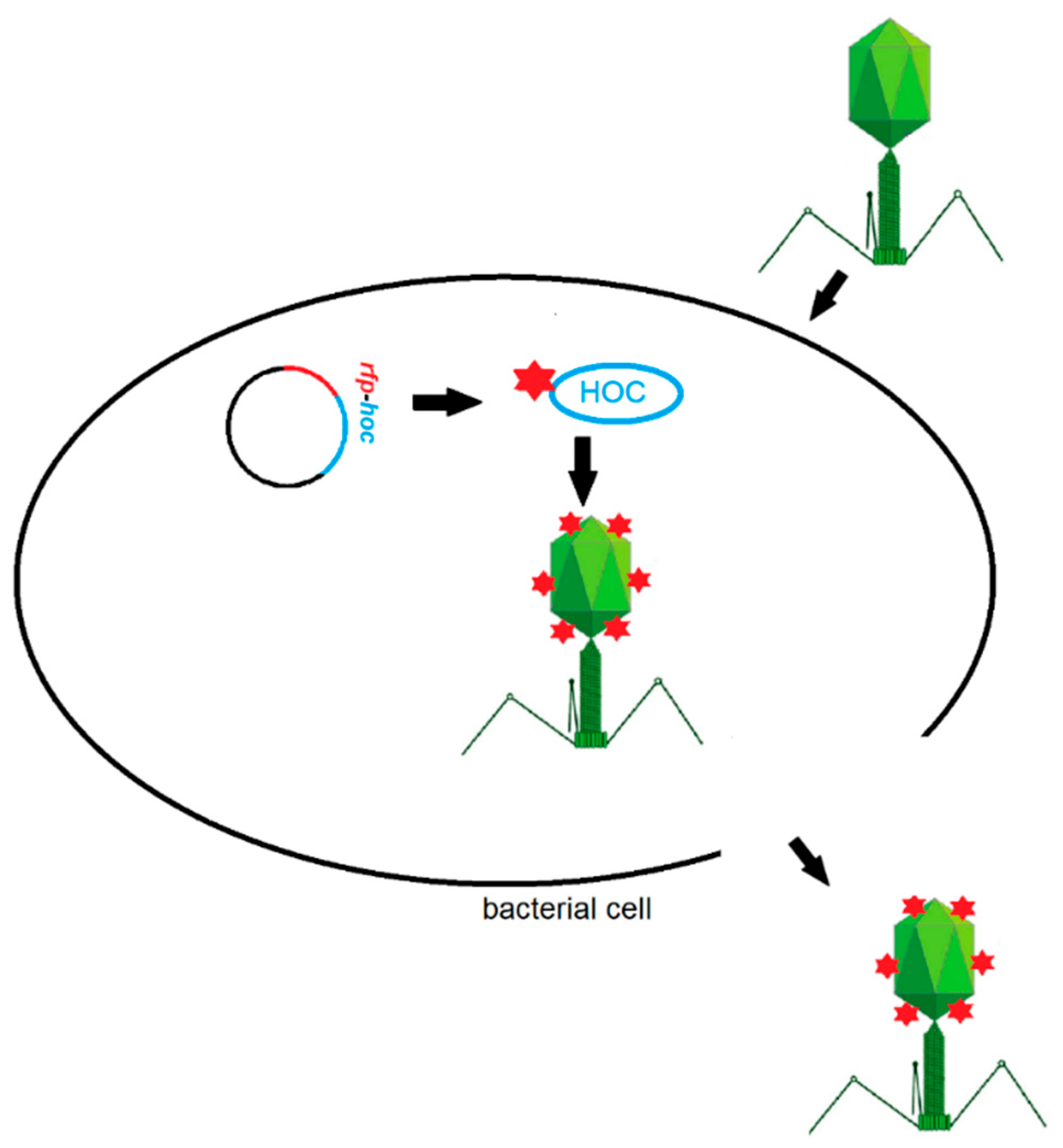
2.1. Construction of the rfp-hoc Fusion in the Expression Vector
2.2. Phage Display
2.3. Purification of Labelled Phage
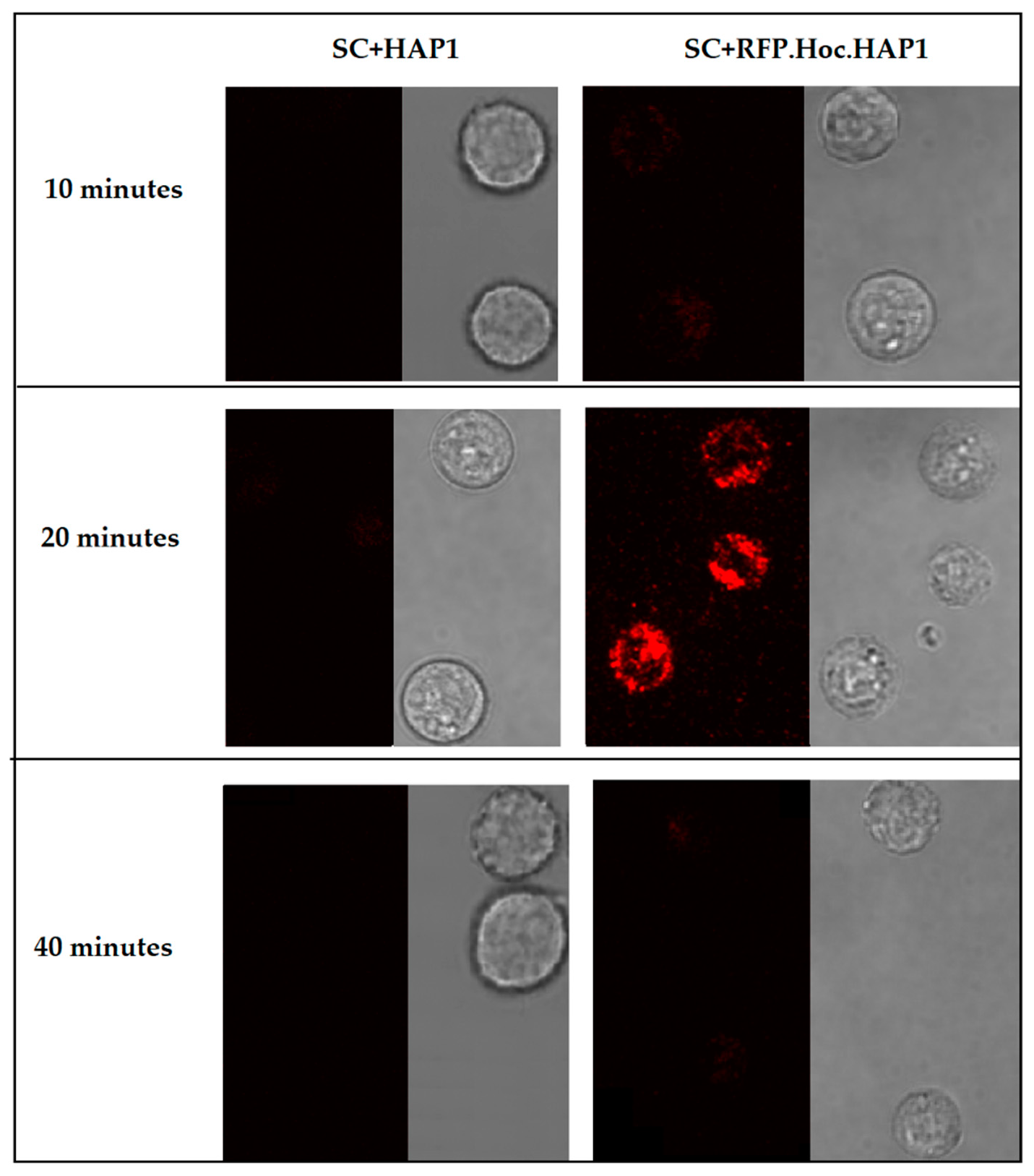
2.4. Phage Imaging in Living Cells
2.5. Ex Vivo Molecular Imaging
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Ethics Statements
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Schooley, R.T.; Biswas, B.; Gill, J.J.; Hernandez-Morales, A.; Lancaster, J.; Lessor, L.; Barr, J.J.; Reed, S.L.; Rohwer, F.; Benler, S.; et al. Development and Use of Personalized Bacteriophage-Based Therapeutic Cocktails To Treat a Patient with a Disseminated Resistant Acinetobacter baumannii Infection. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef]
- Huh, H.; Wong, S.; Jean, J.S.; Slavcev, R. Bacteriophage interactions with mammalian tissue: Therapeutic applications. Adv. Drug Deliv. Rev. 2019, 145, 4–17. [Google Scholar] [CrossRef]
- Jariah, R.O.; Hakim, M.S. Interaction of phages, bacteria, and the human immune system: Evolutionary changes in phage therapy. Rev. Med. Virol. 2019, 29, e2055. [Google Scholar] [CrossRef]
- Dąbrowska, K. Phage therapy: What factors shape phage pharmacokinetics and bioavailability? Systematic and critical review. Med. Res. Rev. 2019, 39, 2000–2025. [Google Scholar] [CrossRef]
- Frenkel, D.; Solomon, B. Filamentous phage as vector-mediated antibody delivery to the brain. Proc. Natl. Acad. Sci. USA 2002, 99, 5675–5679. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.W.; Green, P.; Parsell, Z. Vero Cell Toxins in Escherichia coli and Related Bacteria: Transfer by Phage and Conjugation and Toxic Action in Laboratory Animals, Chickens and Pigs. J. Gen. Microbiol. 1983, 129, 3121–3137. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.W.; Huggins, M.B.; Shaw, K.M. The Control of Experimental Escherichia coli Diarrhoea in Calves by Means of Bacteriophages. J. Gen. Microbiol. 1987, 133, 1111–1126. [Google Scholar] [CrossRef] [PubMed]
- Malik, D.J.; Sokolov, I.J.; Vinner, G.K.; Mancuso, F.; Cinquerrui, S.; Vladisavljevic, G.T.; Clokie, M.R.; Garton, N.J.; Stapley, A.G.; Kirpichnikova, A. Formulation, stabilisation and encapsulation of bacteriophage for phage therapy. Adv. Colloid Interface Sci. 2017, 249, 100–133. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, J.W.; Yang, L.; De Plano, L.M.; Stackhouse, M.A.; Petrenko, V.A. Evolution of a Landscape Phage Library in a Mouse Xenograft Model of Human Breast Cancer. Viruses 2019, 11, 988. [Google Scholar] [CrossRef]
- Pleiko, K.; Põšnograjeva, K.; Haugas, M.; Paiste, P.; Tobi, A.; Kurm, K.; Riekstina, U.; Teesalu, T. In vivo phage display: Identification of organ-specific peptides using deep sequencing and differential profiling across tissues. Nucleic Acids Res. 2021. [Google Scholar] [CrossRef]
- Bruttin, A.; Brüssow, H. Human Volunteers Receiving Escherichia coli Phage T4 Orally: A Safety Test of Phage Therapy. Antimicrob. Agents Chemother. 2005, 49, 2874–2878. [Google Scholar] [CrossRef] [PubMed]
- Monsur, K.A.; Rahman, M.A.; Huq, F.; Islam, M.N.; Northrup, R.S.; Hirschhorn, N. Effect of massive doses of bacteriophage on excretion of vibrios, duration of diarrhoea and output of stools in acute cases of cholera. Bull. World Health Organ. 1970, 42, 723–732. [Google Scholar] [PubMed]
- Newton, J.R.; Kelly, K.A.; Mahmood, U.; Weissleder, R.; Deutscher, S.L. In Vivo Selection of Phage for the Optical Imaging of PC-3 Human Prostate Carcinoma in Mice. Neoplasia 2006, 8, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Kaźmierczak, Z.; Piotrowicz, A.; Owczarek, B.; Hodyra, K.; Miernikiewicz, P.; Lecion, D.; Harhala, M.; Górski, A.; Dąbrowska, K. Molecular imaging of T4 phage in mammalian tissues and cells. Bacteriophage 2014, 4, e28364. [Google Scholar] [CrossRef] [PubMed]
- Swenson, E.S.; Price, J.G.; Brazelton, T.; Krause, D.S. Limitations of Green Fluorescent Protein as a Cell Lineage Marker. Stem Cells 2007, 25, 2593–2600. [Google Scholar] [CrossRef]
- Jiang, X.; Kalajzic, Z.; Maye, P.; Braut, A.; Bellizzi, J.; Mina, M.; Rowe, D.W. Histological Analysis of GFP Expression in Murine Bone. J. Histochem. Cytochem. 2005, 53, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Boratynski, J.; Syber, D.; Weber, B.D.; Marzanna, L.-S.; Pozniak, G.; Gorski, A. Preparation of endotoxin-free bacteriophages. Cell Mol. Biol. Lett. 2004, 9, 253–259. [Google Scholar] [PubMed]
- Oślizło, A.; Miernikiewicz, P.; Piotrowicz, A.; Owczarek, B.; Kopciuch, A.; Figura, G.; Dąbrowska, K. Purification of phage display-modified bacteriophage T4 by affinity chromatography. BMC Biotechnol. 2011, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Ceglarek, I.; Piotrowicz, A.; Lecion, D.; Miernikiewicz, P.; Owczarek, B.; Hodyra, K.; Harhala, M.; Górski, A.; Dabrowska, K. A novel approach for separating bacteriophages from other bacteriophages using affinity chromatography and phage display. Sci. Rep. 2013, 3, 3220. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowska, K.; Zembala, M.; Boratynski, J.; Switala-Jelen, K.; Wietrzyk, J.; Opolski, A.; Szczaurska, K.; Kujawa, M.; Godlewska, J.; Górski, A. Hoc protein regulates the biological effects of T4 phage in mammals. Arch. Microbiol. 2007, 187, 489–498. [Google Scholar] [CrossRef]
- Chang, R.Y.K.; Wallin, M.; Lin, Y.; Leung, S.S.Y.; Wang, H.; Morales, S.; Chan, H.-K. Phage therapy for respiratory infections. Adv. Drug Deliv. Rev. 2018, 133, 76–86. [Google Scholar] [CrossRef]
- Merril, C.R.; Biswas, B.; Carlton, R.; Jensen, N.C.; Creed, G.J.; Zullo, S.; Adhya, S. Long-circulating bacteriophage as antibacterial agents. Proc. Natl. Acad. Sci. USA 1996, 93, 3188–3192. [Google Scholar] [CrossRef]
- Vitiello, C.L.; Merril, C.R.; Adhya, S. An amino acid substitution in a capsid protein enhances phage survival in mouse circulatory system more than a 1000-fold. Virus Res. 2005, 114, 101–103. [Google Scholar] [CrossRef]
- Hodyra-Stefaniak, K.; Lahutta, K.; Majewska, J.; Kaźmierczak, Z.; Lecion, D.; Harhala, M.; Kęska, W.; Owczarek, B.; Jończyk-Matysiak, E.; Kłopot, A.; et al. Bacteriophages engineered to display foreign peptides may become short-circulating phages. Microb. Biotechnol. 2019, 12, 730–741. [Google Scholar] [CrossRef] [PubMed]
- Hodyra, K.; Dąbrowska, K. Molecular and Chemical Engineering of Bacteriophages for Potential Medical Applications. Arch. Immunol. Ther. Exp. 2015, 63, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Jończyk-Matysiak, E.; Weber-Dąbrowska, B.; Owczarek, B.; Międzybrodzki, R.; Łusiak-Szelachowska, M.; Łodej, N.; Górski, A. Phage-Phagocyte Interactions and Their Implications for Phage Application as Therapeutics. Viruses 2017, 9, 150. [Google Scholar] [CrossRef]
- Barfoot, R.; Denham, S.; Gyure, L.A.; Hall, J.G.; Hobbs, S.M.; Jackson, L.E.; Robertson, D. Some properties of dendritic macrophages from peripheral lymph. Immunology 1989, 68, 233–239. [Google Scholar] [PubMed]
- Geier, M.R.; Trigg, M.E.; Merril, C.R. Fate of Bacteriophage Lambda in Non-immune Germ-free Mice. Nat. Cell Biol. 1973, 246, 221–223. [Google Scholar] [CrossRef] [PubMed]
- Uhr, J.W.; Weissman, G. Intracellular Distribution and Degradation of Bacteriophage in Mammalian Tissues. J. Immunol. 1965, 94, 544–550. [Google Scholar]
- Inchley, C.J. The actvity of mouse Kupffer cells following intravenous injection of T4 bacteriophage. Clin. Exp. Immunol. 1969, 5, 173–187. [Google Scholar]
- Tiwari, B.R.; Kim, S.; Rahman, M.; Kim, J. Antibacterial efficacy of lytic Pseudomonas bacteriophage in normal and neutropenic mice models. J. Microbiol. 2011, 49, 994–999. [Google Scholar] [CrossRef]
- Chow, M.Y.T.; Chang, R.Y.K.; Li, M.; Wang, Y.; Lin, Y.; Morales, S.; McLachlan, A.J.; Kutter, E.; Li, J.; Chan, H.-K. Pharmacokinetics and Time-Kill Study of Inhaled Antipseudomonal Bacteriophage Therapy in Mice. Antimicrob. Agents Chemother. 2020, 65, e01470-20. [Google Scholar] [CrossRef] [PubMed]
- Sako, Y.; Yanagida, T. Single-molecule visualization in cell biology. Nat. Rev. Mol. Cell Biol. 2003, SS1–SS5. [Google Scholar]
- Rey-Barroso, L.; Roldán, M.; Burgos-Fernández, F.J.; Gassiot, S.; Llobet, A.R.; Isola, I.; Vilaseca, M. Spectroscopic Evaluation of Red Blood Cells of Thalassemia Patients with Confocal Microscopy: A Pilot Study. Sensors 2020, 20, 4039. [Google Scholar] [CrossRef] [PubMed]
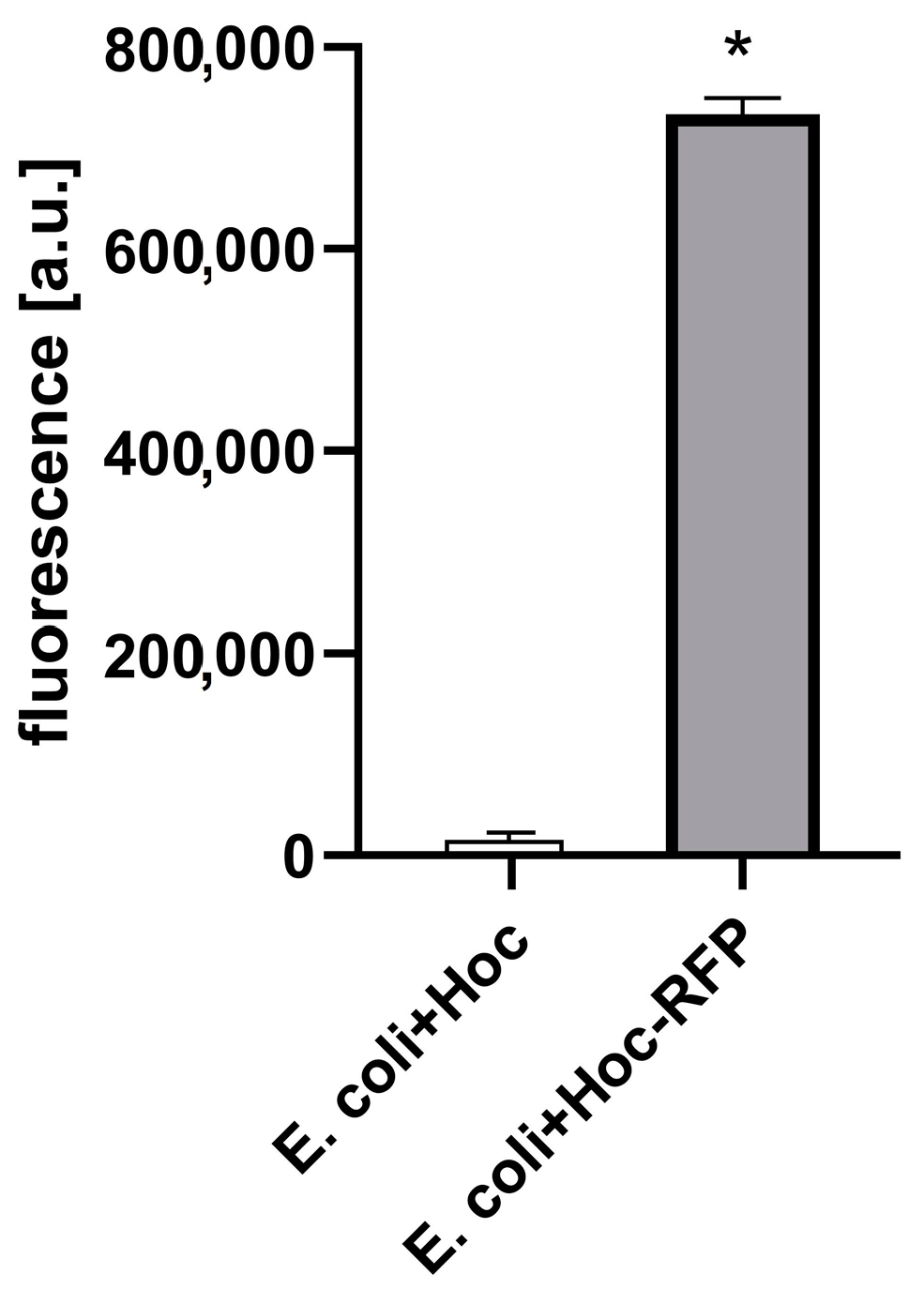
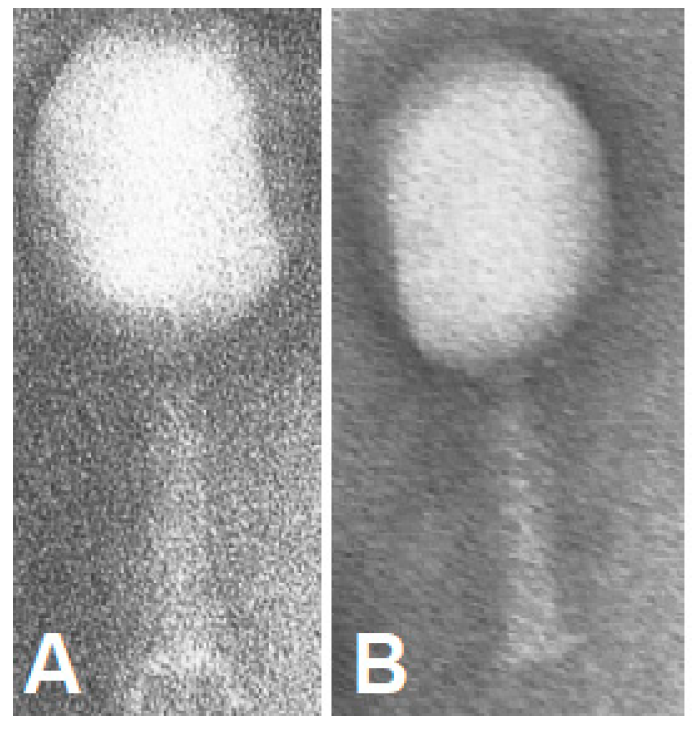
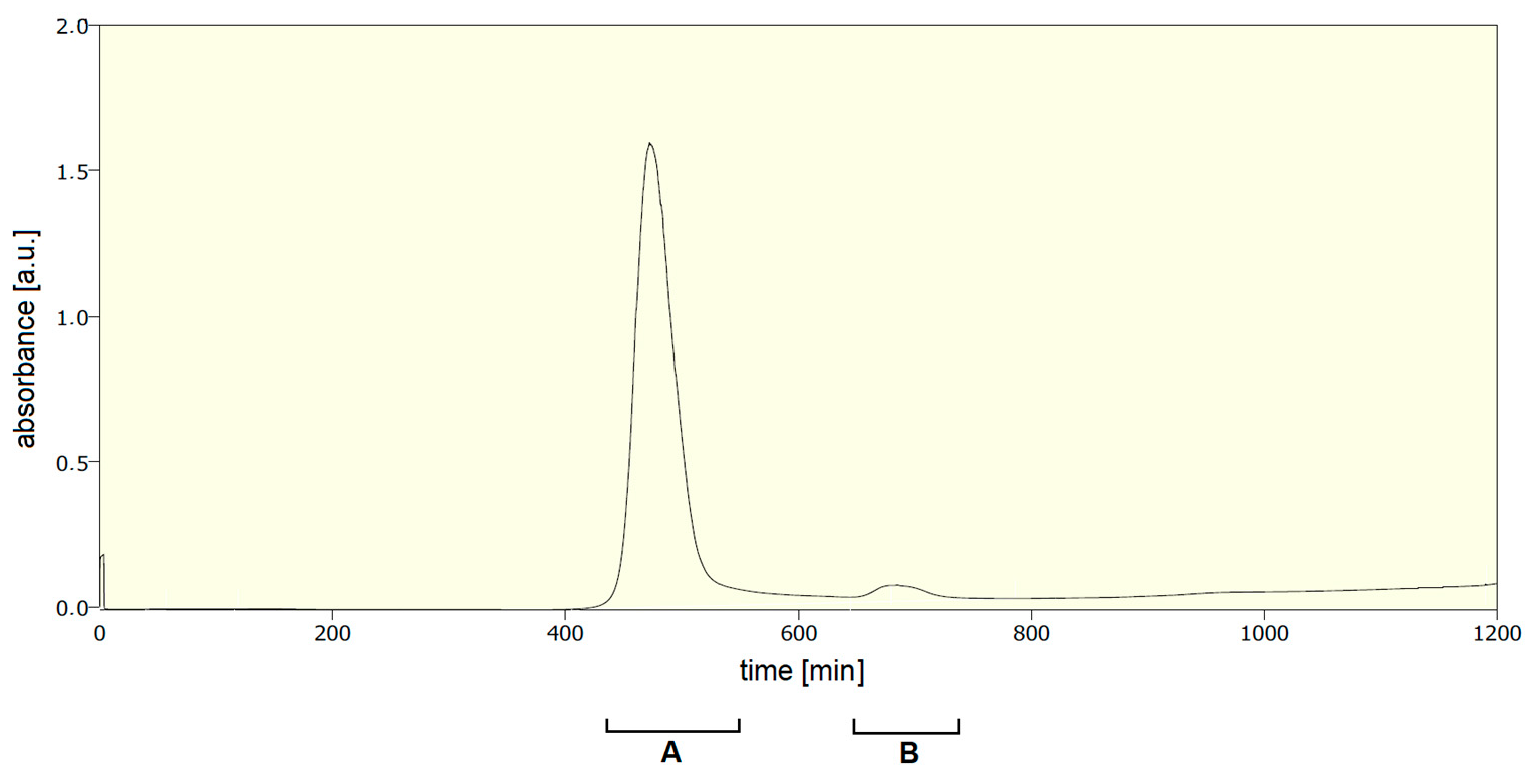




| Fraction | Titer | Fluorescence [a.u.] |
|---|---|---|
| control | 5 × 1011 PFU/mL | 1021 |
| A (labelled phages) | 5 × 1011 PFU/mL | 5,124,785 |
| B (non-incorporated proteins) | 2 × 102 PFU/mL | 8,274,121 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaźmierczak, Z.; Majewska, J.; Milczarek, M.; Owczarek, B.; Dąbrowska, K. Circulation of Fluorescently Labelled Phage in a Murine Model. Viruses 2021, 13, 297. https://doi.org/10.3390/v13020297
Kaźmierczak Z, Majewska J, Milczarek M, Owczarek B, Dąbrowska K. Circulation of Fluorescently Labelled Phage in a Murine Model. Viruses. 2021; 13(2):297. https://doi.org/10.3390/v13020297
Chicago/Turabian StyleKaźmierczak, Zuzanna, Joanna Majewska, Magdalena Milczarek, Barbara Owczarek, and Krystyna Dąbrowska. 2021. "Circulation of Fluorescently Labelled Phage in a Murine Model" Viruses 13, no. 2: 297. https://doi.org/10.3390/v13020297
APA StyleKaźmierczak, Z., Majewska, J., Milczarek, M., Owczarek, B., & Dąbrowska, K. (2021). Circulation of Fluorescently Labelled Phage in a Murine Model. Viruses, 13(2), 297. https://doi.org/10.3390/v13020297






