Detection of a Novel Reassortant H9N9 Avian Influenza Virus in Free-Range Ducks in Bangladesh
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling and Surveillance for AIVs
2.2. Virus Isolation from Samples
2.3. Subtype Detection and Sequencing
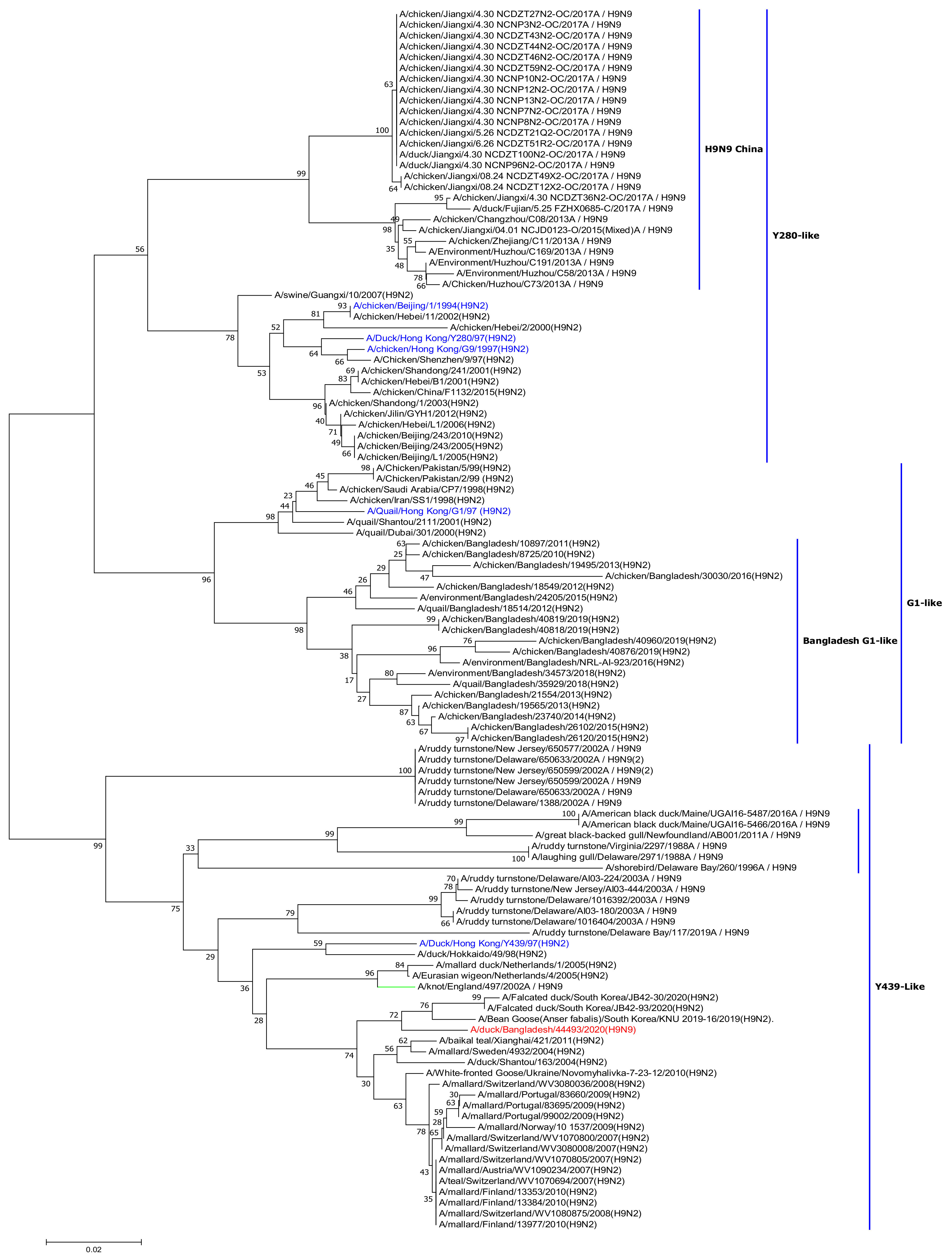
2.4. Phylogenetic Analysis
3. Results and Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Webster, R.G.; Bean, W.J.; Gorman, O.T.; Chambers, T.M.; Kawaoka, Y. Evolution and ecology of influenza A viruses. Microbiol. Rev. 1992, 56, 152–179. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Cao, B.; Hu, Y.; Feng, Z.; Wang, D.; Hu, W.; Chen, J.; Jie, Z.; Qiu, H.; Xu, K.; et al. Human infection with a novel avian-origin influenza A (H7N9) virus. N. Engl. J. Med. 2013, 368, 1888–1897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gilbert, M.; Chaitaweesub, P.; Parakamawongsa, T.; Premashthira, S.; Tiensin, T.; Kalpravidh, W.; Wagner, H.; Slingenbergh, J. Free-grazing ducks and highly pathogenic avian influenza, Thailand. Emerg. Infect. Dis. 2006, 12, 227–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, D.; Shi, W.; Shi, Y.; Wang, D.; Xiao, H.; Li, W.; Bi, Y.; Wu, Y.; Li, X.; Yan, J.; et al. Origin and diversity of novel avian influenza A H7N9 viruses causing human infection: Phylogenetic, structural, and coalescent analyses. Lancet 2013, 381, 1926–1932. [Google Scholar] [CrossRef]
- Barman, S.; Marinova-Petkova, A.; Hasan, M.K.; Akhtar, S.; El-Shesheny, R.; Turner, J.C.; Franks, J.; Walker, D.; Seiler, J.; Friedman, K.; et al. Role of domestic ducks in the emergence of a new genotype of highly pathogenic H5N1 avian influenza A viruses in Bangladesh. Emerg. Microbes Infect. 2017, 6, e72. [Google Scholar] [CrossRef] [Green Version]
- El-Shesheny, R.; Barman, S.; Feeroz, M.M.; Hasan, M.K.; Jones-Engel, L.; Franks, J.; Turner, J.; Seiler, P.; Walker, D.; Friedman, K.; et al. Genesis of Influenza A(H5N8) Viruses. Emerg. Infect. Dis. 2017, 23, 1368–1371. [Google Scholar] [CrossRef]
- Islam, M.R.; Haque, M.E.; Giasuddin, M.; Chowdhury, E.H.; Samad, M.A.; Parvin, R.; Nooruzzaman, M.; Rahman, M.M.; Monoura, P. New introduction of clade 2.3.2.1 avian influenza virus (H5N1) into Bangladesh. Transbound. Emerg. Dis. 2012, 59, 460–463. [Google Scholar] [CrossRef]
- Marinova-Petkova, A.; Feeroz, M.M.; Rabiul Alam, S.M.; Kamrul Hasan, M.; Akhtar, S.; Jones-Engel, L.; Walker, D.; McClenaghan, L.; Rubrum, A.; Franks, J.; et al. Multiple introductions of highly pathogenic avian influenza H5N1 viruses into Bangladesh. Emerg. Microbes Infect. 2014, 3, e11. [Google Scholar] [CrossRef]
- Barman, S.; Turner, J.C.M.; Hasan, M.K.; Akhtar, S.; El-Shesheny, R.; Franks, J.; Walker, D.; Seiler, P.; Friedman, K.; Kercher, L.; et al. Continuing evolution of highly pathogenic H5N1 viruses in Bangladeshi live poultry markets. Emerg. Microbes Infect. 2019, 8, 650–661. [Google Scholar] [CrossRef]
- El-Shesheny, R.; Franks, J.; Turner, J.; Seiler, P.; Walker, D.; Friedman, K.; Mukherjee, N.; Kercher, L.; Hasan, M.K.; Feeroz, M.M.; et al. Continued Evolution of H5Nx Avian Influenza Viruses in Bangladeshi Live Poultry Markets: Pathogenic Potential in Poultry and Mammalian Models. J. Virol. 2020, 94, e01141-20. [Google Scholar] [CrossRef]
- World Health Organization. Antigenic and Genetic Characteristics of Zoonotic Influenza A Viruses and Development of Candidate Vaccine Viruses for Pandemic Preparedness. 2021. Available online: https://cdn.who.int/media/docs/default-source/influenza/who-influenza-recommendations/vcm-southern-hemisphere-recommendation-2022/202110_zoonotic_vaccinevirusupdate.pdf?sfvrsn=8f87a5f1_11 (accessed on 12 November 2021).
- Peacock, T.H.P.; James, J.; Sealy, J.E.; Iqbal, M. A Global Perspective on H9N2 Avian Influenza Virus. Viruses 2019, 11, 620. [Google Scholar] [CrossRef] [Green Version]
- Shanmuganatham, K.; Feeroz, M.M.; Jones-Engel, L.; Smith, G.J.; Fourment, M.; Walker, D.; McClenaghan, L.; Alam, S.M.; Hasan, M.K.; Seiler, P.; et al. Antigenic and molecular characterization of avian influenza A(H9N2) viruses, Bangladesh. Emerg. Infect. Dis. 2013, 19, 1393. [Google Scholar] [CrossRef]
- Turner, J.C.; Feeroz, M.M.; Hasan, M.K.; Akhtar, S.; Walker, D.; Seiler, P.; Barman, S.; Franks, J.; Jones-Engel, L.; McKenzie, P.; et al. Insight into live bird markets of Bangladesh: An overview of the dynamics of transmission of H5N1 and H9N2 avian influenza viruses. Emerg. Microbes Infect. 2017, 6, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Negovetich, N.J.; Feeroz, M.M.; Jones-Engel, L.; Walker, D.; Alam, S.M.; Hasan, K.; Seiler, P.; Ferguson, A.; Friedman, K.; Barman, S.; et al. Live bird markets of Bangladesh: H9N2 viruses and the near absence of highly pathogenic H5N1 influenza. PLoS ONE 2011, 6, e19311. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Manual on Animal Influenza Diagnosis and Surveillance. 2nd ed. 2002. Available online: http://whqlibdoc.who.int/hq/2002/WHO_CDS_CSR_NCS_2002.5.pdf (accessed on 10 April 2021).
- World Organization for Animal Health OIE. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. Available online: https://www.oie.int/en/what-we-do/standards/codes-and-manuals/terrestrial-manual-online-access/ (accessed on 10 April 2021).
- Hall, T. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [Green Version]
- El-Shesheny, R.; Feeroz, M.M.; Krauss, S.; Vogel, P.; McKenzie, P.; Webby, R.J.; Webster, R.G. Replication and pathogenic potential of influenza A virus subtypes H3, H7, and H15 from free-range ducks in Bangladesh in mammals. Emerg. Microbes Infect. 2018, 7, 70. [Google Scholar] [CrossRef] [Green Version]
- Horimoto, T.; Kawaoka, Y. Reverse genetics provides direct evidence for a correlation of hemagglutinin cleavability and virulence of an avian influenza A virus. J. Virol. 1994, 68, 3120–3128. [Google Scholar] [CrossRef] [Green Version]
- Veits, J.; Weber, S.; Stech, O.; Breithaupt, A.; Graber, M.; Gohrbandt, S.; Bogs, J.; Hundt, J.; Teifke, J.P.; Mettenleiter, T.C.; et al. Avian influenza virus hemagglutinins H2, H4, H8, and H14 support a highly pathogenic phenotype. Proc. Natl. Acad. Sci. USA 2012, 109, 2579–2584. [Google Scholar] [CrossRef] [Green Version]
- Senne, D.A.; Panigrahy, B.; Kawaoka, Y.; Pearson, J.E.; Süss, J.; Lipkind, M.; Kida, H.; Webster, R.G. Survey of the hemagglutinin (HA) cleavage site sequence of H5 and H7 avian influenza viruses: Amino acid sequence at the HA cleavage site as a marker of pathogenicity potential. Avian Dis. 1996, 40, 425–437. [Google Scholar] [CrossRef]
- Wan, H.; Sorrell, E.M.; Song, H.; Hossain, M.J.; Ramirez-Nieto, G.; Monne, I.; Stevens, J.; Cattoli, G.; Capua, I.; Chen, L.M.; et al. Replication and transmission of H9N2 influenza viruses in ferrets: Evaluation of pandemic potential. PLoS ONE 2008, 3, e2923. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Shi, J.; Guo, J.; Deng, G.; Zhang, Q.; Wang, J.; He, X.; Wang, K.; Chen, J.; Li, Y.; et al. Genetics, receptor binding property, and transmissibility in mammals of naturally isolated H9N2 Avian Influenza viruses. PLoS Pathog. 2014, 10, e1004508. [Google Scholar] [CrossRef]
- Katz, J.M.; Lu, X.; Tumpey, T.M.; Smith, C.B.; Shaw, M.W.; Subbarao, K. Molecular correlates of influenza A H5N1 virus pathogenesis in mice. J. Virol. 2000, 74, 10807–10810. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Ishaq, M.; Prudence, M.; Xi, X.; Hu, T.; Liu, Q.; Guo, D. Single mutation at the amino acid position 627 of PB2 that leads to increased virulence of an H5N1 avian influenza virus during adaptation in mice can be compensated by multiple mutations at other sites of PB2. Virus Res. 2009, 144, 123–129. [Google Scholar] [CrossRef]
- Yamaji, R.; Yamada, S.; Le, M.Q.; Li, C.; Chen, H.; Qurnianingsih, E.; Nidom, C.A.; Ito, M.; Sakai-Tagawa, Y.; Kawaoka, Y. Identification of PB2 mutations responsible for the efficient replication of H5N1 influenza viruses in human lung epithelial cells. J. Virol. 2015, 89, 3947–3956. [Google Scholar] [CrossRef] [Green Version]
- Hulse-Post, D.J.; Franks, J.; Boyd, K.; Salomon, R.; Hoffmann, E.; Yen, H.L.; Webby, R.J.; Walker, D.; Nguyen, T.D.; Webster, R.G. Molecular changes in the polymerase genes (PA and PB1) associated with high pathogenicity of H5N1 influenza virus in mallard ducks. J. Virol. 2007, 81, 8515–8524. [Google Scholar] [CrossRef] [Green Version]
- Schmolke, M.; Manicassamy, B.; Pena, L.; Sutton, T.; Hai, R.; Varga, Z.T.; Hale, B.G.; Steel, J.; Perez, D.R.; Garcia-Sastre, A. Differential contribution of PB1-F2 to the virulence of highly pathogenic H5N1 influenza A virus in mammalian and avian species. PLoS Pathog. 2011, 7, e1002186. [Google Scholar] [CrossRef] [Green Version]
- Subbarao, E.K.; London, W.; Murphy, B.R. A single amino acid in the PB2 gene of influenza A virus is a determinant of host range. J. Virol. 1993, 67, 1761–1764. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Zhang, Y.; Shinya, K.; Deng, G.; Jiang, Y.; Li, Z.; Guan, Y.; Tian, G.; Li, Y.; Shi, J.; et al. Identification of amino acids in HA and PB2 critical for the transmission of H5N1 avian influenza viruses in a mammalian host. PLoS Pathog. 2009, 5, e1000709. [Google Scholar] [CrossRef] [Green Version]
- Xiao, C.; Ma, W.; Sun, N.; Huang, L.; Li, Y.; Zeng, Z.; Wen, Y.; Zhang, Z.; Li, H.; Li, Q.; et al. PB2-588 V promotes the mammalian adaptation of H10N8, H7N9 and H9N2 avian influenza viruses. Sci. Rep. 2016, 6, 19474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conenello, G.M.; Zamarin, D.; Perrone, L.A.; Tumpey, T.; Palese, P. A single mutation in the PB1-F2 of H5N1 (HK/97) and 1918 influenza A viruses contributes to increased virulence. PLoS Pathog. 2007, 3, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
- Yamayoshi, S.; Yamada, S.; Fukuyama, S.; Murakami, S.; Zhao, D.; Uraki, R.; Watanabe, T.; Tomita, Y.; Macken, C.; Neumann, G.; et al. Virulence-affecting amino acid changes in the PA protein of H7N9 influenza A viruses. J. Virol. 2014, 88, 3127–3134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maines, T.R.; Chen, L.M.; Van Hoeven, N.; Tumpey, T.M.; Blixt, O.; Belser, J.A.; Gustin, K.M.; Pearce, M.B.; Pappas, C.; Stevens, J.; et al. Effect of receptor binding domain mutations on receptor binding and transmissibility of avian influenza H5N1 viruses. Virology 2011, 413, 139–147. [Google Scholar] [CrossRef]
- Chutinimitkul, S.; van Riel, D.; Munster, V.J.; van den Brand, J.M.; Rimmelzwaan, G.F.; Kuiken, T.; Osterhaus, A.D.; Fouchier, R.A.; de Wit, E. In Vitro assessment of attachment pattern and replication efficiency of H5N1 influenza A viruses with altered receptor specificity. J. Virol. 2010, 84, 6825–6833. [Google Scholar] [CrossRef] [Green Version]
- Wan, H.; Perez, D.R. Amino acid 226 in the hemagglutinin of H9N2 influenza viruses determines cell tropism and replication in human airway epithelial cells. J. Virol. 2007, 81, 5181–5191. [Google Scholar] [CrossRef] [Green Version]
- Auewarakul, P.; Suptawiwat, O.; Kongchanagul, A.; Sangma, C.; Suzuki, Y.; Ungchusak, K.; Louisirirotchanakul, S.; Lerdsamran, H.; Pooruk, P.; Thitithanyanont, A.; et al. An avian influenza H5N1 virus that binds to a human-type receptor. J. Virol. 2007, 81, 9950–9955. [Google Scholar] [CrossRef] [Green Version]
- Ilyushina, N.A.; Seiler, J.P.; Rehg, J.E.; Webster, R.G.; Govorkova, E.A. Effect of neuraminidase inhibitor-resistant mutations on pathogenicity of clade 2.2 A/Turkey/15/06 (H5N1) influenza virus in ferrets. PLoS Pathog. 2010, 6, e1000933. [Google Scholar] [CrossRef]
- Sleeman, K.; Guo, Z.; Barnes, J.; Shaw, M.; Stevens, J.; Gubareva, L.V. R292K substitution and drug susceptibility of influenza A(H7N9) viruses. Emerg. Infect. Dis. 2013, 19, 1521–1524. [Google Scholar] [CrossRef] [Green Version]
- Abed, Y.; Goyette, N.; Boivin, G. Generation and characterization of recombinant influenza A (H1N1) viruses harboring amantadine resistance mutations. Antimicrob. Agents Chemother. 2005, 49, 556–559. [Google Scholar] [CrossRef] [Green Version]
- Bean, W.J.; Threlkeld, S.C.; Webster, R.G. Biologic potential of amantadine-resistant influenza A virus in an avian model. J. Infect. Dis. 1989, 159, 1050–1056. [Google Scholar] [CrossRef]
- Cheung, C.L.; Rayner, J.M.; Smith, G.J.; Wang, P.; Naipospos, T.S.; Zhang, J.; Yuen, K.Y.; Webster, R.G.; Peiris, J.S.; Guan, Y.; et al. Distribution of amantadine-resistant H5N1 avian influenza variants in Asia. J. Infect. Dis. 2006, 193, 1626–1629. [Google Scholar] [CrossRef] [Green Version]
- Jiao, P.; Tian, G.; Li, Y.; Deng, G.; Jiang, Y.; Liu, C.; Liu, W.; Bu, Z.; Kawaoka, Y.; Chen, H. A single-amino-acid substitution in the NS1 protein changes the pathogenicity of H5N1 avian influenza viruses in mice. J. Virol. 2008, 82, 1146–1154. [Google Scholar] [CrossRef] [Green Version]
- Seo, S.H.; Hoffmann, E.; Webster, R.G. Lethal H5N1 influenza viruses escape host anti-viral cytokine responses. Nat. Med. 2002, 8, 950–954. [Google Scholar] [CrossRef]
- Li, Z.; Jiang, Y.; Jiao, P.; Wang, A.; Zhao, F.; Tian, G.; Wang, X.; Yu, K.; Bu, Z.; Chen, H. The NS1 gene contributes to the virulence of H5N1 avian influenza viruses. J. Virol. 2006, 80, 11115–11123. [Google Scholar] [CrossRef] [Green Version]
- Stallknecht, D.E.; Luttrell, M.P.; Poulson, R.; Goekjian, V.; Niles, L.; Dey, A.; Krauss, S.; Webster, R.G. Detection of avian influenza viruses from shorebirds: Evaluation of surveillance and testing approaches. J. Wildl. Dis. 2012, 48, 382–393. [Google Scholar] [CrossRef] [Green Version]
- Han, J.; Wang, L.; Liu, J.; Jin, M.; Hao, F.; Zhang, P.; Zhang, Z.; Wen, D.; Wu, X.; Liu, G.; et al. Cocirculation of three hemagglutinin and two neuraminidase subtypes of avian influenza viruses in Huzhou, China, April 2013: Implication for the origin of the novel H7N9 virus. J. Virol. 2014, 88, 6506–6511. [Google Scholar] [CrossRef] [Green Version]
- Parvin, R.; Nooruzzaman, M.; Kabiraj, C.K.; Begum, J.A.; Chowdhury, E.H.; Islam, M.R.; Harder, T. Controlling Avian Influenza Virus in Bangladesh: Challenges and Recommendations. Viruses 2020, 12, 751. [Google Scholar] [CrossRef]


| Gene * | Accession No. | Virus | % Identity |
|---|---|---|---|
| PB2 | MT020147.1 | A/duck/Mongolia/926/2019(H5N3) | 99.1 |
| MW749040.1 † | A/duck/Bangladesh/44478/2020(H10N9) | 100 | |
| PB1 | MN208036.1 | A/northern shoveler/Egypt/MB-D-690C/2016(H7N3) | 99.63 |
| MW749040.1 | A/duck/Bangladesh/44478/2020(H10N9) | 99.96 | |
| PA | MW188628.1 | A/duck/Mongolia/447/2018(H4N6) | 99.1 |
| MW749040.1 | A/duck/Bangladesh/44478/2020(H10N9) | 100 | |
| HA | EPI_ISL_418175 | A/Bean Goose(Anser fabalis)/South Korea/KNU 2019-16/2019(H9N2) | 97.62 |
| NP | MN208011.1 | A/teal/Egypt/MB-D-487OP/2016(H7N3) | 98.72 |
| MW749040.1 | A/duck/Bangladesh/44478/2020(H10N9) | 100 | |
| NA | MW116667.1 | A/Anas platyrhynchos/South Korea/JB31-96/2019(H11N9) | 98.47 |
| M | MW188600.1 | A/duck/Mongolia/345/2018(H4N6) | 99.21 |
| MW749040.1 | A/duck/Bangladesh/44478/2020(H10N9) | 99.80 | |
| NS | MT020282.1 | A/duck/Mongolia/961/2019(H3N8) | 99.77 |
| MW466161.1 | A/environment/Bangladesh/42635/2020(H10N7) | 100 |
| Viral Protein | Amino Acid | A/duck/Bangladesh/44493/2020 | Functional Relevance | References |
|---|---|---|---|---|
| PB2 | E627K | E | Mammalian host adaptation | [32] |
| D701N | D | Increase polymerase activity and viral replication in mammalian cells | [33] | |
| L89V | V | Enhanced polymerase activity, increased virulence in mice | [28] | |
| G309D | D | Enhanced polymerase activity, increased virulence in mice | [28] | |
| T339K | K | Enhanced polymerase activity, increased virulence in mice | [28] | |
| A588V | A | Mammalian host adaptation | [34] | |
| PB1-F2 | N66S | S | Increases virulence, replication efficiency, and the antiviral response in mammals | [31,35] |
| PA | V100A | V | Contributed to the virulence and mammalian adaptation | |
| S409N | S | Contributed to the virulence and mammalian adaptation | [36] | |
| A515T | T | Increased polymerase activity, increased virulence in mammals and birds | [30] | |
| HA | E198D | E | Enhanced mammalian receptor binding | [37] |
| Q234L | Q | Preferential binding to human Sialic acid α2–6 receptor | [38,39] | |
| G236S | G | Preferential binding to human Sialic acid α2–6 receptor | [38] | |
| I155T | T | Enhanced mammalian receptor binding | [40] | |
| NA | E119V | E | Oseltamivir resistance | [41] |
| H275Y | H | Oseltamivir resistance | [41] | |
| R293K | R | Oseltamivir resistance | [42] | |
| N295S | N | Oseltamivir resistance | [41] | |
| M2 | L26P | L | Reduced susceptibility to amantadine | [43,44] |
| V27A/I | V | Reduced susceptibility to amantadine | [43,44] | |
| A30T | A | Reduced susceptibility to amantadine | [44,45] | |
| S31N | S | Reduced susceptibility to amantadine | [44,45] | |
| G34E | G | Reduced susceptibility to amantadine | [43] | |
| NS1 | P42S | S | Increased virulence and pathogenicity in mammals | [46] |
| D92E | D | Increased virulence and pathogenicity in mammals | [47] | |
| V149A | A | Increased virulence and pathogenicity in mammals | [48] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Shesheny, R.; Turner, J.C.M.; Walker, D.; Franks, J.; Seiler, P.; Barman, S.; Feeroz, M.M.; Hasan, M.K.; Akhtar, S.; Mukherjee, N.; et al. Detection of a Novel Reassortant H9N9 Avian Influenza Virus in Free-Range Ducks in Bangladesh. Viruses 2021, 13, 2357. https://doi.org/10.3390/v13122357
El-Shesheny R, Turner JCM, Walker D, Franks J, Seiler P, Barman S, Feeroz MM, Hasan MK, Akhtar S, Mukherjee N, et al. Detection of a Novel Reassortant H9N9 Avian Influenza Virus in Free-Range Ducks in Bangladesh. Viruses. 2021; 13(12):2357. https://doi.org/10.3390/v13122357
Chicago/Turabian StyleEl-Shesheny, Rabeh, Jasmine C. M. Turner, David Walker, John Franks, Patrick Seiler, Subrata Barman, Mohammed M. Feeroz, Md Kamrul Hasan, Sharmin Akhtar, Nabanita Mukherjee, and et al. 2021. "Detection of a Novel Reassortant H9N9 Avian Influenza Virus in Free-Range Ducks in Bangladesh" Viruses 13, no. 12: 2357. https://doi.org/10.3390/v13122357
APA StyleEl-Shesheny, R., Turner, J. C. M., Walker, D., Franks, J., Seiler, P., Barman, S., Feeroz, M. M., Hasan, M. K., Akhtar, S., Mukherjee, N., Kercher, L., McKenzie, P., Webster, R. G., & Webby, R. J. (2021). Detection of a Novel Reassortant H9N9 Avian Influenza Virus in Free-Range Ducks in Bangladesh. Viruses, 13(12), 2357. https://doi.org/10.3390/v13122357







