Ocular Histopathological Findings in Semi-Domesticated Eurasian Tundra Reindeer (Rangifer tarandus tarandus) with Infectious Keratoconjunctivitis after Experimental Inoculation with Cervid Herpesvirus 2
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Collection and Processing of Eye Tissues for Histological Examination
2.3. Quantitative Real-Time PCR (qPCR)
3. Results
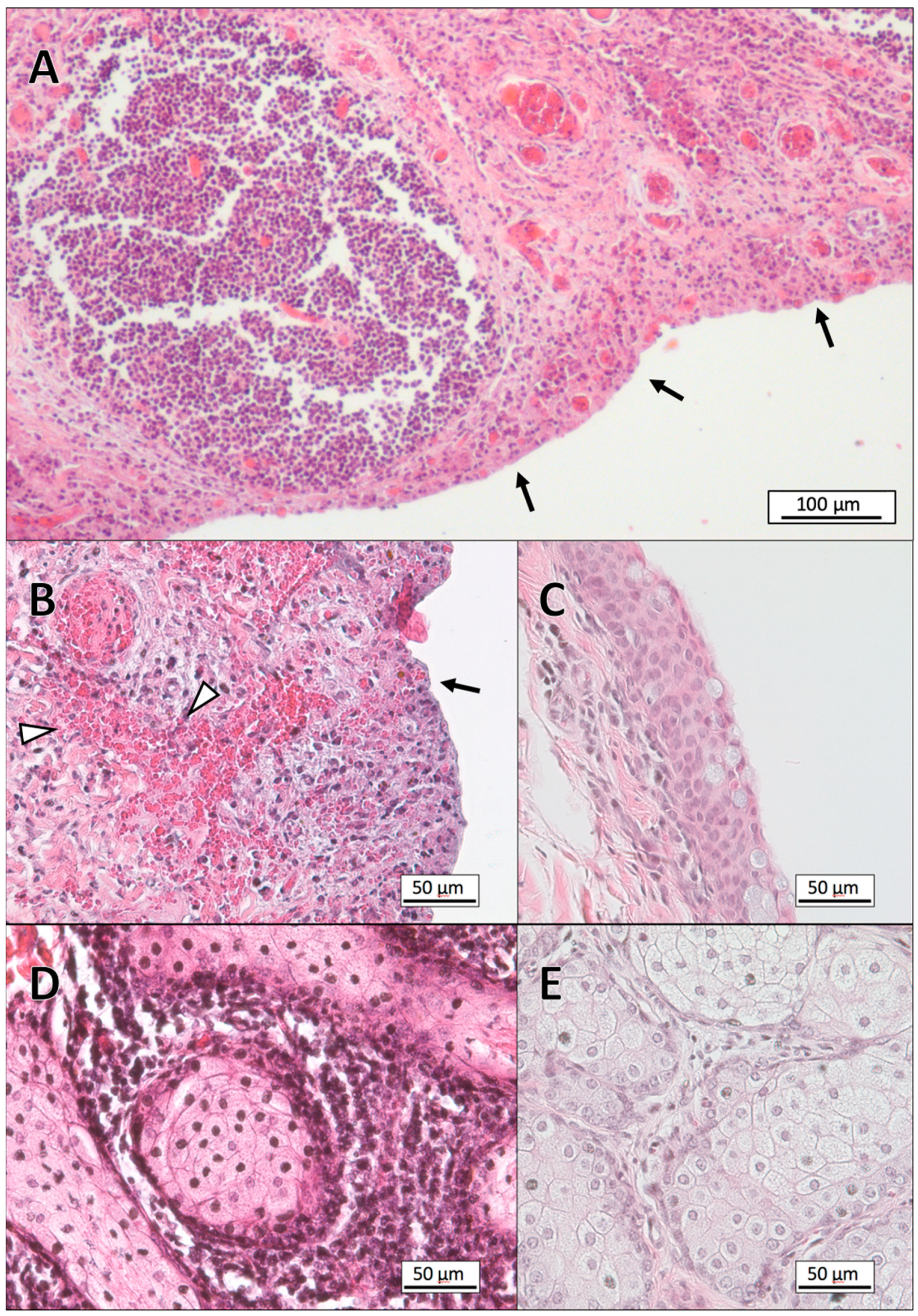
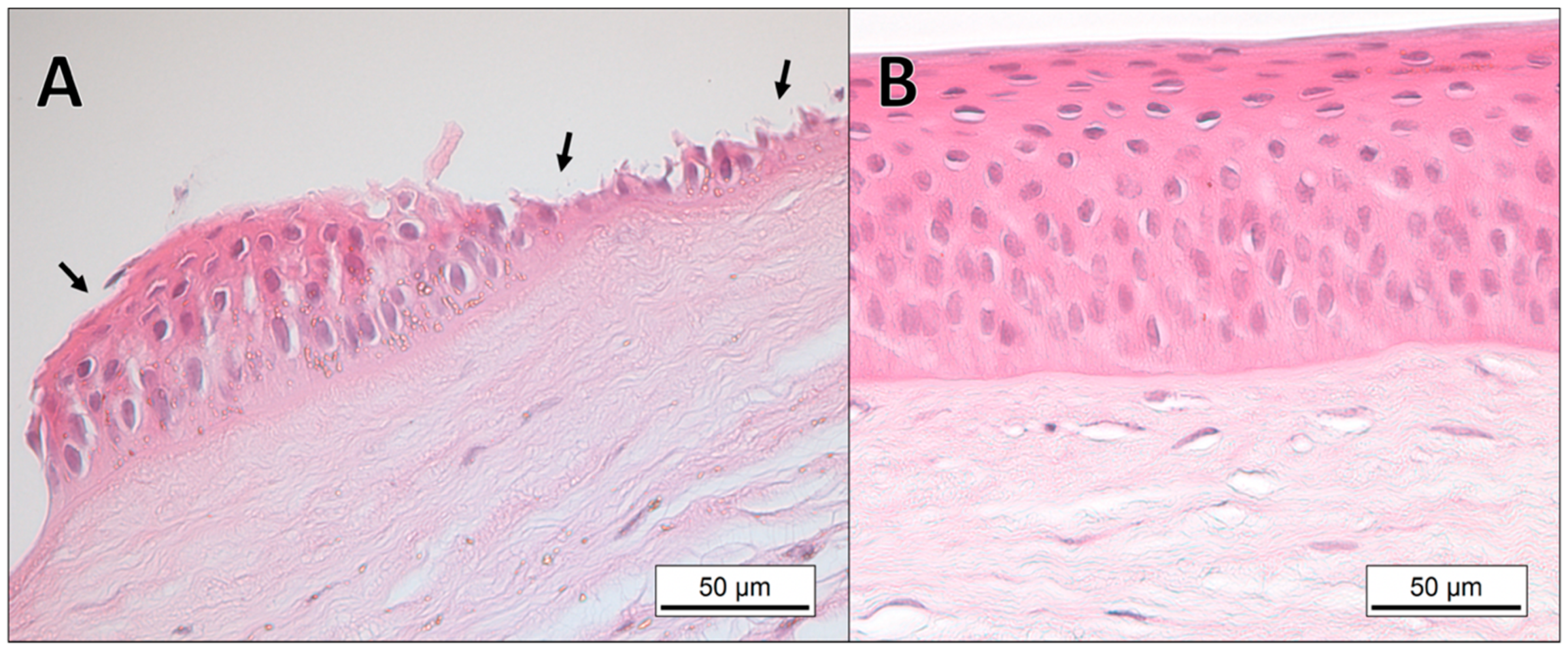
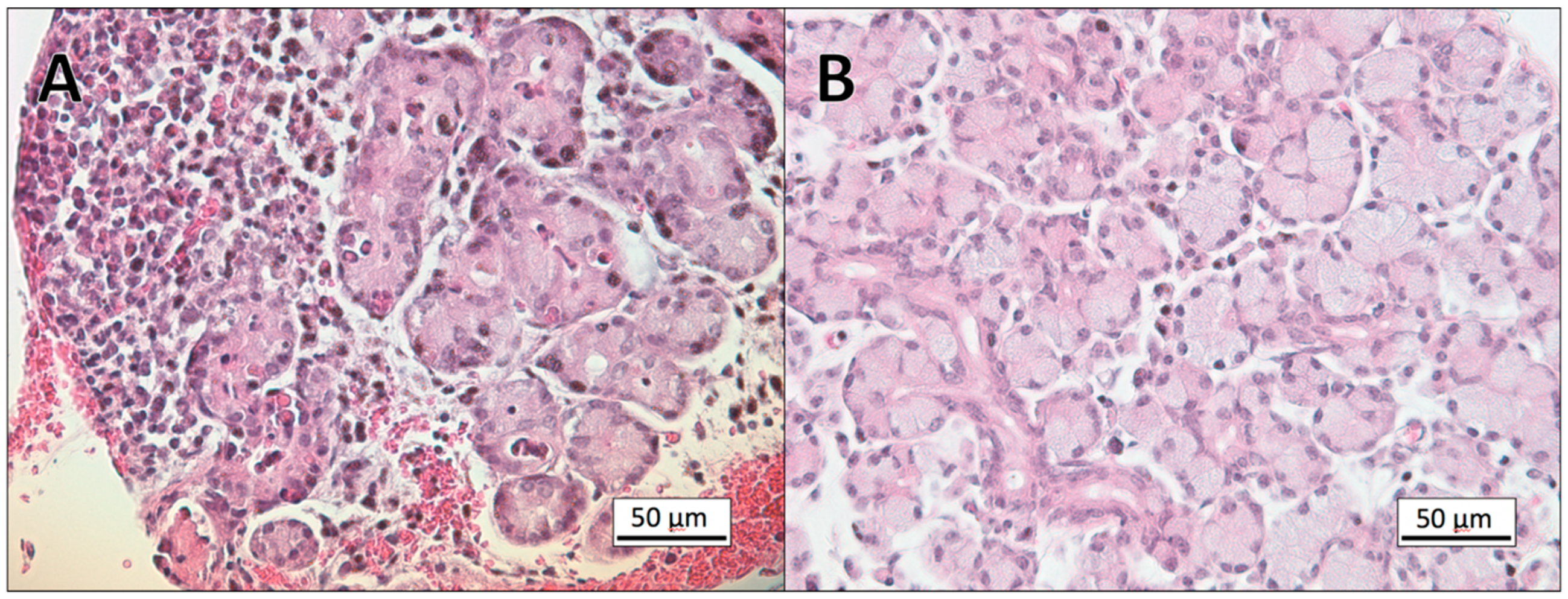
3.1. Histopathology
3.2. Gram-Staining of Tissue Sections
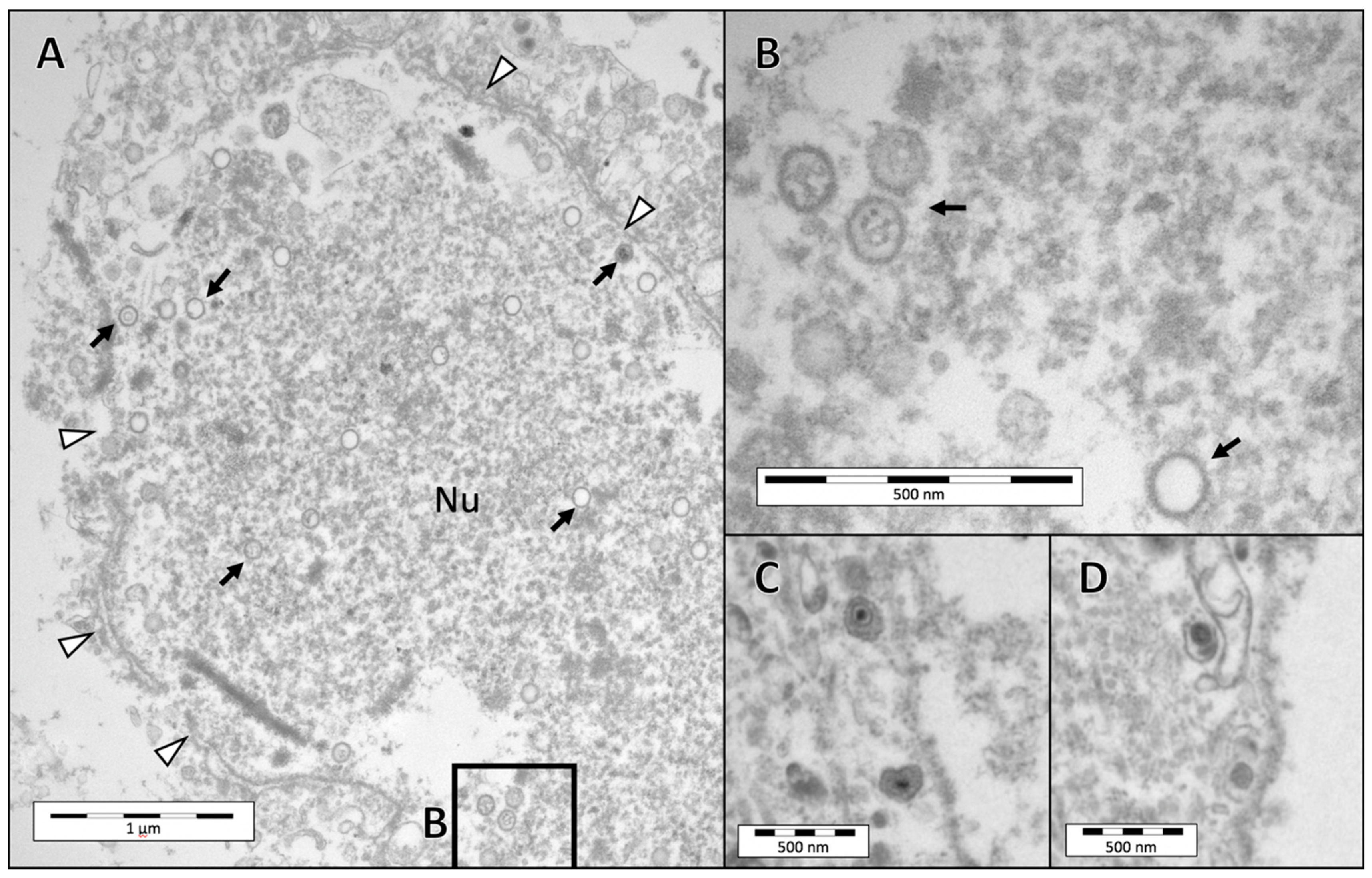
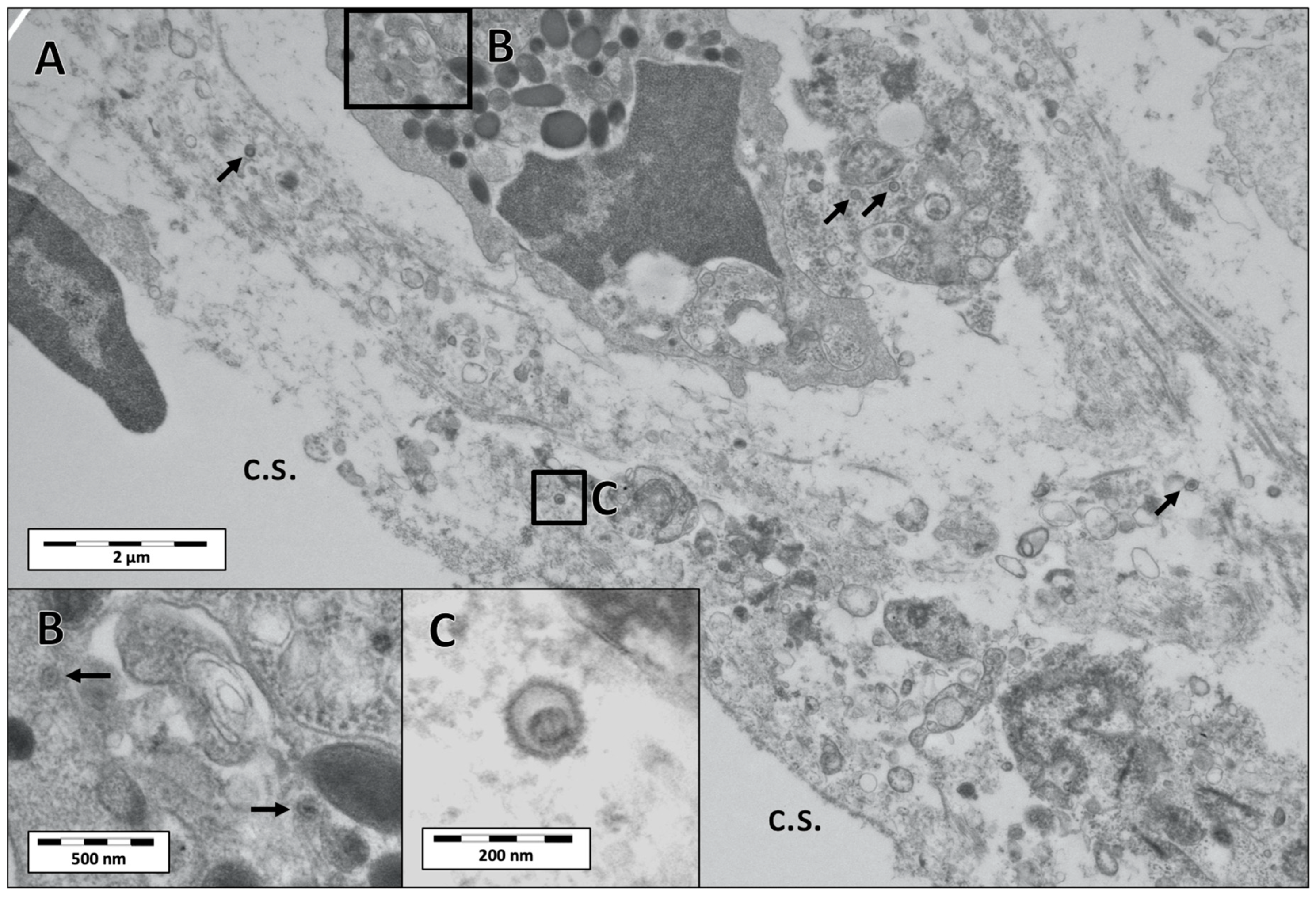
3.3. Electron Microscopy
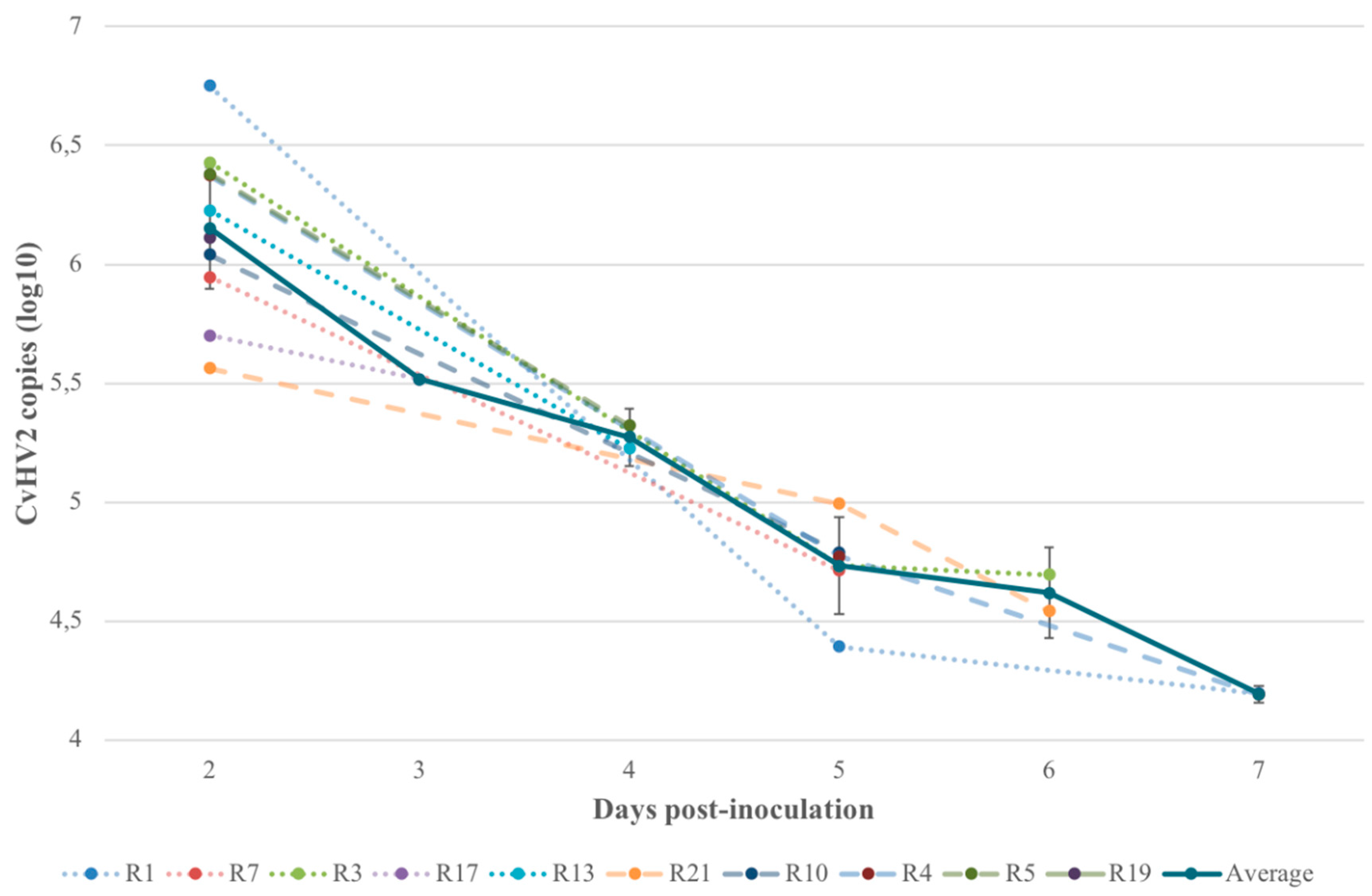
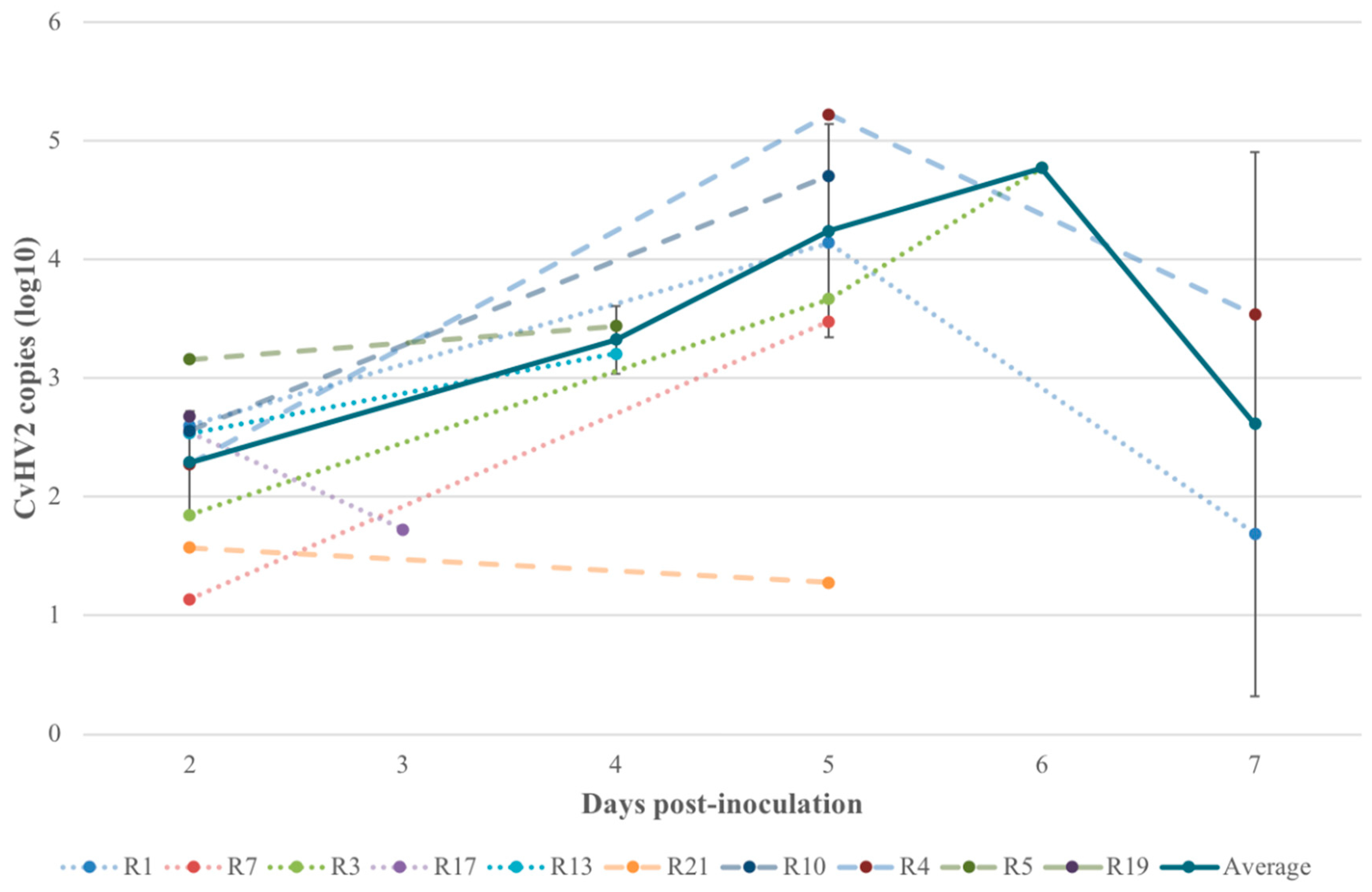
3.4. Quantitative Real-Time PCR (qPCR)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Angelos, J.A. Moraxella bovoculi and Infectious Bovine Keratoconjunctivitis: Cause or Coincidence? Vet. Clin. N. Am. Food Anim. Pr. 2010, 26, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Belloy, L.; Janovsky, M.; Vilei, E.M.; Pilo, P.; Giacometti, M.; Frey, J. Molecular Epidemiology of Mycoplasma conjunctivae in Caprinae: Transmission across Species in Natural Outbreaks. Appl. Environ. Microbiol. 2003, 69, 1913–1919. [Google Scholar] [CrossRef] [PubMed]
- Egwu, G.O.; Faull, W.B.; Bradbury, J.M.; Clarkson, M.J. Ovine infectious keratoconjunctivitis: A microbiological study of clinically unaffected and affected sheep’s eyes with special reference to Mycoplasma conjunctivae. Vet. Rec. 1989, 125, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Galvão, K.N.; Angelos, J.A. Ulcerative blepharitis and conjunctivitis in adult dairy cows and association with Moraxella bovoculi. Can. Vet. J. 2010, 51, 400–402. [Google Scholar]
- Gortázar Zaragoza, C.; Fernández-de-Luco, D.; Frölich, K. Keratoconjunctivitis in a free-ranging red deer (Cervus elaphus) population in Spain. Z. Jagdwiss. 1998, 44, 257–261. [Google Scholar] [CrossRef]
- Gupta, S.; Chahota, R.; Bhardwaj, B.; Priyanka, P.; Verma, S.; Sharma, M. Identification of Chlamydiae and Mycoplasma species in ruminants with ocular infections. Lett. Appl. Microbiol. 2015, 60, 135–139. [Google Scholar] [CrossRef]
- Marco, I.; Mentaberre, G.; Ballesteros, C.; Bischof, D.F.; Lavín, S.; Vilei, E.M. First Report of Mycoplasma conjunctivae from Wild Caprinae with Infectious Keratoconjunctivitis in the Pyrenees (NE Spain). J. Wildl. Dis. 2009, 45, 238–241. [Google Scholar] [CrossRef]
- Mavrot, F.; Vilei, E.M.; Marreros, N.; Signer, C.; Frey, J.; Ryser-Degiorgis, M.-P. Occurrence, quantification, and genotyping of Mycoplasma conjunctivae in wild Caprinae with and without infectious keratoconjunctivitis. J. Wildl. Dis. 2012, 48, 619–631. [Google Scholar] [CrossRef]
- Meagher, M.; Quinn, W.J.; Stackhouse, L. Chlamydial-Caused infectious keratoconjunctivitis in Bighorn sheep of Yellowstone National Park. J. Wildl. Dis. 1992, 28, 171–176. [Google Scholar] [CrossRef]
- Åkerstedt, J.; Hofshagen, M. Bacteriological Investigation of Infectious Keratoconjunctivitis in Norwegian Sheep. Acta Vet. Scand. 2004, 45, 19–26. [Google Scholar] [CrossRef]
- Bergman, A. Contagious keratitis in reindeer. Skand. Vet. 1912, 2, 145–177. [Google Scholar]
- Tryland, M.; Stubsjøen, S.M.; Agren, E.; Johansen, B.; Kielland, C. Herding conditions related to infectious keratoconjunctivitis in semi-domesticated reindeer: A questionnaire-based survey among reindeer herders. Acta Vet. Scand. 2016, 58, 22. [Google Scholar] [CrossRef] [PubMed]
- Rehbinder, C.; Nilsson, A. An outbreak of kerato-conjunctivitis among corralled, supplementary fed, semi-domestic reindeer calves. Rangifer 1995, 15, 9–14. [Google Scholar] [CrossRef][Green Version]
- Tryland, M.; Das Neves, C.G.; Sunde, M.; Mørk, T. Cervid Herpesvirus 2, the Primary Agent in an Outbreak of Infectious Keratoconjunctivitis in Semidomesticated Reindeer. J. Clin. Microbiol. 2009, 47, 3707–3713. [Google Scholar] [CrossRef] [PubMed]
- Sánchez Romano, J.; Mørk, T.; Laaksonen, S.; Ågren, E.; Nymo, I.H.; Sunde, M.; Tryland, M. Infectious keratoconjunctivitis in semi-domesticated Eurasian tundra reindeer (Rangifer tarandus tarandus): Microbiological study of clinically affected and unaffected animals with special reference to cervid herpesvirus 2. BMC Vet. Res. 2018, 14, 15. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.J.; Eberle, R.; Ehlers, B.; Hayward, G.S.; McGeoch, D.J.; Minson, A.C.; Pellett, P.E.; Roizman, B.; Studdert, M.J.; Thiry, E. The order Herpesvirales. Arch. Virol. 2009, 154, 171–177. [Google Scholar] [CrossRef]
- Das Neves, C.G.; Roger, M.; Yoccoz, N.G.; Rimstad, E.; Tryland, M. Evaluation of three commercial bovine ELISA kits for detection of antibodies against Alphaherpesviruses in reindeer (Rangifer tarandus tarandus). Acta Vet. Scand. 2009, 51, 9. [Google Scholar] [CrossRef]
- Ek-Kommonen, C.; Veijalainen, P.; Rantala, M.; Neuvonen, E. Neutralizing antibodies to bovine herpesvirus 1 in reindeer. Acta Vet. Scand. 1982, 23, 565. [Google Scholar]
- Kautto, A.H.; Alenius, S.; Mossing, T.; Becher, P.; Belák, S.; Larska, M. Pestivirus and alphaherpesvirus infections in Swedish reindeer (Rangifer tarandus tarandus L.). Vet. Microbiol. 2012, 156, 64–71. [Google Scholar] [CrossRef]
- Tryland, M.; Sánchez Romano, J.; Marcin, N.; Nymo, I.H.; Josefsen, T.D.; Sørensen, K.K.; Mørk, T. Cervid herpesvirus 2 and not Moraxella bovoculi caused keratoconjunctivitis in experimentally inoculated semi-domesticated Eurasian tundra reindeer. Acta Vet. Scand. 2017, 59, 23. [Google Scholar] [CrossRef]
- Winqvist, G.; Rehbinder, C. Fine structure of the reindeer cornea in normal conditions and in keratitis. Acta Vet. Scand. 1973, 14, 292–300. [Google Scholar] [PubMed]
- Rehbinder, C. Clinical and epizootiological studies on keratitis in reindeer. Acta Vet. Scand. 1977, 66, 1–27. [Google Scholar]
- Rockborn, G.; Rehbinder, C.; Klingeborn, B.; Leffler, M.; Klintevall, K.; Nikkilä, T.; Landén, A.; Nordkvist, M. The demonstration of a herpesvirus, related to bovine herpesvirus 1, in reindeer with ulcerative and necrotizing lesions of the upper alimentary tract and nose. Rangifer 1990, 10, 373–384. [Google Scholar] [CrossRef][Green Version]
- Ek-Kommonen, C.; Pelkonen, S.; Nettleton, P.F. Isolation of a herpesvirus serologically related to bovine herpesvirus 1 from a reindeer (Rangifer tarandus). Acta Vet. Scand. 1986, 27, 299–301. [Google Scholar] [PubMed]
- McDowell, E.M.; Trump, B.F. Histologic fixatives suitable for light and electron microscopy. Arch. Pathol. Lab. Med. 1976, 100, 405–414. [Google Scholar] [PubMed]
- Becerra, S.C.; Roy, D.C.; Sanchez, C.J.; Christy, R.J.; Burmeister, D.M. An optimized staining technique for the detection of Gram positive and Gram negative bacteria within tissue. BMC Res. Notes 2016, 9, 216. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; O’Keefe, J.; Orr, D.; Loth, L.; Banks, M.; Wakeley, P.; West, D.; Card, R.; Ibata, G.; Van Maanen, K.; et al. Validation of a real-time PCR assay for the detection of bovine herpesvirus 1 in bovine semen. J. Virol. Methods 2007, 144, 103–108. [Google Scholar] [CrossRef]
- Das Neves, C.G.; Mørk, T.; Thiry, J.; Godfroid, J.; Rimstad, E.; Thiry, E.; Tryland, M. Cervid herpesvirus 2 experimentally reactivated in reindeer can produce generalized viremia and abortion. Virus Res. 2009, 145, 321–328. [Google Scholar] [CrossRef]
- Das Neves, C.G.; Mørk, T.; Godfroid, J.; Sørensen, K.K.; Breines, E.; Hareide, E.; Thiry, J.; Rimstad, E.; Thiry, E.; Tryland, M. Experimental Infection of Reindeer with Cervid Herpesvirus 2. Clin. Vaccine Immunol. 2009, 16, 1758–1765. [Google Scholar] [CrossRef]
- Dickey, A.M.; Loy, J.D.; Bono, J.L.; Smith, T.P.L.; Apley, M.D.; Lubbers, B.V.; DeDonder, K.D.; Capik, S.F.; Larson, R.L.; White, B.J.; et al. Large genomic differences between Moraxella bovoculi isolates acquired from the eyes of cattle with infectious bovine keratoconjunctivitis versus the deep nasopharynx of asymptomatic cattle. Vet. Res. 2016, 47, 31. [Google Scholar] [CrossRef]
- Dickey, A.M.; Schuller, G.; Loy, J.D.; Clawson, M.L. Whole genome sequencing of Moraxella bovoculi reveals high genetic diversity and evidence for interspecies recombination at multiple loci. PLoS ONE 2018, 13, e0209113. [Google Scholar] [CrossRef] [PubMed]
| Inoculum | ID | General Histopathological Evaluation | Euthanasia | qPCR | |||||
|---|---|---|---|---|---|---|---|---|---|
| Upper eyelid 1 | Lower eyelid 1 | Cornea 2 | Lacrimal gland 1 | Ct 3 | Viral copies 3 | ||||
| CvHV2 (group 1; n = 5) | R19 | N/A | 4 | 2 | 1 | Day 2 p.i. | 19.59 | 1.29 x 106 | |
| R5 | 4 | 5 | 4 | 4 | Day 4 p.i. | 22.10 | 2.10 x 105 | ||
| R10 | 4 | 5 | 3 | 3 | Day 5 p.i. | 23.78 | 6.15 x 104 | ||
| R21 | 5 | 5 | 3 | 4 | Day 6 p.i. | 24.58 | 3.48 x 104 | ||
| R4 | 5 | 5 | 4 | 2 | Day 7 p.i. | 25.69 | 1.55 x 104 | ||
| CvHV2 and M. bovoculi (group 2; n = 5) | R17 | 4 | 4 | 1 | Day 3 p.i. | 21.50 | 3.28 x 105 | ||
| R13 | 4 | 5 | 4 | 1 | Day 4 p.i. | 22.37 | 2.10 x 105 | ||
| R7 | 4 | 4 | 4 | 1 | Day 5 p.i. | 24.03 | 5.15 x 104 | ||
| R3 | 4 | 5 | 3 | 4 | Day 6 p.i. | 24.09 | 4.96 x 104 | ||
| R1 | 5 | 5 | 4 | 4 | Day 7 p.i. | 25.67 | 1.57 x 104 | ||
| M. bovoculi (group 3; n = 5) | R16 | 1 | 1 | 1 | 1 | Day 3 p.i. | N/A | N/A | |
| R6 | 1 | 1 | 1 | 1 | Day 6 p.i. | N/A | N/A | ||
| R8 | 1 | 1 | 1 | 1 | Day 13 p.i. | N/A | N/A | ||
| R9 | 1 | 1 | 1 | 1 | Day 13 p.i. | N/A | N/A | ||
| R15 | 1 | 1 | 1 | 1 | Day 13 p.i. | N/A | N/A | ||
| Control (group 4; n = 3) | R2 | 1 | 1 | 1 | 1 | Day 15 p.i. | N/A | N/A | |
| R18 | 1 | 1 | 1 | 1 | Day 15 p.i. | N/A | N/A | ||
| R20 | 1 | 1 | 1 | 1 | Day 15 p.i. | N/A | N/A | ||
| 1,2 Histopathological evaluation was graded in a 5-degrees scale system: | |||||||||
| 1 1 = No pathological findings, normal tissue 2 = Mild lesions (e.g. mild erosions, lymphoid follicle reaction, no/little inflammatory cell infiltration), 3 = General moderate inflammation 4 = General moderate inflammation with focal severe inflammation 5 = General severe inflammation. | 2 1 = No pathological findings, normal tissue, 2 = Mild, focal lesions (e.g. mild erosions, mild edema, mild infiltration of cells at the limbus) 3 = Mild, widespread lesions (e.g. erosions of the corneal epithelium, corneal edema) 4 = Mild, widespread lesions with focal severe erosions with edema and cellular reaction at the corneal lumbus 5 = Corneal ulcer (humane end-point) and severe inflammatory reaction. | ||||||||
| 3 Viral copies (qPCR) in eye swab samples of the inoculated eye at the day of euthanasia (two–seven days p.i.). Quantification of viral DNA copies was performed in animals inoculated with CvHV2, alone or in combination with M. bovoculi. PCR without quantification was previously performed in all experimental reindeer by Tryland et al. [20]. | |||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez Romano, J.; Sørensen, K.K.; Larsen, A.K.; Mørk, T.; Tryland, M. Ocular Histopathological Findings in Semi-Domesticated Eurasian Tundra Reindeer (Rangifer tarandus tarandus) with Infectious Keratoconjunctivitis after Experimental Inoculation with Cervid Herpesvirus 2. Viruses 2020, 12, 1007. https://doi.org/10.3390/v12091007
Sánchez Romano J, Sørensen KK, Larsen AK, Mørk T, Tryland M. Ocular Histopathological Findings in Semi-Domesticated Eurasian Tundra Reindeer (Rangifer tarandus tarandus) with Infectious Keratoconjunctivitis after Experimental Inoculation with Cervid Herpesvirus 2. Viruses. 2020; 12(9):1007. https://doi.org/10.3390/v12091007
Chicago/Turabian StyleSánchez Romano, Javier, Karen K. Sørensen, Anett K. Larsen, Torill Mørk, and Morten Tryland. 2020. "Ocular Histopathological Findings in Semi-Domesticated Eurasian Tundra Reindeer (Rangifer tarandus tarandus) with Infectious Keratoconjunctivitis after Experimental Inoculation with Cervid Herpesvirus 2" Viruses 12, no. 9: 1007. https://doi.org/10.3390/v12091007
APA StyleSánchez Romano, J., Sørensen, K. K., Larsen, A. K., Mørk, T., & Tryland, M. (2020). Ocular Histopathological Findings in Semi-Domesticated Eurasian Tundra Reindeer (Rangifer tarandus tarandus) with Infectious Keratoconjunctivitis after Experimental Inoculation with Cervid Herpesvirus 2. Viruses, 12(9), 1007. https://doi.org/10.3390/v12091007







