Comparative Protective Efficacies of Novel Avian Paramyxovirus-Vectored Vaccines against Virulent Infectious Bronchitis Virus in Chickens
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells and Viruses
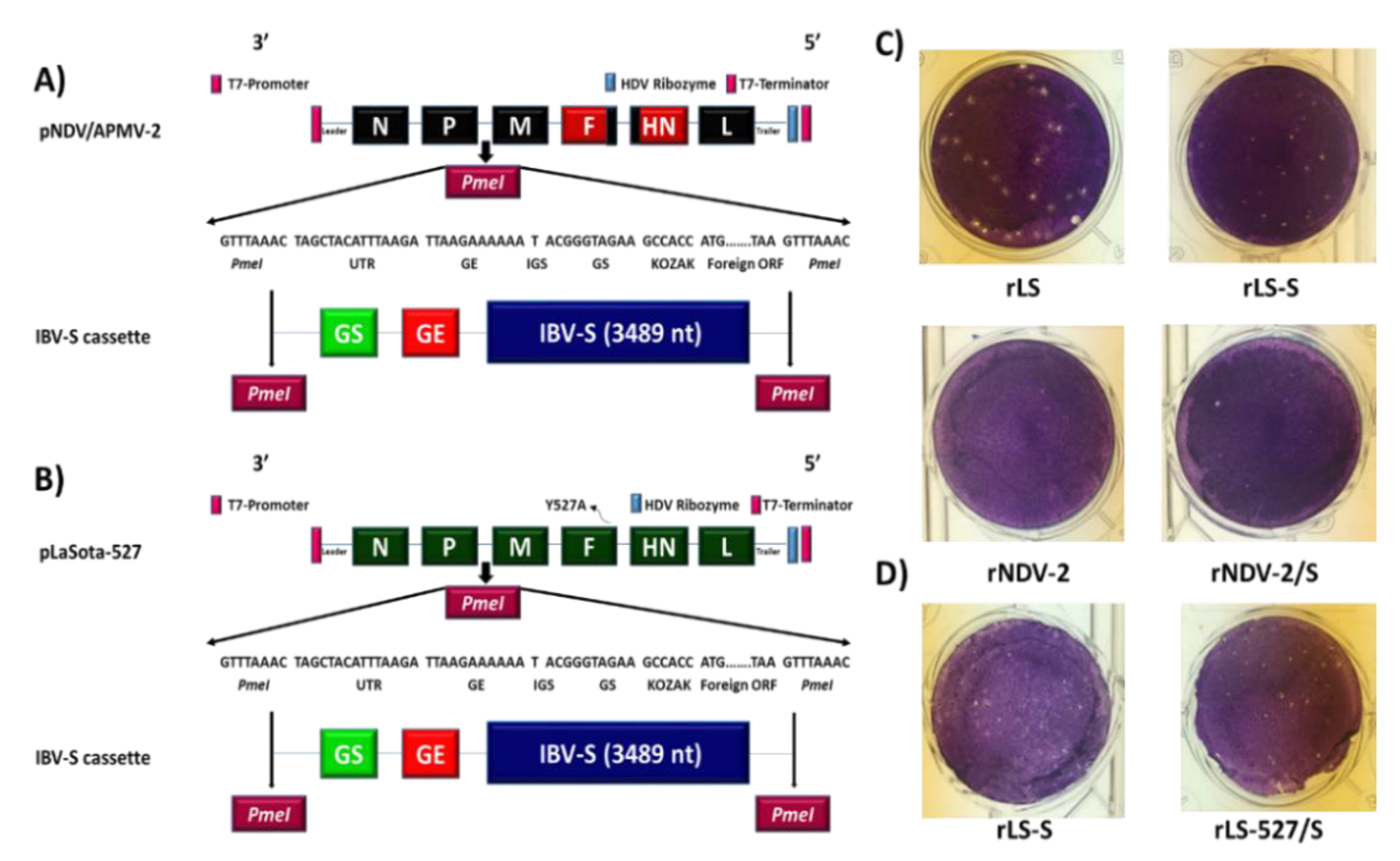
2.2. Generation and Growth Characteristics of rNDV/APMV-2 and rLaSota-527 Expressing S Protein of IBV
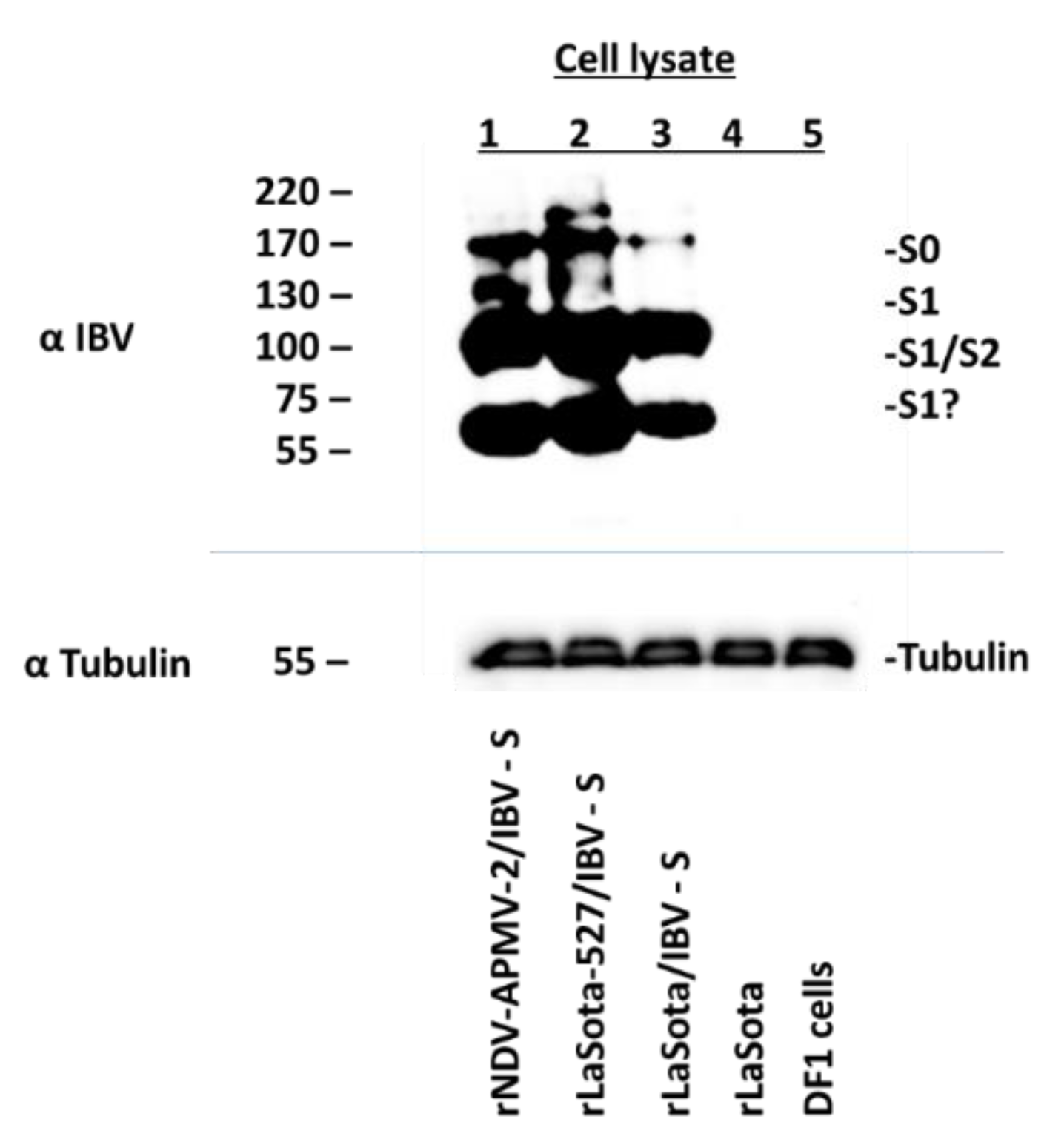
2.3. Expression of S Protein by rNDV-APMV-2/IBV-S and rLaSota-527/IBV-S
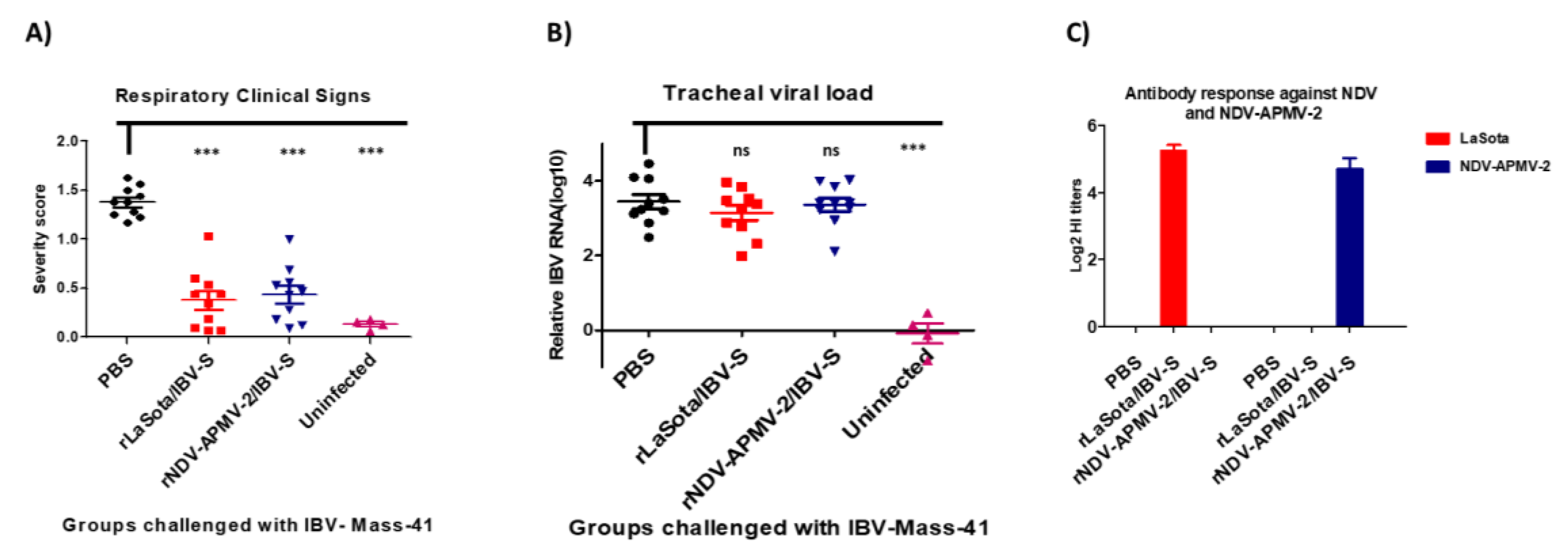
2.4. The Protective Efficacies of rNDV-APMV-2/IBV-S and rLaSota/IBV-S Expressing S Protein against IBV Challenge in SPF Chickens
2.5. The Protective Efficacies of rNDV-APMV-2 and rLaSota Expressing S Protein against IBV Challenge in Broiler Chickens
2.6. Statistical Analysis
3. Results
3.1. Generation and Growth Characterization of rNDV/APMV-2 or rLaSota-527 Expressing S Protein of IBV
3.2. Expression of the S Protein of IBV by rNDV-APMV-2/IBV-S, rLaSota-527/IBV-S and rLaSota/IBV-S
3.3. The Protective Efficacies of rNDV-APMV-2/IBV-S and rLaSota/IBV-S against IBV Challenge in SPF Chickens
3.4. The Protective Efficacies of rNDV-APMV-2/IBV-S and rLaSota/IBV-S against IBV Challenge in Broiler Chickens
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- OIE Terrestrial Manual; Chapter 2.3.3. Avian Infectious Bronchitis. Version adopted by the World Assembly of Delegates of the OIE in May 2013. In OIE terrestrial manual 2018. Available online: https://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/3.03.02_AIB.pdf (accessed on 25 May 2020).
- Cavanagh, D.; Gelb, J.J. Infectious bronchitis. In Diseases of Poultry; Saif, Y.M., Ed.; Blackwell Publishing: Ames, IA, USA, 2008; pp. 117–135. [Google Scholar]
- Masters, P.S.; Perlmon, S. Coronaviruses. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 825–858. [Google Scholar]
- Cavanagh, D. Coronavirus avian infectious bronchitis virus. Vet. Res. 2007, 38, 281–297. [Google Scholar] [CrossRef] [PubMed]
- Collisson, E.W.; Pei, J.; Dzielawa, J.; Seo, S.H. Cytotoxic T lymphocytes are critical in the control of infectious bronchitis virus in poultry. Dev. Comp. Immunol. 2000, 24, 187–200. [Google Scholar] [CrossRef]
- Ladman, B.S.; Pope, C.R.; Ziegler, A.F. Protection of chickens after live and inactivated virus vaccination against challenge with nephropathogenic infectious bronchitis virus PA/Wolgemuth/98. Avian Dis. 2002, 46, 938–944. [Google Scholar] [CrossRef]
- Meeusen, E.N.T.; Walker, J.; Peters, A.; Pastoret, P.P.; Jungersen, G. Current status of veterinary vaccines. Clin. Microbiol. Rev. 2007, 20, 489–510. [Google Scholar] [CrossRef]
- Bande, F.; Arshad, S.S.; Bejo, M.H.; Moeini, H.; Omar, A.R. Progress and challenges toward the development of vaccines against avian infectious bronchitis. J. Immunol. Res. 2015, 2015, 424860. [Google Scholar] [CrossRef]
- McKinley, E.T.; Hilt, D.A.; Jackwood, M.W. Avian coronavirus infectious bronchitis attenuated live vaccines undergo selection of subpopulations and mutations following vaccination. Vaccine 2008, 26, 1274–1284. [Google Scholar] [CrossRef]
- Lee, S.W.; Markham, P.F.; Coppo, M.J.; Legione, A.R.; Markham, J.F.; Noormohammadi, A.H.; Browning, G.F.; Ficorilli, N.; Hartley, C.A.; Devlin, J.M. Attenuated vaccines can recombine to form virulent field viruses. Science 2012, 337, 188. [Google Scholar] [CrossRef]
- Wakenell, P.S.; Sharma, J.M.; Slocombe, R.F. Embryo vaccination of chickens with infectious bronchitis virus: Histologic and ultrastructural lesion response and immunologic response to vaccination. Avian Dis. 1995, 39, 752–765. [Google Scholar] [CrossRef] [PubMed]
- Tarpey, I.; Orbell, S.J.; Britton, P. Safety and efficacy of an infectious bronchitis virus used for chicken embryo vaccination. Vaccine 2006, 24, 6830–6838. [Google Scholar] [CrossRef]
- Wit, J.J.; Cook, K.A. Factors influencing the outcome of infectious bronchitis vaccination and challenge experiments. Avian Pathol. 2014, 43, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.M.; Zhao, Y.; Gaoetal, H.B. Evaluation of recombinant fowl pox virus expressing infectious bronchitis virus S1 gene and chicken interferon-γ gene for immune protection against heterologous strains. Vaccine 2011, 29, 1576–1582. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.A.; Pooley, C.; Ignjatovic, J.; Tyack, S.G.A. Recombinant fowl adenovirus expressing the S1 gene of infectious bronchitis virus protects against challenge with infectious bronchitis virus. Vaccine 2003, 21, 2730–2736. [Google Scholar] [CrossRef]
- Toro, H.; Zhao, W.; Breedlove, C.; Zhang, Z.; Van Santen, V.; Yu, Q. Infectious bronchitis virus S2 expressed from recombinant virus confers broad protection against challenge. Avian Dis. 2014, 58, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Sun, J.; Qi, T.; Zhao, W.; Han, Z.; Yang, X.; Liu, S. Recombinant Newcastle disease virus expressing the infectious bronchitis virus S1 gene protects chickens against Newcastle disease virus and infectious bronchitis virus challenge. Vaccine 2017, 35, 2435–2442. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Yang, M.F.; Cui, B.A. Construction and immunogenicity of a recombinant fowl pox vaccine co-expressing S1 glycoprotein of infectious bronchitis virus and chicken IL-18. Vaccine 2010, 28, 8112–8119. [Google Scholar] [CrossRef]
- Shirvani, E.; Paldurai, A.; Manoharan, V.K.; Varghese, B.P.; Samal, S.K. A Recombinant Newcastle Disease Virus (NDV) Expressing S Protein of Infectious Bronchitis Virus (IBV) Protects Chickens against IBV and NDV. Sci. Rep. 2018, 10, 8. [Google Scholar] [CrossRef]
- International Committee on Taxonomy of Viruses (ICTV) 2019. Available online: https://talk.ictvonline.org/taxonomy (accessed on 25 May 2020).
- Samal, S.K. Newcastle Disease Virus. In Avian Virology-Current Research and Future Trends; Samal, S.K., Ed.; Norfolk Caister Academic Press: Norfolk, UK, 2019; pp. 43–85. [Google Scholar]
- OIE Terrestrial Manual; Chapter 3.3.14. Newcastle Disease. Version adopted by the World Assembly of Delegates of the OIE in May 2018. Available online: https://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/3.03.14_NEWCASTLE_DIS.pdf (accessed on 25 May 2020).
- Kim, S.H.; Samal, S.K. Reverse Genetics for Newcastle Disease Virus as a Vaccine Vector. Curr. Protoc. Microbiol. 2018, 48, 18.5.1–18.5.12. [Google Scholar] [CrossRef]
- Shirvani, E.; Varghese, B.P.; Paldouri, A.; Samal, S.K. A recombinant avian paramyxovirus type-3 (APMV-3) expressing HA protein of highly pathogenic avian influenza virus (HPAIV) protects chickens against HPAIV. Sci. Rep. 2020, 10, 2221. [Google Scholar] [CrossRef]
- Yoshida, A.; Samal, S.K. Avian Paramyxovirus Type-3 as a Vaccine Vector: Identification of a Genome Location for High Level Expression of a Foreign Gene. Front. Microbiol. 2017, 8, 693. [Google Scholar] [CrossRef]
- Tsunekuni, R.; Hikono, H.; Tanikawa, T.; Kurata, R.; Nakaya, T.; Saito, T. Recombinant Avian Paramyxovirus Serotypes 2, 6, and 10 as Vaccine Vectors for Highly Pathogenic Avian Influenza in Chickens with Antibodies Against Newcastle Disease Virus. Avian Dis. 2017, 61, 296–306. [Google Scholar] [CrossRef]
- Kim, S.H.; Xiao, S.; Shive, H.; Collins, P.L.; Samal, S.K. Replication, Neurotropism, and Pathogenicity of Avian Paramyxovirus Serotypes 1–9 in Chickens and Ducks. PLoS ONE 2012, 7, e34927. [Google Scholar] [CrossRef] [PubMed]
- Subbiaha, M.; Xiaoa, S.; Khattara, S.K.; Diasa, F.M.; Collins, P.L.; Samal, S.K. Pathogenesis of two strains of Avian Paramyxovirus serotype 2, Yucaipa and Bangor, in chickens and turkeys. Avian Dis. 2010, 54, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Nayak, B.; Dias, F.M.; Kumar, S.; Paldurai, A.; Collins, P.L.; Samal, S.K. Avian paramyxovirus serotypes 2-9 (APMV -2-9) vary in the ability to induce protective immunity in chickens against challenge with virulent Newcastle disease virus (APMV-1). Vaccine 2012, 30, 2220–2227. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, S.H.; Paldurai, A.; Samal, S.K. A novel chimeric Newcastle disease virus vectored vaccine against highly pathogenic avian influenza virus. Virology 2017, 503, 31–36. [Google Scholar] [CrossRef]
- Kim, S.H.; Subbiah, M.; Samuel, A.S.; Collins, P.L.; Samal, S.K. Roles of the fusion and hemagglutinin-neuraminidase proteins in replication, tropism, and pathogenicity of avian paramyxoviruses. J. Virol. 2011, 85, 8582–8596. [Google Scholar] [CrossRef]
- Warke, A.; Appleby, L.; Mundt, E.S. Prevalence of antibodies to different avian paramyxoviruses in commercial poultry in the United States. Avian Dis. 2008, 52, 694–697. [Google Scholar] [CrossRef]
- Manoharan, V.K.; Khattar, S.K.; Paldurai, A.; Kim, S.H.; Samal, S.K. A Y527A mutation in the fusion protein of Newcastle disease virus strain LaSota leads to a hyperfusogenic virus with increased replication and immunogenicity. J. Gen. Virol. 2016, 97, 287–292. [Google Scholar] [CrossRef]
- Huang, Z.; Krishnamurthy, S.; Panda, A.; Samal, S.K. High-level expression of a foreign gene from the most 3′-proximal locus of a recombinant Newcastle disease virus. J. Gen. Virol. 2001, 82, 1729–1736. [Google Scholar] [CrossRef]
- Albini, B.; Wick, G.; Rose, E.; Orlans, E. Immunoglobulin production in chicken Harderian glands. Int. Arch. Allergy Appl. Immunol. 1974, 47, 23–34. [Google Scholar] [CrossRef]
- Maslak, D.; Reynolds, D. B cells and T-lymphocyte subsets of the head-associated lymphoid tissues of the chicken. Avian Dis. 1995, 39, 736–742. [Google Scholar] [CrossRef]
- Niedorf, H.; Wolters, B. Development of the Harderian gland in the chicken: Light and electron microscopic investigations. Investig. Cell Pathol. 1978, 1, 205–215. [Google Scholar]
- Reemers, S.S.; Leenen, D.V.; Koerkamp, M.J.G.; Haarlem, D.V.; Haar, P.V.D.; Eden, W.D.; Vervelde, L. Early host responses to avian influenza A virus are prolonged and enhanced at transcriptional level depending on maturation of the immune system. Mol. Immunol. 2010, 47, 1675–1685. [Google Scholar] [CrossRef]
- Watabe, M.; Glick, B. Graft versus host response as influenced by the origin of the cell, age of chicken, and cellular interactions. Poult. Sci. 1983, 62, 1317–1324. [Google Scholar] [CrossRef] [PubMed]
- Wells, L.L.; Lowry, V.K.; Deloach, J.R.; Kogut, M.H. Agedependent phagocytosis and bactericidal activities of the chicken heterophil. Dev. Comp. Immunol. 1998, 22, 103–109. [Google Scholar] [CrossRef]
- Zegpi, R.A.; Gulley, S.L.; Van Santen, V.L.; Joiner, K.S.; Toro, H. Infectious Bronchitis Virus Vaccination at Day 1 of Age Further Limits Cross Protection. Avian Dis. 2019, 63, 302–309. [Google Scholar] [CrossRef]
- Van Ginkel, F.W.; Van Santen, V.L.; Gulley, S.L.; Toro, H. Infectious bronchitis virus in the chicken Harderian gland and lachrymal fluid: Viral load, infectivity, immune cell responses, and effects of viral immunodeficiency. Avian Dis. 2008, 52, 608–617. [Google Scholar] [CrossRef]
- Cavanagh, D.; Davis, P.J.; Mockett, A.P.A. Amino acids within hypervariable region 1 of avian coronavirus IBV (Massachusetts serotype) spike glycoprotein are associated with neutralization epitopes. Virus Res. 1998, 11, 141–150. [Google Scholar] [CrossRef]
- DiNapoli, J.M.; Kotelkin, A.; Yang, L.; Elankumaran, S.; Murphy, B.R.; Samal, S.K.; Collins, P.L.; Burkreyev, A. Newcastle disease virus, a host range-restricted virus, as a vaccine for intranasal immunization against emerging pathogens. Proc. Natl. Acad. Sci. USA 2007, 104, 9788–9793. [Google Scholar] [CrossRef]
- Zegpi, R.A.; He, L.; Yu, Q.; Joiner, K.S.; Van Santen, V.L.; Toro, H. Limited Protection Conferred by Recombinant Newcastle Disease Virus Expressing Infectious Bronchitis Spike Protein. Avian Dis. 2020, 64, 53–59. [Google Scholar] [CrossRef]
- Hohdatsu, T.; Yamada, M.; Tominaga, R.; Makino, K.; Kida, K.; Koyama, H. Antibody-dependent enhancement of feline infectious peritonitis virus infection in feline alveolar macrophages and human monocyte cell line U937 by serum of cats experimentally or naturally infected with feline coronavirus. J. Vet. Med. Sci. 1998, 60, 49–55. [Google Scholar] [CrossRef]
- Wang, S.F.; Tseng, S.P.; Yen, C.H.; Yang, J.U.; Tsao, C.H.; Shen, C.W.; Chen, K.H.; Liu, F.T.; Liu, W.T.; Chen, Y.M.A.; et al. Antibody-dependent SARS coronavirus infection is mediated by antibodies against spike proteins. Biochem. Biophys. Res. Commun. 2014, 451, 208–214. [Google Scholar] [CrossRef] [PubMed]
| Groups | Prime Immunization One-Day-Old | Boost-Immunization Three-Week-Old |
|---|---|---|
| 1 | PBS | PBS |
| 2 | rNDV-APMV-2/IBV-S | - |
| 3 | rLaSota/IBV-S | - |
| 4 | rNDV-APMV-2/IBV-S | rLaSota/IBV-S |
| 5 | rLaSota/IBV-S | rNDV-APMV-2/IBV-S |
| 6 | rLaSota/IBV-S | rLaSota/IBV-S |
| 7 | - | rLaSota/IBV-S |
| 8 | - | live IBV Vaccine |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shirvani, E.; Samal, S.K. Comparative Protective Efficacies of Novel Avian Paramyxovirus-Vectored Vaccines against Virulent Infectious Bronchitis Virus in Chickens. Viruses 2020, 12, 697. https://doi.org/10.3390/v12070697
Shirvani E, Samal SK. Comparative Protective Efficacies of Novel Avian Paramyxovirus-Vectored Vaccines against Virulent Infectious Bronchitis Virus in Chickens. Viruses. 2020; 12(7):697. https://doi.org/10.3390/v12070697
Chicago/Turabian StyleShirvani, Edris, and Siba K. Samal. 2020. "Comparative Protective Efficacies of Novel Avian Paramyxovirus-Vectored Vaccines against Virulent Infectious Bronchitis Virus in Chickens" Viruses 12, no. 7: 697. https://doi.org/10.3390/v12070697
APA StyleShirvani, E., & Samal, S. K. (2020). Comparative Protective Efficacies of Novel Avian Paramyxovirus-Vectored Vaccines against Virulent Infectious Bronchitis Virus in Chickens. Viruses, 12(7), 697. https://doi.org/10.3390/v12070697




