Translation-Independent Roles of RNA Secondary Structures within the Replication Protein Coding Region of Turnip Crinkle Virus
Abstract
1. Introduction
2. Materials and Methods
2.1. Constructs
2.2. Agro-Infiltration
2.3. RNA Extraction and Northern Blotting
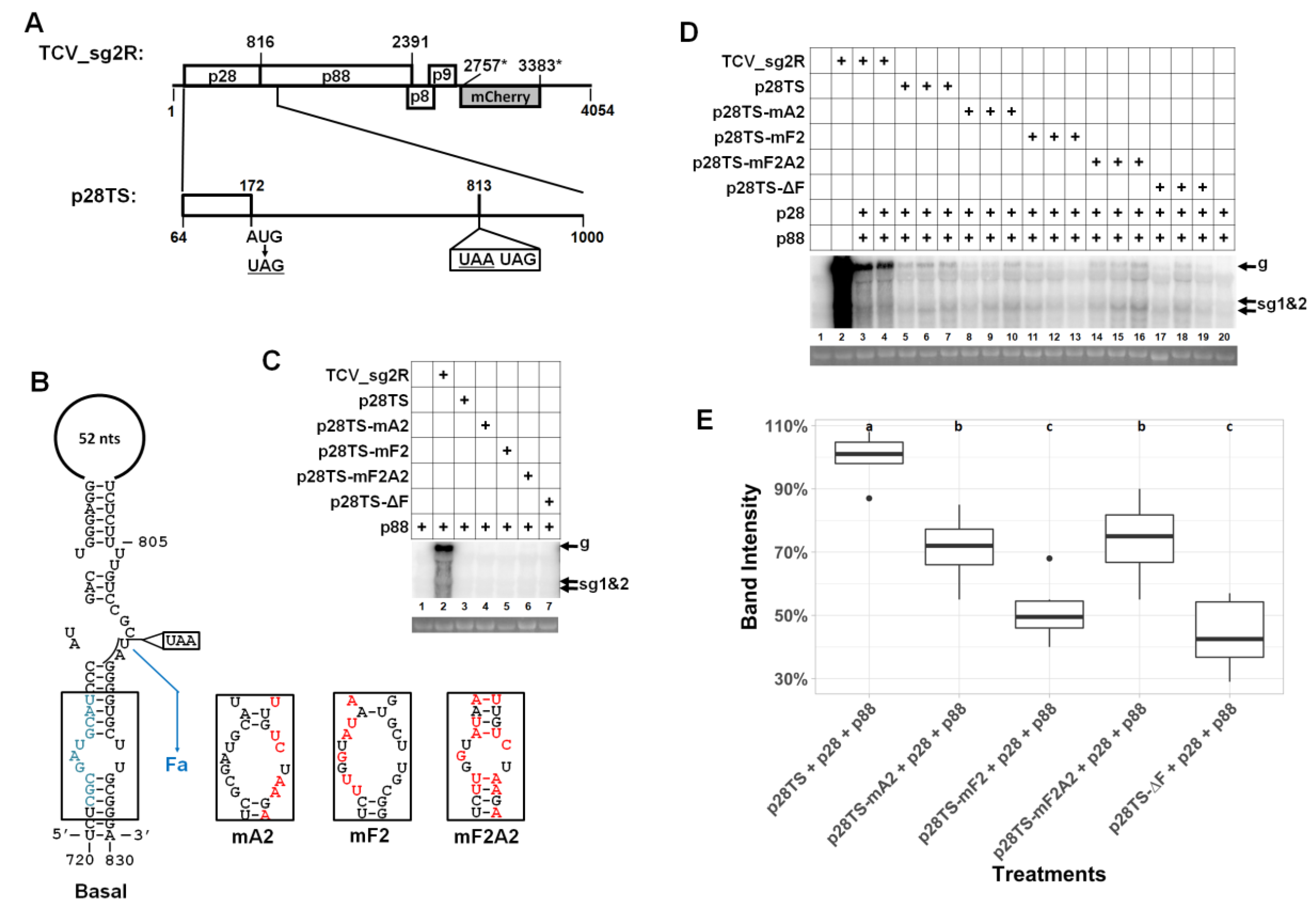
3. Results
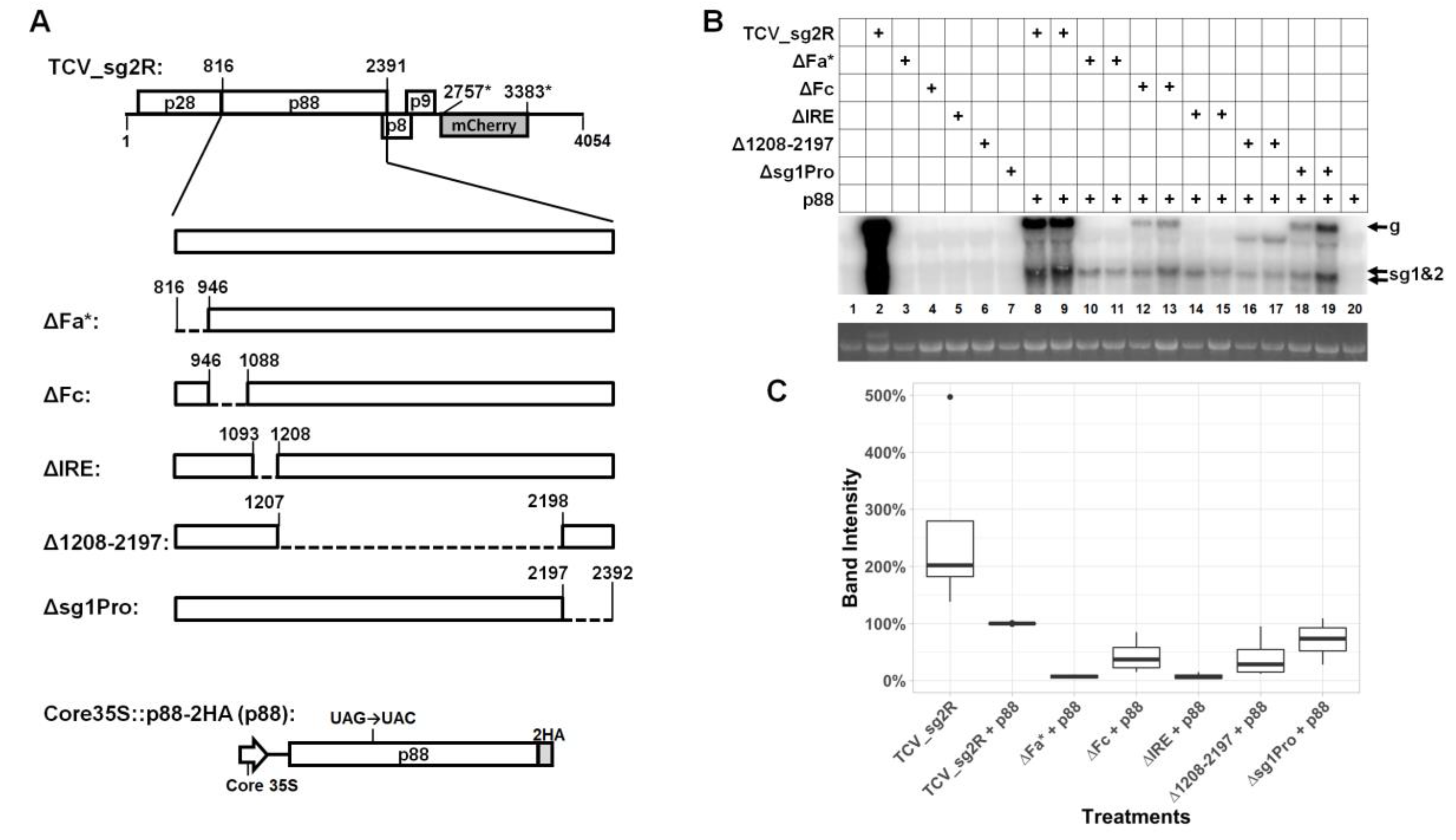
3.1. Much of the p88 Coding Sequence is Dispensable for TCV gRNA Accumulation in Infected Cells
3.2. Two Short Sections of the p88 Coding Sequence Are Essential for Robust Accumulation of TCV gRNA but not sgRNA
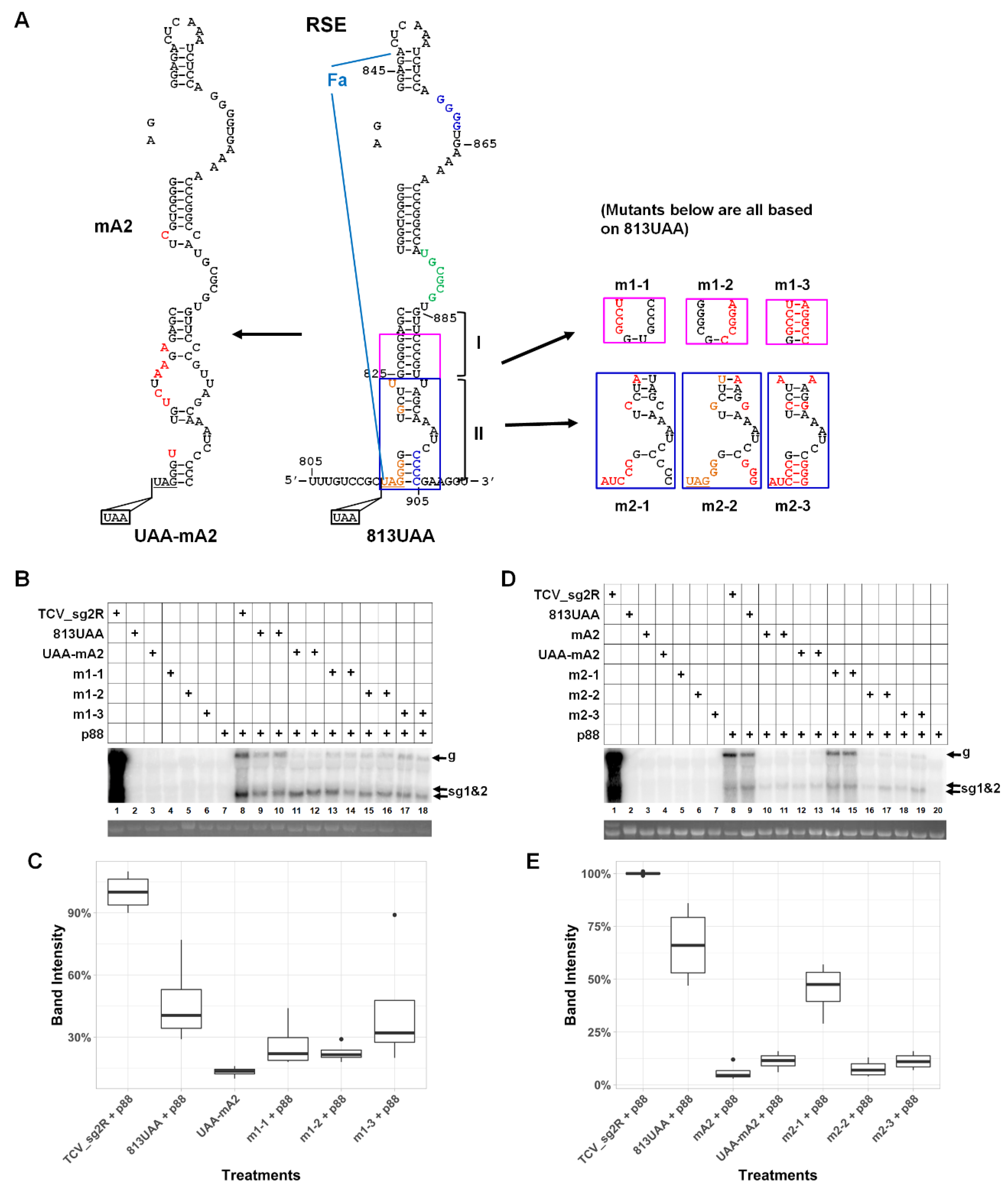
3.3. An Eight Base-Pair (bp) Stem within the Lower Half of RSE Modestly Contributes to TCV gRNA Accumulation
3.4. A Highly Conserved Sequence Motif in the Vicinity of p28 Stop Codon Contributes Little to TCV gRNA Abundance
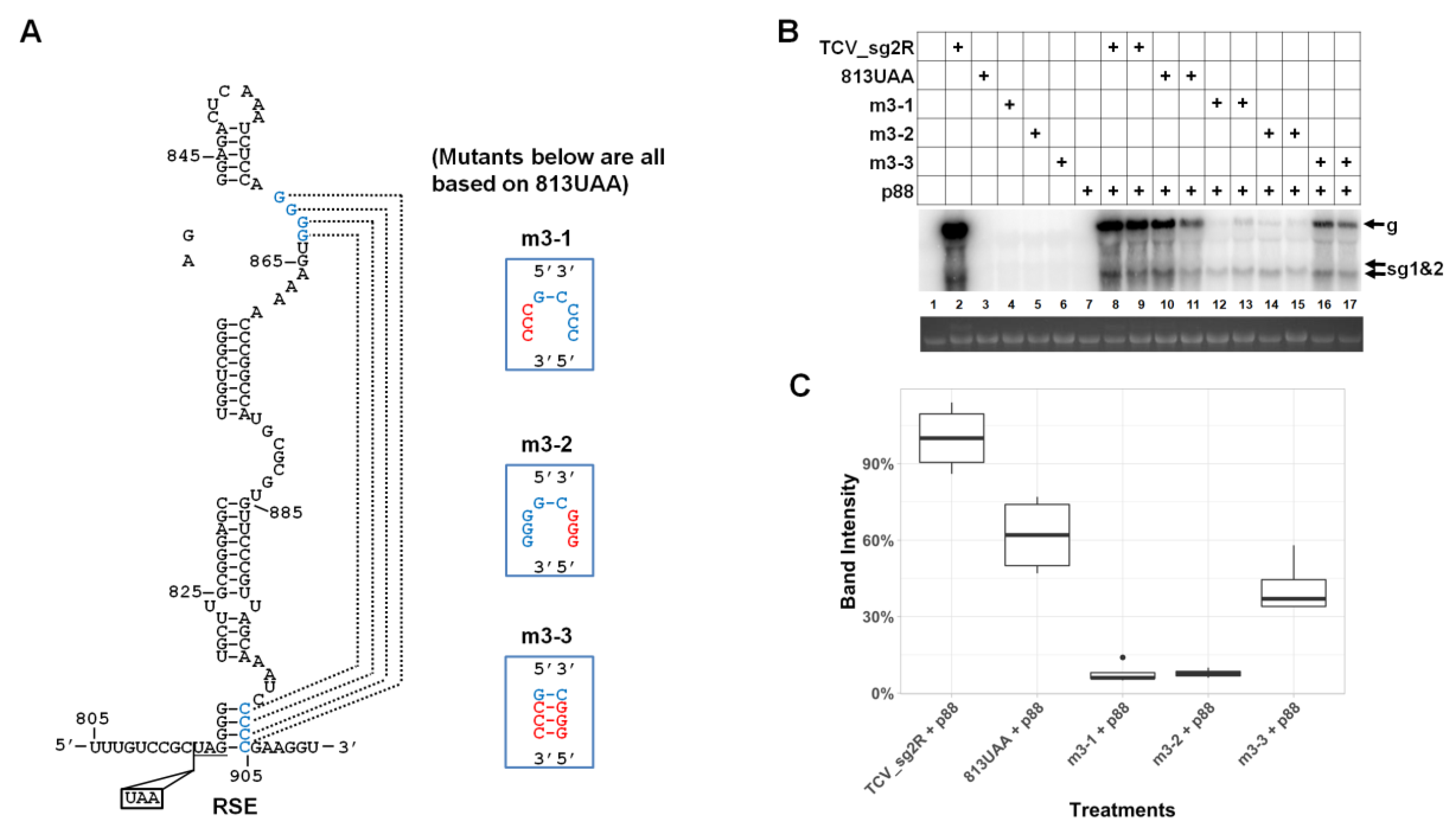
3.5. The Lower-Right Side of Stem II Plays a Dominant Role in TCV gRNA Accumulation
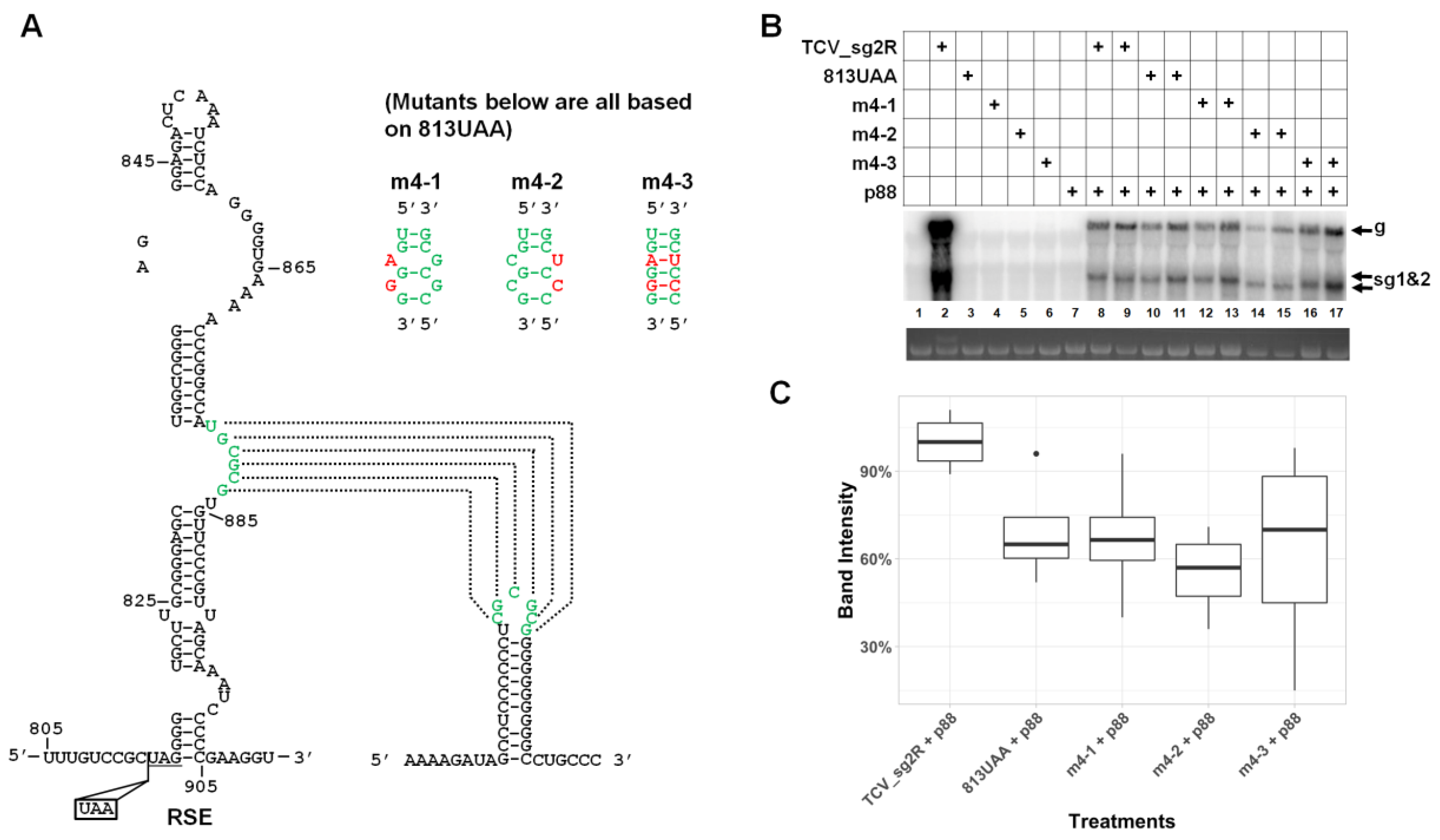
3.6. Robust Accumulation of TCV gRNA Depends on the Integrity of a Previously Identified Pseudoknot within RSE
3.7. The Long-Distance Kissing Loop Interaction between RSE and the 3′ Terminal Stem–Loop does not Contribute to TCV gRNA Abundance
3.8. A Sequence Element within p28 Coding Sequence has a Modest Role in TCV gRNA Accumulation
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Newburn, L.R.; White, K.A. Cis-acting RNA elements in positive-strand RNA plant virus genomes. Virology 2015, 479–480, 434–443. [Google Scholar] [CrossRef]
- Doudna, J.A.; Sarnow, P. Translation Initiation by Viral Internal Ribosome Entry Sites. Transl. Control Biol. Med. 2007, 48, 129–153. [Google Scholar]
- Newburn, L.R.; White, K.A. A trans-activator-like structure in RCNMV RNA1 evokes the origin of the trans-activator in RNA2. PLoS Pathog. 2020, 16, e1008271. [Google Scholar] [CrossRef]
- Simon, A.E.; Miller, W.A. 3′ cap-independent translation enhancers of plant viruses. Annu. Rev. Microbiol. 2013, 67, 21–42. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, B.L.; Lee, P.K.K.; White, K.A. Internal RNA Replication Elements are Prevalent in Tombusviridae. Front. Microbiol. 2012, 3, 279. [Google Scholar] [CrossRef] [PubMed]
- Cimino, P.A.; Nicholson, B.L.; Wu, B.; Xu, W.; White, K.A. Multifaceted Regulation of Translational Readthrough by RNA Replication Elements in a Tombusvirus. PLoS Pathog. 2011, 7, e1002423. [Google Scholar] [CrossRef] [PubMed]
- Kuhlmann, M.M.; Chattopadhyay, M.; Stupina, V.A.; Gao, F.; Simon, A.E. An RNA Element That Facilitates Programmed Ribosomal Readthrough in Turnip Crinkle Virus Adopts Multiple Conformations. J. Virol. 2016, 90, 8575–8591. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Ye, X.; Willie, K.; Lin, J.; Zhang, X.; Redinbaugh, M.G.; Simon, A.E.; Morris, T.J.; Qu, F. The Capsid Protein of Turnip Crinkle Virus Overcomes Two Separate Defense Barriers To Facilitate Systemic Movement of the Virus in Arabidopsis. J. Virol. 2010, 84, 7793–7802. [Google Scholar] [CrossRef] [PubMed]
- Qu, F.; Ren, T.; Morris, T.J. The Coat Protein of Turnip Crinkle Virus Suppresses Posttranscriptional Gene Silencing at an Early Initiation Step. J. Virol. 2003, 77, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Sun, R.; Guo, Q.; Zhang, X.-F.; Qu, F. Repression of turnip crinkle virus replication by its replication protein p88. Virology 2019, 526, 165–172. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, X.; Singh, J.; Li, D.; Qu, F. Temperature-Dependent Survival of Turnip Crinkle Virus-Infected Arabidopsis Plants Relies on an RNA Silencing-Based Defense That Requires DCL2, AGO2, and HEN1. J. Virol. 2012, 86, 6847–6854. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Sun, R.; Guo, Q.; Zhang, S.; Meulia, T.; Halfmann, R.; Li, D.; Qu, F. A self-perpetuating repressive state of a viral replication protein blocks superinfection by the same virus. PLOS Pathog. 2017, 13, e1006253. [Google Scholar] [CrossRef] [PubMed]
- Wei, N.; Hacker, D.L.; Morris, T.J. Characterization of an internal element in turnip crinkle virus RNA involved in both coat protein binding and replication. Virology 1992, 190, 346–355. [Google Scholar] [CrossRef]
- Zhang, X.-F.; Guo, J.; Zhang, X.; Meulia, T.; Paul, P.; Madden, L.V.; Li, D.; Qu, F. Random Plant Viral Variants Attain Temporal Advantages During Systemic Infections and in Turn Resist other Variants of the Same Virus. Sci. Rep. 2015, 5, 15346. [Google Scholar] [CrossRef] [PubMed]
- Wei, N.; Heaton, L.; Morris, T.; Harrison, S. Structure and assembly of turnip crinkle virus. J. Mol. Boil. 1990, 214, 85–95. [Google Scholar] [CrossRef]
- Wang, J.; Simon, A.E. Analysis of the Two Subgenomic RNA Promoters for Turnip Crinkle Virusin Vivoandin Vitro. Virology 1997, 232, 174–186. [Google Scholar] [CrossRef]
- Malpica-López, N.; Rajeswaran, R.; Beknazariants, D.; Seguin, J.; Golyaev, V.; Farinelli, L.; Pooggin, M.M. Revisiting the Roles of Tobamovirus Replicase Complex Proteins in Viral Replication and Silencing Suppression. Mol. Plant Microbe Interact. 2018, 31, 125–144. [Google Scholar] [CrossRef]
- Wu, B.; Oliveri, S.; Mandic, J.; White, K.A. Evidence for a Premature Termination Mechanism of Subgenomic mRNA Transcription in a Carmovirus. J. Virol. 2010, 84, 7904–7907. [Google Scholar] [CrossRef]
- Wu, B.; White, K.A. Uncoupling RNA virus replication from transcription via the polymerase: Functional and evolutionary insights. EMBO J. 2007, 26, 5120–5130. [Google Scholar] [CrossRef]
- Ertel, K.J.; Benefield, D.; Castaño-Diez, D.; Pennington, J.G.; Horswill, M.; Boon, J.A.D.; Otegui, M.S.; Ahlquist, P. Cryo-electron tomography reveals novel features of a viral RNA replication compartment. eLife 2017, 6, e25940. [Google Scholar] [CrossRef]
- Kawamura-Nagaya, K.; Ishibashi, K.; Huang, Y.-P.; Miyashita, S.; Ishikawa, M. Replication protein of tobacco mosaic virus cotranslationally binds the 5′ untranslated region of genomic RNA to enable viral replication. Proc. Natl. Acad. Sci. USA 2014, 111, E1620–E1628. [Google Scholar] [CrossRef] [PubMed]
- White, K.; Skuzeski, J.M.; Li, W.; Wei, N.; Morris, T. Immunodetection, Expression Strategy and Complementation of Turnip Crinkle Virus p28 and p88 Replication Components. Virology 1995, 211, 525–534. [Google Scholar] [CrossRef] [PubMed][Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, R.; Zhang, S.; Zheng, L.; Qu, F. Translation-Independent Roles of RNA Secondary Structures within the Replication Protein Coding Region of Turnip Crinkle Virus. Viruses 2020, 12, 350. https://doi.org/10.3390/v12030350
Sun R, Zhang S, Zheng L, Qu F. Translation-Independent Roles of RNA Secondary Structures within the Replication Protein Coding Region of Turnip Crinkle Virus. Viruses. 2020; 12(3):350. https://doi.org/10.3390/v12030350
Chicago/Turabian StyleSun, Rong, Shaoyan Zhang, Limin Zheng, and Feng Qu. 2020. "Translation-Independent Roles of RNA Secondary Structures within the Replication Protein Coding Region of Turnip Crinkle Virus" Viruses 12, no. 3: 350. https://doi.org/10.3390/v12030350
APA StyleSun, R., Zhang, S., Zheng, L., & Qu, F. (2020). Translation-Independent Roles of RNA Secondary Structures within the Replication Protein Coding Region of Turnip Crinkle Virus. Viruses, 12(3), 350. https://doi.org/10.3390/v12030350





