1. Introduction
The detection of the genome of pathogens has become the gold standard technique for direct diagnosis because of excellent sensitivity and specificity, and due to its capacity to provide a result within hours [
1,
2]. Nonetheless, there are several factors that merit to be mastered in order to obtain results that can be steadily validated. Among those factors, ensuring and maintaining the quality of the components of the reaction mix, in particular oligonucleotide primers and fluorescent probe. It is important to distinguish basic research context from diagnostic context. The latter can hardly suffer delays in result validation. It is important to underline that clinical microbiology laboratories are now frequently grouping assays for viruses, bacteria, fungi, and parasites not only for diagnosis but also for further characterization of pathogen through genotyping or resistance detection. This obviously rapidly leads to a large number of assays to run daily or weekly, and as a consequence a rather large number of potential pitfalls [
3]. Although failure of one of the mix components is easily detected when the positive control does not provide adequate results, such a situation has an important impact on the laboratory throughput due to delayed results, reordering reagents, increased laboratory costs, increasing technical workload, and feeling insecure concerning the capacity of biologists to provide results and of clinicians to obtain results timely. Whether this can appear as a minor problem for laboratories using few in-house assays, it can rapidly become hectic when a larger number of in-house assays are used for routine diagnostic purpose. There are several causes for failures linked to primers and/or probes such as light exposure that deteriorates fluorescence of the probe, repeated freeze-thaw cycles resulting in DNA degradation, mistakes in final concentrations, or pipetting errors when the reaction mix is prepared [
4]. Such problems have been at least partially solved in commercial kits through serial aliquoting and lyophilization or ambient-temperature stable reagents. Ready-to-use reagents reduce the risk of human errors. Lyophilized reagents are more stable than liquid formulations. The combination of both measures aims at improving the quality of the results. We describe here a process of preparation of oligonucleotide primers and hydrolysis probe in a single tube at predefined optimized concentrations (P&P for Primers and Probe(s)) that are stabilized via lyophilization (Lyoph-P&P). We have compared the performances of two selected assays (Lyoph-P&P vs. the classic protocol using frozen reagents) and have studied the long-term stability of Lyoph-P&P in native and rehydrated formulations. Selected assays target two emerging viruses that are listed on the blueprint of the WHO as to be considered for preparedness and response actions [
5]: chikungunya virus (CHIKV), a single-stranded positive-sense RNA alphavirus, and Rift Valley fever phlebovirus (RVFV), a tri-segmented, single-stranded negative-sense RNA phlebovirus.
4. Discussion
Real-time molecular techniques are now the reference methods for the direct diagnosis of pathogens. Increasingly, automation has developed in order to reduce the number of steps prone to human errors, and now the tendency is towards random access tests where all steps are automated until biological validation. However, this approach, developed by diagnostics companies such as Hologic, Roche, Abbott, Cepheid, BioMerieux among others, focus on marketable tests meaning that a certain amount of assays has to be expected in the business plan before such assays are developed. Commercially developed assays need to be registered by regulation agencies before they are available on the market; in many cases, this leads to delays that are not compatible with preparedness and response activities, as witnessed by the current situation with the novel coronavirus. Moreover, often the development and licensing of a novel assay is conditioned by the size and volume of anticipated future market which is not necessarily considered as profitable. Lastly, the price for such assay is almost always not compatible with daily use in laboratories of developing countries.
Obviously, a large number of microbial targets will never be addressed by such random access technologies due to their lack of marketability although they might be major human, veterinary, or plant pathogens. It is worrisome that this situation is contradictory with the principle of preparedness and response to emerging pathogens [
5].
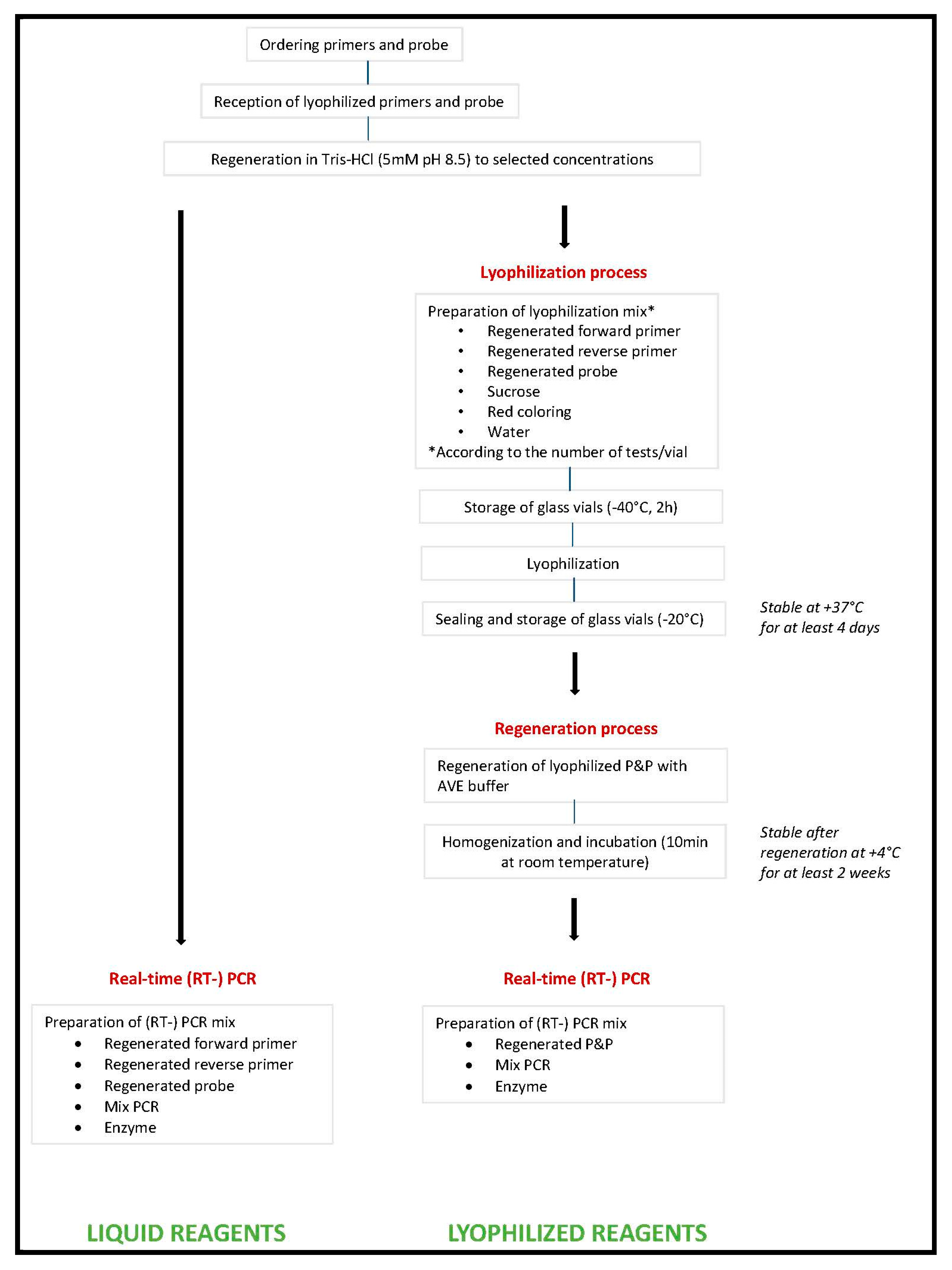
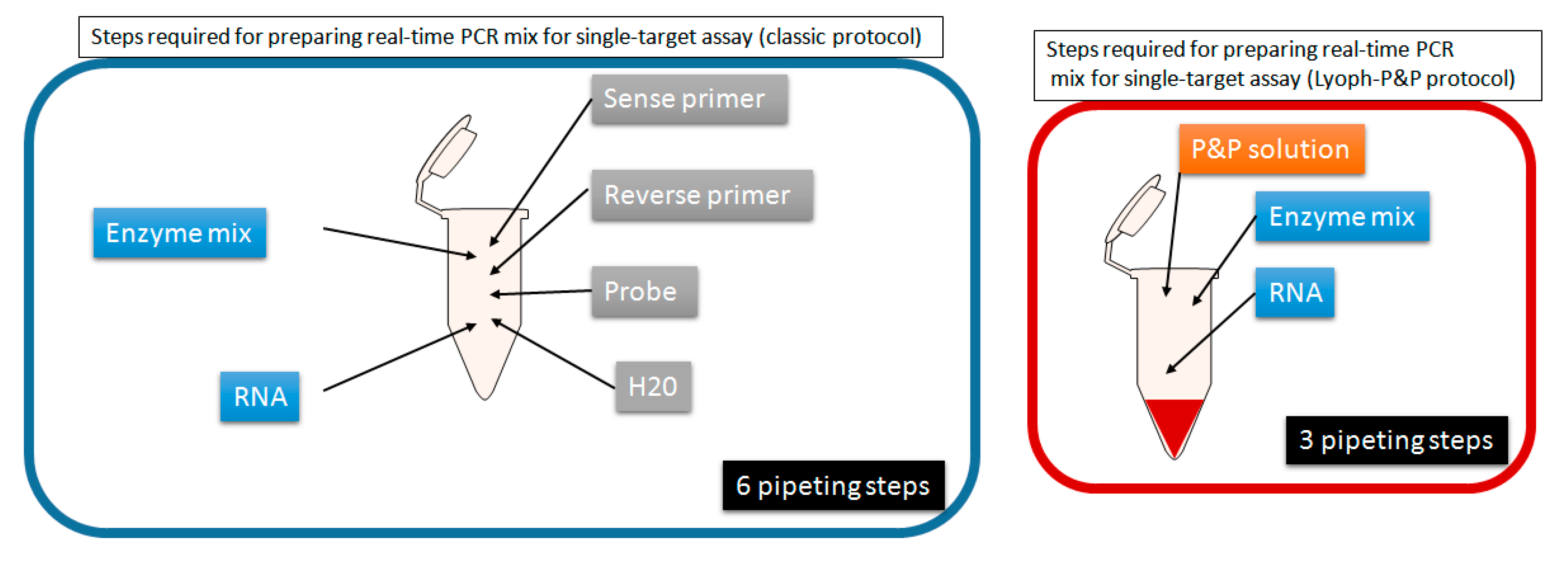
Although real-time molecular techniques are now implemented worldwide, laboratories still face technical problems due to the large number of parameters and reagents to manage, the stability of respective reagents, and the multiple steps from patient to result [
7]. Among the parameters to consider in the process of clinical diagnostics, primers and probe are among those that require the largest number of steps to operate from the stage of ordering the reagents (primers and probe(s)) to the launching the PCR or RT-PCR reaction onto the thermal cycler. Even in the simplest format, two oligonucleotide primers and one probe are ordered from manufacturing companies. Upon reception, each of these three tubes has to be rehydrated and/ or diluted to prepare a stock solution (usually 100 µM) and a working solution (usually 10 µM), both stored at −20 °C for stability. Then for each experiment, specific volumes of each of the three working solutions have to be manipulated to prepare the PCR mix solution which is then distributed into individual reaction tubes or plates. In contrast, the enzyme mix is now mostly commercialized in a 2X solution which requires to perform few steps until distribution into the reaction tubes. Last the “to be tested” solution of total nucleic acids, RNA, or DNA is distributed. Since the manipulation of primers and probe requires the largest number of steps, we selected it as target for simplification (
Figure 1 and
Figure 2).
The aim was (i) to produce a ready-to-use Primers & Probe mix (P&P) for each pathogen to be tested, (ii) to validate the resulting P&P in its Lyophilized form (Lyoph-P&P), (iii) to optimize the whole process and to make it available and usable easily to laboratories willing to adopt the same approach. The ultimate objective was to produce reagents amenable to any laboratory having the capacity to perform real-time molecular detection of pathogens for diagnostic purpose.
The assays that were selected for comparative evaluation in this study have been thoroughly evaluated for the respective detection of CHIKV and RVFV; they have also been used in external quality assessment studies conducted by the European or international level [
6,
8,
9,
10,
11].
The most important aspect was to compare the analytical sensitivity of the Lyoph-P&P assays against the results obtained when the primers and probe were prepared extemporaneously using the classic liquid format. For the two assays included in this study (CHIKV and RVFV), the analytical sensitivity of the Lyoph-P&P is not only equal to that observed with the liquid formulation, but even much better for CHIKV (0.56 copies/µL vs. 14 copies/µL), and slightly better for RVFV (more replicates detected for the last dilution providing positive results). The results observed with clinical samples tested for CHIKV RNA confirm the data obtained in analytical sensitivity studies. Detection of CHIKV RNA using Lyoph-P&P provides results in clinical samples that are at least equal and often better than those obtained with the extemporaneously prepared liquid formulation used as reference. Because of the low number of available clinical samples and due to the highly restrictive MicroOrganisms and Toxins (MOT) French regulation, it was not possible to perform the parallel study for RVFV. However, such comparative studies were done for a substantial number of assays that are routinely processed in the Clinical Microbiology Laboratory of the IHU Méditerranée Infection serving all beds of the Public Hospitals System of Marseille, France (
Supplementary Table S1). Although this has to be addressed systematically when other assays will be transferred from the liquid formulation towards to Lyoph-P&P, these results are very promising and should engage in this direction for the detection of other pathogens.
Although all experiments described here were done using the Superscript® III Platinium® One-Step Quantitative RT-PCR kit (ThermoFischer), we have also used other enzymes such as the one-step qRT-PCR LightCycler® Multiplex RNA Virus Master (Roche) that have provided similar results (data not shown).
As indicated in the
Table 2, the concentration of primers and probe (to be lyophilized) had to be adjusted sometimes in order to obtain sensitivity comparable to that observed with extemporaneously prepared liquid preparation. The correction factor was determined empirically (1.25 and 1.50); interestingly, correction was not systematically necessary, and it could also be needed for one component of the reaction only, as shown with the RVFV assay.
It is important to underline that the rehydration of the Lyoph-P&P must be done as recommended in the protocol. Alternative protocols are likely to result in disappointing performances.
Despite different formats can be prepared as indicated in
Table 3;
Table 4, the question of the stability of Lyoph-P&P after rehydration is important to assess the versatility and flexibility of this solution. Indeed, at laboratory level, it is likely that one or two different formats (number of tests per vial) will be either prepared or ordered; as a consequence, the time during which rehydrated material can be stored without affecting the expected performances of the assay is a key factor. Interestingly, 7-day or 14-day storage at +4 °C had absolutely no deleterious effect on the performances; moreover, in some occurrences, sensitivity was even better after storage than after extemporaneous rehydration. Stability upon +4 °C storage after rehydration is important for the end-users because it prevents discarding reagents; this is not only important economically, but also renders the routine activity more comfortable when a large number of different pathogens are included in detection panels. The fact that rehydrated Lyoph-P&P was stable for at least 14 days after rehydration if stored at 4 °C is interesting because it allows to prepare or order vials containing greater number of tests without fearing the loss of material that is synonymous of increased costs. Stability of rehydrated material warrants versatility of the procedures, thus allowing to prepare/order vials containing 48- or 96-reactions. The same tendency was observed for the two assays suggesting that this phenomenon is not virus-dependent and may be expected with other detection assays.
Assessing the stability during shipping by maintaining the Lyoph-P&P at 37 °C up to 7 days intended to mimic degraded conditions potentially occurring during transportation at a given temperature, and also to address the possibility to perform shipping at ambient temperature. The excellent results observed at day 2 and day 4 support the possibility of ambient temperature shipping for this non-infectious material using rapid delivery companies such as WorldCourrier, UPS, DHL, FedEx, or similar ones that are capable to guarantee delivery within 4 days to almost any place in the world. Again, the promising results observed with CHIKV and RVFV must be confirmed for supplementary assays that will be developed. As examples of its versatility, the described procedure was used to prepare Lyoph-P&P with other RT-qPCR assays from the literature targeting Zika, dengue, and chikungunya viruses [
12,
13,
14]; the corresponding Lyoph-P&P were shipped to overseas laboratories which were satisfied with the resulting performances on their own diagnostic platform (Thirion, unpublished data).
The opportunity to dispense with cold chain is also important to consider for economic reasons.
The last point to consider in the comparative analysis between liquid and Lyoph-P&P formulations deals with the interpretation of the PCR curves. The signal observed with low-copy samples close to the LOD, beyond Ct 35, is frequently weak as shown by low RFU level (
Supplementary Data, dataset#1); interpretation of such results is frequently difficult and as a consequence often induces repeated testing for confirmation. The stronger the intensity of the signal, the easier the discrimination between clear positives and uncertain results. A detailed analysis denotes that for low copy samples, the intensity of the signals is clearly higher with Lyoph-P&P compared with the liquid formulation: approximately 200–1200 RFU vs. 2500–4900 RFU for RVFV, and approximately 120-470 RFU vs. 430-790 RFU for CHIKV (Suppl Data, dataset#1).
Recently, increased robustness of real-time PCR assays has been achieved by combining two targets in a unique reaction tube in order to prevent false negative results that may arise from point mutations/deletions/insertions frequently observed with emerging pathogens, even more frequently with pathogens with RNA genome [
14,
15,
16,
17]. This tendency implies the need to increase the number of different primers and probe within a single assay, which renders the preparation of the reaction mix even more prone to human errors. Whether or not this tendency should expand, Lyoph-P&P would be even more attracting for diagnostic activities in routine clinical microbiology laboratories. The recipient laboratory will have to perform minimal validation steps before the Lyoph-P&P can be included in the routine diagnostic activity.
In conclusions, the advantages of Lyoph-P&P reside (i) in its stability for shipping and storage, (ii) in the drastically reduced number of manipulations to prepare the ultimate reaction tube/plate to be placed in the thermocycler, (iii) in its flexibility in terms of number of reactions per prepared vial (1 to 96, even 1 to 384). Utilization of Lyoph-P&P is an easy manner to transfer diagnostic capacity between laboratories.