Fatal Interstitial Pneumonia Associated with Bovine Coronavirus in Cows from Southern Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Outbreak Description
2.2. Histopathological Analysis
2.3. Bacteriological and Parasitological Analysis
2.4. Viral Nucleic Acids Extraction Procedures
2.5. Real Time Virus Identification
2.6. BCoV Sequencing and Strain Characterization
2.7. Virus Isolation
3. Results
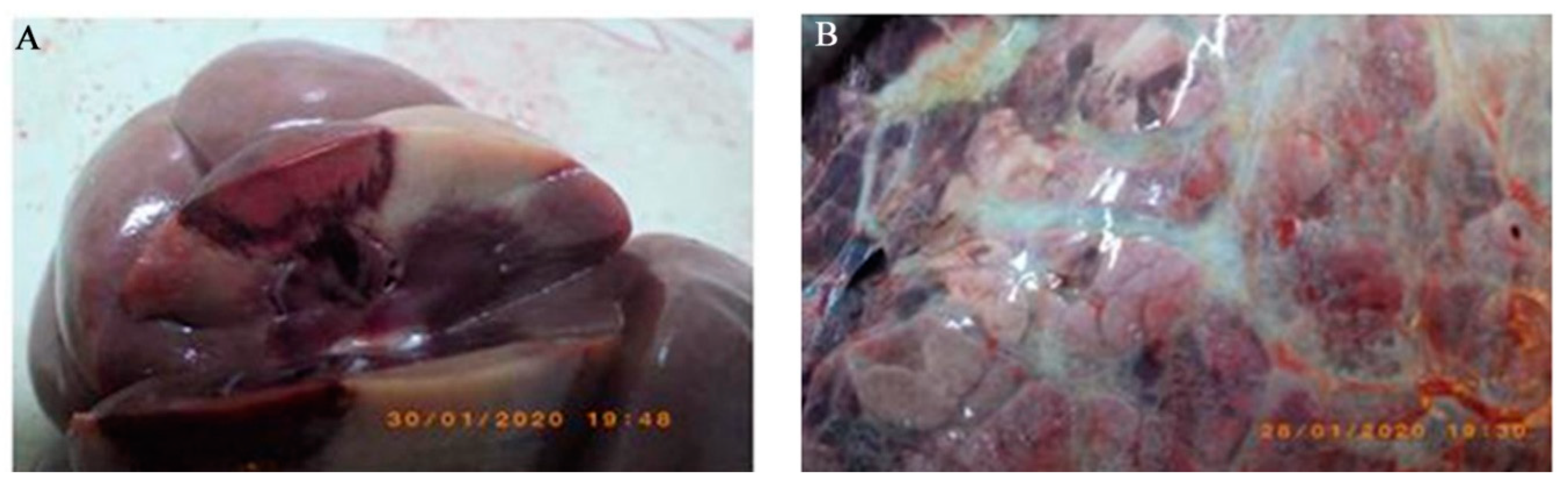
3.1. Necroscopy Findings
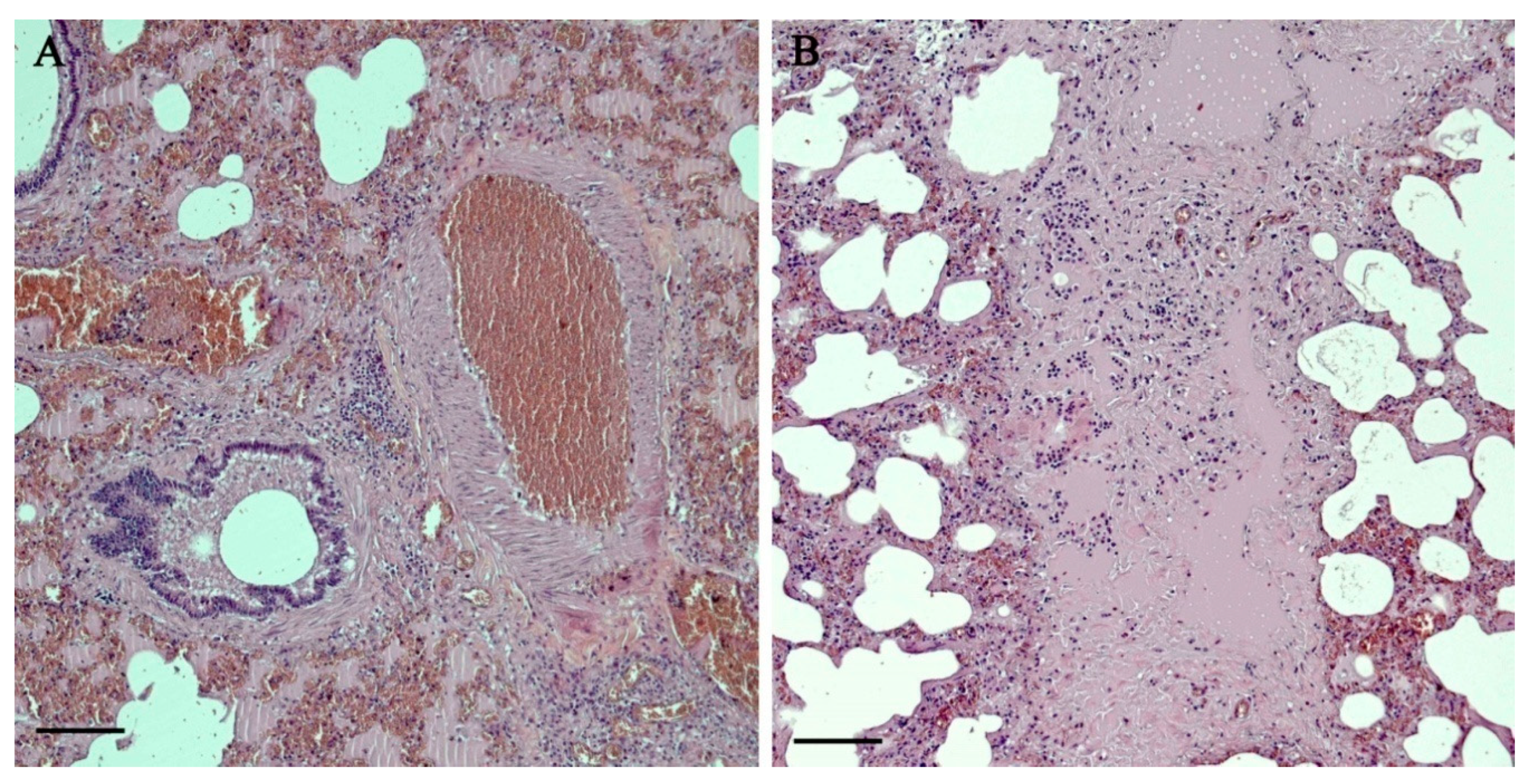
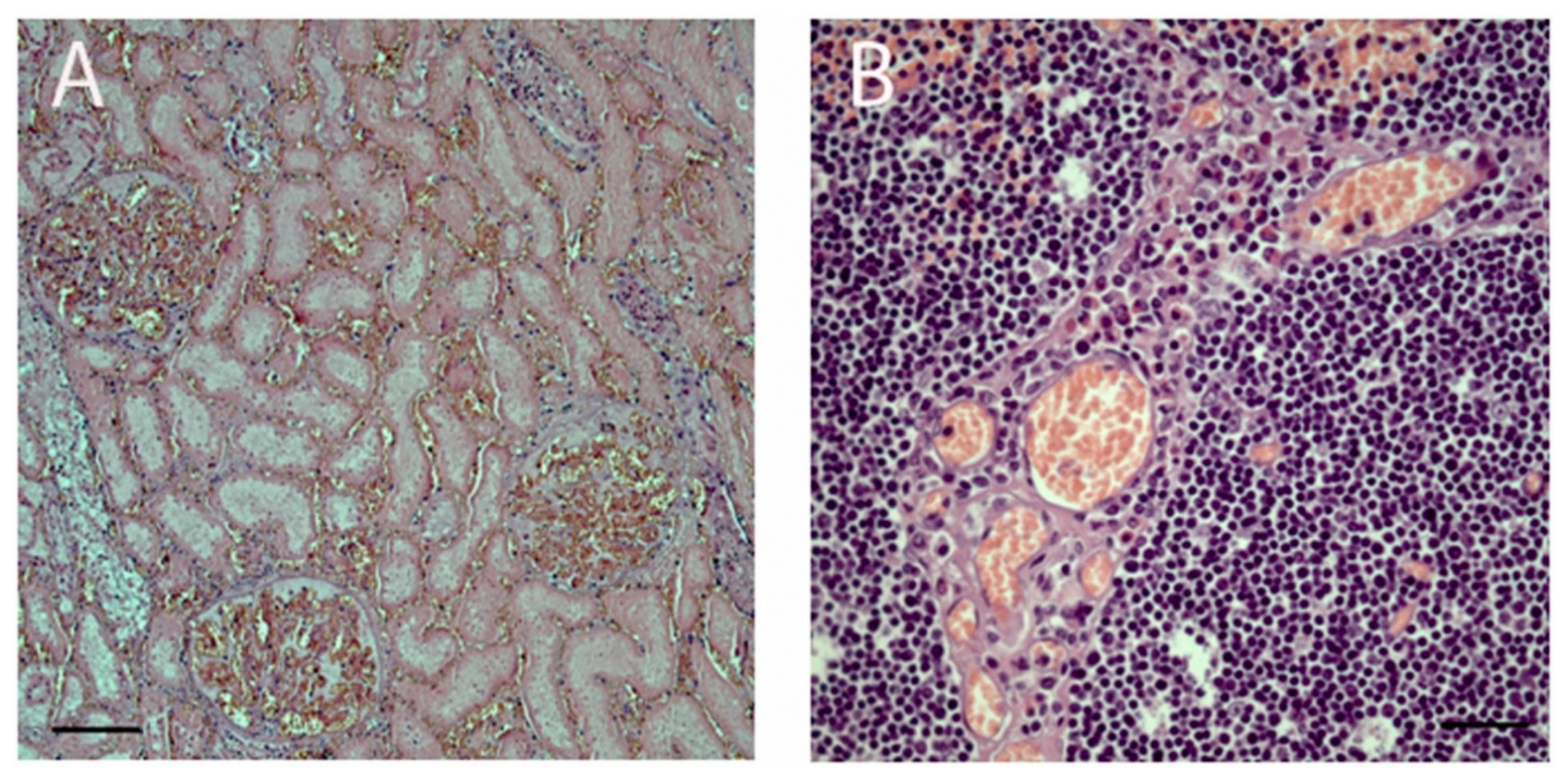
3.2. Histopathology
3.3. Microbiology and Virology
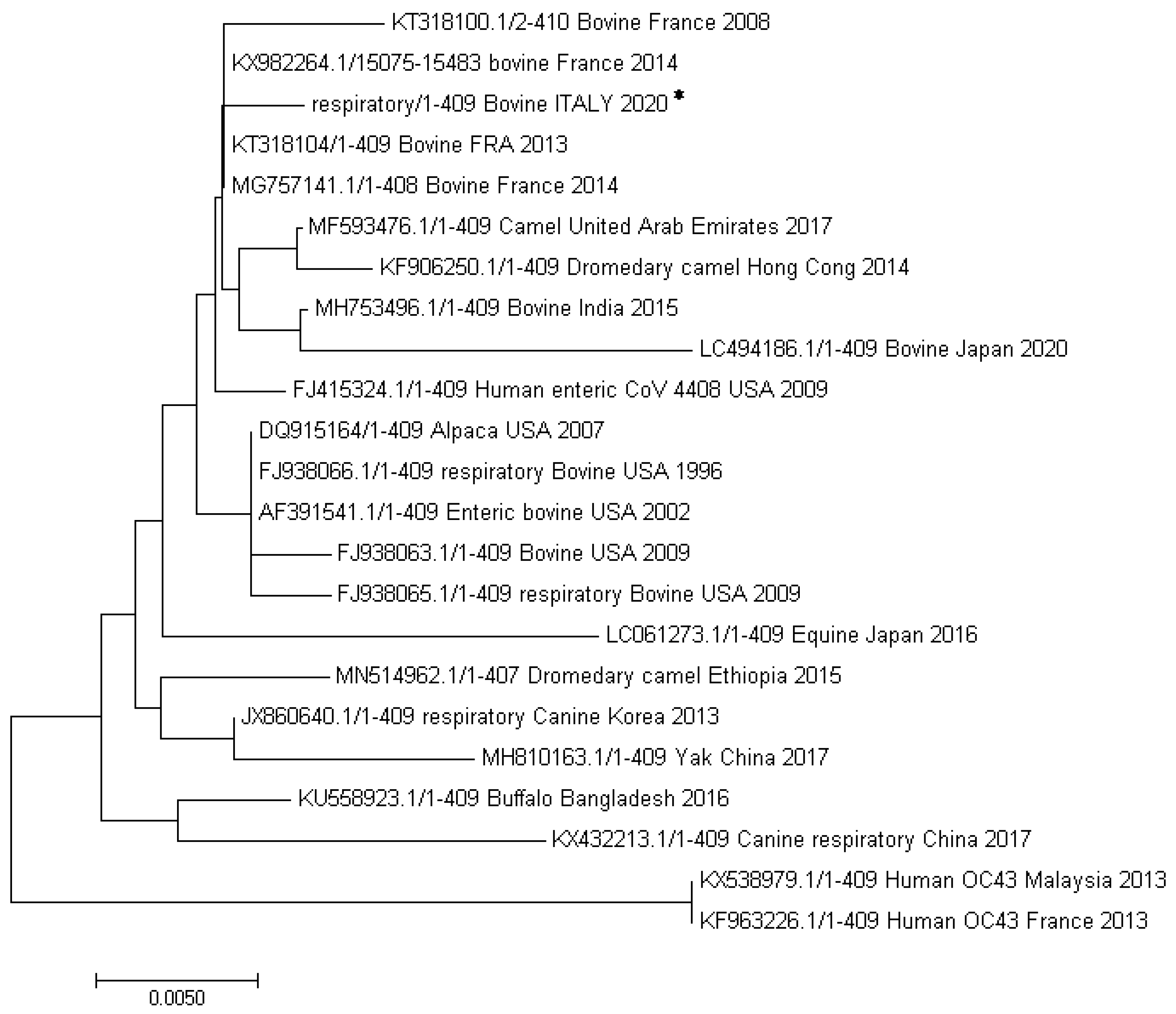
3.4. Sequencing Analysis of BCoV
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Decaro, N.; Lorusso, A. Novel Human Coronavirus (SARS-CoV-2): A Lesson from Animal Coronaviruses. Vet. Microbiol. 2020, 244, 108693. [Google Scholar] [CrossRef] [PubMed]
- Amer, H.M. Bovine-Like Coronaviruses in Domestic and Wild Ruminants. Anim. Health Res. Rev. 2018, 19, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Moneim, A.S.; Abdelwhab, E.M. Evidence for SARS-CoV-2 Infection of Animal Hosts. Pathogens 2020, 9, 529. [Google Scholar] [CrossRef] [PubMed]
- Saif, L.J. Animal Coronaviruses: What can they Teach Us about the Severe Acute Respiratory Syndrome? Rev. Sci. Tech. 2004, 23, 643–660. [Google Scholar] [CrossRef] [PubMed]
- Saif, L.J.; Jung, K. Comparative Pathogenesis of Bovine and Porcine Respiratory Coronaviruses in the Animal Host Species and SARS-CoV-2 in Humans. J. Clin. Microbiol. 2020, 58. [Google Scholar] [CrossRef]
- Stavrinides, J.; Guttman, D.S. Mosaic Evolution of the Severe Acute Respiratory Syndrome Coronavirus. J. Virol. 2004, 78, 76–82. [Google Scholar] [CrossRef]
- Hon, C.C.; Lam, T.Y.; Shi, Z.L.; Drummond, A.J.; Yip, C.W.; Zeng, F.; Lam, P.Y.; Leung, F.C. Evidence of the Recombinant Origin of a Bat Severe Acute Respiratory Syndrome (SARS)-Like Coronavirus and its Implications on the Direct Ancestor of SARS Coronavirus. J. Virol. 2008, 82, 1819–1826. [Google Scholar] [CrossRef]
- Corman, V.M.; Ithete, N.L.; Richards, L.R.; Schoeman, M.C.; Preiser, W.; Drosten, C.; Drexler, J.F. Rooting the Phylogenetic Tree of Middle East Respiratory Syndrome Coronavirus by Characterization of a Conspecific Virus from an African Bat. J. Virol. 2014, 88, 11297–11303. [Google Scholar] [CrossRef]
- Decaro, N.; Campolo, M.; Desario, C.; Cirone, F.; D’Abramo, M.; Lorusso, E.; Greco, G.; Mari, V.; Colaianni, M.L.; Elia, G.; et al. Respiratory Disease Associated with Bovine Coronavirus Infection in Cattle Herds in Southern Italy. J. Vet. Diagn. Investig. 2008, 20, 28–32. [Google Scholar] [CrossRef]
- Decaro, N.; Martella, V.; Elia, G.; Campolo, M.; Mari, V.; Desario, C.; Lucente, M.S.; Lorusso, A.; Greco, G.; Corrente, M.; et al. Biological and Genetic Analysis of a Bovine-Like Coronavirus Isolated from Water Buffalo (Bubalus Bubalis) Calves. Virology 2008, 370, 213–222. [Google Scholar] [CrossRef]
- Ellis, J. What is the Evidence that Bovine Coronavirus is a Biologically Significant Respiratory Pathogen in Cattle? Can. Vet. J. 2019, 60, 147–152. [Google Scholar] [PubMed]
- Decaro, N.; Elia, G.; Campolo, M.; Desario, C.; Mari, V.; Radogna, A.; Colaianni, M.L.; Cirone, F.; Tempesta, M.; Buonavoglia, C. Detection of Bovine Coronavirus using a TaqMan-Based Real-Time RT-PCR Assay. J. Virol. Methods 2008, 151, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Drosten, C.; Gunther, S.; Preiser, W.; van der Werf, S.; Brodt, H.R.; Becker, S.; Rabenau, H.; Panning, M.; Kolesnikova, L.; Fouchier, R.A.; et al. Identification of a Novel Coronavirus in Patients with Severe Acute Respiratory Syndrome. N. Engl. J. Med. 2003, 348, 1967–1976. [Google Scholar] [CrossRef] [PubMed]
- Takiuchi, E.; Stipp, D.T.; Alfieri, A.F.; Alfieri, A.A. Improved detection of bovine coronavirus N gene in faeces of calves infected naturally by a semi-nested PCR assay and an internal control. J. Virol. Methods 2006, 131, 148–154. [Google Scholar] [CrossRef]
- Amoroso, M.G.; Corrado, F.; De Carlo, E.; Lucibelli, M.G.; Martucciello, A.; Guarino, A.; Galiero, G. Bubaline Herpesvirus 1 Associated with Abortion in a Mediterranean Water Buffalo. Res. Vet. Sci. 2013, 94, 813–816. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The Neighbor-Joining Method: A New Method for Reconstructing Phylogenetic Trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Salem, E.; Dhanasekaran, V.; Cassard, H.; Hause, B.; Maman, S.; Meyer, G.; Ducatez, M.F. Global transmission, spatial segregation, and recombination determine the long-term evolution and epidemiology of bovine coronaviruses. Viruses 2020, 12, 534. [Google Scholar] [CrossRef]
- Hasoksuz, M.; Lathrop, S.L.; Gadfield, K.L.; Saif, L.J. Isolation of Bovine Respiratory Coronaviruses from Feedlot Cattle and Comparison of their Biological and Antigenic Properties with Bovine Enteric Coronaviruses. Am. J. Vet. Res. 1999, 60, 1227–1233. [Google Scholar]
- Fulton, R.W.; Blood, K.S.; Panciera, R.J.; Payton, M.E.; Ridpath, J.F.; Confer, A.W.; Saliki, J.T.; Burge, L.T.; Welsh, R.D.; Johnson, B.J.; et al. Lung Pathology and Infectious Agents in Fatal Feedlot Pneumonias and Relationship with Mortality, Disease Onset, and Treatments. J. Vet. Diagn. Investig. 2009, 21, 464–477. [Google Scholar] [CrossRef]
- Francoz, D.; Buczinski, S.; Bélanger, A.; Forté, G.; Labrecque, O.; Tremblay, D.; Wellemans, V.; Dubuc, J. Respiratory Pathogens in Quebec Dairy Calves and their Relationship with Clinical Status, Lung Consolidation, and Average Daily Gain. J. Vet. Intern. Med. 2015, 29, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Mitra, N.; Cernicchiaro, N.; Torres, S.; Li, F.; Hause, B.M. Metagenomic Characterization of the Virome Associated with Bovine Respiratory Disease in Feedlot Cattle Identified Novel Viruses and Suggests an Etiologic Role for Influenza D Virus. J. Gen. Virol. 2016, 97, 1771–1784. [Google Scholar] [CrossRef] [PubMed]
- Workman, A.M.; Kuehn, L.A.; McDaneld, T.G.; Clawson, M.L.; Chitko-McKown, C.G.; Loy, J.D. Evaluation of the Effect of Serum Antibody Abundance Against Bovine Coronavirus on Bovine Coronavirus Shedding and Risk of Respiratory Tract Disease in Beef Calves from Birth through the First Five Weeks in a Feedlot. Am. J. Vet. Res. 2017, 78, 1065–1076. [Google Scholar] [CrossRef] [PubMed]
- Storz, J.; Purdy, C.W.; Lin, X.; Burrell, M.; Truax, R.E.; Briggs, R.E.; Frank, G.H.; Loan, R.W. Isolation of Respiratory Bovine Coronavirus, Other Cytocidal Viruses, and Pasteurella Spp from Cattle Involved in Two Natural Outbreaks of Shipping Fever. J. Am. Vet. Med. Assoc. 2000, 216, 1599–1604. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.O.; Hoet, A.E.; Loerch, S.C.; Wittum, T.E.; Saif, L.J. Evaluation of Concurrent Shedding of Bovine Coronavirus Via the Respiratory Tract and Enteric Route in Feedlot Cattle. Am. J. Vet. Res. 2001, 62, 1436–1441. [Google Scholar] [CrossRef]
- Fulton, R.W.; d’Offay, J.M.; Landis, C.; Miles, D.G.; Smith, R.A.; Saliki, J.T.; Ridpath, J.F.; Confer, A.W.; Neill, J.D.; Eberle, R.; et al. Detection and Characterization of Viruses as Field and Vaccine Strains in Feedlot Cattle with Bovine Respiratory Disease. Vaccine 2016, 34, 3478–3492. [Google Scholar] [CrossRef]
- Fulton, R.W.; Step, D.L.; Wahrmund, J.; Burge, L.J.; Payton, M.E.; Cook, B.J.; Burken, D.; Richards, C.J.; Confer, A.W. Bovine Coronavirus (BCV) Infections in Transported Commingled Beef Cattle and Sole-Source Ranch Calves. Can. J. Vet. Res. 2011, 75, 191–199. [Google Scholar]
- Reynolds, D.J.; Debney, T.G.; Hall, G.A.; Thomas, L.H.; Parsons, K.R. Studies on the Relationship between Coronaviruses from the Intestinal and Respiratory Tracts of Calves. Arch. Virol. 1985, 85, 71–83. [Google Scholar] [CrossRef]
- Saif, L.J.; Redman, D.R.; Moorhead, P.D.; Theil, K.W. Experimentally Induced Coronavirus Infections in Calves: Viral Replication in the Respiratory and Intestinal Tracts. Am. J. Vet. Res. 1986, 47, 1426–1432. [Google Scholar]
- Cho, K.O.; Hasoksuz, M.; Nielsen, P.R.; Chang, K.O.; Lathrop, S.; Saif, L.J. Cross-Protection Studies between Respiratory and Calf Diarrhea and Winter Dysentery Coronavirus Strains in Calves and RT-PCR and Nested PCR for their Detection. Arch. Virol. 2001, 146, 2401–2419. [Google Scholar] [CrossRef]
- Park, S.J.; Kim, G.Y.; Choy, H.E.; Hong, Y.J.; Saif, L.J.; Jeong, J.H.; Park, S.I.; Kim, H.H.; Kim, S.K.; Shin, S.S.; et al. Dual Enteric and Respiratory Tropisms of Winter Dysentery Bovine Coronavirus in Calves. Arch. Virol. 2007, 152, 1885–1900. [Google Scholar] [CrossRef] [PubMed]
- McNulty, M.S.; Bryson, D.G.; Allan, G.M.; Logan, E.F. Coronavirus Infection of the Bovine Respiratory Tract. Vet. Microbiol. 1984, 9, 425–434. [Google Scholar] [CrossRef]
- Kapil, S.; Trent, A.M.; Goyal, S.M. Excretion and Persistence of Bovine Coronavirus in Neonatal Calves. Arch. Virol. 1990, 115, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Chouljenko, V.N.; Lin, X.Q.; Storz, J.; Kousoulas, K.G.; Gorbalenya, A.E. Comparison of Genomic and Predicted Amino Acid Sequences of Respiratory and Enteric Bovine Coronaviruses Isolated from the Same Animal with Fatal Shipping Pneumonia. J. Gen. Virol. 2001, 82, 2927–2933. [Google Scholar] [CrossRef]
- Tsunemitsu, H.; el-Kanawati, Z.R.; Smith, D.R.; Reed, H.H.; Saif, L.J. Isolation of Coronaviruses Antigenically Indistinguishable from Bovine Coronavirus from Wild Ruminants with Diarrhea. J. Clin. Microbiol. 1995, 33, 3264–3269. [Google Scholar] [CrossRef]
- Zhong, N.S.; Wong, G.W. Epidemiology of Severe Acute Respiratory Syndrome (SARS): Adults and Children. Paediatr. Respir. Rev. 2004, 5, 270–274. [Google Scholar] [CrossRef]
- Decaro, N.; Desario, C.; Elia, G.; Mari, V.; Lucente, M.S.; Cordioli, P.; Colaianni, M.L.; Martella, V.; Buonavoglia, C. Serological and Molecular Evidence that Canine Respiratory Coronavirus is Circulating in Italy. Vet. Microbiol. 2007, 121, 225–230. [Google Scholar] [CrossRef]
- Lorusso, A.; Desario, C.; Mari, V.; Campolo, M.; Lorusso, E.; Elia, G.; Martella, V.; Buonavoglia, C.; Decaro, N. Molecular Characterization of a Canine Respiratory Coronavirus Strain Detected in Italy. Virus Res. 2009, 141, 96–100. [Google Scholar] [CrossRef]
- Woo, P.C.; Lau, S.K.; Wernery, U.; Wong, E.Y.; Tsang, A.K.; Johnson, B.; Yip, C.C.; Lau, C.C.; Sivakumar, S.; Cai, J.P.; et al. Novel Betacoronavirus in Dromedaries of the Middle East, 2013. Emerg. Infect. Dis. 2014, 20, 560–572. [Google Scholar] [CrossRef]
- Zhang, X.M.; Herbst, W.; Kousoulas, K.G.; Storz, J. Biological and Genetic Characterization of a Hemagglutinating Coronavirus Isolated from a Diarrhoeic Child. J. Med. Virol. 1994, 44, 152–161. [Google Scholar] [CrossRef]
- Vijgen, L.; Keyaerts, E.; Moes, E.; Thoelen, I.; Wollants, E.; Lemey, P.; Vandamme, A.M.; Van Ranst, M. Complete Genomic Sequence of Human Coronavirus OC43: Molecular Clock Analysis Suggests a Relatively Recent Zoonotic Coronavirus Transmission Event. J. Virol. 2005, 79, 1595–1604. [Google Scholar] [CrossRef] [PubMed]
- Vijgen, L.; Keyaerts, E.; Lemey, P.; Maes, P.; Van Reeth, K.; Nauwynck, H.; Pensaert, M.; Van Ranst, M. Evolutionary History of the Closely Related Group 2 Coronaviruses: Porcine Hemagglutinating Encephalomyelitis Virus, Bovine Coronavirus, and Human Coronavirus OC43. J. Virol. 2006, 80, 7270–7274. [Google Scholar] [CrossRef] [PubMed]
- Forni, D.; Cagliani, R.; Clerici, M.; Sironi, M. Molecular Evolution of Human Coronavirus Genomes. Trends Microbiol. 2017, 25, 35–48. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amoroso, M.G.; Lucifora, G.; Degli Uberti, B.; Serra, F.; De Luca, G.; Borriello, G.; De Domenico, A.; Brandi, S.; Cuomo, M.C.; Bove, F.; et al. Fatal Interstitial Pneumonia Associated with Bovine Coronavirus in Cows from Southern Italy. Viruses 2020, 12, 1331. https://doi.org/10.3390/v12111331
Amoroso MG, Lucifora G, Degli Uberti B, Serra F, De Luca G, Borriello G, De Domenico A, Brandi S, Cuomo MC, Bove F, et al. Fatal Interstitial Pneumonia Associated with Bovine Coronavirus in Cows from Southern Italy. Viruses. 2020; 12(11):1331. https://doi.org/10.3390/v12111331
Chicago/Turabian StyleAmoroso, Maria Grazia, Giuseppe Lucifora, Barbara Degli Uberti, Francesco Serra, Giovanna De Luca, Giorgia Borriello, Alessandro De Domenico, Sergio Brandi, Maria Concetta Cuomo, Francesca Bove, and et al. 2020. "Fatal Interstitial Pneumonia Associated with Bovine Coronavirus in Cows from Southern Italy" Viruses 12, no. 11: 1331. https://doi.org/10.3390/v12111331
APA StyleAmoroso, M. G., Lucifora, G., Degli Uberti, B., Serra, F., De Luca, G., Borriello, G., De Domenico, A., Brandi, S., Cuomo, M. C., Bove, F., Riccardi, M. G., Galiero, G., & Fusco, G. (2020). Fatal Interstitial Pneumonia Associated with Bovine Coronavirus in Cows from Southern Italy. Viruses, 12(11), 1331. https://doi.org/10.3390/v12111331





