Detection of SARS-CoV-2 from Saliva as Compared to Nasopharyngeal Swabs in Outpatients
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Saliva Collection
2.2. SARS-CoV-2 RNA Detection
2.3. Supplemental SARS-CoV-2 Testing of Positive Saliva Samples with Paired Negative NPS
2.4. Statistical Analysis
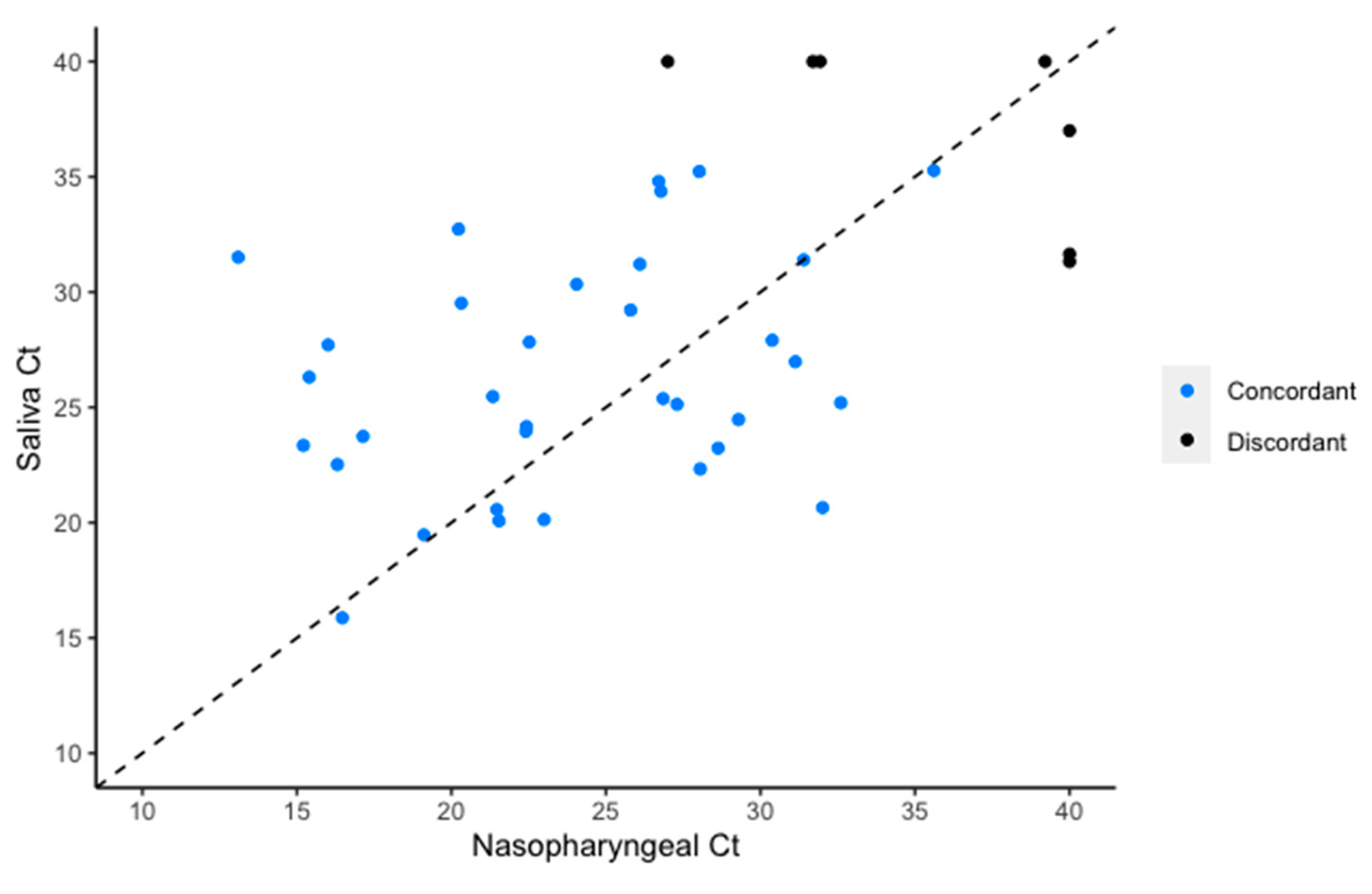
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Santosh, T.S.; Parmar, R.; Anand, H.; Srikanth, K.; Saritha, M. A Review of Salivary Diagnostics and Its Potential Implication in Detection of Covid-19. Cureus 2020, 12, e7708. [Google Scholar] [CrossRef]
- Hong, K.H.; Lee, S.W.; Kim, T.S.; Huh, H.J.; Lee, J.; Kim, S.Y.; Park, J.-S.; Kim, G.J.; Sung, H.; Roh, K.H.; et al. Guidelines for Laboratory Diagnosis of Coronavirus Disease 2019 (COVID-19) in Korea. Ann. Lab. Med. 2020, 40, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Azzi, L.; Carcano, G.; Gianfagna, F.; Grossi, P.; Gasperina, D.D.; Genoni, A.; Fasano, M.; Sessa, F.; Tettamanti, L.; Carinci, F.; et al. Saliva is a reliable tool to detect SARS-CoV-2. J. Infect. 2020, 81, e45–e50. [Google Scholar] [CrossRef]
- Khurshid, Z.; Zohaib, S.; Joshi, C.; Moin, S.F.; Zafar, M.S.; Speicher, D.J. Saliva as a non-invasive sample for the detection of SARS-CoV-2: A systematic review. medRxiv 2020. [Google Scholar] [CrossRef]
- Ott, I.; Simões, M.S.; Watkins, A.; Boot, M.; Kalinich, C.C.; Harden, C.A.; Wyllie, A.L.; Casanovas-Massana, A.; Moore, A.J.; Muenker, C.; et al. Simply saliva: Stability of SARS-CoV-2 detection negates the need for expensive collection devices. medRxiv 2020. [Google Scholar] [CrossRef]
- Becker, D.; Sandoval, E.; Amin, A.; De Hoff, P.; Diets, A.; Leonetti, N.; Lim, Y.W.; Elliott, C.; Laurent, L.; Grzymski, J.; et al. Saliva is less sensitive than nasopharyngeal swabs for COVID-19 detection in the community setting. medRxiv 2020. [Google Scholar] [CrossRef]
- Williams, E.; Bond, K.; Zhang, B.; Putland, M.; Williamson, D. Saliva as a Noninvasive Specimen for Detection of SARS-CoV-2. J. Clin. Microbiol. 2020, 58. [Google Scholar] [CrossRef] [PubMed]
- Caulley, L.; Corsten, M.; Eapen, L.; Whelan, B.J.; Angel, J.B.; Antonation, M.K.; Bastien, N.; Poliquin, G.; Johnson-Obaseki, S. Salivary Detection of COVID-19. Ann. Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wyllie, A.L.; Fournier, J.; Casanovas-Massana, A.; Campbell, M.; Tokuyama, M.; Vijayakumar, P.; Petrone, M.E. Faculty Opinions recommendation of Saliva is more sensitive for SARS-CoV-2 detection in COVID-19 patients than nasopharyngeal swabs. Faculty Opinions Post-Publication Peer Review of the Biomedical Literature 2020. Medrxiv 2020. [Google Scholar] [CrossRef]
- Peeters, E.; Singh, S.K.D.A.; Vandesompele, J.; Mestdagh, P.; Hutse, V.; Arbyn, M. Rapid systematic review of the sensitivity of SARS-CoV-2 molecular testing on saliva compared to nasopharyngeal swabs. medRxiv 2020. [Google Scholar] [CrossRef]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance 2020, 25, 2000045. [Google Scholar] [CrossRef] [PubMed]
- Vermeiren, C.; Marchand-Senécal, X.; Sheldrake, E.; Bulir, D.; Smieja, M.; Chong, S.; Forbes, J.D.; Katz, K. Comparison of Copan ESwab and FLOQSwab for COVID-19 Diagnosis: Working around a Supply Shortage. J. Clin. Microbiol. 2020, 58. [Google Scholar] [CrossRef] [PubMed]
- Czumbel, L.M.; Kiss, S.; Farkas, N.; Mandel, I.; Hegyi, A.; Ákos, N.; Lohinai, Z.; Szakács, Z.; Hegyi, P.; Steward, M.C.; et al. Saliva as a Candidate for COVID-19 Diagnostic Testing: A Meta-Analysis. Front. Med. 2020, 7, 465. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Rashid, F.A.; Sabri, F.S.A.H.; Jamil, N.N.; Zain, R.; Hashim, R.; Amran, F.; Kok, H.T.; Samad, A.A.; Ahmad, N. Comparing nasopharyngeal swab and early morning saliva for the identification of SARS-CoV-2. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Iwasaki, S.; Fujisawa, S.; Nakakubo, S.; Kamada, K.; Yamashita, Y.; Fukumoto, T.; Sato, K.; Oguri, S.; Taki, K.; Senjo, H.; et al. Comparison of SARS-CoV-2 detection in nasopharyngeal swab and saliva. J. Infect. 2020, 81, e145–e147. [Google Scholar] [CrossRef]
- To, K.K.-W.; Tsang, O.T.-Y.; Yip, C.C.-Y.; Chan, K.-H.; Wu, T.-C.; Chan, J.M.-C.; Leung, W.-S.; Chik, T.S.-H.; Choi, C.Y.-C.; Kandamby, D.H.; et al. Consistent Detection of 2019 Novel Coronavirus in Saliva. Clin. Infect. Dis. 2020, 71, 841–843. [Google Scholar] [CrossRef]
- Jamal, A.J.; Mozafarihashjin, M.; Coomes, E.; Powis, J.; Li, A.X.; Paterson, A.; Anceva-Sami, S.; Barati, S.; Crowl, G.; Faheem, A.; et al. Sensitivity of Nasopharyngeal Swabs and Saliva for the Detection of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 2020, 25, ciaa848. [Google Scholar] [CrossRef]
- Wyllie, A.L.; Fournier, J.; Casanovas-Massana, A.; Campbell, M.; Tokuyama, M.; Vijayakumar, P.; Warren, J.L.; Geng, B.; Muenker, M.C.; Moore, A.J.; et al. Saliva or Nasopharyngeal Swab Specimens for Detection of SARS-CoV-2. N. Engl. J. Med. 2020, 383, 1283–1286. [Google Scholar] [CrossRef]
- Liu, Y.; Yan, L.-M.; Wan, L.; Xiang, T.-X.; Le, A.; Liu, J.-M.; Peiris, M.; Poon, L.L.M.; Zhang, W. Viral dynamics in mild and severe cases of COVID-19. Lancet Infect. Dis. 2020, 20, 656–657. [Google Scholar] [CrossRef]
- Long, Q.; Tang, X.-J.; Shi, Q.-L.; Li, Q.; Deng, H.-J.; Yuan, J.; Hu, J.-L.; Xu, W.; Zhang, Y.; Lv, F.-J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus–Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061. [Google Scholar] [CrossRef] [PubMed]
| Saliva Sample | Nasopharyngeal Swab | Total | |
|---|---|---|---|
| Positive | Negative | ||
| Positive | 39 | 3 | 42 |
| Negative | 4 | 383 | 387 |
| Total | 43 | 386 | 429 |
| Results | NPS CT Values | Saliva CT Values | ||||||
|---|---|---|---|---|---|---|---|---|
| NPS | Saliva | Target 1 * | Target 2 † | Internal Control | Target 1 ‡ | Target 2 § | Internal Control | |
| Sample 1 | positive | negative | 27.0 | 26.8 | 25.1 | ND | ND | 35.3 |
| Sample 2 | positive | negative | 39.2 | 38.9 | 26.1 | ND | ND | 35.0 |
| Sample 3 | positive | negative | 31.7 | 33.5 | 21.2 | ND | ND | 34.5 |
| Sample 4 | positive | negative | 31.9 | 31.8 | 20.7 | ND | ND | 22.5 |
| Sample 5 | negative | positive | ND | ND | 21.5 | ND | 37.0 | 36.8 |
| Sample 6 | negative | positive | ND | ND | 24.2 | 29.4 | 31.7 | 38.6 |
| Sample 7 | negative | positive | ND | ND | 27.6 | 30.1 | 31.3 | 36.2 |
| Room Temperature (18–24 ℃) | |||||||||||||
| Saliva Sample | Day 0 (Ct) | Day 3 (Ct) | Day7 (Ct) | Day 3 Δ (Ct) | Day 7 Δ (Ct) | ||||||||
| ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | ORF1a/b | E gene | |
| S01 | 24.4 | 25.2 | 36.1 | 24.9 | 25.7 | 37.0 | 25.5 | 26.4 | 36.9 | 0.5 | 0.4 | 1.1 | 1.2 |
| S02 | 25.3 | 26.0 | 36.9 | 24.0 | 24.7 | 36.2 | 24.5 | 25.5 | 37.1 | −1.4 | −1.3 | −0.8 | −0.6 |
| S03 | 24.2 | 25.0 | 35.7 | 21.8 | 22.7 | 34.0 | 22.6 | 23.3 | 33.1 | −2.4 | −2.3 | −1.6 | −1.7 |
| S04 | 23.4 | 24.1 | 35.2 | 22.4 | 23.3 | 35.9 | 23.9 | 24.6 | 36.1 | −1.0 | −0.8 | 0.5 | 0.5 |
| S05 | 24.7 | 25.5 | 36.1 | 23.1 | 23.9 | 36.1 | 23.1 | 23.8 | 35.2 | −1.7 | −1.7 | −1.7 | −1.7 |
| S06 | 24.5 | 25.3 | 37.1 | 24.4 | 25.1 | 37.1 | 26.6 | 27.3 | 36.8 | −0.2 | −0.1 | 2.1 | 2.0 |
| S07 | 23.4 | 23.8 | 36.0 | 22.9 | 23.8 | 34.1 | 21.9 | 22.5 | 33.8 | −0.5 | 0.0 | −1.5 | −1.3 |
| S08 | 23.5 | 24.4 | 35.5 | 24.0 | 24.9 | 35.2 | 24.4 | 25.2 | 34.8 | 0.4 | 0.5 | 0.9 | 0.8 |
| S09 | 23.6 | 24.1 | 34.5 | 22.2 | 22.9 | 33.6 | 22.6 | 23.5 | 33.4 | −1.5 | −1.2 | −1.0 | −0.6 |
| Refrigerated (2–8 ℃) | |||||||||||||
| Saliva Sample | Day 0 | Day3 | Day7 | Day 3 Δ | Day 7 Δ | ||||||||
| ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | ORF1a/b | E gene | |
| S01 | 24.6 | 25.4 | 36.8 | 24.7 | 25.5 | 35.7 | 26.4 | 27.1 | 36.3 | 0.2 | 0.1 | 1.8 | 1.7 |
| S02 | 25.6 | 26.3 | 38.7 | 22.8 | 23.8 | 34.2 | 25.3 | 26.0 | 36.9 | −2.8 | −2.5 | −0.3 | −0.3 |
| S03 | 24.0 | 24.6 | 35.6 | 24.0 | 24.9 | 35.6 | 23.1 | 23.9 | 35.1 | 0.1 | 0.3 | −0.9 | −0.7 |
| S04 | 23.8 | 24.4 | 37.7 | 23.5 | 24.3 | 34.6 | 22.9 | 23.7 | 35.7 | −0.3 | −0.1 | −0.8 | −0.7 |
| S05 | 25.1 | 25.9 | 35.6 | 24.6 | 25.3 | 36.5 | 24.4 | 25.1 | 36.6 | −0.5 | −0.6 | −0.7 | −0.8 |
| S06 | 24.1 | 25.0 | 36.1 | 25.3 | 26.0 | 36.6 | 25.7 | 26.6 | 37.7 | 1.2 | 1.0 | 1.6 | 1.6 |
| S07 | 25.6 | 26.1 | 35.1 | 23.2 | 23.8 | 35.0 | 23.2 | 23.8 | 34.7 | −2.4 | −2.3 | −2.4 | −2.3 |
| S08 | 25.5 | 26.5 | 36.2 | 24.5 | 25.4 | 35.0 | 26.2 | 26.9 | 36.7 | −1.0 | −1.0 | 0.7 | 0.4 |
| S09 | 23.3 | 23.9 | 35.1 | 23.5 | 24.2 | 37.7 | 23.6 | 24.4 | 33.9 | 0.1 | 0.4 | 0.3 | 0.5 |
| Frozen (−20 ℃) | |||||||||||||
| Day 0 ORF1a/b | Day3 | Day7 | Day 3 Δ | Day 7 Δ | |||||||||
| E gene | Internal Control | ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | Internal Control | ORF1a/b | E gene | ORF1a/b | E gene | ||
| S01 | 24.6 | 25.5 | 36.3 | 24.2 | 25.1 | 36.3 | 26.0 | 26.7 | 35.9 | −0.4 | −0.3 | 1.4 | 1.3 |
| S02 | 25.4 | 26.2 | 35.4 | 25.4 | 26.2 | 38.1 | 25.7 | 26.5 | 36.9 | 0.0 | 0.0 | 0.3 | 0.3 |
| S03 | 25.6 | 26.5 | 35.1 | 25.1 | 26.1 | 34.7 | 25.8 | 26.7 | 35.2 | −0.5 | −0.3 | 0.2 | 0.2 |
| S04 | 23.9 | 24.6 | 35.4 | 23.2 | 24.0 | 36.0 | 23.6 | 24.4 | 34.4 | −0.7 | −0.6 | −0.3 | −0.2 |
| S05 | 25.6 | 26.4 | 36.9 | 23.9 | 24.6 | 36.2 | 27.5 | 28.4 | 36.7 | −1.7 | −1.8 | 1.9 | 2.0 |
| S06 | 24.7 | 25.4 | 36.1 | 23.6 | 24.5 | 35.8 | 27.2 | 28.1 | 36.1 | −1.1 | −0.9 | 2.6 | 2.8 |
| S07 | 24.8 | 25.3 | 35.3 | 24.5 | 25.2 | 34.3 | 26.9 | 27.7 | 36.1 | −0.3 | −0.2 | 2.1 | 2.4 |
| S08 | 24.7 | 25.7 | 35.1 | 24.8 | 25.9 | 36.7 | 27.6 | 28.6 | 36.1 | 0.1 | 0.2 | 2.9 | 3.0 |
| S09 | 24.2 | 24.7 | 35.9 | 22.7 | 23.6 | 34.1 | 25.1 | 26.1 | 34.1 | −1.4 | −1.1 | 1.0 | 1.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kandel, C.; Zheng, J.; McCready, J.; Serbanescu, M.A.; Racher, H.; Desaulnier, M.; Powis, J.E.; Vojdani, K.; Finlay, L.; Sheldrake, E.; et al. Detection of SARS-CoV-2 from Saliva as Compared to Nasopharyngeal Swabs in Outpatients. Viruses 2020, 12, 1314. https://doi.org/10.3390/v12111314
Kandel C, Zheng J, McCready J, Serbanescu MA, Racher H, Desaulnier M, Powis JE, Vojdani K, Finlay L, Sheldrake E, et al. Detection of SARS-CoV-2 from Saliva as Compared to Nasopharyngeal Swabs in Outpatients. Viruses. 2020; 12(11):1314. https://doi.org/10.3390/v12111314
Chicago/Turabian StyleKandel, Christopher, Jennifer Zheng, Janine McCready, Mihaela Anca Serbanescu, Hilary Racher, Melissa Desaulnier, Jeff E Powis, Kyle Vojdani, Laura Finlay, Elena Sheldrake, and et al. 2020. "Detection of SARS-CoV-2 from Saliva as Compared to Nasopharyngeal Swabs in Outpatients" Viruses 12, no. 11: 1314. https://doi.org/10.3390/v12111314
APA StyleKandel, C., Zheng, J., McCready, J., Serbanescu, M. A., Racher, H., Desaulnier, M., Powis, J. E., Vojdani, K., Finlay, L., Sheldrake, E., Vermeiren, C., Katz, K., McGeer, A., Kozak, R., & Goneau, L. W. (2020). Detection of SARS-CoV-2 from Saliva as Compared to Nasopharyngeal Swabs in Outpatients. Viruses, 12(11), 1314. https://doi.org/10.3390/v12111314






