Marek’s Disease Virus Cluster 3 miRNAs Restrict Virus’ Early Cytolytic Replication and Pathogenesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells
2.2. Construction of Cluster 3 miRNA Deletion and Revertant Viruses
2.3. In Vitro Growth Kinetics
2.4. Indirect Immunofluorescence Assay (IFA) and MDV Plaque Size Measurement
2.5. Immunohistochemistry (IHC) Assay
2.6. Nucleic Acid Isolation and Quantitative Polymerase Chain Reaction (qPCR)
2.7. Gene Expression Analysis
2.8. Virus Reactivation Assay
2.9. Evaluation of 686BAC ∆miR-C3 Pathogenesis in Chickens
2.10. Lymphoid Organ Atrophy
2.11. Pathogenesis Study
2.12. Data and Statistical Analysis
3. Results
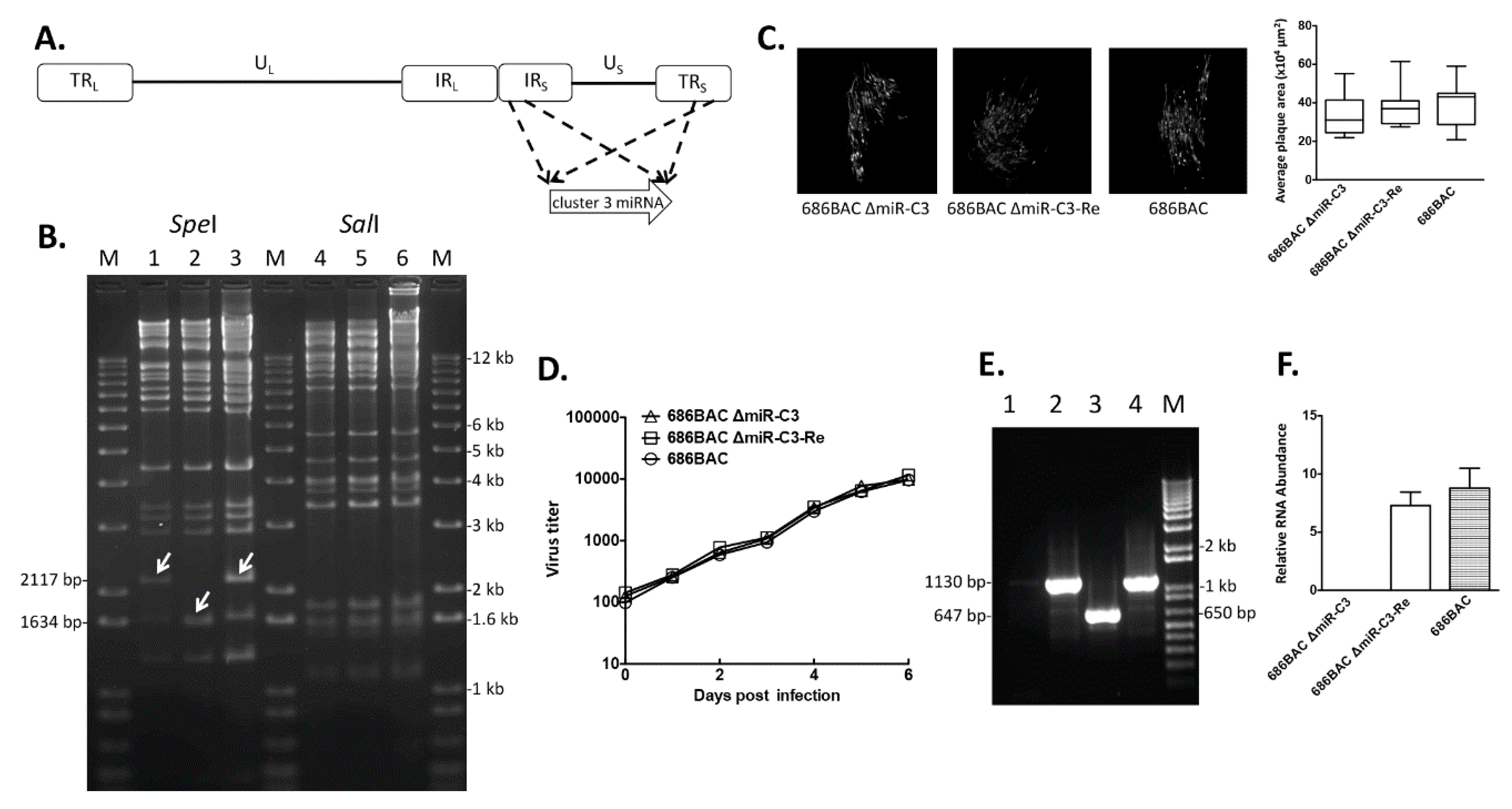
3.1. Construction and In Vitro Characterization of Cluster 3 miRNA Deletion and Revertant Viruses
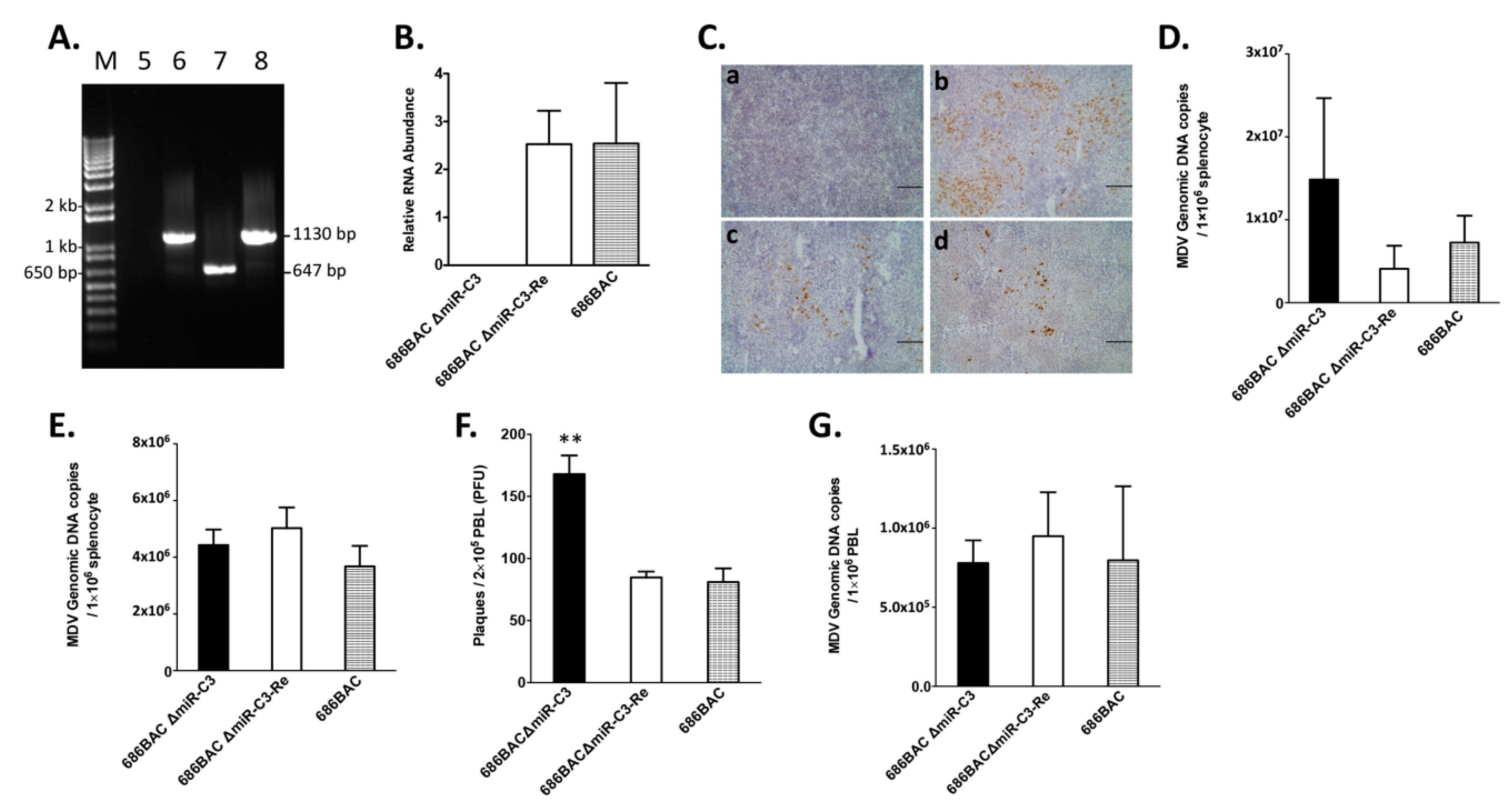
3.2. Deletion of Cluster 3 miRNAs Increases Viral Load during Early Cytolytic Phase
3.3. Deletion of Cluster 3 miRNAs Significantly Increases Virus Reactivation from Peripheral Blood Lymphocytes (PBL)
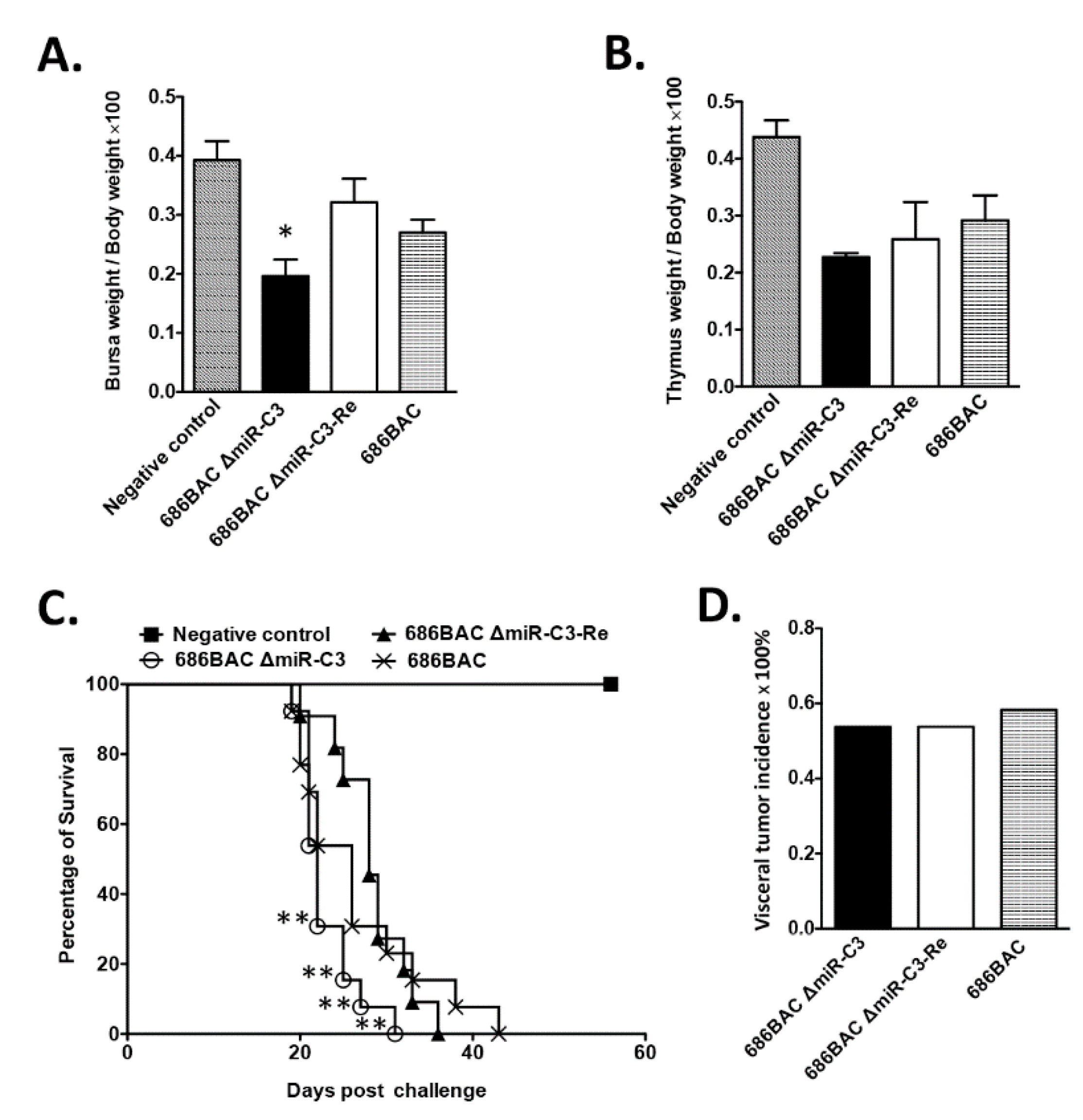
3.4. Deletion of Cluster 3 miRNAs Modulates MDV Pathogenesis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Cullen, B.R. Five questions about viruses and microRNAs. PLoS Pathog. 2010, 6, e1000787. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, R.P.; Sullivan, C.S. Virus-encoded microRNAs: An overview and a look to the future. PLoS Pathog. 2012, 8, e1003018. [Google Scholar] [CrossRef] [PubMed]
- Skalsky, R.L.; Cullen, B.R. Viruses, microRNAs, and host interactions. Annu. Rev. Microbiol. 2010, 64, 123–141. [Google Scholar] [CrossRef]
- Cai, X.; Lu, S.; Zhang, Z.; Gonzalez, C.M.; Damania, B.; Cullen, B.R. Kaposi’s sarcoma-associated herpesvirus expresses an array of viral microRNAs in latently infected cells. Proc. Natl. Acad. Sci. USA 2005, 102, 5570–5575. [Google Scholar] [CrossRef]
- Cui, C.; Griffiths, A.; Li, G.; Silva, L.M.; Kramer, M.F.; Gaasterland, T.; Wang, X.J.; Coen, D.M. Prediction and identification of herpes simplex virus 1-encoded microRNAs. J. Virol. 2006, 80, 5499–5508. [Google Scholar] [CrossRef]
- Yao, Y.; Zhao, Y.; Xu, H.; Smith, L.P.; Lawrie, C.H.; Watson, M.; Nair, V. MicroRNA profile of Marek’s disease virus-transformed T-cell line MSB-1: Predominance of virus-encoded microRNAs. J. Virol. 2008, 82, 4007–4015. [Google Scholar] [CrossRef]
- Cosmopoulos, K.; Pegtel, M.; Hawkins, J.; Moffett, H.; Novina, C.; Middeldorp, J.; Thorley-Lawson, D.A. Comprehensive profiling of Epstein-Barr virus microRNAs in nasopharyngeal carcinoma. J. Virol. 2009, 83, 2357–2367. [Google Scholar] [CrossRef]
- Feederle, R.; Linnstaedt, S.D.; Bannert, H.; Lips, H.; Bencun, M.; Cullen, B.R.; Delecluse, H.J. A viral microRNA cluster strongly potentiates the transforming properties of a human herpesvirus. PLoS Pathog. 2011, 7, e1001294. [Google Scholar] [CrossRef]
- Samols, M.A.; Hu, J.; Skalsky, R.L.; Renne, R. Cloning and identification of a microRNA cluster within the latency-associated region of Kaposi’s sarcoma-associated herpesvirus. J. Virol. 2005, 79, 9301–9305. [Google Scholar] [CrossRef]
- Feederle, R.; Haar, J.; Bernhardt, K.; Linnstaedt, S.D.; Bannert, H.; Lips, H.; Cullen, B.R.; Delecluse, H.J. The members of an Epstein-Barr virus microRNA cluster cooperate to transform B lymphocytes. J. Virol. 2011, 85, 9801–9810. [Google Scholar] [CrossRef] [PubMed]
- Calnek, B.W. Pathogenesis of Marek’s disease virus infection. In Marek’s Disease; Current Topics in Microbiology and Immunology; Springer: Berlin, Germany, 2001; Volume 255, pp. 25–55. [Google Scholar]
- Lupiani, B.; Lee, L.F.; Cui, X.; Gimeno, I.; Anderson, A.; Morgan, R.W.; Silva, R.F.; Witter, R.L.; Kung, H.J.; Reddy, S.M. Marek’s disease virus-encoded Meq gene is involved in transformation of lymphocytes but is dispensable for replication. Proc. Natl. Acad. Sci. USA 2004, 101, 11815–11820. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.M.; Lupiani, B.; Gimeno, I.M.; Silva, R.F.; Lee, L.F.; Witter, R.L. Rescue of a pathogenic Marek’s disease virus with overlapping cosmid DNAs: Use of a pp38 mutant to validate the technology for the study of gene function. Proc. Natl. Acad. Sci. USA 2002, 99, 7054–7059. [Google Scholar] [CrossRef] [PubMed]
- Tahiri-Alaoui, A.; Smith, L.P.; Kgosana, L.; Petherbridge, L.J.; Nair, V. Identification of a neurovirulence factor from Marek’s disease virus. Avian Dis. 2013, 7 (Suppl. 2), 387–394. [Google Scholar] [CrossRef]
- Kamil, J.P.; Tischer, B.K.; Trapp, S.; Nair, V.K.; Osterrieder, N.; Kung, H.J. vLIP, a viral lipase homologue, is a virulence factor of Marek’s disease virus. J. Virol. 2005, 79, 6984–6996. [Google Scholar] [CrossRef]
- Jarosinski, K.W.; Osterrieder, N.; Nair, V.K.; Schat, K.A. Attenuation of Marek’s disease virus by deletion of open reading frame RLORF4 but not RLORF5a. J. Virol. 2005, 79, 11647–11659. [Google Scholar] [CrossRef]
- Parcells, M.S.; Lin, S.F.; Dienglewicz, R.L.; Majerciak, V.; Robinson, D.R.; Chen, H.C.; Wu, Z.; Dubyak, G.R.; Brunovskis, P.; Hunt, H.D.; et al. Marek’s disease virus (MDV) encodes an interleukin-8 homolog (vIL-8): Characterization of the vIL-8 protein and a vIL-8 deletion mutant MDV. J. Virol. 2001, 75, 5159–5173. [Google Scholar] [CrossRef]
- Cui, X.; Lee, L.F.; Reed, W.M.; Kung, H.J.; Reddy, S.M. Marek’s disease virus-encoded vIL-8 gene is involved in early cytolytic infection but dispensable for establishment of latency. J. Virol. 2004, 78, 4753–4760. [Google Scholar] [CrossRef]
- Trapp, S.; Parcells, M.S.; Kamil, J.P.; Schumacher, D.; Tischer, B.K.; Kumar, P.M.; Nair, V.K.; Osterrieder, N. A virus-encoded telomerase RNA promotes malignant T cell lymphomagenesis. J. Exp. Med. 2006, 203, 1307–1317. [Google Scholar] [CrossRef]
- Kaufer, B.B.; Trapp, S.; Jarosinski, K.W.; Osterrieder, N. Herpesvirus telomerase RNA(vTR)-dependent lymphoma formation does not require interaction of vTR with telomerase reverse transcriptase (TERT). PLoS Pathog. 2010, 6, e1001073. [Google Scholar] [CrossRef]
- Burnside, J.; Bernberg, E.; Anderson, A.; Lu, C.; Meyers, B.C.; Green, P.J.; Jain, N.; Isaacs, G.; Morgan, R.W. Marek’s disease virus encodes MicroRNAs that map to meq and the latency-associated transcript. J. Virol. 2006, 80, 8778–8786. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xu, H.; Yao, Y.; Smith, L.P.; Kgosana, L.; Green, J.; Petherbridge, L.; Baigent, S.J.; Nair, V. Critical role of the virus-encoded microRNA-155 ortholog in the induction of Marek’s disease lymphomas. PLoS Pathog. 2011, 7, e1001305. [Google Scholar] [CrossRef] [PubMed]
- Strassheim, S.; Stik, G.; Rasschaert, D.; Laurent, S. mdv1-miR-M7-5p, located in the newly identified first intron of the latency-associated transcript of Marek’s disease virus, targets the immediate-early genes ICP4 and ICP27. J. Gen. Virol. 2012, 93 Pt 8, 1731–1742. [Google Scholar] [CrossRef]
- Reddy, S.M.; Sun, A.; Khan, O.A.; Lee, L.F.; Lupiani, B. Cloning of a very virulent plus, 686 strain of Marek’s disease virus as a bacterial artificial chromosome. Avian Dis. 2013, 57 (Suppl. 2), 469–473. [Google Scholar] [CrossRef] [PubMed]
- Tischer, B.K.; von Einem, J.; Kaufer, B.; Osterrieder, N. Two-step red-mediated recombination for versatile high-efficiency markerless DNA manipulation in Escherichia coli. Biotechniques 2006, 40, 191–197. [Google Scholar] [PubMed]
- Liao, Y.; Lupiani, B.; Bajwa, K.; Khan, O.A.; Izumiya, Y.; Reddy, S.M. Role of Marek’s Disease Virus (MDV)-Encoded US3 Serine/Threonine Protein Kinase in Regulating MDV Meq and Cellular CREB Phosphorylation. J. Virol. 2020, 94. [Google Scholar] [CrossRef]
- Berthault, C.; Larcher, T.; Hartle, S.; Vautherot, J.F.; Trapp-Fragnet, L.; Denesvre, C. Atrophy of primary lymphoid organs induced by Marek’s disease virus during early infection is associated with increased apoptosis, inhibition of cell proliferation and a severe B-lymphopenia. Vet. Res. 2018, 49, 31. [Google Scholar] [CrossRef]
- Witter, R.L.; Calnek, B.W.; Buscaglia, C.; Gimeno, I.M.; Schat, K.A. Classification of Marek’s disease viruses according to pathotype: Philosophy and methodology. Avian Pathol. J. Wvpa 2005, 34, 75–90. [Google Scholar] [CrossRef]
- Faiz, N.M.; Cortes, A.L.; Guy, J.S.; Fletcher, O.J.; Cimino, T.; Gimeno, I.M. Evaluation of factors influencing the development of late Marek’s disease virus-induced immunosuppression: Virus pathotype and host sex. Avian Pathol. 2017, 46, 376–385. [Google Scholar] [CrossRef]
- Gimeno, I.M.; Witter, R.L.; Hunt, H.D.; Reddy, S.M.; Neumann, U. Differential attenuation of the induction by Marek’s disease virus of transient paralysis and persistent neurological disease: A model for pathogenesis studies. Avian Pathol. 2001, 30, 397–409. [Google Scholar] [CrossRef]
- Gimeno, I.M.; Witter, R.L.; Hunt, H.D.; Reddy, S.M.; Lee, L.F.; Silva, R.F. The pp38 gene of Marek’s disease virus (MDV) is necessary for cytolytic infection of B cells and maintenance of the transformed state but not for cytolytic infection of the feather follicle epithelium and horizontal spread of MDV. J. Virol. 2005, 79, 4545–4549. [Google Scholar] [CrossRef] [PubMed]
- Perng, G.C.; Jones, C. Towards an understanding of the herpes simplex virus type 1 latency-reactivation cycle. Interdiscip. Perspect. Infect. Dis. 2010, 2010, 262415. [Google Scholar] [CrossRef] [PubMed]
- Bellare, P.; Ganem, D. Regulation of KSHV lytic switch protein expression by a virus-encoded microRNA: An evolutionary adaptation that fine-tunes lytic reactivation. Cell Host Microbe 2009, 6, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Ziegelbauer, J.M.; Sullivan, C.S.; Ganem, D. Tandem array-based expression screens identify host mRNA targets of virus-encoded microRNAs. Nat. Genet. 2009, 41, 130–134. [Google Scholar] [CrossRef]
| Primer Name | Sequence (5′ to 3′) | Purpose |
|---|---|---|
| Cluster 3 KanR-F | GGAATAAACGTTGTGATACGCGATCGAGTTTTCGTGGCATATTCCTACGGAGGATGACGACGATAAGTAGGG | Forward primer for amplification of KanR cassette gene with MDV sequences flanking MDV cluster 3 miRNAs |
| Cluster 3 KanR-R | CGTTTACTTCCTAAGTCATCGCTCTTTAGTTGGGAGGAAAGTTTCCTAGACCGTAGGAATATGCCACGAAAACTCGATCGCGTATCACAACGTTTATTCCCAACCAATTAACCAATTCTGATTAG | Reverse primer for amplification of KanR cassette gene with MDV sequences flanking MDV cluster 3 miRNAs |
| Cluster 3-F | GTCCTCGTTGAATAGATGA | Amplification for MDV cluster 3 miRNAs with its flanking sequences of MDV |
| Cluster 3-R | AGGGGCGCATATACAGTC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, Y.; Zhuang, G.; Sun, A.; Khan, O.A.; Lupiani, B.; Reddy, S.M. Marek’s Disease Virus Cluster 3 miRNAs Restrict Virus’ Early Cytolytic Replication and Pathogenesis. Viruses 2020, 12, 1317. https://doi.org/10.3390/v12111317
Liao Y, Zhuang G, Sun A, Khan OA, Lupiani B, Reddy SM. Marek’s Disease Virus Cluster 3 miRNAs Restrict Virus’ Early Cytolytic Replication and Pathogenesis. Viruses. 2020; 12(11):1317. https://doi.org/10.3390/v12111317
Chicago/Turabian StyleLiao, Yifei, Guoqing Zhuang, Aijun Sun, Owais A. Khan, Blanca Lupiani, and Sanjay M. Reddy. 2020. "Marek’s Disease Virus Cluster 3 miRNAs Restrict Virus’ Early Cytolytic Replication and Pathogenesis" Viruses 12, no. 11: 1317. https://doi.org/10.3390/v12111317
APA StyleLiao, Y., Zhuang, G., Sun, A., Khan, O. A., Lupiani, B., & Reddy, S. M. (2020). Marek’s Disease Virus Cluster 3 miRNAs Restrict Virus’ Early Cytolytic Replication and Pathogenesis. Viruses, 12(11), 1317. https://doi.org/10.3390/v12111317





