Viral Equine Encephalitis, a Growing Threat to the Horse Population in Europe?
Abstract
1. Introduction
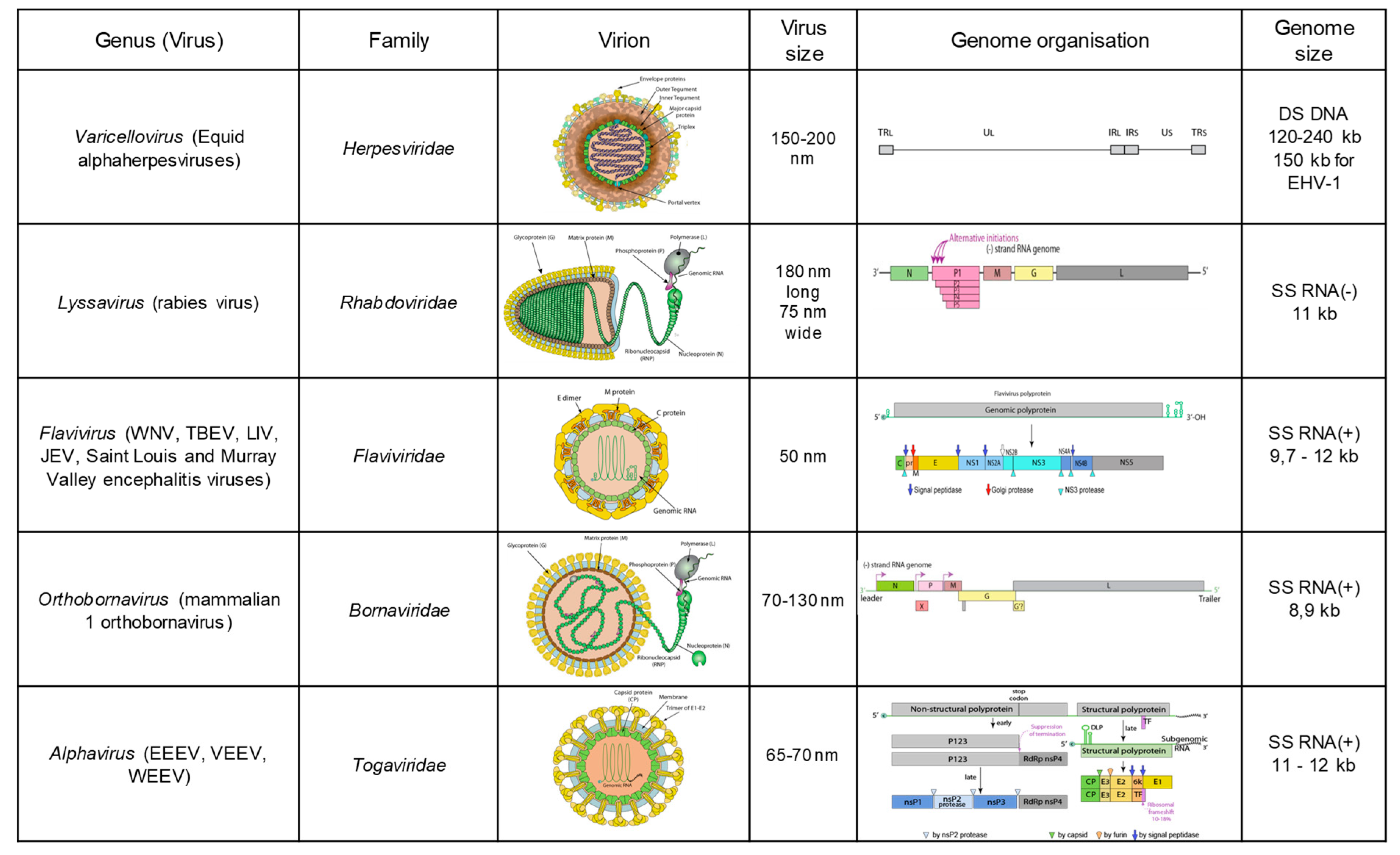
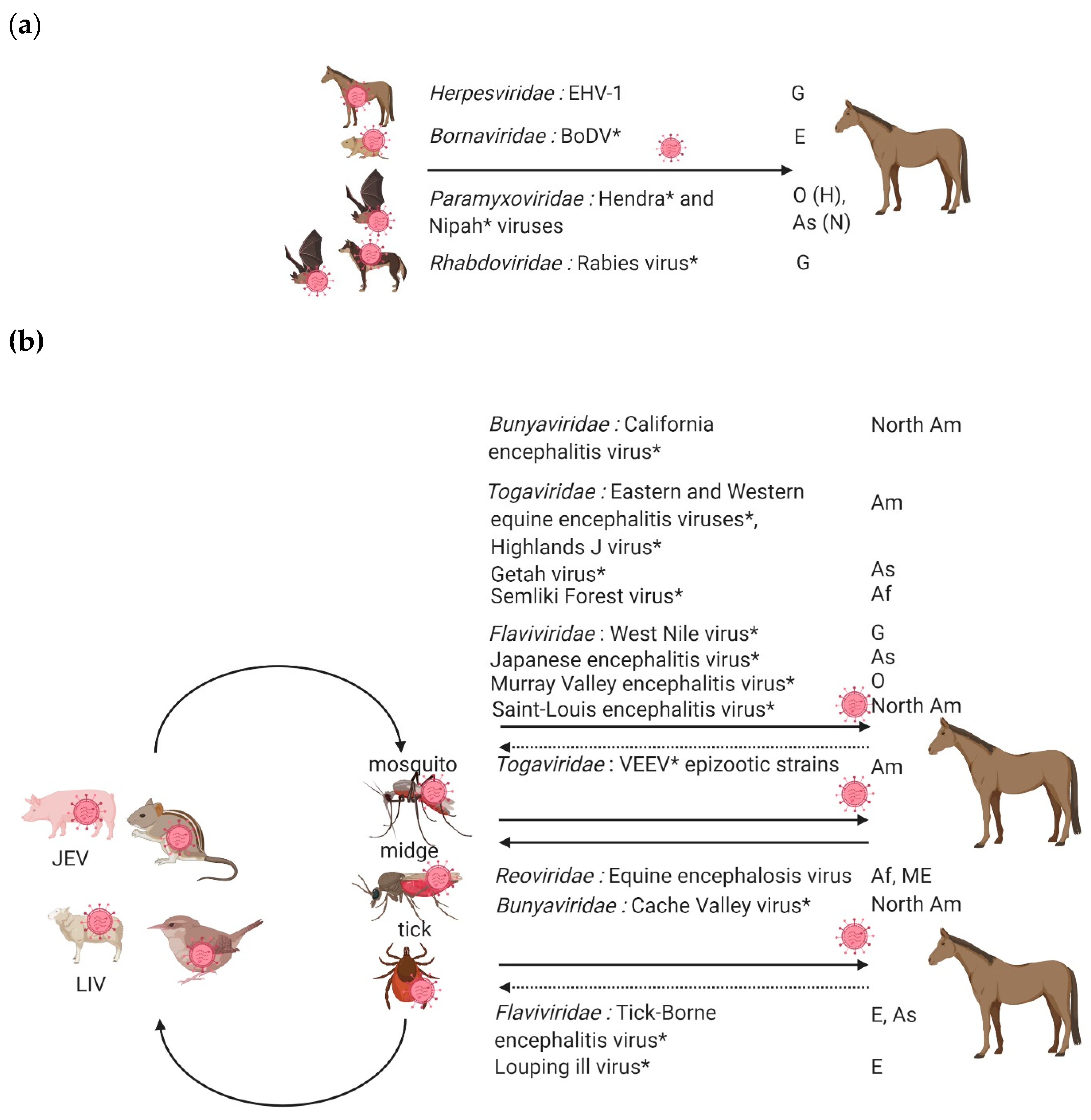
2. Equine Encephalitis Viruses Enzootic in Europe
2.1. Equid Herpesviruses
2.2. Rabies Virus
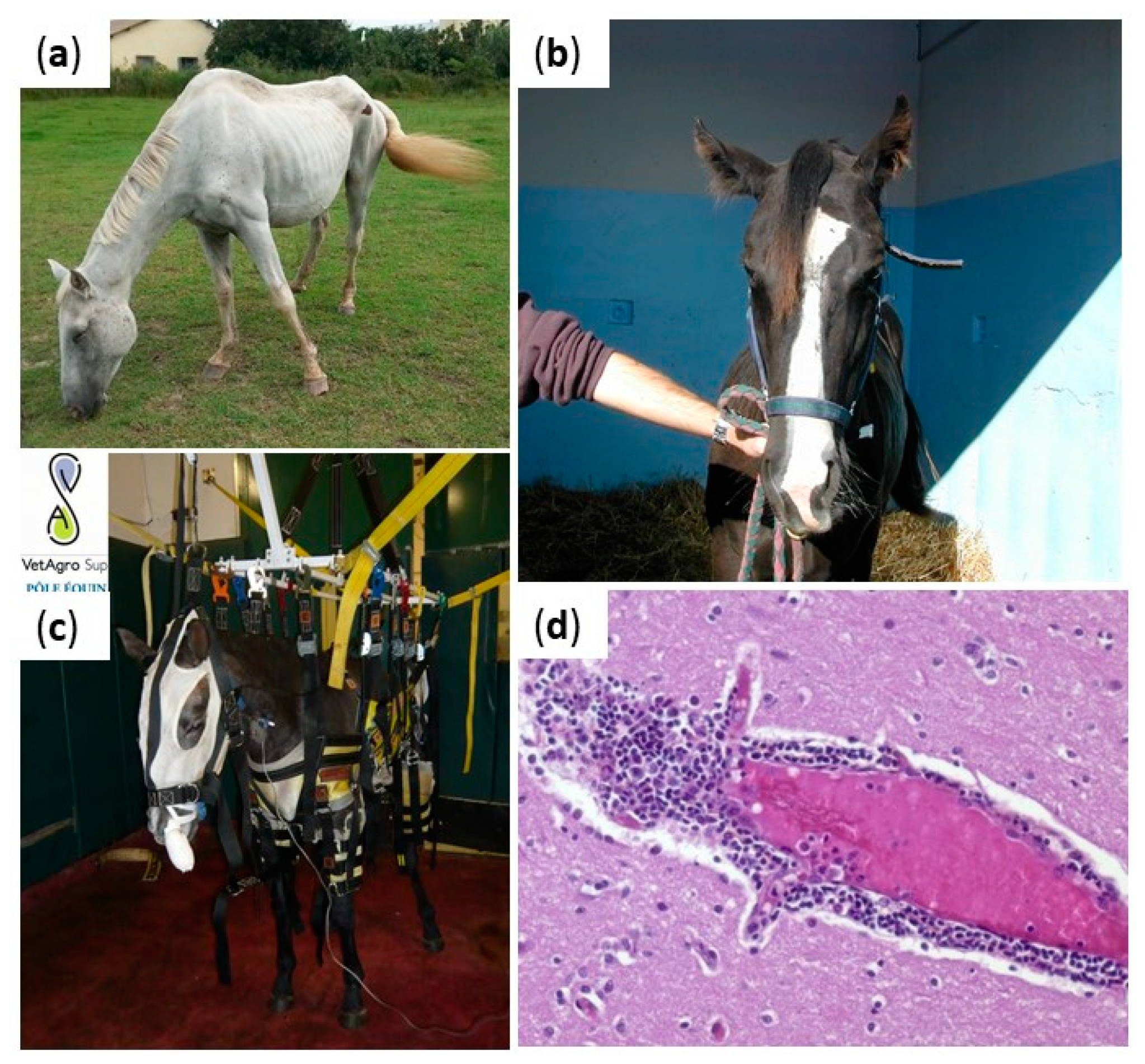
2.3. Borna Disease Virus
2.4. Enzootic Flaviviruses: West Nile Virus, Tick-Borne Encephalitis Virus and Louping Ill Virus
Transmission and Epidemiology
WNV
TBEV and LIV
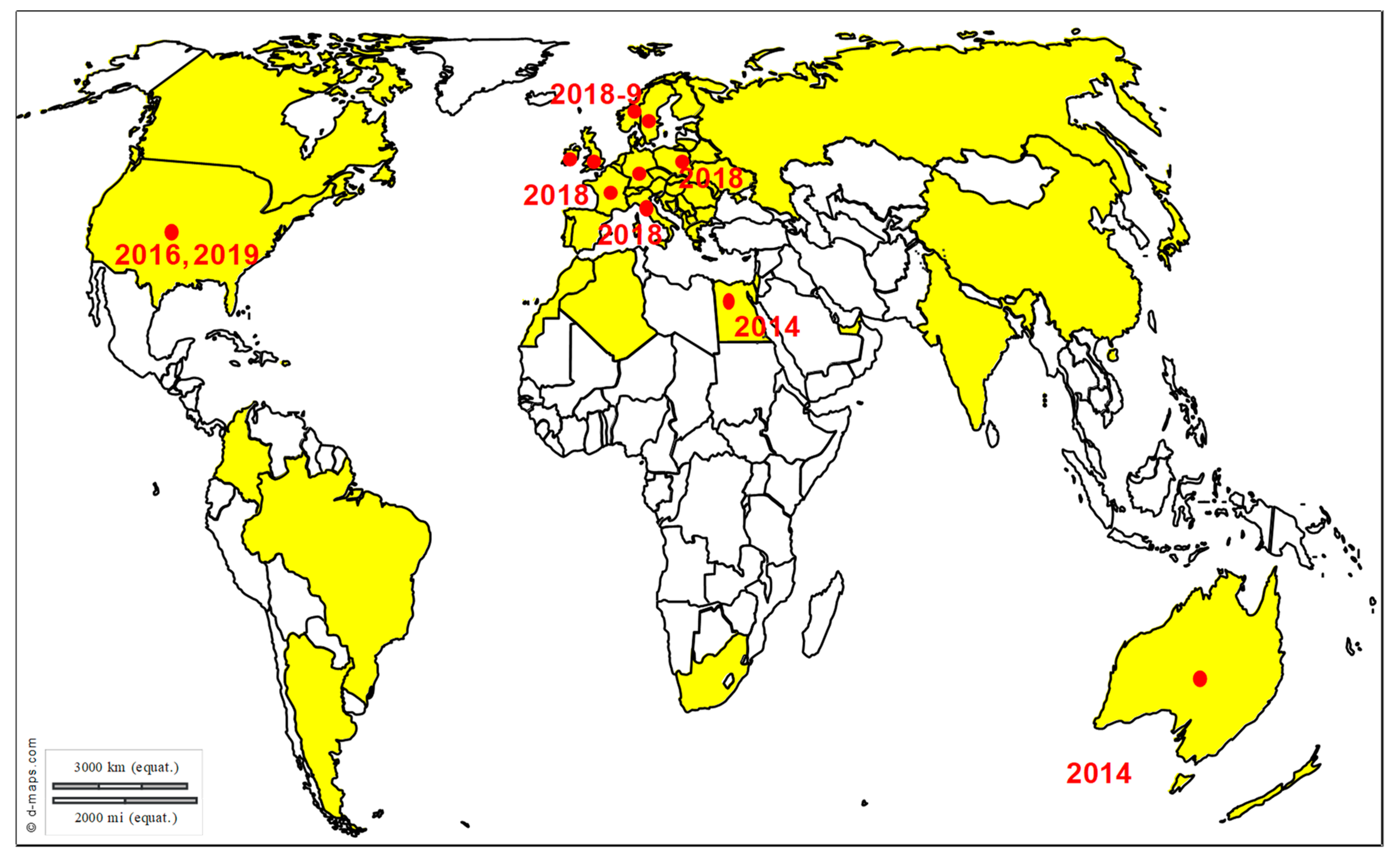
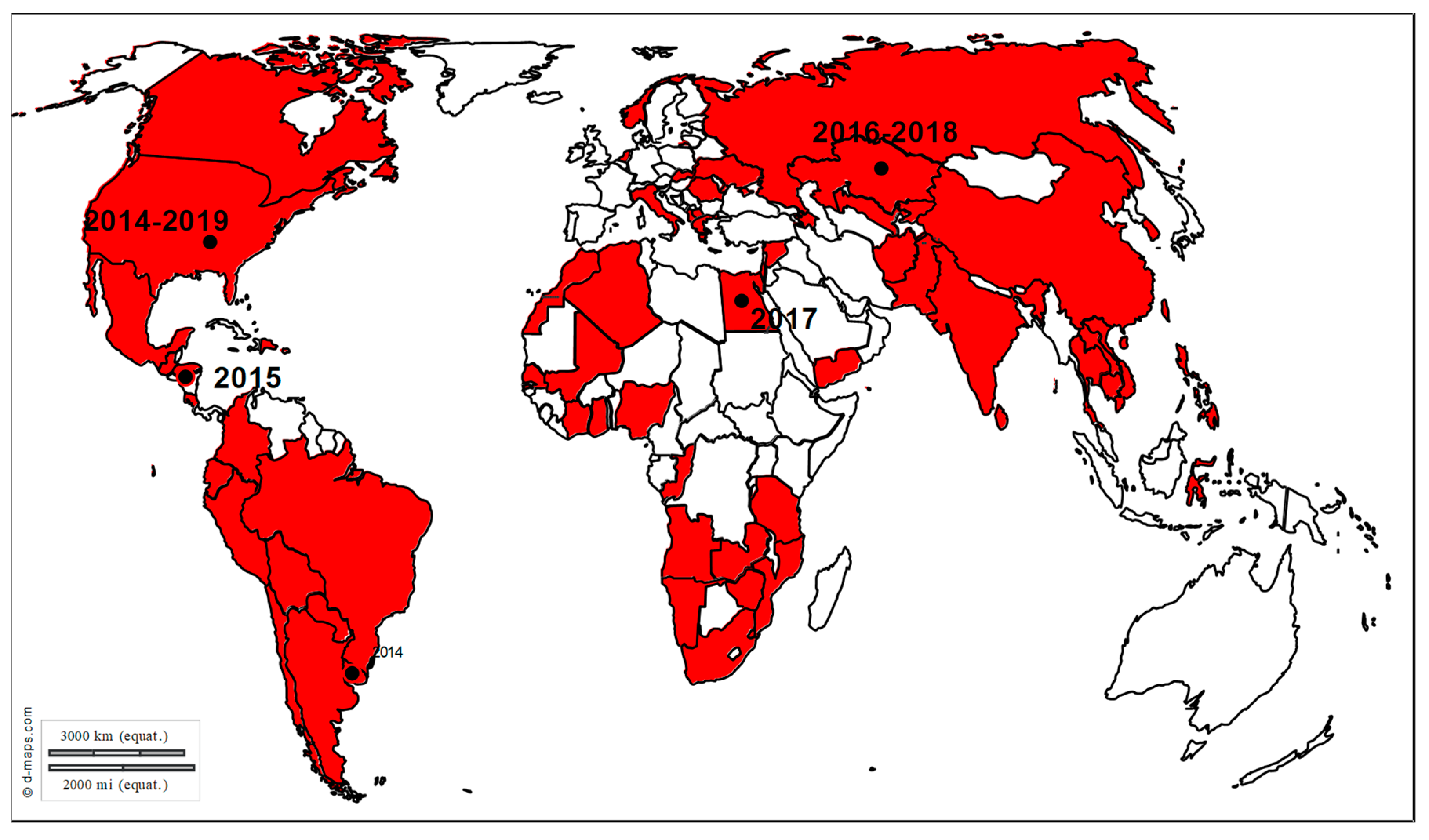
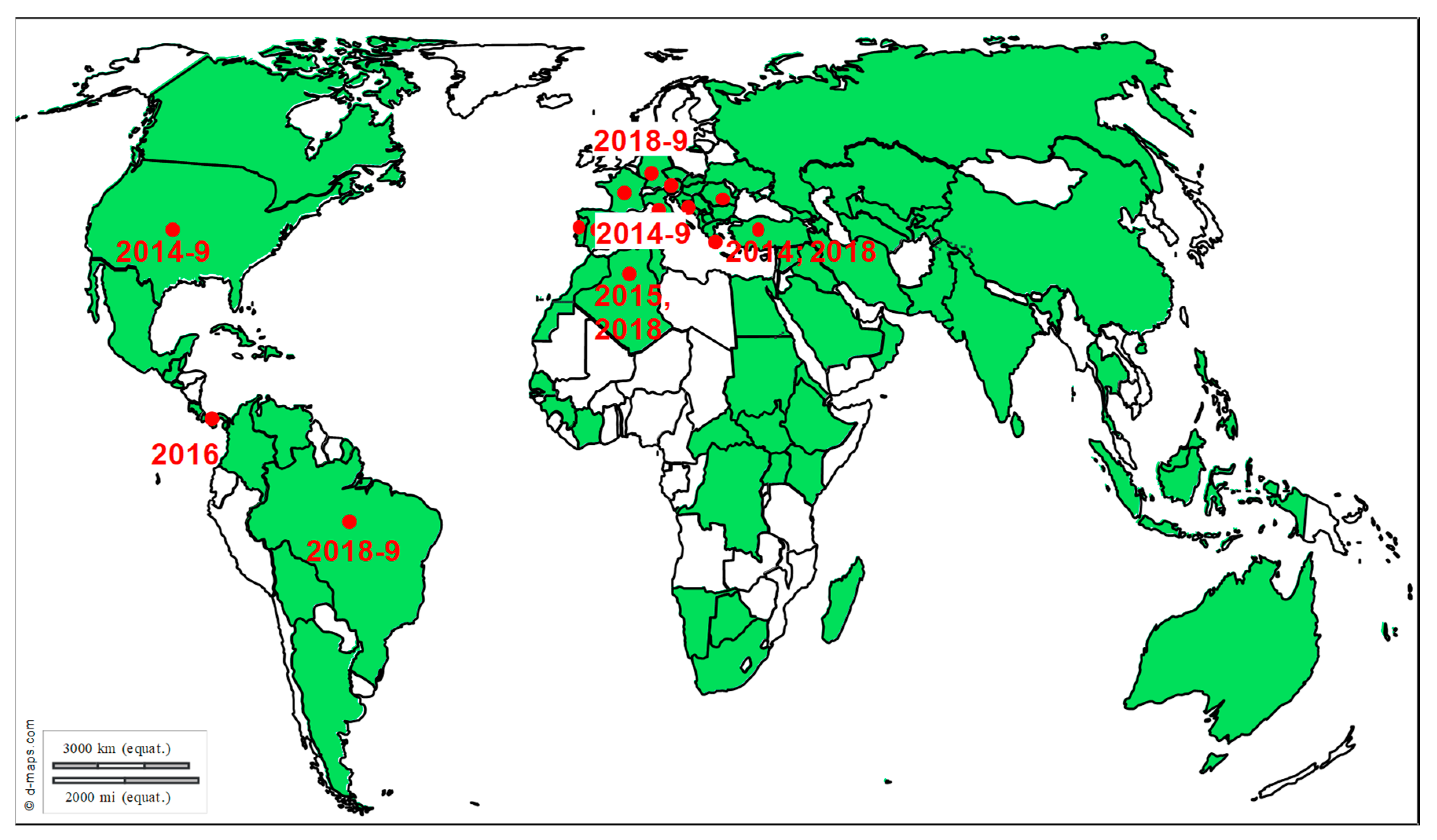
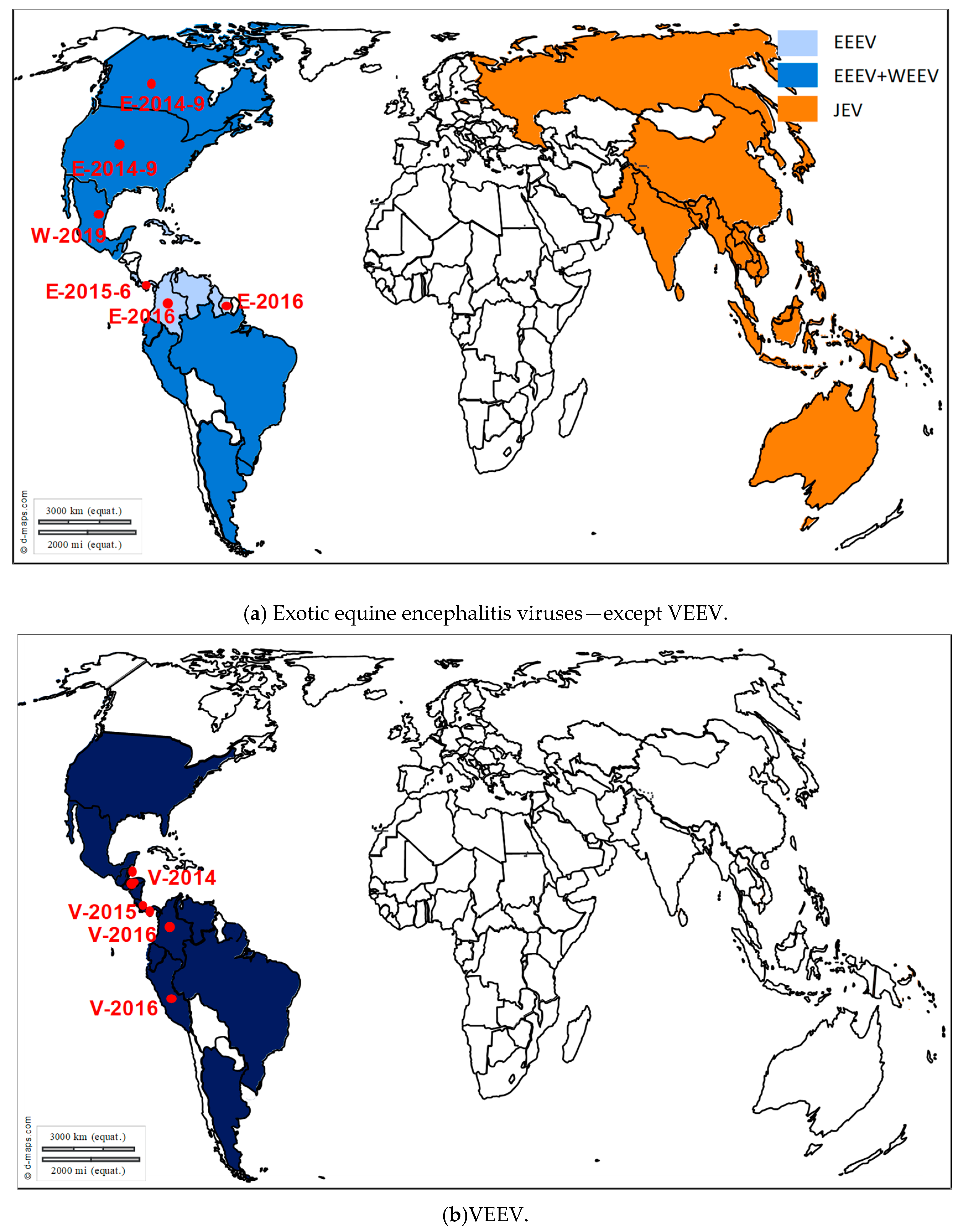
3. Exotic Equine Encephalitis Viruses in Europe
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pellegrini-Masini, A.; Livesey, L.C. Meningitis and encephalomyelitis in horses. Vet. Clin. N. Am. Equine Pract. 2006, 22, 553–589. [Google Scholar] [CrossRef] [PubMed]
- Leblond, A.; Villard, I.; Leblond, L.; Sabatier, P.; Sasco, A.J. A retrospective evaluation of the causes of death of 448 insured French horses in 1995. Vet. Res. Commun. 2000, 24, 85–102. [Google Scholar] [CrossRef] [PubMed]
- Tyler, C.M.; Davis, R.E.; Begg, A.P.; Hutchins, D.R.; Hodgson, D.R. A survey of neurological diseases in horses. Aust. Vet. J. 1993, 70, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Laugier, C.T.; Tapprest, J. Fréquence de la pathologie nerveuse et de ses différentes causes dans un effectif de 4319 chevaux autopsiés. Bull. Epidémiologique St. Anim. Et Aliment. Spécial Équidé 2012, 19, 9. [Google Scholar]
- Kumar, B.; Manuja, A.; Gulati, B.R.; Virmani, N.; Tripathi, B.N. Zoonotic Viral Diseases of Equines and Their Impact on Human and Animal Health. Open Virol. J. 2018, 12, 80–98. [Google Scholar] [CrossRef]
- Walker, P.J.; Siddell, S.G.; Lefkowitz, E.J.; Mushegian, A.R.; Dempsey, D.M.; Dutilh, B.E.; Harrach, B.; Harrison, R.L.; Hendrickson, R.C.; Junglen, S.; et al. Changes to virus taxonomy and the International Code of Virus Classification and Nomenclature ratified by the International Committee on Taxonomy of Viruses (2019). Arch. Virol. 2019, 164, 2417–2429. [Google Scholar] [CrossRef]
- Viralzone, SIB Swiss Institute of Bioinformatics. Available online: www.expasy.org/viralzone (accessed on 15 December 2019).
- Mayhew, I.G.; de Lahunta, A.; Whitlock, R.H.; Krook, L.; Tasker, J.B. Spinal cord disease in the horse. Cornell. Vet. 1978, 68, 1–207. [Google Scholar]
- Porter, R.S.; Leblond, A.; Lecollinet, S.; Tritz, P.; Cantile, C.; Kutasi, O.; Zientara, S.; Pradier, S.; van Galen, G.; Speybroek, N.; et al. Clinical diagnosis of West Nile Fever in Equids by classification and regression tree (CART) analysis and comparative study of clinical appearance in three European countries. Transbound. Emerg. Dis. 2011, 58, 197–205. [Google Scholar] [CrossRef]
- Van Galen, G.; Leblond, A.; Tritz, P.; Martinelle, L.; Pronost, S.; Saegerman, C. A retrospective study on equine herpesvirus type-1 associated myeloencephalopathy in France (2008–2011). Vet. Microbiol. 2015, 179, 304–309. [Google Scholar] [CrossRef]
- Pronost, S.; Legrand, L.; Pitel, P.H.; Wegge, B.; Lissens, J.; Freymuth, F.; Richard, E.; Fortier, G. Outbreak of equine herpesvirus myeloencephalopathy in France: A clinical and molecular investigation. Transbound. Emerg. Dis. 2012, 59, 256–263. [Google Scholar] [CrossRef]
- Knox, J.; Cowan, R.U.; Doyle, J.S.; Ligtermoet, M.K.; Archer, J.S.; Burrow, J.N.; Tong, S.Y.; Currie, B.J.; Mackenzie, J.S.; Smith, D.W.; et al. Murray Valley encephalitis: A review of clinical features, diagnosis and treatment. Med. J. Aust. 2012, 196, 322–326. [Google Scholar] [CrossRef]
- RESPE. Available online: http://www.respe.net (accessed on 15 December 2019).
- EQUINELLA. Available online: http://www.equinella.ch (accessed on 15 December 2019).
- World Organisation for Animal Health (OIE), Animal Health Information. Available online: https://www.oie.int/wahis_2/public/wahid.php/Diseaseinformation/Immsummary (accessed on 15 December 2019).
- Promed, International Society for Infectious Diseases. Available online: https://promedmail.org/ (accessed on 15 December 2019).
- ECDC. Epidemiological Update: West Nile Virus Transmission Season in Europe. Available online: https://www.ecdc.europa.eu/en/news-events/epidemiological-update-west-nile-virus-transmission-season-europe-2018 (accessed on 28 November 2019).
- Dunowska, M. A review of equid herpesvirus 1 for the veterinary practitioner. Part B: Pathogenesis and epidemiology. N. Z. Vet. J. 2014, 62, 179–188. [Google Scholar] [CrossRef] [PubMed]
- BioRender. Available online: https//app.biorender.com/ (accessed on 15 December 2019).
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antiviral Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [PubMed]
- Chapman, G.E.; Baylis, M.; Archer, D.; Daly, J.M. The challenges posed by equine arboviruses. Equine Vet. J. 2018, 50, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.J.; Eberle, R.; Ehlers, B.; Hayward, G.S.; McGeoch, D.J.; Minson, A.C.; Pellett, P.E.; Roizman, B.; Studdert, M.J.; Thiry, E. The order Herpesvirales. Arch. Virol. 2009, 154, 171–177. [Google Scholar] [CrossRef]
- Abdelgawad, A.; Damiani, A.; Ho, S.Y.; Strauss, G.; Szentiks, C.A.; East, M.L.; Osterrieder, N.; Greenwood, A.D. Zebra Alphaherpesviruses (EHV-1 and EHV-9): Genetic Diversity, Latency and Co-Infections. Viruses 2016, 8, 262. [Google Scholar] [CrossRef]
- Garvey, M.; Suarez, N.M.; Kerr, K.; Hector, R.; Moloney-Quinn, L.; Arkins, S.; Davison, A.J.; Cullinane, A. Equid herpesvirus 8: Complete genome sequence and association with abortion in mares. PLoS ONE 2018, 13, e0192301. [Google Scholar] [CrossRef]
- Saxegaard, F. Isolation and identification of equine rhinopneumonitis virus (equine abortion virus) from cases of abortion and paralysis. Nord. Vet. Med. 1966, 18, 504–516. [Google Scholar]
- Ma, G.; Azab, W.; Osterrieder, N. Equine herpesviruses type 1 (EHV-1) and 4 (EHV-4)—Masters of co-evolution and a constant threat to equids and beyond. Vet. Microbiol. 2013, 167, 123–134. [Google Scholar] [CrossRef]
- Chesters, P.M.; Allsop, R.; Purewal, A.; Edington, N. Detection of latency-associated transcripts of equid herpesvirus 1 in equine leukocytes but not in trigeminal ganglia. J. Virol. 1997, 71, 3437–3443. [Google Scholar]
- Oladunni, F.S.; Horohov, D.W.; Chambers, T.M. EHV-1: A constant threat to the horse industry. Front. Microbiol. 2019, 10, 2668. [Google Scholar] [CrossRef]
- Telford, E.A.; Watson, M.S.; McBride, K.; Davison, A.J. The DNA sequence of equine herpesvirus-1. Virology 1992, 189, 304–316. [Google Scholar] [CrossRef]
- Dayaram, A.; Franz, M.; Schattschneider, A.; Damiani, A.M.; Bischofberger, S.; Osterrieder, N.; Greenwood, A.D. Long term stability and infectivity of herpesviruses in water. Sci. Rep. 2017, 7, 46559. [Google Scholar] [CrossRef] [PubMed]
- Foote, C.E.; Love, D.N.; Gilkerson, J.R.; Whalley, J.M. Detection of EHV-1 and EHV-4 DNA in unweaned Thoroughbred foals from vaccinated mares on a large stud farm. Equine Vet. J. 2004, 36, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Kydd, J.H.; Lunn, D.P.; Osterrieder, K. Report of the Fourth International Havemeyer Workshop on Equid Herpesviruses (EHV) EHV-1, EHV-2 and EHV-5. Equine Vet. J. 2019, 51, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Vissani, M.A.; Becerra, M.L.; Olguin Perglione, C.; Tordoya, M.S.; Mino, S.; Barrandeguy, M. Neuropathogenic and non-neuropathogenic genotypes of Equid Herpesvirus type 1 in Argentina. Vet. Microbiol. 2009, 139, 361–364. [Google Scholar] [CrossRef]
- Paillot, R.C.; Case, R.; Ross, J.; Newton, R.; Nugent, J. Equine Herpes Virus-1: Virus, Immunity and Vaccines. TOVSJ 2008, 2, 68–91. [Google Scholar] [CrossRef]
- Crabb, B.S.; Studdert, M.J. Equine herpesviruses 4 (equine rhinopneumonitis virus) and 1 (equine abortion virus). Adv. Virus Res. 1995, 45, 153–190. [Google Scholar]
- Edington, N.; Smyth, B.; Griffiths, L. The role of endothelial cell infection in the endometrium, placenta and foetus of equid herpesvirus 1 (EHV-1) abortions. J. Comp. Pathol. 1991, 104, 379–387. [Google Scholar] [CrossRef]
- Whitwell, K.E.; Blunden, A.S. Pathological findings in horses dying during an outbreak of the paralytic form of Equid herpesvirus type 1 (EHV-1) infection. Equine Vet. J. 1992, 24, 13–19. [Google Scholar] [CrossRef]
- Slater, J.D.; Lunn, D.P.; Horohov, D.W.; Antczak, D.F.; Babiuk, L.; Breathnach, C.; Chang, Y.W.; Davis-Poynter, N.; Edington, N.; Ellis, S.; et al. Report of the equine herpesvirus-1 Havermeyer Workshop, San Gimignano, Tuscany, June 2004. Vet. Immunol. Immunopathol. 2006, 111, 3–13. [Google Scholar] [CrossRef]
- Van Maanen, C. Equine herpesvirus 1 and 4 infections: An update. Vet. Q. 2002, 24, 58–78. [Google Scholar] [CrossRef] [PubMed]
- Kydd, J.H.; Slater, J.; Osterrieder, N.; Antczak, D.F.; Lunn, D.P. Report of the Second Havemeyer EHV-1 Workshop, Steamboat Springs, Colorado, USA, September 2008. Equine Vet. J. 2010, 42, 572–575. [Google Scholar] [CrossRef] [PubMed]
- Lunn, D.P.; Davis-Poynter, N.; Flaminio, M.J.; Horohov, D.W.; Osterrieder, K.; Pusterla, N.; Townsend, H.G. Equine herpesvirus-1 consensus statement. J. Vet. Intern. Med. 2009, 23, 450–461. [Google Scholar] [CrossRef] [PubMed]
- Goehring, L.S.; van Winden, S.C.; van Maanen, C.; Sloet van Oldruitenborgh-Oosterbaan, M.M. Equine herpesvirus type 1-associated myeloencephalopathy in The Netherlands: A four-year retrospective study (1999–2003). J. Vet. Intern. Med. 2006, 20, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Pronost, S.; Leon, A.; Legrand, L.; Fortier, C.; Miszczak, F.; Freymuth, F.; Fortier, G. Neuropathogenic and non-neuropathogenic variants of equine herpesvirus 1 in France. Vet. Microbiol. 2010, 145, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Sutton, G.; Garvey, M.; Cullinane, A.; Jourdan, M.; Fortier, C.; Moreau, P.; Foursin, M.; Gryspeerdt, A.; Maisonnier, V.; Marcillaud-Pitel, C.; et al. Molecular Surveillance of EHV-1 Strains Circulating in France during and after the Major 2009 Outbreak in Normandy Involving Respiratory Infection, Neurological Disorder, and Abortion. Viruses 2019, 11, 916. [Google Scholar] [CrossRef]
- Allen, G.P. Risk factors for development of neurologic disease after experimental exposure to equine herpesvirus-1 in horses. Am. J. Vet. Res. 2008, 69, 1595–1600. [Google Scholar] [CrossRef]
- Goodman, L.B.; Loregian, A.; Perkins, G.A.; Nugent, J.; Buckles, E.L.; Mercorelli, B.; Kydd, J.H.; Palu, G.; Smith, K.C.; Osterrieder, N.; et al. A point mutation in a herpesvirus polymerase determines neuropathogenicity. PLoS Pathog. 2007, 3, e160. [Google Scholar] [CrossRef]
- Goehring, L.S.; van Maanen, C.; Berendsen, M.; Cullinane, A.; de Groot, R.J.; Rottier, P.J.; Wesselingh, J.J.; Sloet van Oldruitenborgh-Oosterbaan, M.M. Experimental infection with neuropathogenic equid herpesvirus type 1 (EHV-1) in adult horses. Vet. J. 2010, 186, 180–187. [Google Scholar] [CrossRef]
- Nugent, J.; Birch-Machin, I.; Smith, K.C.; Mumford, J.A.; Swann, Z.; Newton, J.R.; Bowden, R.J.; Allen, G.P.; Davis-Poynter, N. Analysis of equid herpesvirus 1 strain variation reveals a point mutation of the DNA polymerase strongly associated with neuropathogenic versus nonneuropathogenic disease outbreaks. J. Virol. 2006, 80, 4047–4060. [Google Scholar] [CrossRef]
- Allen, G.P.; Breathnach, C.C. Quantification by real-time PCR of the magnitude and duration of leucocyte-associated viraemia in horses infected with neuropathogenic vs. non-neuropathogenic strains of EHV-1. Equine Vet. J. 2006, 38, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Allen, G.P. Development of a real-time polymerase chain reaction assay for rapid diagnosis of neuropathogenic strains of equine herpesvirus-1. J. Vet. Diagn. Investig. 2007, 19, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Perkins, G.A.; Goodman, L.B.; Tsujimura, K.; Van de Walle, G.R.; Kim, S.G.; Dubovi, E.J.; Osterrieder, N. Investigation of the prevalence of neurologic equine herpes virus type 1 (EHV-1) in a 23-year retrospective analysis (1984–2007). Vet. Microbiol. 2009, 139, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Pronost, S.; Cook, R.F.; Fortier, G.; Timoney, P.J.; Balasuriya, U.B. Relationship between equine herpesvirus-1 myeloencephalopathy and viral genotype. Equine Vet. J. 2010, 42, 672–674. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.R.; Heldens, J. Equine herpesviruses 1 (EHV-1) and 4 (EHV-4)—Epidemiology, disease and immunoprophylaxis: A brief review. Vet. J. 2005, 170, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Goodman, L.B.; Wagner, B.; Flaminio, M.J.; Sussman, K.H.; Metzger, S.M.; Holland, R.; Osterrieder, N. Comparison of the efficacy of inactivated combination and modified-live virus vaccines against challenge infection with neuropathogenic equine herpesvirus type 1 (EHV-1). Vaccine 2006, 24, 3636–3645. [Google Scholar] [CrossRef] [PubMed]
- Garre, B.; Gryspeerdt, A.; Croubels, S.; De Backer, P.; Nauwynck, H. Evaluation of orally administered valacyclovir in experimentally EHV1-infected ponies. Vet. Microbiol. 2009, 135, 214–221. [Google Scholar] [CrossRef]
- Thieulent, C.J.; Hue, E.S.; Fortier, C.I.; Dallemagne, P.; Zientara, S.; Munier-Lehmann, H.; Hans, A.; Fortier, G.D.; Pitel, P.H.; Vidalain, P.O.; et al. Screening and evaluation of antiviral compounds against Equid alpha-herpesviruses using an impedance-based cellular assay. Virology 2019, 526, 105–116. [Google Scholar] [CrossRef]
- Vissani, M.A.; Thiry, E.; Dal Pozzo, F.; Barrandeguy, M. Antiviral agents against equid alphaherpesviruses: Current status and perspectives. Vet. J. 2016, 207, 38–44. [Google Scholar] [CrossRef]
- Nolden, T.; Banyard, A.C.; Finke, S.; Fooks, A.R.; Hanke, D.; Hoper, D.; Horton, D.L.; Mettenleiter, T.C.; Muller, T.; Teifke, J.P.; et al. Comparative studies on the genetic, antigenic and pathogenic characteristics of Bokeloh bat lyssavirus. J. Gen. Virol. 2014, 95, 1647–1653. [Google Scholar] [CrossRef]
- Malerczyk, C.; Freuling, C.; Gniel, D.; Giesen, A.; Selhorst, T.; Muller, T. Cross-neutralization of antibodies induced by vaccination with Purified Chick Embryo Cell Vaccine (PCECV) against different Lyssavirus species. Hum. Vaccin. Immunother. 2014, 10, 2799–2804. [Google Scholar] [CrossRef] [PubMed]
- Gunawardena, P.S.; Marston, D.A.; Ellis, R.J.; Wise, E.L.; Karawita, A.C.; Breed, A.C.; McElhinney, L.M.; Johnson, N.; Banyard, A.C.; Fooks, A.R. Lyssavirus in Indian Flying Foxes, Sri Lanka. Emerg. Infect. Dis. 2016, 22, 1456–1459. [Google Scholar] [CrossRef] [PubMed]
- Hayman, D.T.; Fooks, A.R.; Marston, D.A.; Garcia, R.J. The Global Phylogeography of Lyssaviruses-Challenging the ‘Out of Africa’ Hypothesis. PLoS Negl. Trop. Dis. 2016, 10, e0005266. [Google Scholar] [CrossRef] [PubMed]
- Rupprecht, C.E.; Turmelle, A.; Kuzmin, I.V. A perspective on lyssavirus emergence and perpetuation. Curr. Opin. Virol. 2011, 1, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.; Vos, A.; Freuling, C.; Tordo, N.; Fooks, A.R.; Muller, T. Human rabies due to lyssavirus infection of bat origin. Vet. Microbiol. 2010, 142, 151–159. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Expert Consultation on Rabies: Third Report; WHO Technical Report Series; World Health Organization: Geneva, Switzerland, 2018; pp. 1–183. [Google Scholar]
- Barecha, C.B.G.; Girzaw, F.; Kandi, V.; Pal, M. Epidemiology and Public Health Significance of Rabies. Perspect. Med. Res. 2017, 5, 55–67. [Google Scholar]
- Green, S.L. Equine rabies. Vet. Clin. N. Am. Equine Pract. 1993, 9, 337–347. [Google Scholar] [CrossRef]
- Sato, G.; Itou, T.; Shoji, Y.; Miura, Y.; Mikami, T.; Ito, M.; Kurane, I.; Samara, S.I.; Carvalho, A.A.; Nociti, D.P.; et al. Genetic and phylogenetic analysis of glycoprotein of rabies virus isolated from several species in Brazil. J. Vet. Med. Sci. 2004, 66, 747–753. [Google Scholar] [CrossRef][Green Version]
- Dyer, J.L.; Yager, P.; Orciari, L.; Greenberg, L.; Wallace, R.; Hanlon, C.A.; Blanton, J.D. Rabies surveillance in the United States during 2013. J. Am. Vet. Med. Assoc. 2014, 245, 1111–1123. [Google Scholar] [CrossRef]
- Ali, Y.; Intisar, S.; Wegdan, H.; Ali, E. Epidemiology of Rabies in Sudan. J. Anim. Vet. Adv. 2006, 5, 266–270. [Google Scholar]
- Weir, D.L.; Annand, E.J.; Reid, P.A.; Broder, C.C. Recent observations on Australian bat lyssavirus tropism and viral entry. Viruses 2014, 6, 909–926. [Google Scholar] [CrossRef] [PubMed]
- WHO. Rabies Bulletin Europe. 2010–2019. Available online: https://www.who-rabies-bulletin.org/ (accessed on 15 December 2019).
- Rech, R.; Barros, C. Neurologic Diseases in Horses. Vet. Clin. N. Am. Equine Pract. 2015, 31, 281–306. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Patil, R.D. Cryptic etiopathological conditions of equine nervous system with special emphasis on viral diseases. Vet. World 2017, 10, 1427–1438. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meyer, E.E.; Morris, P.G.; Elcock, L.H.; Weil, J. Hindlimb hyperesthesia associated with rabies in two horses. J. Am. Vet. Med. Assoc. 1986, 188, 629–632. [Google Scholar]
- OIE. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2019, Chapter 3.1.17; OIE: Paris, France, 2019; pp. 1–35. [Google Scholar]
- Appolinario, C.; Allendorf, S.D.; Vicente, A.F.; Ribeiro, B.D.; Fonseca, C.R.; Antunes, J.M.; Peres, M.G.; Kotait, I.; Carrieri, M.L.; Megid, J. Fluorescent antibody test, quantitative polymerase chain reaction pattern and clinical aspects of rabies virus strains isolated from main reservoirs in Brazil. Braz. J. Infect. Dis. 2015, 19, 479–485. [Google Scholar] [CrossRef]
- Muller, F.T.; Freuling, C.M. Rabies control in Europe: An overview of past, current and future strategies. Rev. Sci. Tech. 2018, 37, 409–419. [Google Scholar] [CrossRef]
- Harvey, A.M.; Watson, J.L.; Brault, S.A.; Edman, J.M.; Moore, S.M.; Kass, P.H.; Wilson, W.D. Duration of serum antibody response to rabies vaccination in horses. J. Am. Vet. Med. Assoc. 2016, 249, 411–418. [Google Scholar] [CrossRef]
- De la Torre, J.C. Molecular biology of borna disease virus: Prototype of a new group of animal viruses. J. Virol. 1994, 68, 7669–7675. [Google Scholar]
- Kuhn, J.H.; Durrwald, R.; Bao, Y.; Briese, T.; Carbone, K.; Clawson, A.N.; deRisi, J.L.; Garten, W.; Jahrling, P.B.; Kolodziejek, J.; et al. Taxonomic reorganization of the family Bornaviridae. Arch. Virol. 2015, 160, 621–632. [Google Scholar] [CrossRef]
- Honkavuori, K.S.; Shivaprasad, H.L.; Williams, B.L.; Quan, P.L.; Hornig, M.; Street, C.; Palacios, G.; Hutchison, S.K.; Franca, M.; Egholm, M.; et al. Novel borna virus in psittacine birds with proventricular dilatation disease. Emerg. Infect. Dis. 2008, 14, 1883–1886. [Google Scholar] [CrossRef]
- Hoffmann, B.; Tappe, D.; Hoper, D.; Herden, C.; Boldt, A.; Mawrin, C.; Niederstrasser, O.; Muller, T.; Jenckel, M.; van der Grinten, E.; et al. A Variegated Squirrel Bornavirus Associated with Fatal Human Encephalitis. N. Engl. J. Med. 2015, 373, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Dunia, D.; Cubitt, B.; de la Torre, J.C. Mechanism of Borna disease virus entry into cells. J. Virol. 1998, 72, 783–788. [Google Scholar] [PubMed]
- Rott, R.; Becht, H. Natural and experimental Borna disease in animals. Curr. Top. Microbiol. Immunol. 1995, 190, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Berg, M.; Johansson, M.; Montell, H.; Berg, A.L. Wild birds as a possible natural reservoir of Borna disease virus. Epidemiol. Infect. 2001, 127, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Bode, L.; Durrwald, R.; Ludwig, H. Borna virus infections in cattle associated with fatal neurological disease. Vet. Rec. 1994, 135, 283–284. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, K.; Tsuge, Y.; Asakawa, M.; Kabaya, H.; Okamoto, M.; Miyasho, T.; Taniyama, H.; Ishihara, C.; de la Torre, J.C.; Ikuta, K. Borna disease virus RNA detected in Japanese macaques (Macaca fuscata). Primates 2008, 49, 57–64. [Google Scholar] [CrossRef]
- Hilbe, M.; Herrsche, R.; Kolodziejek, J.; Nowotny, N.; Zlinszky, K.; Ehrensperger, F. Shrews as reservoir hosts of borna disease virus. Emerg. Infect. Dis. 2006, 12, 675–677. [Google Scholar] [CrossRef] [PubMed]
- Kinnunen, P.M.; Billich, C.; Ek-Kommonen, C.; Henttonen, H.; Kallio, R.K.; Niemimaa, J.; Palva, A.; Staeheli, P.; Vaheri, A.; Vapalahti, O. Serological evidence for Borna disease virus infection in humans, wild rodents and other vertebrates in Finland. J. Clin. Virol. 2007, 38, 64–69. [Google Scholar] [CrossRef]
- Lundgren, A.L.; Zimmermann, W.; Bode, L.; Czech, G.; Gosztonyi, G.; Lindberg, R.; Ludwig, H. Staggering disease in cats: Isolation and characterization of the feline Borna disease virus. J. Gen. Virol. 1995, 76, 2215–2222. [Google Scholar] [CrossRef]
- Malkinson, M.; Weisman, Y.; Ashash, E.; Bode, L.; Ludwig, H. Borna disease in ostriches. Vet. Rec. 1993, 133, 304. [Google Scholar] [CrossRef]
- Durrwald, R.; Kolodziejek, J.; Muluneh, A.; Herzog, S.; Nowotny, N. Epidemiological pattern of classical Borna disease and regional genetic clustering of Borna disease viruses point towards the existence of to-date unknown endemic reservoir host populations. Microbes Infect. 2006, 8, 917–929. [Google Scholar] [CrossRef] [PubMed]
- Staeheli, P.; Sauder, C.; Hausmann, J.; Ehrensperger, F.; Schwemmle, M. Epidemiology of Borna disease virus. J. Gen. Virol. 2000, 81, 2123–2135. [Google Scholar] [CrossRef] [PubMed]
- Weissenbock, H.; Bago, Z.; Kolodziejek, J.; Hager, B.; Palmetzhofer, G.; Durrwald, R.; Nowotny, N. Infections of horses and shrews with Bornaviruses in Upper Austria: A novel endemic area of Borna disease. Emerg. Microbes Infect. 2017, 6, e52. [Google Scholar] [CrossRef] [PubMed]
- Bourg, M.; Herzog, S.; Encarnacao, J.A.; Nobach, D.; Lange-Herbst, H.; Eickmann, M.; Herden, C. Bicolored white-toothed shrews as reservoir for borna disease virus, Bavaria, Germany. Emerg. Infect. Dis. 2013, 19, 2064–2066. [Google Scholar] [CrossRef] [PubMed]
- Nobach, D.; Bourg, M.; Herzog, S.; Lange-Herbst, H.; Encarnacao, J.A.; Eickmann, M.; Herden, C. Shedding of Infectious Borna Disease Virus-1 in Living Bicolored White-Toothed Shrews. PLoS ONE 2015, 10, e0137018. [Google Scholar] [CrossRef]
- Ludwig, H.; Bode, L.; Gosztonyi, G. Borna disease: A persistent virus infection of the central nervous system. Prog. Med. Virol. 1988, 35, 107–151. [Google Scholar]
- Herzog, S.; Pfeuffer, I.; Haberzettl, K.; Feldmann, H.; Frese, K.; Bechter, K.; Richt, J.A. Molecular characterization of Borna disease virus from naturally infected animals and possible links to human disorders. Arch. Virol. Suppl. 1997, 13, 183–190. [Google Scholar]
- Hagiwara, K.; Kamitani, W.; Takamura, S.; Taniyama, H.; Nakaya, T.; Tanaka, H.; Kirisawa, R.; Iwai, H.; Ikuta, K. Detection of Borna disease virus in a pregnant mare and her fetus. Vet. Microbiol. 2000, 72, 207–216. [Google Scholar] [CrossRef]
- Okamoto, M.; Hagiwara, K.; Kamitani, W.; Sako, T.; Hirayama, K.; Kirisawa, R.; Tsuji, M.; Ishihara, C.; Iwai, H.; Kobayashi, T.; et al. Experimental vertical transmission of Borna disease virus in the mouse. Arch. Virol. 2003, 148, 1557–1568. [Google Scholar] [CrossRef]
- Chalmers, R.M.; Thomas, D.R.; Salmon, R.L. Borna disease virus and the evidence for human pathogenicity: A systematic review. QJM 2005, 98, 255–274. [Google Scholar] [CrossRef]
- Nakamura, Y.; Takahashi, H.; Shoya, Y.; Nakaya, T.; Watanabe, M.; Tomonaga, K.; Iwahashi, K.; Ameno, K.; Momiyama, N.; Taniyama, H.; et al. Isolation of Borna disease virus from human brain tissue. J. Virol. 2000, 74, 4601–4611. [Google Scholar] [CrossRef] [PubMed]
- Brnic, D.; Stevanovic, V.; Cochet, M.; Agier, C.; Richardson, J.; Montero-Menei, C.N.; Milhavet, O.; Eloit, M.; Coulpier, M. Borna disease virus infects human neural progenitor cells and impairs neurogenesis. J. Virol. 2012, 86, 2512–2522. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Scordel, C.; Huttin, A.; Cochet-Bernoin, M.; Szelechowski, M.; Poulet, A.; Richardson, J.; Benchoua, A.; Gonzalez-Dunia, D.; Eloit, M.; Coulpier, M. Borna disease virus phosphoprotein impairs the developmental program controlling neurogenesis and reduces human GABAergic neurogenesis. PLoS Pathog. 2015, 11, e1004859. [Google Scholar] [CrossRef] [PubMed]
- Schwemmle, M.; Jehle, C.; Formella, S.; Staeheli, P. Sequence similarities between human bornavirus isolates and laboratory strains question human origin. Lancet 1999, 354, 1973–1974. [Google Scholar] [CrossRef]
- Hornig, M.; Briese, T.; Licinio, J.; Khabbaz, R.F.; Altshuler, L.L.; Potkin, S.G.; Schwemmle, M.; Siemetzki, U.; Mintz, J.; Honkavuori, K.; et al. Absence of evidence for bornavirus infection in schizophrenia, bipolar disorder and major depressive disorder. Mol. Psychiatry 2012, 17, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Korn, K.; Coras, R.; Bobinger, T.; Herzog, S.M.; Lucking, H.; Stohr, R.; Huttner, H.B.; Hartmann, A.; Ensser, A. Fatal Encephalitis Associated with Borna Disease Virus 1. N. Engl. J. Med. 2018, 379, 1375–1377. [Google Scholar] [CrossRef]
- Schlottau, K.; Forth, L.; Angstwurm, K.; Hoper, D.; Zecher, D.; Liesche, F.; Hoffmann, B.; Kegel, V.; Seehofer, D.; Platen, S.; et al. Fatal Encephalitic Borna Disease Virus 1 in Solid-Organ Transplant Recipients. N. Engl. J. Med. 2018, 379, 1377–1379. [Google Scholar] [CrossRef]
- Richt, J.A.; Grabner, A.; Herzog, S. Borna disease in horses. Vet. Clin. N. Am. Equine Pract. 2000, 16, 579–595. [Google Scholar] [CrossRef]
- Ludwig, H.; Bode, L. Borna disease virus: New aspects on infection, disease, diagnosis and epidemiology. Rev. Sci. Tech. 2000, 19, 259–288. [Google Scholar] [CrossRef]
- Beck, C.; Jimenez-Clavero, M.A.; Leblond, A.; Durand, B.; Nowotny, N.; Leparc-Goffart, I.; Zientara, S.; Jourdain, E.; Lecollinet, S. Flaviviruses in Europe: Complex circulation patterns and their consequences for the diagnosis and control of West Nile disease. Int. J. Environ. Res. Public Health 2013, 10, 6049–6083. [Google Scholar] [CrossRef]
- Melian, E.B.; Hinzman, E.; Nagasaki, T.; Firth, A.E.; Wills, N.M.; Nouwens, A.S.; Blitvich, B.J.; Leung, J.; Funk, A.; Atkins, J.F.; et al. NS1’ of flaviviruses in the Japanese encephalitis virus serogroup is a product of ribosomal frameshifting and plays a role in viral neuroinvasiveness. J. Virol. 2010, 84, 1641–1647. [Google Scholar] [CrossRef] [PubMed]
- Pradier, S.; Lecollinet, S.; Leblond, A. West Nile virus epidemiology and factors triggering change in its distribution in Europe. Rev. Sci. Tech. 2012, 31, 829–844. [Google Scholar] [CrossRef] [PubMed]
- Gardner, I.A.; Wong, S.J.; Ferraro, G.L.; Balasuriya, U.B.; Hullinger, P.J.; Wilson, W.D.; Shi, P.Y.; MacLachlan, N.J. Incidence and effects of West Nile virus infection in vaccinated and unvaccinated horses in California. Vet. Res. 2007, 38, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Zeller, H.G.; Schuffenecker, I. West Nile virus: An overview of its spread in Europe and the Mediterranean basin in contrast to its spread in the Americas. Eur. J. Clin. Microbiol. Infect. Dis. 2004, 23, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Autorino, G.L.; Battisti, A.; Deubel, V.; Ferrari, G.; Forletta, R.; Giovannini, A.; Lelli, R.; Murri, S.; Scicluna, M.T. West Nile virus epidemic in horses, Tuscany region, Italy. Emerg. Infect. Dis. 2002, 8, 1372–1378. [Google Scholar] [CrossRef]
- Ceianu, C.S.; Ungureanu, A.; Nicolescu, G.; Cernescu, C.; Nitescu, L.; Tardei, G.; Petrescu, A.; Pitigoi, D.; Martin, D.; Ciulacu-Purcarea, V.; et al. West nile virus surveillance in Romania: 1997–2000. Viral Immunol. 2001, 14, 251–262. [Google Scholar] [CrossRef]
- Murgue, B.; Murri, S.; Triki, H.; Deubel, V.; Zeller, H.G. West Nile in the Mediterranean basin: 1950–2000. Ann. N. Y. Acad. Sci. 2001, 951, 117–126. [Google Scholar] [CrossRef]
- Chancey, C.; Grinev, A.; Volkova, E.; Rios, M. The global ecology and epidemiology of West Nile virus. BioMed Res. Int. 2015, 2015, 376230. [Google Scholar] [CrossRef]
- Bakonyi, T.; Hubalek, Z.; Rudolf, I.; Nowotny, N. Novel flavivirus or new lineage of West Nile virus, central Europe. Emerg. Infect. Dis. 2005, 11, 225–231. [Google Scholar] [CrossRef]
- Hernandez-Triana, L.M.; Jeffries, C.L.; Mansfield, K.L.; Carnell, G.; Fooks, A.R.; Johnson, N. Emergence of west nile virus lineage 2 in europe: A review on the introduction and spread of a mosquito-borne disease. Front. Public Health 2014, 2, 271. [Google Scholar] [CrossRef]
- Michel, F.; Sieg, M.; Fischer, D.; Keller, M.; Eiden, M.; Reuschel, M.; Schmidt, V.; Schwehn, R.; Rinder, M.; Urbaniak, S.; et al. Evidence for West Nile Virus and Usutu Virus Infections in Wild and Resident Birds in Germany, 2017 and 2018. Viruses 2019, 11, 674. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, A.F.; Hall-Mendelin, S.; Webb, C.E.; Tan, C.S.; Frentiu, F.D.; Prow, N.A.; Hall, R.A. Role of enhanced vector transmission of a new West Nile virus strain in an outbreak of equine disease in Australia in 2011. Parasites Vectors 2014, 7, 586. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, J.A.; Wang, T.; Barrett, A.D. Virulence determinants of West Nile virus: How can these be used for vaccine design? Future Virol. 2017, 12, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Langevin, S.A.; Bowen, R.A.; Reisen, W.K.; Andrade, C.C.; Ramey, W.N.; Maharaj, P.D.; Anishchenko, M.; Kenney, J.L.; Duggal, N.K.; Romo, H.; et al. Host competence and helicase activity differences exhibited by West Nile viral variants expressing NS3-249 amino acid polymorphisms. PLoS ONE 2014, 9, e100802. [Google Scholar] [CrossRef]
- Brault, A.C.; Huang, C.Y.; Langevin, S.A.; Kinney, R.M.; Bowen, R.A.; Ramey, W.N.; Panella, N.A.; Holmes, E.C.; Powers, A.M.; Miller, B.R. A single positively selected West Nile viral mutation confers increased virogenesis in American crows. Nat. Genet. 2007, 39, 1162–1166. [Google Scholar] [CrossRef]
- Dridi, M.; Van Den Berg, T.; Lecollinet, S.; Lambrecht, B. Evaluation of the pathogenicity of West Nile virus (WNV) lineage 2 strains in a SPF chicken model of infection: NS3-249Pro mutation is neither sufficient nor necessary for conferring virulence. Vet. Res. 2015, 46, 130. [Google Scholar] [CrossRef][Green Version]
- Randolph, S.E.; Gern, L.; Nuttall, P.A. Co-feeding ticks: Epidemiological significance for tick-borne pathogen transmission. Parasitol. Today 1996, 12, 472–479. [Google Scholar] [CrossRef]
- Labuda, M.; Jones, L.D.; Williams, T.; Nuttall, P.A. Enhancement of tick-borne encephalitis virus transmission by tick salivary gland extracts. Med. Vet. Entomol. 1993, 7, 193–196. [Google Scholar] [CrossRef]
- Holzmann, H.; Aberle, S.W.; Stiasny, K.; Werner, P.; Mischak, A.; Zainer, B.; Netzer, M.; Koppi, S.; Bechter, E.; Heinz, F.X. Tick-borne encephalitis from eating goat cheese in a mountain region of Austria. Emerg. Infect. Dis. 2009, 15, 1671–1673. [Google Scholar] [CrossRef]
- Jones, L.D.; Gaunt, M.; Hails, R.S.; Laurenson, K.; Hudson, P.J.; Reid, H.; Henbest, P.; Gould, E.A. Transmission of louping ill virus between infected and uninfected ticks co-feeding on mountain hares. Med. Vet. Entomol. 1997, 11, 172–176. [Google Scholar] [CrossRef]
- Jeffries, C.L.; Mansfield, K.L.; Phipps, L.P.; Wakeley, P.R.; Mearns, R.; Schock, A.; Bell, S.; Breed, A.C.; Fooks, A.R.; Johnson, N. Louping ill virus: An endemic tick-borne disease of Great Britain. J. Gen. Virol. 2014, 95, 1005–1014. [Google Scholar] [CrossRef] [PubMed]
- Timoney, P.J.; Donnelly, W.J.; Clements, L.O.; Fenlon, M. Encephalitis caused by louping ill virus in a group of horses in Ireland. Equine Vet. J. 1976, 8, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Klaus, C.; Horugel, U.; Hoffmann, B.; Beer, M. Tick-borne encephalitis virus (TBEV) infection in horses: Clinical and laboratory findings and epidemiological investigations. Vet. Microbiol. 2013, 163, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Waldvogel, A.; Matile, H.; Wegmann, C.; Wyler, R.; Kunz, C. Tick-borne encephalitis in the horse. Schweiz. Arch. Tierheilkd. 1981, 123, 227–233. [Google Scholar] [PubMed]
- Rushton, J.O.; Lecollinet, S.; Hubalek, Z.; Svobodova, P.; Lussy, H.; Nowotny, N. Tick-borne encephalitis virus in horses, Austria, 2011. Emerg. Infect. Dis. 2013, 19, 635–637. [Google Scholar] [CrossRef] [PubMed]
- Csank, T.; Drzewniokova, P.; Korytar, L.; Major, P.; Gyuranecz, M.; Pistl, J.; Bakonyi, T. A Serosurvey of Flavivirus Infection in Horses and Birds in Slovakia. Vector Borne Zoonotic Dis. 2018, 18, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Suss, J. Tick-borne encephalitis 2010: Epidemiology, risk areas, and virus strains in Europe and Asia-An overview. Ticks Tick-Borne Dis. 2011, 2, 2–15. [Google Scholar] [CrossRef]
- Jaaskelainen, A.E.; Sironen, T.; Murueva, G.B.; Subbotina, N.; Alekseev, A.N.; Castren, J.; Alitalo, I.; Vaheri, A.; Vapalahti, O. Tick-borne encephalitis virus in ticks in Finland, Russian Karelia and Buryatia. J. Gen. Virol. 2010, 91, 2706–2712. [Google Scholar] [CrossRef]
- Balseiro, A.; Royo, L.J.; Martinez, C.P.; Fernandez de Mera, I.G.; Hofle, U.; Polledo, L.; Marreros, N.; Casais, R.; Marin, J.F. Louping ill in goats, Spain, 2011. Emerg. Infect. Dis. 2012, 18, 976–978. [Google Scholar] [CrossRef]
- Ytrehus, B.; Vainio, K.; Dudman, S.G.; Gilray, J.; Willoughby, K. Tick-borne encephalitis virus and louping-ill virus may co-circulate in Southern Norway. Vector Borne Zoonotic Dis. (Larchmt. N.Y.) 2013, 13, 762–768. [Google Scholar] [CrossRef]
- Mustafa, Y.M.; Meuren, L.M.; Coelho, S.V.A.; de Arruda, L.B. Pathways Exploited by Flaviviruses to Counteract the Blood-Brain Barrier and Invade the Central Nervous System. Front. Microbiol. 2019, 10, 525. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Lo, Y.; Chapagain, M.; Lum, S.; Kumar, M.; Gurjav, U.; Luo, H.; Nakatsuka, A.; Nerurkar, V.R. West Nile virus infection modulates human brain microvascular endothelial cells tight junction proteins and cell adhesion molecules: Transmigration across the in vitro blood-brain barrier. Virology 2009, 385, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Dai, J.; Bai, F.; Kong, K.F.; Wong, S.J.; Montgomery, R.R.; Madri, J.A.; Fikrig, E. Matrix metalloproteinase 9 facilitates West Nile virus entry into the brain. J. Virol. 2008, 82, 8978–8985. [Google Scholar] [CrossRef] [PubMed]
- Palus, M.; Vancova, M.; Sirmarova, J.; Elsterova, J.; Perner, J.; Ruzek, D. Tick-borne encephalitis virus infects human brain microvascular endothelial cells without compromising blood-brain barrier integrity. Virology 2017, 507, 110–122. [Google Scholar] [CrossRef]
- Cho, H.; Diamond, M.S. Immune responses to West Nile virus infection in the central nervous system. Viruses 2012, 4, 3812–3830. [Google Scholar] [CrossRef]
- Samuel, M.A.; Wang, H.; Siddharthan, V.; Morrey, J.D.; Diamond, M.S. Axonal transport mediates West Nile virus entry into the central nervous system and induces acute flaccid paralysis. Proc. Natl. Acad. Sci. USA 2007, 104, 17140–17145. [Google Scholar] [CrossRef]
- Xiao, S.Y.; Guzman, H.; Zhang, H.; Travassos da Rosa, A.P.; Tesh, R.B. West Nile virus infection in the golden hamster (Mesocricetus auratus): A model for West Nile encephalitis. Emerg. Infect. Dis. 2001, 7, 714–721. [Google Scholar] [CrossRef]
- Potokar, M.; Jorgacevski, J.; Zorec, R. Astrocytes in Flavivirus Infections. Int. J. Mol. Sci. 2019, 20, 691. [Google Scholar] [CrossRef]
- Palus, M.; Bily, T.; Elsterova, J.; Langhansova, H.; Salat, J.; Vancova, M.; Ruzek, D. Infection and injury of human astrocytes by tick-borne encephalitis virus. J. Gen. Virol. 2014, 95, 2411–2426. [Google Scholar] [CrossRef]
- Donadieu, E.; Lowenski, S.; Servely, J.L.; Laloy, E.; Lilin, T.; Nowotny, N.; Richardson, J.; Zientara, S.; Lecollinet, S.; Coulpier, M. Comparison of the neuropathology induced by two West Nile virus strains. PLoS ONE 2013, 8, e84473. [Google Scholar] [CrossRef]
- Hyde, J.; Nettleton, P.; Marriott, L.; Willoughby, K. Louping ill in horses. Vet. Rec. 2007, 160, 532. [Google Scholar] [CrossRef] [PubMed]
- Beck, C.; Lowenski, S.; Durand, B.; Bahuon, C.; Zientara, S.; Lecollinet, S. Improved reliability of serological tools for the diagnosis of West Nile fever in horses within Europe. PLoS Negl. Trop. Dis. 2017, 11, e0005936. [Google Scholar] [CrossRef] [PubMed]
- Chaskopoulou, A.; L’Ambert, G.; Petric, D.; Bellini, R.; Zgomba, M.; Groen, T.A.; Marrama, L.; Bicout, D.J. Ecology of West Nile virus across four European countries: Review of weather profiles, vector population dynamics and vector control response. Parasites Vectors 2016, 9, 482. [Google Scholar] [CrossRef]
- Gossner, C.M.; Marrama, L.; Carson, M.; Allerberger, F.; Calistri, P.; Dilaveris, D.; Lecollinet, S.; Morgan, D.; Nowotny, N.; Paty, M.C.; et al. West Nile virus surveillance in Europe: Moving towards an integrated animal-human-vector approach. Euro Surveill. 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. EPAR Product Information: Equip WNV 2015. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/veterinary/000137/WC500063683.pdf (accessed on 12 September 2019).
- Minke, J.M.; Siger, L.; Karaca, K.; Austgen, L.; Gordy, P.; Bowen, R.; Renshaw, R.W.; Loosmore, S.; Audonnet, J.C.; Nordgren, B. Recombinant canarypoxvirus vaccine carrying the prM/E genes of West Nile virus protects horses against a West Nile virus-mosquito challenge. Arch. Virol. Suppl. 2004, 18, 221–230. [Google Scholar]
- El Garch, H.; Minke, J.M.; Rehder, J.; Richard, S.; Edlund Toulemonde, C.; Dinic, S.; Andreoni, C.; Audonnet, J.C.; Nordgren, R.; Juillard, V. A West Nile virus (WNV) recombinant canarypox virus vaccine elicits WNV-specific neutralizing antibodies and cell-mediated immune responses in the horse. Vet. Immunol. Immunopathol. 2008, 123, 230–239. [Google Scholar] [CrossRef]
- Guy, B.; Guirakhoo, F.; Barban, V.; Higgs, S.; Monath, T.P.; Lang, J. Preclinical and clinical development of YFV 17D-based chimeric vaccines against dengue, West Nile and Japanese encephalitis viruses. Vaccine 2010, 28, 632–649. [Google Scholar] [CrossRef]
- Durand, B.; Lecollinet, S.; Beck, C.; Martinez-Lopez, B.; Balenghien, T.; Chevalier, V. Identification of hotspots in the European union for the introduction of four zoonotic arboviroses by live animal trade. PLoS ONE 2013, 8, e70000. [Google Scholar] [CrossRef]
- Pfeffer, M.; Dobler, G. Emergence of zoonotic arboviruses by animal trade and migration. Parasites Vectors 2010, 3, 35. [Google Scholar] [CrossRef]
- Diaz, A.; Coffey, L.L.; Burkett-Cadena, N.; Day, J.F. Reemergence of St. Louis Encephalitis Virus in the Americas. Emerg. Infect. Dis. 2018, 24. [Google Scholar] [CrossRef]
- Mansfield, K.L.; Hernandez-Triana, L.M.; Banyard, A.C.; Fooks, A.R.; Johnson, N. Japanese encephalitis virus infection, diagnosis and control in domestic animals. Vet. Microbiol. 2017, 201, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Prow, N.A.; Tan, C.S.; Wang, W.; Hobson-Peters, J.; Kidd, L.; Barton, A.; Wright, J.; Hall, R.A.; Bielefeldt-Ohmann, H. Natural exposure of horses to mosquito-borne flaviviruses in south-east Queensland, Australia. Int. J. Environ. Res. Public Health 2013, 10, 4432–4443. [Google Scholar] [CrossRef] [PubMed]
- Morita, K.; Nabeshima, T.; Buerano, C.C. Japanese encephalitis. Rev. Sci. Tech. 2015, 34, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Ravanini, P.; Huhtamo, E.; Ilaria, V.; Crobu, M.G.; Nicosia, A.M.; Servino, L.; Rivasi, F.; Allegrini, S.; Miglio, U.; Magri, A.; et al. Japanese encephalitis virus RNA detected in Culex pipiens mosquitoes in Italy. Euro Surveill. 2012, 17. [Google Scholar] [CrossRef] [PubMed]
- Preziuso, S.; Mari, S.; Mariotti, F.; Rossi, G. Detection of Japanese Encephalitis Virus in bone marrow of healthy young wild birds collected in 1997-2000 in Central Italy. Zoonoses Public Health 2018, 65, 798–804. [Google Scholar] [CrossRef]
- Reisen, W.K.; Lothrop, H.D.; Wheeler, S.S.; Kennsington, M.; Gutierrez, A.; Fang, Y.; Garcia, S.; Lothrop, B. Persistent West Nile virus transmission and the apparent displacement St. Louis encephalitis virus in southeastern California, 2003–2006. J. Med. Entomol. 2008, 45, 494–508. [Google Scholar] [CrossRef]
- Fang, Y.; Reisen, W.K. Previous infection with West Nile or St. Louis encephalitis viruses provides cross protection during reinfection in house finches. Am. J. Trop. Med. Hyg. 2006, 75, 480–485. [Google Scholar] [CrossRef]
- Spinsanti, L.I.; Diaz, L.A.; Glatstein, N.; Arselan, S.; Morales, M.A.; Farias, A.A.; Fabbri, C.; Aguilar, J.J.; Re, V.; Frias, M.; et al. Human outbreak of St. Louis encephalitis detected in Argentina, 2005. J. Clin. Virol. 2008, 42, 27–33. [Google Scholar] [CrossRef]
- Rico-Hesse, R. Venezuelan equine encephalomyelitis. Vet. Clin. N. Am. Equine Pract. 2000, 16, 553–563. [Google Scholar] [CrossRef]
- Bergren, N.A.; Auguste, A.J.; Forrester, N.L.; Negi, S.S.; Braun, W.A.; Weaver, S.C. Western equine encephalitis virus: Evolutionary analysis of a declining alphavirus based on complete genome sequences. J. Virol. 2014, 88, 9260–9267. [Google Scholar] [CrossRef]
- Brault, A.C.; Powers, A.M.; Holmes, E.C.; Woelk, C.H.; Weaver, S.C. Positively charged amino acid substitutions in the e2 envelope glycoprotein are associated with the emergence of venezuelan equine encephalitis virus. J. Virol. 2002, 76, 1718–1730. [Google Scholar] [CrossRef] [PubMed]
- Arechiga-Ceballos, N.; Aguilar-Setien, A. Alphaviral equine encephalomyelitis (Eastern, Western and Venezuelan). Rev. Sci. Tech. 2015, 34, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Deardorff, E.R.; Forrester, N.L.; Travassos-da-Rosa, A.P.; Estrada-Franco, J.G.; Navarro-Lopez, R.; Tesh, R.B.; Weaver, S.C. Experimental infection of potential reservoir hosts with Venezuelan equine encephalitis virus, Mexico. Emerg. Infect. Dis. 2009, 15, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, P.V.; Paessler, S.; Carrara, A.S.; Baron, S.; Poast, J.; Wang, E.; Moncayo, A.C.; Anishchenko, M.; Watts, D.; Tesh, R.B.; et al. Variation in interferon sensitivity and induction among strains of eastern equine encephalitis virus. J. Virol. 2005, 79, 11300–11310. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C.; Powers, A.M.; Brault, A.C.; Barrett, A.D. Molecular epidemiological studies of veterinary arboviral encephalitides. Vet. J. 1999, 157, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Zientara, S.; Weyer, C.T.; Lecollinet, S. African horse sickness. Rev. Sci. Tech. 2015, 34, 315–327. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.; Mellor, P. Bluetongue in Europe: Vectors, epidemiology and climate change. Parasitol. Res. 2008, 103, S69–S77. [Google Scholar] [CrossRef]
- Aharonson-Raz, K.; Steinman, A.; Bumbarov, V.; Maan, S.; Maan, N.S.; Nomikou, K.; Batten, C.; Potgieter, C.; Gottlieb, Y.; Mertens, P.; et al. Isolation and phylogenetic grouping of equine encephalosis virus in Israel. Emerg. Infect. Dis. 2011, 17, 1883–1886. [Google Scholar] [CrossRef]
- Reed, S.M.; Toribio, R.E. Equine herpesvirus 1 and 4. Vet. Clin. N. Am. Equine Pract. 2004, 20, 631–642. [Google Scholar] [CrossRef]
- Slater, J.D.; Borchers, K.; Thackray, A.M.; Field, H.J. The trigeminal ganglion is a location for equine herpesvirus 1 latency and reactivation in the horse. J. Gen. Virol. 1994, 75, 2007–2016. [Google Scholar] [CrossRef]
- Roehrig, J.T. West nile virus in the United States—A historical perspective. Viruses 2013, 5, 3088–3108. [Google Scholar] [CrossRef] [PubMed]
- Beck, C.; Despres, P.; Paulous, S.; Vanhomwegen, J.; Lowenski, S.; Nowotny, N.; Durand, B.; Garnier, A.; Blaise-Boisseau, S.; Guitton, E.; et al. A High-Performance Multiplex Immunoassay for Serodiagnosis of Flavivirus-Associated Neurological Diseases in Horses. BioMed Res. Int. 2015, 2015, 678084. [Google Scholar] [CrossRef] [PubMed]
- Cleton, N.B.; van Maanen, K.; Bergervoet, S.A.; Bon, N.; Beck, C.; Godeke, G.J.; Lecollinet, S.; Bowen, R.; Lelli, D.; Nowotny, N.; et al. A Serological Protein Microarray for Detection of Multiple Cross-Reactive Flavivirus Infections in Horses for Veterinary and Public Health Surveillance. Transbound. Emerg. Dis. 2017, 64, 1801–1812. [Google Scholar] [CrossRef] [PubMed]
- Van Eeden, C.; Williams, J.H.; Gerdes, T.G.; van Wilpe, E.; Viljoen, A.; Swanepoel, R.; Venter, M. Shuni virus as cause of neurologic disease in horses. Emerg. Infect. Dis. 2012, 18, 318–321. [Google Scholar] [CrossRef]
- Altan, E.; Li, Y.; Sabino-Santos, G., Jr.; Sawaswong, V.; Barnum, S.; Pusterla, N.; Deng, X.; Delwart, E. Viruses in Horses with Neurologic and Respiratory Diseases. Viruses 2019, 11, 942. [Google Scholar] [CrossRef]
- Cohen, M.L. Changing patterns of infectious disease. Nature 2000, 406, 762–767. [Google Scholar] [CrossRef]
- Martinet, J.P.; Ferte, H.; Failloux, A.B.; Schaffner, F.; Depaquit, J. Mosquitoes of North-Western Europe as Potential Vectors of Arboviruses: A Review. Viruses 2019, 11, 1059. [Google Scholar] [CrossRef]
- Cunze, S.; Koch, L.K.; Kochmann, J.; Klimpel, S. Aedes albopictus and Aedes japonicus—Two invasive mosquito species with different temperature niches in Europe. Parasites Vectors 2016, 9, 573. [Google Scholar] [CrossRef]
- Vogels, C.B.; Goertz, G.P.; Pijlman, G.P.; Koenraadt, C.J. Vector competence of European mosquitoes for West Nile virus. Emerg. Microbes Infect. 2017, 6, e96. [Google Scholar] [CrossRef]
- De Wispelaere, M.; Despres, P.; Choumet, V. European Aedes albopictus and Culex pipiens Are Competent Vectors for Japanese Encephalitis Virus. PLoS Negl. Trop. Dis. 2017, 11, e0005294. [Google Scholar] [CrossRef]
- Huber, K.; Jansen, S.; Leggewie, M.; Badusche, M.; Schmidt-Chanasit, J.; Becker, N.; Tannich, E.; Becker, S.C. Aedes japonicus japonicus (Diptera: Culicidae) from Germany have vector competence for Japan encephalitis virus but are refractory to infection with West Nile virus. Parasitol. Res. 2014, 113, 3195–3199. [Google Scholar] [CrossRef] [PubMed]

| Virus | Diagnostic Assays | Advantages and Shortcomings |
|---|---|---|
| EHV-1 | Direct assays: PCR, virus isolation | Direct virus detection and typing (SNP-PCR) is possible from easily accessible samples (nasal swabs and blood). |
| Serology: VNT, CFT or ELISA | Due to highly prevalent and lifelong infection, diagnostic assays should be interpreted with care. Serology will be informative if serial serum samples can be obtained. | |
| Rabies virus | Direct assays: DFA, dRIT, RT-PCR | Direct virus detection is possible only from the brain of dead animals. |
| BoDV | Direct assays: RT-PCR, IHC | Due to limited antibody response induced after BoDV infection, definitive diagnostic will be made only after direct virus detection from the brain of dead animals. |
| WNV/Flaviviruses | Indirect assays preferred: ELISA, IFA, HIA, VNT | Rapid serological screening tests (competition ELISA, IFA) are very sensitive but present a low diagnostic specificity; they should be interpreted with care and confronted with results from confirmatory serological assays (VNT, MIA). |
| Direct assays: RT-PCR, virus isolation | Direct virus detection is possible from the brain of dead animals and when positive, indicates recent virus infection. |
| Virus | Vaccine Types Available in Europe | Protection Provided |
|---|---|---|
| EHV-1 | Inactivated: BIOEQUIN® H (BIOVETA), PNEUMEQUINE® (Boehringer Ingelheim), EQUIP® EHV 1,4 (Zoetis) Live attenuated: PREVACCINOL® (MSD Animal Health), licensed in Germany | Insufficient individual protection against EHM but allows for decreased virus transmission in the vaccinated population, after 2 primes at a 1-month interval (3–4 months with the live attenuated vaccine) and boosts every 6 to 12 months. |
| Rabies virus | Inactivated: ENDURACELL® R MONO and VERSIGUARD® Rabies (Zoetis), NOBIVAC® Rabies (MSD), RABIGEN® mono (Virbac) and RABISIN® (Boehringer Ingelheim) | Good protection at the individual level provided after a unique prime and boosts performed every year or every 2 years. |
| WNV | Inactivated: EQUIP® WNV (Zoetis) Recombinant: EQUILIS® West Nile (MSD) and PROTEQ® West Nile (Boehringer Ingelheim) | Good protection at the individual level provided after 2 primes at a 1-month interval and boosts performed every year. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lecollinet, S.; Pronost, S.; Coulpier, M.; Beck, C.; Gonzalez, G.; Leblond, A.; Tritz, P. Viral Equine Encephalitis, a Growing Threat to the Horse Population in Europe? Viruses 2020, 12, 23. https://doi.org/10.3390/v12010023
Lecollinet S, Pronost S, Coulpier M, Beck C, Gonzalez G, Leblond A, Tritz P. Viral Equine Encephalitis, a Growing Threat to the Horse Population in Europe? Viruses. 2020; 12(1):23. https://doi.org/10.3390/v12010023
Chicago/Turabian StyleLecollinet, Sylvie, Stéphane Pronost, Muriel Coulpier, Cécile Beck, Gaelle Gonzalez, Agnès Leblond, and Pierre Tritz. 2020. "Viral Equine Encephalitis, a Growing Threat to the Horse Population in Europe?" Viruses 12, no. 1: 23. https://doi.org/10.3390/v12010023
APA StyleLecollinet, S., Pronost, S., Coulpier, M., Beck, C., Gonzalez, G., Leblond, A., & Tritz, P. (2020). Viral Equine Encephalitis, a Growing Threat to the Horse Population in Europe? Viruses, 12(1), 23. https://doi.org/10.3390/v12010023







