Diversity and Host Specificity Revealed by Biological Characterization and Whole Genome Sequencing of Bacteriophages Infecting Salmonella enterica
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Bacteriophage Isolation and Purification
2.3. Host Range Determination
2.4. Phage DNA isolation
2.5. DNA Sequencing and Annotation Workflow
2.6. Genomic Analysis
3. Results
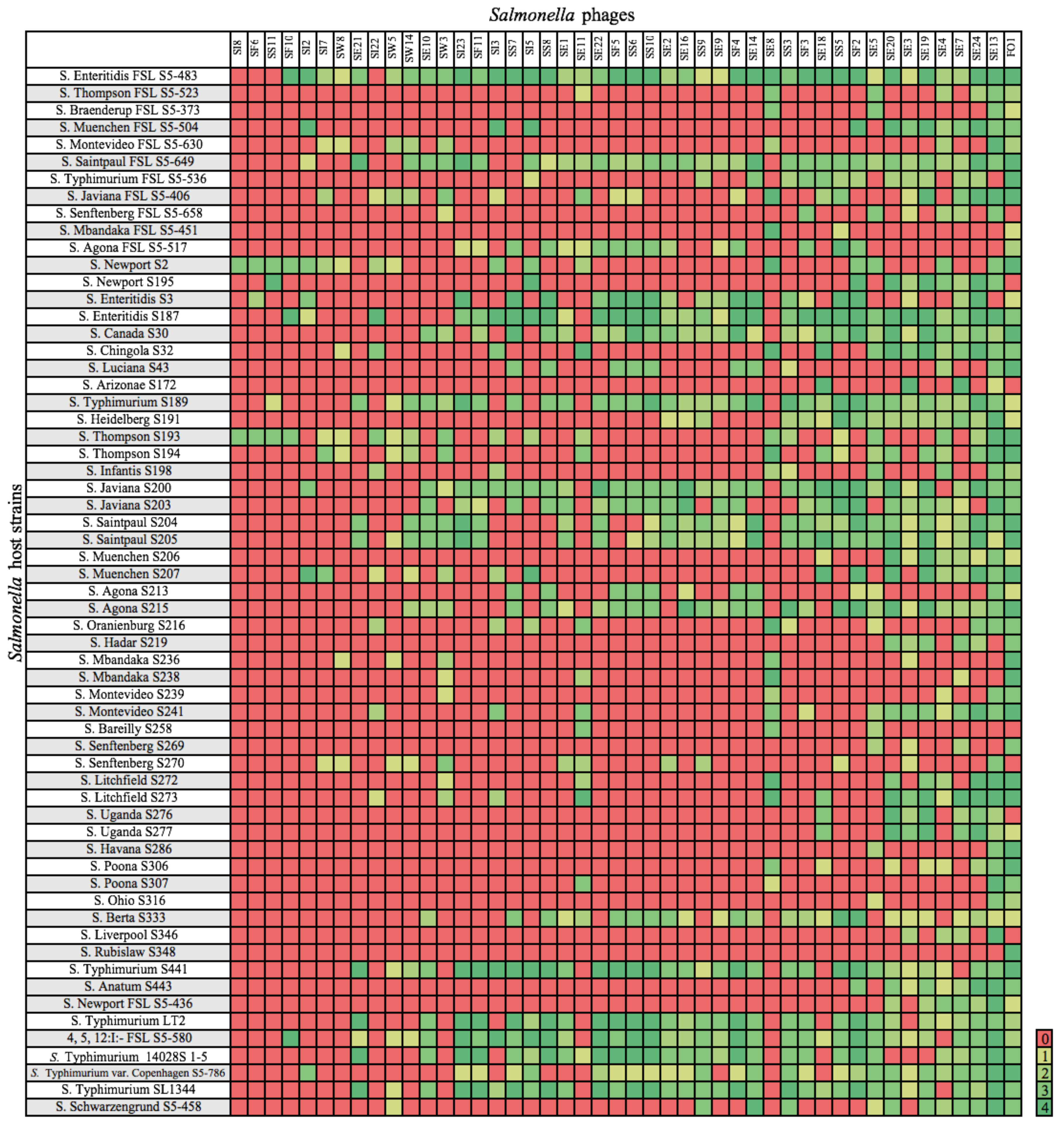
3.1. Host Range of Phages Infecting Salmonella
3.2. General Genomic Characterization
3.3. Phage Classifications
3.4. Identification of Putative Phage tRNAs
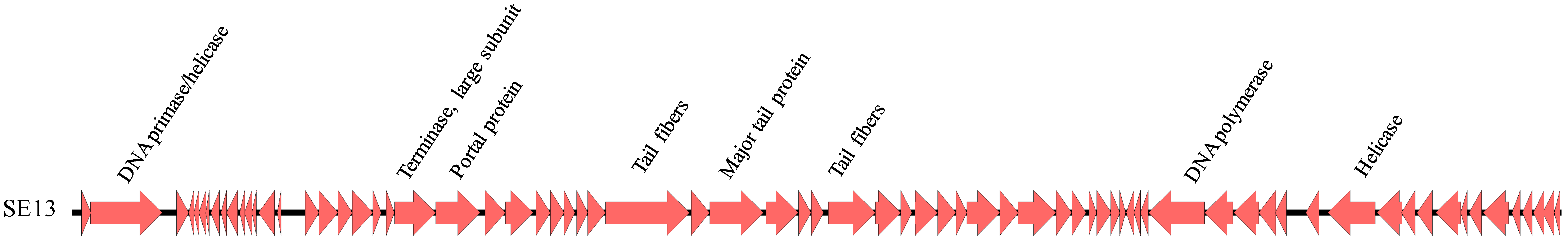
3.5. Genomic Analysis of SE13
3.6. Genome Size, G+C content and Identification of DNA Metabolism-Related Genes
3.7. Identification of Genes Encoding for Putative Virulence Factors
4. Discussion
4.1. Host Range of Phages Infecting Salmonella
4.2. General Genomic Characterization
4.3. Phage Classifications
4.4. Identification of Putative Phage tRNAs
4.5. Genomic Analysis of SE13
4.6. Genome size, G+C Content and Identification of DNA Metabolism-Related Genes
4.7. Identification of Genes Encoding for Putative Virulence Factors
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hendrix, R.W. Bacteriophages: Evolution of the majority. Theor. Popul. Biol. 2002, 61, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Ackermann, H.W.; Kropinski, A.M.; Lavigne, R.; Sutton, J.M.; Reynolds, D.M. Comparative analysis of 37 Acinetobacter bacteriophages. Viruses 2018, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, B.F. Bacteriophages as anti-infective agents: Recent developments and regulatory challenges. Exp. Rev. Anti-Infect. Ther. 2012, 10, 533–535. [Google Scholar] [CrossRef] [PubMed]
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2017, 5, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Mikalová, L.; Bosák, J.; Hříbková, H.; Dědičová, D.; Benada, O.; Šmarda, J.; Šmajs, D. Novel temperate phages of Salmonella enterica subsp. salamae and subsp. diarizonae and their activity against pathogenic S. enterica subsp. enterica isolates. PLoS ONE 2017, 12, e0170734. [Google Scholar] [CrossRef]
- Seed, K.D. Battling phages: How bacteria defend against viral attack. PLoS Pathog. 2015, 11, e1004847. [Google Scholar] [CrossRef] [PubMed]
- Clokie, M.R.J.; Millard, A.D.; Letarov, A.V.; Heaphy, S. Phages in nature. Bacteriophage 2011, 1, 31–45. [Google Scholar] [CrossRef]
- Cortés, P.; Spricigo, D.A.; Bardina, C.; Llagostera, M. Remarkable diversity of Salmonella bacteriophages in swine and poultry. FEMS Microbiol. Lett. 2015, 362, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Goodridge, L.; Fong, K.; Wang, S.; Delaquis, P. Bacteriophage-based weapons for the war against foodborne pathogens. Curr. Opin. Food Sci. 2018, 20, 69–75. [Google Scholar] [CrossRef]
- Hagens, S.; Loessner, M.J. Application of bacteriophages for detection and control of foodborne pathogens. Appl. Microbiol. Biotechnol. 2007, 76, 513–519. [Google Scholar] [CrossRef]
- Abedon, S.T. Lysis from without. Bacteriophage 2011, 1, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Majowicz, S.; Musto, J.; Scallan, E.; Angulo, F.; Kirk, M.; O’Brien, S.J.; Jones, T.F.; Fazil, A.; Hoekstra, R.M. International Collaboration on Enteric Disease ‘Burden of Illness’ Studies. The global burden of nontyphoidal Salmonella gastroenteritis. Clin. Infect. Dis. 2010, 50, 882–889. [Google Scholar] [CrossRef] [PubMed]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States—Major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Reports of Selected Salmonella Outbreak Investigations. Available online: https://www.cdc.gov/salmonella/outbreaks.html (accessed on 2 November 2018).
- Fong, K.; Wang, S. Strain-specific survival of Salmonella enterica in peanut oil, peanut shell, and chia seeds. J. Food Prot. 2016, 79, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Finke, J.F.; Winget, D.M.; Chan, A.M.; Suttle, C.A. Variation in the genetic repertoire of viruses infecting Micromonas pusilla reflects horizontal gene transfer and links to their environmental distribution. Viruses 2017, 9, 116. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Lin, Y.R.; Lu, M.W.; Sung, P.J.; Wang, W.H.; Lin, C.S. Genome sequences characterizing five mutations in RNA polymerase and major capsid of phages φA318 and φAs51 of Vibrio alginolyticus with different burst efficiencies. BMC Genom. 2014, 15, 505. [Google Scholar] [CrossRef] [PubMed]
- Marinelli, L.J.; Fitz-Gibbon, S.; Hayes, C.; Bowman, C.; Inkeles, M.; Loncaric, A.; Russell, D.A.; Jacobs-Sera, D.; Cokus, S.; Pellegrini, M.; et al. Propionibacterium acnes bacteriophages display limited genetic diversity and broad killing activity against bacterial skin isolates. mBio 2012, 3, e00279-12. [Google Scholar] [CrossRef] [PubMed]
- Pope, W.H.; Bowman, C.A.; Russell, D.A.; Jacobs-Sera, D.; Asai, D.J.; Cresawn, S.G.; Jacobs, W.R.; Hendrix, R.W.; Lawrence, J.G.; Hatfull, G.F. Whole genome comparison of a large collection of mycobacteriophages reveals a continuum of phage genetic diversity. eLIFE 2015, 4, e06416. [Google Scholar] [CrossRef] [PubMed]
- Switt, A.I.M.; Orsi, R.H.; Den Bakker, H.C.; Vongkamjan, K.; Altie, C.; Wiedmann, M. Genomic characterization provides new insight into Salmonella phage diversity. BMC Genom. 2013, 14, 481. [Google Scholar]
- Fong, K.; LaBossiere, B.; Switt, A.I.M.; Delaquis, P.; Goodridge, L.; Levesque, R.C.; Danyluk, M.D.; Wang, S. Characterization of four novel bacteriophages isolated from British Columbia for control of non-typhoidal Salmonella in vitro and on sprouting alfalfa seeds. Front. Microbiol. 2017, 8, 2193. [Google Scholar] [CrossRef]
- Kutter, E. Phage host range and efficiency ofplating. In Bacteriophage; Clokie, M.R.J., Kropinski, A.M., Eds.; Humana Press: New York, NY, USA, 2009. [Google Scholar]
- Ferrari, R.G.; Rosario, D.K.A.; Cunha-Neto, A.; Mano, S.B.; Figueiredo, S.F.; Conte-Jubior, C.A. Worldwide epidemiology of Salmonella serovars in animal-based foods: A meta-analysis. Appl. Environ. Microbiol. 2019, 85, e00591-19. [Google Scholar] [CrossRef] [PubMed]
- Snyder, T.R.; Boktor, S.W.; M’Ikanatha, N.M. Salmonellosis outbreaks by food vehicle, serotype, season, and geographical location, United States, 1998 to 2015. J. Food Prot. 2019, 82, 1191–1199. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.H. Bacteriophages; Interscience Publishers: New York, NY, USA, 1959. [Google Scholar]
- Ellis, A.; Preston, M.; Borczyk, A.; Miller, B.; Stone, P.; Hatton, B.; Shagla, A.; Hockin, J. A community outbreak of Salmonella Berta associated with a soft cheese product. Epidemiol. Infect. 1998, 120, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Khan Mirzaei, M.; Nilsson, A.S. Isolation of phages for phage therapy: A comparison of spot tests and efficiency of plating analyses for determination of host range and efficacy. PLoS ONE 2015, 10, e0118557. [Google Scholar] [CrossRef] [PubMed]
- Moineau, S.; Pandian, S.; Klaenhammer, T.R. Evolution of a lytic bacteriophage via DNA acquisition from the Lactococcus lactis chromosome. Appl. Environ. Microbiol. 1994, 60, 1832–1841. [Google Scholar] [PubMed]
- Boisvert, S.; Laviolette, F.; Corbeil, J. Ray: Simultaneous assembly of reads from a mix of high-throughput sequencing technologies. J. Comput. Biol. 2010, 17, 1519–1533. [Google Scholar] [CrossRef] [PubMed]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef]
- Dutilh, B.E.; Cassman, N.; McNair, K.; Sanchez, S.E.; Silva, G.G.Z.; Boling, L.; Barr, J.J.; Speth, D.R.; Seguritan, V.; Aziz, R.K.; et al. A highly abundant bacteriophage discovered in the unknown sequences of human faecal metagenomes. Nat. Commun. 2014, 5, 4498. [Google Scholar] [CrossRef]
- Laslett, D.; Canback, B. ARAGORN, a program to detect tRNA genes and tmRNA genes in nucleotide sequences. Nucleic Acids Res. 2004, 32, 11–16. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Ragonnet-Cronin, M.; Lycett, S.J.; Hodcroft, E.; Hue, S.; Fearnhill, E.; Dunn, D.; Delpech, V.; Leigh Brown, A.J.; Lycett, S. Automated analysis of phylogenetic clusters. BMC Bioinform. 2013, 14, 317. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.J.; Petty, N.K.; Beatson, S.A. Easyfig: A genome comparison visualizer. Bioinformatics 2011, 27, 1009–1010. [Google Scholar] [CrossRef] [PubMed]
- Colavecchio, A.; D’Souza, Y.; Tompkins, E.; Jeukens, J.; Freschi, L.; Emond-Rheault, J.G.; Kukavica-Ibrulj, I.; Boyle, B.; Bekal, S.; Tamber, S.; et al. Prophage integrase typing is a useful indicator of genomic diversity in Salmonella enterica. Front. Microbiol. 2017, 8, 1283. [Google Scholar] [CrossRef] [PubMed]
- Felix, A.; Callow, B.R. Typing of paratyphoid B bacilli by means of Vi bacteriophage. Br. Med. J. 1943, 2, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, J.; Suissa, M.; Wyse, J.; Cohen, I.; Weiser, I.; Reznick, S.; Lubinsky-Mink, S.; Stewart, G.; Ulitzur, S. Detection of bacteria using foreign DNA: The development of a bacteriophage reagent for Salmonella. Int. J. Food Microbiol. 2002, 74, 229–238. [Google Scholar] [CrossRef]
- Welkos, S.; Schreiber, M.; Baer, H. Identification of Salmonella with the O-1 bacteriophage. Appl. Microbiol. 1974, 28, 618–622. [Google Scholar] [PubMed]
- Wichels, A.; Biel, S.S.; Gelderblom, H.R.; Brinkhoff, T.; Muyzer, G.; Schütt, C. Bacteriophage diversity in the North Sea. Appl. Environ. Microbiol. 1998, 64, 4128–4133. [Google Scholar]
- Murphy, J.; Bottacini, F.; Mohony, J.; Kelleher, P.; Neve, H.; Zomer, A.; Nauta, A.; van Sinderen, D. Comparative genomics and functional analysis of the 936 group of lactococcal Siphoviridae phages. Sci. Rep. 2016, 6, 21345. [Google Scholar] [CrossRef]
- Weber-Dabrowska, B.; Jończyk-Matysiak, E.; Żaczek, M.; Łobocka, M.; Łusiak-Szelachowska, M.; Górski, A. Bacteriophage procurement for therapeutic purposes. Front. Microbiol. 2016, 7, 1177. [Google Scholar] [CrossRef]
- Galanis, E.; Lo Fo Wong, D.M.; Patrick, M.E.; Binsztein, N.; Cieslik, A.; Chalermchikit, T.; Aidara-Kane, A.; Ellis, A.; Angulo, F.J.; Wegener, H.C.; et al. Web-based surveillance and global Salmonella distribution, 2000–2002. Emerg. Infect. Dis. 2006, 12, 381–388. [Google Scholar] [CrossRef]
- Díaz-Muñoz, S.L.; Koskella, B. Bacteria-phage interactions in natural environments. Adv. Appl. Microbiol. 2014, 89, 135–183. [Google Scholar] [PubMed]
- Parmar, K.; Dafale, N.; Pal, R.; Tikariha, H.; Purohit, H. An insight into phage diversity at environmental habitats using comparative metagenomics approach. Curr. Microbiol. 2018, 75, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Deveau, H.; Labrie, S.J.; Chopin, M.C.; Moineau, S. Biodiversity and classification of Lactococcal phages. Appl. Environ. Microbiol. 2006, 72, 4338–4346. [Google Scholar] [CrossRef] [PubMed]
- Adriaenssens, E.M.; Cowan, D.A. Using signature genes as tools to assess environmental viral ecology and diversity. Appl. Environ. Microbiol. 2014, 80, 4470–4480. [Google Scholar] [CrossRef] [PubMed]
- Grose, J.H.; Casjens, S.R. Understanding the enormous diversity of bacteriophages: The tailed phages that infect the bacterial family Enterobacteriaceae. Virology 2014, 468–480, 421–443. [Google Scholar] [CrossRef] [PubMed]
- Casjens, S.R.; Grose, J.H. Contributions of P2-and P22-like prophages to understanding the enormous diversity and abundance of tailed bacteriophages. Virology 2016, 496, 255–276. [Google Scholar] [CrossRef]
- Born, Y.; Knecht, L.E.; Eigenmann, M.; Bolliger, M.; Klumpp, J.; Fieseler, L. A major-capsid-protein-based multiplex PCR assay for rapid identification of selected virulent bacteriophage types. Arch. Virol. 2019, 164, 819–830. [Google Scholar] [CrossRef]
- Adriaenssens, E.M.; Brister, J.R. How to name and classify your phage: An informal guide. Viruses 2017, 9, 70. [Google Scholar] [CrossRef]
- Simmonds, P.; Aiewasakun, P. Virus classification—Where do you draw the line? Arch. Virol. 2018, 163, 2037–2046. [Google Scholar] [CrossRef]
- Bodier-Montagutelli, E.; Morello, E.; l’Hostis, G.; Guillon, A.; Dalloneau, E.; Respaud, R.; Pallaoro, N.; Blois, H.; Vecellio, L.; Gabard, J.; et al. Inhaled phage therapy: A promising and challenging approach to treat bacterial respiratory infections. Expert Opin. Drug Deliv. 2017, 14, 959–972. [Google Scholar] [CrossRef]
- Shin, H.; Lee, J.H.; Kim, H.; Choi, Y.; Heu, S.; Ryu, S. Receptor diversity and host interaction of bacteriophages infecting Salmonella enterica serovar Typhimurium. PLoS ONE 2012, 7, e43392. [Google Scholar] [CrossRef] [PubMed]
- Rohde, C.; Resch, G.; Pirnay, J.P.; Blasdel, B.G.; Debarbieux, L.; Gelman, D.; Górski, A.; Hazan, R.; Huys, I.; Kakabadze, E.; et al. Expert opinion on three phage therapy related topics: Bacterial phage resistance, phage training and prophages in bacterial production strains. Viruses 2018, 10, 178. [Google Scholar] [CrossRef] [PubMed]
- Delesalle, V.A.; Tanke, N.T.; Vill, A.C.; Krukonis, G.P. Testing hypotheses for the presence of tRNA genes in mycobacteriophage genomes. Bacteriophage 2016, 6, e1219441. [Google Scholar] [CrossRef] [PubMed]
- Bailly-Bechet, M.; Vergassola, M.; Rocha, E. Causes for the intriguing presence of tRNAs in phages. Genome Res. 2007, 17, 1486–1495. [Google Scholar] [CrossRef] [PubMed]
- Kunisawa, T. Functional role of mycobacteriophage transfer RNAs. J. Theor. Biol. 2000, 205, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Cheetham, B.F.; Katz, M.E. A role for bacteriophages in the evolution and transfer of bacterial virulence determinants. Mol. Microbiol. 1995, 18, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Bardina, C.; Colom, J.; Spricigo, D.; Otero, J.; Sánchez-Osuna, M.; Cortés, P.; Llagostera, M. Genomics of three new bacteriophages useful in the biocontrol of Salmonella. Front. Microbiol. 2016, 7, 545. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.W.; Kim, J.W.; Jung, T.S.; Woo, G.J. wksl3, a new biocontrol agent for Salmonella enterica serovars Enteritidis and Typhimurium in foods: Characterization, application, sequence analysis, and oral acute toxicity study. Appl. Environ. Microbiol. 2013, 79, 1956–1968. [Google Scholar] [CrossRef] [PubMed]
- Weigel, C.; Seitz, H. Bacteriophage replication modules. FEMS Microbiol. Rev. 2006, 30, 321–381. [Google Scholar] [CrossRef] [PubMed]
- Karkman, A.; Do, T.T.; Walsh, F.; Virta, M.P.J. Antibiotic-resistance genes in waste water. Trends Microbiol. 2018, 26, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Moreirinha, C.; Lewicka, M.; Almeida, P.; Clemente, C.; Cunha, A.; Delgadillo, I.; Romalde, J.L.; Nunes, M.L.; Almeida, A. Bacteriophages with potential to inactivate Salmonella Typhimurium: Use of single phage suspensions and phage cocktails. Virus Res. 2016, 220, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Dupuis, M.E.; Moineau, S. Genome organization and characterization of the virulent lactococcal phage 1358 and its similarities to Listeria phages. Appl. Environ. Microbiol. 2010, 76, 1623–1632. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.S.; Kutter, E.; Mosig, G.; Arisaka, F.; Kunisawa, T.; Rüger, W. Bacteriophage T4 genome. Microbiol. Mol. Biol. Rev. 2003, 67, 86–156. [Google Scholar] [CrossRef] [PubMed]
- Howard-Varona, C.; Hargreaves, K.R.; Abedon, S.T.; Sullivan, M.B. Lysogeny in nature: Mechanisms, impact and ecology of temperate phages. ISME J. 2017, 11, 1511–1520. [Google Scholar] [CrossRef] [PubMed]
- Goh, S. Phage transduction. Methods Mol. Biol. 2016, 476, 177–185. [Google Scholar]
- Paradiso, R.; Riccardi, M.G.; Orsini, M.; Galiero, G.; Borriello, G. Complete genome sequences of three bacteriophages infecting Salmonella enterica serovar Enteritidis. Genome Announc. 2016, 4, e00939-16. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, S.M.; Brannon, M.K.; Dasgupta, N.; Pier, M.; Sgambati, N.; Miller, A.K.; Selgrade, S.E.; Miller, S.I.; Denton, M.; Conway, S.P.; et al. PmrB mutations promote polymyxin resistance of Pseudomonas aeruginosa isolated from colistin-treated cystic fibrosis patients. Antimicrob. Agents Chemother. 2012, 56, 1019–1030. [Google Scholar] [CrossRef]
- Abraham, N.; Kwon, D.H. A single amino acid substitution in PmrB is associated with polymyxin B resistance in clinical isolates of Pseudomonas aeruginosa. FEMS Microbiol. Lett. 2009, 298, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Karpe, Y.A.; Kanade, G.D.; Pingale, K.D.; Arankalle, V.A.; Banerjee, K. Genomic characterization of Salmonella bacteriophages isolated from India. Virus Genes. 2016, 52, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Oh, B.; Moyer, C.L.; Hendrix, R.W.; Duda, R.L. The delta domain of the HK97 major capsid protein is essential for assembly. Virology 2014, 456–457, 171–178. [Google Scholar] [CrossRef]
- Shahrbabak, S.S.; Khodabandehlou, Z.; Shahverdi, A.R.; Skurnik, M.; Ackermann, H.W.; Varjosalo, M.; Yazdi, M.T.; Sepehizadeh, Z. Isolation, characterization and complete genome sequence of PhaxI: A phage of Escherichia coli O157:H7. Microbiology 2013, 159, 1629–1638. [Google Scholar] [CrossRef] [PubMed]
- Moreno Switt, A.I.; den Bakker, H.C.; Cummings, C.A.; Rodriguez-Rivera, L.D.; Govoni, G.; Raneiri, M.L.; Degoricija, L.; Brown, S.; Hoelzer, K.; Peters, J.E.; et al. Identification and characterization of novel Salmonella mobile elements involved in the dissemination of genes linked to virulence and transmission. PLoS ONE 2012, 7, e41247. [Google Scholar] [CrossRef]
- York, G.M.; Walker, G.C. The succinyl and acetyl modifications of succinoglycan influence susceptibility of succinoglycan to cleavage by the Rhizobium meliloti glycanases ExoK and ExsH. J. Bacteriol. 1998, 95, 4912–4917. [Google Scholar]
- Aird, E.L.H.; Brightwell, G.; Jones, M.A.; Johnston, A.W.B. Identification of the exo loci required for exopolysaccharide synthesis in Agrobacterium radiobacter NCIB11883. Microbiology 1991, 137, 2287–2297. [Google Scholar]


| Newly Isolated Phage | Cluster | Closest Related Phage (NCBI Best Match) | Genus |
|---|---|---|---|
| SE21 | 1 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SE22 | 1 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SI23 | 1 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SF10 | 2 | Salmonella phage ST160 | Lederbergvirus |
| SI5 | 2 | Salmonella phage ST160 | Lederbergvirus |
| SI3 | 2 | Salmonella phage ST160 | Lederbergvirus |
| SS3 | 3 | Salmonella phage GG32 | Cvivirinae |
| SS9 | 3 | Salmonella phage GG32 | Cvivirinae |
| SS5 | 4 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SS1 | 4 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SS7 | 4 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SS6 | 4 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SF2 | 4 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SS8 | 4 | Salmonella phage ST3 | Guernseyvirinae |
| SI22 | 5 | Salmonella phage FSL SP-004 | Peduovirinae |
| SW9 | 5 | Salmonella phage FSL SP-004 | Peduovirinae |
| SI7 | 6 | N/A | N/A |
| SW5 | 6 | N/A | N/A |
| SW3 | 6 | N/A | N/A |
| SE7 | 7 | Salmonella phage S147 | Tequintavirus |
| SE20 | 7 | Salmonella phage Seabear | Tequintavirus |
| SE24 | 7 | Salmonella phage S126 | Tequintavirus |
| SF3 | 8 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SF11 | 8 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SE16 | 8 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SE1 | 8 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SE10 | 8 | Salmonella phage 103203_sal5 | Lederbergvirus |
| SI8 | 9 | Salmonella phage ST160 | Lederbergvirus |
| SF6 | 9 | Salmonella phage SE1 | Lederbergvirus |
| SE14 | Singleton | Salmonella phage S115 | Cvivirinae |
| SI1 | 10 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SF4 | 10 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SF5 | 10 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SS10 | 10 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SI2 | 10 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SS4 | 10 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SF1 | 10 | Salmonella phage vB_SenS_SB3 | Guernseyvirinae |
| SE4 | Singleton | Salmonella phage ZCSE2 | N/A |
| SE5 | Singleton | Erwinia phage phiEa21-4 | Ounavirinae |
| SE13 | Singleton | Salmonella phage BP63 | N/A |
| SE11 | 11 | Salmonella phage SP01 | Tequintavirus |
| SE8 | 11 | Salmonella phage SP01 | Tequintavirus |
| SE19 | 12 | Salmonella phage SP01 | Tequintavirus |
| SE18 | 12 | Salmonella phage BSP22A | Tequintavirus |
| SE3 | 12 | Salmonella phage S147 | Tequintavirus |
| Morphotype Classification | tRNA Genes | arnC | vriC | exoZ | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Myoviridae | Siphoviridae | Podoviridae | Unclassified | Phage | Number | Phage | Number | Phage | Number | Phage | Number | Phage | Number | Phage | Cluster | Source | Phage | Cluster | Source | Phage | Cluster | Source |
| SE4 | SE3 | SE1 | SI7 | SE1 | 0 | SF1 | 0 | SI1 | 0 | SS1 | 0 | SW3 | 0 | SE21 | 1 | E | SS3 | 3 | S | SI3 | 2 | I |
| SE5 | SE7 | SE10 | SW3 | SE3 | 29 | SF2 | 0 | SI2 | 0 | SS3 | 4 | SW5 | 0 | SE22 | 1 | I | SS9 | 3 | S | SI5 | 2 | I |
| SE13 | SE8 | SE16 | SW5 | SE4 | 0 | SF3 | 0 | SI3 | 1 | SS4 | 0 | SW9 | 0 | SI23 | 1 | E | SE14 | Singleton | E | SF10 | 2 | F |
| SE14 | SE11 | SE21 | SE5 | 27 | SF4 | 0 | SI5 | 1 | SS5 | 0 | SE16 | 10 | E | SI8 | 11 | I | ||||||
| SI22 | SE18 | SE22 | SE7 | 29 | SF5 | 0 | SI7 | 0 | SS6 | 0 | SF11 | 10 | F | SF6 | 11 | F | ||||||
| SS3 | SE19 | SF3 | SE8 | 22 | SF6 | 1 | SI8 | 1 | SS7 | 0 | SE10 | 10 | E | |||||||||
| SS9 | SE20 | SF6 | SE10 | 0 | SF10 | 1 | SI22 | 0 | SS8 | 0 | SF3 | 10 | F | |||||||||
| SW9 | SE24 | SF10 | SE11 | 22 | SF11 | 0 | SI23 | 0 | SS9 | 4 | SE1 | 10 | E | |||||||||
| SF1* | SF11 | SE13 | 0 | SS10 | 0 | |||||||||||||||||
| SF2 | SI3 | SE14 | 4 | |||||||||||||||||||
| SF4 | SI5 | SE16 | 0 | |||||||||||||||||||
| SF5 | SI8 | SE18 | 28 | |||||||||||||||||||
| SI1* | SI23 | SE19 | 29 | |||||||||||||||||||
| SI2 | SE20 | 29 | ||||||||||||||||||||
| SS1* | SE21 | 0 | ||||||||||||||||||||
| SS4* | SE22 | 0 | ||||||||||||||||||||
| SS5 | SE24 | 29 | ||||||||||||||||||||
| SS6 | ||||||||||||||||||||||
| SS7 | ||||||||||||||||||||||
| SS8 | ||||||||||||||||||||||
| SS10 | ||||||||||||||||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fong, K.; Tremblay, D.M.; Delaquis, P.; Goodridge, L.; Levesque, R.C.; Moineau, S.; Suttle, C.A.; Wang, S. Diversity and Host Specificity Revealed by Biological Characterization and Whole Genome Sequencing of Bacteriophages Infecting Salmonella enterica. Viruses 2019, 11, 854. https://doi.org/10.3390/v11090854
Fong K, Tremblay DM, Delaquis P, Goodridge L, Levesque RC, Moineau S, Suttle CA, Wang S. Diversity and Host Specificity Revealed by Biological Characterization and Whole Genome Sequencing of Bacteriophages Infecting Salmonella enterica. Viruses. 2019; 11(9):854. https://doi.org/10.3390/v11090854
Chicago/Turabian StyleFong, Karen, Denise M. Tremblay, Pascal Delaquis, Lawrence Goodridge, Roger C. Levesque, Sylvain Moineau, Curtis A. Suttle, and Siyun Wang. 2019. "Diversity and Host Specificity Revealed by Biological Characterization and Whole Genome Sequencing of Bacteriophages Infecting Salmonella enterica" Viruses 11, no. 9: 854. https://doi.org/10.3390/v11090854
APA StyleFong, K., Tremblay, D. M., Delaquis, P., Goodridge, L., Levesque, R. C., Moineau, S., Suttle, C. A., & Wang, S. (2019). Diversity and Host Specificity Revealed by Biological Characterization and Whole Genome Sequencing of Bacteriophages Infecting Salmonella enterica. Viruses, 11(9), 854. https://doi.org/10.3390/v11090854




