Serum LPS Associated with Hantavirus and Dengue Disease Severity in Barbados
Abstract
1. Introduction
Objectives
2. Methods
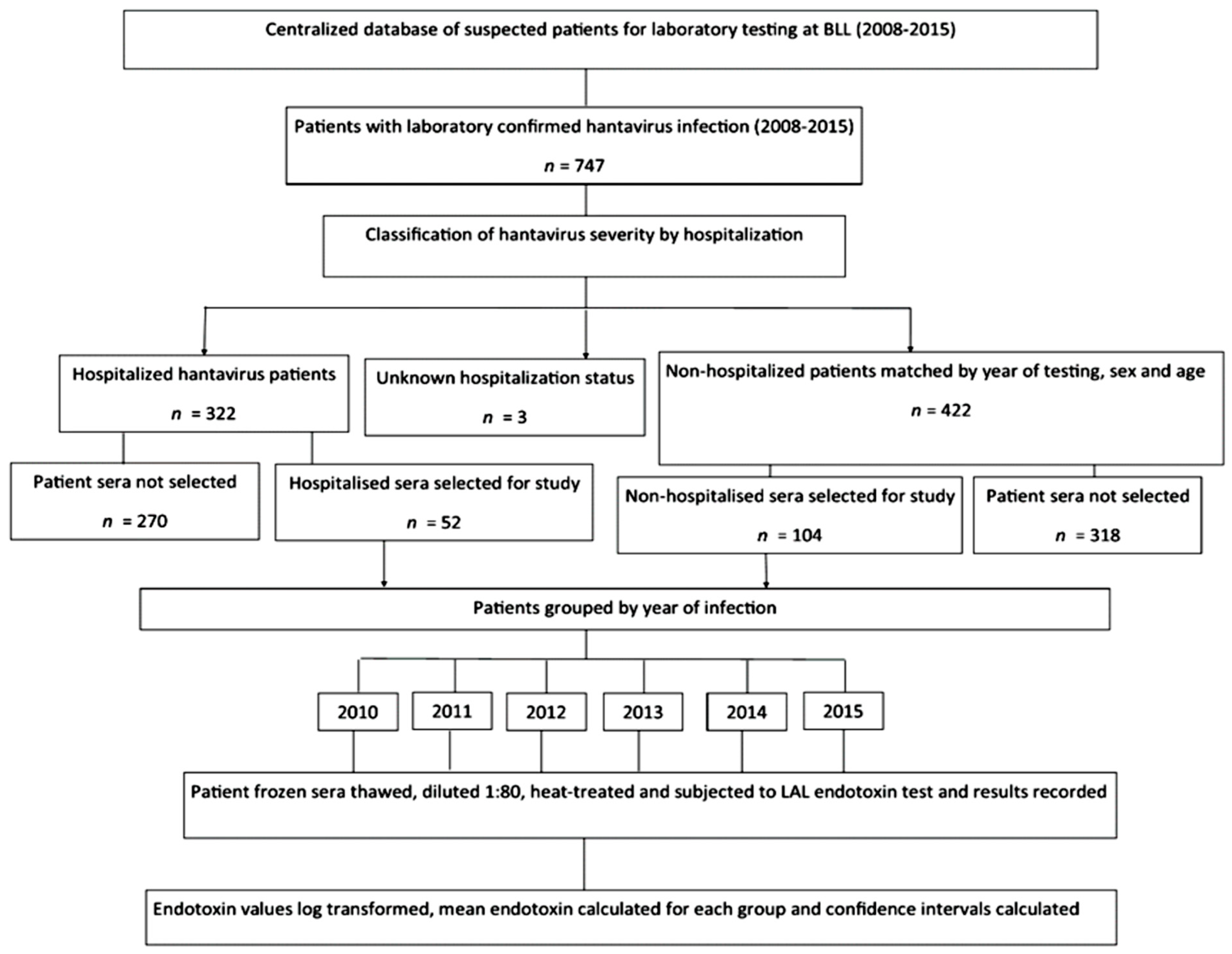
2.1. Hantavirus Serum Endotoxin Study: Cases and Controls Selection
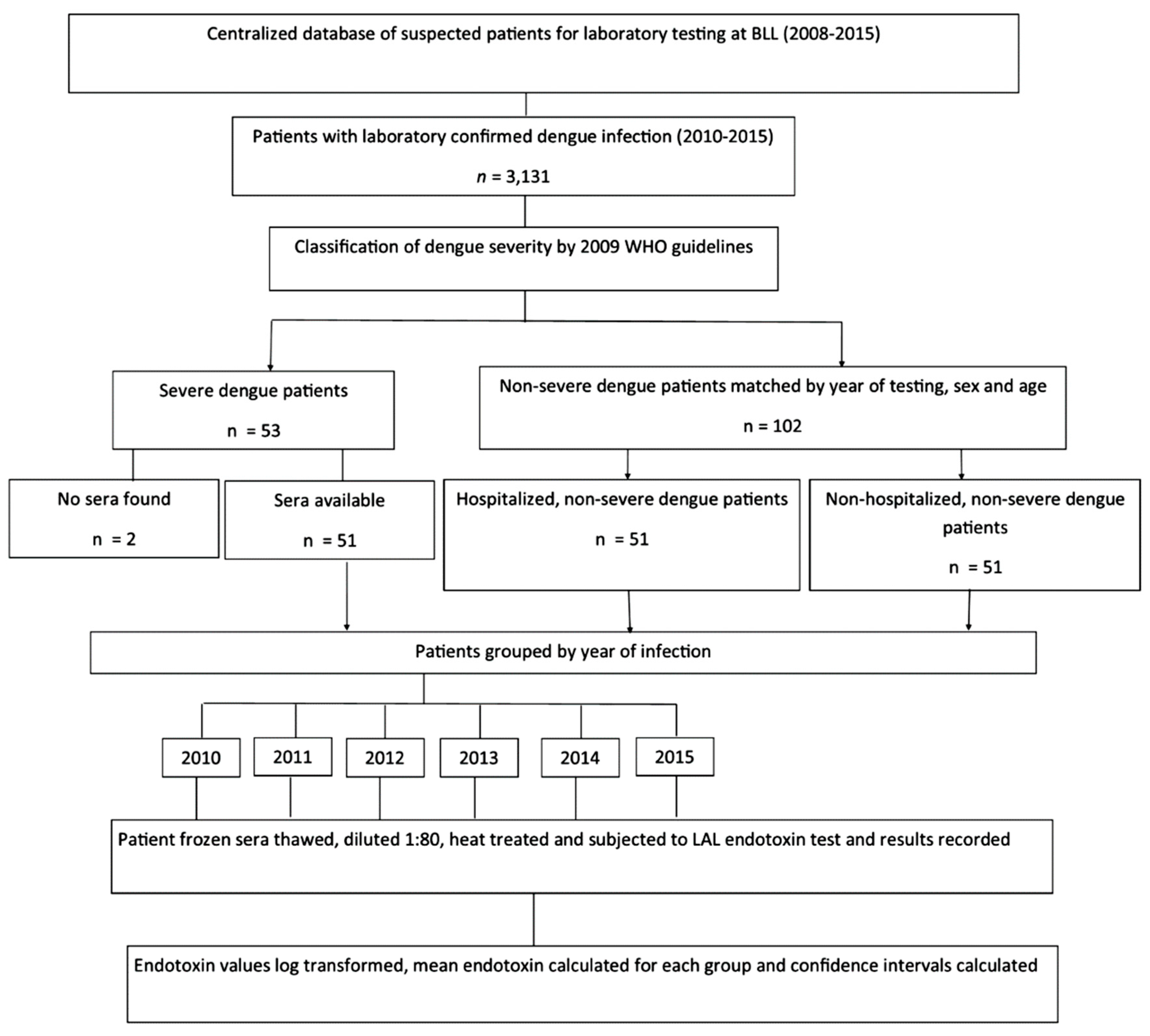
2.2. Serum Endotoxin Study: Dengue Patient Selection
2.3. Analysis of Clinical Laboratory Parameters and Dengue Disease Severity
2.4. Endotoxin Determination in Dengue—and Hantavirus-Infected Patient Sera
2.5. Data Treatment and Statistical Analysis
3. Results
3.1. The Association of Serum Endotoxin Levels and Hantavirus Disease Severity
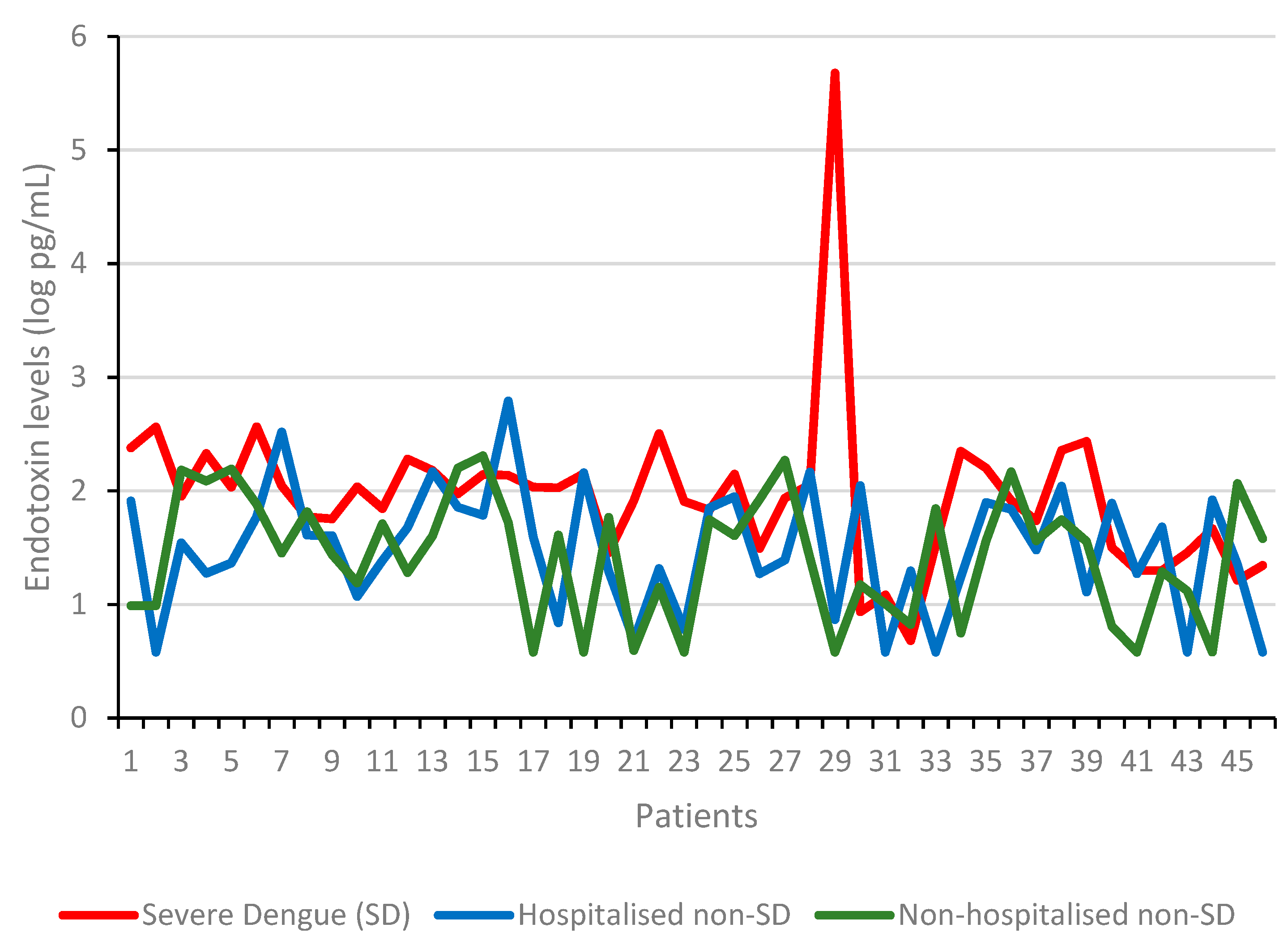
3.2. The Association of Serum Endotoxin Levels and Dengue Disease Severity
3.3. Severe Dengue vs. Hospitalized Non-Severe Dengue
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stanaway, J.D.; Shepard, D.S.; Undurraga, E.A.; Halasa, Y.A.; Coffeng, L.E.; Brady, O.J.; Hay, S.I.; Bedi, N.; Bensenor, I.M.; Castañeda-Orjuela, C.A. The global burden of dengue: An analysis from the Global Burden of Disease Study 2013. Lancet Infect. Dis. 2016, 16, 712–723. [Google Scholar] [CrossRef]
- Manigold, T.; Vial, P. Human hantavirus infections: Epidemiology, clinical features, pathogenesis and immunology. Swiss Med. Wkly. 2014, 144, w13937. [Google Scholar] [CrossRef] [PubMed]
- Muyangwa, M.; Martynova, E.V.; Khaiboullina, S.F.; Morzunov, S.P.; Rizvanov, A.A. Hantaviral proteins: Structure, functions, and role in hantavirus infection. Front. Microbiol. 2015, 6, 1326. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-A.; Kim, W.-k.; No, J.S.; Lee, S.-H.; Lee, S.-Y.; Kim, J.H.; Kho, J.H.; Lee, D.; Song, D.H.; Gu, S.H. Genetic diversity and reassortment of Hantaan virus tripartite RNA genomes in nature, the Republic of Korea. PLoS Negl. Trop. Dis. 2016, 10, e0004650. [Google Scholar] [CrossRef] [PubMed]
- Bi, Z.; Formenty, P.B.; Roth, C.E. Hantavirus infection: A review and global update. J. Infect. Dev. Ctries. 2008, 2, 3–23. [Google Scholar] [CrossRef] [PubMed]
- Hammon, W.M.; Rundnick, A.; Sather, G. Viruses associated with epidemic hemorrhagic fevers of the Philippines and Thailand. Science 1960, 131, 1102–1103. [Google Scholar] [CrossRef] [PubMed]
- Hammon, W.M.; Sather, G.E. Virological Findings in the 1960 Hemorrhagic Fever Epidemic (Dengue) in Thailand. Am. J. Trop. Med. Hyg. 1964, 13, 629–641. [Google Scholar] [CrossRef]
- Oliver, J.; Macdowell, M. The renal lesion in epidemic hemorrhagic fever. J. Clin. Investig. 1957, 36, 99–133. [Google Scholar] [CrossRef]
- Bellon, M.M.; MacLean, J.D. A point source dengue outbreak in Canadian tourists in Barbados. Can. Commun. Dis. Rep. 1998, 24, 161–164. [Google Scholar] [PubMed]
- Branch, S.L.; Levett, P.N. Evaluation of four methods for detection of immunoglobulin M antibodies to dengue virus. Clin. Diagn. Lab. Immunol. 1999, 6, 555–557. [Google Scholar]
- Levett, P.N.; Branch, S.L.; Edwards, C.N. Detection of dengue infection in patients investigated for leptospirosis in Barbados. Am. J. Trop. Med. Hyg. 2000, 62, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Gittens-St Hilaire, M.; Clarke-Greenidge, N. An analysis of the subtypes of dengue fever infections in Barbados 2003–2007 by reverse transcriptase polymerase chain reaction. Virol. J. 2008, 5, 152. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Gittens-St Hilaire, M.; Nielsen, A.L. Epidemiological trends and clinical manifestations of Dengue among children in one of the English-speaking Caribbean countries. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Gittens-St Hilaire, M.; Clarke-Greenidge, N.; Nielsen, A.L. Epidemiological Trend and Clinical Observations among Children and Adults with Dengue in Barbados. West. Indian Med. J. 2015, 64, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Nielsen, A.L. Trends in the patterns of IgM and IgG antibodies in febrile persons with suspected dengue in Barbados, an English-speaking Caribbean country, 2006–2013. J. Infect. Public Health 2015, 8, 583–592. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kumar, A.; Gittens-St Hilair, M.; Jason, V.; Ugwuagu, C.; Krishnamurthy, K. The clinical characteristics and outcome of children hospitalized with dengue in Barbados, an English Caribbean country. J. Infect. Dev. Ctries 2015, 9, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Chadee, D.; Mahabir, R.S.; Sutherland, J.M. Dengue Fever Epidemiology and Control in the Caribbean: A Status Report (2012). Caribb Med. J. 2012, 74, 17–21. [Google Scholar]
- Douglas, K.O.; Martina, B.; Samuels, A.; Gittens-St. Hilaire, M. Epidemiology and Molecular Investigations of Clinical Dengue Infections in Barbados (2008–2016). Am. J. Trop. Med. Hyg. 2019. (manuscript in preparation for publication). [Google Scholar]
- Sharma, S.K.; Seth, T.; Mishra, P.; Gupta, N.; Agrawal, N.; Broor, S.; Mahapatra, M.; Saxena, R. Clinical profile of dengue infection in patients with hematological diseases. Mediterr. J. Hematol. Infect. Dis. 2011, 3, 2011039. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Halstead, S.B. Dengue. Lancet 2007, 370, 1644–1652. [Google Scholar] [CrossRef]
- Huy, N.T.; van Giang, T.; Thuy, D.H.D.; Kikuchi, M.; Hien, T.T.; Zamora, J.; Hirayama, K. Factors associated with dengue shock syndrome: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2013, 7, e2412. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-W.; Lee, I.-K.; Wang, L.; Chen, R.-F.; Yang, K.D. The usefulness of clinical-practice-based laboratory data in facilitating the diagnosis of dengue illness. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Horstick, O.; Martinez, E.; Guzman, M.G.; Martin, J.L.; Ranzinger, S.R. WHO dengue case classification 2009 and its usefulness in practice: An expert consensus in the Americas. Pathog. Glob. Health 2015, 109, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Deen, J.L.; Harris, E.; Wills, B.; Balmaseda, A.; Hammond, S.N.; Rocha, C.; Dung, N.M.; Hung, N.T.; Hien, T.T.; Farrar, J.J. The WHO dengue classification and case definitions: Time for a reassessment. Lancet 2006, 368, 170–173. [Google Scholar] [CrossRef]
- Groen, J.; Koraka, P.; Edwards, C.N.; Branch, S.L.; Douglas, K.O.; Osterhaus, A.D.; Levett, P.N. Serological evidence of hantavirus in humans and rodents in Barbados. J. Infect. 2002, 45, 109–110. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Krishnamurthy, K.; Nielsen, A.L. Hantavirus infection among children hospitalized for febrile illness suspected to be dengue in Barbados. J. Infect. Public Health 2016, 9, 81–87. [Google Scholar] [CrossRef]
- Sharma, R.N.; Guerrero, A.; Seligson, E.O.; Tiwari, K. Antibodies Against Seoul Hantavirus in Brown rats (Rattus norvegicus) from Grenada, West Indies. J. Anim. Res. 2019, 9, 45–49. [Google Scholar] [CrossRef]
- Keenan, L.M.; Chikweto, A.; Sharma, R.N.; Stone, D.M. Seroprevalence of hantavirus in Rattus norvegicus in Grenada, West Indies. West. Indian Vet. J. 2008, 8, 67–71. [Google Scholar]
- Adesiyun, A.; Dookeran, S.; Stewart-Johnson, A.; Rahaman, S.; Bissessar, S.; Thompson, N. Serological Evidence of Hantavirus Infection in Farm and Abattoir Workers in Trinidad—A Preliminary Study. J. Agromed. 2011, 16, 194–199. [Google Scholar] [CrossRef]
- Rovida, F.; Percivalle, E.; Sarasini, A.; Chichino, G.; Baldanti, F. Imported hantavirus cardiopulmonary syndrome in an Italian traveller returning from Cuba. New Microbiol 2013, 36, 103–105. [Google Scholar]
- Matheus, S.; Kallel, H.; Mayence, C.; Bremand, L.; Houcke, S.; Rousset, D.; Lacoste, V.; de Thoisy, B.; Hommel, D.; Lavergne, A. Hantavirus pulmonary syndrome caused by Maripa virus in French Guiana, 2008–2016. Emerg. Infect. Dis. 2017, 23, 1722. [Google Scholar] [CrossRef] [PubMed]
- Gnauck, A.; Lentle, R.G.; Kruger, M.C. The characteristics and function of bacterial lipopolysaccharides and their endotoxic potential in humans. Int. Rev. Immunol. 2015, 35, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Cadieux, J.; Kuzio, J.; Milazzo, F.; Kropinski, A. Spontaneous release of lipopolysaccharide by Pseudomonas aeruginosa. J. Bacteriol. 1983, 155, 817–825. [Google Scholar] [PubMed]
- Williams, K.L. Endotoxins: Pyrogens, LAL Testing and Depyrogenation; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Marshall, J.C. Lipopolysaccharide: An endotoxin or an exogenous hormone? Clin. Infect. Dis. 2005, 41, S470–S480. [Google Scholar] [CrossRef] [PubMed]
- Lundin, J.I.; Checkoway, H. Endotoxin and cancer. Environ. Health Perspect. 2009, 117, 1344. [Google Scholar] [CrossRef] [PubMed]
- Elsbach, P.; Weiss, J. The bactericidal/permeability-increasing protein (BPI), a potent element in host-defense against gram-negative bacteria and lipopolysaccharide. Immunobiology 1993, 187, 417–429. [Google Scholar] [CrossRef]
- Ghoshal, S.; Witta, J.; Zhong, J.; de Villiers, W.; Eckhardt, E. Chylomicrons promote intestinal absorption of lipopolysaccharides. J. Lipid Res. 2009, 50, 90–97. [Google Scholar] [CrossRef]
- Gupta, K.; Subramanian, H.; Ali, H. Modulation of host defense peptide-mediated human mast cell activation by LPS. Innate Immun. 2016, 22, 21–30. [Google Scholar] [CrossRef]
- Aird, W.C. The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome. Blood 2003, 101, 3765–3777. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Wang, S.-Y. Activation of terminally differentiated human monocytes/macrophages by dengue virus: Productive infection, hierarchical production of innate cytokines and chemokines, and the synergistic effect of lipopolysaccharide. J. Virol. 2002, 76, 9877–9887. [Google Scholar] [CrossRef]
- Maleki, K.T.; García, M.; Iglesias, A.; Alonso, D.; Ciancaglini, M.; Hammar, U.; Ljunggren, H.-G.; Schierloh, P.; Martínez, V.P.; Klingström, J. Serum Markers Associated with Severity and Outcome of Hantavirus Pulmonary Syndrome. J. Infect. Dis. 2019, 219, 1832–1840. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Wang, S.-Y.; King, C.-C. Bacterial lipopolysaccharide inhibits dengue virus infection of primary human monocytes/macrophages by blockade of virus entry via a CD14-dependent mechanism. J. Virol. 1999, 73, 2650–2657. [Google Scholar] [PubMed]
- Lustig, S.; Danenberg, H.; Kafri, Y.; Kobiler, D.; Ben-Nathan, D. Viral neuroinvasion and encephalitis induced by lipopolysaccharide and its mediators. J. Exp. Med. 1992, 176. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, J.M.; Douek, D.C. Microbial translocation across the GI tract. Annu. Rev. Immunol. 2012, 30, 149. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, J.M.; Price, D.A.; Schacker, T.W.; Asher, T.E.; Silvestri, G.; Rao, S.; Kazzaz, Z.; Bornstein, E.; Lambotte, O.; Altmann, D. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat. Med. 2006, 12, 1365–1371. [Google Scholar] [CrossRef]
- Van de Weg, C.A.; Koraka, P.; van Gorp, E.C.; Mairuhu, A.T.; Supriatna, M.; Soemantri, A.; van de Vijver, D.A.; Osterhaus, A.D.; Martina, B.E. Lipopolysaccharide levels are elevated in dengue virus infected patients and correlate with disease severity. J. Clin. Virol. 2012, 53, 38–42. [Google Scholar] [CrossRef]
- Jakab, F.; Sebők, J.; Szántó, Z.; Hang, D.; Imre, M.; Németh, V.; Madai, M.; Oldal, M.; Kovács, T.; Wittmann, I. Dobrava–Belgrade hantavirus infection mimics acute appendicitis. J. Clin. Virol. 2011, 50, 164–166. [Google Scholar] [CrossRef]
- Latus, J.; Fritzenkötter, M.; Schmidt-Chanasit, J.; Tenner-Racz, K.; Leibold, T.; Kimmel, M.; Ott, G.; Ting, E.; Alscher, M.D.; Braun, N. Hantavirus and acute appendicitis—The diagnosis behind the diagnosis? J. Clin. Virol. 2012, 53, 156–158. [Google Scholar] [CrossRef]
- Latus, J.; Tenner-Racz, K.; Racz, P.; Kitterer, D.; Cadar, D.; Ott, G.; Alscher, M.D.; Schmidt-Chanasit, J.; Braun, N. Detection of Puumala hantavirus antigen in human intestine during acute hantavirus infection. PLoS ONE 2014, 9, e98397. [Google Scholar] [CrossRef][Green Version]
- Centers for Disease Control and Prevention. Hantavirus Infection, Non-Hantavirus Pulmonary Syndrome. 2015 Case Definition. Available online: https://wwwn.cdc.gov/nndss/conditions/hantavirus-infection-non-hantavirus-pulmonary-syndrome/case-definition/2015/ (accessed on 2 May 2019).
- Douglas, K.O.; Cayol, C.; Forbes, K.; Samuels, A.; Iheozor-Ejiofor, R.; Vapalahti, O.; Sironen, T.; Gittens-St. Hilaire, M. Epidemiology of Clinical Hantavirus Infections and Reservoir Investigations of rodents in Barbados. Viruses 2019. (manuscript accepted with minor changes). [Google Scholar]
- Bennett, B.; Underwood, R. 283. Note: On McNemar’s Test for the 2 × 2 Table and Its Power Function. Biometrics 1970, 26, 339–343. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistics notes: The use of transformation when comparing two means. Br. Med. J. 1996, 312, 1153. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.M.; Altman, D.G. Transformations, means, and confidence intervals. Br. Med. J. 1996, 312, 1079. [Google Scholar] [CrossRef] [PubMed]
- Krautkrämer, E.; Zeier, M.; Plyusnin, A. Hantavirus infection: An emerging infectious disease causing acute renal failure. Kidney Int. 2013, 83, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Braun, N.; Haap, M.; Overkamp, D.; Kimmel, M.; Alscher, M.D.; Lehnert, H.; Haas, C.S. Characterization and outcome following Puumala virus infection: A retrospective analysis of 75 cases. Nephrol. Dial. Transplant. 2010, 25, 2997–3003. [Google Scholar] [CrossRef] [PubMed]
- Vaheri, A.; Strandin, T.; Hepojoki, J.; Sironen, T.; Henttonen, H.; Mäkelä, S.; Mustonen, J. Uncovering the mysteries of hantavirus infections. Nat. Rev. Microbiol. 2013, 11, 539–550. [Google Scholar] [CrossRef] [PubMed]
- Witkowski, P.T.; Perley, C.C.; Brocato, R.L.; Hooper, J.W.; Jürgensen, C.; Schulzke, J.-D.; Krüger, D.H.; Bücker, R. Gastrointestinal Tract As Entry Route for Hantavirus Infection. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Vaheri, A.; Henttonen, H.; Voutilainen, L.; Mustonen, J.; Sironen, T.; Vapalahti, O. Hantavirus infections in Europe and their impact on public health. Rev. Med. Virol. 2013, 23, 35–49. [Google Scholar] [CrossRef]
- Zapata, J.C.; Cox, D.; Salvato, M.S. The role of platelets in the pathogenesis of viral hemorrhagic fevers. PLoS Negl. Trop. Dis. 2014, 8, e2858. [Google Scholar] [CrossRef] [PubMed]
- Van de Weg, C.A.; Pannuti, C.S.; de Araujo, E.S.; van den Ham, H.J.; Andeweg, A.C.; Boas, L.S.; Felix, A.C.; Carvalho, K.I.; de Matos, A.M.; Levi, J.E.; et al. Microbial translocation is associated with extensive immune activation in dengue virus infected patients with severe disease. PLoS Negl. Trop. Dis. 2013, 7, e2236. [Google Scholar] [CrossRef]
- Carrasco, L.R.; Leo, Y.S.; Cook, A.R.; Lee, V.J.; Thein, T.L.; Go, C.J.; Lye, D.C. Predictive tools for severe dengue conforming to World Health Organization 2009 criteria. PLoS Negl. Trop. Dis. 2014, 8, e2972. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhou, Y.; Peng, H.; Zhang, X.; Zhou, F.; Liu, Z.; Chen, X. Predictive symptoms and signs of severe dengue disease for patients with dengue fever: A meta-analysis. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Liu, J.-W.; Yang, K.D. Fatal dengue hemorrhagic fever in adults: Emphasizing the evolutionary pre-fatal clinical and laboratory manifestations. PLoS Negl. Trop. Dis. 2012, 6, e1532. [Google Scholar] [CrossRef] [PubMed]
- Vejchapipat, P.; Theamboonlers, A.; Chongsrisawat, V.; Poovorawan, Y. An evidence of intestinal mucosal injury in dengue infection. Southeast. Asian J. Trop. Med. Public Health 2006, 37, 79. [Google Scholar] [PubMed]
- Usawattanakul, W.; Nimmannitya, S.; Sarabenjawong, K.; Tharavanij, S. Endotoxin and dengue haemorrhagic fever. Southeast. Asian J. Trop. Med. Public Health 1986, 17, 8–12. [Google Scholar] [PubMed]
- Lam, P.K.; van Ngoc, T.; Thuy, T.T.T.; van, N.T.H.; Thuy, T.T.N.; Tam, D.T.H.; Dung, N.M.; Tien, N.T.H.; Kieu, N.T.T.; Simmons, C. The value of daily platelet counts for predicting dengue shock syndrome: Results from a prospective observational study of 2301 Vietnamese children with dengue. PLoS Negl. Trop. Dis. 2017, 11, e0005498. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control; World Health Organization: Geneva, Switherland, 2009. [Google Scholar]
- Chuansumrit, A.; Puripokai, C.; Butthep, P.; Wongtiraporn, W.; Sasanakul, W.; Tangnararatchakit, K.; Chunhakan, S.; Yoksan, S. Laboratory predictors of dengue shock syndrome during the febrile stage. Southeast. Asian J. Trop. Med. Public Health 2010, 41, 326. [Google Scholar] [PubMed]
- Potts, J.A.; Gibbons, R.V.; Rothman, A.L.; Srikiatkhachorn, A.; Thomas, S.J.; Supradish, P.-o.; Lemon, S.C.; Libraty, D.H.; Green, S.; Kalayanarooj, S. Prediction of dengue disease severity among pediatric Thai patients using early clinical laboratory indicators. PLoS Negl. Trop. Dis. 2010, 4, e769. [Google Scholar] [CrossRef] [PubMed]
- Pongpan, S.; Wisitwong, A.; Tawichasri, C.; Patumanond, J. Prognostic indicators for dengue infection severity. Int. J. Clin. Pediatrics 2013, 2, 12–18. [Google Scholar] [CrossRef]
- Wiwanitkit, V.; Manusvanich, P. Can hematocrit and platelet determination on admission predict shock in hospitalized children with dengue hemorrhagic fever? A clinical observation from a small outbreak. Clin. Appl. Thromb. Hemost. 2004, 10, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Assinger, A. Platelets and infection–an emerging role of platelets in viral infection. Front. Immunol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Murgue, B.; Cassar, O.; Guigon, M.; Chungue, E. Dengue virus inhibits human hematopoietic progenitor growth in vitro. J. Infect. Dis. 1997, 175, 1497–1501. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.S.; King, C.C.; Huang, H.S.; Shih, Y.L.; Lee, C.C.; Tsai, W.J.; Yu, C.C.; Chang, H.H. Antiplatelet autoantibodies elicited by dengue virus non-structural protein 1 cause thrombocytopenia and mortality in mice. J. Thromb. Haemost. 2007, 5, 2291–2299. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-F.; Lei, H.Y.; Liu, C.C.; Liu, H.S.; Yeh, T.M.; Anderson, R.; Lin, Y.S. Patient and mouse antibodies against dengue virus nonstructural protein 1 cross-react with platelets and cause their dysfunction or depletion. Am. J. Infect. Dis. 2008, 4, 69–75. [Google Scholar]
- Hottz, E.D.; Lopes, J.F.; Freitas, C.; Valls-de-Souza, R.; Oliveira, M.F.; Bozza, M.T.; da Poian, A.T.; Weyrich, A.S.; Zimmerman, G.A.; Bozza, F.A. Platelets mediate increased endothelium permeability in dengue through NLRP3-inflammasome activation. Blood 2013, 122, 3405–3414. [Google Scholar] [CrossRef] [PubMed]
- Funahara, Y.; Wirawan, R. Features of DIC in dengue hemorrhagic fever. In Disseminated Intravascular Coagulation; Karger Publishers: Berlin, Germany, 1983; Volume 49, pp. 201–211. [Google Scholar]
- Hottz, E.D.; Oliveira, M.F.; Nunes, P.C.; Nogueira, R.M.R.; Valls-de-Souza, R.; da Poian, A.T.; Weyrich, A.S.; Zimmerman, G.A.; Bozza, P.T.; Bozza, F.A. Dengue induces platelet activation, mitochondrial dysfunction and cell death through mechanisms that involve DC-SIGN and caspases. J. Thromb. Haemost. 2013, 11, 951–962. [Google Scholar] [CrossRef] [PubMed]
- Gunawardena, D.; Jayaweera, S.; Madhubhashini, G.; Lokumarakkala, D.D.; Senanayake, S.J. Reliability of Parameters of Complete Blood Count with Different Storage Conditions. J. Clin. Lab. Anal. 2017, 31, e22042. [Google Scholar] [CrossRef]
- Kulkarni, D.; Sharma, B. Dengue Fever-Induced Cold-Agglutinin Syndrome; SAGE Publications Sage UK: London, UK, 2014. [Google Scholar]
- Albuquerque, P.L.; Júnior, G.B.S.; Diógenes, S.S.; Silva, H.F. Dengue and aplastic anemia–A rare association. Travel Med. Infect. Dis. 2009, 7, 118–120. [Google Scholar] [CrossRef]
- Kurukularatne, C.; Dimatatac, F.; Teo, D.; Lye, D.C.; Leo, Y.S. When less is more: Can we abandon prophylactic platelet transfusion in Dengue fever. Ann. Acad. Med. Singap. 2011, 40, 539–545. [Google Scholar]
- Lee, T.-H.; Wong, J.G.; Leo, Y.-S.; Thein, T.-L.; Ng, E.-L.; Lee, L.K.; Lye, D.C. Potential harm of prophylactic platelet transfusion in adult dengue patients. PLoS Negl. Trop. Dis. 2016, 10, e0004576. [Google Scholar] [CrossRef]
- Lye, D.C.; Lee, V.J.; Sun, Y.; Leo, Y.S. Lack of efficacy of prophylactic platelet transfusion for severe thrombocytopenia in adults with acute uncomplicated dengue infection. Clin. Infect. Dis. 2009, 48, 1262–1265. [Google Scholar] [CrossRef] [PubMed]
- Modhiran, N.; Watterson, D.; Muller, D.A.; Panetta, A.K.; Sester, D.P.; Liu, L.; Hume, D.A.; Stacey, K.J.; Young, P.R. Dengue virus NS1 protein activates cells via Toll-like receptor 4 and disrupts endothelial cell monolayer integrity. Sci. Transl. Med. 2015, 7, ra142–ra304. [Google Scholar] [CrossRef] [PubMed]
| Non-Severe DF | Severe Dengue (SD) | Difference | |
|---|---|---|---|
| Mean | 0.705 * | ||
| Sample size | 100 | 50 | |
| Standard Deviation | 2.08 | 0.6147 | |
| Variance | 4.3264 | 0.377856 |
| Hospitalised Hantavirus Patients | Non-Hospitalised Hantavirus Patients | Chi2 | p Value | ||
|---|---|---|---|---|---|
| Number | n = | 52 | 104 | ||
| Sex | Male | 17 (32.7%) | 34 (32.7%) | ||
| Female | 35 (67.5%) | 70 (67.5%) | |||
| Symptoms | Fever | 69.2% | 87.5% | 10.69 Ψ | <0.01 |
| Headache | 53.8% | 74.0% | 8.68 Ψ | <0.01 | |
| Gastrointestinal related symptoms | 57.7% | 53.8% | 0.68 | >0.01 | |
| Joint pain | 34.6% | 53.8% | 7.31 | >0.01 | |
| Myalgia | 28.8% | 39.4% | 2.23 | >0.01 | |
| Retroorbital pain | 26.9% | 29.8% | 0.22 | >0.01 | |
| Respiratory symptoms | 25.0% | 6.7% | 12.05 Ψ | <0.01 | |
| Rash | 11.5% | 10.6% | 0.05 | >0.01 | |
| Thrombocytopenia | 11.5% | 0% | 9.95 Ψ | <0.01 | |
| Liver involvement | 9.6% | 8.7% | 0.06 | >0.01 | |
| Bleeding/haemorrhages | 5.8% | 2.9% | 1.05 | >0.01 | |
| Hantavirus IgM ELISA positive | 52/52 (100%) | 104/104 (100%) | |||
| Hantavirus IgG ELISA positive | 2/52 (3.9%) | 6/104 (5.8%) | |||
| Endotoxin | Mean (pg/mL) | 76.02 (48.10–120.16) Ψ | 24.33 (18.07–32.76) | ||
| Standard Deviation | 4.12 | 3.68 |
| Severe Dengue | Hospitalised Non-Severe Dengue | Non-Hospitalised Non-Severe Dengue | ||
|---|---|---|---|---|
| Number | 51 | 51 | 51 | |
| Sex | Male | 27 (52.9%) | 27 (52.9%) | 27 (52.9%) |
| Female | 24 (47.1%) | 24 (47.1%) | 24 (47.1%) | |
| Endotoxin | Mean (pg/mL) | 86.24 (48.56–153.15) | 31.55 (20.63–48.20) | 26.96 (17.60–41.30) |
| 99% CI | (48.56, 153.15) † | (20.62, 48.20) † | (17.60, 41.30) |
| Number of Patients | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameter | Normal Range | Severe Dengue (SD) | Hospitalised Non-SD | SD OOR + Non-SD OOR | SD OOR + Non-SD IR | SD IR + Non-SD OOR | SD + Non-SD IR | McNemar’s Test p Value | Bonferroni Correction p Value |
| Sodium (Na) | 134–144 mmol/L | 25 | 25 | 5 | 4 | 5 | 11 | 0.7389 | 1 |
| Chloride (Cl) | 94–104 mmol/L | 25 | 25 | 11 | 2 | 4 | 8 | 0.4142 | 1 |
| Urea | 2.5–7.1 mmol/L | 15 | 15 | 6 | 1 | 3 | 5 | 0.3173 | 1 |
| Creatinine | 46–87 µmol/L | 16 | 16 | 4 | 2 | 7 | 3 | 0.0956 | 1 |
| T-Bilirubin | 3–22 µmol/L | 13 | 13 | 3 | 4 | 3 | 3 | 0.7055 | 1 |
| D-Bilirubin | < 5.1 µmol/L | 1 | 1 | 0 | 1 | 0 | 0 | 0.3173 | 1 |
| ALT | 3–35 IU/L | 8 | 8 | 3 | 2 | 3 | 0 | 0.6547 | 1 |
| AST | 10–42 U/L | 11 | 11 | 6 | 4 | 1 | 0 | 0.1797 | 1 |
| Albumin | 39–49 g/L | 4 | 4 | 2 | 0 | 2 | 2 | 0.1573 | 1 |
| Potassium (K) | 2.8–4.1 mmol/L | 23 | 23 | 3 | 7 | 6 | 6 | 0.7815 | 1 |
| Total CO2 | 22–29 mmol/L | 16 | 16 | 6 | 4 | 3 | 3 | 0.7055 | 1 |
| Anion Gap | 4–16 | 16 | 16 | 4 | 2 | 6 | 4 | 0.1573 | 1 |
| Uric Acid | 2 (2.1)–7 (8.5) mmol/L | 0 | 0 | 0 | 0 | 0 | 0 | N/A | N/A |
| Alkaline Phosphatase | 39–106 IU/L | 11 | 11 | 1 | 1 | 0 | 9 | 0.3173 | 1 |
| Corrected Calcium | 2.15–2.51 mmol/L | 2 | 2 | 0 | 0 | 0 | 2 | N/A | N/A |
| Magnesium (Mg) | 0.85–1.1 mmol/L | 1 | 1 | 0 | 1 | 0 | 0 | 0.3173 | 1 |
| Haemoglobin | 11.5–16.5 g/dL | 20 | 20 | 3 | 5 | 5 | 7 | 1 | 1 |
| Haematocrit | 37–47% | 20 | 20 | 2 | 7 | 6 | 5 | 0.7815 | 1 |
| MCH | 27–32 g/dL | 20 | 20 | 2 | 7 | 6 | 5 | 1 | 1 |
| MCV | 76–96 g/dL | 20 | 20 | 0 | 4 | 6 | 10 | 0.5271 | 1 |
| RDWCV | 11.5–14.5% | 20 | 20 | 0 | 6 | 5 | 9 | 0.0253 ** | 1 |
| BASO% | 0–2% | 14 | 14 | 2 | 4 | 3 | 4 | 0.7055 | 1 |
| NEUT% | 37–92% | 15 | 15 | 0 | 4 | 6 | 10 | 0.3173 | 1 |
| MONO% | 0–12% | 18 | 18 | 1 | 3 | 7 | 7 | 0.2059 | 1 |
| LYM% | 10–58.5% | 18 | 18 | 0 | 0 | 5 | 12 | 0.3173 | 1 |
| EOS% | 0–7% | 18 | 18 | 0 | 0 | 0 | 18 | N/a | 1 |
| WBC | 4–11 g/dL | 18 | 18 | 2 | 4 | 3 | 4 | 0.5637 | 1 |
| MCHC | 30–35 g/dL | 18 | 18 | 0 | 6 | 2 | 10 | 0.1573 | 1 |
| Red blood cells | 3.8–5.8 cells/μL | 18 | 18 | 2 | 1 | 3 | 8 | 0.3173 | 1 |
| Platelets | 150–450 × 109/L | 17 | 17 | 8 | 5 | 0 | 4 | 0.0253 ** | 1 |
| Basophils | 0.00–0.2 × 109/L | 15 | 15 | 1 | 3 | 7 | 7 | 0.3173 | 1 |
| Neutrophils | 2–7.8 × 109/L | 15 | 15 | 1 | 5 | 5 | 3 | 1 | 1 |
| Monocytes | 0.0–0.9 × 109/L | 18 | 18 | 1 | 3 | 1 | 13 | 1 | 1 |
| Lymphocytes | 0.6–4.1 × 109/L | 18 | 18 | 1 | 2 | 2 | 13 | 1 | 1 |
| Eosinophils | 0.0–0.7% | 18 | 18 | 0 | 0 | 0 | 18 | N/A | N/A |
| IR Range | 2–3 | 3 | 3 | 0 | 0 | 0 | 3 | N/A | N/A |
| PTT | 23–39 s | 4 | 4 | 1 | 3 | 0 | 0 | 0.0833 | 1 |
| PT | 10.9–13.0 s | 4 | 4 | 3 | 1 | 0 | 0 | 0.3173 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Douglas, K.O.; Samuels, T.A.; Gittens-St. Hilaire, M. Serum LPS Associated with Hantavirus and Dengue Disease Severity in Barbados. Viruses 2019, 11, 838. https://doi.org/10.3390/v11090838
Douglas KO, Samuels TA, Gittens-St. Hilaire M. Serum LPS Associated with Hantavirus and Dengue Disease Severity in Barbados. Viruses. 2019; 11(9):838. https://doi.org/10.3390/v11090838
Chicago/Turabian StyleDouglas, Kirk Osmond, Thelma Alafia Samuels, and Marquita Gittens-St. Hilaire. 2019. "Serum LPS Associated with Hantavirus and Dengue Disease Severity in Barbados" Viruses 11, no. 9: 838. https://doi.org/10.3390/v11090838
APA StyleDouglas, K. O., Samuels, T. A., & Gittens-St. Hilaire, M. (2019). Serum LPS Associated with Hantavirus and Dengue Disease Severity in Barbados. Viruses, 11(9), 838. https://doi.org/10.3390/v11090838





