Human Norovirus Histo-Blood Group Antigen (HBGA) Binding Sites Mediate the Virus Specific Interactions with Lettuce Carbohydrates
Abstract
1. Introduction
2. Materials and Methods
2.1. Production and Purification of HuNoV VLPs
2.2. Generation of GII.4 Mutant VLP
2.3. Extraction of Plant Cell Wall Material (CWM)
2.4. Detection of HuNoV VLP Binding to CWM Using Enzyme-Linked Immunosorbent Assay (ELISA)
2.5. Porcine Gastric Mucin and Human B-type HBGA Saliva ELISA
2.6. Detection of HuNoV VLP Binding to Lettuce Tissues Using Immunofluorescence Assay (IFA)
2.7. Statistical Analyses
3. Results
3.1. Binding of Historical GII.4 VLPs to Lettuce CWM Correlated with Their PGM Binding
3.2. Loss of HBGA Binding Profile for GII.4 D374A and G443A VLP Mutants
3.3. Amino Acid Residues 374 and 443 are Essential for GII.4 Binding to Lettuce
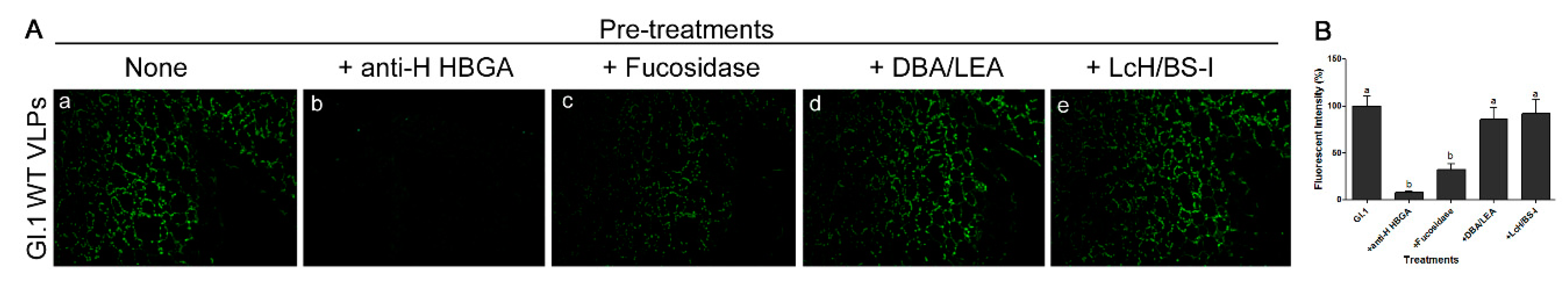
3.4. HuNoV GI.1 Binds to Lettuce Tissues Through a Fucose Moiety on H-like HBGA
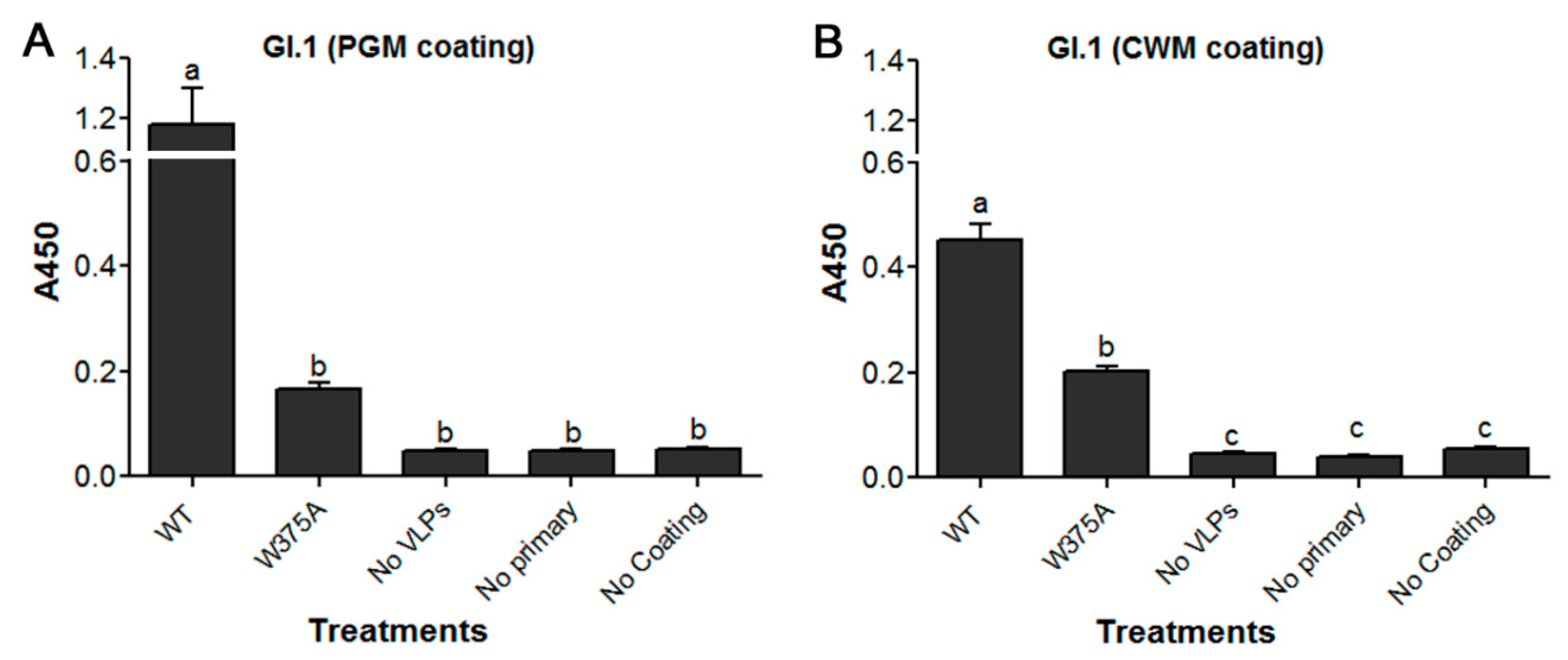
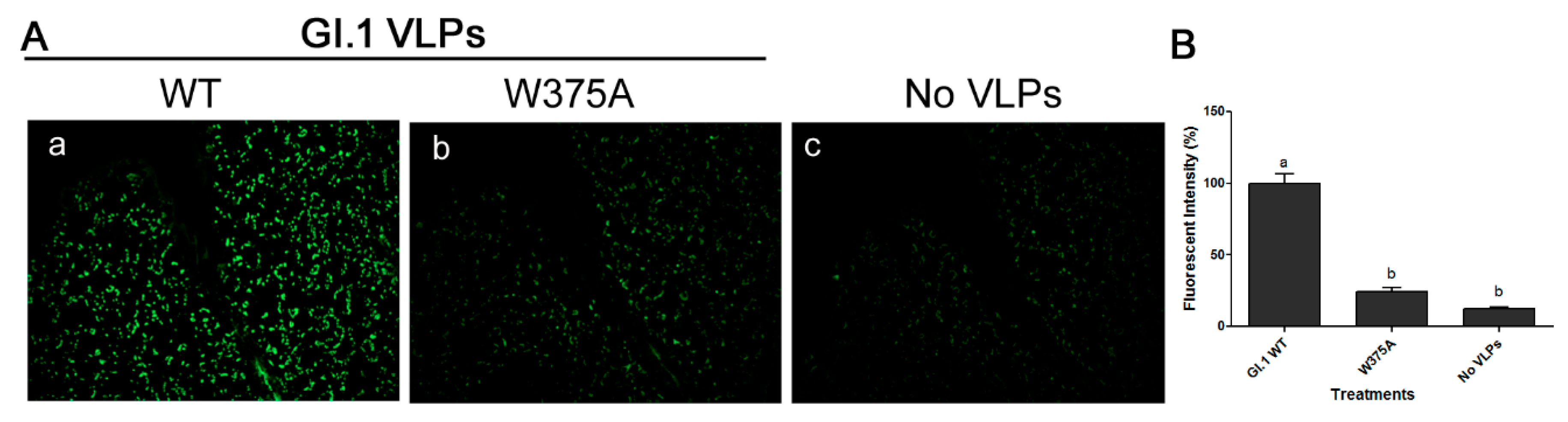
3.5. Amino Acid at Position 375 is Important for GI.1 Binding to Lettuce
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dewey-Mattia, D.; Manikonda, K.; Hall, A.J.; Wise, M.E.; Crowe, S.J. Surveillance for Foodborne Disease Outbreaks—United States, 2009–2015. MMWR Surveill. Summ. 2018, 67, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States—Major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- CDC. Burden of Norovirus Illness in the U.S. Available online: https://www.cdc.gov/norovirus/trends-outbreaks/burden-US.html (accessed on 30 June 2019).
- Hall, A.J.; Wikswo, M.E.; Pringle, K.; Gould, L.H.; Parashar, U.D.; Division of Viral Diseases, N.C.f.I.; Respiratory Diseases, C.D.C. Vital signs: Foodborne norovirus outbreaks—United States, 2009–2012. MMWR Morb. Mortal. Wkly. Rep. 2014, 63, 491–495. [Google Scholar] [PubMed]
- Mallory, M.L.; Lindesmith, L.C.; Graham, R.L.; Baric, R.S. GII.4 Human Norovirus: Surveying the Antigenic Landscape. Viruses 2019, 11, 177. [Google Scholar] [CrossRef] [PubMed]
- Hoa Tran, T.N.; Trainor, E.; Nakagomi, T.; Cunliffe, N.A.; Nakagomi, O. Molecular epidemiology of noroviruses associated with acute sporadic gastroenteritis in children: Global distribution of genogroups, genotypes and GII.4 variants. J. Clin. Virol. 2013, 56, 185–193. [Google Scholar] [CrossRef]
- Atmar, R.L.; Baehner, F.; Cramer, J.P.; Lloyd, E.; Sherwood, J.; Borkowski, A.; Mendelman, P.M.; Group, N.O.R.S. Antibody persistence to two Virus-Like Particle norovirus vaccine candidate formulations in healthy adults: One-year follow-up with memory probe vaccination. J. Infect. Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.; Liebowitz, D.; Lin, K.; Kasparek, K.; Pasetti, M.F.; Garg, S.J.; Gottlieb, K.; Trager, G.; Tucker, S.N. Safety and immunogenicity of an oral tablet norovirus vaccine, a phase I randomized, placebo-controlled trial. JCI Insight 2018, 3. [Google Scholar] [CrossRef]
- Atmar, R.L.; Ramani, S.; Estes, M.K. Human noroviruses: Recent advances in a 50-year history. Curr. Opin. Infect. Dis. 2018, 31, 422–432. [Google Scholar] [CrossRef]
- Bartsch, S.M.; Lopman, B.A.; Ozawa, S.; Hall, A.J.; Lee, B.Y. Global Economic Burden of Norovirus Gastroenteritis. PLoS ONE 2016, 11, e0151219. [Google Scholar] [CrossRef]
- European Food Safety Authority; European Center for Disease Prevention and Control. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2010. Euro Surveill. 2012, 17, 10. [Google Scholar]
- Gil, M.I.; Selma, M.V.; Suslow, T.; Jacxsens, L.; Uyttendaele, M.; Allende, A. Pre- and postharvest preventive measures and intervention strategies to control microbial food safety hazards of fresh leafy vegetables. Crit. Rev. Food Sci. Nutr. 2015, 55, 453–468. [Google Scholar] [CrossRef] [PubMed]
- Taban, B.M.; Halkman, A.K. Do leafy green vegetables and their ready-to-eat [RTE] salads carry a risk of foodborne pathogens? Anaerobe 2011, 17, 286–287. [Google Scholar] [CrossRef] [PubMed]
- Esseili, M.A.; Gao, X.; Tegtmeier, S.; Saif, L.J.; Wang, Q. Abiotic stress and Phyllosphere Bacteria Influence the Survival of Human Norovirus and its surrogates on Preharvest Leafy Greens. Appl. Environ. Microbiol. 2015, 10, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Esseili, M.A.; Chin, A.; Saif, L.; Miller, S.A.; Qu, F.; Lewis Ivey, M.L.; Wang, Q. Postharvest Survival of Porcine Sapovirus, a Human Norovirus Surrogate, on Phytopathogen-Infected Leafy Greens. J. Food Prot. 2015, 78, 1472–1480. [Google Scholar] [CrossRef] [PubMed]
- Esseili, M.A.; Wang, Q.; Saif, L.J. Binding of human GII.4 norovirus virus-like particles to carbohydrates of romaine lettuce leaf cell wall materials. Appl. Environ. Microbiol. 2012, 78, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Esseili, M.A.; Meulia, T.; Saif, L.J.; Wang, Q. Tissue Distribution and Visualization of Internalized Human Norovirus in Leafy Greens. Appl. Environ. Microbiol. 2018, 84, e00292-18. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Esseili, M.A.; Lu, Z.; Saif, L.J.; Wang, Q. Recognition of Histo-Blood Group Antigen-Like Carbohydrates in Lettuce by Human GII.4 Norovirus. Appl. Environ. Microbiol. 2016, 82, 2966–2974. [Google Scholar] [CrossRef]
- Marionneau, S.; Cailleau-Thomas, A.; Rocher, J.; Le Moullac-Vaidye, B.; Ruvoen, N.; Clement, M.; Le Pendu, J. ABH and Lewis histo-blood group antigens, a model for the meaning of oligosaccharide diversity in the face of a changing world. Biochimie 2001, 83, 565–573. [Google Scholar] [CrossRef]
- Ettayebi, K.; Crawford, S.E.; Murakami, K.; Broughman, J.R.; Karandikar, U.; Tenge, V.R.; Neill, F.H.; Blutt, S.E.; Zeng, X.L.; Qu, L.; et al. Replication of human noroviruses in stem cell-derived human enteroids. Science 2016, 353, 1387–1393. [Google Scholar] [CrossRef]
- Donaldson, E.F.; Lindesmith, L.C.; Lobue, A.D.; Baric, R.S. Viral shape-shifting: Norovirus evasion of the human immune system. Nat. Rev. Microbiol. 2010, 8, 231–241. [Google Scholar] [CrossRef]
- Choi, J.M.; Hutson, A.M.; Estes, M.K.; Prasad, B.V. Atomic resolution structural characterization of recognition of histo-blood group antigens by Norwalk virus. Proc. Natl. Acad. Sci. USA 2008, 105, 9175–9180. [Google Scholar] [CrossRef] [PubMed]
- Debbink, K.; Donaldson, E.F.; Lindesmith, L.C.; Baric, R.S. Genetic mapping of a highly variable norovirus GII.4 blockade epitope: Potential role in escape from human herd immunity. J. Virol. 2012, 86, 1214–1226. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, E.F.; Lindesmith, L.C.; Lobue, A.D.; Baric, R.S. Norovirus pathogenesis: Mechanisms of persistence and immune evasion in human populations. Immunol. Rev. 2008, 225, 190–211. [Google Scholar] [CrossRef] [PubMed]
- Shanker, S.; Choi, J.M.; Sankaran, B.; Atmar, R.L.; Estes, M.K.; Prasad, B.V. Structural analysis of histo-blood group antigen binding specificity in a norovirus GII.4 epidemic variant: Implications for epochal evolution. J. Virol. 2011, 85, 8635–8645. [Google Scholar] [CrossRef] [PubMed]
- Bu, W.; Mamedova, A.; Tan, M.; Xia, M.; Jiang, X.; Hegde, R.S. Structural basis for the receptor binding specificity of Norwalk virus. J. Virol. 2008, 82, 5340–5347. [Google Scholar] [CrossRef] [PubMed]
- Debbink, K.; Lindesmith, L.C.; Donaldson, E.F.; Baric, R.S. Norovirus immunity and the great escape. PLoS Pathog. 2012, 8, e1002921. [Google Scholar] [CrossRef] [PubMed]
- Lindesmith, L.C.; Debbink, K.; Swanstrom, J.; Vinje, J.; Costantini, V.; Baric, R.S.; Donaldson, E.F. Monoclonal antibody-based antigenic mapping of norovirus GII.4-2002. J. Virol. 2012, 86, 873–883. [Google Scholar] [CrossRef]
- Lindesmith, L.C.; Donaldson, E.F.; Lobue, A.D.; Cannon, J.L.; Zheng, D.P.; Vinje, J.; Baric, R.S. Mechanisms of GII.4 norovirus persistence in human populations. PLoS Med. 2008, 5, e31. [Google Scholar] [CrossRef]
- Lindesmith, L.C.; Donaldson, E.; Leon, J.; Moe, C.L.; Frelinger, J.A.; Johnston, R.E.; Weber, D.J.; Baric, R.S. Heterotypic humoral and cellular immune responses following Norwalk virus infection. J. Virol. 2010, 84, 1800–1815. [Google Scholar] [CrossRef]
- Takanashi, S.; Wang, Q.; Chen, N.; Shen, Q.; Jung, K.; Zhang, Z.; Yokoyama, M.; Lindesmith, L.C.; Baric, R.S.; Saif, L.J. Characterization of emerging GII.g/GII.12 noroviruses from a gastroenteritis outbreak in the United States in 2010. J. Clin. Microbiol. 2011, 49, 3234–3244. [Google Scholar] [CrossRef]
- Jung, K.; Wang, Q.; Kim, Y.; Scheuer, K.; Zhang, Z.; Shen, Q.; Chang, K.O.; Saif, L.J. The effects of simvastatin or interferon-alpha on infectivity of human norovirus using a gnotobiotic pig model for the study of antivirals. PLoS ONE 2012, 7, e41619. [Google Scholar] [CrossRef] [PubMed]
- Lindesmith, L.C.; Donaldson, E.F.; Beltramello, M.; Pintus, S.; Corti, D.; Swanstrom, J.; Debbink, K.; Jones, T.A.; Lanzavecchia, A.; Baric, R.S. Particle conformation regulates antibody access to a conserved GII.4 norovirus blockade epitope. J. Virol. 2014, 88, 8826–8842. [Google Scholar] [CrossRef] [PubMed]
- Tian, P.; Brandl, M.; Mandrell, R. Porcine gastric mucin binds to recombinant norovirus particles and competitively inhibits their binding to histo-blood group antigens and Caco-2 cells. Lett. Appl. Microbiol. 2005, 41, 315–320. [Google Scholar] [CrossRef] [PubMed]
- de Rougemont, A.; Ruvoen-Clouet, N.; Simon, B.; Estienney, M.; Elie-Caille, C.; Aho, S.; Pothier, P.; Le Pendu, J.; Boireau, W.; Belliot, G. Qualitative and quantitative analysis of the binding of GII.4 norovirus variants onto human blood group antigens. J. Virol. 2011, 85, 4057–4070. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Lou, Z.; Tan, M.; Chen, Y.; Liu, Y.; Zhang, Z.; Zhang, X.C.; Jiang, X.; Li, X.; Rao, Z. Structural basis for the recognition of blood group trisaccharides by norovirus. J. Virol. 2007, 81, 5949–5957. [Google Scholar] [CrossRef]
- Chen, Y.; Tan, M.; Xia, M.; Hao, N.; Zhang, X.C.; Huang, P.; Jiang, X.; Li, X.; Rao, Z. Crystallography of a Lewis-binding norovirus, elucidation of strain-specificity to the polymorphic human histo-blood group antigens. PLoS Pathog. 2011, 7, e1002152. [Google Scholar] [CrossRef] [PubMed]
- Ethelberg, S.; Lisby, M.; Bottiger, B.; Schultz, A.C.; Villif, A.; Jensen, T.; Olsen, K.E.; Scheutz, F.; Kjelso, C.; Muller, L. Outbreaks of gastroenteritis linked to lettuce, Denmark, January 2010. Euro Surveill. 2010, 15, 6. [Google Scholar]
- Makary, P.; Maunula, L.; Niskanen, T.; Kuusi, M.; Virtanen, M.; Pajunen, S.; Ollgren, J.; Minh, N.N.T. Multiple norovirus outbreaks among workplace canteen users in Finland, July 2006. Epidemiol. Infect. 2009, 137, 402–407. [Google Scholar] [CrossRef]
- Oogane, T.; Hirata, A.; Funatogawa, K.; Kobayashi, K.; Sato, T.; Kimura, H. Food poisoning outbreak caused by norovirus GII/4 in school lunch, Tochigi prefecture, Japan. Jpn. J. Infect. Dis. 2008, 61, 423–424. [Google Scholar]
- Schmid, D.; Stuger, H.P.; Lederer, I.; Pichler, A.M.; Kainz-Arnfelser, G.; Schreier, E.; Allerberger, F. A foodborne norovirus outbreak due to manually prepared salad, Austria 2006. Infection 2007, 35, 232–239. [Google Scholar] [CrossRef]
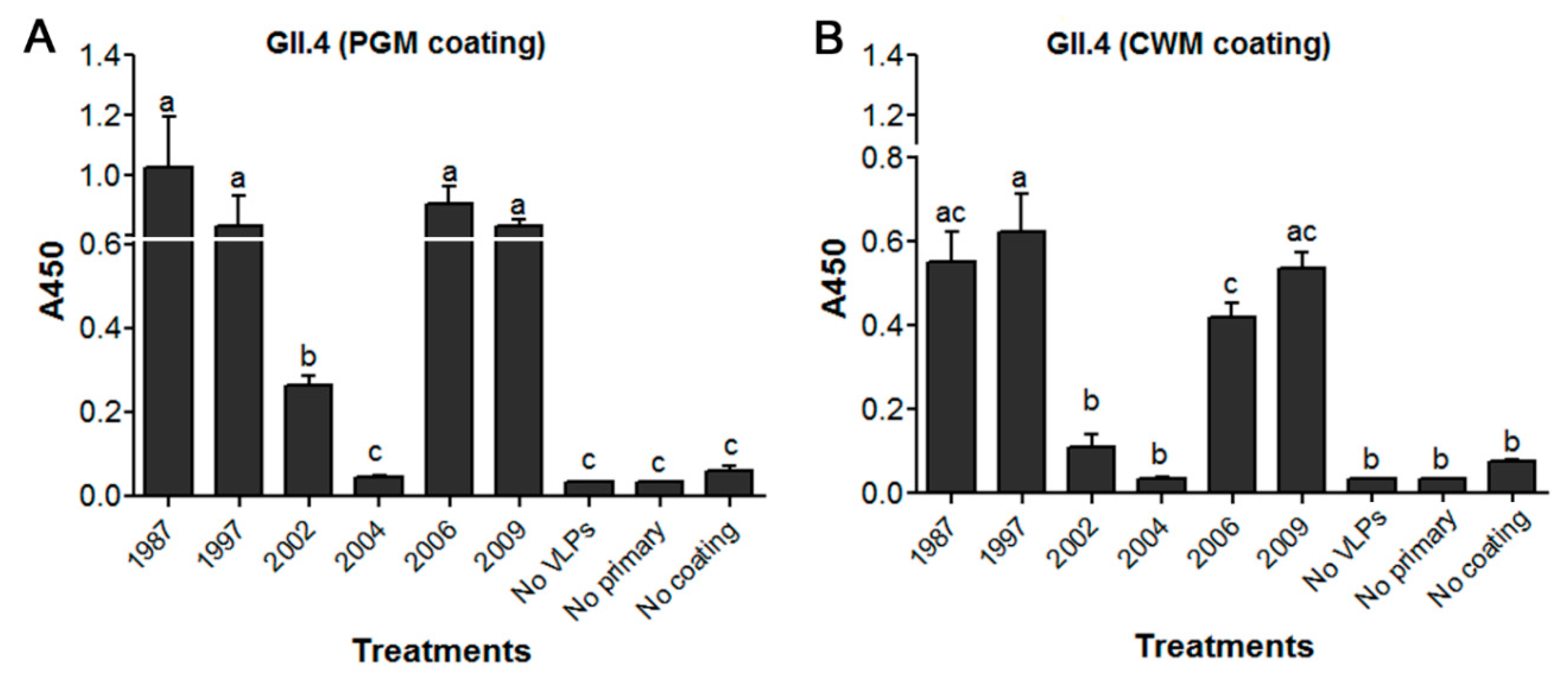
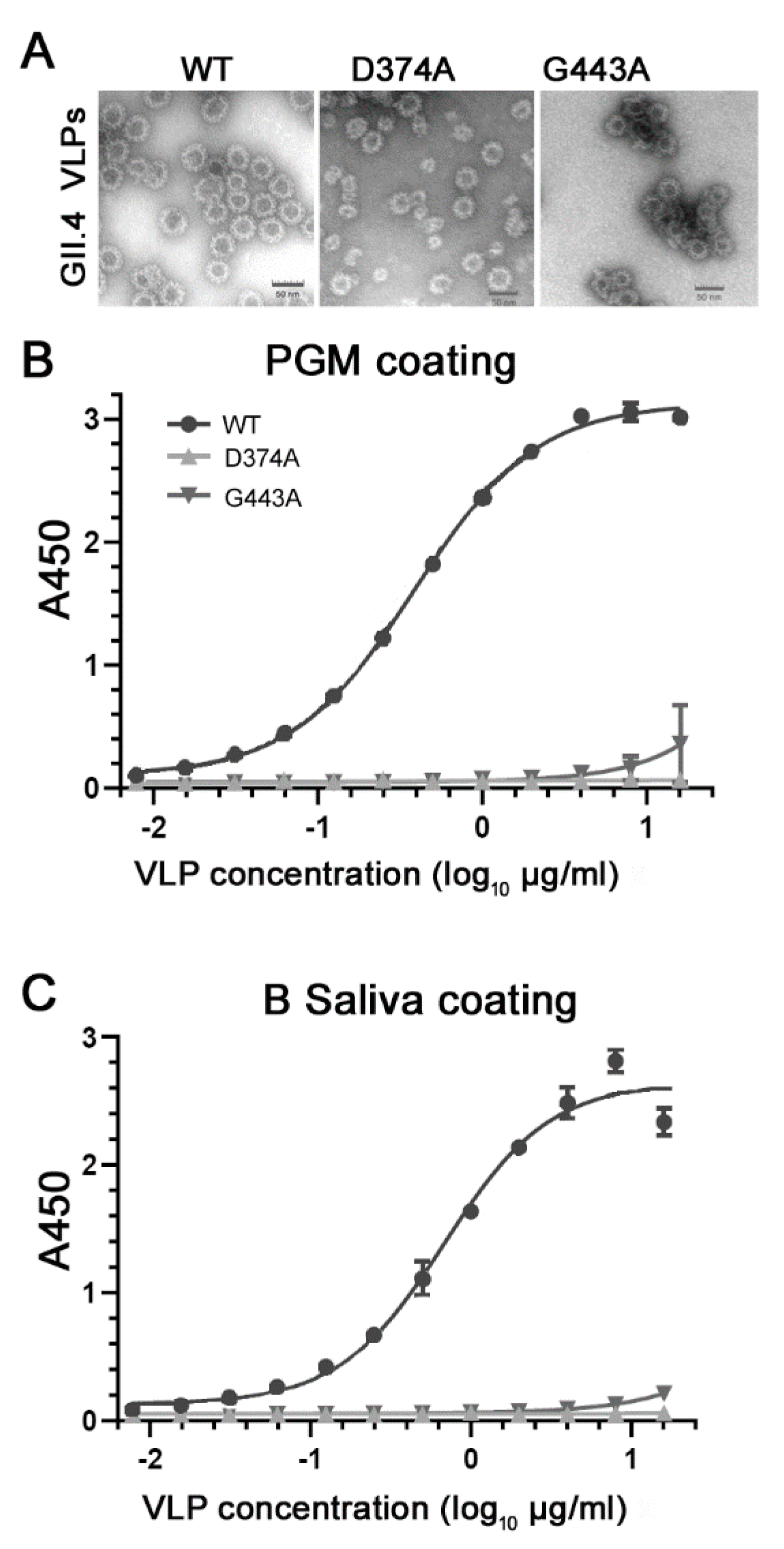
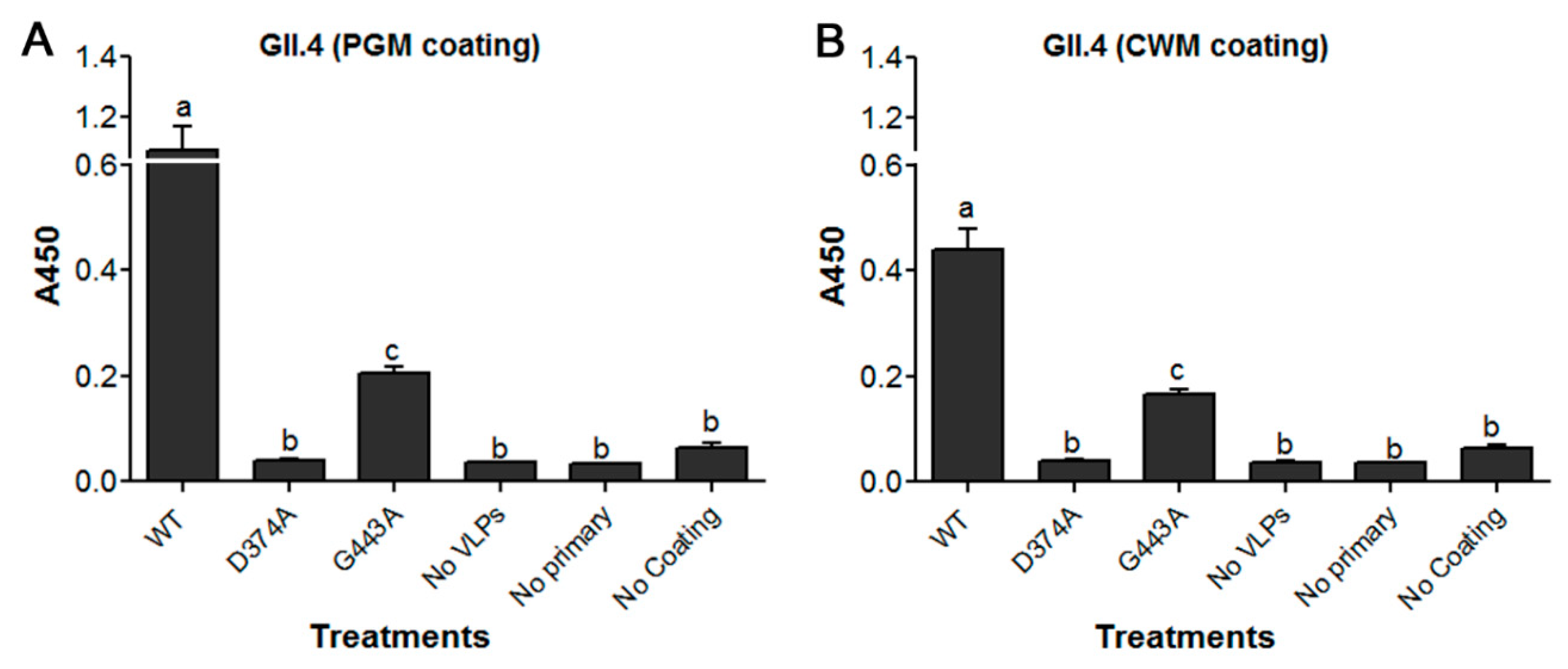
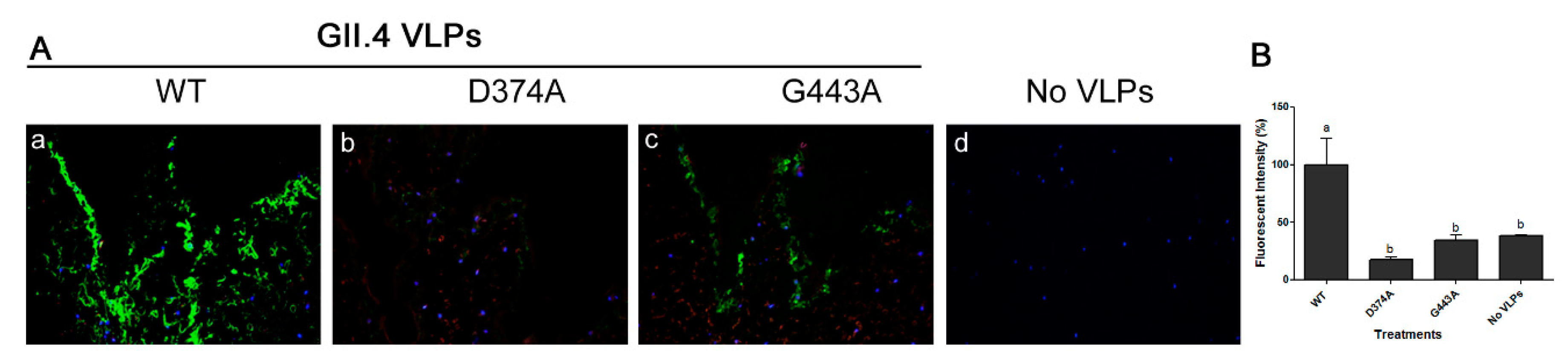
| Genotype | Year | Strain | GenBank Accession No. | VLP Variant | VLP Name | HBGA Profile |
|---|---|---|---|---|---|---|
| GI.1 | 1968 | Norwalk | M87661 | Hu/US/1968/GI.1/Norwalk | GI.1.1968 | H1, H3 and A |
| GII.4 | 1987 | Camberwell | AAK50355.1 | Hu/GII.4/MD145-12/1987/US | GII.4.1987 | H3 and Ley |
| 1997 | US95/96 | AFJ04707.1 | Hu/GII.4/1997/USA | GII.4.1997 | H3, A, B, Ley and Leb | |
| 2002 | Farmington Hill | AFJ04708.1 | Hu/GII.4/Farmington Hills/2002/USA | GII.4.2002 | Ley, H3 and B | |
| 2004 | Hunter | AAZ31376.2 | Hu/GII.4/Hunter 284E/2004/AU | GII.4.2004 | None | |
| 2006 | Minerva | AFJ04709.1 | Hu/GII.4/Minerva/2006/USA | GII.4.2006 | H3, B and A | |
| 2009 | HS194 | GU325839 | Hu/GII.4/Minerva/2006/US | GII.4.HS194 | H3, B, H1, Ley, A and Leb |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esseili, M.A.; Gao, X.; Boley, P.; Hou, Y.; Saif, L.J.; Brewer-Jensen, P.; Lindesmith, L.C.; Baric, R.S.; Atmar, R.L.; Wang, Q. Human Norovirus Histo-Blood Group Antigen (HBGA) Binding Sites Mediate the Virus Specific Interactions with Lettuce Carbohydrates. Viruses 2019, 11, 833. https://doi.org/10.3390/v11090833
Esseili MA, Gao X, Boley P, Hou Y, Saif LJ, Brewer-Jensen P, Lindesmith LC, Baric RS, Atmar RL, Wang Q. Human Norovirus Histo-Blood Group Antigen (HBGA) Binding Sites Mediate the Virus Specific Interactions with Lettuce Carbohydrates. Viruses. 2019; 11(9):833. https://doi.org/10.3390/v11090833
Chicago/Turabian StyleEsseili, Malak A., Xiang Gao, Patricia Boley, Yixuan Hou, Linda J. Saif, Paul Brewer-Jensen, Lisa C. Lindesmith, Ralph S. Baric, Robert L. Atmar, and Qiuhong Wang. 2019. "Human Norovirus Histo-Blood Group Antigen (HBGA) Binding Sites Mediate the Virus Specific Interactions with Lettuce Carbohydrates" Viruses 11, no. 9: 833. https://doi.org/10.3390/v11090833
APA StyleEsseili, M. A., Gao, X., Boley, P., Hou, Y., Saif, L. J., Brewer-Jensen, P., Lindesmith, L. C., Baric, R. S., Atmar, R. L., & Wang, Q. (2019). Human Norovirus Histo-Blood Group Antigen (HBGA) Binding Sites Mediate the Virus Specific Interactions with Lettuce Carbohydrates. Viruses, 11(9), 833. https://doi.org/10.3390/v11090833







