A Methodology for Determining Which Diseases Warrant Care in a High-Level Containment Care Unit †
Abstract
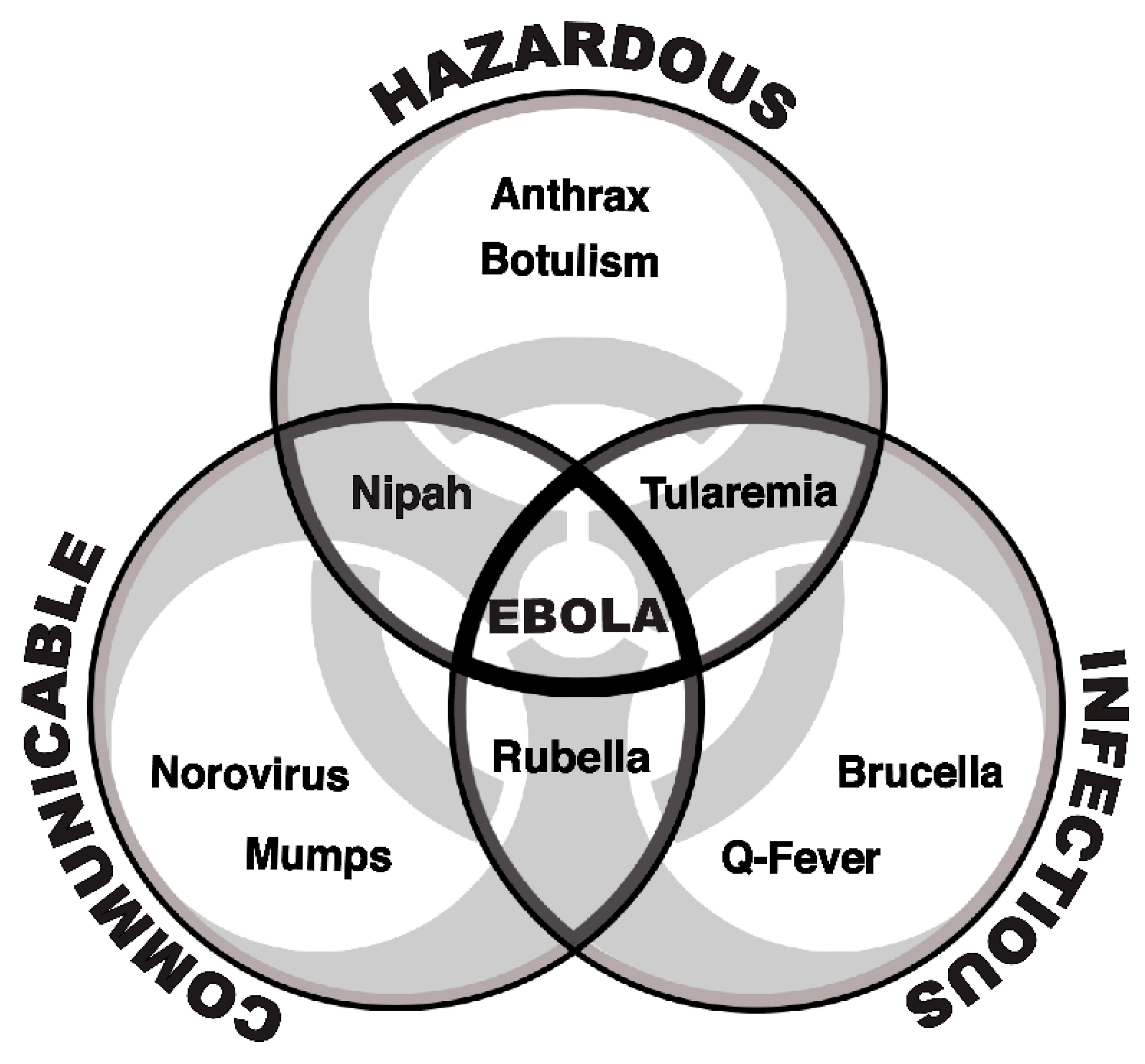
1. Introduction
2. Background
3. Methods
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Smith, P.W.; Anderson, A.O.; Christopher, G.W.; Cieslak, T.J.; Devreede, G.J.; Fosdick, G.A.; Greiner, C.B.; Hauser, J.M.; Hinrichs, S.H.; Huebner, K.D.; et al. Designing a biocontainment unit to care for patients with serious communicable diseases: A consensus statement. Biosecur. Bioterror. 2006, 4, 351–365. [Google Scholar] [CrossRef] [PubMed]
- Bell, B.P.; Damon, I.K.; Jernigan, D.B.; Jernigan, D.B.; Kenyon, T.A.; Nichol, S.T.; O’Connor, J.P.; Tappero, J.W. Overview Control Strategies, and Lessons Learned in the CDC Response to the 2014–2016 Ebola Epidemic. MMWR Suppl. 2016, 65 (Suppl. 3), 4–11. [Google Scholar] [CrossRef] [PubMed]
- Uyeki, T.M.; Mehta, A.K.; Davey, R.T.; Liddell, A.M.; Wolf, T.; Vetter, P.; Schmiedel, S.; Grünewald, T.; Jacobs, M.; Arribas, J.R.; et al. Clinical management of Ebola virus disease in the United States and Europe. N. Engl. J. Med. 2016, 374, 636–646. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Krumkamp, R.; Reintjes, R. Controlling SARS: A review on China’s response compared with other SARS-affected countries. Trop. Med. Int. Health 2009, 14, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Herstein, J.J.; Biddinger, P.D.; Kraft, C.S.; Saiman, L.; Gibbs, S.G.; Smith, P.W.; Hewlett, A.L.; Lowe, J.L. Initial costs of Ebola treatment centers in the United States. Emerg. Infect. Dis. 2016, 22, 350–352. [Google Scholar] [CrossRef]
- Togertt, W.D.; Benenson, A.S.; Gochenour, W.S. Airborne Q-fever. Bacteriol. Rev. 1961, 25, 285–293. [Google Scholar]
- Edmunds, W.J.; Gay, N.J.; Kretzschmar, M.; Peabody, R.G.; Wachman, H. The pre-vaccination epidemiology of measles, mumps, and rubella in Europe: implications for modeling studies. Epidemiol. Infect. 2000, 125, 635–650. [Google Scholar] [CrossRef]
- Bui, T.; Kocher, J.; Li, Y.; Wen, K.; Li, G.; Liu, F.; Yang, X.; LeRoith, T.; Tan, M.; Xia, M.; et al. Median infectious dose of human norovirus GII.4 in gnotobiotic pigs is decreased by simvastatin treatment and increased by age. J. Gen. Virol. 2013, 94, 2005–2016. [Google Scholar] [CrossRef]
- Watson, A.; Kair, D. Information on which to base risk assessments of risk from environments contaminated with anthrax spores. Epidemiol. Infect. 1994, 113, 479–490. [Google Scholar] [CrossRef]
- United States Army Medical Research Institute of Infectious Diseases. In Medical Management of Biological Casualties Handbook, 8th ed.; United States Army Medical Research Institute of Infectious Diseases: Frederick, MD, USA, 2014.
- Stuart, B.M.; Pullen, R.L. Tularemic pneumonia: Review of the American literature and report of 15 additional cases. Am. J. Med. Sci. 1945, 210, 223–236. [Google Scholar] [CrossRef]
- Egan, J.R.; Hall, I.M.; Leach, S. Modeling inhalational tularemia: Deliberate release and public health response. Biosecur. Bioterror. 2011, 9, 331–343. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Measles. Available online: https://www.who.int/news-room/fact-sheets/detail/measles (accessed on 3 July 2019).
- Cieslak, T.J.; Herstein, J.J.; Kortepeter, M.G. Communicable Diseases and Emerging Pathogens: The Past, Present, and Future of High-Level Containment Care. In Planning for Bioemergencies: A Guide for Healthcare Facilities; [Chapter 1]; Hewlett, A., Murthy, A.R.K., Eds.; Springer: New York, NY, USA, 2018. [Google Scholar]
- Franz, D.R.; Jahrling, P.B.; McClain, D.J.; Hoover, D.L.; Byrne, W.R.; Pavlin, J.A.; Christopher, G.W.; Eitzen, E.M., Jr. Clinical recognition and management of patients exposed to biological warfare agents. Clin. Lab. Med. 2001, 21, 435–473. [Google Scholar] [CrossRef]
- Camacho, A.; Kucharski, A.J.; Funk, S.; Breman, J.; Piot, P.; Edmunds, W.J. Potential for large outbreaks of Ebola virus disease. Epidemics 2014, 9, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Chowell, G.; Hengartner, N.W.; Castillo-Chavez, C.; Fenimore, P.W.; Hyman, J.M. The basic reproductive number of Ebola and the effects of public health measures: The cases of Congo and Uganda. J. Theor. Biol. 2004, 229, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Chowell, G.; Nishiura, H. Transmission dynamics and control of Ebola virus disease (EVD): A review. BMC Med. 2014, 12, 196. [Google Scholar] [CrossRef]
- Lekone, P.E.; Finkenstadt, B.F. Statistical inference in a stochastic epidemic SEIR model with control intervention: Ebola as a case study. Biometrics 2006, 62, 1170–1177. [Google Scholar] [CrossRef] [PubMed]
- Gomes, M.F.; Pastore, Y.; Piontti, A.; Rossi, L.; Chao, D.; Longini, I.; Halloran, M.E.; Vespignani, A. Assessing the international spreading risk associated with the 2014 west african ebola outbreak. PLoS Curr. 2014, 6. [Google Scholar] [CrossRef]
- Althaus, C.L. Estimating the Reproduction Number of Ebola Virus (EBOV) during the 2014 Outbreak in West Africa. PLoS Curr. 2014, 6. [Google Scholar] [CrossRef]
- Khan, A.; Naveed, M.; Dur-E-Ahmad, M.; Imran, M. Estimating the basic reproductive ratio for the Ebola outbreak in Liberia and Sierra Leone. Infect. Dis. Poverty 2015, 4, 13. [Google Scholar] [CrossRef]
- Ajelli, M.; Merler, S. Transmission potential and design of adequate control measures for Marburg hemorrhagic fever. PLoS ONE 2012, 7, e50948. [Google Scholar] [CrossRef]
- Zhao, S. Large-scale Lassa fever outbreaks in Nigeria: Quantifying the association between disease reproduction number and local rainfall. BioRxiv 2019. [Google Scholar] [CrossRef]
- Hay, S.I.; Battle, K.E.; Pigott, D.M.; Smith, D.L.; Moyes, C.L.; Bhatt, S.; Collier, N.; Myers, M.F.; George, D.B.; Gething, P.W. Global mapping of infectious disease. Philos. Trans. R. Soc. Lond B Biol. Sci. 2013, 368, 20120250. [Google Scholar] [CrossRef] [PubMed]
- LeDuc, J.W. Epidemiology of hemorrhagic fever viruses. Rev. Infect. Dis. 1989, 11 (Suppl. 4), S730–S735. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, C.A. Crimean-Congo hemorrhagic fever. Antivir. Res. 2004, 64, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Kwok, K.O.; Tang, A.; Wei, V.W.I.; Park, W.H.; Yeoh, E.K.; Riley, S. Epidemic Models of Contact Tracing: Systematic Review of Transmission Studies of Severe Acute Respiratory Syndrome and Middle East Respiratory Syndrome. Comput. Struct. Biotechnol. J. 2019, 17, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Wallinga, J.; Teunis, P. Different epidemic curves for severe acute respiratory syndrome reveal similar impacts of control measures. Am. J. Epidemiol. 2004, 160, 509–516. [Google Scholar] [CrossRef]
- Lipsitch, M.; Cohen, T.; Cooper, B.; Robins, J.M.; Ma, S.; James, L.; Gopalakrishna, G.; Chew, S.K.; Tan, C.C.; Samore, M.H.; et al. Transmission dynamics and control of severe acute respiratory syndrome. Science 2003, 300, 1966–1970. [Google Scholar] [CrossRef] [PubMed]
- Riley, S.; Fraser, C.; Donnelly, C.A.; Ghani, A.C.; Abu-Raddad, L.J.; Hedley, A.J.; Leung, G.M.; Ho, L.M.; Lam, T.H.; Thach, T.Q.; et al. Transmission dynamics of the etiological agent of SARS in Hong Kong: Impact of public health interventions. Science 2003, 300, 1961–1966. [Google Scholar] [CrossRef]
- Nishiura, H.; Miyamatsu, Y.; Chowell, G.; Saitoh, M. Assessing the risk of observing multiple generations of Middle East respiratory syndrome (MERS) cases given an imported case. Eurosurveillance 2015, 20. [Google Scholar] [CrossRef]
- Breban, R.; Riou, J.; Fontanet, A. Interhuman transmissibility of Middle East respiratory syndrome coronavirus: Estimation of pandemic risk. Lancet 2013, 382, 694–699. [Google Scholar] [CrossRef]
- Majumder, M.S.; Rivers, C.; Lofgren, E.; Fisman, D. Estimation of MERS-Coronavirus Reproductive Number and Case Fatality Rate for the Spring 2014 Saudi Arabia Outbreak: Insights from Publicly Available Data. PLoS Curr. 2014, 6. [Google Scholar] [CrossRef]
- Hsieh, Y.H. 2015 Middle East Respiratory Syndrome Coronavirus (MERS-CoV) nosocomial outbreak in South Korea: Insights from modeling. PeerJ 2015, 3, e1505. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.J. Estimation of basic reproduction number of the Middle East respiratory syndrome coronavirus (MERS-CoV) during the outbreak in South Korea, 2015. Biomed. Eng. Online 2017, 16, 79. [Google Scholar] [CrossRef] [PubMed]
- Bui, C.; Bethmont, A.; Chughtai, A.A.; Gardner, L.; Sarkar, S.; Hassan, S.; Seale, H.; MacIntyre, C.R. A Systematic Review of the Comparative Epidemiology of Avian and Human Influenza A H5N1 and H7N9—Lessons and Unanswered Questions. Transbound. Emerg. Dis. 2016, 63, 602–620. [Google Scholar] [CrossRef] [PubMed]
- Nicas, M.; Hubbard, A.E.; Jones, R.M.; Reingold, A.L. The infectious dose of variola (smallpox) virus. Appl. Biosaf. 2004, 9, 118–127. [Google Scholar] [CrossRef]
- Rosenbloom, M.; Leikin, J.B.; Vogel, S.N.; Chaudry, Z.A. Biological and chemical agents: A brief synopsis. Am. J. Ther. 2002, 9, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Gani, R.; Leach, S. Transmission potential of smallpox in contemporary populations. Nature 2001, 414, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Fine, P.E. Herd immunity: History, theory, practice. Epidemiol. Rev. 1993, 15, 265–302. [Google Scholar] [CrossRef]
- Blumberg, S.; Lloyd-Smith, J.O. Inference of R(0) and transmission heterogeneity from the size distribution of stuttering chains. PLoS Comput. Biol. 2013, 9, e1002993. [Google Scholar] [CrossRef]
- Nikolay, B.; Salje, H.; Hossain, M.J.; Khan, A.K.M.D.; Sazzad, H.M.S.; Rahman, M.; Daszak, P.; Ströher, U.; Pulliam, J.R.C.; Kilpatrick, A.M.; et al. Transmission of Nipah Virus—14 Years of Investigations in Bangladesh. N. Engl. J. Med. 2019, 380, 1804–1814. [Google Scholar] [CrossRef]
- Gani, R.; Leach, S. Epidemiologic determinants for modeling pneumonic plague outbreaks. Emerg. Infect. Dis. 2004, 10, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H.; Schwehm, M.; Kakehashi, M.; Eichner, M. Transmission potential of primary pneumonic plague: Time inhomogeneous evaluation based on historical documents of the transmission network. J. Epidemiol. Community Health 2006, 60, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Tsuzuki, S.; Lee, H.; Miura, F.; Chan, Y.H.; Jung, S.M.; Akhmetzhanov, A.R.; Nishiura, H. Dynamics of the pneumonic plague epidemic in Madagascar, August to October 2017. Eur. Surveill. 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Randremanana, R.; Andrianaivoarimanana, V.; Nikolay, B.; Ramasindrazana, B.; Paireau, J.; Ten Bosch, Q.A. Epidemiological characteristics of an urban plague epidemic in Madagascar, August-November, 2017, an outbreak report. Lancet Infect. Dis. 2019, 19, 537–545. [Google Scholar] [CrossRef]
- Majumder, M.S.; Cohn, E.L.; Santillana, M.; Brownstein, J.S. Estimation of Pneumonic Plague Transmission in Madagascar, August-November 2017. PLoS Curr. 2018, 10. [Google Scholar] [CrossRef]
- Pfyffer, G.E.; Palicova, F. Mycobacterium: General Characteristics, Laboratory Detection, and Staining Procedures. In Manual of Clinical Microbiology, 9th ed.; Murray, P.R., Ed.; ASM Press: Washington, DC, USA, 2007; pp. 543–572. [Google Scholar]
- Basu, S.; Friedland, G.H.; Medlock, J.; Andrews, J.R.; Shah, N.S.; Gandhi, N.R.; Moll, A.; Moodley, P.; Sturm, A.W.; Galvani, A.P. Averting epidemics of extensively drug-resistant tuberculosis. Proc. Natl. Acad. Sci. USA 2009, 106, 7672–7677. [Google Scholar] [CrossRef] [PubMed]
| Pathogen | Mechanism of PTP Spread | ID50 | R0 | References |
|---|---|---|---|---|
| Ebola | Blood & Body Fluids | 1-10 aerosolized organisms | 1.3–2.53 | [15,16,17,18,19,20,21,22] |
| Marburg | Blood & Body Fluids | 1-10 aerosolized organisms | 1.59 | [11,23] |
| Lassa | Blood & Body Fluids | 1-10 aerosolized organisms | 1.23–1.33 | [11,24] |
| Lujo | Scant data; Presumably Blood & Body Fluids | No data | No data | |
| Junin | Blood & Body Fluids | No data | <1 | [25,26] |
| Machupo | Blood & Body Fluids | No data | <1 | [21,22] |
| Guanarito | Scant data; Presumably Blood & Body Fluids | No data | No data | |
| Sabia | No data | No data | <1 | [21] |
| CCHF | Blood & Body Fluids | No data | <1 | [21,27] |
| SARS | Respiratory Droplets; Possibly Droplet Nuclei | No data | 2.2–3.6 | [28,29,30,31] |
| MERS | Respiratory Droplets; Possibly Droplet Nuclei | No data | 0.60–11.5 1 | [32,33,34,35,36] |
| H5N1 Influenza | Respiratory Droplets; Possibly Droplet Nuclei | 1000 viral particles 2 | 1.14 | [37] |
| H7N9 Influenza | Respiratory Droplets; Possibly Droplet Nuclei | 1000 viral particles | 0.1–0.47 | [37] |
| Smallpox | Droplet Nuclei, Scabs | 1–100 | 3.5–7.0 | [38,39,40,41] |
| Monkeypox | Respiratory Droplets; Possibly Droplet Nuclei and Scabs | No data | 0.32 | [42] |
| Nipah | Respiratory Droplets | No data | 0.33 | [43] |
| Hendra | No Data | No data | No data | |
| Pneumonic Plague | Respiratory Droplets | 100 to 500 organisms by inhalation | 1.3–3.5 | [44,45,46,47,48] |
| XDR-TB | Droplet Nuclei | <10 bacilli 3 | 1.97 | [49,50] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cieslak, T.J.; Herstein, J.J.; Kortepeter, M.G.; Hewlett, A.L. A Methodology for Determining Which Diseases Warrant Care in a High-Level Containment Care Unit. Viruses 2019, 11, 773. https://doi.org/10.3390/v11090773
Cieslak TJ, Herstein JJ, Kortepeter MG, Hewlett AL. A Methodology for Determining Which Diseases Warrant Care in a High-Level Containment Care Unit. Viruses. 2019; 11(9):773. https://doi.org/10.3390/v11090773
Chicago/Turabian StyleCieslak, Theodore J., Jocelyn J. Herstein, Mark G. Kortepeter, and Angela L. Hewlett. 2019. "A Methodology for Determining Which Diseases Warrant Care in a High-Level Containment Care Unit" Viruses 11, no. 9: 773. https://doi.org/10.3390/v11090773
APA StyleCieslak, T. J., Herstein, J. J., Kortepeter, M. G., & Hewlett, A. L. (2019). A Methodology for Determining Which Diseases Warrant Care in a High-Level Containment Care Unit. Viruses, 11(9), 773. https://doi.org/10.3390/v11090773




