The Clinical Perspective on Hepatitis E
Abstract
1. Introduction
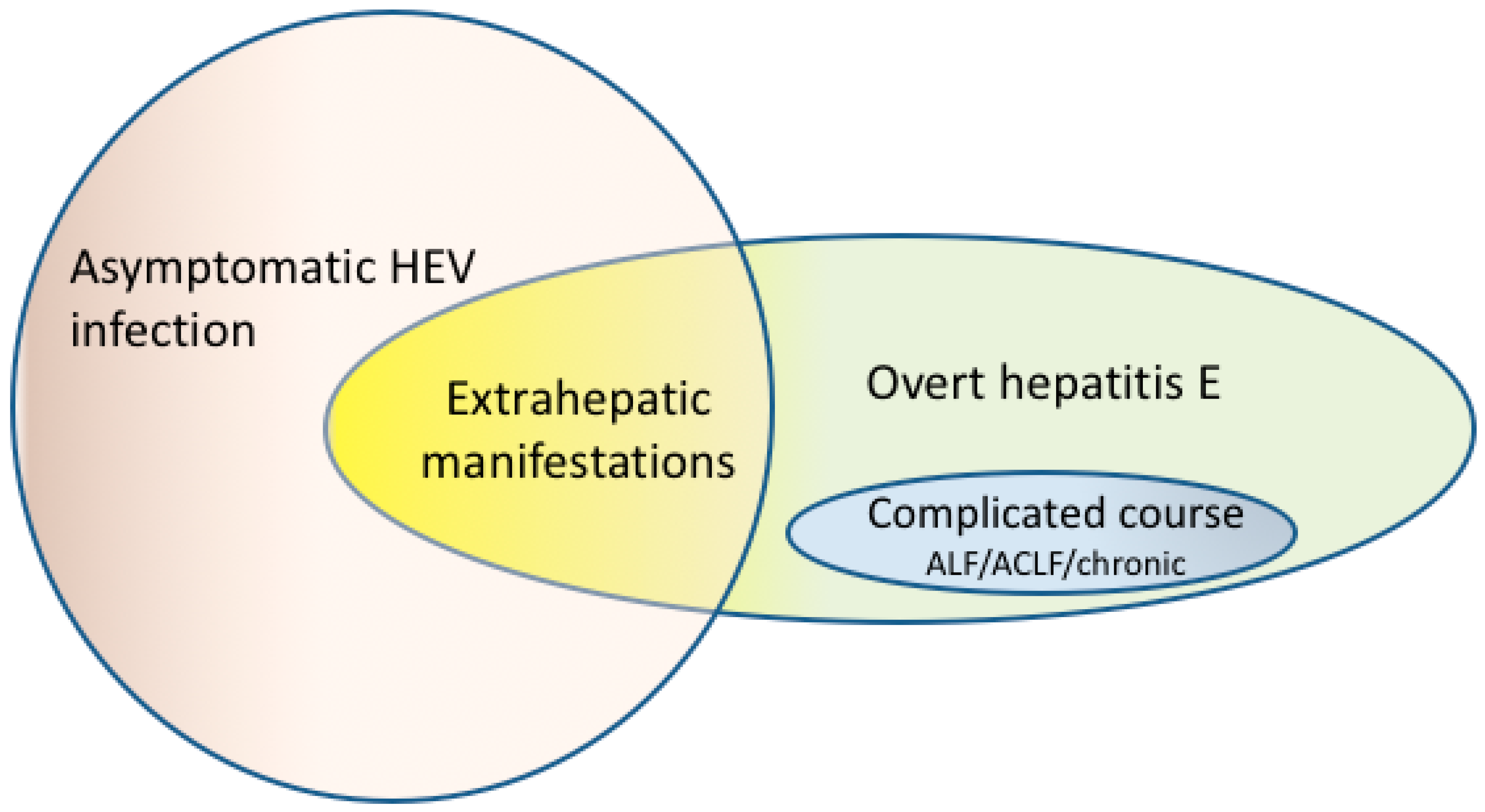
2. Acute Hepatitis E
2.1. Autochthonous HEV Infections in Immunocompetent Individuals in Industrialized Countries
2.2. Hepatitis E as Cause of ACLF
2.3. Hepatitis E in the Tropics
2.4. Hepatitis E during Pregnancy
2.5. Acute and Chronic HEV Infections in Organ Transplant Recipients
3. Hepatitis E in HIV Infected Patients
4. Chronic Hepatitis E beyond Transplant Recipients or HIV Infected Patients
5. Extrahepatic Manifestations of Hepatitis E
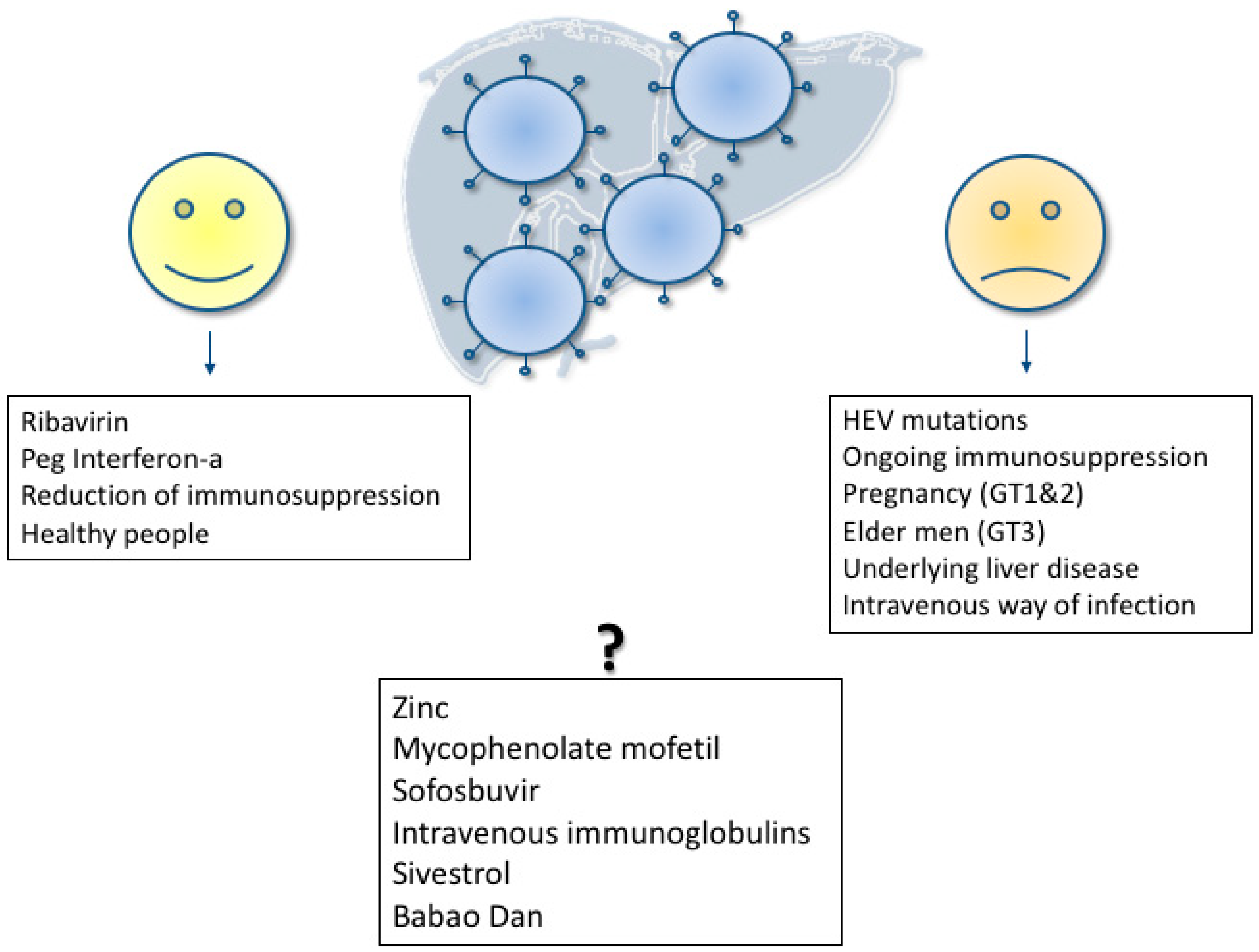
6. Treatment of Acute Hepatitis E
7. Treatment of Chronic Hepatitis E
8. Vaccine
9. Conclusions and Recommendations
Funding
Conflicts of Interest
References
- Adlhoch, C.; Avellon, A.; Baylis, S.A.; Ciccaglione, A.R.; Couturier, E.; de Sousa, R.; Epstein, J.; Ethelberg, S.; Faber, M.; Feher, A.; et al. Hepatitis e virus: Assessment of the epidemiological situation in humans in europe, 2014/15. J. Clin. Virol. 2016, 82, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Pischke, S.; Hartl, J.; Pas, S.D.; Lohse, A.W.; Jacobs, B.C.; Van der Eijk, A.A. Hepatitis e virus: Infection beyond the liver? J. Hepatol. 2017, 66, 1082–1095. [Google Scholar] [CrossRef] [PubMed]
- Hoofnagle, J.H.; Nelson, K.E.; Purcell, R.H. Hepatitis e. N. Engl. J. Med. 2012, 367, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Rein, D.B.; Stevens, G.A.; Theaker, J.; Wittenborn, J.S.; Wiersma, S.T. The global burden of hepatitis e virus genotypes 1 and 2 in 2005. Hepatology 2012, 55, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Imagawa, T.; Sugiyama, R.; Shiota, T.; Li, T.C.; Yoshizaki, S.; Wakita, T.; Ishii, K. Evaluation of heating conditions for inactivation of hepatitis e virus genotypes 3 and 4. J. Food Prot. 2018, 81, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Wedemeyer, H.; Pischke, S.; Manns, M.P. Pathogenesis and treatment of hepatitis e virus infection. Gastroenterology 2012, 142, 1388–1397. [Google Scholar] [CrossRef] [PubMed]
- Terio, V.; Bottaro, M.; Pavoni, E.; Losio, M.N.; Serraino, A.; Giacometti, F.; Martella, V.; Mottola, A.; di Pinto, A.; Tantillo, G. Occurrence of hepatitis a and e and norovirus gi and gii in ready-to-eat vegetables in italy. Int. J. Food Microbiol. 2017, 249, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Maunula, L.; Kaupke, A.; Vasickova, P.; Soderberg, K.; Kozyra, I.; Lazic, S.; van der Poel, W.H.; Bouwknegt, M.; Rutjes, S.; Willems, K.A.; et al. Tracing enteric viruses in the european berry fruit supply chain. Int. J. Food Microbiol. 2013, 167, 177–185. [Google Scholar] [CrossRef]
- Oeser, C.; Vaughan, A.; Said, B.; Ijaz, S.; Tedder, R.; Haywood, B.; Warburton, F.; Charlett, A.; Elson, R.; Morgan, D. Epidemiology of hepatitis e in england and wales: A 10-year retrospective surveillance study, 2008–2017. J. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Hunter, J.G.; Madden, R.G.; Stone, A.M.; Osborne, N.; Wheeler, B.; Vine, L.; Dickson, A.; Barlow, M.; Lewis, J.; Bendall, R.P.; et al. Coastal clustering of hev; Cornwall, UK. Eur. J. Gastroenterol. Hepatol. 2016, 28, 323–327. [Google Scholar] [CrossRef]
- Purpari, G.; Macaluso, G.; Di Bella, S.; Gucciardi, F.; Mira, F.; di Marco, P.; Lastra, A.; Petersen, E.; La Rosa, G.; Guercio, A. Molecular characterization of human enteric viruses in food, water samples, and surface swabs in sicily. Int. J. Infect. Dis. 2019, 80, 66–72. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, Z.; Crossan, C.; Craft, J.; Scobie, L. First report of the presence of hepatitis e virus in scottish-harvested shellfish purchased at retail level. Food Environ. Virol. 2018, 10, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Romalde, J.L.; Rivadulla, E.; Varela, M.F.; Barja, J.L. An overview of 20 years of studies on the prevalence of human enteric viruses in shellfish from galicia, spain. J. Appl. Microbiol. 2018, 124, 943–957. [Google Scholar] [CrossRef] [PubMed]
- Pischke, S.; Hiller, J.; Lutgehetmann, M.; Polywka, S.; Rybczynski, M.; Ayuk, F.; Lohse, A.W. Blood-borne hepatitis e virus transmission: A relevant risk for immunosuppressed patients. Clin. Infect. Dis. 2016, 63, 569–570. [Google Scholar] [CrossRef] [PubMed]
- Mallet, V.; Sberro-Soussan, R.; Vallet-Pichard, A.; Roque-Afonso, A.M.; Pol, S. Transmission of hepatitis e virus by plasma exchange: A case report. Ann. Intern. Med. 2016, 164, 851–852. [Google Scholar] [CrossRef] [PubMed]
- Mullhaupt, B.; Niederhauser, C. Hepatitis e blood donor screening-more than a mere drop in the ocean? J. Hepatol. 2018, 69, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Horvatits, T.; Westholter, D.; Peine, S.; Schulze Zur Wiesch, J.; Lohse, A.W.; Lutgehetmann, M.; Pischke, S. Lack of evidence for human serum albumin as major source of hev infections. Transfus. Med. 2018, 28, 470–471. [Google Scholar] [CrossRef] [PubMed]
- Westholter, D.; Hiller, J.; Denzer, U.; Polywka, S.; Ayuk, F.; Rybczynski, M.; Horvatits, T.; Gundlach, S.; Blocker, J.; Schulze Zur Wiesch, J.; et al. Hev-positive blood donations represent a relevant infection risk for immunosuppressed recipients. J. Hepatol. 2018, 69, 36–42. [Google Scholar] [CrossRef]
- Riveiro-Barciela, M.; Sauleda, S.; Quer, J.; Salvador, F.; Gregori, J.; Piron, M.; Rodriguez-Frias, F.; Buti, M. Red blood cell transfusion-transmitted acute hepatitis e in an immunocompetent subject in europe: A case report. Transfusion 2017, 57, 244–247. [Google Scholar] [CrossRef]
- Riveiro-Barciela, M.; Bes, M.; Quer, J.; Valcarcel, D.; Piriz, S.; Gregori, J.; Llorens, M.; Salcedo, M.T.; Piron, M.; Esteban, R.; et al. Thrombotic thrombocytopenic purpura relapse induced by acute hepatitis e transmitted by cryosupernatant plasma and successfully controlled with ribavirin. Transfusion 2018, 58, 2501–2505. [Google Scholar] [CrossRef]
- Woo, P.C.; Lau, S.K.; Teng, J.L.; Tsang, A.K.; Joseph, M.; Wong, E.Y.; Tang, Y.; Sivakumar, S.; Xie, J.; Bai, R.; et al. New hepatitis e virus genotype in camels, the middle east. Emerg. Infect. Dis. 2014, 20, 1044–1048. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.B.; Simmonds, P.; Izopet, J.; Oliveira-Filho, E.F.; Ulrich, R.G.; Johne, R.; Koenig, M.; Jameel, S.; Harrison, T.J.; Meng, X.J.; et al. Proposed reference sequences for hepatitis e virus subtypes. J. Gen. Virol. 2016, 97, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.G.; Williams, N.J.; Jennings, D.; Chantrey, J.; Verin, R.; Grierson, S.; McElhinney, L.M.; Bennett, M. First detection of hepatitis e virus (orthohepevirus c) in wild brown rats (rattus norvegicus) from great britain. Zoonoses Public Health 2019. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, S.; Yip, C.C.Y.; Wu, S.; Cai, J.; Zhang, A.J.; Leung, K.H.; Chung, T.W.H.; Chan, J.F.W.; Chan, W.M.; Teng, J.L.L.; et al. Rat hepatitis e virus as cause of persistent hepatitis after liver transplant. Emerg. Infect. Dis. 2018, 24, 2241–2250. [Google Scholar] [CrossRef] [PubMed]
- Andonov, A.; Robbins, M.; Borlang, J.; Cao, J.; Hattchete, T.; Stueck, A.; Deschaumbault, Y.; Murnaghan, K.; Varga, J.; Johnston, B. Rat hepatitis e virus linked to severe acute hepatitis in an immunocompetent patient. J. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Hartl, J.; Otto, B.; Madden, R.G.; Webb, G.; Woolson, K.L.; Kriston, L.; Vettorazzi, E.; Lohse, A.W.; Dalton, H.R.; Pischke, S. Hepatitis e seroprevalence in europe: A meta-analysis. Viruses 2016, 8, 211. [Google Scholar] [CrossRef] [PubMed]
- Horvatits, T.; Ozga, A.K.; Westholter, D.; Hartl, J.; Manthey, C.F.; Lutgehetmann, M.; Rauch, G.; Kriston, L.; Lohse, A.W.; Bendall, R.; et al. Hepatitis e seroprevalence in the americas: A systematic review and meta-analysis. Liver Int. 2018, 38, 1951–1964. [Google Scholar] [CrossRef]
- Helsen, N.; Debing, Y.; Paeshuyse, J.; Dallmeier, K.; Boon, R.; Coll, M.; Sancho-Bru, P.; Claes, C.; Neyts, J.; Verfaillie, C.M. Stem cell-derived hepatocytes: A novel model for hepatitis e virus replication. J. Hepatol. 2016, 64, 565–573. [Google Scholar] [CrossRef]
- Allweiss, L.; Gass, S.; Giersch, K.; Groth, A.; Kah, J.; Volz, T.; Rapp, G.; Schobel, A.; Lohse, A.W.; Polywka, S.; et al. Human liver chimeric mice as a new model of chronic hepatitis e virus infection and preclinical drug evaluation. J. Hepatol. 2016, 64, 1033–1040. [Google Scholar] [CrossRef]
- Sayed, I.M.; Verhoye, L.; Cocquerel, L.; Abravanel, F.; Foquet, L.; Montpellier, C.; Debing, Y.; Farhoudi, A.; Wychowski, C.; Dubuisson, J.; et al. Study of hepatitis e virus infection of genotype 1 and 3 in mice with humanised liver. Gut 2017, 66, 920–929. [Google Scholar] [CrossRef]
- Suneetha, P.V.; Pischke, S.; Schlaphoff, V.; Grabowski, J.; Fytili, P.; Gronert, A.; Bremer, B.; Markova, A.; Jaroszewicz, J.; Bara, C.; et al. Hepatitis e virus (hev)-specific t-cell responses are associated with control of hev infection. Hepatology 2012, 55, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Halliday, J.S.; Swadling, L.; Madden, R.G.; Bendall, R.; Hunter, J.G.; Maggs, J.; Simmonds, P.; Smith, D.B.; Vine, L.; et al. Characterization of the specificity, functionality, and durability of host t-cell responses against the full-length hepatitis e virus. Hepatology 2016, 64, 1934–1950. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Abravanel, F.; Selves, J.; Garrouste, C.; Esposito, L.; Lavayssiere, L.; Cointault, O.; Ribes, D.; Cardeau, I.; Nogier, M.B.; et al. Influence of immunosuppressive therapy on the natural history of genotype 3 hepatitis-e virus infection after organ transplantation. Transplantation 2010, 89, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Rostaing, L.; Abravanel, F.; Garrouste, C.; Esposito, L.; Cardeau-Desangles, I.; Mansuy, J.M.; Selves, J.; Peron, J.M.; Otal, P.; et al. Pegylated interferon-alpha for treating chronic hepatitis e virus infection after liver transplantation. Clin. Infect. Dis. 2010, 50, e30–e33. [Google Scholar] [CrossRef] [PubMed]
- Haagsma, E.B.; Riezebos-Brilman, A.; van den Berg, A.P.; Porte, R.J.; Niesters, H.G. Treatment of chronic hepatitis e in liver transplant recipients with pegylated interferon alpha-2b. Liver Transpl. 2010, 16, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Rostaing, L.; Abravanel, F.; Garrouste, C.; Lhomme, S.; Esposito, L.; Basse, G.; Cointault, O.; Ribes, D.; Nogier, M.B.; et al. Ribavirin therapy inhibits viral replication on patients with chronic hepatitis e virus infection. Gastroenterology 2010, 139, 1612–1618. [Google Scholar] [CrossRef] [PubMed]
- Mallet, V.; Nicand, E.; Sultanik, P.; Chakvetadze, C.; Tesse, S.; Thervet, E.; Mouthon, L.; Sogni, P.; Pol, S. Brief communication: Case reports of ribavirin treatment for chronic hepatitis e. Ann. Intern. Med. 2010, 153, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Pischke, S.; Hardtke, S.; Bode, U.; Birkner, S.; Chatzikyrkou, C.; Kauffmann, W.; Bara, C.L.; Gottlieb, J.; Wenzel, J.; Manns, M.P.; et al. Ribavirin treatment of acute and chronic hepatitis e: A single-centre experience. Liver Int. 2013, 33, 722–726. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Izopet, J.; Tripon, S.; Bismuth, M.; Hillaire, S.; Dumortier, J.; Radenne, S.; Coilly, A.; Garrigue, V.; D’Alteroche, L.; et al. Ribavirin for chronic hepatitis e virus infection in transplant recipients. N. Engl. J. Med. 2014, 370, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.C.; Zhang, J.; Zhang, X.F.; Zhou, C.; Wang, Z.Z.; Huang, S.J.; Wang, H.; Yang, C.L.; Jiang, H.M.; Cai, J.P.; et al. Efficacy and safety of a recombinant hepatitis e vaccine in healthy adults: A large-scale, randomised, double-blind placebo-controlled, phase 3 trial. Lancet 2010, 376, 895–902. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. Easl clinical practice guidelines on hepatitis e virus infection. J. Hepatol. 2018, 68, 1256–1271. [Google Scholar] [CrossRef] [PubMed]
- Hartl, J.; Kreuels, B.; Polywka, S.; Addo, M.; Luethgehetmann, M.; Dandri, M.; Dammermann, W.; Sterneck, M.; Lohse, A.W.; Pischke, S. Comparison of autochthonous and imported cases of hepatitis a or hepatitis e. Z. Gastroenterol. 2015, 53, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Sebode, M.; Pischke, S.; Lutgehetmann, M.; Polywka, S.; Quaas, A.; Lohse, A.W.; Wege, H. New foe treated with old guns-supportive role of steroids in the treatment of acute severe hepatitis e. BMC Gastroenterol. 2014, 14, 191. [Google Scholar] [CrossRef] [PubMed]
- Pischke, S.; Gisa, A.; Suneetha, P.V.; Wiegand, S.B.; Taubert, R.; Schlue, J.; Wursthorn, K.; Bantel, H.; Raupach, R.; Bremer, B.; et al. Increased hev seroprevalence in patients with autoimmune hepatitis. PLoS ONE 2014, 9, e85330. [Google Scholar] [CrossRef] [PubMed]
- Davern, T.J.; Chalasani, N.; Fontana, R.J.; Hayashi, P.H.; Protiva, P.; Kleiner, D.E.; Engle, R.E.; Nguyen, H.; Emerson, S.U.; Purcell, R.H.; et al. Acute hepatitis e infection accounts for some cases of suspected drug-induced liver injury. Gastroenterology 2011, 141, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Fontana, R.J.; Engle, R.E.; Scaglione, S.; Araya, V.; Shaikh, O.; Tillman, H.; Attar, N.; Purcell, R.H.; Lee, W.M.; US Acute Liver Failure Study Group. The role of hepatitis e virus infection in adult americans with acute liver failure. Hepatology 2016, 64, 1870–1880. [Google Scholar] [CrossRef] [PubMed]
- Nagasaki, F.; Ueno, Y.; Kanno, N.; Okamoto, H.; Shimosegawa, T. A case of acute hepatitis with positive autoantibodies who actually had hepatitis e virus infection. Hepatol. Res. 2005, 32, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Peron, J.M.; Bureau, C.; Poirson, H.; Mansuy, J.M.; Alric, L.; Selves, J.; Dupuis, E.; Izopet, J.; Vinel, J.P. Fulminant liver failure from acute autochthonous hepatitis e in france: Description of seven patients with acute hepatitis e and encephalopathy. J. Viral Hepat. 2007, 14, 298–303. [Google Scholar] [CrossRef]
- Kumar, A.; Saraswat, V.A. Hepatitis e and acute-on-chronic liver failure. J. Clin. Exp. Hepatol. 2013, 3, 225–230. [Google Scholar] [CrossRef]
- Hernaez, R.; Sola, E.; Moreau, R.; Gines, P. Acute-on-chronic liver failure: An update. Gut 2017, 66, 541–553. [Google Scholar] [CrossRef]
- Moreau, R.; Jalan, R.; Gines, P.; Pavesi, M.; Angeli, P.; Cordoba, J.; Durand, F.; Gustot, T.; Saliba, F.; Domenicali, M.; et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology 2013, 144, 1426–1437. [Google Scholar] [CrossRef] [PubMed]
- Blasco-Perrin, H.; Madden, R.G.; Stanley, A.; Crossan, C.; Hunter, J.G.; Vine, L.; Lane, K.; Devooght-Johnson, N.; McLaughlin, C.; Petrik, J.; et al. Hepatitis e virus in patients with decompensated chronic liver disease: A prospective uk/french study. Aliment. Pharmacol. Ther. 2015, 42, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Shalimar; Kedia, S.; Gunjan, D.; Sonika, U.; Mahapatra, S.J.; Nayak, B.; Kaur, H.; Acharya, S.K. Acute liver failure due to hepatitis e virus infection is associated with better survival than other etiologies in indian patients. Dig. Dis. Sci. 2017, 62, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Shalimar; Kedia, S.; Mahapatra, S.J.; Nayak, B.; Gunjan, D.; Thakur, B.; Acharya, S.K. Severity and outcome of acute-on-chronic liver failure is dependent on the etiology of acute hepatic insults: Analysis of 368 patients. J. Clin. Gastroenterol. 2017, 51, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Shimakawa, Y.; Njai, H.F.; Takahashi, K.; Berg, L.; Ndow, G.; Jeng-Barry, A.; Ceesay, A.; Tamba, S.; Opoku, E.; Taal, M.; et al. Hepatitis e virus infection and acute-on-chronic liver failure in west africa: A case-control study from the gambia. Aliment. Pharmacol. Ther. 2016, 43, 375–384. [Google Scholar] [CrossRef]
- Vento, S.; Garofano, T.; Renzini, C.; Cainelli, F.; Casali, F.; Ghironzi, G.; Ferraro, T.; Concia, E. Fulminant hepatitis associated with hepatitis a virus superinfection in patients with chronic hepatitis c. N. Engl. J. Med. 1998, 338, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Pischke, S.; Wedemeyer, H. Hepatitis e virus infection: Multiple faces of an underestimated problem. J. Hepatol. 2013, 58, 1045–1046. [Google Scholar] [CrossRef]
- Caron, M.; Enouf, V.; Than, S.C.; Dellamonica, L.; Buisson, Y.; Nicand, E. Identification of genotype 1 hepatitis e virus in samples from swine in cambodia. J. Clin. Microbiol. 2006, 44, 3440–3442. [Google Scholar] [CrossRef]
- Azman, A.S.; Ciglenecki, I.; Oeser, C.; Said, B.; Tedder, R.S.; Ijaz, S. The incubation period of hepatitis e genotype 1: Insights from pooled analyses of travellers. Epidemiol. Infect. 2018, 146, 1533–1536. [Google Scholar] [CrossRef]
- Kamar, N.; Dalton, H.R.; Abravanel, F.; Izopet, J. Hepatitis e virus infection. Clin. Microbiol. Rev. 2014, 27, 116–138. [Google Scholar] [CrossRef]
- Aggarwal, R.; Kini, D.; Sofat, S.; Naik, S.R.; Krawczynski, K. Duration of viraemia and faecal viral excretion in acute hepatitis e. Lancet 2000, 356, 1081–1082. [Google Scholar] [CrossRef]
- Naik, A.; Gupta, N.; Goel, D.; Ippagunta, S.K.; Sharma, R.K.; Aggarwal, R. Lack of evidence of hepatitis e virus infection among renal transplant recipients in a disease-endemic area. J. Viral Hepat. 2013, 20, e138–e140. [Google Scholar] [CrossRef] [PubMed]
- Feldt, T.; Sarfo, F.S.; Zoufaly, A.; Phillips, R.O.; Burchard, G.; van Lunzen, J.; Jochum, J.; Chadwick, D.; Awasom, C.; Claussen, L.; et al. Hepatitis e virus infections in hiv-infected patients in ghana and cameroon. J. Clin. Virol. 2013, 58, 18–23. [Google Scholar] [CrossRef]
- Agarwala, P.; Gupta, E.; Choudhary, M.C.; Pamecha, V. Absence of chronic hepatitis e virus infection in liver transplant recipients: Report from a hyperendemic region. Indian J. Gastroenterol. 2018, 37, 160–163. [Google Scholar] [CrossRef]
- Robins, A.E.M.; Bowden, D.J.; Gelson, W.T.H. Chronic genotype 1 hepatitis e infection from immunosuppression for ileo-colonic crohn’s disease. Oxf. Med. Case Rep. 2018, 2018, omy059. [Google Scholar] [CrossRef] [PubMed]
- Ankcorn, M.; Haywood, B.; Tedder, R.; Ijaz, S. Response to: ’Chronic genotype 1 hepatitis e infection from immunosuppression for ileo-colonic crohn’s disease’. Oxf. Med. Case Rep. 2019, 2019, omy125. [Google Scholar] [CrossRef]
- Navaneethan, U.; Al Mohajer, M.; Shata, M.T. Hepatitis e and pregnancy: Understanding the pathogenesis. Liver Int. 2008, 28, 1190–1199. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Bu, Q.; Gong, W.; Li, H.; Wang, L.; Li, S.; Sridhar, S.; Cy Woo, P.; Wang, L. Hepatitis e virus infection and its associated adverse feto-maternal outcomes among pregnant women in qinhuangdao, china. J. Matern. Fetal Neonatal Med. 2019. [Google Scholar] [CrossRef]
- Jilani, N.; Das, B.C.; Husain, S.A.; Baweja, U.K.; Chattopadhya, D.; Gupta, R.K.; Sardana, S.; Kar, P. Hepatitis e virus infection and fulminant hepatic failure during pregnancy. J. Gastroenterol. Hepatol. 2007, 22, 676–682. [Google Scholar] [CrossRef] [PubMed]
- McGovern, B.H.; Ditelberg, J.S.; Taylor, L.E.; Gandhi, R.T.; Christopoulos, K.A.; Chapman, S.; Schwartzapfel, B.; Rindler, E.; Fiorino, A.M.; Zaman, M.T.; et al. Hepatic steatosis is associated with fibrosis, nucleoside analogue use, and hepatitis c virus genotype 3 infection in hiv-seropositive patients. Clin. Infect. Dis. 2006, 43, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Horvatits, T.; Pischke, S. Hev in pregnancy: Understanding the crucial role of steroid hormones. Liver Int. 2019, 39, 621–622. [Google Scholar] [CrossRef] [PubMed]
- Bose, P.D.; Das, B.C.; Kumar, A.; Gondal, R.; Kumar, D.; Kar, P. High viral load and deregulation of the progesterone receptor signaling pathway: Association with hepatitis e-related poor pregnancy outcome. J. Hepatol. 2011, 54, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Daga, M.K.; Kumar, A.; Husain, S.A.; Kar, P. Role of oestrogen and its receptors in hev-associated feto-maternal outcomes. Liver Int. 2019, 39, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Bose, P.D.; Das, B.C.; Hazam, R.K.; Kumar, A.; Medhi, S.; Kar, P. Evidence of extrahepatic replication of hepatitis e virus in human placenta. J. Gen. Virol. 2014, 95, 1266–1271. [Google Scholar] [CrossRef] [PubMed]
- Kar, P.; Sengupta, A. A guide to the management of hepatitis e infection during pregnancy. Expert Rev. Gastroenterol. Hepatol. 2019, 13, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Tabatabai, J.; Wenzel, J.J.; Soboletzki, M.; Flux, C.; Navid, M.H.; Schnitzler, P. First case report of an acute hepatitis e subgenotype 3c infection during pregnancy in germany. J. Clin. Virol. 2014, 61, 170–172. [Google Scholar] [CrossRef] [PubMed]
- Anty, R.; Ollier, L.; Peron, J.M.; Nicand, E.; Cannavo, I.; Bongain, A.; Giordanengo, V.; Tran, A. First case report of an acute genotype 3 hepatitis e infected pregnant woman living in south-eastern france. J. Clin. Virol. 2012, 54, 76–78. [Google Scholar] [CrossRef]
- Riveiro-Barciela, M.; Buti, M.; Homs, M.; Campos-Varela, I.; Cantarell, C.; Crespo, M.; Castells, L.; Tabernero, D.; Quer, J.; Esteban, R.; et al. Cirrhosis, liver transplantation and hiv infection are risk factors associated with hepatitis e virus infection. PLoS ONE 2014, 9, e103028. [Google Scholar] [CrossRef]
- Kamar, N.; Selves, J.; Mansuy, J.M.; Ouezzani, L.; Peron, J.M.; Guitard, J.; Cointault, O.; Esposito, L.; Abravanel, F.; Danjoux, M.; et al. Hepatitis e virus and chronic hepatitis in organ-transplant recipients. N. Engl. J. Med. 2008, 358, 811–817. [Google Scholar] [CrossRef]
- Gerolami, R.; Moal, V.; Colson, P. Chronic hepatitis e with cirrhosis in a kidney-transplant recipient. N. Engl. J. Med. 2008, 358, 859–860. [Google Scholar] [CrossRef]
- Pischke, S.; Stiefel, P.; Franz, B.; Bremer, B.; Suneetha, P.V.; Heim, A.; Ganzenmueller, T.; Schlue, J.; Horn-Wichmann, R.; Raupach, R.; et al. Chronic hepatitis e in heart transplant recipients. Am. J. Transpl. 2012, 12, 3128–3133. [Google Scholar] [CrossRef] [PubMed]
- Pischke, S.; Greer, M.; Hardtke, S.; Bremer, B.; Gisa, A.; Lehmann, P.; Haverich, A.; Welte, T.; Manns, M.P.; Wedemeyer, H.; et al. Course and treatment of chronic hepatitis e virus infection in lung transplant recipients. Transpl. Infect. Dis. 2014, 16, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Ingiliz, P.; Mayr, C.; Obermeier, M.; Herbst, H.; Polywka, S.; Pischke, S. Persisting hepatitis e virus infection leading to liver cirrhosis despite recovery of the immune system in an hiv-infected patient. Clin. Res. Hepatol. Gastroenterol. 2016, 40, e23–e25. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Pischke, S. Acute and persistent hepatitis e virus genotype 3 and 4 infection: Clinical features, pathogenesis, and treatment. Cold Spring Harb. Perspect. Med. 2018, 9, a031872. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, G.; Pan, Q.; Zhao, J. Chronic hepatitis e in a renal transplant recipient: The first report of genotype 4 hepatitis e virus caused chronic infection in organ recipient. Gastroenterology 2018, 154, 1199–1201. [Google Scholar] [CrossRef] [PubMed]
- Legrand-Abravanel, F.; Kamar, N.; Sandres-Saune, K.; Lhomme, S.; Mansuy, J.M.; Muscari, F.; Sallusto, F.; Rostaing, L.; Izopet, J. Hepatitis e virus infection without reactivation in solid-organ transplant recipients, france. Emerg. Infect. Dis. 2011, 17, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Rostaing, L.; Legrand-Abravanel, F.; Izopet, J. How should hepatitis e virus infection be defined in organ-transplant recipients? Am. J. Transpl. 2013, 13, 1935–1936. [Google Scholar] [CrossRef] [PubMed]
- Meisner, S.; Polywka, S.; Memmler, M.; Nashan, B.; Lohse, A.W.; Sterneck, M.; Pischke, S. Definition of chronic hepatitis e after liver transplant conforms to convention. Am. J. Transpl. 2015, 15, 3011–3012. [Google Scholar] [CrossRef]
- Dalton, H.R.; Bendall, R.P.; Keane, F.E.; Tedder, R.S.; Ijaz, S. Persistent carriage of hepatitis e virus in patients with hiv infection. N. Engl. J. Med. 2009, 361, 1025–1027. [Google Scholar] [CrossRef]
- Kaba, M.; Richet, H.; Ravaux, I.; Moreau, J.; Poizot-Martin, I.; Motte, A.; Nicolino-Brunet, C.; Dignat-George, F.; Menard, A.; Dhiver, C.; et al. Hepatitis e virus infection in patients infected with the human immunodeficiency virus. J. Med. Virol. 2011, 83, 1704–1716. [Google Scholar] [CrossRef]
- Kuniholm, M.H.; Ong, E.; Hogema, B.M.; Koppelman, M.; Anastos, K.; Peters, M.G.; Seaberg, E.C.; Chen, Y.; Nelson, K.E.; Linnen, J.M. Acute and chronic hepatitis e virus infection in human immunodeficiency virus-infected U.S. Women. Hepatology 2016, 63, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Pischke, S.; Peron, J.M.; von Wulffen, M.; von Felden, J.; Honer Zu Siederdissen, C.; Fournier, S.; Lutgehetmann, M.; Iking-Konert, C.; Bettinger, D.; Par, G.; et al. Chronic hepatitis e in rheumatology and internal medicine patients: A retrospective multicenter european cohort study. Viruses 2019, 11, 186. [Google Scholar] [CrossRef] [PubMed]
- Mallet, V.; Bruneau, J.; Zuber, J.; Alanio, C.; Leclerc-Mercier, S.; Roque-Afonso, A.M.; Kraft, A.R.M.; Couronne, L.; Roulot, D.; Wedemeyer, H.; et al. Hepatitis e virus-induced primary cutaneous cd30 (+) T cell lymphoproliferative disorder. J. Hepatol. 2017, 67, 1334–1339. [Google Scholar] [CrossRef] [PubMed]
- Mallet, V.; van Bommel, F.; Doerig, C.; Pischke, S.; Hermine, O.; Locasciulli, A.; Cordonnier, C.; Berg, T.; Moradpour, D.; Wedemeyer, H.; et al. Management of viral hepatitis in patients with haematological malignancy and in patients undergoing haemopoietic stem cell transplantation: Recommendations of the 5th european conference on infections in leukaemia (ecil-5). Lancet Infect. Dis. 2016, 16, 606–617. [Google Scholar] [CrossRef]
- Woolson, K.L.; Forbes, A.; Vine, L.; Beynon, L.; McElhinney, L.; Panayi, V.; Hunter, J.G.; Madden, R.G.; Glasgow, T.; Kotecha, A.; et al. Extra-hepatic manifestations of autochthonous hepatitis e infection. Aliment. Pharmacol. Ther. 2014, 40, 1282–1291. [Google Scholar] [CrossRef] [PubMed]
- Versluis, J.; Pas, S.D.; Agteresch, H.J.; de Man, R.A.; Maaskant, J.; Schipper, M.E.; Osterhaus, A.D.; Cornelissen, J.J.; van der Eijk, A.A. Hepatitis e virus: An underestimated opportunistic pathogen in recipients of allogeneic hematopoietic stem cell transplantation. Blood 2013, 122, 1079–1086. [Google Scholar] [CrossRef] [PubMed]
- Von Felden, J.; Laurent, A.; Pischke, S.; Aitken, C.; Schlabe, S.; Spengler, U.; Giordani, M.T.; Schnitzler, P.; Bettinger, D.; Thimme, R.; et al. The burden of hepatitis e among patients with haematological malignancy: A retrospective european cohort study. J. Hepatol. 2019. [Google Scholar] [CrossRef]
- Dalton, H.R.; van Eijk, J.J.J.; Cintas, P.; Madden, R.G.; Jones, C.; Webb, G.W.; Norton, B.; Pique, J.; Lutgens, S.; Devooght-Johnson, N.; et al. Hepatitis e virus infection and acute non-traumatic neurological injury: A prospective multicentre study. J. Hepatol. 2017, 67, 925–932. [Google Scholar] [CrossRef]
- Van Eijk, J.J.J.; Dalton, H.R.; Ripellino, P.; Madden, R.G.; Jones, C.; Fritz, M.; Gobbi, C.; Melli, G.; Pasi, E.; Herrod, J.; et al. Clinical phenotype and outcome of hepatitis e virus-associated neuralgic amyotrophy. Neurology 2017, 89, 909–917. [Google Scholar] [CrossRef]
- Wang, L.; Gao, F.; Lin, G.; Yuan, Y.; Huang, Y.; Hao, H.; Zhuang, H.; Wang, L. Association of hepatitis e virus infection and myasthenia gravis: A pilot study. J. Hepatol. 2018, 68, 1318–1320. [Google Scholar] [CrossRef]
- Van den Berg, B.; van der Eijk, A.A.; Pas, S.D.; Hunter, J.G.; Madden, R.G.; Tio-Gillen, A.P.; Dalton, H.R.; Jacobs, B.C. Guillain-barre syndrome associated with preceding hepatitis e virus infection. Neurology 2014, 82, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, S.; Wu, J.; Jiang, Y.; Zhang, H.; Li, S.; Liu, H.; Yang, C.; Tang, H.; Guo, N.; et al. Hepatitis e virus infection in acute non-traumatic neuropathy: A large prospective case-control study in China. EBioMedicine 2018, 36, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Horvatits, T.; Pischke, S. Extrahepatic manifestations and hev, the genotype matters. EBioMedicine 2018, 36, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Soomro, M.H.; Shi, R.; She, R.; Yang, Y.; Hu, F.; Li, H. Antigen detection and apoptosis in mongolian gerbil’s kidney experimentally intraperitoneally infected by swine hepatitis e virus. Virus Res. 2016, 213, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Zhao, C.; Huang, W.; Harrison, T.J.; Zhang, H.; Geng, K.; Wang, Y. Detection and assessment of infectivity of hepatitis e virus in urine. J. Hepatol. 2016, 64, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Del Bello, A.; Guilbeau-Frugier, C.; Josse, A.G.; Rostaing, L.; Izopet, J.; Kamar, N. Successful treatment of hepatitis e virus-associated cryoglobulinemic membranoproliferative glomerulonephritis with ribavirin. Transpl. Infect. Dis. 2015, 17, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Long, F.; Yu, W.; Situ, J.; Fu, L.; He, Z.; Dong, H.; Yang, C.; Li, Y.; Yang, F.; et al. High prevalence of hepatitis e virus in semen of infertile male and causes testis damage. Gut 2018, 67, 1199–1201. [Google Scholar] [CrossRef] [PubMed]
- Horvatits, T.; Varwig-Janssen, D.; Schulze Zur Wiesch, J.; Lübke, R.; Reucher, S.; Frerk, S.; Addo, M.; Schneider, S.W.; Lohse, A.W.; Luetgehethmann, M.; et al. No link between male infertility and hev genotype 3 infection. Gut 2019. [Google Scholar] [CrossRef]
- Goyal, R.; Kumar, A.; Panda, S.K.; Paul, S.B.; Acharya, S.K. Ribavirin therapy for hepatitis e virus-induced acute on chronic liver failure: A preliminary report. Antivir. Ther. 2012, 17, 1091–1096. [Google Scholar] [CrossRef]
- Sinclair, S.M.; Jones, J.K.; Miller, R.K.; Greene, M.F.; Kwo, P.Y.; Maddrey, W.C. The ribavirin pregnancy registry: An interim analysis of potential teratogenicity at the mid-point of enrollment. Drug Saf. 2017, 40, 1205–1218. [Google Scholar] [CrossRef]
- Gerolami, R.; Borentain, P.; Raissouni, F.; Motte, A.; Solas, C.; Colson, P. Treatment of severe acute hepatitis e by ribavirin. J. Clin. Virol. 2011, 52, 60–62. [Google Scholar] [CrossRef] [PubMed]
- Peron, J.M.; Dalton, H.; Izopet, J.; Kamar, N. Acute autochthonous hepatitis e in western patients with underlying chronic liver disease: A role for ribavirin? J. Hepatol. 2011, 54, 1323–1324. [Google Scholar] [CrossRef] [PubMed]
- Peron, J.M.; Abravanel, F.; Guillaume, M.; Gerolami, R.; Nana, J.; Anty, R.; Pariente, A.; Renou, C.; Bureau, C.; Robic, M.A.; et al. Treatment of autochthonous acute hepatitis e with short-term ribavirin: A multicenter retrospective study. Liver Int. 2016, 36, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Garrouste, C.; Haagsma, E.B.; Garrigue, V.; Pischke, S.; Chauvet, C.; Dumortier, J.; Cannesson, A.; Cassuto-Viguier, E.; Thervet, E.; et al. Factors associated with chronic hepatitis in patients with hepatitis e virus infection who have received solid organ transplants. Gastroenterology 2011, 140, 1481–1489. [Google Scholar] [CrossRef] [PubMed]
- Van Tong, H.; Hoan, N.X.; Wang, B.; Wedemeyer, H.; Bock, C.T.; Velavan, T.P. Hepatitis e virus mutations: Functional and clinical relevance. EBioMedicine 2016, 11, 31–42. [Google Scholar] [CrossRef]
- Debing, Y.; Gisa, A.; Dallmeier, K.; Pischke, S.; Bremer, B.; Manns, M.; Wedemeyer, H.; Suneetha, P.V.; Neyts, J. A mutation in the hepatitis e virus rna polymerase promotes its replication and associates with ribavirin treatment failure in organ transplant recipients. Gastroenterology 2014, 147, 1008–1011. [Google Scholar] [CrossRef] [PubMed]
- Todt, D.; Gisa, A.; Radonic, A.; Nitsche, A.; Behrendt, P.; Suneetha, P.V.; Pischke, S.; Bremer, B.; Brown, R.J.; Manns, M.P.; et al. In vivo evidence for ribavirin-induced mutagenesis of the hepatitis e virus genome. Gut 2016, 65, 1733–1743. [Google Scholar] [CrossRef]
- Todt, D.; Walter, S.; Brown, R.J.; Steinmann, E. Mutagenic effects of ribavirin on hepatitis e virus-viral extinction versus selection of fitness-enhancing mutations. Viruses 2016, 8, 283. [Google Scholar] [CrossRef]
- Dao Thi, V.L.; Debing, Y.; Wu, X.; Rice, C.M.; Neyts, J.; Moradpour, D.; Gouttenoire, J. Sofosbuvir inhibits hepatitis e virus replication in vitro and results in an additive effect when combined with ribavirin. Gastroenterology 2016, 150, 82–85 e84. [Google Scholar] [CrossRef]
- Van der Valk, M.; Zaaijer, H.L.; Kater, A.P.; Schinkel, J. Sofosbuvir shows antiviral activity in a patient with chronic hepatitis e virus infection. J. Hepatol. 2017, 66, 242–243. [Google Scholar] [CrossRef]
- Todesco, E.; Mazzola, A.; Akhavan, S.; Abravanel, F.; Poynard, T.; Roque-Afonso, A.M.; Peytavin, G.; Marcelin, A.G.; Calmus, Y.; Lecuyer, L.; et al. Chronic hepatitis e in a heart transplant patient: Sofosbuvir and ribavirin regimen not fully effective. Antivir. Ther. 2018, 23, 463–465. [Google Scholar] [CrossRef] [PubMed]
- Cornberg, M.; Pischke, S.; Mueller, T.; Behrendt, P.; Piecha, F.; Benckert, J.; Smith, A.; Koch, A.; Lohse, A.W.; Hardtke, S.; et al. Efficacy and safety of sofosbuvir monotherapy in patients with chronic hepatitis e-the hepnet sofe pilot study. J. Hepatol. 2019, 70, e625–e853. [Google Scholar] [CrossRef]
- Biedenkopf, N.; Lange-Grunweller, K.; Schulte, F.W.; Weisser, A.; Muller, C.; Becker, D.; Becker, S.; Hartmann, R.K.; Grunweller, A. The natural compound silvestrol is a potent inhibitor of ebola virus replication. Antivir. Res. 2017, 137, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Liu, L.; Li, M.; Wang, L.; Zhang, M.; Luo, Z.; Sridhar, S.; Woo, P.C.Y.; Wang, L. Evaluation of antiviral efficacy of chinese traditional medicine babao dan in rabbits infected with hepatitis e virus. J. Gen. Virol. 2018, 99, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Horvatits, T.; Schübel, N.; Kamar, N.; Polywka, S.; Luetgehethmann, M.; Rutter, K.; Schulze Zur Wiesch, J.; Manns, M.; Lohse, A.W.; Roque Afonso, A.M.; et al. Zinc/ribavirin: A possible treatment option in chronically hev genotype 3 infected patients without svr under ribavirin monotherapy. J. Hepatol. 2019, 70, e625–e853. [Google Scholar] [CrossRef]
- Shrestha, M.P.; Scott, R.M.; Joshi, D.M.; Mammen, M.P., Jr.; Thapa, G.B.; Thapa, N.; Myint, K.S.; Fourneau, M.; Kuschner, R.A.; Shrestha, S.K.; et al. Safety and efficacy of a recombinant hepatitis e vaccine. N. Engl. J. Med. 2007, 356, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zhu, F.C.; Huang, S.J.; Zhang, X.F.; Wang, Z.Z.; Zhang, J.; Xia, N.S. Safety of the hepatitis e vaccine for pregnant women: A preliminary analysis. Hepatology 2012, 55, 2038. [Google Scholar] [CrossRef]
- Khuroo, M.S.; Kamili, S.; Jameel, S. Vertical transmission of hepatitis e virus. Lancet 1995, 345, 1025–1026. [Google Scholar] [CrossRef]
- Kumar, A.; Beniwal, M.; Kar, P.; Sharma, J.B.; Murthy, N.S. Hepatitis e in pregnancy. Int. J. Gynaecol. Obs. 2004, 85, 240–244. [Google Scholar] [CrossRef]

| HEV Genotype | Area | Sources of Infection * | Comment |
|---|---|---|---|
| GT 1 | Tropical developing countries of Asia and Africa | Contaminated drinking water | No zoonotic relevance, no chronic infections described, fulminant courses in pregnant women with a mortality of up to 20% in the last trimester, association with pancreatitis but not with neurological symptoms assumed |
| GT 2 | Tropical countries of Africa or Mexico/Central America | Contaminated drinking water | No zoonotic relevance, no chronic infections described |
| GT 3 | Industrialized nations, worldwide distributed, autochthonous in Europe, North and South America, Australia, large parts of Asia | Foodborne zoonosis (mainly contact or consumption of inadequately cooked pork) shellfish deer Strawberries Vegetables (spinach, rocket) Blood transfusions | Chronic HEV infections described in several cohorts of immunosuppressed patients, frequently observed neurological symptoms (in particular Neuralgic amyotrophy) in association with HEV infections, no association with male infertility |
| GT 4 | Mainly in Asia, recently single cases in Europe | Foodborne zoonosis (mainly contact or consumption of inadequately cooked pork) shellfishdeer | Chronic HEV infections described in single immunosuppressed patients, no clear evidence for an association with neurological symptoms, association with preterm birth and abortion, eventually associated with male infertility |
| GT 5 | Japan | Wild boar | Relevance for humans still unclear |
| GT 6 | Japan | Wild boar | Relevance for humans still unclear |
| GT 7 | Middle East | Dromedary camels (one-humped camels) | Chronic infection in a liver transplant recipient who regularly consumed camel meat and milk |
| GT 8 | Middle East | Bactrian camels (two-humped camels) | Relevance for humans still unclear |
| Rat hepatitis E | Hong Kong | Rats | Genetically distinct from the classical HEV genotypes and thus not classified regularly. Two known cases of human infections, one of them chronic in a transplant recipient |
| Avian hepatitis E | Worldwide | Birds | Genetically distinct from the classical HEV genotypes. No known human cases |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horvatits, T.; Schulze zur Wiesch, J.; Lütgehetmann, M.; Lohse, A.W.; Pischke, S. The Clinical Perspective on Hepatitis E. Viruses 2019, 11, 617. https://doi.org/10.3390/v11070617
Horvatits T, Schulze zur Wiesch J, Lütgehetmann M, Lohse AW, Pischke S. The Clinical Perspective on Hepatitis E. Viruses. 2019; 11(7):617. https://doi.org/10.3390/v11070617
Chicago/Turabian StyleHorvatits, Thomas, Julian Schulze zur Wiesch, Marc Lütgehetmann, Ansgar W. Lohse, and Sven Pischke. 2019. "The Clinical Perspective on Hepatitis E" Viruses 11, no. 7: 617. https://doi.org/10.3390/v11070617
APA StyleHorvatits, T., Schulze zur Wiesch, J., Lütgehetmann, M., Lohse, A. W., & Pischke, S. (2019). The Clinical Perspective on Hepatitis E. Viruses, 11(7), 617. https://doi.org/10.3390/v11070617






