Porcine Epidemic Diarrhea Virus (PEDV) ORF3 Interactome Reveals Inhibition of Virus Replication by Cellular VPS36 Protein
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Line and Plasmid Construction
2.2. Co-Immunoprecipitation
2.3. In-gel Digestion and Mass Spectrometry
2.4. Western Blot Assay
2.5. Virus Infection
2.6. Confocal Microscopy
2.7. Virus Titration
2.8. Cell viability (CCK-8) Analysis
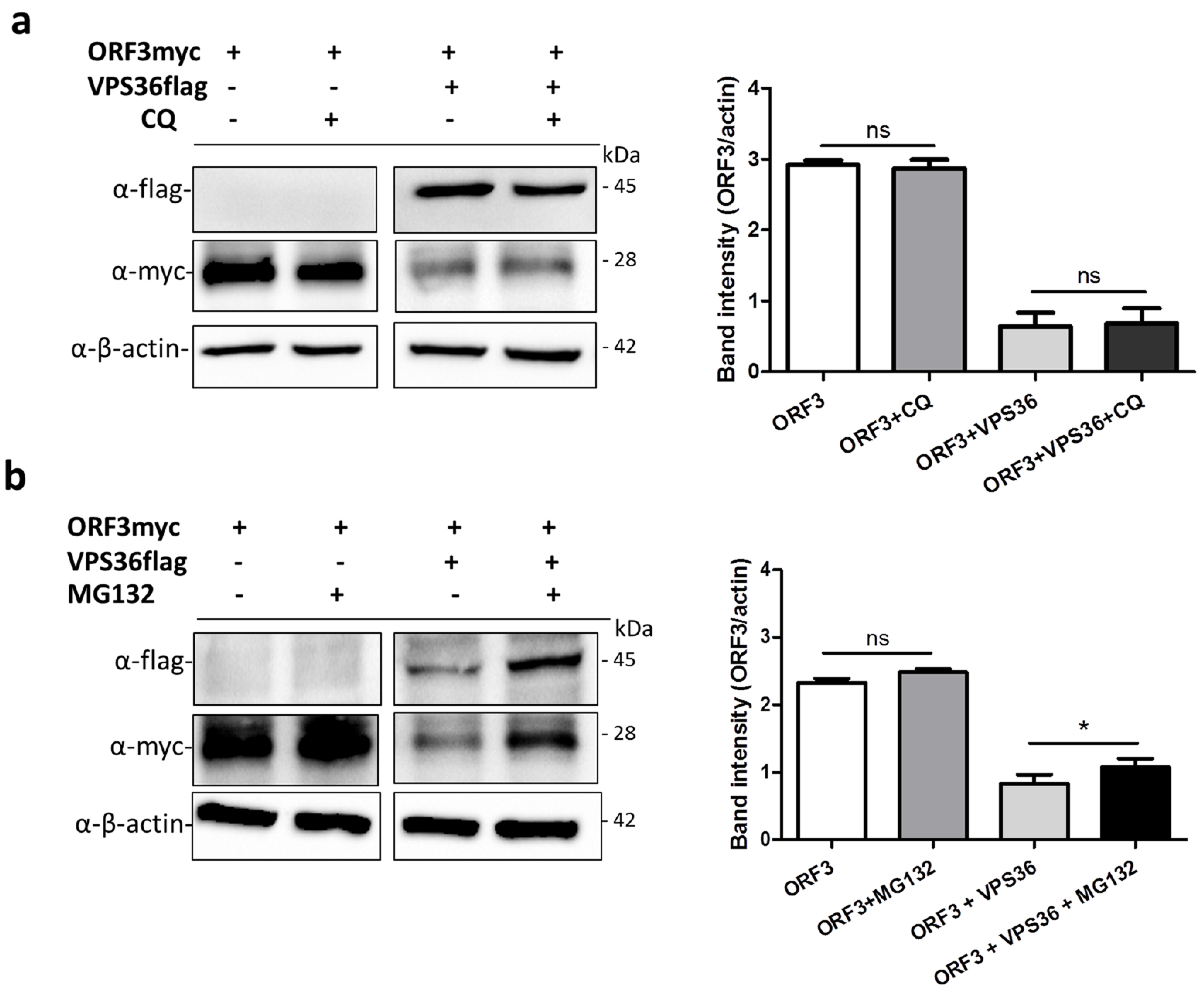
2.9. Lysosomal Inhibitor Treatment
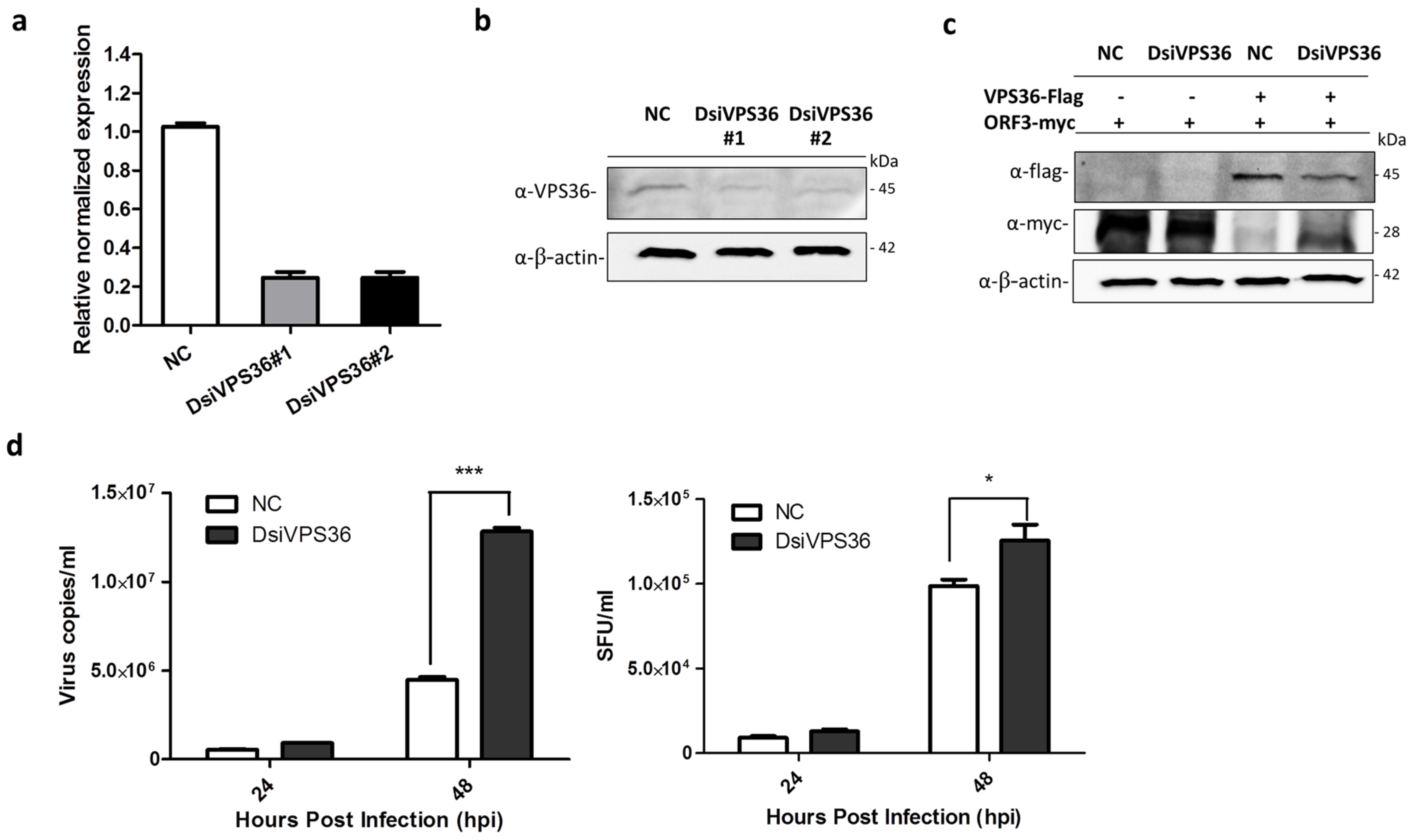
2.10. VPS36 Knock-Down by Small Interfering RNA Assay
2.11. Quantitative RT-PCR (RT-qPCR)
2.12. Statistical Analysis
3. Results
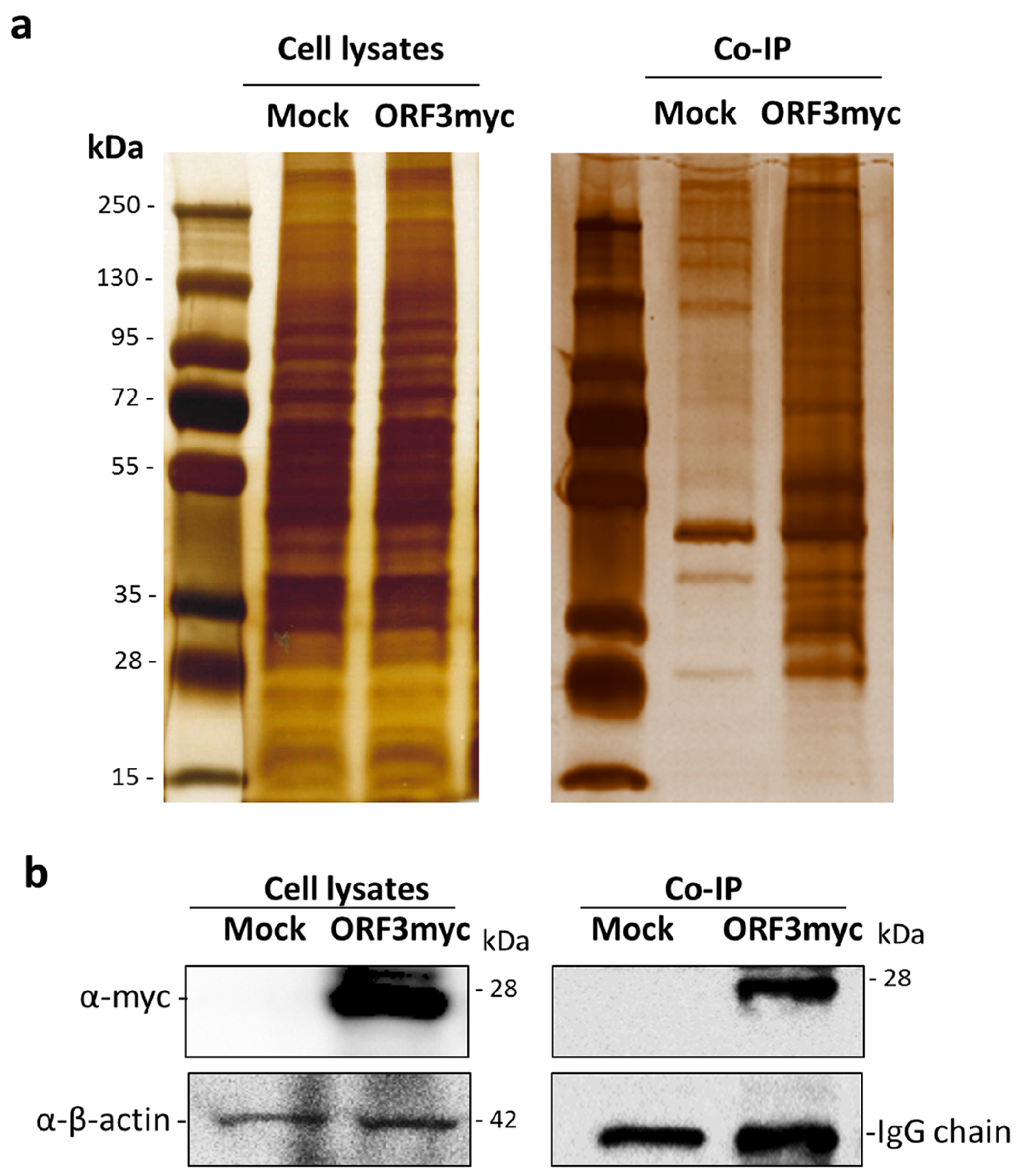
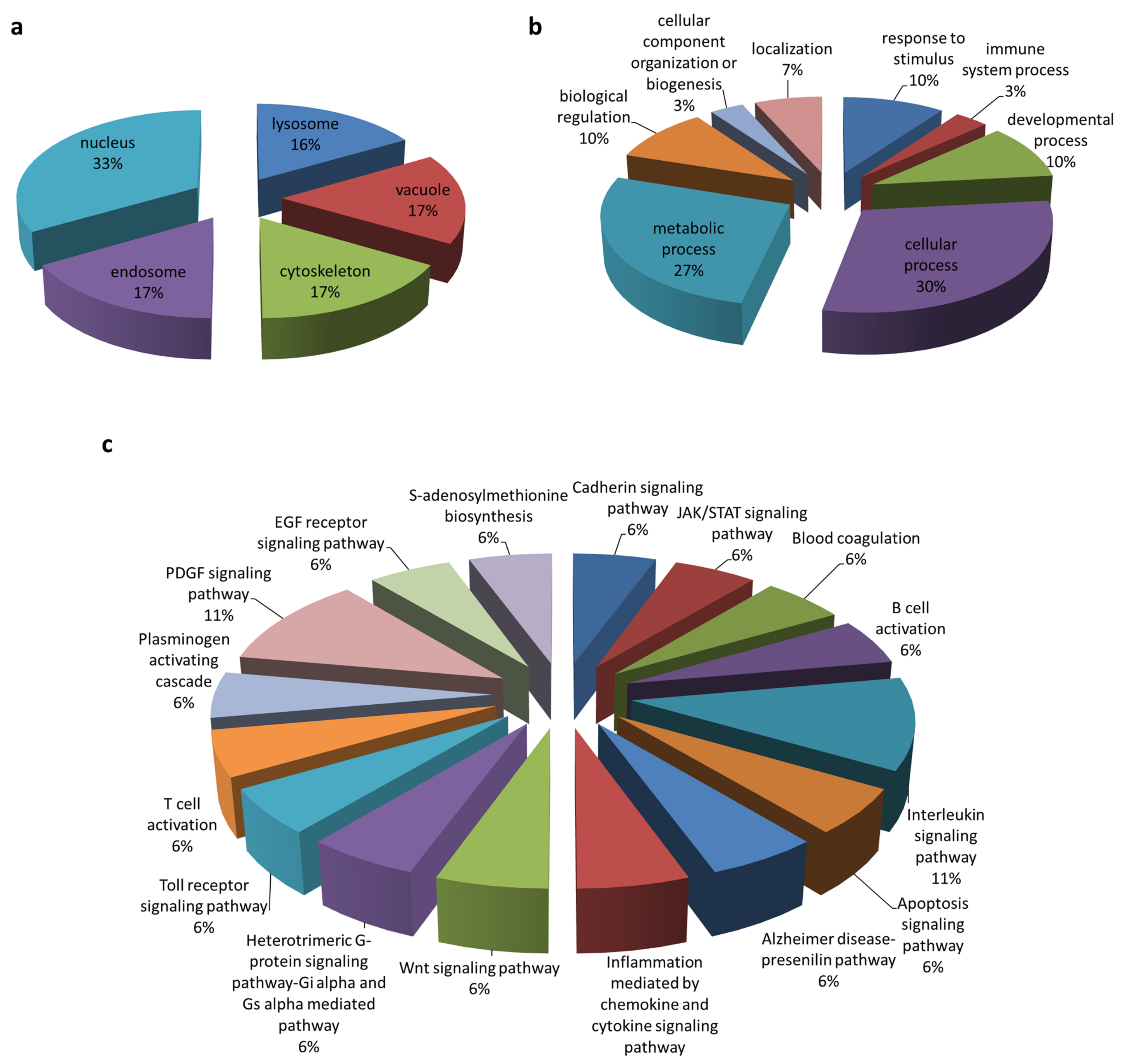
3.1. Identification of Cellular Protein Partners of PEDV ORF3
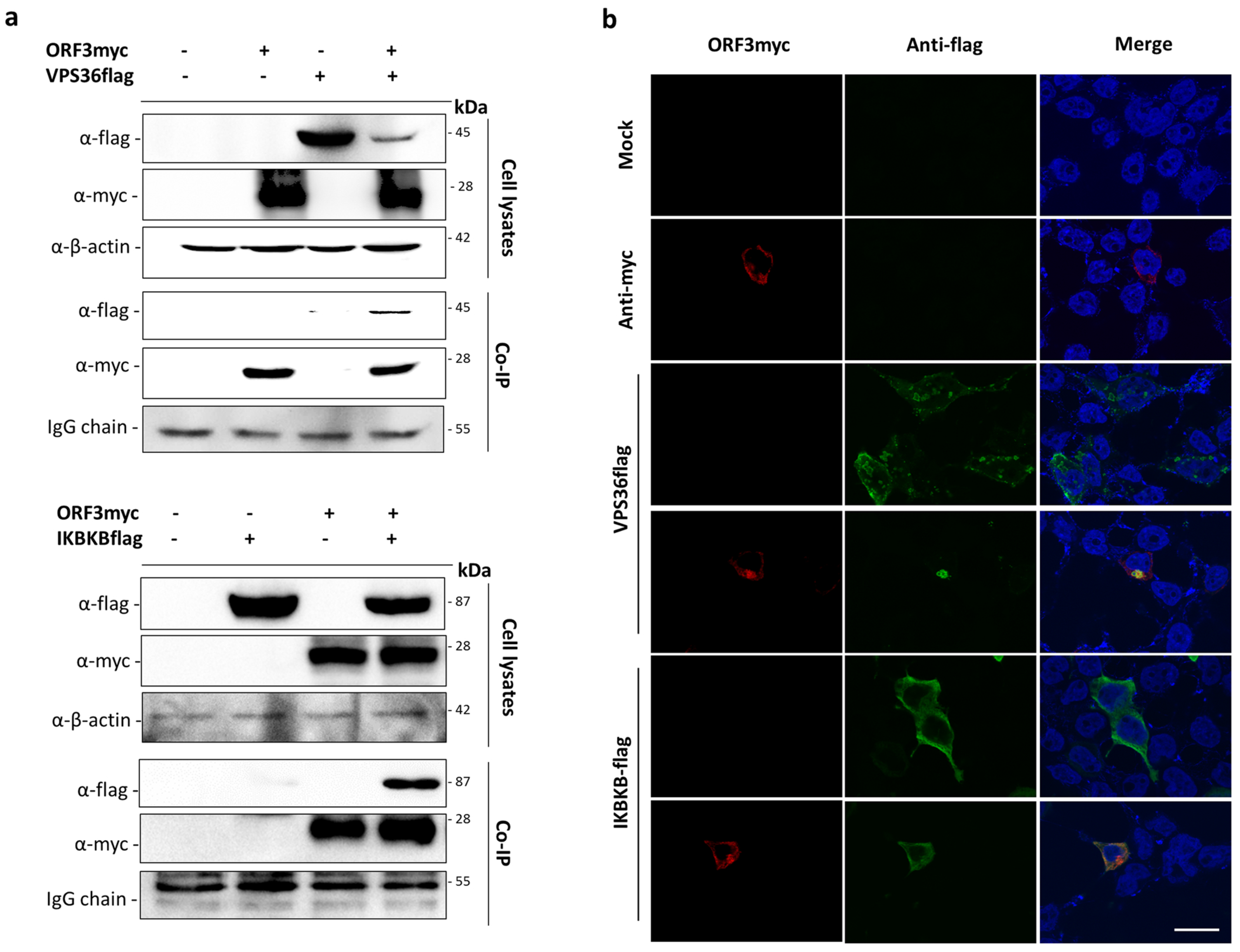
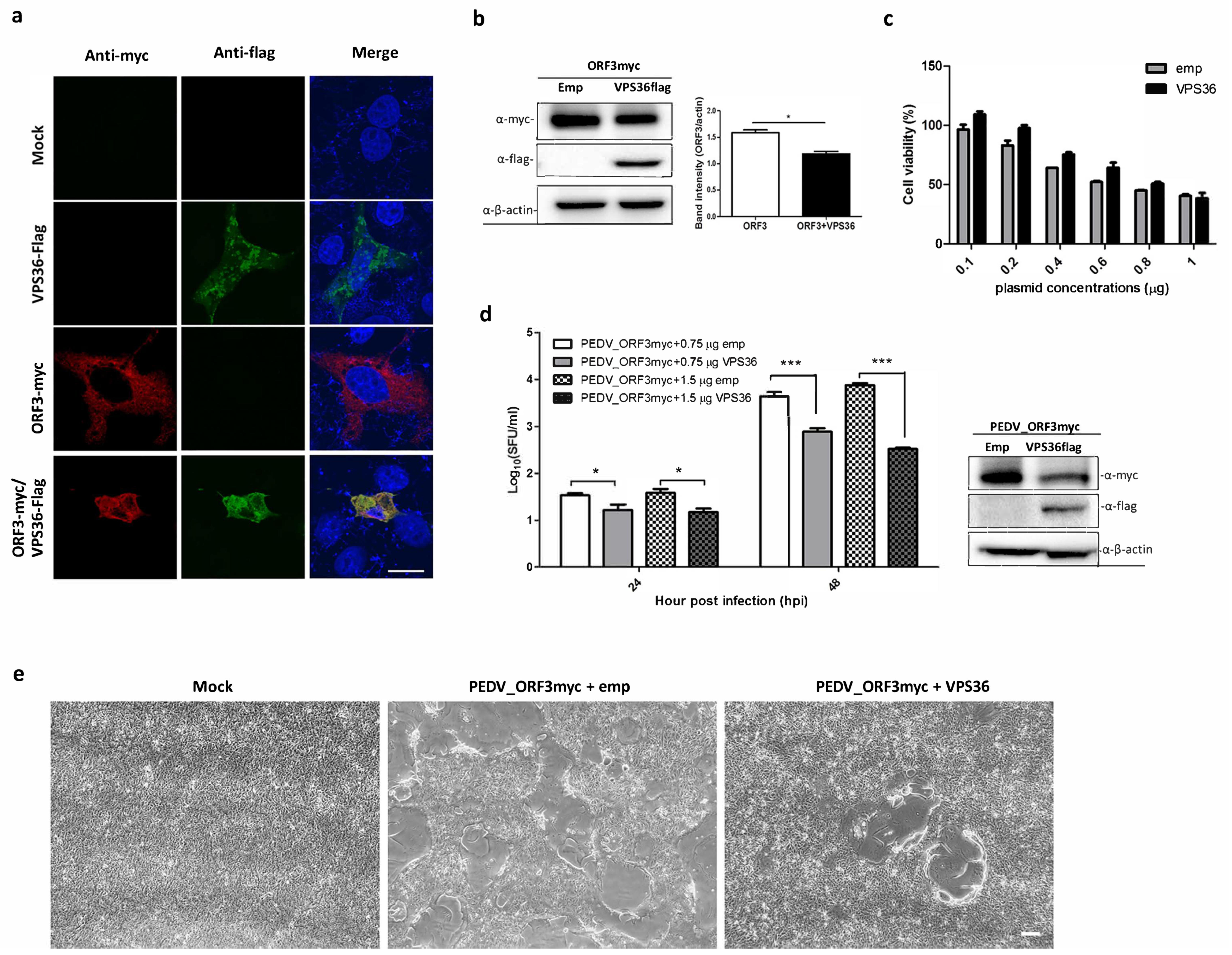
3.2. Validation of ORF3myc Interaction
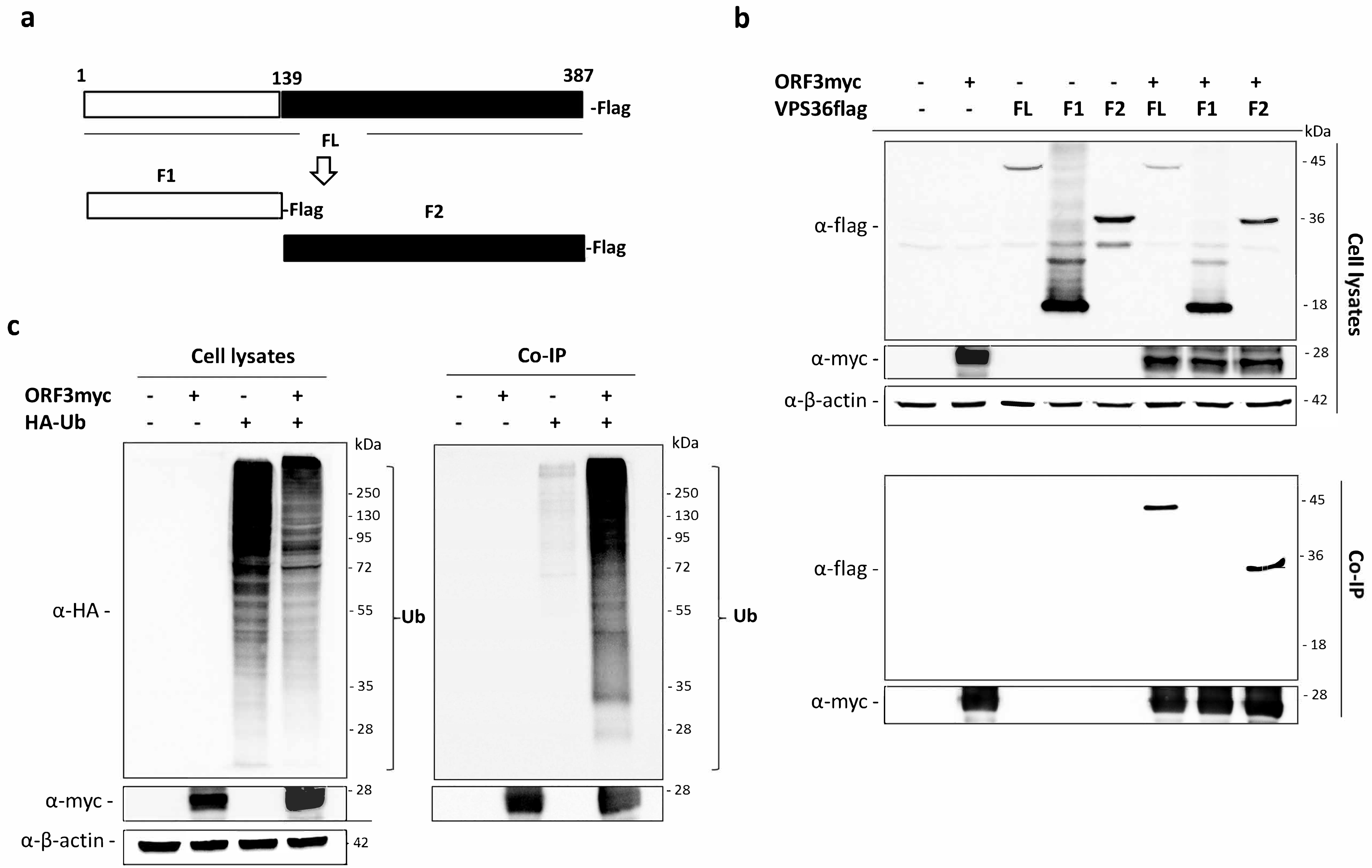
3.3. The GLUE Domain of VPS36 Is Not Required for Binding to Ubiquitinated ORF3
3.4. Overexpression of VPS36 Protein Suppresses PEDV Replication in VeroE6-APN Cells
3.5. Recovery of PEDVAV12_ORF3myc Growth in VPS36 Knock-Down Cells
3.6. ORF3 Degradation Was Not Recovered in VPS36-Overexpressed Cells Treated with Lysosome Inhibitor
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Debouck, P.; Pensaert, M. Experimental infection of pigs with a new porcine enteric coronavirus, CV777. Am. J. Vet. Res. 1980, 41, 219–223. [Google Scholar]
- Sun, R.Q.; Cai, R.J.; Chen, Y.Q.; Liang, P.S.; Chen, D.K.; Song, C.X. Outbreak of porcine epidemic diarrhea in suckling piglets, china. Emerg. Infect. Dis. 2012, 18, 161–163. [Google Scholar] [CrossRef]
- Kocherhans, R.; Bridgen, A.; Ackermann, M.; Tobler, K. Completion of the porcine epidemic diarrhoea coronavirus (PEDV) genome sequence. Virus Genes 2001, 23, 137–144. [Google Scholar] [CrossRef]
- De Haan, C.A.M.; Rottier, P.J.M. Molecular interactions in the assembly of coronaviruses. In Advances in virus research; Academic Press: Cambridge, MA, USA, 2005; Volume 64, pp. 165–230. [Google Scholar]
- Park, S.J.; Moon, H.J.; Luo, Y.; Kim, H.K.; Kim, E.M.; Yang, J.S.; Song, D.S.; Kang, B.K.; Lee, C.S.; Park, B.K. Cloning and further sequence analysis of the ORF3 gene of wild- and attenuated-type porcine epidemic diarrhea viruses. Virus Genes 2008, 36, 95–104. [Google Scholar] [CrossRef]
- Wang, K.; Lu, W.; Chen, J.; Xie, S.; Shi, H.; Hsu, H.; Yu, W.; Xu, K.; Bian, C.; Fischer, W.B.; et al. PEDV ORF3 encodes an ion channel protein and regulates virus production. FEBS Lett 2012, 586, 384–391. [Google Scholar] [CrossRef]
- Wongthida, P.; Liwnaree, B.; Wanasen, N.; Narkpuk, J.; Jongkaewwattana, A. The role of ORF3 accessory protein in replication of cell-adapted porcine epidemic diarrhea virus (PEDV). Arch. Virol. 2017, 162, 2553–2563. [Google Scholar] [CrossRef]
- Ye, S.; Li, Z.; Chen, F.; Li, W.; Guo, X.; Hu, H.; He, Q. Porcine epidemic diarrhea virus ORF3 gene prolongs S-phase, facilitates formation of vesicles and promotes the proliferation of attenuated PEDV. Virus Genes 2015, 51, 385–392. [Google Scholar] [CrossRef]
- Kaewborisuth, C.; He, Q.; Jongkaewwattana, A. The accessory protein ORF3 contributes to porcine epidemic diarrhea virus replication by direct binding to the spike protein. Viruses 2018, 10, 399. [Google Scholar] [CrossRef]
- Jitoboam, K.; Phaonakrop, N.; Libsittikul, S.; Thepparit, C.; Roytrakul, S.; Smith, D.R. Actin interacts with dengue virus 2 and 4 envelope proteins. Plos One 2016, 11, e0151951. [Google Scholar] [CrossRef]
- Wu, W.; Tran, K.C.; Teng, M.N.; Heesom, K.J.; Matthews, D.A.; Barr, J.N.; Hiscox, J.A. The interactome of the human respiratory syncytial virus NS1 protein highlights multiple effects on host cell biology. J. Virol. 2012, 86, 7777–7789. [Google Scholar] [CrossRef]
- Kuo, R.L.; Chen, C.J.; Tam, E.H.; Huang, C.G.; Li, L.H.; Li, Z.H.; Su, P.C.; Liu, H.P.; Wu, C.C. Interactome analysis of NS1 protein encoded by influenza a H7N9 virus reveals an inhibitory role of NS1 in host mRNA maturation. J. Proteome Res. 2018, 17, 1474–1484. [Google Scholar] [CrossRef]
- Jengarn, J.; Wongthida, P.; Wanasen, N.; Frantz, P.N.; Wanitchang, A.; Jongkaewwattana, A. Genetic manipulation of porcine epidemic diarrhoea virus recovered from a full-length infectious cDNA clone. J. Gen Virol. 2015, 96, 2206–2218. [Google Scholar] [CrossRef]
- Taylor, R.T.; Best, S.M. Assessing ubiquitination of viral proteins: Lessons from flavivirus NS5. Methods 2011, 55, 166–171. [Google Scholar] [CrossRef]
- Shevchenko, A.; Tomas, H.; Havlis, J.; Olsen, J.V.; Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 2006, 1, 2856–2860. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆CT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Perkins, D.N.; Pappin, D.J.; Creasy, D.M.; Cottrell, J.S. Probability-based protein identification by searching sequence databases using mass spectrometry data. Electrophoresis 1999, 20, 3551–3567. [Google Scholar] [CrossRef]
- Mi, H.; Muruganujan, A.; Thomas, P.D. Panther in 2013: Modeling the evolution of gene function, and other gene attributes, in the context of phylogenetic trees. Nucleic Acids Res. 2013, 41, D377–D386. [Google Scholar] [CrossRef]
- Mi, H.; Poudel, S.; Muruganujan, A.; Casagrande, J.T.; Thomas, P.D. Panther version 10: Expanded protein families and functions, and analysis tools. Nucleic Acids Res. 2016, 44, D336–D342. [Google Scholar] [CrossRef]
- Kumthip, K.; Yang, D.; Li, N.L.; Zhang, Y.; Fan, M.; Sethuraman, A.; Li, K. Pivotal role for the ESCRT-II complex subunit EAP30/SNF8 in IRF3-dependent innate antiviral defense. PLoS Pathog. 2017, 13, e1006713. [Google Scholar] [CrossRef]
- Stieler, J.T.; Prange, R. Involvement of ESCRT-II in hepatitis B virus morphogenesis. PLOS ONE 2014, 9, e91279. [Google Scholar] [CrossRef]
- Meng, B.; Ip, N.C.; Prestwood, L.J.; Abbink, T.E.; Lever, A.M. Evidence that the endosomal sorting complex required for transport-II (ESCRT-II) is required for efficient human immunodeficiency virus-1 (HIV-1) production. Retrovirology 2015, 12, 72. [Google Scholar] [CrossRef]
- Alam, S.L.; Langelier, C.; Whitby, F.G.; Koirala, S.; Robinson, H.; Hill, C.P.; Sundquist, W.I. Structural basis for ubiquitin recognition by the human ESCRT-II Eap45 GLUE domain. Nat. Struct. Mol. Biol. 2006, 13, 1029–1030. [Google Scholar] [CrossRef]
- Slagsvold, T.; Aasland, R.; Hirano, S.; Bache, K.G.; Raiborg, C.; Trambaiolo, D.; Wakatsuki, S.; Stenmark, H. Eap45 in mammalian ESCRT-II binds ubiquitin via a phosphoinositide-interacting GLUE domain. J. Biol. Chem. 2005, 280, 19600–19606. [Google Scholar] [CrossRef]
- Darniot, M.; Schildgen, V.; Schildgen, O.; Sproat, B.; Kleines, M.; Ditt, V.; Pitoiset, C.; Pothier, P.; Manoha, C. RNA interference in vitro and in vivo using DsiRNA targeting the nucleocapsid N mRNA of human metapneumovirus. Antivir. Res. 2012, 93, 364–373. [Google Scholar] [CrossRef]
- Burkard, C.; Verheije, M.H.; Wicht, O.; van Kasteren, S.I.; van Kuppeveld, F.J.; Haagmans, B.L.; Pelkmans, L.; Rottier, P.J.M.; Bosch, B.J.; de Haan, C.A.M. Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner. PLoS Pathog. 2014, 10, e1004502. [Google Scholar] [CrossRef]
- Teo, H.; Perisic, O.; Gonzalez, B.; Williams, R.L. ESCRT-II, an endosome-associated complex required for protein sorting: Crystal structure and interactions with ESCRT-III and membranes. Dev. Cell 2004, 7, 559–569. [Google Scholar] [CrossRef]
- Ooi, E.E.; Chew, J.S.; Loh, J.P.; Chua, R.C. In vitro inhibition of human influenza A virus replication by chloroquine. Virol. J. 2006, 3, 39. [Google Scholar] [CrossRef]
- Delvecchio, R.; Higa, L.M.; Pezzuto, P.; Valadão, A.L.; Garcez, P.P.; Monteiro, F.L.; Loiola, E.C.; Dias, A.A.; Silva, F.J.M.; Aliota, M.T.; et al. Chloroquine, an endocytosis blocking agent, inhibits Zika virus infection in different cell models. Viruses 2016, 8, 322. [Google Scholar] [CrossRef]
- Gomez-Navarro, N.; Miller, E. Protein sorting at the ER-Golgi interface. J. Cell Biol. 2016, 215, 769. [Google Scholar] [CrossRef]
- Thomas, P.D.; Campbell, M.J.; Kejariwal, A.; Mi, H.; Karlak, B.; Daverman, R.; Diemer, K.; Muruganujan, A.; Narechania, A. Panther: A library of protein families and subfamilies indexed by function. Genome Res. 2003, 13, 2129–2141. [Google Scholar] [CrossRef]
- Zhang, Q.; Ma, J.; Yoo, D. Inhibition of NF-kappaB activity by the porcine epidemic diarrhea virus nonstructural protein 1 for innate immune evasion. Virology 2017, 510, 111–126. [Google Scholar] [CrossRef]
- Frankel, E.B.; Audhya, A. ESCRT-dependent cargo sorting at multivesicular endosomes. Semin. Cell Dev. Biol. 2018, 74, 4–10. [Google Scholar] [CrossRef]
- Mageswaran, S.K.; Dixon, M.G.; Curtiss, M.; Keener, J.P.; Babst, M. Binding to any ESCRT can mediate ubiquitin-independent cargo sorting. Traffic 2014, 15, 212–229. [Google Scholar] [CrossRef]
| Protein Name; Gene Name | Binding Ratio |
|---|---|
| Human Glutathione Transferase O2; GSTP1 | 18.25905 |
| ADAMTS-like protein 3; ADAMTSL3 | 18.0177 |
| Sorting nexin-13; SNX13 | 17.84919 |
| Zinc finger protein 443; ZNF443 | 17.36122 |
| NDUFA13 protein; NDUFA13 | 17.18651 |
| Frizzled-6; FZD6 | 17.17375 |
| S-adenosylmethionine synthase isoform type-2; MAT2A | 16.98951 |
| Protein ripply3; RIPPLY3 | 16.88675 |
| JAW1-related protein MRVI1B; MRVI1B | 16.86521 |
| Calmodulin isoform 2; CALM1 | 16.71276 |
| Splicing factor, suppressor of white-apricot homolog; SFSWAP | 16.53506 |
| Signal transducer and activator of transcription 4; STAT4 | 16.40851 |
| Pre-mRNA 3’-end-processing factor FIP1; FIP1L1 | 16.19049 |
| Plasminogen; PLAU | 15.85092 |
| Inhibitor of nuclear factor kappa-B kinase subunit beta; IKBKB | 15.5677 |
| Mucolipin-2; MCOLN2 | 15.31623 |
| protein TANC1 isoform X25; TANC1 | 15.10605 |
| Vacuolar protein-sorting-associated protein 36; VPS36 | 14.34693 |
| Probable E3 ubiquitin-protein ligase MARCH10; MARCH10 | 13.37567 |
| Centrosomal protein of 44 kDa; CEP44 | 13.13018 |
| ATP synthase mitochondrial F1 complex assembly factor 2 isoform X4; ATPAF2 | 12.90607 |
| Xaa-Pro aminopeptidase 3; XPNPEP3 | 10.94451 |
| DsiRNAs/Primers | Sequence |
|---|---|
| DsiRNAs | |
| DsiVPS36#1 | 5’-GUCAUGGUAAUUGAGCUUCAGUCUC-3’ |
| 3’-CGCAGUACCAUUAACUCGAAGUCAGAG-5’ | |
| DsiVPS36#2 | 5’-AAGAUGAGACCAUCAGGUUUAAAUC-3’ |
| 3’-UCUUCUACUCUGGUAGUCCAAAUUUAG-5’ | |
| Negative control DsiRNA (NC) | 5’-CGUUAAUCGCGUAUAAUACGCGUAT-3’ |
| 3’- CAGCAAUUAGCGCAUAUUAUGCGCAUA-5’ | |
| Primers for RT-qPCR | |
| VPS36_F | 5’-GCCCTGGAGACAGTTTCAGA-3’ |
| VPS36_R | 5’-CTGAGTCATCACGGCAAAGA-3’ |
| GAPDH_F | 5’-CCCTTCATTGACCTCAACTACAT-3’ |
| GAPDH_R | 5’-ACGATACCAAAGTTGTCATGG-3’ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaewborisuth, C.; Yingchutrakul, Y.; Roytrakul, S.; Jongkaewwattana, A. Porcine Epidemic Diarrhea Virus (PEDV) ORF3 Interactome Reveals Inhibition of Virus Replication by Cellular VPS36 Protein. Viruses 2019, 11, 382. https://doi.org/10.3390/v11040382
Kaewborisuth C, Yingchutrakul Y, Roytrakul S, Jongkaewwattana A. Porcine Epidemic Diarrhea Virus (PEDV) ORF3 Interactome Reveals Inhibition of Virus Replication by Cellular VPS36 Protein. Viruses. 2019; 11(4):382. https://doi.org/10.3390/v11040382
Chicago/Turabian StyleKaewborisuth, Challika, Yodying Yingchutrakul, Sittiruk Roytrakul, and Anan Jongkaewwattana. 2019. "Porcine Epidemic Diarrhea Virus (PEDV) ORF3 Interactome Reveals Inhibition of Virus Replication by Cellular VPS36 Protein" Viruses 11, no. 4: 382. https://doi.org/10.3390/v11040382
APA StyleKaewborisuth, C., Yingchutrakul, Y., Roytrakul, S., & Jongkaewwattana, A. (2019). Porcine Epidemic Diarrhea Virus (PEDV) ORF3 Interactome Reveals Inhibition of Virus Replication by Cellular VPS36 Protein. Viruses, 11(4), 382. https://doi.org/10.3390/v11040382






