A Functional Ubiquitin-Proteasome System is Required for Efficient Replication of New World Mayaro and Una Alphaviruses
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell culture, Virus Strains Propagation and Reagents
2.2. Cell Viability Assay
2.3. Plaque Assay
2.4. Immunofluorescence and Confocal Microscopy Analysis
2.5. Protein Analysis
2.6. Design, Generation and Validation of Polyclonal Antibodies Against MAYV E1 and nsP1 Protein
2.7. Statistical Analysis
3. Results
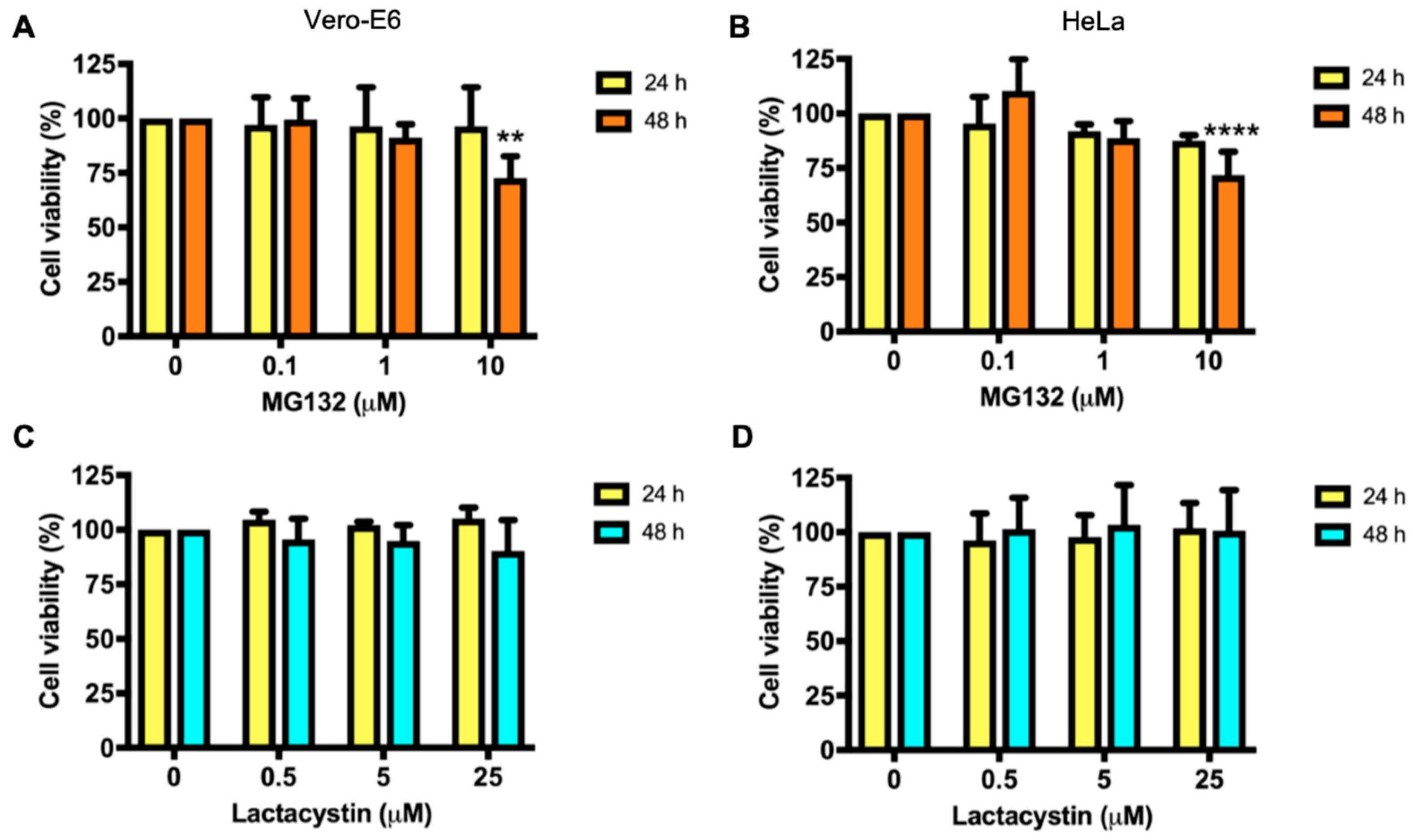
3.1. Effect of Proteasome Inhibitors in Vero-E6 and HeLa Cells
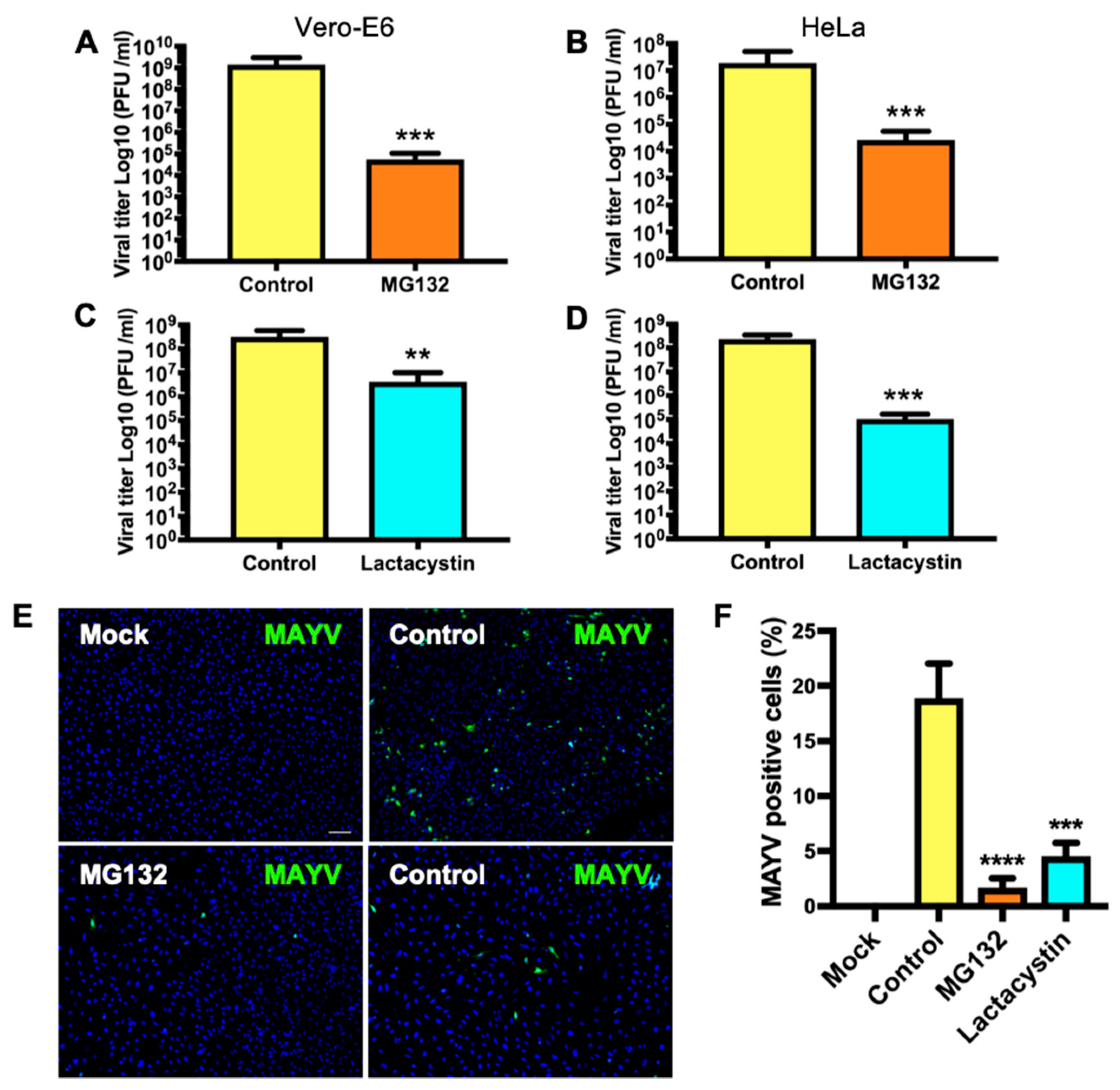
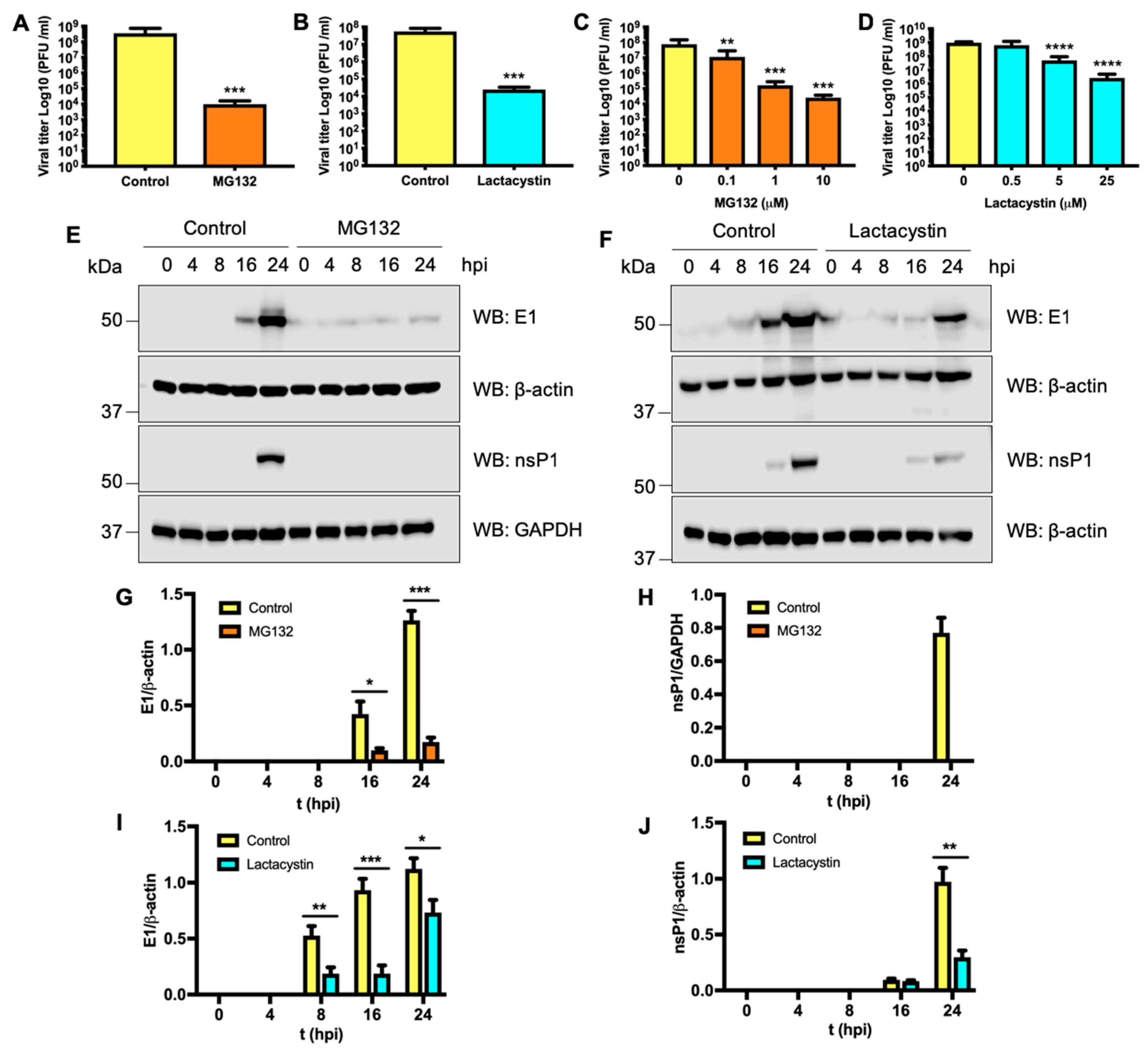
3.2. Treatment with Proteasome Inhibitors MG132 or Lactacystin Reduces MAYV Progeny Production in Vero-E6 and HeLa Cells
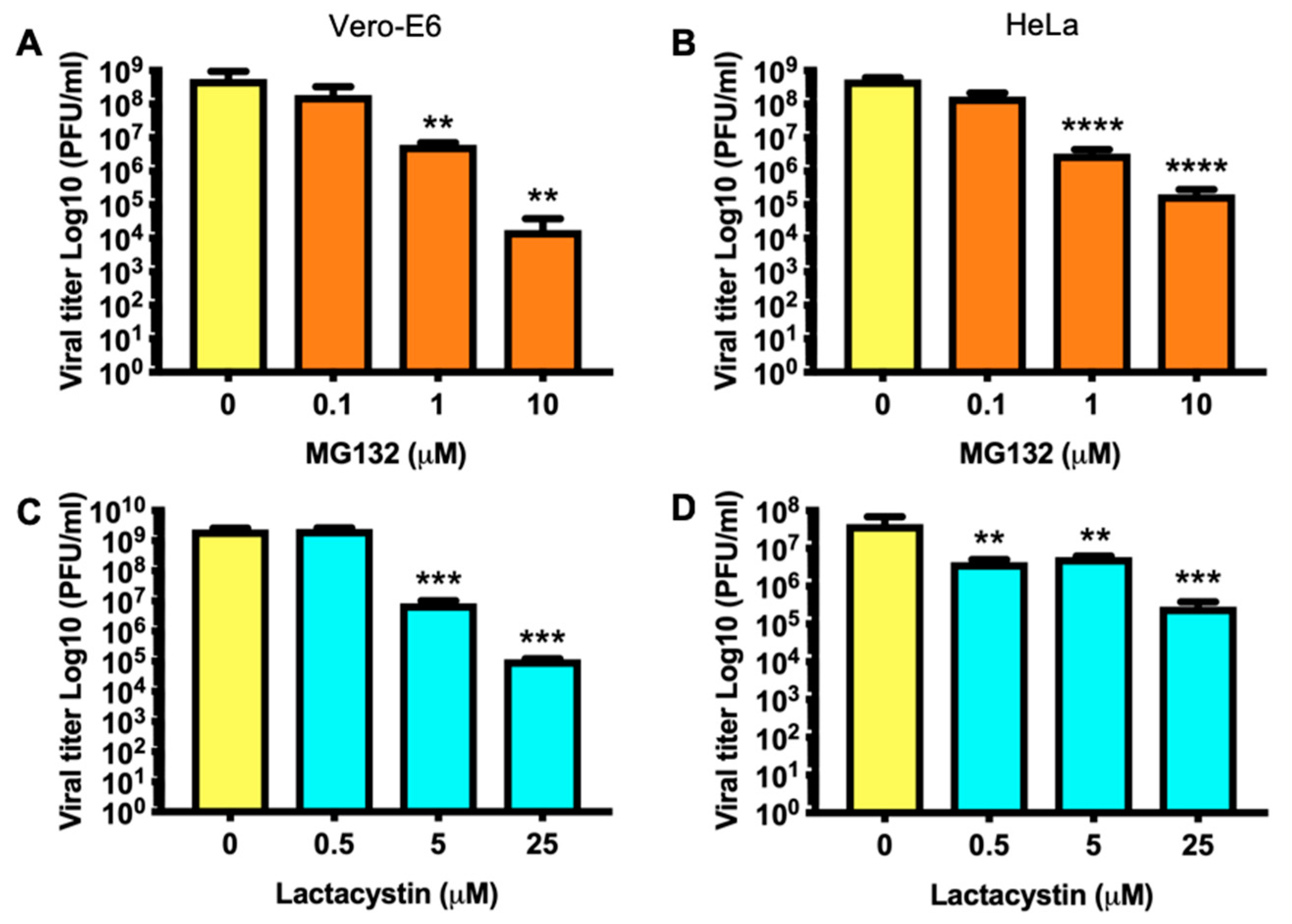
3.3. Decrease in MAYV Titers in Response to Proteasome Inhibitors Occurs in a Dose-Dependent Manner
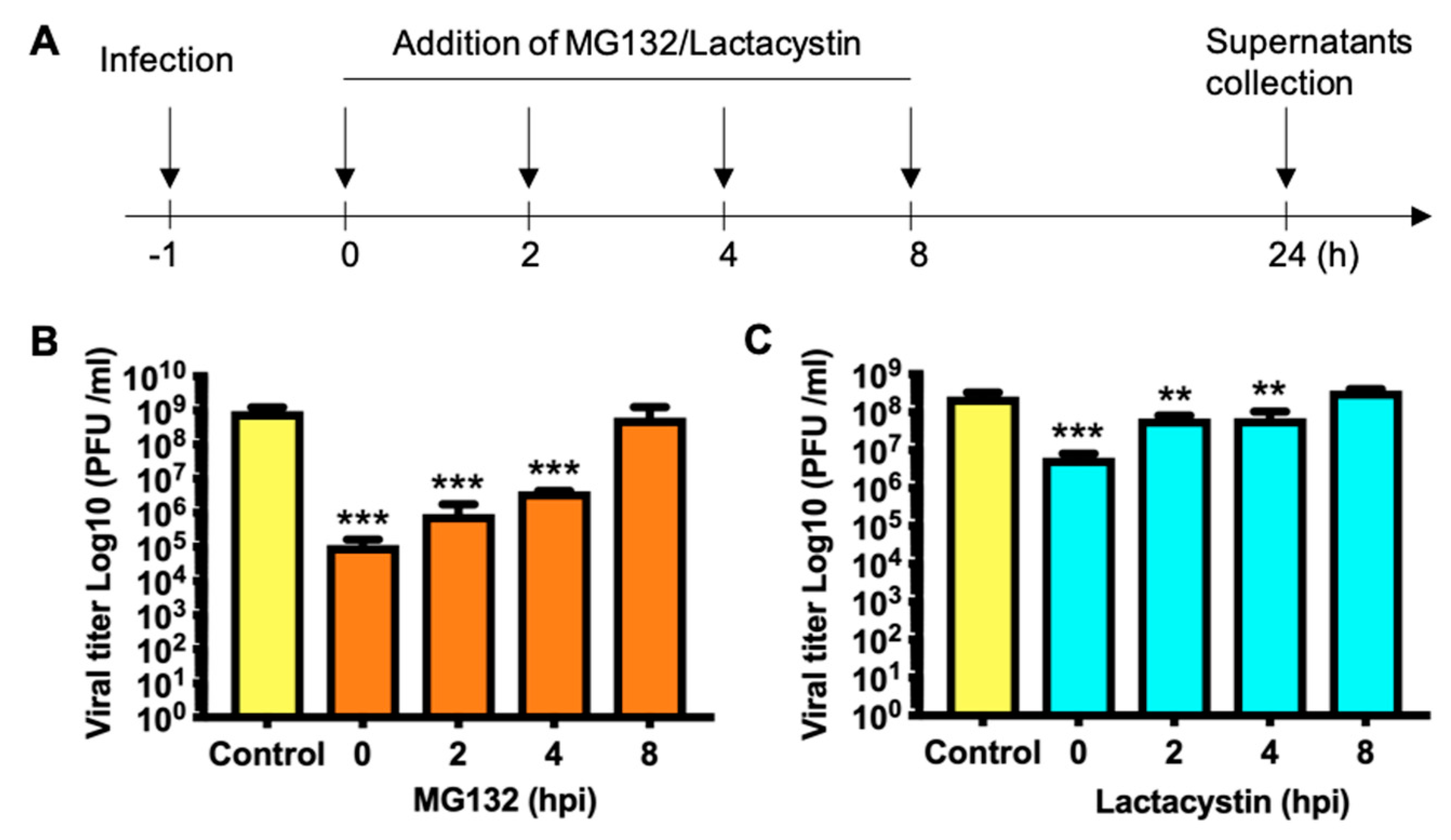
3.4. Proteasome Activity is Necessary in the First Stage of the MAYV Replication Cycle
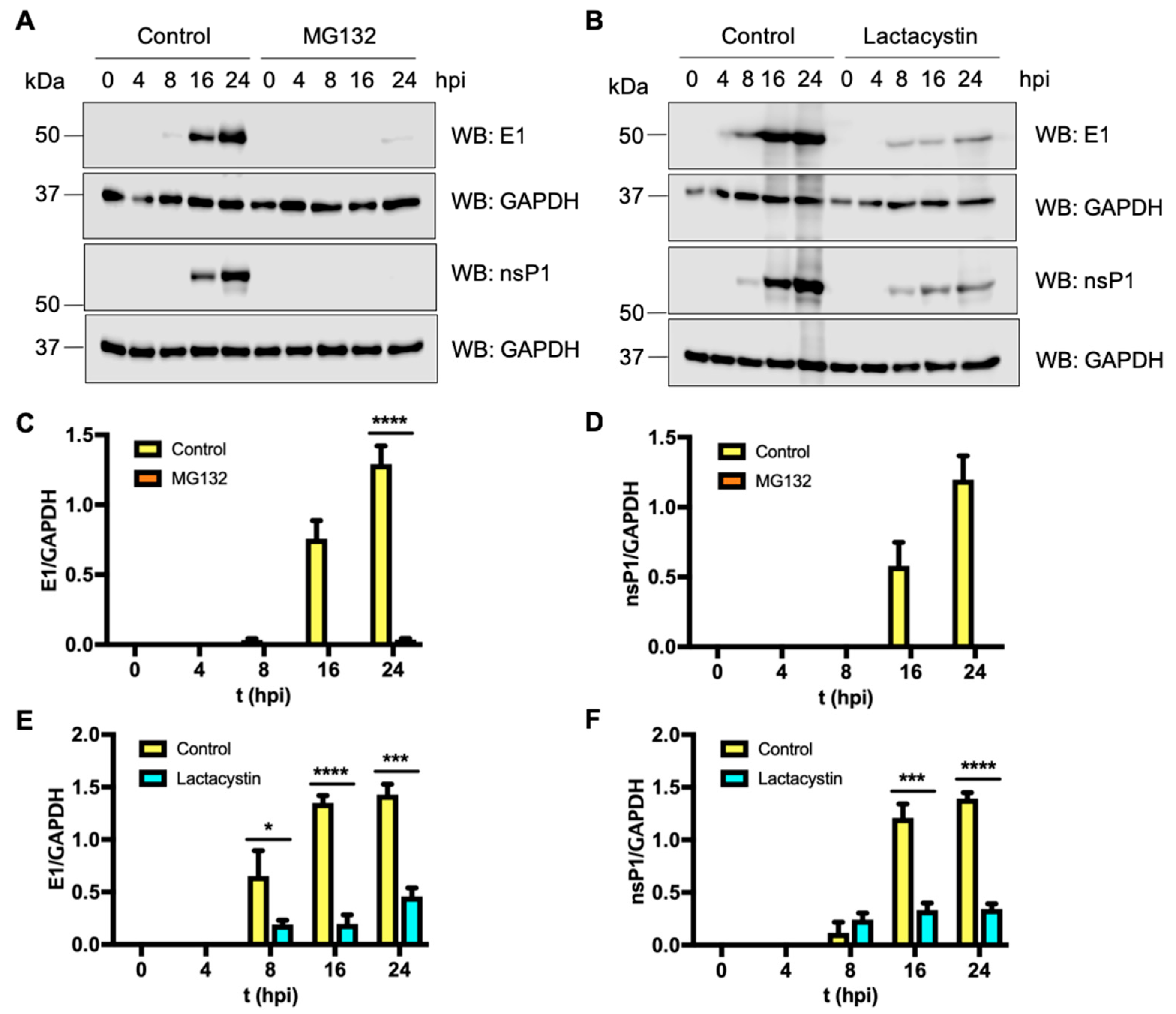
3.5. Proteasome Inhibition Impairs MAYV Viral Protein Synthesis
3.6. Proteasome Activity is also Required for UNAV Replication
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weaver, S.C. Urbanization and geographic expansion of zoonotic arboviral diseases: Mechanisms and potential strategies for prevention. Trends Microbiol. 2013, 21, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, P.F.C.; Calisher, C.H. Emergence of Human Arboviral Diseases in the Americas, 2000–2016. Vector-Borne Zoonotic Dis. 2016, 16, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antiviral Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [PubMed]
- Marcondes, C.B.; Contigiani, M.; Gleiser, R.M. Emergent and reemergent arboviruses in South America and the Caribbean: Why so many and why now? J. Med. Entomol. 2017, 54, 509–532. [Google Scholar] [CrossRef]
- De Figueiredo, M.L.; Figueiredo, L.T. Emerging alphaviruses in the americas: Chikungunya and mayaro. Rev. Soc. Bras. Med. Trop. 2014, 47, 677–683. [Google Scholar] [CrossRef]
- Acosta-Ampudia, Y.; Monsalve, D.M.; Rodriguez, Y.; Pacheco, Y.; Anaya, J.-M.; Ramirez-Santana, C. Mayaro: an emerging viral threat? Emerg. Microbes Infect. 2018, 7, 163. [Google Scholar] [CrossRef]
- Anderson, C.R.; Downs, W.G.; Wattley, G.H.; Ahin, N.W.; Reese, A.A. Mayaro virus: a new human disease agent. II. Isolation from blood of patients in Trinidad, B.W.I. Am. J. Trop. Med. Hyg. 1957, 6, 1012–1016. [Google Scholar] [CrossRef]
- Long, K.C.; Ziegler, S.A.; Thangamani, S.; Hausser, N.L.; Kochel, T.J.; Higgs, S.; Tesh, R.B. Experimental transmission of Mayaro virus by Aedes aegypti. Am. J. Trop. Med. Hyg. 2011, 85, 750–757. [Google Scholar] [CrossRef]
- Wiggins, K.; Eastmond, B.; Alto, B.W. Transmission potential of Mayaro virus in Florida Aedes aegypti and Aedes albopictus mosquitoes. Med. Vet. Entomol. 2018, 32, 436–442. [Google Scholar] [CrossRef]
- Tesh, R.B.; Watts, D.M.; Russell, K.L.; Damodaran, C.; Calampa, C.; Cabezas, C.; Ramirez, G.; Vasquez, B.; Hayes, C.G.; Rossi, C.A.; et al. Mayaro Virus Disease: An Emerging Mosquito-Borne Zoonosis in Tropical South America. Clin. Infect. Dis. 1999, 28, 67–73. [Google Scholar] [CrossRef]
- Santiago, F.W.; Halsey, E.S.; Siles, C.; Vilcarromero, S.; Guevara, C.; Silvas, J.A.; Ramal, C.; Ampuero, J.S.; Aguilar, P.V. Long-Term Arthralgia after Mayaro Virus Infection Correlates with Sustained Pro-inflammatory Cytokine Response. PLoS Negl. Trop. Dis. 2015, 9, e0004104. [Google Scholar] [CrossRef]
- LeDuc, J.W.; Pinheiro, F.P.; Travassos Da Rosa, A.P.A. An outbreak of Mayaro virus disease in Belterra, Brazil. II. Epidemiology. Am. J. Trop. Med. Hyg. 1981, 30, 682–688. [Google Scholar] [CrossRef]
- Mourão, M.P.G.; de Bastos, M.S.; de Figueiredo, R.P.; Gimaque, J.B.L.; dos Santos Galusso, E.; Kramer, V.M.; de Oliveira, C.M.C.; Naveca, F.G.; Figueiredo, L.T.M. Mayaro Fever in the City of Manaus, Brazil, 2007–2008. Vector-Borne Zoonotic Dis. 2012, 12, 42–46. [Google Scholar]
- Halsey, E.S.; Siles, C.; Guevara, C.; Vilcarromero, S.; Jhonston, E.J.; Ramal, C.; Aguilar, P.V.; Ampuero, J.S. Mayaro virus infection, Amazon Basin region, Peru, 2010–2013. Emerg. Infect. Dis. 2013, 19, 1839–1842. [Google Scholar] [CrossRef]
- De Thoisy, B.; Gardon, J.; Alba Salas, R.; Morvan, J.; Kazanji, M. Mayaro virus in wild mammals, French Guiana. Emerg. Infect. Dis. 2003, 9, 1326–1329. [Google Scholar] [CrossRef]
- Torres, J.R.; Russell, K.L.; Vasquez, C.; Barrera, R.; Tesh, R.B.; Salas, R.; Watts, D.M. Family cluster of Mayaro fever, Venezuela. Emerg. Infect. Dis. 2004, 10, 1304–1306. [Google Scholar] [CrossRef]
- Muñoz, M.; Navarro, J.C. Virus Mayaro: un arbovirus reemergente en Venezuela y Latinoamérica. Biomédica 2012, 32, 288–302. [Google Scholar] [CrossRef]
- Izurieta, R.; Macaluso, M.; Watts, D.; Tesh, R.; Guerra, B.; Cruz, L.; Galwankar, S.; Vermund, S. Hunting in the rainforest and mayaro virus infection: An emerging alphavirus in Ecuador. J. Glob. Infect. Dis. 2011, 3, 317–323. [Google Scholar] [CrossRef]
- Galindo, P.; Srihongse, S.; De Rodaniche, E.; Grayson, M.A. An ecological survey for arboviruses in Almirante, Panama, 1959–1962. Am. J. Trop. Med. Hyg. 1966, 15, 385–400. [Google Scholar] [CrossRef]
- Carrera, J.P.; Bagamian, K.H.; Travassos Da Rosa, A.P.; Wang, E.; Beltran, D.; Gundaker, N.D.; Armien, B.; Arroyo, G.; Sosa, N.; Pascale, J.M.; et al. Human and equine infection with alphaviruses and flaviviruses in panamá during 2010: A cross-Sectional study of household contacts during an encephalitis outbreak. Am. J. Trop. Med. Hyg. 2018, 98, 1798–1804. [Google Scholar] [CrossRef]
- Lednicky, J.; Beau De Rochars, V.M.; Elbadry, M.; Loeb, J.; Telisma, T.; Chavannes, S.; Anilis, G.; Cella, E.; Ciccozzi, M.; Okech, B.; et al. Mayaro virus in child with acute febrile illness, Haiti, 2015. Emerg. Infect. Dis. 2016, 22, 2000–2002. [Google Scholar] [CrossRef]
- Hotez, P.J.; Murray, K.O. Dengue, West Nile virus, chikungunya, Zika-and now Mayaro? PLoS Negl. Trop. Dis. 2017, 11, e0005462. [Google Scholar] [CrossRef]
- Lavergne, A.; de Thoisy, B.; Lacoste, V.; Pascalis, H.; Pouliquen, J.F.; Mercier, V.; Tolou, H.; Dussart, P.; Morvan, J.; Talarmin, A.; et al. Mayaro virus: Complete nucleotide sequence and phylogenetic relationships with other alphaviruses. Virus Res. 2006, 117, 283–290. [Google Scholar] [CrossRef]
- Powers, A.M.; Aguilar, P.V.; Chandler, L.J.; Brault, A.C.; Meakins, T.A.; Watts, D.; Russell, K.L.; Olson, J.; Vasconcelos, P.F.C.; Da Rosa, A.T.; et al. Genetic relationships among Mayaro and Una viruses suggest distinct patterns of transmission. Am. J. Trop. Med. Hyg. 2006, 75, 461–469. [Google Scholar] [CrossRef]
- Causey, O.R.; Casals, J.; Shope, R.E.; Udomsakdi, S. Aura and Una, Two New Group A Arthropod-Borne Viruses. Am. J. Trop. Med. Hyg. 1963, 12, 777–781. [Google Scholar] [CrossRef]
- Peralta, P.H.; Shelokov, A. Isolation and characterization of arboviruses from Almirante, Republic of Panama. Am. J. Trop. Med. Hyg. 1966, 15, 369–378. [Google Scholar] [CrossRef]
- Haas, R.A.; Arron-Leeuwin, A.E. Arboviruses isolated from mosquitos and man in Surinam. Trop. Geogr. Med. 1975, 27, 409–412. [Google Scholar]
- Walder, R.; Suarez, O.M.; Calisher, C.H. Arbovirus studies in southwestern Venezuela during 1973–1981. II. Isolations and further studies of Venezuelan and eastern equine encephalitis, Una, Itaqui, and Moju viruses. Am. J. Trop. Med. Hyg. 1984, 33, 483–491. [Google Scholar] [CrossRef]
- Díaz, L.A.; del Pilar Díaz, M.; Almirón, W.R.; Contigiani, M.S. Infection by UNA virus (Alphavirus; Togaviridae) and risk factor analysis in black howler monkeys (Alouatta caraya) from Paraguay and Argentina. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 1039–1041. [Google Scholar] [CrossRef]
- Diaz, L.A.; Spinsanti, L.I.; Almiron, W.R.; Contigiani, M.S. Una virus: First report of human infection in Argentina. Rev. Inst. Med. Trop. Sao Paulo 2003, 45, 109–110. [Google Scholar] [CrossRef]
- Cardozo, F.; Konigheim, B.; Albrieu-Llinás, G.; Rivarola, M.E.; Aguilar, J.; Rojas, A.; Quaglia, A.I.; Paez, M.; Guillén, Y.; Diaz, A.; et al. Alphaviruses: Serological Evidence of Human Infection in Paraguay (2012–2013). Vector-Borne Zoonotic Dis. 2018, 18, 266–272. [Google Scholar] [CrossRef]
- Isaacson, M.K.; Ploegh, H.L. Ubiquitination, Ubiquitin-like Modifiers, and Deubiquitination in Viral Infection. Cell Host Microbe 2009, 5, 559–570. [Google Scholar] [CrossRef]
- Tang, Q.; Wu, P.; Chen, H.; Li, G. Pleiotropic roles of the ubiquitin-proteasome system during viral propagation. Life Sci. 2018, 207, 350–354. [Google Scholar] [CrossRef]
- Clague, M.J.; Urbé, S. Ubiquitin: Same molecule, different degradation pathways. Cell 2010, 143, 682–685. [Google Scholar] [CrossRef]
- Swatek, K.N.; Komander, D. Ubiquitin modifications. Cell Res. 2016, 26, 399–422. [Google Scholar] [CrossRef]
- Viswanathan, K.; Früh, K.; DeFilippis, V. Viral hijacking of the host ubiquitin system to evade interferon responses. Curr. Opin. Microbiol. 2010, 13, 517–523. [Google Scholar] [CrossRef]
- Contin, R.; Arnoldi, F.; Mano, M.; Burrone, O.R. Rotavirus Replication Requires a Functional Proteasome for Effective Assembly of Viroplasms. J. Virol. 2011, 85, 2781–2792. [Google Scholar] [CrossRef]
- Karpe, Y.A.; Meng, X.-J. Hepatitis E Virus Replication Requires an Active Ubiquitin-Proteasome System. J. Virol. 2012, 86, 5948–5952. [Google Scholar] [CrossRef]
- Barrado-Gil, L.; Galindo, I.; Martínez-Alonso, D.; Viedma, S.; Alonso, C. The ubiquitin-proteasome system is required for African swine fever replication. PLoS ONE 2017, 12, e0189741. [Google Scholar] [CrossRef]
- Cheng, S.; Yan, W.; Gu, W.; He, Q. The ubiquitin-proteasome system is required for the early stages of porcine circovirus type 2 replication. Virology 2014, 456–457, 198–204. [Google Scholar] [CrossRef]
- Widjaja, I.; de Vries, E.; Tscherne, D.M.; Garcia-Sastre, A.; Rottier, P.J.M.; de Haan, C.A.M. Inhibition of the Ubiquitin-Proteasome System Affects Influenza A Virus Infection at a Postfusion Step. J. Virol. 2010, 84, 9625–9631. [Google Scholar] [CrossRef] [PubMed]
- Lupfer, C.; Pastey, M.K. Decreased replication of human respiratory syncytial virus treated with the proteasome inhibitor MG-132. Virus Res. 2010, 149, 36–41. [Google Scholar] [CrossRef]
- Akhrymuk, I.; Kulemzin, S.V.; Frolova, E.I. Evasion of the Innate Immune Response: the Old World Alphavirus nsP2 Protein Induces Rapid Degradation of Rpb1, a Catalytic Subunit of RNA Polymerase II. J. Virol. 2012, 86, 7180–7191. [Google Scholar] [CrossRef] [PubMed]
- Amaya, M.; Keck, F.; Lindquist, M.; Voss, K.; Scavone, L.; Kehn-Hall, K.; Roberts, B.; Bailey, C.; Schmaljohn, C.; Narayanan, A. The ubiquitin proteasome system plays a role in Venezuelan equine encephalitis virus infection. PLoS ONE 2015, 10, e0124792. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Goldberg, A.L. Proteasome inhibitors: Valuable new tools for cell biologists. Trends Cell Biol. 1998, 8, 397–403. [Google Scholar] [CrossRef]
- Fenteany, G.; Schreiber, S.L. Lactacystin, proteasome function, and cell fate. J. Biol. Chem. 1998, 273, 8545–8548. [Google Scholar] [CrossRef]
- González-Santamaría, J.; Campagna, M.; García, M.A.; Marcos-Villar, L.; González, D.; Gallego, P.; Lopitz-Otsoa, F.; Guerra, S.; Rodríguez, M.S.; Esteban, M.; et al. Regulation of vaccinia virus E3 protein by small ubiquitin-like modifier proteins. J. Virol. 2011, 85, 12890–12900. [Google Scholar]
- Thrower, J.S.; Hoffman, L.; Rechsteiner, M.; Pickart, C.M. Recognition of the polyubiquitin proteolytic signal. EMBO J. 2000, 19, 94–102. [Google Scholar] [CrossRef]
- Karpe, Y.A.; Pingale, K.D.; Kanade, G.D. Activities of proteasome and m-calpain are essential for Chikungunya virus replication. Virus Genes 2016, 52, 716–721. [Google Scholar] [CrossRef] [PubMed]
- La Frazla, S.; Amici, C.; Santoro, M.G. Antiviral activity of proteasome inhibitors in herpes simplex virus-1 infection: Role of nuclear factor-κB. Antivir. Ther. 2006, 11, 995–1004. [Google Scholar]
- Watanabe, H.; Tanaka, Y.; Shimazu, Y.; Sugahara, F.; Kuwayama, M.; Hiramatsu, A.; Kiyotani, K.; Yoshida, T.; Sakaguchi, T. Cell-specific inhibition of paramyxovirus maturation by proteasome inhibitors. Microbiol. Immunol. 2005, 49, 835–844. [Google Scholar] [CrossRef] [PubMed]
- Harty, R.N.; Brown, M.E.; McGettigan, J.P.; Wang, G.; Jayakar, H.R.; Huibregtse, J.M.; Whitt, M.A.; Schnell, M.J. Rhabdoviruses and the Cellular Ubiquitin-Proteasome System: a Budding Interaction. J. Virol. 2001, 75, 10623–10629. [Google Scholar] [CrossRef]
- Schubert, U.; Ott, D.E.; Chertova, E.N.; Welker, R.; Tessmer, U.; Princiotta, M.F.; Bennink, J.R.; Krausslich, H.-G.; Yewdell, J.W. Proteasome inhibition interferes with Gag polyprotein processing, release, and maturation of HIV-1 and HIV-2. Proc. Natl. Acad. Sci. 2000, 97, 13057–13062. [Google Scholar] [CrossRef]
- Satheshkumar, P.S.; Anton, L.C.; Sanz, P.; Moss, B. Inhibition of the Ubiquitin-Proteasome System Prevents Vaccinia Virus DNA Replication and Expression of Intermediate and Late Genes. J. Virol. 2009, 83, 2469–2479. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Schmitt, S.; Frezza, M.; Kanwar, J.; Dou, Q.P. Bortezomib as the First Proteasome Inhibitor Anticancer Drug: Current Status and Future Perspectives. Curr. Cancer Drug Targets 2011, 11, 239–253. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Llamas-González, Y.Y.; Campos, D.; Pascale, J.M.; Arbiza, J.; González-Santamaría, J. A Functional Ubiquitin-Proteasome System is Required for Efficient Replication of New World Mayaro and Una Alphaviruses. Viruses 2019, 11, 370. https://doi.org/10.3390/v11040370
Llamas-González YY, Campos D, Pascale JM, Arbiza J, González-Santamaría J. A Functional Ubiquitin-Proteasome System is Required for Efficient Replication of New World Mayaro and Una Alphaviruses. Viruses. 2019; 11(4):370. https://doi.org/10.3390/v11040370
Chicago/Turabian StyleLlamas-González, Yessica Y., Dalkiria Campos, Juan M. Pascale, Juan Arbiza, and José González-Santamaría. 2019. "A Functional Ubiquitin-Proteasome System is Required for Efficient Replication of New World Mayaro and Una Alphaviruses" Viruses 11, no. 4: 370. https://doi.org/10.3390/v11040370
APA StyleLlamas-González, Y. Y., Campos, D., Pascale, J. M., Arbiza, J., & González-Santamaría, J. (2019). A Functional Ubiquitin-Proteasome System is Required for Efficient Replication of New World Mayaro and Una Alphaviruses. Viruses, 11(4), 370. https://doi.org/10.3390/v11040370




