Epidemiology of Norovirus Outbreaks Reported to the Public Health Emergency Event Surveillance System, China, 2014–2017
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Outbreak Definitions
2.3. Data Management and Analysis
2.4. Ethics Statement
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
Appendix A
References
- Ahmed, S.M.; Hall, A.J.; Robinson, A.E.; Verhoef, L.; Premkumar, P.; Parashar, U.D.; Koopmans, M.; Lopman, B.A. Global prevalence of norovirus in cases of gastroenteritis: A systematic review and meta-analysis. Lancet Infect. Dis. 2014, 14, 725–730. [Google Scholar] [CrossRef]
- Patel, M.M.; Hall, A.J.; Vinjé, J.; Parashar, U.D. Noroviruses: A comprehensive review. J. Clin. Virol. 2009, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Pires, S.M.; Fischer-Walker, C.L.; Lanata, C.F.; Brecht, D.; Hall, A.J.; Kirk, M.D.; Duarte, A.S.R.; Black, R.E.; Angulo, F.J. Aetiology-Specific Estimates of the Global and Regional Incidence and Mortality of Diarrhoeal Diseases Commonly Transmitted through Food. PLoS ONE 2015, 10, e0142927. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.I.; Parashar, U.D.; Estes, M.K. Norovirus gastroenteritis. N. Engl. J. Med. 2009, 361, 1776–1785. [Google Scholar] [CrossRef] [PubMed]
- Atmar, R.L.; Opekun, A.R.; Gilger, M.A.; Estes, M.K.; Crawford, S.E.; Neill, F.H.; Sasirekha, R.; Heather, H.; Jennifer, F.; Graham, D.Y. Determination of the 50% human infectious dose for Norwalk virus. J. Infect. Dis. 2014, 209, 1016. [Google Scholar] [CrossRef]
- Teunis, P.; Moe, C.P.; E-Miller, S.; Lindesmith, L.; Baric, R.; Le-Pendu, J.; Calderon, R. Norwalk virus: How infectious is it? J. Med. Virol. 2010, 80, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.J.; Wikswo, M.E.; Manikonda, K.; Roberts, V.A.; Yoder, J.S.; Gould, L.H. Acute gastroenteritis surveillance through the National Outbreak Reporting System, United States. Emerg. Infect. Dis. 2013, 19, 1305–1309. [Google Scholar] [CrossRef]
- Atmar, R.L.; Opekun, A.R.; Gilger, M.A.; Estes, M.K.; Crawford, S.E.; Neill, F.H.; Graham, D.Y. Norwalk virus shedding after experimental human infection. Emerg. Infect. Dis. 2008, 14, 1553–1557. [Google Scholar] [CrossRef]
- Shang, X.; Fu, X.; Zhang, P.; Sheng, M.; Song, J.; He, F.; Qiu, Y.; Wu, H.; Lu, Q.; Feng, Y. An outbreak of norovirus-associated acute gastroenteritis associated with contaminated barrelled water in many schools in Zhejiang, China. PLoS ONE 2017, 12, e0171307. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.L.; Zhen, S.S.; Wang, J.X.; Zhang, C.J.; Qiu, C.; Wang, S.M.; Jiang, X.; Wang, X.Y. Burden of acute gastroenteritis caused by norovirus in China: A systematic review. J. Infect. 2017, 75, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, L.; Lai, S.; Li, Z.; Sun, Q.; Zhang, P. Surveillance and early warning systems of infectious disease in China: From 2012 to 2014. Int. J. Health Plan. Manag. 2017, 32, 329–338. [Google Scholar] [CrossRef]
- National Public Health Emergency Event Information Report and Management Regulations 2005. Available online: http://61.49.18.65/uploadfile/200629113745905.doc (accessed on 12 March 2019).
- Huang, X.Y.; Su, J.; Lu, Q.C.; Li, S.Z.; Zhao, J.Y.; Li, M.L.; Li, Y.; Shen, X.J.; Zhang, B.F.; Wang, H.F. A large outbreak of acute gastroenteritis caused by the human norovirus GII.17 strain at a university in Henan Province, China. Infect. Dis. Poverty 2017, 6, 6. [Google Scholar] [CrossRef]
- Wang, X.; Yong, W.; Shi, L.; Qiao, M.; He, M.; Zhang, H.; Guo, B.; Xie, G.; Zhang, M.; Jin, M. An outbreak of multiple norovirus strains on a cruise ship in China, 2014. J. Appl. Microbiol. 2016, 120, 226–233. [Google Scholar] [CrossRef]
- Li, J.S.; Meng, Q.; Dong, X.G.; Yang, J.Y.; Yang, X.X.; Wei, X.X.; Wang, Z.E.; Feng, H.R.; Wu, Q.R.; Li, R.X. Norovirus outbreaks in Fengtai District, Beijing, China, 2014. Arch. Virol. 2016, 161, 2855–2858. [Google Scholar] [CrossRef]
- Alfano-Sobsey, E.; Sweat, D.; Hall, A.; Breedlove, F.; Rodriguez, R.; Greene, S.; Pierce, A.; Sobsey, M.; Davies, M.; Ledford, S.L. Norovirus outbreak associated with undercooked oysters and secondary household transmission. Epidemiol. Infect. 2012, 140, 276–282. [Google Scholar] [CrossRef]
- Liao, Q.H.; Ran, L.; Jin, M.; Cui, S.H.; Yuan, J.; Ma, H.L.; Ban, H.Q.; Sun, L.M.; Luo, L.; Liu, N.; et al. Guidelines on outbreak investigation, prevention and control of norovirus infection (2015). Zhonghua Yu Fang Yi Xue Za Zhi 2016, 50, 7–16. [Google Scholar]
- Lopman, B.A.; Reacher, M.H.; Yvonne, V.D.; François-Xavier, H.; David, B.; Marion, K. Viral gastroenteritis outbreaks in Europe, 1995–2000. Emerg. Infect. Dis. 2003, 9, 90–96. [Google Scholar] [CrossRef]
- Rohayem, J. Norovirus seasonality and the potential impact of climate change. Clin. Microbiol. Infect. 2009, 15, 524–527. [Google Scholar] [CrossRef]
- Greer, A.L.; Drews, S.J.; Fisman, D.N. Why “Winter” Vomiting Disease? Seasonality, Hydrology, and Norovirus Epidemiology in Toronto, Canada. Ecohealth 2009, 6, 192–199. [Google Scholar] [CrossRef]
- Lin, Q.; Wang, A.; Zhong, X.; Liang, J.; Zhuang, Y.; Chen, R.; Li, B.; Huang, Q.; Zhang, Y. Norovirus infection surveillance in early warning of outbreaks in population in oyster farming areas. J. Hyg. Res. 2017, 46, 201–212. [Google Scholar]
- Kumazaki, M.; Usuku, S. Norovirus genotype distribution in outbreaks of acute gastroenteritis among children and older people: An 8-year study. BMC Infect. Dis. 2016, 16, 643. [Google Scholar] [CrossRef]
- Wu, F.T.; Chen, H.C.; Yen, C.; Wu, C.Y.; Katayama, K.; Park, Y.; Hall, A.J.; Vinjé, J.; Huang, J.C.; Wu, H.S. Epidemiology and molecular characteristics of norovirus GII.4 Sydney outbreaks in Taiwan, January 2012–December 2013. J. Med. Virol. 2015, 87, 1462–1470. [Google Scholar] [CrossRef]
- Wheatley, A.D. High rate and changing molecular epidemiology pattern of norovirus infections in sporadic cases and outbreaks of gastroenteritis in Hong Kong. J. Med. Virol. 2010, 73, 113–117. [Google Scholar]
- Everardo, V.; Barclay, L.; Gregoricus, N.; Shirley, S.H.; Lee, D.; Vinjé, J. Genotypic and epidemiologic trends of norovirus outbreaks in the United States, 2009 to 2013. J. Clin. Microbiol. 2014, 52, 147–155. [Google Scholar]
- Friesema, I.H.M.; Vennema, H.; Heijne, J.C.M.; de Jager, C.M.; Morroy, G.; van den Kerkhof, J.H.; de Coster, E.J.; Wolters, B.A.; ter Waarbeek, H.L.; Fanoy, E.B.; et al. Norovirus outbreaks in nursing homes: The evaluation of infection control measures. Epidemiol. Infect. 2009, 137, 1722–1733. [Google Scholar] [CrossRef]
- Barret, A.S.; Silva, J.D.; Ambert-Balay, K.; Delmas, G.; Bone, A.; Thiolet, J.M.; Vaillant, V. Surveillance for outbreaks of gastroenteritis in elderly long-term care facilities in France, November 2010 to May 2012. Eurosurveillance 2014, 19, 20859. [Google Scholar] [CrossRef]
- Li, J.; Gao, X.; Ye, Y.L.; Wan, T.; Zang, H.; Mo, P.H.; Song, C.L. An acute gastroenteritis outbreak associated with person-to-person transmission in a primary school in Shanghai: First report of a GI.5 norovirus outbreak in China. BMC Infect. Dis. 2018, 18, 316. [Google Scholar] [CrossRef]
- Shen, J.C.; Lin, J.F.; Gao, J.; Yao, W.T.; Wen, D.; Liu, G.T.; Han, J.K.; Ma, H.L.; Zhang, L.J.; Zhu, B.P. A norovirus-borne outbreak caused by contaminated bottled spring water in a school, Zhejiang province. Zhonghua Liu Xing Bing Xue Za Zhi 2011, 32, 800–803. [Google Scholar]
- Criterion for Reporting Infectious Diseases in Schools and Child-Care Institutions (for Trial Implementation). Available online: http://www.nhc.gov.cn/zhjcj/s5853/201304/5765acfb22604d91aaf167bfc1247fbd.shtml (accessed on 12 March 2019).
- Ao, Y.; Wang, J.; Ling, H.; He, Y.; Dong, X.; Wang, X.; Peng, J.; Zhang, H.; Jin, M.; Duan, Z. Norovirus GII.P16/GII.2-Associated Gastroenteritis, China, 2016. Emerg. Infect. Dis. 2017, 23, 1172–1175. [Google Scholar] [CrossRef]
- Ao, Y.; Cong, X.; Jin, M.; Sun, X.; Wei, X.; Wang, J.; Zhang, Q.; Song, J.; Yu, J.; Cui, J. Genetic Analysis of Re-Emerging GII.P16-GII.2 Noroviruses in 2016–2017 in China. J. Infect. Dis. 2018, 218, 133–143. [Google Scholar] [CrossRef]
- Matsushima, Y.; Ishikawa, M.; Shimizu, T.; Komane, A.; Kasuo, S.; Shinohara, M.; Nagasawa, K.; Kimura, H.; Ryo, A.; Okabe, N. Genetic analyses of GII.17 norovirus strains in diarrheal disease outbreaks from December 2014 to March 2015 in Japan reveal a novel polymerase sequence and amino acid substitutions in the capsid region. Eurosurveillance 2015, 26, 21173. [Google Scholar] [CrossRef]
- Lee, C.C.; Feng, Y.; Chen, S.Y.; Tsai, C.N.; Lai, M.W.; Chiu, C.H. Emerging norovirus GII.17 in Taiwan. Clin. Infect. Dis. 2015, 61, 1762–1764. [Google Scholar]
- Jung, S.; Hwang, B.M.; Jung, H.; Chung, G.; Yoo, C.K.; Lee, D.Y. Emergence of Norovirus GII.17-associated Outbreak and Sporadic Cases in Korea from 2014 to 2015. Osong Public Health Res. Perspect. 2017, 8, 86–90. [Google Scholar] [CrossRef]
- Jin, M.; Zhou, Y.K.; Xie, H.P.; Fu, J.G.; He, Y.Q.; Zhang, S.; Jing, H.B.; Kong, X.Y.; Sun, X.M.; Li, H.Y. Characterization of the new GII.17 variant that emerged recently as the predominant strain in China. J. Gen. Virol. 2016, 97, 2620–2632. [Google Scholar] [CrossRef]
- Niendorf, S.; Jacobsen, S.; Faber, M.; Eishübinger, A.M.; Hofmann, J.; Zimmermann, O.; Höhne, M.; Bock, C.T. Steep rise in norovirus cases and emergence of a new recombinant strain GII.P16-GII.2, Germany, winter 2016. Eurosurveillance 2017, 22, 30447. [Google Scholar] [CrossRef]
- Liu, L.T.; Kuo, T.Y.; Wu, C.Y.; Liao, W.T.; Hall, A.J.; Wu, F.T. Recombinant GII.P16-GII.2 Norovirus, Taiwan, 2016. Emerg. Infect. Dis. 2017, 23, 1180–1183. [Google Scholar] [CrossRef]
- Nagasawa, K.; Matsushima, Y.; Motoya, T.; Mizukoshi, F.; Ueki, Y.; Sakon, N.; Murakami, K.; Shimizu, T.; Okabe, N.; Nagata, N. Phylogeny and Immunoreactivity of Norovirus GII.P16-GII.2, Japan, Winter 2016–17. Emerg. Infect. Dis. 2018, 24, 144–148. [Google Scholar] [CrossRef]
- Kwok, K.; Niendorf, S.; Lee, N.; Hung, T.N.; Chan, L.Y.; Jacobsen, S.; Nelson, E.A.S.; Leung, T.F.; Lai, R.W.M.; Chan, P.K.S. Increased Detection of Emergent Recombinant Norovirus GII.P16-GII.2 Strains in Young Adults, Hong Kong, China, 2016–2017. Emerg. Infect. Dis. 2017, 23, 1852–1855. [Google Scholar] [CrossRef]
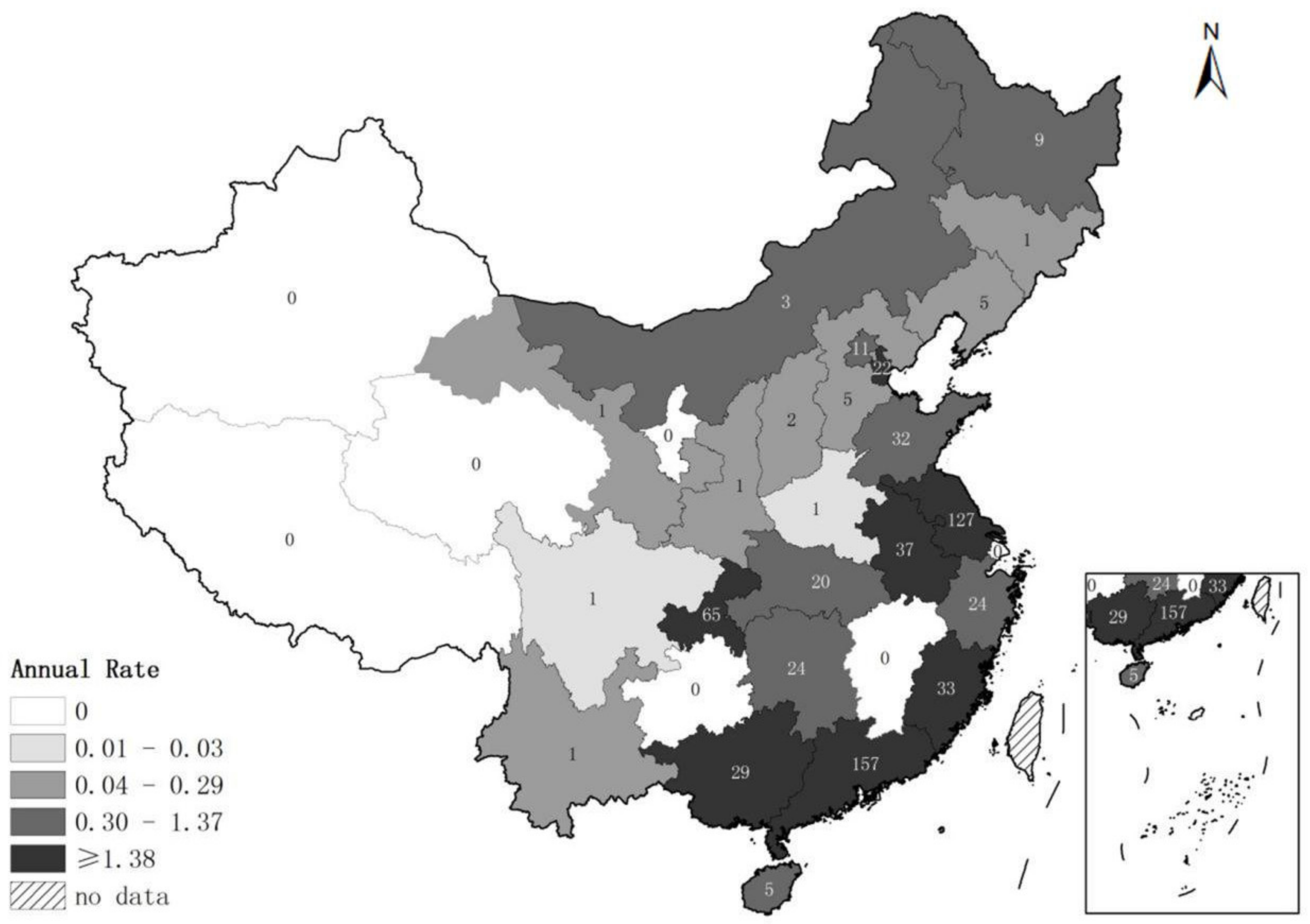
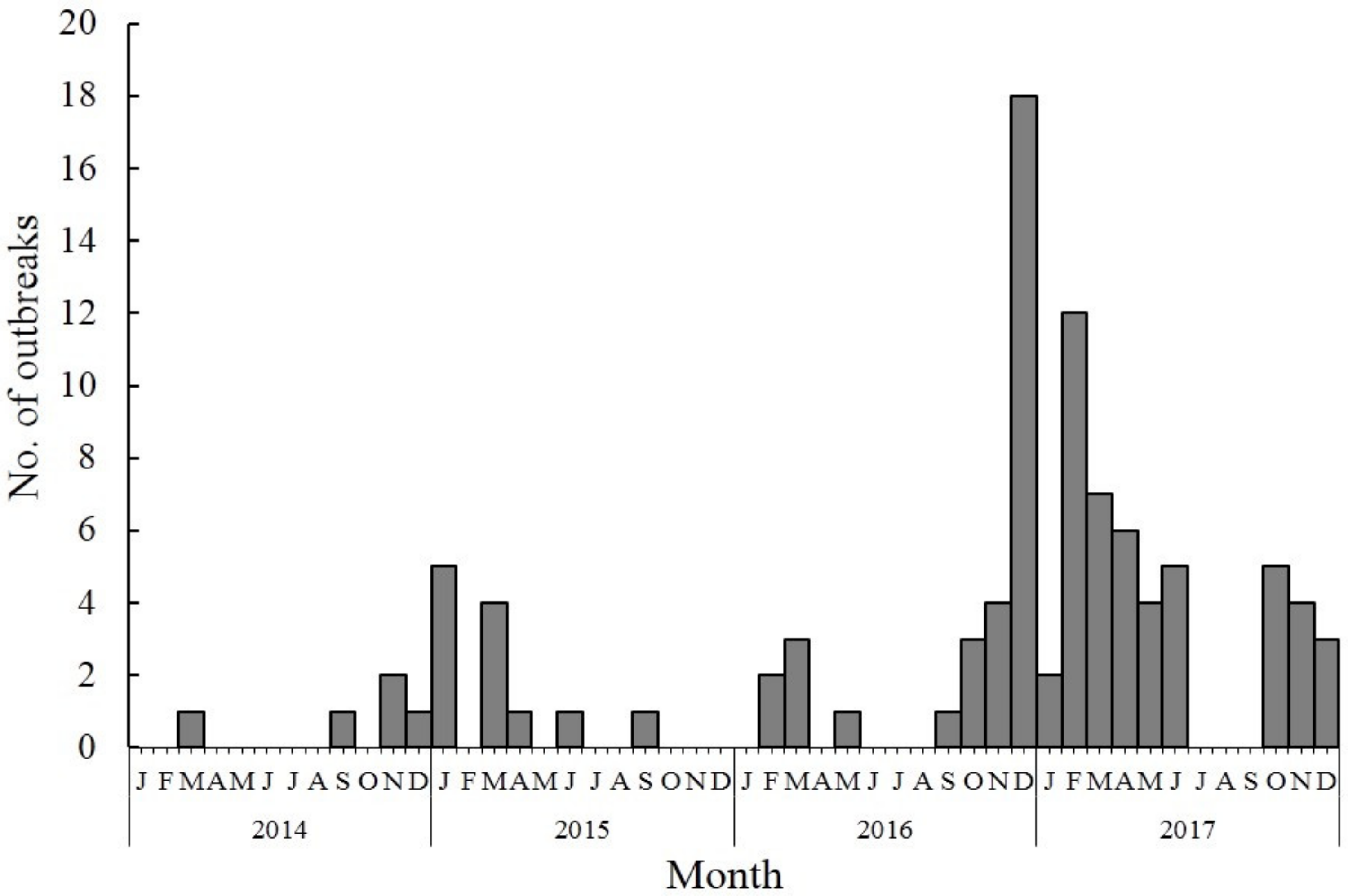
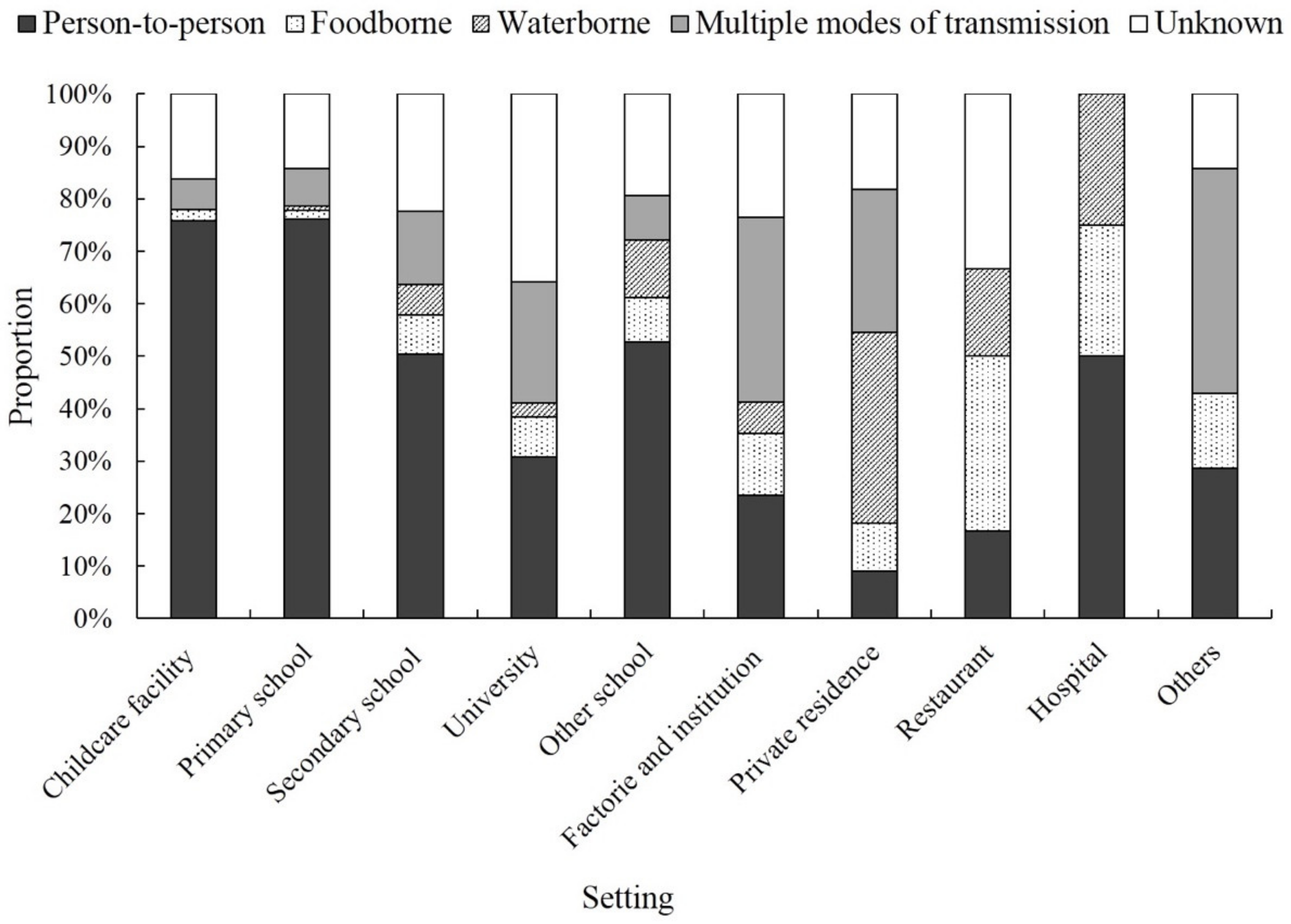
| Etiology | No. of Outbreaks (%) | Total No. of Cases | Total No. of Persons Exposed | Attack Rate (%), Median (IQR) | Total No. of Deaths |
|---|---|---|---|---|---|
| Single etiology outbreaks | 635 (91.8) | 31,313 | 1,331,092 | 3.7 (1.8, 8.0) | 2 |
| Norovirus | 611 (88.3) | 30,249 | 1,245,725 | 3.7 (1.8, 8.0) | 0 |
| Non-norovirus 1 | 24 (3.5) | 1064 | 85,367 | 3.7 (1.7, 8.8) | 2 |
| Multiple etiology outbreaks | 8 (1.1) | 993 | 13,905 | 10.4 (5.0, 17.6) | 0 |
| Norovirus and other etiologies 2 | 5 (0.7) | 599 | 9718 | 8.6 (6.1, 17.5) | 0 |
| Etiologies other than norovirus 3 | 3 (0.4) | 394 | 4187 | 12.3 (6.9, 15.7) | 0 |
| Not identified | 49 (7.1) | 1493 | 47,608 | 5.1 (1.9, 10.1) | 1 |
| Total | 692 | 33,799 | 1,392,605 | 3.8 (1.8, 8.3) | 3 |
| Year | Total No. of Outbreaks | Total No. of Cases | Total No. of Deaths | No. of Reported Provinces | Outbreak Size (cases), Median (IQR) | Outbreak Duration (days), Median (IQR) 1 |
|---|---|---|---|---|---|---|
| 2014 | 58 | 4672 | 0 | 10 | 48 (26, 91) | 5.9 (2.8, 8.1) |
| 2015 | 100 | 5861 | 0 | 14 | 39 (23, 63) | 5.8 (2.6, 8.2) |
| 2016 | 135 | 5253 | 0 | 14 | 31 (21, 49) | 4.0 (2.0, 8.6) |
| 2017 | 323 | 15,062 | 0 | 21 | 34 (24, 58) | 4.8 (2.3, 8.8) |
| Total | 616 | 30,848 | 0 | 24 | 34 (23, 60) | 4.7 (2.3, 8.5) |
| Outbreak Characteristic | No. of Total Outbreaks | No. of Total Cases | Attack Rate (%), Median (IQR) | Outbreak Size (cases), Median (IQR) 1 | Outbreak Duration (days), Median (IQR) 2 |
|---|---|---|---|---|---|
| Transmission Mode | |||||
| Person-to-person | 387 (62.8) | 16,884 | 3.6 (1.9, 7.5) | 31 (22, 52) | 5.3 (2.6, 8.9) |
| Foodborne | 29 (4.7) | 1947 | 7.8 (2.2, 18.9) | 53 (38, 86) | 3.4 (1.3, 5.4) |
| Waterborne | 21 (3.4) | 1616 | 3.6 (1.2, 5.9) | 47 (31, 107) | 6.9 (3.6, 9.2) |
| Multiple | 66 (10.7) | 4665 | 4.9 (1.4, 9.5) | 48 (26, 84) | 4.5 (2, 8.1) |
| Unknown | 113 (18.3) | 5736 | 3.3 (1.6, 8.3) | 38 (24, 67) | 3.4 (2, 7.3) |
| Exposure Setting | |||||
| Childcare facility | 136 (22.1) | 3639 | 8.4 (5.2, 16.2) | 24 (17, 32) | 2.3 (1.2, 4.7) |
| Primary school | 239 (38.8) | 10,353 | 2.9 (1.7, 5.7) | 34 (23, 54) | 5 (2.4, 8.2) |
| Secondary school | 121 (19.6) | 7586 | 2.7 (1.5, 5.6) | 52 (34, 74) | 7.4 (4.2, 12.8) |
| University | 39 (6.3) | 5081 | 1.1 (0.6, 2.1) | 79 (57, 139) | 10.4 (7.3, 16.3) |
| Other school 3 | 36 (5.8) | 1819 | 2.8 (1.3, 5.3) | 36 (25, 61) | 3.3 (2.6, 9.5) |
| Factory and institute | 17 (2.8) | 799 | 7.8 (4.2, 15.9) | 34 (29, 71) | 2.7 (1.1, 7.6) |
| Restaurant | 6 (1.0) | 524 | 24.2 (7.5, 40.7) | 70 (29, 164) | - |
| Private residence | 11 (1.8) | 456 | 3.2 (0.7, 8.0) | 37 (27, 42) | 6.6 (2.4, 9.6) |
| Hospital | 4 (0.6) | 297 | 14.8 (6.4, 46.4) | 55 (27, 142) | - |
| Others 4 | 7 (1.1) | 294 | 10.0 (2.2, 18.2) | 28 (20, 67) | 2.6 (1.8, 4.7) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lian, Y.; Wu, S.; Luo, L.; Lv, B.; Liao, Q.; Li, Z.; Rainey, J.J.; Hall, A.J.; Ran, L. Epidemiology of Norovirus Outbreaks Reported to the Public Health Emergency Event Surveillance System, China, 2014–2017. Viruses 2019, 11, 342. https://doi.org/10.3390/v11040342
Lian Y, Wu S, Luo L, Lv B, Liao Q, Li Z, Rainey JJ, Hall AJ, Ran L. Epidemiology of Norovirus Outbreaks Reported to the Public Health Emergency Event Surveillance System, China, 2014–2017. Viruses. 2019; 11(4):342. https://doi.org/10.3390/v11040342
Chicago/Turabian StyleLian, Yiyao, Shuyu Wu, Li Luo, Bin Lv, Qiaohong Liao, Zhongjie Li, Jeanette J. Rainey, Aron J. Hall, and Lu Ran. 2019. "Epidemiology of Norovirus Outbreaks Reported to the Public Health Emergency Event Surveillance System, China, 2014–2017" Viruses 11, no. 4: 342. https://doi.org/10.3390/v11040342
APA StyleLian, Y., Wu, S., Luo, L., Lv, B., Liao, Q., Li, Z., Rainey, J. J., Hall, A. J., & Ran, L. (2019). Epidemiology of Norovirus Outbreaks Reported to the Public Health Emergency Event Surveillance System, China, 2014–2017. Viruses, 11(4), 342. https://doi.org/10.3390/v11040342





