Final Consumer Options to Control and Prevent Foodborne Norovirus Infections
Abstract
1. Introduction
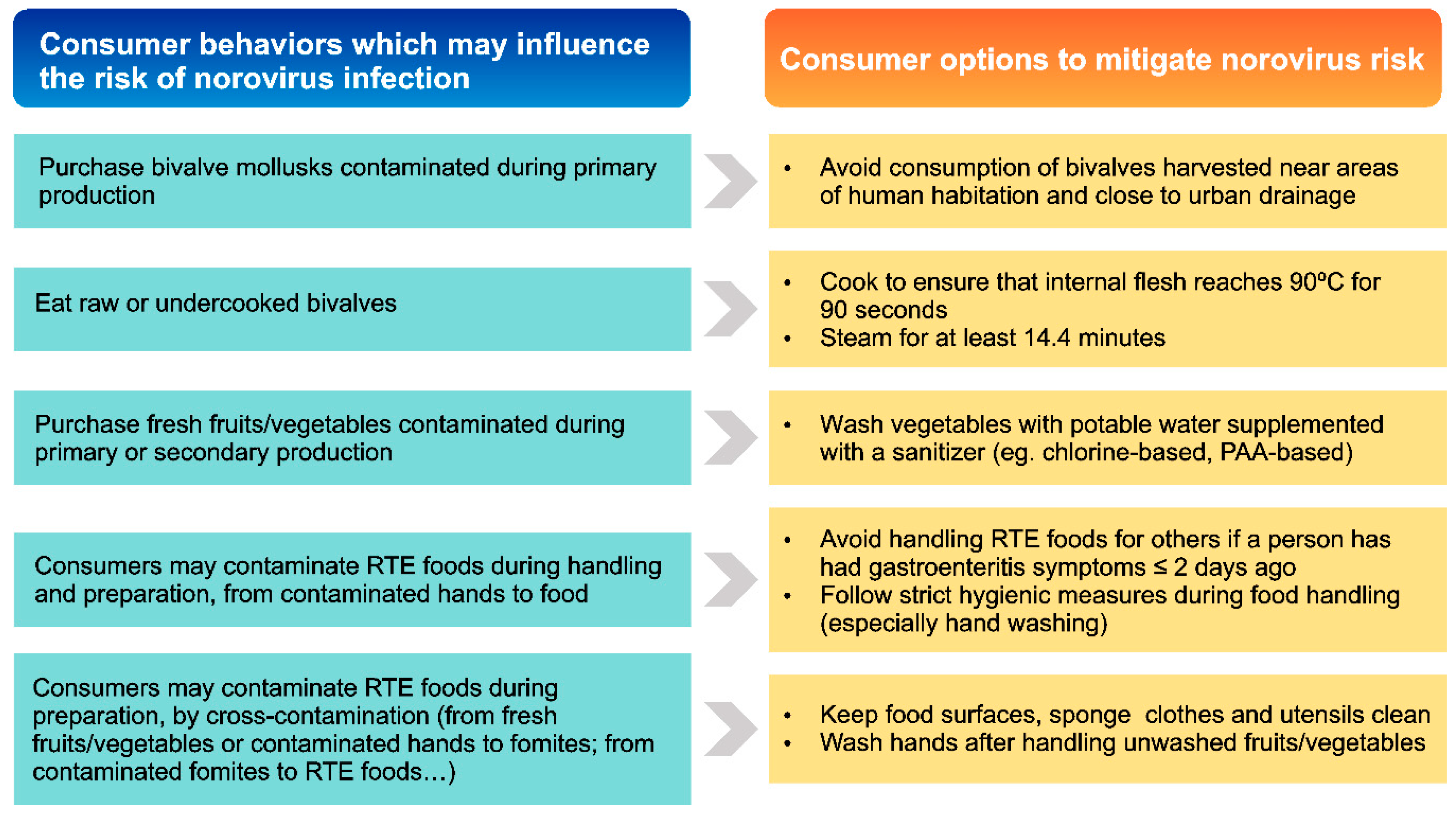
2. Food Choice: Is It Possible to Prevent NoV by Selecting Products with Low Risk?
3. Survival of NoV on Fruits and Vegetables and Elimination during Washing and Sanitation
4. Safe Cooking Methods for Bivalve Mollusks to Ensure NoV Inactivation
5. Spread of NoV by Infected Food Handlers and Lack of Personal Hygiene
5.1. NoV Asymptomatic Carriers
5.2. Hand Hygiene and Disinfection
5.3. Environmental and Fomite Contamination
5.4. Food Contamination by Contact with Fomites and Handling
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vinje, J. Advances in laboratory methods for detection and typing of norovirus. J. Clin. Microbiol. 2015, 53, 373–381. [Google Scholar] [CrossRef]
- Robilotti, E.; Deresinski, S.; Pinsky, B.A. Norovirus. Clin. Microbiol. Rev. 2015, 28, 134–164. [Google Scholar] [PubMed]
- Lopman, B.A.; Steele, D.; Kirkwood, C.D.; Parashar, U.D. The vast and varied global burden of norovirus: Prospects for prevention and control. PLoS Med. 2016, 13, e1001999. [Google Scholar] [CrossRef] [PubMed]
- Havelaar, A.H.; Kirk, M.D.; Torgerson, P.R.; Gibb, H.J.; Hald, T.; Lake, R.J.; Praet, N.; Bellinger, D.C.; de Silva, N.R.; Gargouri, N.; et al. World health organization global estimates and regional comparisons of the burden of foodborne disease in 2010. PLoS Med. 2015, 12, e1001923. [Google Scholar] [CrossRef]
- Belliot, G.; Lopman, B.A.; Ambert-Balay, K.; Pothier, P. The burden of norovirus gastroenteritis: An important foodborne and healthcare-related infection. Clin. Microbiol. Infect. 2014, 20, 724–730. [Google Scholar] [CrossRef] [PubMed]
- Havelaar, A.H.; Galindo, A.V.; Kurowicka, D.; Cooke, R.M. Attribution of foodborne pathogens using structured expert elicitation. Foodborne Pathog. Dis. 2008, 5, 649–659. [Google Scholar] [CrossRef]
- ECDC. The european union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2015. EFSA J. 2016, 14, 4634. [Google Scholar]
- ECDC. The european union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2016. EFSA J. 2017, 15, 5077. [Google Scholar]
- ECDC. The european union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2017. EFSA J. 2018, 16, 5500. [Google Scholar]
- Bernard, H.; Faber, M.; Wilking, H.; Haller, S.; Hohle, M.; Schielke, A.; Ducomble, T.; Siffczyk, C.; Merbecks, S.S.; Fricke, G.; et al. Large multistate outbreak of norovirus gastroenteritis associated with frozen strawberries, germany, 2012. Euro Surveill. 2014, 19, 20719. [Google Scholar] [CrossRef]
- Blanco, A.; Guix, S.; Fuster, N.; Fuentes, C.; Bartolome, R.; Cornejo, T.; Pinto, R.M.; Bosch, A. Norovirus in bottled water associated with gastroenteritis outbreak, spain, 2016. Emerg. Infect. Dis. 2017, 23, 1531–1534. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on an update on the present knowledge on the occurrence and control of foodborne viruses. EFSA J. 2011, 9, 2190. [Google Scholar]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on norovirus (nov) in oysters: Methods, limits and control options. EFSA J. 2012, 10, 2500. [Google Scholar]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on the risk posed by pathogens in food of non-animal origin. Part 2 (salmonella and norovirus in berries). EFSA J. 2014, 12, 3706. [Google Scholar]
- EFSA Panel on Biological Hazards (BIOHAZ). Evaluation of heat treatments, different from those currently established in the eu legislation, that could be applied to live bivalve molluscs from b and c production areas, that have not been submitted to purification or relaying, in order to eliminate pathogenic microorganisms. EFSA J. 2015, 13, 4332. [Google Scholar]
- RASFF. The Rapid Alert System for Food and Feed 2017 Annual Report. Available online: https://ec.europa.eu/food/sites/food/files/safety/docs/rasff_annual_report_2017.pdf (accessed on 8 April 2019).
- Bosch, A.; Gkogka, E.; Le Guyader, F.S.; Loisy-Hamon, F.; Lee, A.; van Lieshout, L.; Marthi, B.; Myrmel, M.; Sansom, A.; Schultz, A.C.; et al. Foodborne viruses: Detection, risk assessment, and control options in food processing. Int. J. Food Microbiol. 2018, 285, 110–128. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, G.T.; Pu, J.; Miura, T.; Ito, H.; Kazama, S.; Konta, Y.; Van Le, A.; Watanabe, T. Oyster contamination with human noroviruses impacted by urban drainage and seasonal flooding in vietnam. Food Environ. Virol. 2018, 10, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, J.; Treguier, C.; Piquet, J.C.; Gachelin, S.; Cochennec-Laureau, N.; Le Saux, J.C.; Garry, P.; Le Guyader, F.S. Improving the efficacy of sewage treatment decreases norovirus contamination in oysters. Int. J. Food Microbiol. 2018, 286, 1–5. [Google Scholar] [CrossRef]
- Campos, C.J.A.; Kershaw, S.; Morgan, O.C.; Lees, D.N. Risk factors for norovirus contamination of shellfish water catchments in england and wales. Int. J. Food Microbiol. 2017, 241, 318–324. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on the risk posed by pathogens in food of non-animal origin. Part 2 (salmonella and norovirus in leafy greens eaten raw as salads). EFSA J. 2014, 12, 3600. [Google Scholar]
- Codex, A. Guidelines on the Application of General Principles of Food Hygiene to the Control of Viruses in Food. Available online: http://www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCAC%2BGL%2B79-2012%252FCXG_079e.pdf (accessed on 8 April 2019).
- Cates, S.C.; Kosa, K.M.; Brophy, J.E.; Hall, A.J.; Fraser, A. Consumer education needed on norovirus prevention and control: Findings from a nationally representative survey of U.S. Adults. J. Food Prot. 2015, 78, 484–490. [Google Scholar] [CrossRef]
- ISO15216–1:2017. Microbiology of the Food Chain—Horizontal Method for Determination of Hepatitis a Virus and Norovirus Using Real-Time rt-PCR—Part 1: Method for Quantification; ISO (International Organization for Standardization): Geneva, Switzerland, 2017. [Google Scholar]
- Lowther, J.A.; Gustar, N.E.; Hartnell, R.E.; Lees, D.N. Comparison of norovirus rna levels in outbreak-related oysters with background environmental levels. J. Food. Prot. 2012, 75, 389–393. [Google Scholar] [CrossRef]
- Woods, J.W.; Calci, K.R.; Marchant-Tambone, J.G.; Burkhardt, W. Detection and molecular characterization of norovirus from oysters implicated in outbreaks in the US. Food Microbiol. 2016, 59, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, J.; Le Saux, J.C.; Lora, M.; Atmar, R.L.; Le Guyader, F.S. Norovirus contamination on french marketed oysters. Int. J. Food Microbiol. 2013, 166, 244–248. [Google Scholar] [CrossRef]
- Lowther, J.A.; Henshilwood, K.; Lees, D.N. Determination of norovirus contamination in oysters from two commercial harvesting areas over an extended period, using semiquantitative real-time reverse transcription pcr. J. Food Prot. 2008, 71, 1427–1433. [Google Scholar] [CrossRef]
- Lowther, J.A.; Gustar, N.E.; Powell, A.L.; Hartnell, R.E.; Lees, D.N. Two-year systematic study to assess norovirus contamination in oysters from commercial harvesting areas in the united kingdom. Appl. Environ. Microbiol. 2012, 78, 5812–5817. [Google Scholar] [CrossRef]
- Lowther, J.A.; Gustar, N.E.; Powell, A.L.; O’Brien, S.; Lees, D.N. A one-year survey of norovirus in uk oysters collected at the point of sale. Food Environ. Virol. 2018, 10, 278–287. [Google Scholar] [CrossRef]
- Ilic, N.; Velebit, B.; Teodorovic, V.; Djordjevic, V.; Karabasil, N.; Vasilev, D.; Djuric, S.; Adzic, B.; Dimitrijevic, M. Influence of environmental conditions on norovirus presence in mussels harvested in montenegro. Food Environ. Virol. 2017, 9, 406–414. [Google Scholar] [CrossRef]
- Suffredini, E.; Corrain, C.; Arcangeli, G.; Fasolato, L.; Manfrin, A.; Rossetti, E.; Biazzi, E.; Mioni, R.; Pavoni, E.; Losio, M.N.; et al. Occurrence of enteric viruses in shellfish and relation to climatic-environmental factors. Lett. Appl. Microbiol. 2008, 47, 467–474. [Google Scholar] [CrossRef]
- Pepe, T.; Ventrone, I.; Suffredini, E.; Ceruso, M.; Croci, L.; Anastasio, A.; Cortesi, M.L. Norovirus monitoring in bivalve molluscs harvested and commercialized in southern italy. J. Food Prot. 2012, 75, 976–981. [Google Scholar] [CrossRef]
- Manso, C.F.; Romalde, J.L. Detection and characterization of hepatitis a virus and norovirus in mussels from galicia (nw spain). Food Environ. Virol. 2013, 5, 110–118. [Google Scholar] [CrossRef]
- Polo, D.; Varela, M.F.; Romalde, J.L. Detection and quantification of hepatitis a virus and norovirus in spanish authorized shellfish harvesting areas. Int. J. Food Microbiol. 2015, 193, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Losio, M.N.; Pavoni, E.; Bilei, S.; Bertasi, B.; Bove, D.; Capuano, F.; Farneti, S.; Blasi, G.; Comin, D.; Cardamone, C.; et al. Microbiological survey of raw and ready-to-eat leafy green vegetables marketed in italy. Int. J. Food Microbiol. 2015, 210, 88–91. [Google Scholar] [CrossRef]
- Terio, V.; Bottaro, M.; Pavoni, E.; Losio, M.N.; Serraino, A.; Giacometti, F.; Martella, V.; Mottola, A.; Di Pinto, A.; Tantillo, G. Occurrence of hepatitis a and e and norovirus gi and gii in ready-to-eat vegetables in italy. Int. J. Food Microbiol. 2017, 249, 61–65. [Google Scholar] [CrossRef]
- Kokkinos, P.; Kozyra, I.; Lazic, S.; Bouwknegt, M.; Rutjes, S.; Willems, K.; Moloney, R.; de Roda Husman, A.M.; Kaupke, A.; Legaki, E.; et al. Harmonised investigation of the occurrence of human enteric viruses in the leafy green vegetable supply chain in three european countries. Food Environ. Virol. 2012, 4, 179–191. [Google Scholar] [CrossRef]
- Purpari, G.; Macaluso, G.; Di Bella, S.; Gucciardi, F.; Mira, F.; Di Marco, P.; Lastra, A.; Petersen, E.; La Rosa, G.; Guercio, A. Molecular characterization of human enteric viruses in food, water samples, and surface swabs in sicily. Int. J. Infect. Dis. 2019, 80, 66–72. [Google Scholar] [CrossRef]
- Baert, L.; Mattison, K.; Loisy-Hamon, F.; Harlow, J.; Martyres, A.; Lebeau, B.; Stals, A.; Van Coillie, E.; Herman, L.; Uyttendaele, M. Review: Norovirus prevalence in belgian, canadian and french fresh produce: A threat to human health? Int. J. Food Microbiol. 2011, 151, 261–269. [Google Scholar] [CrossRef]
- Mattison, K.; Harlow, J.; Morton, V.; Cook, A.; Pollari, F.; Bidawid, S.; Farber, J.M. Enteric viruses in ready-to-eat packaged leafy greens. Emerg. Infect. Dis. 2010, 16, 1815–1817. [Google Scholar] [CrossRef] [PubMed]
- Loutreul, J.; Cazeaux, C.; Levert, D.; Nicolas, A.; Vautier, S.; Le Sauvage, A.L.; Perelle, S.; Morin, T. Prevalence of human noroviruses in frozen marketed shellfish, red fruits and fresh vegetables. Food Environ. Virol. 2014, 6, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Callejon, R.M.; Rodriguez-Naranjo, M.I.; Ubeda, C.; Hornedo-Ortega, R.; Garcia-Parrilla, M.C.; Troncoso, A.M. Reported foodborne outbreaks due to fresh produce in the united states and european union: Trends and causes. Foodborne Pathog. Dis. 2015, 12, 32–38. [Google Scholar] [CrossRef]
- Li, D.; Stals, A.; Tang, Q.J.; Uyttendaele, M. Detection of noroviruses in shellfish and semiprocessed fishery products from a belgian seafood company. J. Food Prot. 2014, 77, 1342–1347. [Google Scholar] [CrossRef]
- Flannery, J.; Keaveney, S.; Dore, W. Use of frna bacteriophages to indicate the risk of norovirus contamination in irish oysters. J. Food Prot. 2009, 72, 2358–2362. [Google Scholar] [CrossRef]
- Suffredini, E.; Magnabosco, C.; Civettini, M.; Rossetti, E.; Arcangeli, G.; Croci, L. Norovirus contamination in different shellfish species harvested in the same production areas. J. Appl. Microbiol. 2012, 113, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Suffredini, E.; Lanni, L.; Arcangeli, G.; Pepe, T.; Mazzette, R.; Ciccaglioni, G.; Croci, L. Qualitative and quantitative assessment of viral contamination in bivalve molluscs harvested in italy. Int. J. Food Microbiol. 2014, 184, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Fusco, G.; Aprea, G.; Galiero, G.; Guarino, A.; Viscardi, M. Escherichia coli, salmonella spp., hepatitis a virus and norovirus in bivalve molluscs in southern italy. Vet. Ital. 2013, 49, 55–58. [Google Scholar] [PubMed]
- Pavoni, E.; Consoli, M.; Suffredini, E.; Arcangeli, G.; Serracca, L.; Battistini, R.; Rossini, I.; Croci, L.; Losio, M.N. Noroviruses in seafood: A 9-year monitoring in italy. Foodborne Pathog. Dis. 2013, 10, 533–539. [Google Scholar] [CrossRef]
- Fusco, G.; Di Bartolo, I.; Cioffi, B.; Ianiro, G.; Palermo, P.; Monini, M.; Amoroso, M.G. Prevalence of foodborne viruses in mussels in southern italy. Food Environ. Virol. 2017, 9, 187–194. [Google Scholar] [CrossRef] [PubMed]
- La Bella, G.; Martella, V.; Basanisi, M.G.; Nobili, G.; Terio, V.; La Salandra, G. Food-borne viruses in shellfish: Investigation on norovirus and hav presence in apulia (se italy). Food Environ. Virol. 2017, 9, 179–186. [Google Scholar] [CrossRef]
- Vilarino, M.L.; Le Guyader, F.S.; Polo, D.; Schaeffer, J.; Krol, J.; Romalde, J.L. Assessment of human enteric viruses in cultured and wild bivalve molluscs. Int. Microbiol. 2009, 12, 145–151. [Google Scholar]
- Brake, F.; Ross, T.; Holds, G.; Kiermeier, A.; McLeod, C. A survey of australian oysters for the presence of human noroviruses. Food Microbiol. 2014, 44, 264–270. [Google Scholar] [CrossRef]
- Torok, V.; Hodgson, K.; McLeod, C.; Tan, J.; Malhi, N.; Turnbull, A. National survey of foodborne viruses in australian oysters at production. Food Microbiol. 2018, 69, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.P.; Zhao, F.; Yao, L.; Li, X.G.; Zhou, D.Q.; Zhang, R.L. The presence of genogroup ii norovirus in retail shellfish from seven coastal cities in china. Food Environ. Virol. 2013, 5, 81–86. [Google Scholar] [CrossRef]
- Nishida, T.; Kimura, H.; Saitoh, M.; Shinohara, M.; Kato, M.; Fukuda, S.; Munemura, T.; Mikami, T.; Kawamoto, A.; Akiyama, M.; et al. Detection, quantitation, and phylogenetic analysis of noroviruses in japanese oysters. Appl. Environ. Microbiol. 2003, 69, 5782–5786. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Benabbes, L.; Ollivier, J.; Schaeffer, J.; Parnaudeau, S.; Rhaissi, H.; Nourlil, J.; Le Guyader, F.S. Norovirus and other human enteric viruses in moroccan shellfish. Food Environ. Virol. 2013, 5, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Montazeri, N.; Maite, M.; Liu, D.; Cormier, J.; Landry, M.; Shackleford, J.; Lampila, L.E.; Achberger, E.C.; Janes, M.E. Surveillance of enteric viruses and microbial indicators in the eastern oysters (crassostrea virginica) and harvest waters along louisiana gulf coast. J. Food Sci. 2015, 80, M1075–M1082. [Google Scholar] [CrossRef]
- DePaola, A.; Jones, J.L.; Woods, J.; Burkhardt, W., 3rd; Calci, K.R.; Krantz, J.A.; Bowers, J.C.; Kasturi, K.; Byars, R.H.; Jacobs, E.; et al. Bacterial and viral pathogens in live oysters: 2007 united states market survey. Appl. Environ. Microbiol. 2010, 76, 2754–2768. [Google Scholar] [CrossRef] [PubMed]
- Marti, E.; Ferrary-Americo, M.; Barardi, C.R.M. Detection of potential infectious enteric viruses in fresh produce by (rt)-qpcr preceded by nuclease treatment. Food Environ. Virol. 2017, 9, 444–452. [Google Scholar] [CrossRef]
- El-Senousy, W.M.; Costafreda, M.I.; Pinto, R.M.; Bosch, A. Method validation for norovirus detection in naturally contaminated irrigation water and fresh produce. Int. J. Food Microbiol. 2013, 167, 74–79. [Google Scholar] [CrossRef]
- Ettayebi, K.; Crawford, S.E.; Murakami, K.; Broughman, J.R.; Karandikar, U.; Tenge, V.R.; Neill, F.H.; Blutt, S.E.; Zeng, X.-L.; Qu, L.; et al. Replication of human noroviruses in stem cell–derived human enteroids. Science 2016, 353, 1387–1393. [Google Scholar] [CrossRef]
- Cook, N.; Knight, A.; Richards, G.P. Persistence and elimination of human norovirus in food and on food contact surfaces: A critical review. J. Food Prot. 2016, 79, 1273–1294. [Google Scholar] [CrossRef] [PubMed]
- Costantini, V.; Morantz, E.K.; Browne, H.; Ettayebi, K.; Zeng, X.L.; Atmar, R.L.; Estes, M.K.; Vinje, J. Human norovirus replication in human intestinal enteroids as model to evaluate virus inactivation. Emerg. Infect. Dis. 2018, 24, 1453–1464. [Google Scholar] [CrossRef]
- Zhou, Z.; Zuber, S.; Cantergiani, F.; Butot, S.; Li, D.; Stroheker, T.; Devlieghere, F.; Lima, A.; Piantini, U.; Uyttendaele, M. Inactivation of viruses and bacteria on strawberries using a levulinic acid plus sodium dodecyl sulfate based sanitizer, taking sensorial and chemical food safety aspects into account. Int. J. Food Microbiol. 2017, 257, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Okakura, Y.; Takahashi, H.; Imamura, M.; Takeuchi, A.; Shidara, H.; Kuda, T.; Kimura, B. Heat-denatured lysozyme could be a novel disinfectant for reducing hepatitis a virus and murine norovirus on berry fruit. Int. J. Food Microbiol. 2018, 266, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Dunkin, N.; Coulter, C.; Weng, S.; Jacangelo, J.G.; Schwab, K.J. Effects of ph variability on peracetic acid reduction of human norovirus gi, gii rna, and infectivity plus rna reduction of selected surrogates. Food Environ. Virol. 2019, 11, 76–89. [Google Scholar] [CrossRef] [PubMed]
- Butot, S.; Putallaz, T.; Sanchez, G. Effects of sanitation, freezing and frozen storage on enteric viruses in berries and herbs. Int. J. Food Microbiol. 2008, 126, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.Y.; Lee, J.S.; Shin, M.H.; Lee, S.H.; Hwang, I.G. Effect of wash treatments on reducing human norovirus on iceberg lettuce and perilla leaf. J. Food Prot. 2011, 74, 1908–1911. [Google Scholar] [CrossRef]
- Baert, L.; Vandekinderen, I.; Devlieghere, F.; Van Coillie, E.; Debevere, J.; Uyttendaele, M. Efficacy of sodium hypochlorite and peroxyacetic acid to reduce murine norovirus 1, b40-8, listeria monocytogenes, and escherichia coli o157:H7 on shredded iceberg lettuce and in residual wash water. J. Food Prot. 2009, 72, 1047–1054. [Google Scholar] [CrossRef]
- Girard, M.; Mattison, K.; Fliss, I.; Jean, J. Efficacy of oxidizing disinfectants at inactivating murine norovirus on ready-to-eat foods. Int. J. Food Microbiol. 2016, 219, 7–11. [Google Scholar] [CrossRef]
- Bozkurt, H.; D’Souza, D.H.; Davidson, P.M. Thermal inactivation of foodborne enteric viruses and their viral surrogates in foods. J. Food Prot. 2015, 78, 1597–1617. [Google Scholar] [CrossRef]
- FDA. Food Code. 2017 Recommendations of the United States Public Health Service Food and Drug Administration; FDA: Silver Spring, MD, USA, 2017.
- Alfano-Sobsey, E.; Sweat, D.; Hall, A.; Breedlove, F.; Rodriguez, R.; Greene, S.; Pierce, A.; Sobsey, M.; Davies, M.; Ledford, S.L. Norovirus outbreak associated with undercooked oysters and secondary household transmission. Epidemiol. Infect. 2012, 140, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Lunestad, B.T.; Maage, A.; Roiha, I.S.; Myrmel, M.; Svanevik, C.S.; Duinker, A. An outbreak of norovirus infection from shellfish soup due to unforeseen insufficient heating during preparation. Food Environ. Virol. 2016, 8, 231–234. [Google Scholar] [CrossRef]
- Hewitt, J.; Greening, G.E. Effect of heat treatment on hepatitis a virus and norovirus in new zealand greenshell mussels (perna canaliculus) by quantitative real-time reverse transcription pcr and cell culture. J. Food Prot. 2006, 69, 2217–2223. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, H.; D’Souza, D.H.; Davidson, P.M. Determination of thermal inactivation kinetics of hepatitis a virus in blue mussel (mytilus edulis) homogenate. Appl. Environ. Microbiol. 2014, 80, 3191–3197. [Google Scholar] [CrossRef] [PubMed]
- Croci, L.; Ciccozzi, M.; De Medici, D.; Di Pasquale, S.; Fiore, A.; Mele, A.; Toti, L. Inactivation of hepatitis a virus in heat-treated mussels. J. Appl. Microbiol. 1999, 87, 884–888. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Ha, S.-D. Thermal inactivation of hepatitis a virus in suspension and in dried mussels (mytilus edulis). Int. J. Food Sci. Technol. 2015, 50, 717–722. [Google Scholar] [CrossRef]
- Croci, L.; Suffredini, E.; Di Pasquale, S.; Cozzi, L. Detection of norovirus and feline calicivirus in spiked molluscs subjected to heat treatments. Food Control 2012, 25, 17–22. [Google Scholar] [CrossRef]
- Todd, E.C.; Greig, J.D.; Bartleson, C.A.; Michaels, B.S. Outbreaks where food workers have been implicated in the spread of foodborne disease. Part 2. Description of outbreaks by size, severity, and settings. J. Food Prot. 2007, 70, 1975–1993. [Google Scholar] [CrossRef]
- WHO. Viruses in Food: Scientific Advice to Support Risk Management Activities: Meeting Report; WHO: Geneva, Switzerland, 2008. [Google Scholar]
- Duret, S.; Pouillot, R.; Fanaselle, W.; Papafragkou, E.; Liggans, G.; Williams, L.; Van Doren, J.M. Quantitative risk assessment of norovirus transmission in food establishments: Evaluating the impact of intervention strategies and food employee behavior on the risk associated with norovirus in foods. Risk Anal. 2017, 37, 2080–2106. [Google Scholar] [CrossRef]
- Amar, C.F.; East, C.L.; Gray, J.; Iturriza-Gomara, M.; Maclure, E.A.; McLauchlin, J. Detection by pcr of eight groups of enteric pathogens in 4,627 faecal samples: Re-examination of the english case-control infectious intestinal disease study (1993–1996). Eur. J. Clin. Microbiol. Infect. Dis. 2007, 26, 311–323. [Google Scholar] [CrossRef]
- Phillips, G.; Tam, C.C.; Rodrigues, L.C.; Lopman, B. Prevalence and characteristics of asymptomatic norovirus infection in the community in england. Epidemiol. Infect. 2010, 138, 1454–1458. [Google Scholar] [CrossRef]
- Okabayashi, T.; Yokota, S.; Ohkoshi, Y.; Ohuchi, H.; Yoshida, Y.; Kikuchi, M.; Yano, K.; Fujii, N. Occurrence of norovirus infections unrelated to norovirus outbreaks in an asymptomatic food handler population. J. Clin. Microbiol. 2008, 46, 1985–1988. [Google Scholar] [CrossRef]
- Yu, J.H.; Kim, N.Y.; Lee, E.J.; Jeon, I.S. Norovirus infections in asymptomatic food handlers in elementary schools without norovirus outbreaks in some regions of incheon, korea. J. Korean Med. Sci. 2011, 26, 734–739. [Google Scholar] [CrossRef][Green Version]
- Utsumi, T.; Lusida, M.I.; Dinana, Z.; Wahyuni, R.M.; Yamani, L.N.; Juniastuti; Soetjipto; Matsui, C.; Deng, L.; Abe, T.; et al. Occurrence of norovirus infection in an asymptomatic population in Indonesia. Infect. Genet. Evol. 2017, 55, 1–7. [Google Scholar] [CrossRef]
- Kaarme, J.; Hickman, R.A.; Nevéus, T.; Blomberg, J.; Öhrmalm, C. Reassuringly low carriage of enteropathogens among healthy swedish children in day care centres. Public Health 2016, 140, 221–227. [Google Scholar] [CrossRef]
- Bucardo, F.; Nordgren, J.; Carlsson, B.; Kindberg, E.; Paniagua, M.; Mollby, R.; Svensson, L. Asymptomatic norovirus infections in nicaraguan children and its association with viral properties and histo-blood group antigens. Pediatr. Infect. Dis. J. 2010, 29, 934–939. [Google Scholar] [CrossRef]
- Garcia, C.; DuPont, H.L.; Long, K.Z.; Santos, J.I.; Ko, G. Asymptomatic norovirus infection in mexican children. J. Clin. Microbiol. 2006, 44, 2997–3000. [Google Scholar] [CrossRef]
- Ayukekbong, J.; Lindh, M.; Nenonen, N.; Tah, F.; Nkuo-Akenji, T.; Bergstrom, T. Enteric viruses in healthy children in cameroon: Viral load and genotyping of norovirus strains. J. Med. Virol. 2011, 83, 2135–2142. [Google Scholar] [CrossRef]
- Kabue, J.P.; Meader, E.; Hunter, P.R.; Potgieter, N. Norovirus prevalence and estimated viral load in symptomatic and asymptomatic children from rural communities of vhembe district, south africa. J. Clin. Virol. 2016, 84, 12–18. [Google Scholar] [CrossRef]
- Atmar, R.L.; Opekun, A.R.; Gilger, M.A.; Estes, M.K.; Crawford, S.E.; Neill, F.H.; Graham, D.Y. Norwalk virus shedding after experimental human infection. Emerg. Infect. Dis. 2008, 14, 1553–1557. [Google Scholar] [CrossRef]
- Kirby, A.E.; Shi, J.; Montes, J.; Lichtenstein, M.; Moe, C.L. Disease course and viral shedding in experimental norwalk virus and snow mountain virus infection. J. Med. Virol. 2014, 86, 2055–2064. [Google Scholar] [CrossRef]
- Sabria, A.; Pinto, R.M.; Bosch, A.; Bartolome, R.; Cornejo, T.; Torner, N.; Martinez, A.; Simon, M.; Dominguez, A.; Guix, S. Norovirus shedding among food and healthcare workers exposed to the virus in outbreak settings. J. Clin. Virol. 2016, 82, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Teunis, P.F.; Sukhrie, F.H.; Vennema, H.; Bogerman, J.; Beersma, M.F.; Koopmans, M.P. Shedding of norovirus in symptomatic and asymptomatic infections. Epidemiol. Infect. 2015, 143, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Phillips, G.; Tam, C.C.; Rodrigues, L.C.; Lopman, B. Risk factors for symptomatic and asymptomatic norovirus infection in the community. Epidemiol. Infect. 2011, 139, 1676–1686. [Google Scholar] [CrossRef]
- Prag, C.; Prag, M.; Fredlund, H. Proton pump inhibitors as a risk factor for norovirus infection. Epidemiol. Infect. 2017, 145, 1617–1623. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heusinkveld, M.; Mughini-Gras, L.; Pijnacker, R.; Vennema, H.; Scholts, R.; van Huisstede-Vlaanderen, K.W.; Kortbeek, T.; Kooistra-Smid, M.; van Pelt, W. Potential causative agents of acute gastroenteritis in households with preschool children: Prevalence, risk factors, clinical relevance and household transmission. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1691–1700. [Google Scholar] [CrossRef] [PubMed]
- Lopman, B.A.; Kolsin, J.M.; Hall, A.J.; Payne, D.C.; Wikswo, M.E.; Dunn, J.R.; Halasa, N.B. Evaluating previous antibiotic use as a risk factor for acute gastroenteritis among children in davidson county, tennessee, 2014–2015. J. Pediatr. Infect. Dis. Soc. 2018, 7, e86–e91. [Google Scholar]
- Monedero, V.; Buesa, J.; Rodríguez-Díaz, J. The interactions between host glycobiology, bacterial microbiota, and viruses in the gut. Viruses 2018, 10, 96. [Google Scholar] [CrossRef]
- Lee, H.; Ko, G. New perspectives regarding the antiviral effect of vitamin a on norovirus using modulation of gut microbiota. Gut Microbes 2017, 8, 616–620. [Google Scholar] [CrossRef]
- Lei, S.; Ramesh, A.; Twitchell, E.; Wen, K.; Bui, T.; Weiss, M.; Yang, X.; Kocher, J.; Li, G.; Giri-Rachman, E.; et al. High protective efficacy of probiotics and rice bran against human norovirus infection and diarrhea in gnotobiotic pigs. Front. Microbiol. 2016, 7, 1699. [Google Scholar] [CrossRef]
- Liu, P.; Escudero, B.; Jaykus, L.A.; Montes, J.; Goulter, R.M.; Lichtenstein, M.; Fernandez, M.; Lee, J.C.; De Nardo, E.; Kirby, A.; et al. Laboratory evidence of norwalk virus contamination on the hands of infected individuals. Appl. Environ. Microbiol. 2013, 79, 7875–7881. [Google Scholar] [CrossRef]
- Park, G.W.; Williamson, K.J.; DeBess, E.; Cieslak, P.R.; Gregoricus, N.; De Nardo, E.; Fricker, C.; Costantini, V.; Vinje, J. High hand contamination rates during norovirus outbreaks in long-term care facilities. Infect. Control Hosp. Epidemiol. 2018, 39, 219–221. [Google Scholar] [CrossRef]
- Mattioli, M.C.; Davis, J.; Mrisho, M.; Boehm, A.B. Quantification of human norovirus gii on hands of mothers with children under the age of five years in Bagamoyo, Tanzania. Am. J. Trop. Med. Hyg. 2015, 93, 478–484. [Google Scholar] [CrossRef][Green Version]
- Kampf, G. Efficacy of ethanol against viruses in hand disinfection. J. Hosp. Infect. 2018, 98, 331–338. [Google Scholar] [CrossRef]
- Ionidis, G.; Hubscher, J.; Jack, T.; Becker, B.; Bischoff, B.; Todt, D.; Hodasa, V.; Brill, F.H.; Steinmann, E.; Steinmann, J. Development and virucidal activity of a novel alcohol-based hand disinfectant supplemented with urea and citric acid. BMC Infect. Dis. 2016, 16, 77. [Google Scholar] [CrossRef]
- Liu, P.; Yuen, Y.; Hsiao, H.M.; Jaykus, L.A.; Moe, C. Effectiveness of liquid soap and hand sanitizer against norwalk virus on contaminated hands. Appl. Environ. Microbiol. 2010, 76, 394–399. [Google Scholar] [CrossRef]
- Tuladhar, E.; Hazeleger, W.C.; Koopmans, M.; Zwietering, M.H.; Duizer, E.; Beumer, R.R. Reducing viral contamination from finger pads: Handwashing is more effective than alcohol-based hand disinfectants. J. Hosp. Infect. 2015, 90, 226–234. [Google Scholar] [CrossRef]
- Grove, S.F.; Suriyanarayanan, A.; Puli, B.; Zhao, H.; Li, M.; Li, D.; Schaffner, D.W.; Lee, A. Norovirus cross-contamination during preparation of fresh produce. Int. J. Food Microbiol. 2015, 198, 43–49. [Google Scholar] [CrossRef]
- Pillet, S.; Berthelot, P.; Gagneux-Brunon, A.; Mory, O.; Gay, C.; Viallon, A.; Lucht, F.; Pozzetto, B.; Botelho-Nevers, E. Contamination of healthcare workers’ mobile phones by epidemic viruses. Clin. Microbiol. Infect. 2016, 22, 456.e1–e6. [Google Scholar] [CrossRef]
- Boxman, I.L.; Verhoef, L.; Dijkman, R.; Hagele, G.; Te Loeke, N.A.; Koopmans, M. Year-round prevalence of norovirus in the environment of catering companies without a recently reported outbreak of gastroenteritis. Appl. Environ. Microbiol. 2011, 77, 2968–2974. [Google Scholar] [CrossRef]
- Boxman, I.L.A.; Verhoef, L.; Hägele, G.; Klunder, K.; te Loeke, N.A.J.M.; Vennema, H.; Jansen, C.C.C.; Koopmans, M. Environmental testing for norovirus in various institutional settings using catering companies as sentinels for norovirus prevalence among the general population. Food Control 2015, 47, 98–102. [Google Scholar] [CrossRef]
- Somura, Y.; Kimoto, K.; Oda, M.; Nagano, M.; Okutsu, Y.; Mori, K.; Akiba, T.; Sadamasu, K. Detection of norovirus in swab specimens of restrooms and kitchens collected for investigation of suspected food poisoning outbreaks in tokyo. Shokuhin Eiseigaku Zasshi 2017, 58, 201–204. [Google Scholar] [CrossRef]
- Maunula, L.; Ronnqvist, M.; Aberg, R.; Lunden, J.; Nevas, M. The presence of norovirus and adenovirus on environmental surfaces in relation to the hygienic level in food service operations associated with a suspected gastroenteritis outbreak. Food Environ. Virol. 2017, 9, 334–341. [Google Scholar] [CrossRef]
- Stobnicka, A.; Gołofit-Szymczak, M.; Wójcik-Fatla, A.; Zając, V.; Korczyńska-Smolec, J.; Górny, R.L. Prevalence of human parainfluenza viruses and noroviruses genomes on office fomites. Food Environ. Virol. 2018, 10, 133–140. [Google Scholar] [CrossRef]
- Escudero, B.I.; Rawsthorne, H.; Gensel, C.; Jaykus, L.A. Persistence and transferability of noroviruses on and between common surfaces and foods. J. Food Prot. 2012, 75, 927–935. [Google Scholar] [CrossRef]
- Randazzo, W.; Khezri, M.; Ollivier, J.; Le Guyader, F.S.; Rodríguez-Díaz, J.; Aznar, R.; Sánchez, G. Optimization of pmaxx pretreatment to distinguish between human norovirus with intact and altered capsids in shellfish and sewage samples. Int. J. Microbiol. 2018, 266, 1–7. [Google Scholar] [CrossRef]
- Manuel, C.S.; Moore, M.D.; Jaykus, L.-A. Predicting human norovirus infectivity—Recent advances and continued challenges. Food Microbiol. 2018, 76, 337–345. [Google Scholar] [CrossRef]
- Stals, A.; Uyttendaele, M.; Baert, L.; Van Coillie, E. Norovirus transfer between foods and food contact materials. J. Food Prot. 2013, 76, 1202–1209. [Google Scholar] [CrossRef]
- Rönnqvist, M.; Aho, E.; Mikkelä, A.; Ranta, J.; Tuominen, P.; Rättö, M.; Maunula, L. Norovirus transmission between hands, gloves, utensils, and fresh produce during simulated food handling. Appl. Environ. Microbiol. 2014, 80, 5403–5410. [Google Scholar] [CrossRef]
- Sharps, C.P.; Kotwal, G.; Cannon, J.L. Human norovirus transfer to stainless steel and small fruits during handling. J. Food Prot. 2012, 75, 1437–1446. [Google Scholar] [CrossRef] [PubMed]
- Tuladhar, E.; Hazeleger, W.C.; Koopmans, M.; Zwietering, M.H.; Duizer, E.; Beumer, R.R. Transfer of noroviruses between fingers and fomites and food products. Int. J. Food Microbiol. 2013, 167, 346–352. [Google Scholar] [CrossRef]
| Country | Type of Shellfish | Production Areas | Dates of Sampling | NoV Prevalence (Total Number of Analyzed Samples) | Prevalence of NoV GI | Prevalence of NoV GII | Data on Viral Load a → % Positive Samples | Ref. |
|---|---|---|---|---|---|---|---|---|
| EUROPE | ||||||||
| Belgium | Oysters, clams and mussels (raw and frozen) | Seafood company | 2012–2013 | 32.3% (n = 65) | 24.6% | 13.8% | 3.3 × 103–1.4 × 104 →42.6% | [44] |
| France | Oysters | ND | ND | 14% (n = 78) | ND | ND | ND | [13] |
| France | Mussels | Thermally-treated shellfish; imported from different countries as frozen | 2008 | 21.7% (n = 83) | 8.4% | 11.4% | ND | [42] |
| France | Oysters | A and B | 2010–2011 | 9% (n = 387) | 1.6% | 8.3% | Range: 9.3 × 101–2.2 × 102 | [27] |
| Ireland | Oysters | A and B | 2005–2007 | 37.1% (n = 167) | ND | ND | ND | [45] |
| Ireland | Oysters | A and B | 2009–2011 | 88.1% (Nov-March) 50.9% (April-Oct) (n = 113) | ND | ND | Average Nov-March: 1.3 × 103 Average April-Oct: 2.1 × 102 | [13] |
| Ireland | Oysters | Point of sale | 2015–2016 | 84% (n = 25) | ND | ND | Geometric mean (95% CI): 69 (40–120) | [30] |
| Italy | Clams and mussels (before and after depuration) | A and B | 2005–2006 | 8.3% (0% in depurated samples) (n = 120) | 3.3% | 5% | ND | [32] |
| Italy | Clams, mussels and oysters | B | 2008–2009 | 51.4% (n = 70) | 37.1% | 48.6% | ND | [46] |
| Italy | Clams, mussels, oysters and others | A and B | 2008–2012 | 51.5% (n = 336) | 26.5% | 45.4% | Average A area: 3.1 × 102 Average B area: 1.9 × 103 | [47] |
| Italy | Mussels and razor shells | A and natural beds | 2011–2012 | 18.7% (n = 59) | 6.8% | 11.9% | ND | [48] |
| Italy | Clams, mussels, oysters and others | A and product at retail | 2003–2011 | 4.2% (n = 4359) | ND | ND | ND | [49] |
| Italy | Mussels and clams | A and B, as well as shellfish from registered and unregistered retailers (street vendors) | 2007–2010 | 57.5% (n = 163) | 29.4% | 56.4% | ND | [33] |
| Italy | Mussels | A and B | 2014–2015 | 23.1% (n = 108) | 9.3% | 20.4% | Average GI: 1.0 × 102 Average GII: 0.6 × 102 (range 1.2 × 102–5.8 × 107) | [50] |
| Italy | Mussels, clams, oysters, and others | A and B | 2013–2015 | 14.2% (n = 253) | 1.6% | 12.2% | ND | [51] |
| Montenegro | Mussels | A and B | 2015–2016 | 43% (n = 72) | 19.4% | 37.5% | Range GI: 10–1.2 × 103 Range GII: 10–1.2 × 104 | [31] |
| Netherlands | Oysters | Point of sale | 2015–2016 | 31% (n = 29) | ND | ND | Geometric mean (95% CI): 49 (40–91) | [30] |
| Spain (Galicia) | Clams, cockles, mussels | B and C | 2005 | 56% (n = 41) | 7.3% | 53.7% | Range: 5.6 × 101–1.5 × 104 | [52] |
| Spain (Galicia) | Mussels | B and C | 2010–2012 | 49.4% (n = 81) | 8.6% | 30.8% | Range: 5.9 × 103–1.6 × 109 | [34] |
| Spain (Galicia) | Clams, cockles, mussels | B | 2011–2012 | 45.2% (n = 168) | 32.1% | 25.6% | <102→9.5 % 102–103→31.6 % 103–104→37.9 % 104–105→20 % >105→1 % | [35] |
| UK | Oysters | B | 2004–2006 | 48% (n = 14) | 41.4% | 40% | ND | [28] |
| UK and Scotland | Oysters | 39 areas, A, B and C | 2009–2011 | 76.2% (n = 844) | 67.4% | 54.5% | <102→63.5 % 102–103→21.9 % 103–104→13.5 % | [29] |
| UK | Oysters | Point of sale | 2015–2016 | 71.7% (n = 434) | ND | ND | Geometric mean (95% CI): 78 (40–277) | [30] |
| ASIA AND AUSTRALIA | ||||||||
| Australia | Oysters | Growing areas with risk of pollution | 2010–2011 | 1.7% (n = 120) | 0% | 1.7% | ND | [53] |
| Australia | Oysters | All major oyster harvest areas within the country | 2014–2015 | <2% (n = 300) | 0% | 0% | ND | [54] |
| China | Clams, mussels, oysters and others | Retail | 2009–2011 | 13.3% (n = 840) | ND | ND | <102→37.4% 102–103→46.1% 103–104→13.9% >104→2.6% | [55] |
| Japan | Oysters | A and B | 2002 | 9% (n = 191) | 7.4% | 1.6% | <102→35.3 % 102–103→41.2 % 103–104→17.6 % >104→5.9 % | [56] |
| Vietnam | Oysters | ND | 2016–2016 | ND (n = 34) | 79% | 42% | Max GI: 2.4 × 105 Mas GII: 2.3 × 104 | [18] |
| AFRICA | ||||||||
| Morocco | Clams, cockles and oysters | ND | 2006–2010 | 29.9% (n = 77) | 20.8% | 18.2% | Average clams: 2.1 × 102 Average cockles: 2.8 × 102 | [57] |
| UNITED STATES (US) | ||||||||
| US (Louisiana) | Oysters | A and B | 2013 | 0.2% (n = 440) | 0% | 0.2% | 3.4 × 103 | [58] |
| US | Oysters | ND | 2007 | 3.9% (n = 338) | 1% | 2.9% | ND | [59] |
| Country of Commercialization | Type of Vegetables | Country of Production | Sampling Period | NoV Prevalence (Total Number of Analyzed Samples) | Data on Viral Load a | Ref. |
|---|---|---|---|---|---|---|
| EUROPE | ||||||
| Belgium, France | Leafy greens | Belgium, Canada, France, US, Mexico, Spain, Poland/Serbia | 2009–2010 | Belgium: 33.3% (n = 6) France: 50% (n = 6) | ND | [40] |
| Belgium, France | Soft red fruits | Belgium, Canada, France, US, Mexico, Spain, Poland/Serbia | 2009–2010 | Belgium: 34.5% (n = 29) France: 6.7% (n = 150) | ND | [40] |
| France | Berries | Serbia, Chile, Bulgaria, Poland, Spain, Morocco, Turkey | ND | 16% (n = 200) | ND | [42] |
| France | Lettuce | Spain, Italy, Belgium, France and Tunisia | ND | 12.4% (n = 210) | ND | [42] |
| Greece, Serbia and Poland | Fresh lettuce | Greece, Serbia and Poland | ND | 1.3% GI (n = 149) 0.8% GII (n = 126) | 6–23 | [38] |
| Italy | Fresh and RTE green vegetables | ND | 2011–2012 | Fresh leafy veg: 0.1% (n = 1372) RTE veg: 0% (n = 1160) | ND | [36] |
| Italy | RTE vegetables | ND | 2014–2015 | 0% (n = 911) | ND | [37] |
| Italy | Raw and RTE vegetables, and frozen berries | ND | 2012–2017 | 2.9% GI (n = 51) | ND | [39] |
| OTHER COUNTRIES | ||||||
| Brazil | Fresh lettuce Green onions Strawberries | ND | 2015–2016 | 0% (n = 12) 0% (n = 12) 0% (n = 12) | ND | [60] |
| Canada | RTE leafy greens | ND | 2009 | 54% (n = 275) | Median: 5.0 × 102 Range: 1.4–9 × 106 | [41] |
| Canada | Leafy greens | Belgium, Canada, France, US, Mexico, Spain, Poland/Serbia | 2009–2010 | 28.2% (n = 641) | ND | [40] |
| Egypt | Green onion, watercress, radish, leek and lettuce | Egypt | 2008–2009 | 20.8–34% (GI; n = 144) 0% (GII; n = 144) | Range: 4.2 × 103–1.6 × 104 | [61] |
| Food Product | Treatment | Log10 Reduction | Observations | Ref. |
|---|---|---|---|---|
| Measurement of NoV Genomes | ||||
| Blueberries, strawberries, raspberries, parsley | Stirring 15 g of food in 200 mL of tap water for 30 s + rinsing in 200 mL of tap water | 0.1–1.5 | Reductions on raspberries and parsley were only 0.1–0.9 log10 | [68] |
| Stirring 15 g of food in 200 mL of tap water containing 200 ppm FC for 30 s + rinsing in 200 mL of tap water | 0–3.4 | Reductions on raspberries and parsley were only 0–1.8 log10 | [68] | |
| Iceberg lettuce and perilla (mint) leaf | Immersion 5 g of food in 900 mL of water for 2 min + 30 s of rinsing water | 0.9–1.3 | Addition of a commercial class I detergent that can be used to wash fruits and vegetables made no difference | [69] |
| Measurement of MNV infectivity | ||||
| Iceberg lettuce | Shaking 50 g of food in 500 mL of tap water for 5 min + spin drying for 1 min | 1.1 | Considerable numbers of viruses were found in residual wash water | [70] |
| Shaking 50 g of food in 500 mL of tap water containing 200 mg/L of sodium hypochlorite for 5 min + spin drying for 1 min | 1.6–2.2 | Effectiveness was greatly influenced by the presence of organic material | [70] | |
| Shaking 50 g of food in 500 mL of tap water containing 80 mg/L of PAA for 5 min + spin drying for 1 min | 0.8 | [70] | ||
| Shaking 50 g of food in 500 mL of tap water containing 200 mg/L of PAA for 5 min + spin drying for 1 min | 2.2 | [70] | ||
| Romaine lettuce, blueberries, strawberries | Immersion of small portions of food in 10–15 mL solution containing 50 ppm FC for 1 min | 0–2.5 | Reductions differed by food (lettuce and strawberries < blueberries) The addition of Feclone™ significantly improved efficacy by >2 log10 | [71] |
| Immersion of small portions of food in 10–15 mL solution containing 85 ppm PAA for 1 min | 2.5–3.5 | The addition of Feclone™ improved efficacy by >2 log10 only in lettuce | [71] | |
| Immersion of small portions of food in 10–15 mL solution containing 20 ppm chlorine dioxide for 1 min | <1 | [71] | ||
| Strawberries | Manual stirring of 25 g of food in 200 mL of water for 2 min + spray rinsing in 200 mL of potable water | 1 | [65] | |
| Manual stirring of 25 g of food in 200 mL of water containing sodium hypochlorite 50 ppm for 2 min + spray rinsing in 200 mL of potable water | 1.5 | Reductions observed for HAV and MS2 bacteriophage were 0.6–1.9 log10 higher | [65] | |
| Manual stirring of 25 g of food in 200 mL of a solution containing 0.5% LVA plus 0.5% SDS solution for 2 min + spray rinsing in 200 mL of potable water | 1.4 | Concentration of 5% LVA plus 2% SDS showed no significantly higher reductions | [65] | |
| Blueberries and mixed berries | Soaking 10–20 g of food in 100 mL of distilled water for 1 min | 1.6–1.8 | As opposed to previous studies, spiking of berries was performed by immersion in a virus-containing solution, without a drying step | [66] |
| Soaking 10–20 g of food in 100 mL of distilled water containing 100 ppm of FC for 1 min | 3.8–4.2 | [66] | ||
| Soaking 10–20 g of food in 100 mL of distilled water containing 1% heat-denatured lysozyme for 1 min | 4.1–4.2 | [66] | ||
| NoV GII (RTqPCR) | HAV (RTqPCR) | HAV (Infectious) | Ref. | |
|---|---|---|---|---|
| D-value 60 °C (min) | 25 | NA | 3.25–7.93 | [76,77,78,79] |
| D-value 80 °C (min) | 4.84 | NA | 3.2 | [76,78] |
| D-value 90 °C (min) | NA | NA | 0.55–0.9 | [15] |
| D-value 100 °C (min) | 0.93–1.3 | 1.58 | 0.38–0.86 | [76,79,80] |
| z-value (°C) | 28 | NA | 12.97–27.5 | [15,76,77] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guix, S.; Pintó, R.M.; Bosch, A. Final Consumer Options to Control and Prevent Foodborne Norovirus Infections. Viruses 2019, 11, 333. https://doi.org/10.3390/v11040333
Guix S, Pintó RM, Bosch A. Final Consumer Options to Control and Prevent Foodborne Norovirus Infections. Viruses. 2019; 11(4):333. https://doi.org/10.3390/v11040333
Chicago/Turabian StyleGuix, Susana, Rosa M. Pintó, and Albert Bosch. 2019. "Final Consumer Options to Control and Prevent Foodborne Norovirus Infections" Viruses 11, no. 4: 333. https://doi.org/10.3390/v11040333
APA StyleGuix, S., Pintó, R. M., & Bosch, A. (2019). Final Consumer Options to Control and Prevent Foodborne Norovirus Infections. Viruses, 11(4), 333. https://doi.org/10.3390/v11040333







