IFNα and IFNγ Impede Marek’s Disease Progression
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells and Viruses
2.2. Chicken Interferons and Antibodies
2.3. In Vitro Assays
2.4. Ethics Statement and Animal Experiments
2.5. Statistical Analyses
3. Results and Discussion
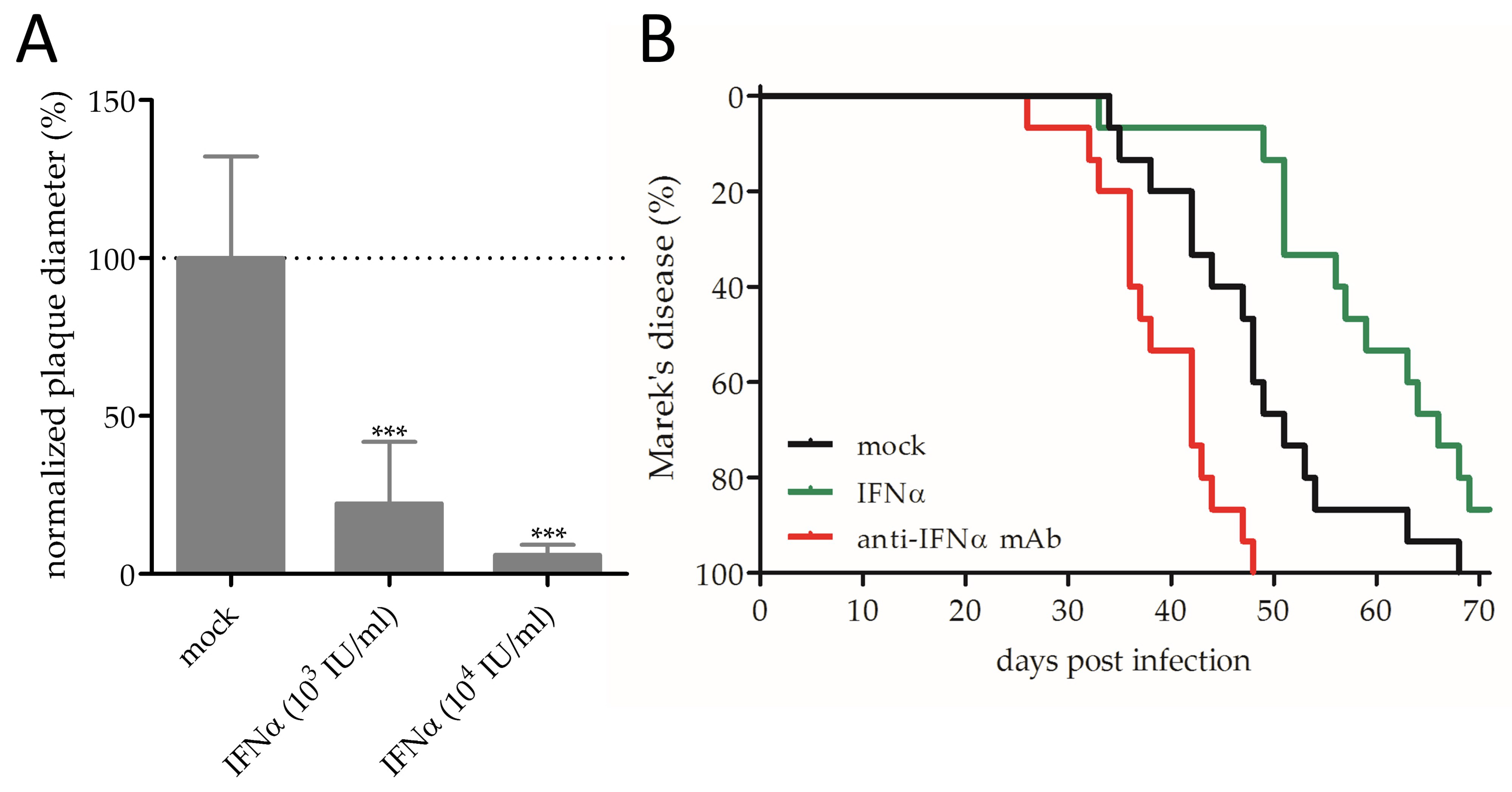
3.1. Effect of IFNα on MDV Replication and Pathogenesis
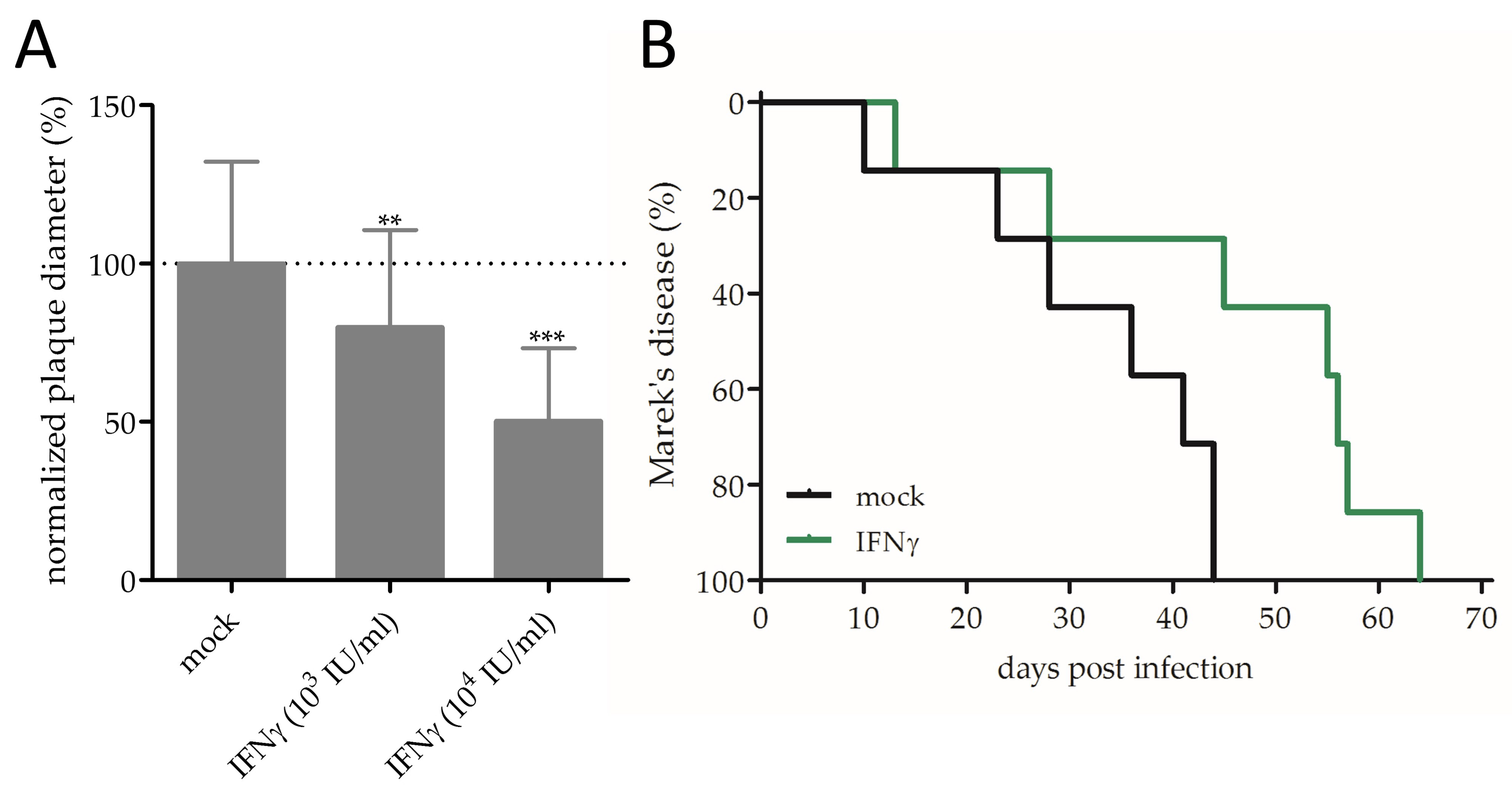
3.2. Effect of IFNγ on MDV Replication and Pathogenesis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Morrow, C.; Fehler, F. Marek’s Disease: A worldwide problem. In Marek’s Disease: An Evolving Problem; Davison, F., Nair, V., Eds.; Elsevier: Amsterdam, The Netherlands, 2004. [Google Scholar] [CrossRef]
- Bertzbach, L.D.; Laparidou, M.; Härtle, S.; Etches, R.J.; Kaspers, B.; Schusser, B.; Kaufer, B.B. Unraveling the role of B cells in the pathogenesis of an oncogenic avian herpesvirus. Proc. Natl. Acad. Sci. USA 2018, 115, 11603–11607. [Google Scholar] [CrossRef] [PubMed]
- Nair, V. Latency and tumorigenesis in Marek’s disease. Avian Dis. 2013, 57, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Bertzbach, L.D.; Kheimar, A.; Ali, F.A.Z.; Kaufer, B.B. Viral Factors Involved in Marek’s Disease Virus (MDV) Pathogenesis. Curr. Clin. Microbiol. Rep. 2018, 5, 238–244. [Google Scholar] [CrossRef]
- Schat, K.A.; Nair, V. Neoplastic Diseases. In Diseases of Poultry; Swayne, D.E., Ed.; John Wiley & Sons Inc: Hoboken, NJ, USA, 2017; pp. 513–673. [Google Scholar] [CrossRef]
- Lee, A.J.; Ashkar, A.A. The Dual Nature of Type I and Type II Interferons. Front. Immunol. 2018, 9, 2061. [Google Scholar] [CrossRef]
- Reuter, A.; Soubies, S.; Härtle, S.; Schusser, B.; Kaspers, B.; Staeheli, P.; Rubbenstroth, D. Antiviral activity of lambda interferon in chickens. J. Virol. 2014, 88, 2835–2843. [Google Scholar] [CrossRef]
- Karpala, A.J.; Morris, K.R.; Broadway, M.M.; McWaters, P.G.; O’Neil, T.E.; Goossens, K.E.; Lowenthal, J.W.; Bean, A.G. Molecular cloning, expression, and characterization of chicken IFN-lambda. J. Interferon Cytokine Res. 2008, 28, 341–350. [Google Scholar] [CrossRef]
- Xing, Z.; Schat, K.A. Expression of cytokine genes in Marek’s disease virus-infected chickens and chicken embryo fibroblast cultures. Immunology 2000, 100, 70–76. [Google Scholar] [CrossRef]
- Bertzbach, L.D.; van Haarlem, D.A.; Härtle, S.; Kaufer, B.B.; Jansen, C.A. Marek’s Disease Virus Infection of Natural Killer Cells. Microorganisms 2019, 7, 588. [Google Scholar] [CrossRef]
- Kaiser, P.; Underwood, G.; Davison, F. Differential cytokine responses following Marek’s disease virus infection of chickens differing in resistance to Marek’s disease. J. Virol. 2003, 77, 762–768. [Google Scholar] [CrossRef]
- Jarosinski, K.W.; Njaa, B.L.; O’Connell, P.H.; Schat, K.A. Pro-inflammatory responses in chicken spleen and brain tissues after infection with very virulent plus Marek’s disease virus. Viral Immunol. 2005, 18, 148–161. [Google Scholar] [CrossRef]
- Abdul-Careem, M.F.; Hunter, B.D.; Lee, L.F.; Fairbrother, J.H.; Haghighi, H.R.; Read, L.; Parvizi, P.; Heidari, M.; Sharif, S. Host responses in the bursa of Fabricius of chickens infected with virulent Marek’s disease virus. Virology 2008, 379, 256–265. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Abdul-Careem, M.F.; Hunter, B.D.; Sarson, A.J.; Parvizi, P.; Haghighi, H.R.; Read, L.; Heidari, M.; Sharif, S. Host responses are induced in feathers of chickens infected with Marek’s disease virus. Virology 2008, 370, 323–332. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baaten, B.J.; Staines, K.A.; Smith, L.P.; Skinner, H.; Davison, T.F.; Butter, C. Early replication in pulmonary B cells after infection with Marek’s disease herpesvirus by the respiratory route. Viral Immunol. 2009, 22, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Heidari, M.; Sarson, A.J.; Huebner, M.; Sharif, S.; Kireev, D.; Zhou, H. Marek’s disease virus-induced immunosuppression: Array analysis of chicken immune response gene expression profiling. Viral Immunol. 2010, 23, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Jarosinski, K.W.; Jia, W.; Sekellick, M.J.; Marcus, P.I.; Schat, K.A. Cellular responses in chickens treated with IFN-alpha orally or inoculated with recombinant Marek’s disease virus expressing IFN-alpha. J. Interferon Cytokine Res. 2001, 21, 287–296. [Google Scholar] [CrossRef]
- Schat, K.; Purchase, H. Cell-Culture Methods. In A Laboratory Manual for the Isolation and Identification of Avian Pathogens, 4th ed.; American Association of Avian Pathologists: Kennett Square, PA, USA, 1998. [Google Scholar]
- Kothlow, S.; Morgenroth, I.; Tregaskes, C.A.; Kaspers, B.; Young, J.R. CD40 ligand supports the long-term maintenance and differntiation of chicken B cells in culture. Dev. Comp. Immunol. 2008, 32, 1015–1026. [Google Scholar] [CrossRef]
- Schermuly, J.; Greco, A.; Härtle, S.; Osterrieder, N.; Kaufer, B.B.; Kaspers, B. In vitro model for lytic replication, latency, and transformation of an oncogenic alphaherpesvirus. Proc. Natl. Acad. Sci. USA 2015, 112, 7279–7284. [Google Scholar] [CrossRef]
- Bertzbach, L.D.; Pfaff, F.; Pauker, V.I.; Kheimar, A.M.; Höper, D.; Härtle, S.; Karger, A.; Kaufer, B.B. The Transcriptional Landscape of Marek’s Disease Virus in Primary Chicken B Cells Reveals Novel Splice Variants and Genes. Viruses 2019, 11, 264. [Google Scholar] [CrossRef]
- Petherbridge, L.; Brown, A.C.; Baigent, S.J.; Howes, K.; Sacco, M.A.; Osterrieder, N.; Nair, V.K. Oncogenicity of virulent Marek’s disease virus cloned as bacterial artificial chromosomes. J. Virol. 2004, 78, 13376–13380. [Google Scholar] [CrossRef]
- Petherbridge, L.; Howes, K.; Baigent, S.J.; Sacco, M.A.; Evans, S.; Osterrieder, N.; Nair, V. Replication-competent bacterial artificial chromosomes of Marek’s disease virus: Novel tools for generation of molecularly defined herpesvirus vaccines. J. Virol. 2003, 77, 8712–8718. [Google Scholar] [CrossRef]
- Schumacher, D.; Tischer, B.K.; Teifke, J.P.; Wink, K.; Osterrieder, N. Generation of a permanent cell line that supports efficient growth of Marek’s disease virus (MDV) by constitutive expression of MDV glycoprotein E. J. Gen. Virol. 2002, 83, 1987–1992. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schultz, U.; Kaspers, B.; Rinderle, C.; Sekellick, M.J.; Marcus, P.I.; Staeheli, P. Recombinant chicken interferon: A potent antiviral agent that lacks intrinsic macrophage activating factor activity. Eur. J. Immunol. 1995, 25, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Weining, K.C.; Schultz, U.; Munster, U.; Kaspers, B.; Staeheli, P. Biological properties of recombinant chicken interferon-gamma. Eur. J. Immunol. 1996, 26, 2440–2447. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Schwarz, H.; Harlin, O.; Ohnemus, A.; Kaspers, B.; Staeheli, P. Synthesis of IFN-beta by virus-infected chicken embryo cells demonstrated with specific antisera and a new bioassay. J. Interferon Cytokine Res. 2004, 24, 179–184. [Google Scholar] [CrossRef]
- Plachy, J.; Weining, K.C.; Kremmer, E.; Puehler, F.; Hala, K.; Kaspers, B.; Staeheli, P. Protective effects of type I and type II interferons toward Rous sarcoma virus-induced tumors in chickens. Virology 1999, 256, 85–91. [Google Scholar] [CrossRef]
- Conradie, A.M.; Bertzbach, L.D.; Bhandari, N.; Parcells, M.; Kaufer, B.B. A Common Live-Attenuated Avian Herpesvirus Vaccine Expresses a Very Potent Oncogene. mSphere 2019, 4. [Google Scholar] [CrossRef]
- Lewis, J.A. Biological Assays for Interferons. In Lymphokines and Interferons: A Practical Approach; Clemens, M.J., Morris, A.G., Gearing, A.J.H., Eds.; IRL Press: Oxford, UK, 1987; pp. 73–87. [Google Scholar]
- Ahmed, M.; Mitchell, L.M.; Puckett, S.; Brzoza-Lewis, K.L.; Lyles, D.S.; Hiltbold, E.M. Vesicular stomatitis virus M protein mutant stimulates maturation of Toll-like receptor 7 (TLR7)-positive dendritic cells through TLR-dependent and -independent mechanisms. J. Virol. 2009, 83, 2962–2975. [Google Scholar] [CrossRef]
- Vengris, V.E.; Mare, C.J. Protection of Chickens against Marek’s Disease Virus JM-V Strain with Statolon and Exogenous Interferon. Avian Dis. 1973, 17. [Google Scholar] [CrossRef]
- Zelnik, V.; Harlin, O.; Fehler, F.; Kaspers, B.; Göbel, T.W.; Nair, V.K.; Osterrieder, N. An enzyme-linked immunosorbent assay (ELISA) for detection of Marek’s disease virus-specific antibodies and its application in an experimental vaccine trial. J. Vet. Med. B Infect Dis. Vet. Public Health 2004, 51, 61–67. [Google Scholar] [CrossRef]
- Levy, A.M.; Heller, E.D.; Leitner, G.; Davidson, I. Effect of native chicken interferon on MDV replication. Acta Virol. 1999, 43, 121–127. [Google Scholar] [PubMed]
- Liu, Y.; Gao, L.; Xu, Z.; Luo, D.; Zhang, Y.; Gao, Y.; Liu, C.; Zhang, Y.; Qi, X.; Cui, H.; et al. Marek’s Disease Virus RLORF4 Inhibits Type I Interferon Production by Antagonizing NF-kappaB Activation. J. Virol. 2019. [Google Scholar] [CrossRef]
- Xing, Z.; Schat, K.A. Inhibitory effects of nitric oxide and gamma interferon on in vitro and in vivo replication of Marek’s disease virus. J. Virol. 2000, 74, 3605–3612. [Google Scholar] [CrossRef] [PubMed]
- Digby, M.R.; Lowenthal, J.W. Cloning and expression of the chicken interferon-gamma gene. J. Interferon Cytokine Res. 1995, 15, 939–945. [Google Scholar] [CrossRef]
- Buza, J.J.; Burgess, S.C. Different signaling pathways expressed by chicken naive CD4(+) T cells, CD4(+) lymphocytes activated with staphylococcal enterotoxin B, and those malignantly transformed by Marek’s disease virus. J. Proteome Res. 2008, 7, 2380–2387. [Google Scholar] [CrossRef]
- Pauker, V.I.; Bertzbach, L.D.; Hohmann, A.; Kheimar, A.; Teifke, J.P.; Mettenleiter, T.C.; Karger, A.; Kaufer, B.B. Imaging Mass Spectrometry and Proteome Analysis of Marek’s Disease Virus-Induced Tumors. mSphere 2019, 4. [Google Scholar] [CrossRef]
- Rausch, M.P.; Hastings, K.T. Diverse cellular and organismal functions of the lysosomal thiol reductase GILT. Mol. Immunol. 2015, 68, 124–128. [Google Scholar] [CrossRef]
- Petrera, E.; Coto, C.E. The synergistic effect of IFN-alpha and IFN-gamma against HSV-2 replication in Vero cells is not interfered by the plant antiviral 1-cinnamoyl-3, 11-dihydroxymeliacarpin. Virol. J. 2006, 3, 45. [Google Scholar] [CrossRef][Green Version]
- Rizza, P.; Capone, I.; Moretti, F.; Proietti, E.; Belardelli, F. IFN-alpha as a vaccine adjuvant: Recent insights into the mechanisms and perspectives for its clinical use. Expert Rev. Vaccines 2011, 10, 487–498. [Google Scholar] [CrossRef]
- Haq, K.; Elawadli, I.; Parvizi, P.; Mallick, A.I.; Behboudi, S.; Sharif, S. Interferon-gamma influences immunity elicited by vaccines against very virulent Marek’s disease virus. Antivir. Res. 2011, 90, 218–226. [Google Scholar] [CrossRef]
- Santhakumar, D.; Rubbenstroth, D.; Martinez-Sobrido, L.; Munir, M. Avian Interferons and Their Antiviral Effectors. Front. Immunol. 2017, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Sid, H.; Schusser, B. Applications of Gene Editing in Chickens: A New Era Is on the Horizon. Front. Genet. 2018, 9, 456. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, P. Advances in avian immunology-prospects for disease control: A review. Avian Pathol. 2010, 39, 309–324. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertzbach, L.D.; Harlin, O.; Härtle, S.; Fehler, F.; Vychodil, T.; Kaufer, B.B.; Kaspers, B. IFNα and IFNγ Impede Marek’s Disease Progression. Viruses 2019, 11, 1103. https://doi.org/10.3390/v11121103
Bertzbach LD, Harlin O, Härtle S, Fehler F, Vychodil T, Kaufer BB, Kaspers B. IFNα and IFNγ Impede Marek’s Disease Progression. Viruses. 2019; 11(12):1103. https://doi.org/10.3390/v11121103
Chicago/Turabian StyleBertzbach, Luca D., Olof Harlin, Sonja Härtle, Frank Fehler, Tereza Vychodil, Benedikt B. Kaufer, and Bernd Kaspers. 2019. "IFNα and IFNγ Impede Marek’s Disease Progression" Viruses 11, no. 12: 1103. https://doi.org/10.3390/v11121103
APA StyleBertzbach, L. D., Harlin, O., Härtle, S., Fehler, F., Vychodil, T., Kaufer, B. B., & Kaspers, B. (2019). IFNα and IFNγ Impede Marek’s Disease Progression. Viruses, 11(12), 1103. https://doi.org/10.3390/v11121103




