Productive Infection of Mouse Mammary Glands and Human Mammary Epithelial Cells by Zika Virus
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Mice
2.3. Virus Strains
2.4. Foci Forming Assay
2.5. Animal Infections
2.6. Cell Infections
2.7. Immunofluorescence
2.8. Flow Cytometry
2.9. Ethics Statement
2.10. Ethics Statement
2.11. Statistical Analysis
3. Results
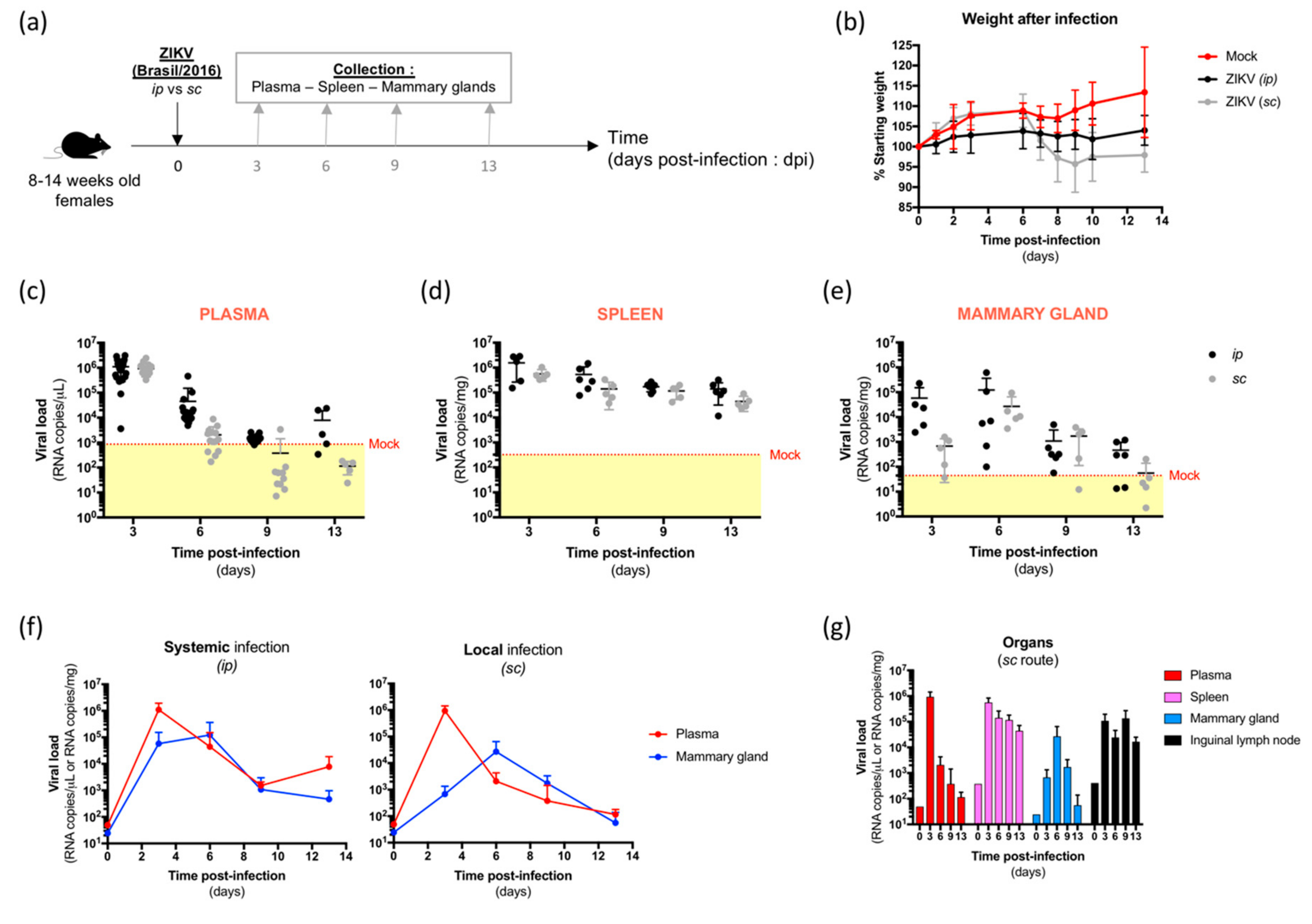
3.1. ZIKV Spreads to the Mammary Glands of Systemically- and Locally-Infected A129 Mice with Differential Kinetics
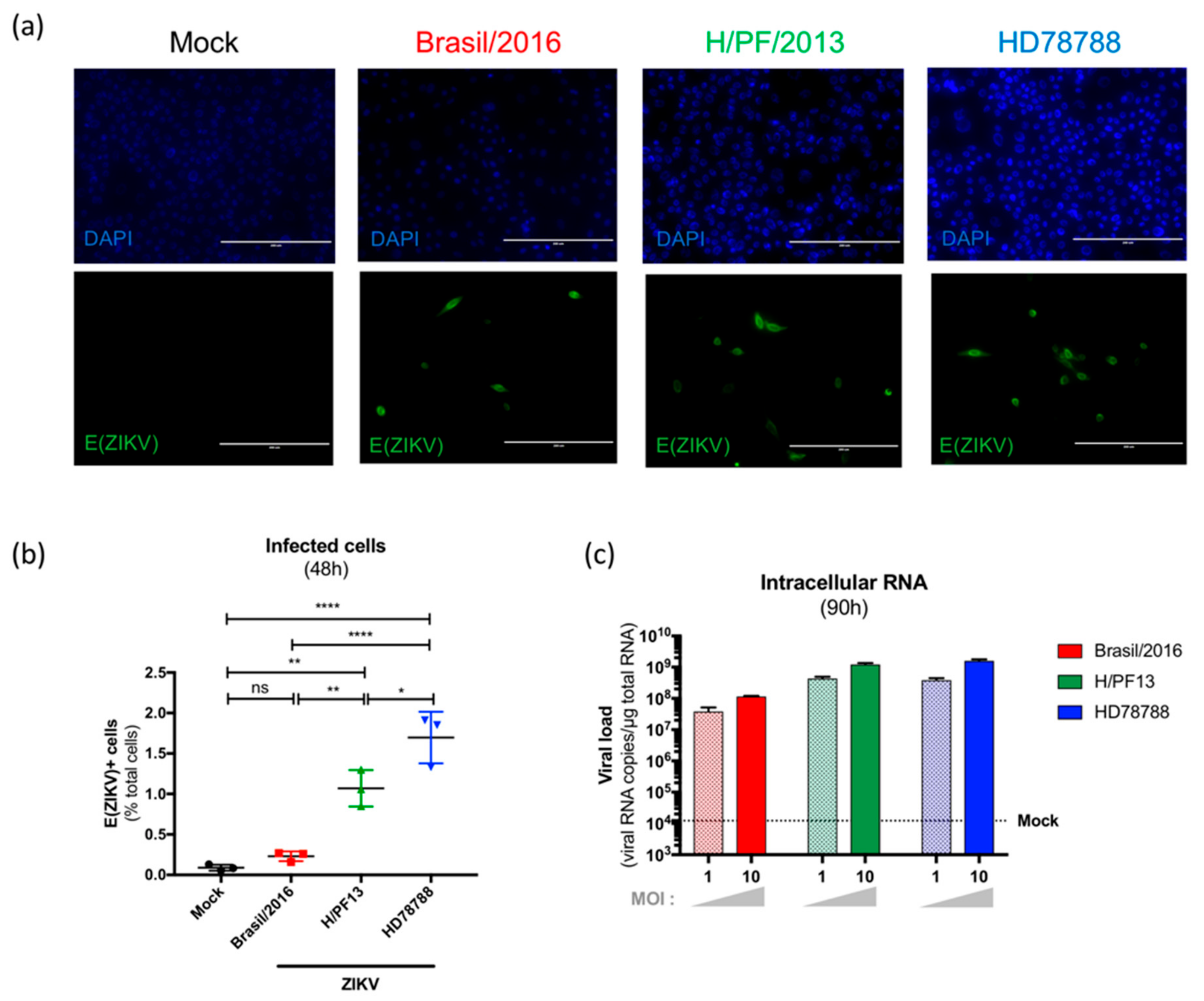
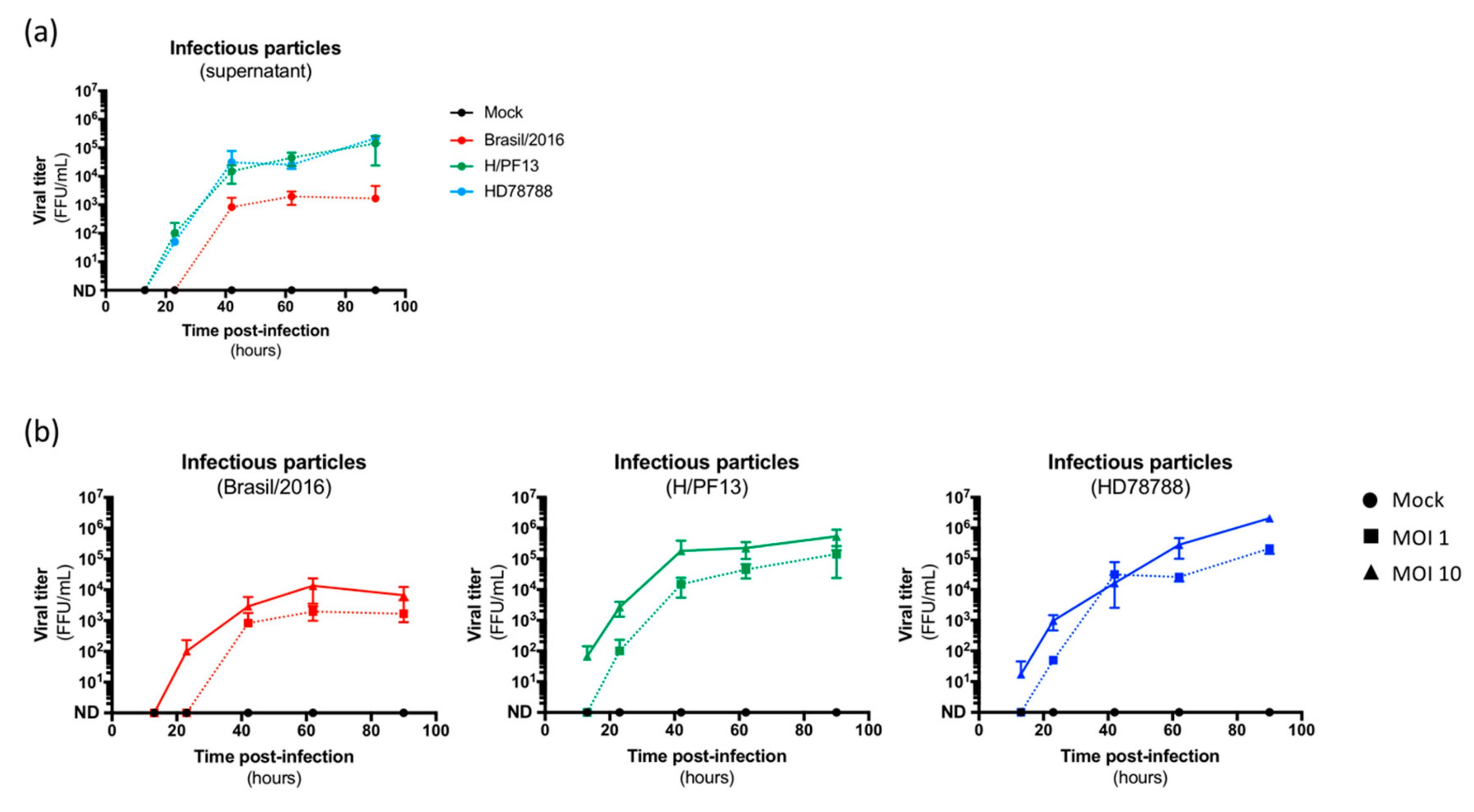
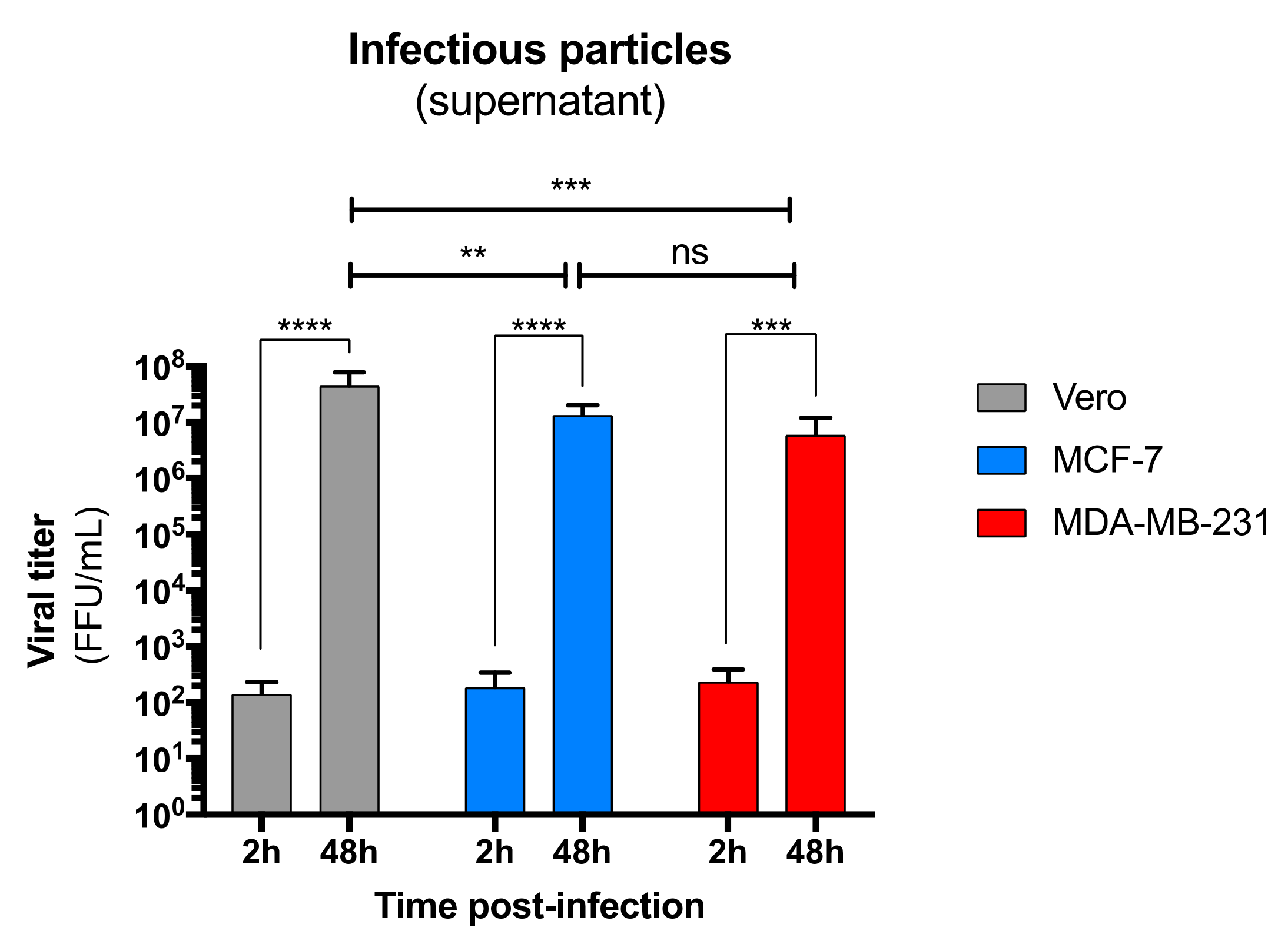
3.2. ZIKV Productively Infects Human Primary Mammary Epithelial Cells
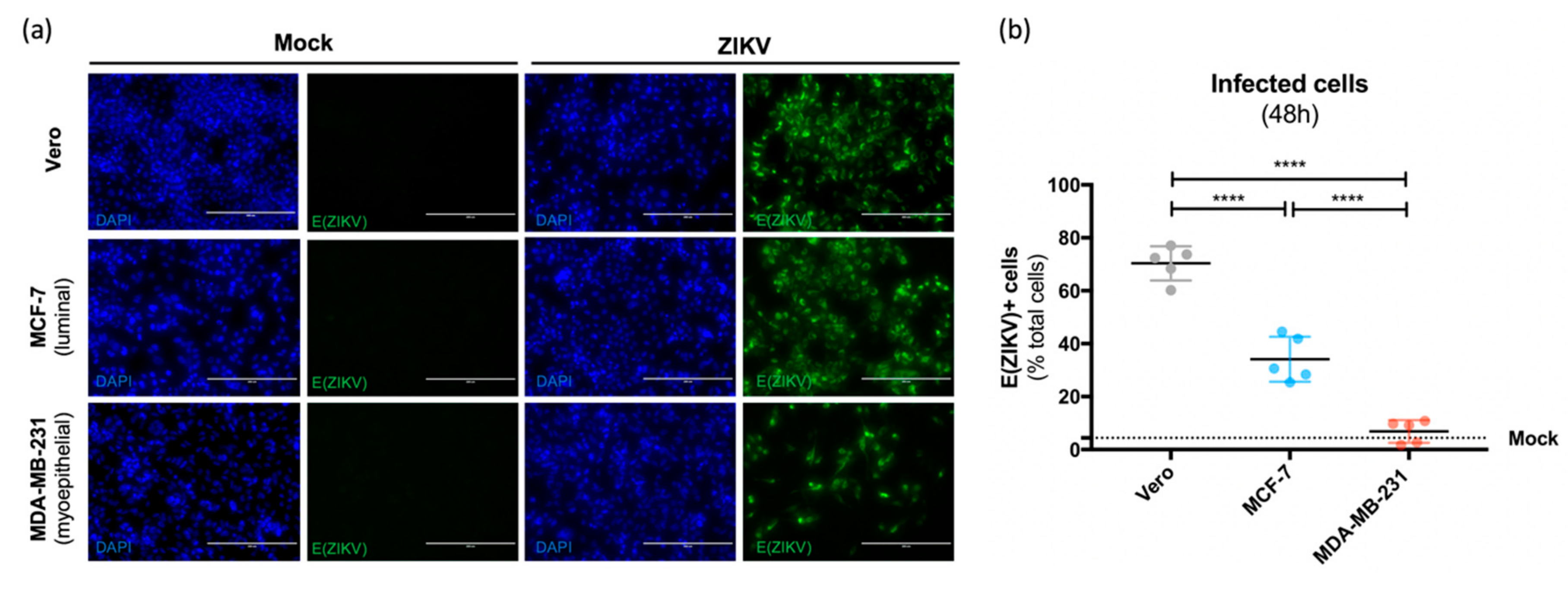
3.3. Both Human Luminal and Myoepithelial Cells of the Mammary Epithelium are Permissive to ZIKV Infection
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Boyer, S.; Calvez, E.; Chouin-Carneiro, T.; Diallo, D.; Failloux, A.-B. An overview of mosquito vectors of Zika virus. Microbes Infect. 2018, 20, 646–660. [Google Scholar] [CrossRef]
- Musso, D.; Roche, C.; Robin, E.; Nhan, T.; Teissier, A.; Cao-Lormeau, V.-M. Potential Sexual Transmission of Zika Virus. Emerg. Infect. Dis. 2015, 21, 359–361. [Google Scholar] [CrossRef]
- D’Ortenzio, E.; Matheron, S.; Yazdanpanah, Y.; de Lamballerie, X.; Hubert, B.; Piorkowski, G.; Maquart, M.; Descamps, D.; Damond, F.; Leparc-Goffart, I. Evidence of Sexual Transmission of Zika Virus. N. Engl. J. Med. 2016, 374, 2195–2198. [Google Scholar] [CrossRef] [PubMed]
- Grischott, F.; Puhan, M.; Hatz, C.; Schlagenhauf, P. Non-vector-borne transmission of Zika virus: A systematic review. Travel Med. Infect. Dis. 2016, 14, 313–330. [Google Scholar] [CrossRef] [PubMed]
- Mansuy, J.M.; Dutertre, M.; Mengelle, C.; Fourcade, C.; Marchou, B.; Delobel, P.; Izopet, J.; Martin-Blondel, G. Zika virus: High infectious viral load in semen, a new sexually transmitted pathogen? Lancet Infect. Dis. 2016, 16, 405. [Google Scholar] [CrossRef]
- Mansuy, J.M.; Suberbielle, E.; Chapuy-Regaud, S.; Mengelle, C.; Bujan, L.; Marchou, B.; Delobel, P.; Gonzalez-Dunia, D.; Malnou, C.E.; Izopet, J.; et al. Zika virus in semen and spermatozoa. Lancet Infect. Dis. 2016, 16, 1106–1107. [Google Scholar] [CrossRef]
- Murray, K.O.; Gorchakov, R.; Carlson, A.R.; Berry, R.; Lai, L.; Natrajan, M.; Garcia, M.N.; Correa, A.; Patel, S.M.; Aagaard, K.; et al. Prolonged Detection of Zika Virus in Vaginal Secretions and Whole Blood. Emerg. Infect. Dis. 2017, 23, 99–101. [Google Scholar] [CrossRef]
- Penot, P.; Brichler, S.; Guilleminot, J.; Lascoux-Combe, C.; Taulera, O.; Gordien, E.; Leparc-Goffart, I.; Molina, J.-M. Infectious Zika virus in vaginal secretions from an HIV-infected woman, France, August 2016. Euro Surveill. 2017, 22, 30444. [Google Scholar] [CrossRef]
- Calvet, G.; Aguiar, R.S.; Melo, A.S.O.; Sampaio, S.A.; de Filippis, I.; Fabri, A.; Araujo, E.S.M.; de Sequeira, P.C.; de Mendonça, M.C.L.; de Oliveira, L.; et al. Detection and sequencing of Zika virus from amniotic fluid of fetuses with microcephaly in Brazil: A case study. Lancet Infect. Dis. 2016, 16, 653–660. [Google Scholar] [CrossRef]
- Campos R de, M.; Cirne-Santos, C.; Meira, G.L.S.; Santos, L.L.R.; de Meneses, M.D.; Friedrich, J.; Jansen, S.; Ribeiro, M.S.; da Cruz, I.C.; Schmidt-Chanasit, J.; et al. Prolonged detection of Zika virus RNA in urine samples during the ongoing Zika virus epidemic in Brazil. J. Clin. Virol. 2016, 77, 69–70. [Google Scholar]
- Kutsuna, S.; Kato, Y.; Takasaki, T.; Moi, M.; Kotaki, A.; Uemura, H.; Matono, T.; Fujiya, Y.; Mawatari, M.; Takeshita, N.; et al. Two cases of Zika fever imported from French Polynesia to Japan, December 2013 to January 2014 (corrected). Euro Surveill. 2014, 19, 20683. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, K.; Meatherall, B.; Zarra, D.; Drebot, M.; MacDonald, J.; Pabbaraju, K.; Wong, S.; Webster, P.; Lindsay, R.; Tellier, R. First case of Zika virus infection in a returning Canadian traveler. Am. J. Trop. Med. Hyg. 2014, 91, 1035–1038. [Google Scholar] [CrossRef] [PubMed]
- Gourinat, A.-C.; O’Connor, O.; Calvez, E.; Goarant, C.; Dupont-Rouzeyrol, M. Detection of Zika Virus in Urine. Emerg. Infect. Dis. 2015, 21, 84–86. [Google Scholar] [CrossRef] [PubMed]
- Barzon, L.; Pacenti, M.; Berto, A.; Sinigaglia, A.; Franchin, E.; Lavezzo, E.; Brugnaro, P.; Palù, G. Isolation of infectious Zika virus from saliva and prolonged viral RNA shedding in a traveller returning from the Dominican Republic to Italy, January 2016. Euro Surveill. 2016, 21, 30159. [Google Scholar] [CrossRef] [PubMed]
- Besnard, M.; Lastere, S.; Teissier, A.; Cao-Lormeau, V.; Musso, D. Evidence of perinatal transmission of Zika virus, French Polynesia, December 2013 and February 2014. Euro Surveill. 2014, 19, 20751. [Google Scholar] [CrossRef]
- Tan, J.J.L.; Balne, P.K.; Leo, Y.-S.; Tong, L.; Ng, L.F.P.; Agrawal, R. Persistence of Zika virus in conjunctival fluid of convalescence patients. Sci. Rep. 2017, 7, 11194. [Google Scholar] [CrossRef]
- Leung, G.H.Y.; Baird, R.W.; Druce, J.; Anstey, N.M. Zika Virus Infection in Australia Following A Monkey Bite In Indonesia. Southeast Asian J. Trop. Med. Public Health 2015, 46, 460–464. [Google Scholar]
- Mann, T.Z.; Haddad, L.B.; Williams, T.R.; Hills, S.L.; Read, J.S.; Dee, D.L.; Dziuban, E.J.; Pérez-Padilla, J.; Jamieson, D.J.; Honein, M.A.; et al. Breast milk transmission of flaviviruses in the context of Zika virus: A systematic review. Paediatr. Perinat. Epidemiol. 2018, 32, 358–368. [Google Scholar] [CrossRef]
- Dupont-Rouzeyrol, M.; Biron, A.; O’Connor, O.; Huguon, E.; Descloux, E. Infectious Zika viral particles in breastmilk. Lancet 2016, 387, 1051. [Google Scholar] [CrossRef]
- Colt, S.; Garcia-Casal, M.N.; Peña-Rosas, J.P.; Finkelstein, J.L.; Rayco-Solon, P.; Weise Prinzo, Z.C.; Mehta, S. Transmission of Zika virus through breast milk and other breastfeeding-related bodily-fluids: A systematic review. PLoS Negl. Trop. Dis. 2017, 11, e0005528. [Google Scholar] [CrossRef]
- Cavalcanti, M.G.; Cabral-Castro, M.J.; Gonçalves, J.L.S.; Santana, L.S.; Pimenta, E.S.; Peralta, J.M. Zika virus shedding in human milk during lactation: An unlikely source of infection? Int. J. Infect. Dis. 2017, 57, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Sotelo, J.R.; Sotelo, A.B.; Sotelo, F.J.B.; Doi, A.M.; Pinho, J.R.R.; de Cassia Oliveira, R.; Bezerra, A.M.P.S.; Deutsch, A.D.; Villas-Boas, L.S.; Felix, A.C.; et al. Persistence of Zika Virus in Breast Milk after Infection in Late Stage of Pregnancy. Emerg. Infect. Dis. 2017, 23, 856–857. [Google Scholar] [CrossRef] [PubMed]
- Pfaender, S.; Vielle, N.J.; Ebert, N.; Steinmann, E.; Alves, M.P.; Thiel, V. Inactivation of Zika virus in human breast milk by prolonged storage or pasteurization. Virus Res. 2017, 228, 58–60. [Google Scholar] [CrossRef] [PubMed]
- Conzelmann, C.; Zou, M.; Groß, R.; Harms, M.; Röcker, A.; Riedel, C.U.; Münch, J.; Müller, J.A. Storage-Dependent Generation of Potent Anti-ZIKV Activity in Human Breast Milk. Viruses 2019, 11, 591. [Google Scholar] [CrossRef] [PubMed]
- Blohm, G.M.; Lednicky, J.A.; Márquez, M.; White, S.K.; Loeb, J.C.; Pacheco, C.A.; Nolan, D.J.; Paisie, T.; Salemi, M.; Rodríguez-Morales, A.J.; et al. Evidence for Mother-to-Child Transmission of Zika Virus Through Breast Milk. Clin. Infect. Dis. 2018, 66, 1120–1121. [Google Scholar] [CrossRef] [PubMed]
- Blohm, G.M.; Lednicky, J.A.; Márquez, M.; White, S.K.; Loeb, J.C.; Pacheco, C.A.; Nolan, D.J.; Paisie, T.; Salemi, M.; Rodríguez-Morales, A.J.; et al. Complete Genome Sequences of Identical Zika virus Isolates in a Nursing Mother and Her Infant. Genome Announc. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Inman, J.L.; Robertson, C.; Mott, J.D.; Bissell, M.J. Mammary gland development: Cell fate specification, stem cells and the microenvironment. Development 2015, 142, 1028–1042. [Google Scholar] [CrossRef]
- Regla-Nava, J.A.; Viramontes, K.M.; Vozdolska, T.; Huynh, A.-T.; Villani, T.; Gardner, G.; Johnson, M.; Ferro, P.J.; Shresta, S.; Kim, K. Detection of Zika virus in mouse mammary gland and breast milk. PLoS Negl. Trop. Dis. 2019, 13, e0007080. [Google Scholar] [CrossRef]
- Muller, U.; Steinhoff, U.; Reis, L.F.; Hemmi, S.; Pavlovic, J.; Zinkernagel, R.M.; Aguet, M. Functional role of type I and type II interferons in antiviral defense. Science 1994, 264, 1918–1921. [Google Scholar] [CrossRef]
- Monel, B.; Compton, A.A.; Bruel, T.; Amraoui, S.; Burlaud-Gaillard, J.; Roy, N.; Guivel-Benhassine, F.; Porrot, F.; Génin, P.; Meertens, L.; et al. Zika virus induces massive cytoplasmic vacuolization and paraptosis-like death in infected cells. EMBO J. 2017, 36, 1653–1668. [Google Scholar] [CrossRef]
- Lazear, H.M.; Govero, J.; Smith, A.M.; Platt, D.J.; Fernandez, E.; Miner, J.J.; Diamond, M.S. A Mouse Model of Zika Virus Pathogenesis. Cell Host Microbe 2016, 19, 720–730. [Google Scholar] [CrossRef] [PubMed]
- Hubert, M. (Université de Paris, Paris, France). Sensitivity and permissivity assays of luminal (MCF-7) and myoepithelial (MDA-MB-231) human cell lines to African (HD78788) and Asian (H/PF13 and Brasil/2016) strains of ZIKV were assessed. Intracellular expression of envelope protein of ZIKV was detected and production of infectious particles in the extracellular medium was demonstrated. Personal communication, 2019. [Google Scholar]
- Grant, A.; Ponia, S.S.; Tripathi, S.; Balasubramaniam, V.; Miorin, L.; Sourisseau, M.; Schwarz, M.C.; Sánchez-Seco, M.P.; Evans, M.J.; Best, S.M.; et al. Zika Virus Targets Human STAT2 to Inhibit Type I Interferon Signaling. Cell Host Microbe 2016, 19, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Metka, K.; Nada, P. The role of exocytosis in the apocrine secretion of milk lipid globules in mouse mammary gland during lactogenesis. Biol. Cell 1992, 75, 211–216. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hubert, M.; Chiche, A.; Legros, V.; Jeannin, P.; Montange, T.; Gessain, A.; Ceccaldi, P.-E.; Vidy, A. Productive Infection of Mouse Mammary Glands and Human Mammary Epithelial Cells by Zika Virus. Viruses 2019, 11, 950. https://doi.org/10.3390/v11100950
Hubert M, Chiche A, Legros V, Jeannin P, Montange T, Gessain A, Ceccaldi P-E, Vidy A. Productive Infection of Mouse Mammary Glands and Human Mammary Epithelial Cells by Zika Virus. Viruses. 2019; 11(10):950. https://doi.org/10.3390/v11100950
Chicago/Turabian StyleHubert, Mathieu, Aurélie Chiche, Vincent Legros, Patricia Jeannin, Thomas Montange, Antoine Gessain, Pierre-Emmanuel Ceccaldi, and Aurore Vidy. 2019. "Productive Infection of Mouse Mammary Glands and Human Mammary Epithelial Cells by Zika Virus" Viruses 11, no. 10: 950. https://doi.org/10.3390/v11100950
APA StyleHubert, M., Chiche, A., Legros, V., Jeannin, P., Montange, T., Gessain, A., Ceccaldi, P.-E., & Vidy, A. (2019). Productive Infection of Mouse Mammary Glands and Human Mammary Epithelial Cells by Zika Virus. Viruses, 11(10), 950. https://doi.org/10.3390/v11100950




