Bacteriophage Application for Difficult-To-Treat Musculoskeletal Infections: Development of a Standardized Multidisciplinary Treatment Protocol
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Identification
2.2. Isolation of Pathogens and Susceptibility Testing
2.3. Intraoperative Administration of Bacteriophages
2.4. Postoperative Administration of Bacteriophages and Patient Follow-up
2.5. Phage Neutralization Assay
2.6. Sequence Analysis of Bacterial Isolates
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Metsemakers, W.J.; Kuehl, R.; Moriarty, T.F.; Richards, R.G.; Verhofstad, M.H.J.; Borens, O.; Kates, S.; Morgenstern, M. Infection after fracture fixation: Current surgical and microbiological concepts. Injury 2018, 49, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Trampuz, A.; Widmer, A.F. Infections associated with orthopedic implants. Curr. Opin. Infect. Dis. 2006, 19, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Boudarel, H.; Mathias, J.D.; Blaysat, B.; Grediac, M. Towards standardized mechanical characterization of microbial biofilms: Analysis and critical review. NPJ Biofilms Microb. 2018, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Kutter, E.; De Vos, D.; Gvasalia, G.; Alavidze, Z.; Gogokhia, L.; Kuhl, S.; Abedon, S.T. Phage therapy in clinical practice: Treatment of human infections. Curr. Pharm. Biotechnol. 2010, 11, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.P.; Oliveira, H.; Melo, L.D.; Sillankorva, S.; Azeredo, J. Bacteriophage-Encoded depolymerases: Their diversity and biotechnological applications. Appl. Microbiol. Biotechnol. 2016, 100, 2141–2151. [Google Scholar] [CrossRef]
- Abedon, S.T.; Thomas-Abedon, C. Phage therapy pharmacology. Curr. Pharm. Biotechnol. 2010, 11, 28–47. [Google Scholar] [CrossRef] [PubMed]
- Curtright, A.J.; Abedon, S.T. Phage therapy: Emergent property pharmacology. J. Bioanal. Biomed. 2011, 6. [Google Scholar] [CrossRef]
- Abedon, S.T.; Kuhl, S.J.; Blasdel, B.G.; Kutter, E.M. Phage treatment of human infections. Bacteriophage 2011, 1, 66–85. [Google Scholar] [CrossRef]
- Moelling, K.; Broecker, F.; Willy, C. A wake-up call: We need phage therapy now. Viruses 2018, 10, 688. [Google Scholar] [CrossRef]
- Furfaro, L.L.; Payne, M.S.; Chang, B.J. Bacteriophage therapy: Clinical trials and regulatory hurdles. Front. Cell. Infect. Microbiol. 2018, 8, 376. [Google Scholar] [CrossRef]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Akanda, Z.Z.; Taha, M.; Abdelbary, H. Current review—The rise of bacteriophage as a unique therapeutic platform in treating peri-Prosthetic joint infections. J. Orthop. Res. 2018, 36, 1051–1060. [Google Scholar] [CrossRef] [PubMed]
- Cisek, A.A.; Dabrowska, I.; Gregorczyk, K.P.; Wyzewski, Z. Phage therapy in bacterial infections treatment: One hundred years after the discovery of bacteriophages. Curr. Microbiol. 2017, 74, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Azam, A.H.; Tanji, Y. Peculiarities of staphylococcus aureus phages and their possible application in phage therapy. Appl. Microbiol. Biotechnol. 2019, 103, 4279–4289. [Google Scholar] [CrossRef] [PubMed]
- Dufour, N.; Delattre, R.; Ricard, J.D.; Debarbieux, L. The lysis of pathogenic escherichia coli by bacteriophages releases less endotoxin than by beta-Lactams. Clin. Infect. Dis. 2017, 64, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- El Haddad, L.; Harb, C.P.; Gebara, M.A.; Stibich, M.A.; Chemaly, R.F. A systematic and critical review of bacteriophage therapy against multidrug-Resistant eskape organisms in humans. Clin. Infect. Dis. 2019, 69, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Knoll, B.M.; Mylonakis, E. Antibacterial bioagents based on principles of bacteriophage biology: An overview. Clin. Infect. Dis. 2014, 58, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Pirnay, J.P.; Verbeken, G.; Ceyssens, P.J.; Huys, I.; De Vos, D.; Ameloot, C.; Fauconnier, A. The magistral phage. Viruses 2018, 10, 64. [Google Scholar] [CrossRef]
- Merabishvili, M.; Pirnay, J.P.; Verbeken, G.; Chanishvili, N.; Tediashvili, M.; Lashkhi, N.; Glonti, T.; Krylov, V.; Mast, J.; Van Parys, L.; et al. Quality-Controlled small-Scale production of a well-Defined bacteriophage cocktail for use in human clinical trials. PLoS ONE 2009, 4, e4944. [Google Scholar] [CrossRef]
- Villarroel, J.; Larsen, M.V.; Kilstrup, M.; Nielsen, M. Metagenomic analysis of therapeutic pyo phage cocktails from 1997 to 2014. Viruses 2017, 9, 328. [Google Scholar] [CrossRef]
- Djebara, S.; Maussen, C.; De Vos, D.; Merabishvili, M.; Damanet, B.; Pang, K.W.; De Leenheer, P.; Strachinaru, I.; Soentjens, P.; Pirnay, J.P. Processing phage therapy requests in a brussels military hospital: Lessons identified. Viruses 2019, 11, 265. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.H. Methods of study of bacterial viruses. In Bacteriophages; Inter-Science Publishers: New York, NY, USA, 1959; pp. 443–522. [Google Scholar]
- Wattam, A.R.; Abraham, D.; Dalay, O.; Disz, T.L.; Driscoll, T.; Gabbard, J.L.; Gillespie, J.J.; Gough, R.; Hix, D.; Kenyon, R.; et al. Patric, the bacterial bioinformatics database and analysis resource. Nucleic Acids Res. 2014, 42, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Darling, A.E.; Mau, B.; Perna, N.T. Progressivemauve: Multiple genome alignment with gene gain, loss and rearrangement. PLoS ONE 2010, 5, e11147. [Google Scholar] [CrossRef] [PubMed]
- Phage Therapy: Current Research and Applications; Caister Academic Press: Norfolk, UK, 2014.
- Chanishvili, N. A Literature Review of the Practical Application of Bacteriophage Research; Nova Science Publishers, Inc.: New York, NY, USA, 2012. [Google Scholar]
- Slopek, S.; Weber-Dabrowska, B.; Dabrowski, M.; Kucharewicz-Krukowska, A. Results of bacteriophage treatment of suppurative bacterial infections in the years 1981–1986. Arch. Immunol. Ther. Exp. 1987, 35, 569–583. [Google Scholar]
- Lang, G.; Kehr, P.; Mathevon, H.; Clavert, J.M.; Sejourne, P.; Pointu, J. Bacteriophage therapy of septic complications of orthopaedic surgery (author’s transl]. Rev. Chir. Orthop. Repar. Appar. Mot. 1979, 65, 33–37. [Google Scholar]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by pseudomonas aeruginosa (phagoburn): A randomised, controlled, double-Blind phase 1/2 trial. Lancet Infect. Dis. 2019, 19, 35–45. [Google Scholar] [CrossRef]
- Patey, O.; McCallin, S.; Mazure, H.; Liddle, M.; Smithyman, A.; Dublanchet, A. Clinical indications and compassionate use of phage therapy: Personal experience and literature review with a focus on osteoarticular infections. Viruses 2018, 11, 18. [Google Scholar] [CrossRef]
- Bearden, C.M.; Agarwal, A.; Book, B.K.; Vieira, C.A.; Sidner, R.A.; Ochs, H.D.; Young, M.; Pescovitz, M.D. Rituximab inhibits the in vivo primary and secondary antibody response to a neoantigen, bacteriophage phix174. Am. J. Transplant. 2005, 5, 50–57. [Google Scholar] [CrossRef]
- Ferry, T.; Leboucher, G.; Fevre, C.; Herry, Y.; Conrad, A.; Josse, J.; Batailler, C.; Chidiac, C.; Medina, M.; Lustig, S.; et al. Salvage debridement, antibiotics and implant retention (“dair”) with local injection of a selected cocktail of bacteriophages: Is it an option for an elderly patient with relapsing staphylococcus aureus prosthetic-Joint infection? Open Forum Infect. Dis. 2018, 5, 269. [Google Scholar] [CrossRef]
- Nir-Paz, R.; Gelman, D.; Khouri, A.; Sisson, B.M.; Fackler, J.; Alkalay-Oren, S.; Khalifa, L.; Rimon, A.; Yerushalmy, O.; Bader, R.; et al. Successful treatment of antibiotic resistant poly-Microbial bone infection with bacteriophages and antibiotics combination. Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Ferry, T.; Boucher, F.; Fevre, C.; Perpoint, T.; Chateau, J.; Petitjean, C.; Josse, J.; Chidiac, C.; L’Hostis, G.; Leboucher, G.; et al. Innovations for the treatment of a complex bone and joint infection due to xdr pseudomonas aeruginosa including local application of a selected cocktail of bacteriophages. J. Antimicrob. Chemother. 2018, 73, 2901–2903. [Google Scholar] [CrossRef] [PubMed]
- Vogt, D.; Sperling, S.; Tkhilaishvili, T.; Trampuz, A.; Pirnay, J.P.; Willy, C. beyond antibiotic therapy—Future antiinfective strategies—Update 2017. Unfallchirurg 2017, 120, 573–584. [Google Scholar] [CrossRef] [PubMed]
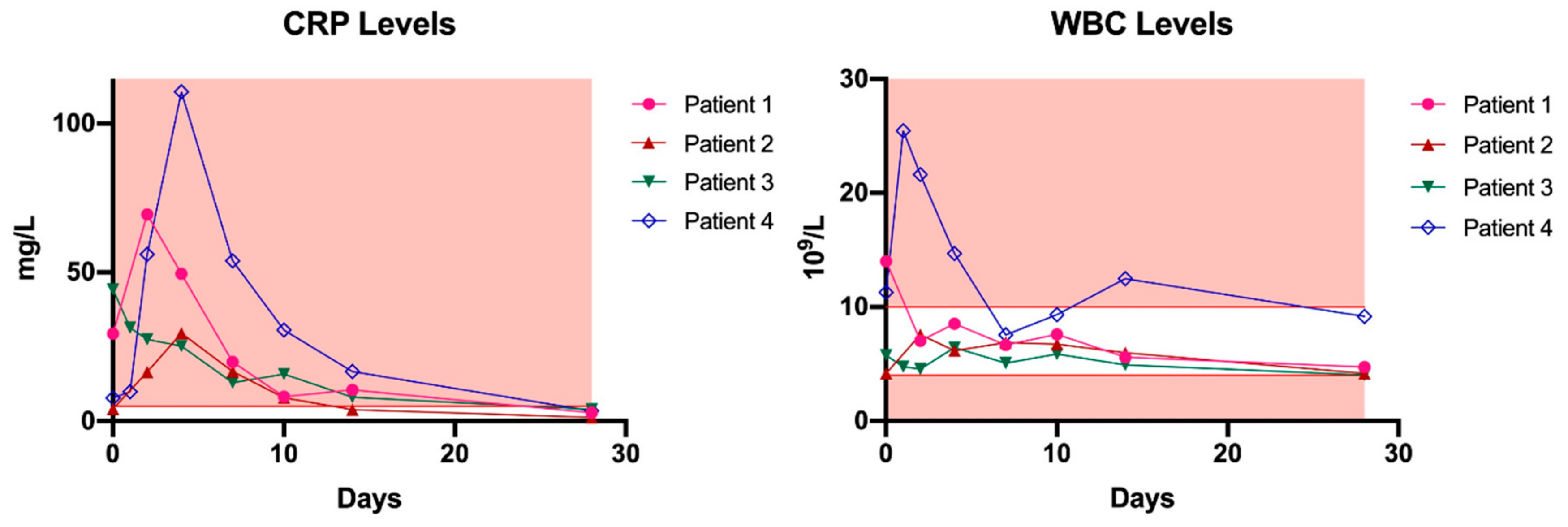
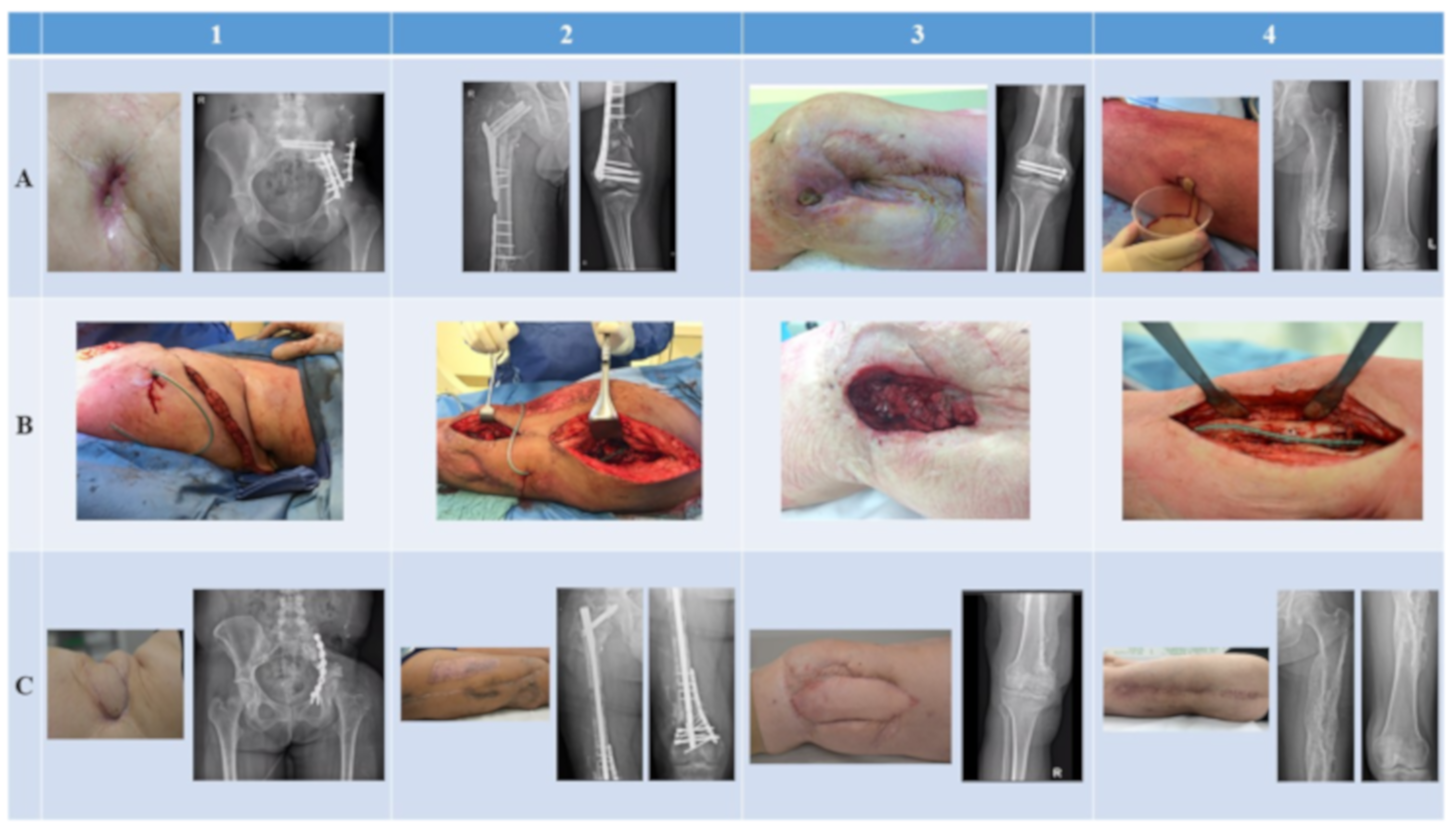
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | |
|---|---|---|---|---|
| Relevant medical history | 2013—Solitary fibrous tumor in left pelvic region. Treated with neoadjuvant radiotherapy and surgical resection; reconstruction with bone grafts, osteosynthesis and free muscle flap. | 2017—Polytrauma after assault, with open segmental fractures of the right femur. Treated with debridements, staged open reductions, proximal and distal fracture fixation of the femur and synthetic bone grafting. Soft tissue coverage with a lateral Gastrocnemius flap. | 1995—Polytrauma after building collapse, with crush lesions of the right upper leg, complex femur fractures, condylar fracture of the knee and compartment syndrome. Treated with plate fixation. | 1981—Polytrauma after traffic accident, with femur fractures. Treated with fracture fixation. |
| Infection onset and evolution | Wound dehiscence and evolution to chronic osteomyelitis of the os ileum with a draining fistula | Non-union distal femur | Postoperative wound problems: multiple episodes of erysipelas, pus drainage, evolution into a draining fistula | Infection of the surgical site with abscess formation and eventually the evolution to osteomyelitis of the femur |
| Diagnosis | 2015 | 2017 | 1995 | 1984 |
| Infected site | Pelvis | Femur | Femur | Femur |
| Isolated pathogen(s) | P. aeruginosaa S. epidermidis | P. aeruginosa S. epidermidis | S. agalactiae S. aureus | E. faecalis |
| Initial/previous treatments | Multiple debridements Removal of the osteosynthesis material Hyperbaric oxygen therapy Multiple courses of antibiotic therapy Temporary coverage of the wound with negative-pressure wound therapy | Multiple debridements Removal of the osteosynthesis material Multiple courses of antibiotic therapy | Multiple debridements Removal of the osteosynthesis material Multiple courses of antibiotic therapy Local (topical) treatment | Multiple debridements Removal of the osteosynthesis material Placement of gentamicin-coated beads Multiple courses of antibiotic therapy Local (topical) treatment |
| Antibiotic therapy used with phages and duration | Vancomycin iv Rifampicin po b Moxifloxacin po c Total duration: 3 months | Vancomycin iv Colistin iv Fosfomycin iv Total duration: 6 weeks | Vancomycin iv ↔ Clindamycin po Moxifloxacin po Total duration: 3 months | Amoxicillin iv ↔ Amoxicillin po Total duration: 3 months |
| Phages used and duration | BFC 1 Total duration: 7 days | BFC 1 Total duration: 10 days | BFC 1 Total duration: 9 days | Pyo bacteriophage Total duration: 7 days |
| AMX | TZP | CAZ | FEP | MEM | LVX | GEN | TOB | AMK | ERY | CLI | SXT | VAN | RIF | CST | OXA | LZD | MIN | PEN | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient 1 | |||||||||||||||||||
| S. epidermidis | R | R | R | S | R | R | R | S | S | R | S | S | |||||||
| P. aeruginosa | R | R | R | R | R | I | S | R | S | ||||||||||
| Patient 2 | |||||||||||||||||||
| S. epidermidis | S | R | R | S | R | S | S | S | S | R | S | S | |||||||
| P. aeruginosa | R | R | R | R | R | R | R | R | R | S | |||||||||
| Patient 3 | |||||||||||||||||||
| S. aureus | R | S | S | S | S | S | S | S | R | ||||||||||
| S. agalactiae | S | S | S | ||||||||||||||||
| Patient 4 | |||||||||||||||||||
| E. faecalis | S | R | |||||||||||||||||
| Patient | Bacterial Strains | Activity of BFC 1 | EOP of BFC 1 | Activity of Pyo Bacteriophage | Titer of Pyo Bacteriophage PFU/mL |
|---|---|---|---|---|---|
| 1 | S. epidermidis | + | 0.1 | NT | NT |
| 2 | P. aeruginosa S. epidermidis | - + | NA 0.001 | NT NT | NT NT |
| 3 | S. aureus | + | 0.7 | NT | NT |
| 4 | E. faecalis | NA | NA | + | 1.00 × 107 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onsea, J.; Soentjens, P.; Djebara, S.; Merabishvili, M.; Depypere, M.; Spriet, I.; De Munter, P.; Debaveye, Y.; Nijs, S.; Vanderschot, P.; et al. Bacteriophage Application for Difficult-To-Treat Musculoskeletal Infections: Development of a Standardized Multidisciplinary Treatment Protocol. Viruses 2019, 11, 891. https://doi.org/10.3390/v11100891
Onsea J, Soentjens P, Djebara S, Merabishvili M, Depypere M, Spriet I, De Munter P, Debaveye Y, Nijs S, Vanderschot P, et al. Bacteriophage Application for Difficult-To-Treat Musculoskeletal Infections: Development of a Standardized Multidisciplinary Treatment Protocol. Viruses. 2019; 11(10):891. https://doi.org/10.3390/v11100891
Chicago/Turabian StyleOnsea, Jolien, Patrick Soentjens, Sarah Djebara, Maia Merabishvili, Melissa Depypere, Isabel Spriet, Paul De Munter, Yves Debaveye, Stefaan Nijs, Paul Vanderschot, and et al. 2019. "Bacteriophage Application for Difficult-To-Treat Musculoskeletal Infections: Development of a Standardized Multidisciplinary Treatment Protocol" Viruses 11, no. 10: 891. https://doi.org/10.3390/v11100891
APA StyleOnsea, J., Soentjens, P., Djebara, S., Merabishvili, M., Depypere, M., Spriet, I., De Munter, P., Debaveye, Y., Nijs, S., Vanderschot, P., Wagemans, J., Pirnay, J.-P., Lavigne, R., & Metsemakers, W.-J. (2019). Bacteriophage Application for Difficult-To-Treat Musculoskeletal Infections: Development of a Standardized Multidisciplinary Treatment Protocol. Viruses, 11(10), 891. https://doi.org/10.3390/v11100891








