Establishment of an Alphavirus-Specific Neutralization Assay to Distinguish Infections with Different Members of the Semliki Forest Complex
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Human Samples
2.3. Ethics Statement
2.4. Plasmids and DNA
2.5. Lentiviral Vector Particle Production
2.6. Transduction of Cells with Lentiviral Vector Particles
2.7. Titration of Lentiviral Vector Particles
2.8. Statistical Analysis
2.9. ELISA
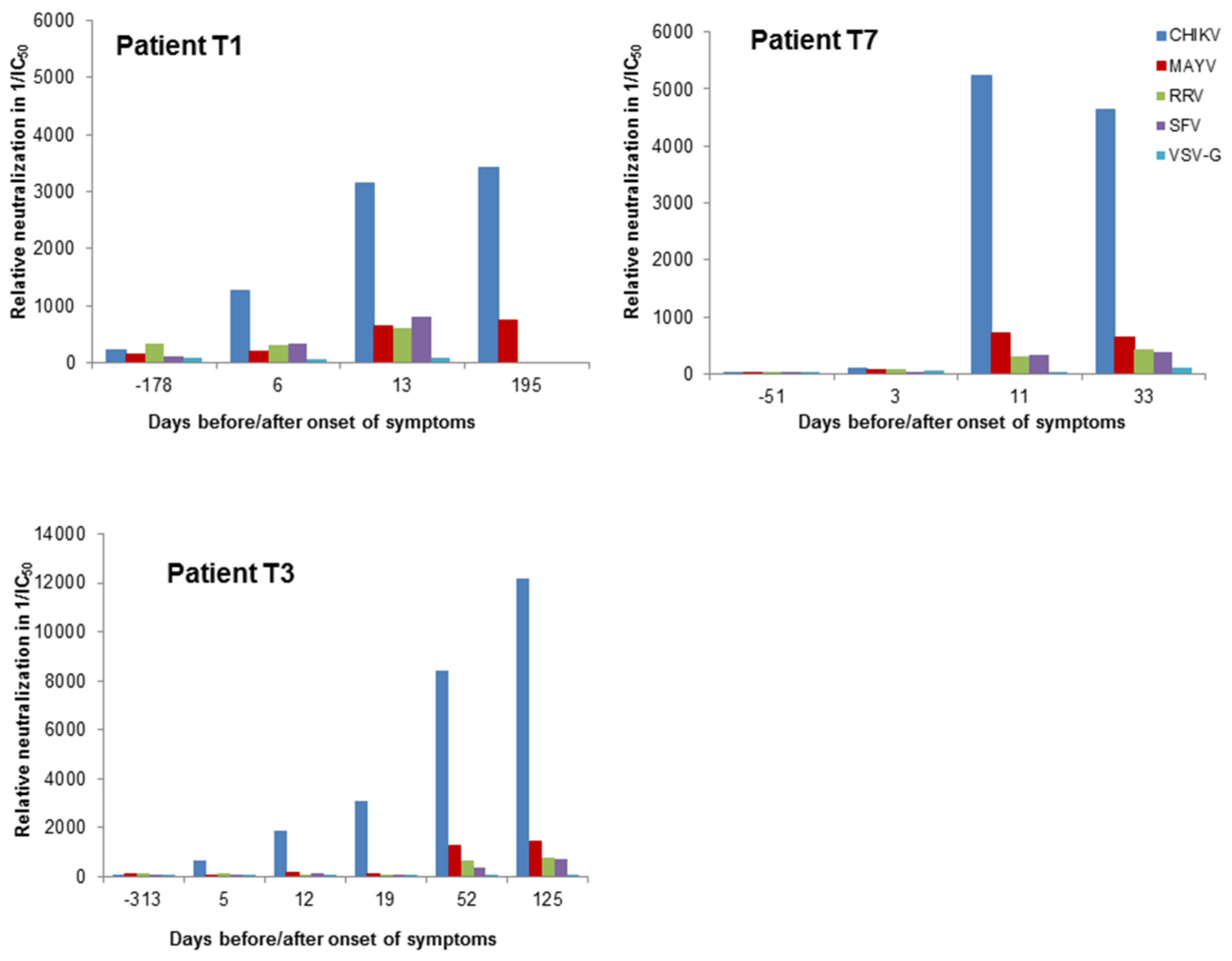
3. Results
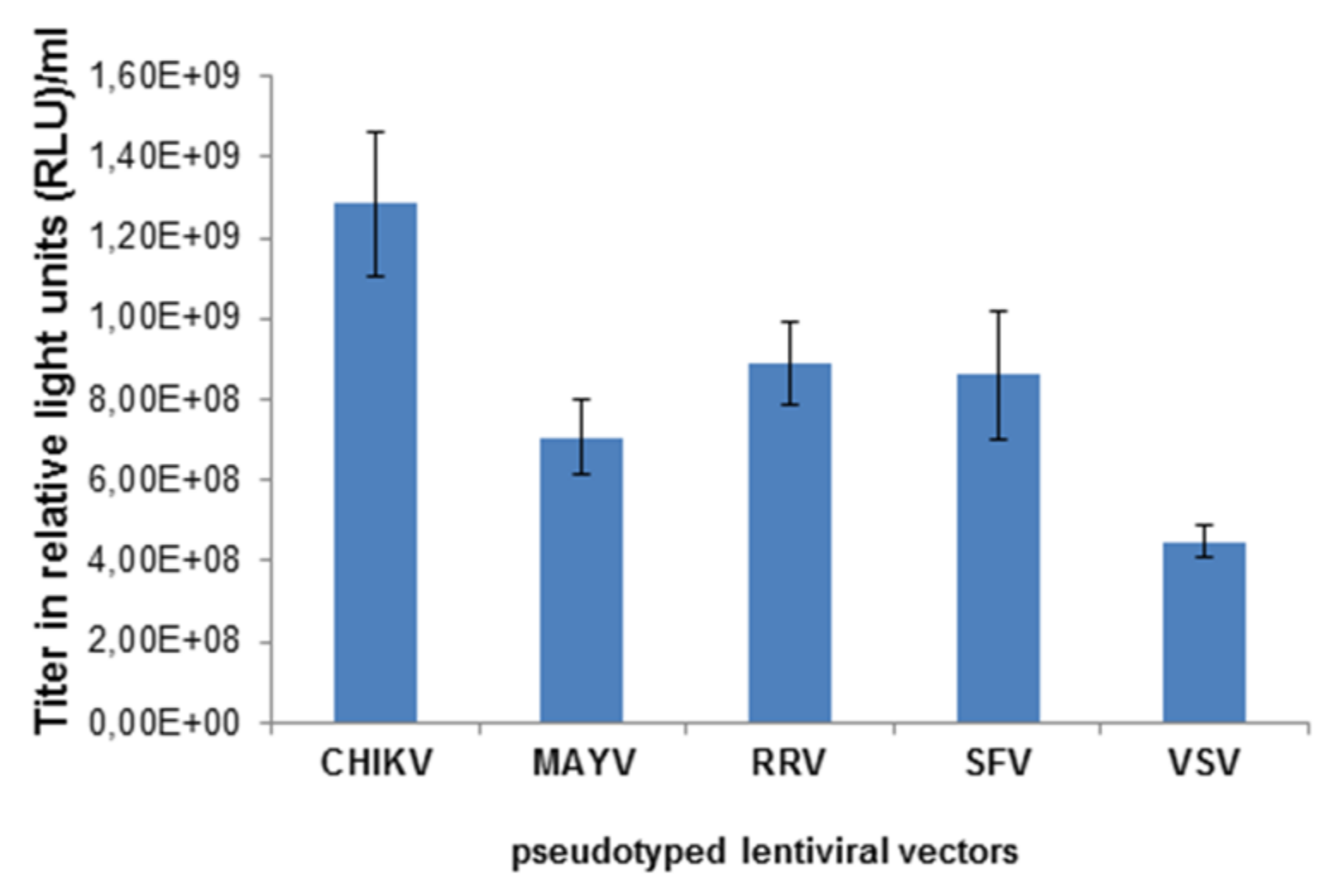
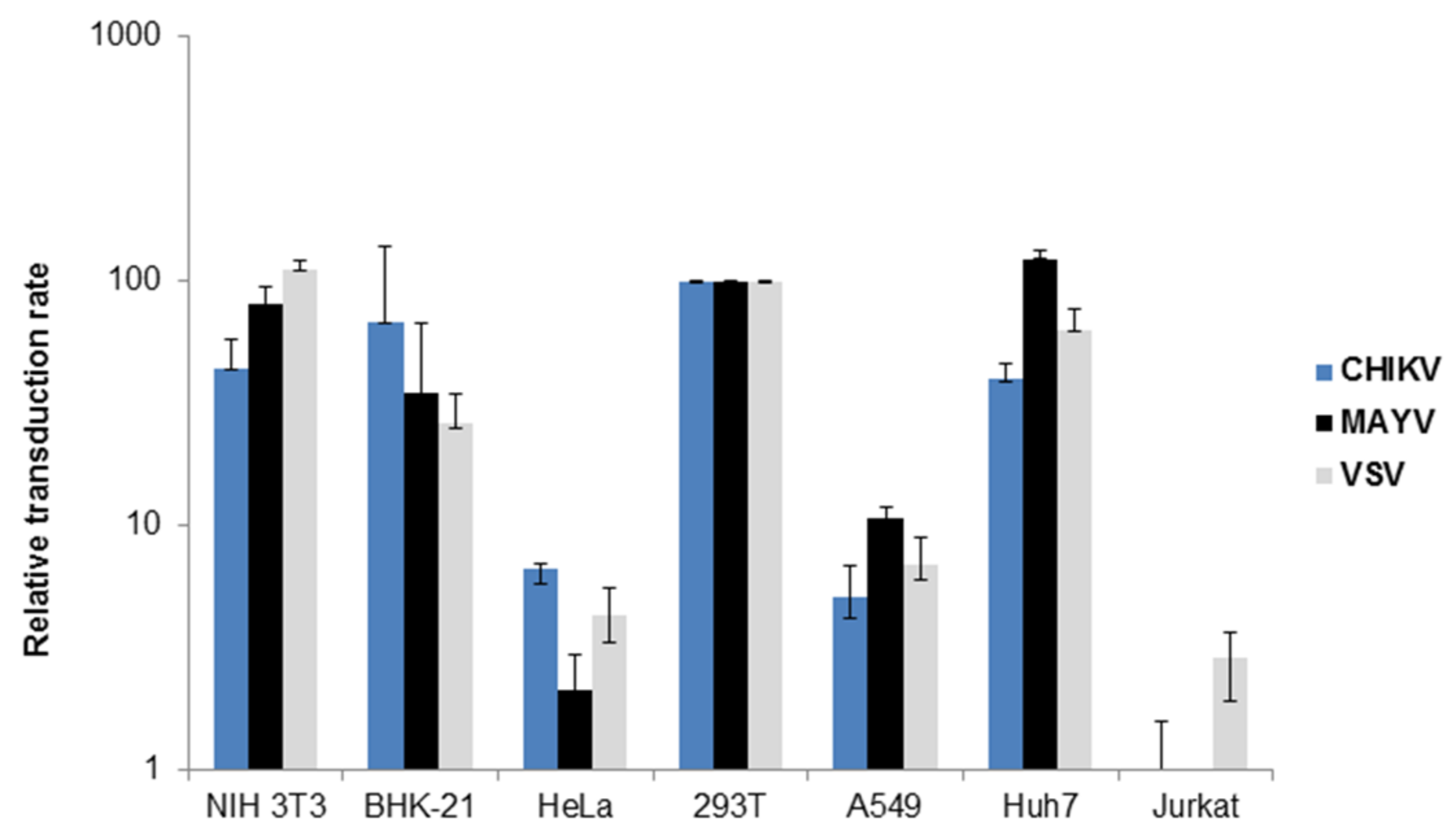
3.1. Characterization of MAYV-Pseudotyped Vector Particles
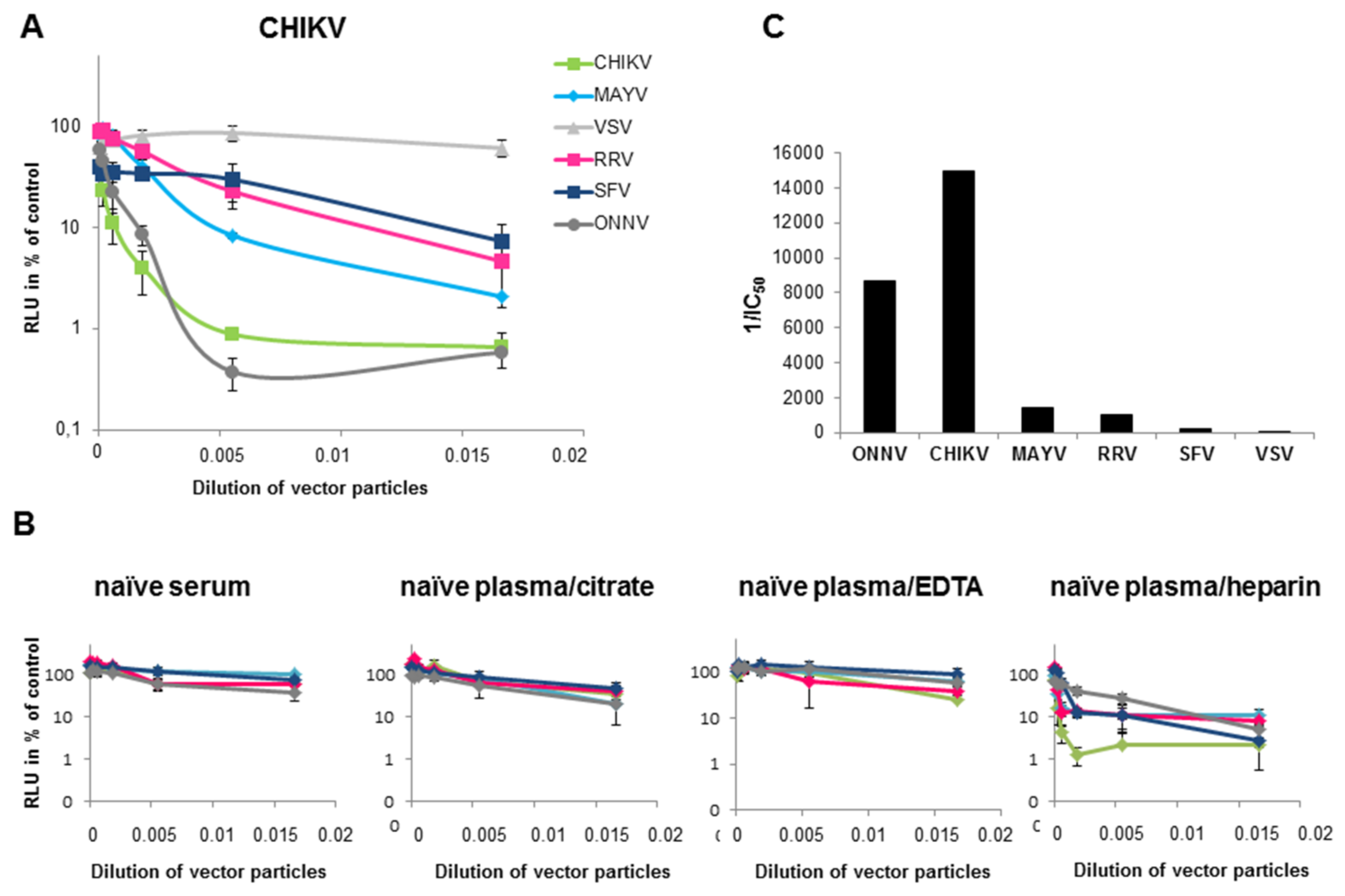
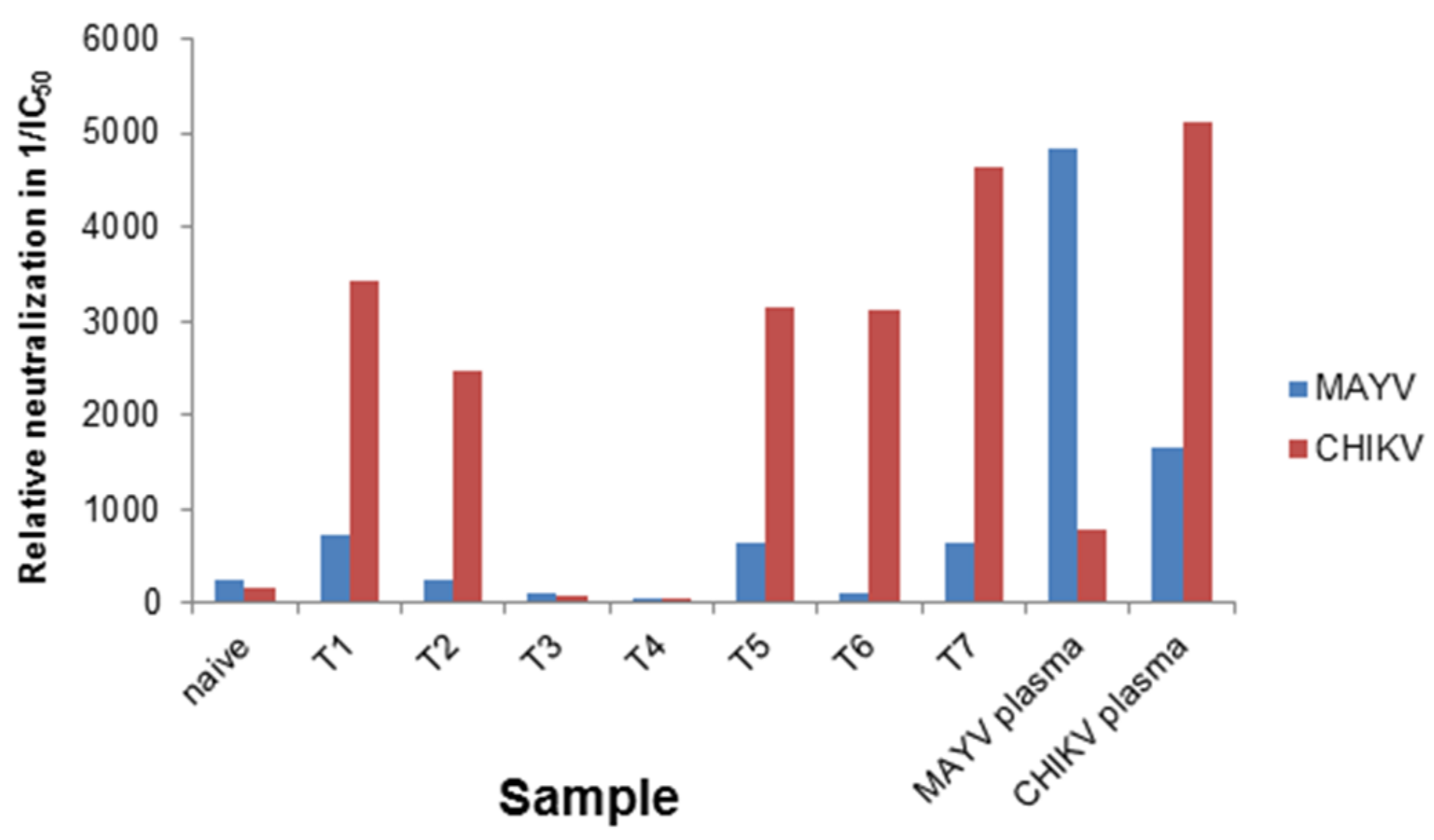
3.2. Determination of Different Alphavirus-Specific Neutralization Activities

4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Atkins, G.J.; Sheahan, B.J.; Dimmock, N.J. Semliki Forest virus infection of mice. A model for genetic and molecular analysis of viral pathogenicity. J. Gen. Virol. 1985, 66, 395–408. [Google Scholar] [CrossRef] [PubMed]
- Tesh, R.B.; Watts, D.M.; Russell, K.L.; Damodaran, C.; Calampa, C.; Cabezas, C.; Ramirez, G.; Vasquez, B.; Hayes, C.G.; Rossi, C.A.; et al. Mayaro virus disease: An emerging mosquito-borne zoonosis in tropical South America. Clin. Infect. Dis. 1999, 28, 67–73. [Google Scholar] [CrossRef]
- Schwartz, O.; Albert, M.L. Biology and pathogenesis of chikungunya virus. Nat. Rev. Microbiol. 2010, 8, 491–500. [Google Scholar] [CrossRef]
- Liu, X.; Tharmarajah, K.; Taylor, A. Ross River virus disease clinical presentation, pathogenesis and current therapeutic strategies. Microbes Infect. 2017, 19, 496–504. [Google Scholar] [CrossRef]
- Anderson, C.R.; Downs, W.G.; Wattley, G.H.; Ahin Nw Reese, A.A. Mayaro virus. A new human disease agent. II. Isolation from blood of patients in Trinidad, B.W.I. Am. J. Trop. Med. Hygiene 1957, 6, 1012–1016. [Google Scholar] [CrossRef]
- Mourao, M.P.G.; Bastos, M.d.S.; Figueiredo, R.P.; de Gimaque, J.B.L.; Galusso, E.d.S.; Kramer, V.M.; de Oliveira, C.M.; Naveca, F.G.; Figueiredo, L.T. Mayaro fever in the city of Manaus, Brazil, 2007–2008. Vector Borne Zoonotic Dis. 2012, 12, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Auguste, A.J.; Liria, J.; Forrester, N.L.; Giambalvo, D.; Moncada, M.; Long, K.C.; Morón, D.; de Manzione, N.; Tesh, R.B.; Halsey, E.S.; et al. Evolutionary and Ecological Characterization of Mayaro Virus Strains Isolated during an Outbreak, Venezuela, 2010. Emerg. Infect. Dis. 2015, 21, 1742–1750. [Google Scholar] [CrossRef] [PubMed]
- Lednicky, J.; Rochars, V.M.B.; de Elbadry, M.; Loeb, J.; Telisma, T.; Chavannes, S.; Anilis, G.; Cella, E.; Ciccozzi, M.; Okech, B.; et al. Mayaro Virus in Child with Acute Febrile Illness, Haiti, 2015. Emerg. Infect. Dis. 2016, 22, 2000–2002. [Google Scholar] [CrossRef]
- Mackay, I.M.; Arden, K.E. Mayaro virus: A forest virus primed for a trip to the city? Microbes Infect. 2016, 18, 724–734. [Google Scholar] [CrossRef]
- Long, K.C.; Ziegler, S.A.; Thangamani, S.; Hausser, N.L.; Kochel, T.J.; Higgs, S.; Tesh, R.B. Experimental transmission of Mayaro virus by Aedes aegypti. Am. J. Trop. Med. Hygiene 2011, 85, 750–757. [Google Scholar] [CrossRef]
- Zeller, H.; van Bortel, W.; Sudre, B. Chikungunya: Its History in Africa and Asia and Its Spread to New Regions in 2013–2014. J. Infect. Dis. 2016, 214, S436–S440. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Sharma, A.K.; Sukumaran, D.; Parida, M.; Dash, P.K. Two novel epistatic mutations (E1:K211E and E2:V264A) in structural proteins of Chikungunya virus enhance fitness in Aedes aegypti. Virology 2016, 497, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Gratz, N.G. Critical review of the vector status of Aedes albopictus. Med. Vet. Entomol. 2004, 18, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Flies, E.J.; Lau, C.L.; Carver, S.; Weinstein, P. Another Emerging Mosquito-Borne Disease? Endemic Ross River Virus Transmission in the Absence of Marsupial Reservoirs. BioScience 2018, 30, 1053. [Google Scholar] [CrossRef]
- Claflin, S.B.; Webb, C.E. Ross River Virus. Many Vectors and Unusual Hosts Make for an Unpredictable Pathogen. PLoS Pathog. 2015, 11, e1005070. [Google Scholar] [CrossRef] [PubMed]
- Moreira, J.; Bressan, C.S.; Brasil, P.; Siqueira, A.M. Epidemiology of acute febrile illness in Latin America. Clin. Microbiol. Infect. 2018, 24, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Possas, C. Zika. What we do and do not know based on the experiences of Brazil. Epidemiol. Health 2016, 38, e2016023. [Google Scholar] [CrossRef]
- Forrester, N.L.; Palacios, G.; Tesh, R.B.; Savji, N.; Guzman, H.; Sherman, M.; Weaver, S.C.; Lipkin, W.I. Genome-scale phylogeny of the alphavirus genus suggests a marine origin. J. Virol. 2012, 86, 2729–2738. [Google Scholar] [CrossRef] [PubMed]
- Partidos, C.D.; Paykel, J.; Weger, J.; Borland, E.M.; Powers, A.M.; Seymour, R.; Weaver, S.C.; Stinchcomb, D.T.; Osorio, J.E. Cross-protective immunity against o’nyong-nyong virus afforded by a novel recombinant chikungunya vaccine. Vaccine 2012, 30, 4638–4643. [Google Scholar] [CrossRef]
- Prat, C.M.; Flusin, O.; Panella, A.; Tenebray, B.; Lanciotti, R.; Leparc-Goffart, I. Evaluation of commercially available serologic diagnostic tests for chikungunya virus. Emerg. Infect. Dis. 2014, 20, 2129–2132. [Google Scholar] [CrossRef]
- Smith, J.L.; Pugh, C.L.; Cisney, E.D.; Keasey, S.L.; Guevara, C.; Ampuero, J.S.; Comach, G.; Gomez, D.; Ochoa-Diaz, M.; Hontz, R.D.; et al. Human Antibody Responses to Emerging Mayaro Virus and Cocirculating Alphavirus Infections Examined by Using Structural Proteins from Nine New and Old World Lineages. mSphere 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Kam, Y.-W.; Pok, K.-Y.; Eng, K.E.; Tan, L.-K.; Kaur, S.; Lee, W.W.L.; Leo, Ye.; Ng, Le.; Ng, L.F.P. Sero-prevalence and cross-reactivity of chikungunya virus specific anti-E2EP3 antibodies in arbovirus-infected patients. PLoS Negl. Trop. Dis. 2015, 9, e3445. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Konig, R.; Niedrig, M.; Emmerich, P.; Schnierle, B.S. A neutralization assay for chikungunya virus infections in a multiplex format. J. Virol. Methods 2014, 201, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Siegert, S.; Thaler, S.; Wagner, R.; Schnierle, B.S. Assessment of HIV-1 entry inhibitors by MLV/HIV-1 pseudotyped vectors. AIDS Res. Ther. 2005, 2, 7. [Google Scholar] [CrossRef] [PubMed]
- Seaman, M.S.; Janes, H.; Hawkins, N.; Grandpre, L.E.; Devoy, C.; Giri, A.; Coffey, R.T.; Harris, L.; Wood, B.; Daniels, M.G.; et al. Tiered categorization of a diverse panel of HIV-1 Env pseudoviruses for assessment of neutralizing antibodies. J. Virol. 2010, 84, 1439–1452. [Google Scholar] [CrossRef] [PubMed]
- Adam, A.; Seidahmed, O.M.E.; Weber, C.; Schnierle, B.; Schmidt-Chanasit, J.; Reiche, S.; Jassoy, C. Low Seroprevalence Indicates Vulnerability of Eastern and Central Sudan to Infection with Chikungunya Virus. Vector Borne Zoonotic Dis. 2016, 16, 290–291. [Google Scholar] [CrossRef] [PubMed]
- Kahl, C.A.; Marsh, J.; Fyffe, J.; Sanders, D.A.; Cornetta, K. Human Immunodeficiency Virus Type 1-Derived Lentivirus Vectors Pseudotyped with Envelope Glycoproteins Derived from Ross River Virus and Semliki Forest Virus. J. Virol. 2004, 78, 1421–1430. [Google Scholar] [CrossRef]
- Salvador, B.; Zhou, Y.; Michault, A.; Muench, M.O.; Simmons, G. Characterization of Chikungunya pseudotyped viruses: Identification of refractory cell lines and demonstration of cellular tropism differences mediated by mutations in E1 glycoprotein. Virology 2009, 393, 33–41. [Google Scholar] [CrossRef]
- Kishishita, N.; Takeda, N.; Anuegoonpipat, A.; Anantapreecha, S. Development of a pseudotyped-lentiviral-vector-based neutralization assay for chikungunya virus infection. J. Clin. Microbiol. 2013, 51, 1389–1395. [Google Scholar] [CrossRef]
- Theilacker, C.; Held, J.; Allering, L.; Emmerich, P.; Schmidt-Chanasit, J.; Kern, W.V.; Panning, M. Prolonged polyarthralgia in a German traveller with Mayaro virus infection without inflammatory correlates. BMC Infect. Dis. 2013, 13, 369. [Google Scholar] [CrossRef]
- Dull, T.; Zufferey, R.; Kelly, M.; Mandel, R.J.; Nguyen, M.; Trono, D.; Naldini, L. A third-generation lentivirus vector with a conditional packaging system. J. Virol. 1997, 72, 8463–8471. [Google Scholar]
- Soneoka, Y.; Cannon, P.M.; Ramsdale, E.E.; Griffiths, J.C.; Romano, G.; Kingsman, S.M.; Kingsman, A.J. A transient three-plasmid expression system for the production of high titer retroviral vectors. Nucleic Acids Res. 1995, 23, 628–633. [Google Scholar] [CrossRef]
- Kahl, C.A.; Pollok, K.; Haneline, L.S.; Cornetta, K. Lentiviral vectors pseudotyped with glycoproteins from Ross River and vesicular stomatitis viruses. Variable transduction related to cell type and culture conditions. Mol. Ther. 2005, 11, 470–482. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Berberich, E.; Rhein C von Henss, L.; Hildt, E.; Schnierle, B.S. Identification of Functional Determinants in the Chikungunya Virus E2 Protein. PLoS Negl. Trop. Dis. 2017, 11, e0005318. [Google Scholar] [CrossRef] [PubMed]
- Jacups, S.P.; Whelan, P.I.; Currie, B.J. Ross River virus and Barmah Forest virus infections. A review of history, ecology, and predictive models, with implications for tropical northern Australia. Vector Borne Zoonotic Dis. 2008, 8, 283–297. [Google Scholar] [CrossRef] [PubMed]
- Update on emerging infections: News from the Centers for Disease Control and Prevention. Notes from the field: Chikungunya virus spreads in the Americas-Caribbean and South America, 2013–2014. Ann. Emerg. Med. 2014, 64, 552–553. [Google Scholar] [CrossRef] [PubMed]
- Marano, G.; Pupella, S.; Pati, I.; Masiello, F.; Franchini, M.; Vaglio, S.; Velati, C.; Liumbruno, G.M. Ten years since the last Chikungunya virus outbreak in Italy. History repeats itself. Blood Transfus. 2017, 15, 489–490. [Google Scholar] [CrossRef]
- Lum, F.-M.; Couderc, T.; Chia, B.-S.; Ong, R.-Y.; Her, Z.; Chow, A.; Leo, Ye.; Kam, Yi.; Rénia, L.; Lecuit, M.; et al. Antibody-mediated enhancement aggravates chikungunya virus infection and disease severity. Sci. Rep. 2018, 8, 1860. [Google Scholar] [CrossRef]
- Fox, J.M.; Long, F.; Edeling, M.A.; Lin, H.; van Duijl-Richter Mareike, K.S.; Fong, R.H.; Kahle, K.M.; Smit, J.M.; Jin, J.; Simmons, G.; et al. Broadly Neutralizing Alphavirus Antibodies Bind an Epitope on E2 and Inhibit Entry and Egress. Cell 2015, 163, 1095–1107. [Google Scholar] [CrossRef]
| Plasma | Detection by ELISA | IC50CHIKV | IC50MAYV | IC50MAYV/IC50CHIKV | ||
|---|---|---|---|---|---|---|
| CHIKV | DENV | ZIKA | ||||
| Naïve | negative | negative | negative | 6.2 × 10−3 | 4.2 × 10−3 | 0.68 |
| T1 | IgM (-), IgG (+) | IgM (-), IgG (+) | negative | 2.9 × 10−4 | 1.3 × 10−3 | 4.68 |
| T2 | IgM (+), IgG (+) | IgM( -), IgG (+) | negative | 4.0 × 10−4 | 4.1 × 10−3 | 10.22 |
| T3 | negative | IgM (-), IgG (+) | IgM (-), IgG (+) | 1.1 × 10−2 | 9.1 × 10−3 | 0.80 |
| T4 | negative | IgG (+) | negative | 2.3 × 10−2 | 2.2 × 10−2 | 0.95 |
| T5 | IgM (+), IgG (+) | IgM (-), IgG (+) | n.d. | 3.1 × 10−4 | 1.5 × 10−3 | 4.97 |
| T6 | IgM (+), IgG (+) | IgM (-), IgG (+) | IgM (-), IgG (+) | 3.2 × 10−4 | 1 × 10−2 | 31.58 |
| T7 | IgM (+), IgG (+) | n.d. | IgG (+) | 2.1 × 10−4 | 1.5 × 10−3 | 7.23 |
| MAYV | IgM (-), IgG (+) | n.d. | n.d. | 1.2 × 10−3 | 2.0 × 10−4 | 0.15 |
| CHIKV | IgM (+), IgG (+) | negative | negative | 1.9 × 10−4 | 6.0 × 10−4 | 3.11 |
| Sample | ||||||
|---|---|---|---|---|---|---|
| A | B | C | D | E | F | |
| RRV ELISA | IgM (-) | IgM (-) | IgM (+) | IgM (-) | IgM (-) | IgM (-) |
| IgG (+) | IgG (+) | IgG (+) | IgG (+) | IgG (-) | IgG (+) | |
| BFV ELISA | IgM (-) | IgM (-) | IgM (-) | IgM (-) | IgM (-) | IgM (-) |
| IgG (-) | IgG (-) | IgG (+) | IgG (+) | IgG (+) | IgG (-) | |
| CHIKV ELISA assay 1 | IgM (-) | IgM (-) | IgM (-) | IgM (-) | IgM (-) | IgM (-) |
| IgG (+) | IgG (+) | IgG (+) | IgG (inc.) | IgG (-) | IgG (+) | |
| CHIKV ELISA assay 2 | IgM (-) | IgM (-) | IgM (-) | IgM (-) | IgM (-) | IgM (-) |
| IgG (-) | IgG (-) | IgG (-) | IgG (-) | IgG (-) | IgG (-) | |
| MAYV ELISA | IgM (-) | IgM (-) | IgM (+) | IgM (-) | IgM (-) | IgM (-) |
| IgG (+) | IgG (+) | IgG (+) | IgG (-) | IgG (-) | IgG (+) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Henss, L.; Yue, C.; Kandler, J.; Faddy, H.M.; Simmons, G.; Panning, M.; Lewis-Ximenez, L.L.; Baylis, S.A.; Schnierle, B.S. Establishment of an Alphavirus-Specific Neutralization Assay to Distinguish Infections with Different Members of the Semliki Forest Complex. Viruses 2019, 11, 82. https://doi.org/10.3390/v11010082
Henss L, Yue C, Kandler J, Faddy HM, Simmons G, Panning M, Lewis-Ximenez LL, Baylis SA, Schnierle BS. Establishment of an Alphavirus-Specific Neutralization Assay to Distinguish Infections with Different Members of the Semliki Forest Complex. Viruses. 2019; 11(1):82. https://doi.org/10.3390/v11010082
Chicago/Turabian StyleHenss, Lisa, Constanze Yue, Joshua Kandler, Helen M. Faddy, Graham Simmons, Marcus Panning, Lia Laura Lewis-Ximenez, Sally A. Baylis, and Barbara S. Schnierle. 2019. "Establishment of an Alphavirus-Specific Neutralization Assay to Distinguish Infections with Different Members of the Semliki Forest Complex" Viruses 11, no. 1: 82. https://doi.org/10.3390/v11010082
APA StyleHenss, L., Yue, C., Kandler, J., Faddy, H. M., Simmons, G., Panning, M., Lewis-Ximenez, L. L., Baylis, S. A., & Schnierle, B. S. (2019). Establishment of an Alphavirus-Specific Neutralization Assay to Distinguish Infections with Different Members of the Semliki Forest Complex. Viruses, 11(1), 82. https://doi.org/10.3390/v11010082





