Heartland Virus Epidemiology, Vector Association, and Disease Potential
Abstract
1. Introduction
2. Virology, Epidemiology, and Clinical Disease
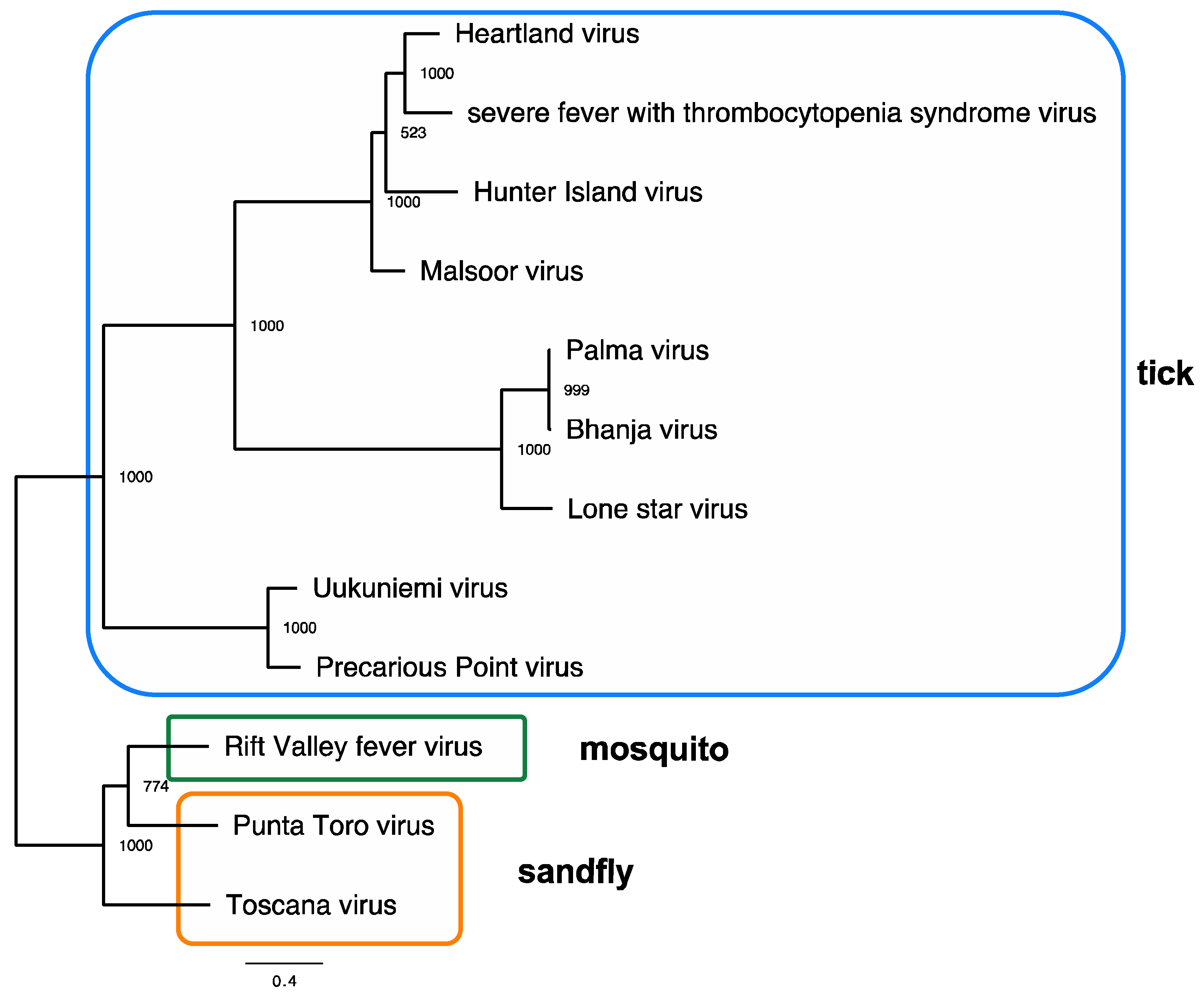
2.1. Virology
2.2. Surveillance and Testing for HRTV Disease
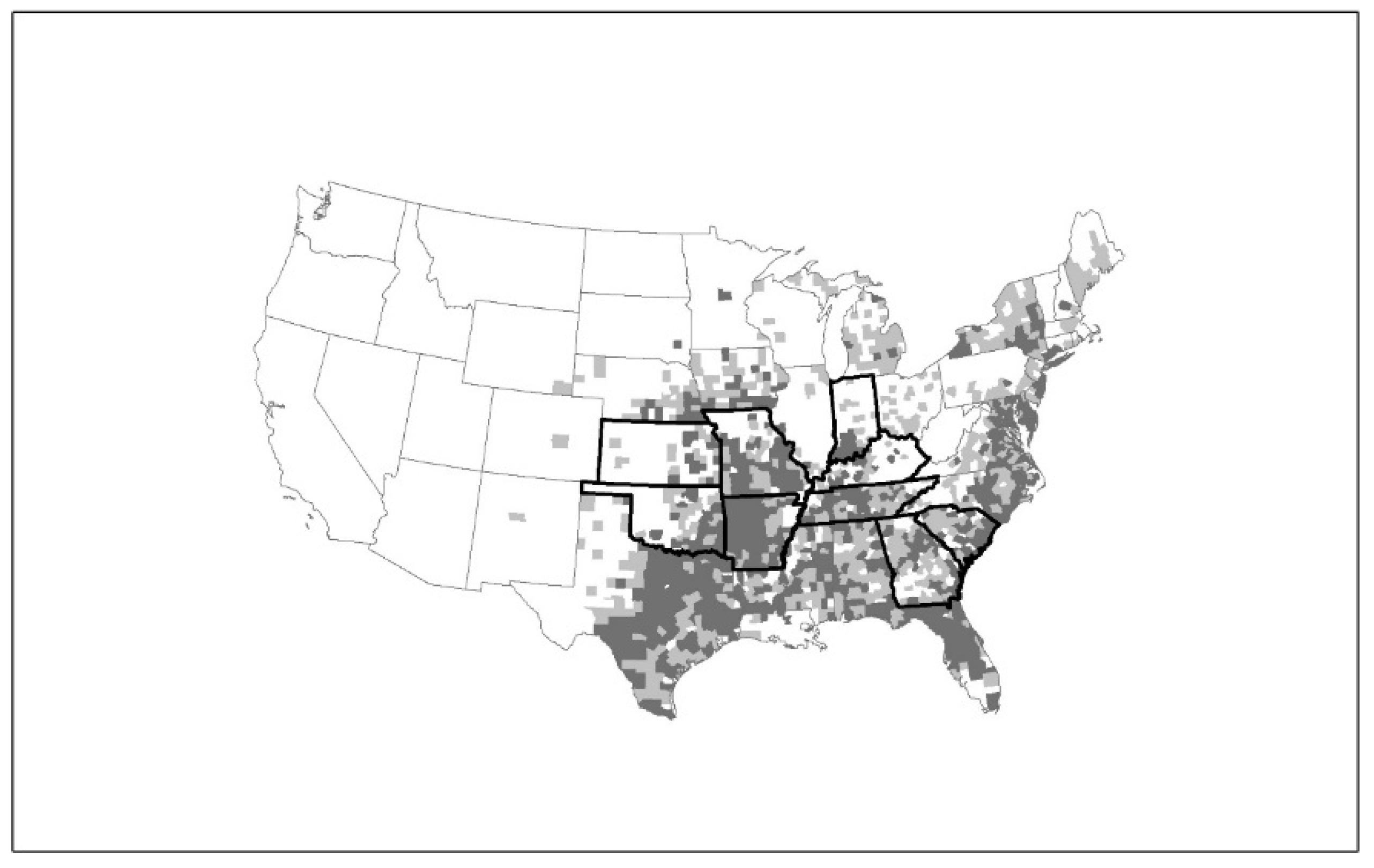
2.3. Epidemiology of HRTV
2.4. Clinical Disease
2.5. Clinical Management
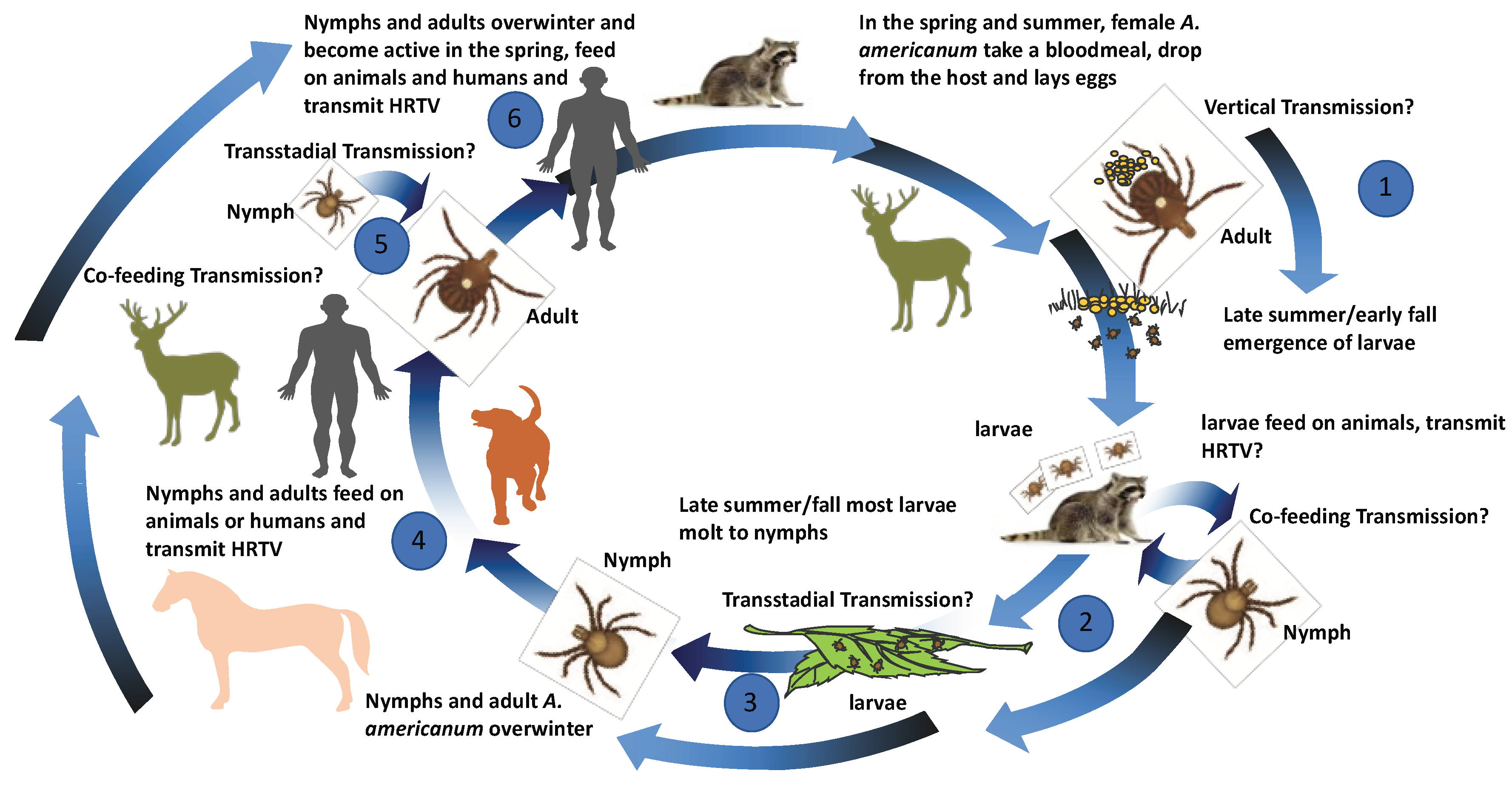
3. Ecological Assessments of HRTV Transmission
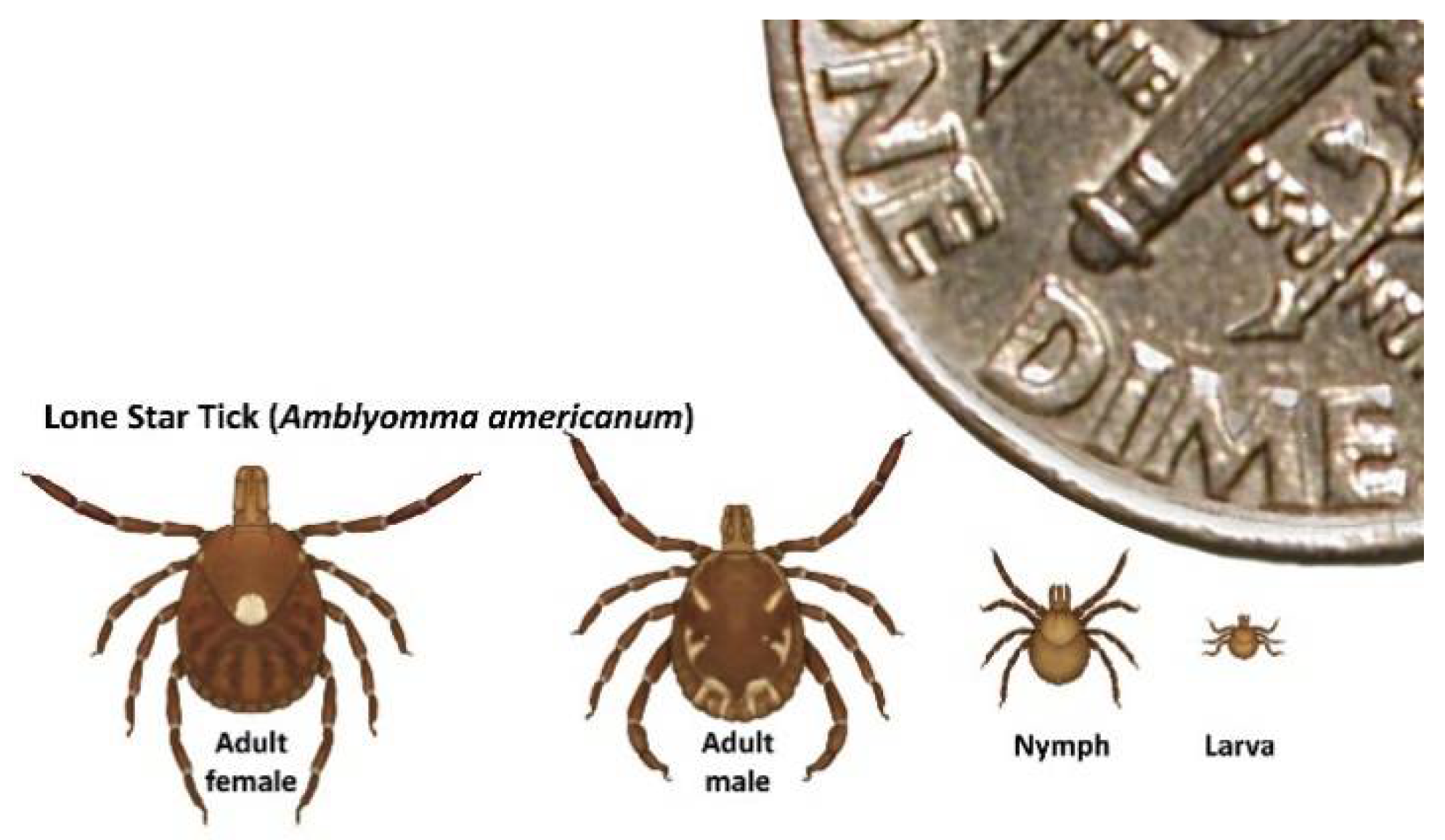
3.1. Detection of HRTV in Field-Collected Ticks
3.2. Experimental Vector Competence Studies with HRTV and A. americanum
3.3. Phleboviral/HRTV-Neutralizing Antibodies in Field-Collected Animals
4. Host Competence and Pathogenesis Assessments
Animal Model Assessments
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- McMullan, L.K.; Folk, S.M.; Kelly, A.J.; MacNeil, A.; Goldsmith, C.S.; Metcalfe, M.G.; Batten, B.C.; Albarino, C.G.; Zaki, S.R.; Rollin, P.E.; et al. A new phlebovirus associated with severe febrile illness in Missouri. N. Engl. J. Med. 2012, 367, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.J.; Liang, M.F.; Zhang, S.Y.; Liu, Y.; Li, J.D.; Sun, Y.L.; Zhang, L.; Zhang, Q.F.; Popov, V.L.; Li, C.; et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N. Engl. J. Med. 2011, 364, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Yamagishi, T.; Shimada, T.; Matsui, T.; Shimojima, M.; Saijo, M.; Oishi, K.; SFTS Epidemiological Research Group-Japan. Epidemiological and Clinical Features of Severe Fever with Thrombocytopenia Syndrome in Japan, 2013–2014. PLoS ONE 2016, 11, e0165207. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.J.; Park, S.W.; Bae, I.G.; Kim, S.H.; Ryu, S.Y.; Kim, H.A.; Jang, H.C.; Hur, J.; Jun, J.B.; Jung, Y.; et al. Severe Fever with Thrombocytopenia Syndrome in South Korea, 2013–2015. PLoS Negl. Trop. Dis. 2016, 10, e0005264. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.J.; Lefkowitz, E.J.; King, A.M.Q.; Harrach, B.; Harrison, R.L.; Knowles, N.J.; Kropinski, A.M.; Krupovic, M.; Kuhn, J.H.; Mushegian, A.R.; et al. Changes to taxonomy and the International Code of Virus Classification and Nomenclature ratified by the International Committee on Taxonomy of Viruses (2017). Arch. Virol. 2017, 162, 2505–2538. [Google Scholar] [CrossRef] [PubMed]
- Walter, C.T.; Barr, J.N. Recent advances in the molecular and cellular biology of bunyaviruses. J. Gen. Virol. 2011, 92, 2467–2484. [Google Scholar] [CrossRef] [PubMed]
- Swei, A.; Russell, B.J.; Naccache, S.N.; Kabre, B.; Veeraraghavan, N.; Pilgard, M.A.; Johnson, B.J.; Chiu, C.Y. The Genome Sequence of Lone Star Virus, a Highly Divergent Bunyavirus Found in the Amblyomma americanum Tick. PLoS ONE 2013, 8, e62083. [Google Scholar] [CrossRef] [PubMed]
- Matsuno, K.; Weisend, C.; Kajihara, M.; Matysiak, C.; Williamson, B.N.; Simuunza, M.; Mweene, A.S.; Takada, A.; Tesh, R.B.; Ebihara, H. Comprehensive molecular detection of tick-borne phleboviruses leads to the retrospective identification of taxonomically unassigned bunyaviruses and the discovery of a novel member of the genus Phlebovirus. J. Virol. 2015, 89, 594–604. [Google Scholar] [CrossRef] [PubMed]
- Mourya, D.T.; Yadav, P.D.; Basu, A.; Shete, A.; Patil, D.Y.; Zawar, D.; Majumdar, T.D.; Kokate, P.; Sarkale, P.; Raut, C.G.; et al. Malsoor virus, a novel bat phlebovirus, is closely related to Severe Fever with Thrombocytopenia Syndrome virus and Heartland virus. J. Virol. 2014, 88, 3605–3609. [Google Scholar] [CrossRef] [PubMed]
- Gauci, P.J.; McAllister, J.; Mitchell, I.R.; St George, T.D.; Cybinski, D.H.; Davis, S.S.; Gubala, A.J. Hunter Island group phlebovirus in ticks, Australia. Emerg. Infect. Dis. 2015, 21, 2246–2248. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Selleck, P.; Yu, M.; Ha, W.; Rootes, C.; Gales, R.; Wise, T.; Crameri, S.; Chen, H.; Broz, I.; et al. Novel phlebovirus with zoonotic potential isolated from ticks, Australia. Emerg. Infect. Dis. 2014, 20, 1040–1043. [Google Scholar] [CrossRef] [PubMed]
- Bosco-Lauth, A.M.; Calvert, A.E.; Root, J.J.; Gidlewski, T.; Bird, B.H.; Bowen, R.A.; Muehlenbachs, A.; Zaki, S.R.; Brault, A.C. Vertebrate Host Susceptibility to Heartland Virus. Emerg. Infect. Dis. 2016, 22, 2070–2077. [Google Scholar] [CrossRef] [PubMed]
- Calvert, A.E.; Brault, A.C. Development and Characterization of Monoclonal Antibodies Directed Against the Nucleoprotein of Heartland Virus. Am. J. Trop. Med. Hyg. 2015, 93, 1338–1340. [Google Scholar] [CrossRef] [PubMed]
- Martin-Folgar, R.; Lorenzo, G.; Boshra, H.; Iglesias, J.; Mateos, F.; Borrego, B.; Brun, A. Development and characterization of monoclonal antibodies against Rift Valley fever virus nucleocapsid protein generated by DNA immunization. MAbs 2010, 2, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Fafetine, J.M.; Tijhaar, E.; Paweska, J.T.; Neves, L.C.; Hendriks, J.; Swanepoel, R.; Coetzer, J.A.; Egberink, H.F.; Rutten, V.P. Cloning and expression of Rift Valley fever virus nucleocapsid (N) protein and evaluation of a N-protein based indirect ELISA for the detection of specific IgG and IgM antibodies in domestic ruminants. Vet. Microbiol. 2007, 121, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Paweska, J.T.; Mortimer, E.; Leman, P.A.; Swanepoel, R. An inhibition enzyme-linked immunosorbent assay for the detection of antibody to Rift Valley fever virus in humans, domestic and wild ruminants. J. Virol. Methods 2005, 127, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Paweska, J.T.; van Vuren, P.J.; Kemp, A.; Buss, P.; Bengis, R.G.; Gakuya, F.; Breiman, R.F.; Njenga, M.K.; Swanepoel, R. Recombinant nucleocapsid-based ELISA for detection of IgG antibody to Rift Valley fever virus in African buffalo. Vet. Microbiol. 2008, 127, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Magurano, F.; Nicoletti, L. Humoral response in Toscana virus acute neurologic disease investigated by viral-protein-specific immunoassays. Clin. Diagn. Lab. Immunol. 1999, 6, 55–60. [Google Scholar] [PubMed]
- Schwarz, T.F.; Gilch, S.; Pauli, C.; Jager, G. Immunoblot detection of antibodies to Toscana virus. J. Med. Virol. 1996, 49, 83–86. [Google Scholar] [CrossRef]
- Swanepoel, R.; Struthers, J.K.; Erasmus, M.J.; Shepherd, S.P.; McGillivray, G.M.; Erasmus, B.J.; Barnard, B.J. Comparison of techniques for demonstrating antibodies to Rift Valley fever virus. J. Hyg. (Lond.) 1986, 97, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Zhang, L.; Zhang, W.; Chi, Y.; Zeng, X.; Li, X.; Qi, X.; Jin, Q.; Zhang, X.; Huang, M.; et al. Human antibody neutralizes Severe Fever with Thrombocytopenia Syndrome virus, an emerging hemorrhagic fever virus. Clin. Vaccine Immunol. 2013, 20, 1426–1432. [Google Scholar] [CrossRef] [PubMed]
- Mousavi-Jazi, M.; Karlberg, H.; Papa, A.; Christova, I.; Mirazimi, A. Healthy individuals’ immune response to the Bulgarian Crimean-Congo hemorrhagic fever virus vaccine. Vaccine 2012, 30, 6225–6229. [Google Scholar] [CrossRef] [PubMed]
- Zivcec, M.; Safronetz, D.; Scott, D.; Robertson, S.; Ebihara, H.; Feldmann, H. Lethal Crimean-Congo hemorrhagic fever virus infection in interferon alpha/beta receptor knockout mice is associated with high viral loads, proinflammatory responses, and coagulopathy. J. Infect. Dis. 2013, 207, 1909–1921. [Google Scholar] [CrossRef] [PubMed]
- Bridgen, A.; Weber, F.; Fazakerley, J.K.; Elliott, R.M. Bunyamwera bunyavirus nonstructural protein NSs is a nonessential gene product that contributes to viral pathogenesis. Proc. Natl. Acad. Sci. USA 2001, 98, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, T.; Peters, C.J.; Makino, S. Rift valley fever virus nonstructural protein NSs promotes viral RNA replication and transcription in a minigenome system. J. Virol. 2005, 79, 5606–5615. [Google Scholar] [CrossRef] [PubMed]
- Bouloy, M.; Janzen, C.; Vialat, P.; Khun, H.; Pavlovic, J.; Huerre, M.; Haller, O. Genetic evidence for an interferon-antagonistic function of rift valley fever virus nonstructural protein NSs. J. Virol. 2001, 75, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Nfon, C.K.; Marszal, P.; Zhang, S.; Weingartl, H.M. Innate immune response to Rift Valley fever virus in goats. PLoS Negl. Trop. Dis. 2012, 6, e1623. [Google Scholar] [CrossRef] [PubMed]
- Pastula, D.M.; Turabelidze, G.; Yates, K.F.; Jones, T.F.; Lambert, A.J.; Panella, A.J.; Kosoy, O.I.; Velez, J.O.; Fisher, M.; Staples, E.; et al. Prevention, Notes from the field: Heartland virus disease—United States, 2012–2013. Morb. Mortal. Wkly. Rep. 2014, 63, 270–271. [Google Scholar]
- Heartland Virus: Statistics & Maps. Available online: https://www.cdc.gov/heartland-virus/statistics/index.html (accessed on 6 July 2018).
- Geographic Distribution of Ticks That Bite Humans. Available online: https://www.cdc.gov/ticks/geographic_distribution.html (accessed on 6 July 2018).
- Chen, Y.; Jia, B.; Huang, R.; Yan, X.; Xiong, Y.; Yong, L.; Chao, W. Occupational Severe Fever With Thrombocytopenia Syndrome Following Needle-Stick Injury. Infect. Control. Hosp. Epidemiol. 2017, 38, 760–762. [Google Scholar] [CrossRef] [PubMed]
- Gai, Z.T.; Zhang, Y.; Liang, M.F.; Jin, C.; Zhang, S.; Zhu, C.B.; Li, C.; Li, X.Y.; Zhang, Q.F.; Bian, P.F.; et al. Clinical progress and risk factors for death in severe fever with thrombocytopenia syndrome patients. J. Infect. Dis. 2012, 206, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Wu, W.; Wang, H.; Du, Y.; Liu, L.; Kang, K.; Huang, X.; Ma, H.; Mu, F.; Zhang, S.; et al. Human-to-human transmission of severe fever with thrombocytopenia syndrome bunyavirus through contact with infectious blood. J. Infect. Dis. 2013, 207, 736–739. [Google Scholar] [CrossRef] [PubMed]
- Gai, Z.; Liang, M.; Zhang, Y.; Zhang, S.; Jin, C.; Wang, S.W.; Sun, L.; Zhou, N.; Zhang, Q.; Sun, Y.; et al. Person-to-person transmission of severe fever with thrombocytopenia syndrome bunyavirus through blood contact. Clin. Infect. Dis. 2012, 54, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Springer, Y.P.; Jarnevich, C.S.; Barnett, D.T.; Monaghan, A.J.; Eisen, R.J. Modeling the Present and Future Geographic Distribution of the Lone Star Tick, Amblyomma americanum (Ixodida: Ixodidae), in the Continental United States. Am. J. Trop. Med. Hyg. 2015, 93, 875–890. [Google Scholar] [CrossRef] [PubMed]
- Childs, J.E.; Paddock, C.D. The ascendancy of Amblyomma americanum as a vector of pathogens affecting humans in the United States. Annu. Rev. Entomol. 2003, 48, 307–337. [Google Scholar] [CrossRef] [PubMed]
- Carlson, A.L.; Pastula, D.M.; Lambert, A.J.; Staples, J.E.; Muehlenbachs, A.; Turabelidze, G.; Eby, C.S.; Keller, J.; Hess, B.; Buller, R.S.; et al. Heartland Virus and Hemophagocytic Lymphohistiocytosis in Immunocompromised Patient, Missouri, USA. Emerg. Infect. Dis. 2018, 24, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Fill, M.A.; Compton, M.L.; McDonald, E.C.; Moncayo, A.C.; Dunn, J.R.; Schaffner, W.; Bhatnagar, J.; Zaki, S.R.; Jones, T.F.; Shieh, W.J. Novel Clinical and Pathologic Findings in a Heartland Virus-Associated Death. Clin. Infect. Dis. 2017, 64, 510–512. [Google Scholar] [PubMed]
- Muehlenbachs, A.; Fata, C.R.; Lambert, A.J.; Paddock, C.D.; Velez, J.O.; Blau, D.M.; Staples, J.E.; Karlekar, M.B.; Bhatnagar, J.; Nasci, R.S.; et al. Heartland virus-associated death in Tennessee. Clin. Infect. Dis. 2014, 59, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.S.; Kim, M.; Lee, J.O.; Kim, H.; Kim, E.S.; Park, K.U.; Kim, H.B.; Song, K.H. Hemophagocytic lymphohistiocytosis associated with SFTS virus infection: A case report with literature review. Medicine 2016, 95, e4476. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.; Kim, K.H.; Lee, S.J.; Park, S.H.; Kim, I.S.; Lee, E.Y.; Yi, J. Bone marrow findings in severe fever with thrombocytopenia syndrome: Prominent haemophagocytosis and its implication in haemophagocytic lymphohistiocytosis. J. Clin. Pathol. 2016, 69, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Liu, L.; Huang, X.; Ma, H.; Zhang, Y.; Du, Y.; Wang, P.; Tang, X.; Wang, H.; Kang, K.; et al. Metagenomic analysis of fever, thrombocytopenia and leukopenia syndrome (FTLS) in Henan Province, China: Discovery of a new bunyavirus. PLoS Pathog. 2011, 7, e1002369. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; He, B.; Huang, S.Y.; Wei, F.; Zhu, X.Q. Severe fever with thrombocytopenia syndrome, an emerging tick-borne zoonosis. Lancet Infect. Dis. 2014, 14, 763–772. [Google Scholar] [CrossRef]
- Liu, W.; Lu, Q.B.; Cui, N.; Li, H.; Wang, L.Y.; Liu, K.; Yang, Z.D.; Wang, B.J.; Wang, H.Y.; Zhang, Y.Y.; et al. Case-fatality ratio and effectiveness of ribavirin therapy among hospitalized patients in china who had severe fever with thrombocytopenia syndrome. Clin. Infect. Dis. 2013, 57, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Lu, Q.B.; Xing, B.; Zhang, S.F.; Liu, K.; Du, J.; Li, X.K.; Cui, N.; Yang, Z.D.; Wang, L.Y.; et al. Epidemiological and clinical features of laboratory-diagnosed severe fever with thrombocytopenia syndrome in China, 2011–17: A prospective observational study. Lancet Infect. Dis. 2018, in press. [Google Scholar] [CrossRef]
- Savage, H.M.; Godsey, M.S., Jr.; Lambert, A.; Panella, N.A.; Burkhalter, K.L.; Harmon, J.R.; Lash, R.R.; Ashley, D.C.; Nicholson, W.L. First detection of Heartland virus (Bunyaviridae: Phlebovirus) from field collected arthropods. Am. J. Trop. Med. Hyg. 2013, 89, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Savage, H.M.; Godsey, M.S., Jr.; Panella, N.A.; Burkhalter, K.L.; Ashley, D.C.; Lash, R.R.; Ramsay, B.; Patterson, T.; Nicholson, W.L. Surveillance for Heartland Virus (Bunyaviridae: Phlebovirus) in Missouri During 2013: First Detection of Virus in Adults of Amblyomma americanum (Acari: Ixodidae). J. Med. Entomol. 2016, 53, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Savage, H.M.; Godsey, M.S., Jr.; Panella, N.A.; Burkhalter, K.L.; Manford, J.; Trevino-Garrison, I.C.; Straily, A.; Wilson, S.; Bowen, J.; Raghavan, R.K. Surveillance for Tick-Borne Viruses Near the Location of a Fatal Human Case of Bourbon Virus (Family Orthomyxoviridae: Genus Thogotovirus) in Eastern Kansas, 2015. J. Med. Entomol. 2018, 55, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.M.; Zhao, L.; Wen, H.L.; Zhang, Z.T.; Liu, J.W.; Fang, L.Z.; Xue, Z.F.; Ma, D.Q.; Zhang, X.S.; Ding, S.J.; et al. Haemaphysalis longicornis Ticks as Reservoir and Vector of Severe Fever with Thrombocytopenia Syndrome Virus in China. Emerg. Infect. Dis. 2015, 21, 1770–1776. [Google Scholar] [CrossRef] [PubMed]
- Godsey, M.S., Jr.; Savage, H.M.; Burkhalter, K.L.; Bosco-Lauth, A.M.; Delorey, M.J. Transmission of Heartland Virus (Bunyaviridae: Phlebovirus) by Experimentally Infected Amblyomma americanum (Acari: Ixodidae). J. Med. Entomol. 2016, 53, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Bao, C.; Zhou, M.; Hu, J.; Tang, F.; Guo, X.; Jiao, Y.; Zhang, W.; Luo, P.; Li, L.; et al. Seroprevalence and risk factors for severe fever with thrombocytopenia syndrome virus infection in Jiangsu Province, China, 2011. Am. J. Trop. Med. Hyg. 2014, 90, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.L.; Wang, X.J.; Li, J.D.; Ding, S.J.; Zhang, Q.F.; Qu, J.; Zhang, S.; Li, C.; Wu, W.; Jiang, M.; et al. Isolation, identification and characterization of SFTS bunyavirus from ticks collected on the surface of domestic animals. Bing Du Xue Bao 2012, 28, 252–257. [Google Scholar] [PubMed]
- Allan, B.F.; Goessling, L.S.; Storch, G.A.; Thach, R.E. Blood meal analysis to identify reservoir hosts for Amblyomma americanum ticks. Emerg. Infect. Dis. 2010, 16, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Mitzel, D.N.; Wolfinbarger, J.B.; Long, R.D.; Masnick, M.; Best, S.M.; Bloom, M.E. Tick-borne flavivirus infection in Ixodes scapularis larvae: Development of a novel method for synchronous viral infection of ticks. Virology 2007, 365, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Troughton, D.R.; Levin, M.L. Life cycles of seven ixodid tick species (Acari: Ixodidae) under standardized laboratory conditions. J. Med. Entomol. 2007, 44, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.D.; Davies, C.R.; Steele, G.M.; Nuttall, P.A. A novel mode of arbovirus transmission involving a nonviremic host. Science 1987, 237, 775–777. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Yanagihara, R.; Gibbs, C.J., Jr.; Amyx, H.L.; Gajdusek, D.C. Differential susceptibility and resistance of immunocompetent and immunodeficient mice to fatal Hantaan virus infection. Arch. Virol. 1985, 86, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Bosco-Lauth, A.M.; Panella, N.A.; Root, J.J.; Gidlewski, T.; Lash, R.R.; Harmon, J.R.; Burkhalter, K.L.; Godsey, M.S.; Savage, H.M.; Nicholson, W.L.; et al. Serological investigation of Heartland virus (Bunyaviridae: Phlebovirus) exposure in wild and domestic animals adjacent to human case sites in Missouri 2012–2013. Am. J. Trop. Med. Hyg. 2015, 92, 1163–1167. [Google Scholar] [CrossRef] [PubMed]
- Riemersma, K.K.; Komar, N. Heartland virus neutralizing antibodies in vertebrate wildlife, United States, 2009–2014. Emerg. Infect. Dis 2015, 21, 1830–1833. [Google Scholar] [CrossRef] [PubMed]
- Xing, Z.; Schefers, J.; Schwabenlander, M.; Jiao, Y.; Liang, M.; Qi, X.; Li, C.; Goyal, S.; Cardona, C.J.; Wu, X.; et al. Novel Bunyavirus in Domestic and Captive Farmed Animals, Minnesota, USA. Emerg. Infect. Dis. 2013, 19, 1487–1489. [Google Scholar] [CrossRef] [PubMed]
- Tugwell, P.; Lancaster, J.L. Results of a tick-host study in northwest Arkansas. J. Kans. Entomol. Soc. 1962, 35, 202–211. [Google Scholar]
- Zhao, L.; Zhai, S.; Wen, H.; Cui, F.; Chi, Y.; Wang, L.; Xue, F.; Wang, Q.; Wang, Z.; Zhang, S.; et al. Severe fever with thrombocytopenia syndrome virus, Shandong Province, China. Emerg. Infect. Dis. 2012, 18, 963–965. [Google Scholar] [CrossRef] [PubMed]
- Cui, F.; Cao, H.X.; Wang, L.; Zhang, S.F.; Ding, S.J.; Yu, X.J.; Yu, H. Clinical and epidemiological study on severe fever with thrombocytopenia syndrome in Yiyuan County, Shandong Province, China. Am. J. Trop. Med. Hyg. 2013, 88, 510–512. [Google Scholar] [CrossRef] [PubMed]
- Japanese Woman Dies from Tick Disease after Cat Bite. Available online: https://www.bbc.com/news/world-asia-40713172 (accessed on 6 July 2018).
- Ding, F.; Guan, X.H.; Kang, K.; Ding, S.J.; Huang, L.Y.; Xing, X.S.; Sha, S.; Liu, L.; Wang, X.J.; Zhang, X.M.; et al. Risk factors for bunyavirus-associated severe Fever with thrombocytopenia syndrome, china. PLoS Negl. Trop. Dis. 2014, 8, e3267. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Kang, J.G.; Oh, S.S.; Chae, J.B.; Cho, Y.K.; Cho, Y.S.; Lee, H.; Chae, J.S. Molecular detection of severe fever with thrombocytopenia syndrome virus (SFTSV) in feral cats from Seoul, Korea. Ticks Tick-Borne Dis. 2017, 8, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Dahlgren, F.S.; Paddock, C.D.; Springer, Y.P.; Eisen, R.J.; Behravesh, C.B. Expanding Range of Amblyomma americanum and Simultaneous Changes in the Epidemiology of Spotted Fever Group Rickettsiosis in the United States. Am. J. Trop. Med. Hyg. 2016, 94, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Qi, X.; Liu, D.; Zeng, X.; Han, Y.; Guo, X.; Shi, Z.; Wang, H.; Zhou, M. Experimental and natural infections of goats with Severe Fever with Thrombocytopenia Syndrome virus: Evidence for ticks as viral vector. PLoS Negl. Trop. Dis. 2015, 9, e0004092. [Google Scholar] [CrossRef] [PubMed]
- Clarke, L.L.; Ruder, M.G.; Mead, D.; Howerth, E.W. Experimental Infection of White-Tailed Deer (Odocoileus virginanus) with Heartland Virus. Am. J. Trop. Med. Hyg. 2018, 98, 1194–1196. [Google Scholar] [CrossRef] [PubMed]
- Matsuno, K.; Orba, Y.; Maede-White, K.; Scott, D.; Feldmann, F.; Liang, M.; Ebihara, H. Animal Models of Emerging Tick-Borne Phleboviruses: Determining Target Cells in a Lethal Model of SFTSV Infection. Front. Microbiol. 2017, 8, 104. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Liang, M.; Ning, J.; Gu, W.; Jiang, H.; Wu, W.; Zhang, F.; Li, C.; Zhang, Q.; Zhu, H.; et al. Pathogenesis of emerging Severe Fever with Thrombocytopenia Syndrome virus in C57/BL6 mouse model. Proc. Natl. Acad. Sci. USA 2012, 109, 10053–10058. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.P.; Cong, M.L.; Li, M.H.; Kang, Y.J.; Feng, Y.M.; Plyusnin, A.; Xu, J.; Zhang, Y.Z. Infection and pathogenesis of Huaiyangshan virus (a novel tick-borne bunyavirus) in laboratory rodents. J. Gen. Virol. 2012, 93, 1288–1293. [Google Scholar] [CrossRef] [PubMed]
- Qu, B.; Qi, X.; Wu, X.; Liang, M.; Li, C.; Cardona, C.J.; Xu, W.; Tang, F.; Li, Z.; Wu, B.; et al. Suppression of the Interferon and NF-kappaB Responses by Severe Fever with Thrombocytopenia Syndrome Virus. J. Virol. 2012, 86, 8388–8401. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wu, B.; Paessler, S.; Walker, D.H.; Tesh, R.B.; Yu, X.J. The pathogenesis of Severe Fever with Thrombocytopenia Syndrome virus infection in alpha/beta interferon knockout mice: Insights into the pathologic mechanisms of a new viral hemorrhagic fever. J. Virol. 2014, 88, 1781–1786. [Google Scholar] [CrossRef] [PubMed]
- Westover, J.B.; Rigas, J.D.; Van Wettere, A.J.; Li, R.; Hickerson, B.T.; Jung, K.H.; Miao, J.; Reynolds, E.S.; Conrad, B.L.; Nielson, S.; et al. Heartland virus infection in hamsters deficient in type I interferon signaling: Protracted disease course ameliorated by favipiravir. Virology 2017, 511, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Gowen, B.B.; Westover, J.B.; Miao, J.; Van Wettere, A.J.; Rigas, J.D.; Hickerson, B.T.; Jung, K.H.; Li, R.; Conrad, B.L.; Nielson, S.; et al. Modeling Severe Fever with Thrombocytopenia Syndrome Virus Infection in Golden Syrian Hamsters: Importance of STAT2 in Preventing Disease and Effective Treatment with Favipiravir. J. Virol. 2017, 91, e01942-16. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Jiang, H.; Liang, M.; Han, Y.; Gu, W.; Zhang, F.; Zhu, H.; Wu, W.; Chen, T.; Li, C.; et al. SFTS virus infection in nonhuman primates. J. Infect. Dis. 2015, 211, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Mendenhall, M.; Wong, M.H.; Skirpstunas, R.; Morrey, J.D.; Gowen, B.B. Punta Toro virus (Bunyaviridae, Phlebovirus) infection in mice: Strain differences in pathogenesis and host interferon response. Virology 2009, 395, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Pifat, D.Y.; Smith, J.F. Punta Toro virus infection of C57BL/6J mice: A model for phlebovirus-induced disease. Microb. Pathog. 1987, 3, 409–422. [Google Scholar] [CrossRef]
- Hermance, M.E.; Thangamani, S. Tick saliva enhances Powassan virus transmission to the host, influencing its dissemination and the course of disease. J. Virol. 2015, 89, 7852–7860. [Google Scholar] [CrossRef] [PubMed]
- Labuda, M.; Jones, L.D.; Williams, T.; Nuttall, P.A. Enhancement of tick-borne encephalitis virus transmission by tick salivary gland extracts. Med. Vet. Entomol. 1993, 7, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.D.; Hodgson, E.; Nuttall, P.A. Enhancement of virus transmission by tick salivary glands. J. Gen. Virol. 1989, 70, 1895–1898. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, L.; Du, J.; Cui, X.M.; Li, H.; Tang, F.; Zhang, P.H.; Hu, J.G.; Tong, Y.G.; Feng, Z.C.; Liu, W. Identification of tick-borne pathogen diversity by metagenomic analysis in Haemaphysalis longicornis from Xinyang, China. Infect. Dis. Poverty 2018, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Exotic Tick Species Confirmed to Have Overwintered in New Jersey. Available online: https://www.state.nj.us/agriculture/news/press/2018/approved/press180420.html (accessed on 6 July 2018).
| Model | Heartland Virus | References | Severe Fever with Thrombocytopenia Virus | References |
|---|---|---|---|---|
| immunocompetent mice | C57bl/6 (i.p. inoculation): no viremia, morbidity/mortality, or clinical signs, seroconversion CD1 (i.c. inoculation): no morbidity/mortality | [12,70] | C57bl/6, BalB/C (i.v., i.m., i.p. inoculation): viremia, lesions, leukopenia through all routes and replication in spleen; thrombocytopenia (i.v. and i.m. routes); no significant morbidity or blood counts in BalB/C, but elicited liver/kidney lesions Newborn KM mice and rats (i.p. and i.c.): lethality and viremia and liver lesions, but adults were refractory CD1 newborn mice (i.c. inoculation): passage adaptation of viruses for consistent morbidity/mortality | [70,71,72,73] |
| immunodeficient mice | AG129 (Type I/II IFNr KO; i.p. inoculation): high viremia and dose-dependent mortality | [12] | IFNAR−/− (s.c. inoculation): detectable SFTSV RNA in serum with morbidity/mortality, reticular tropism in spleen | [70,73,74] |
| hamsters | Syrian: no detectable viremia or serum RNA, seroconversion; STAT2 KO (s.c. inoculation) showed viremia and morbidity/mortality | [12,75] | Syrian (i.v., i.m., i.p., i.c. inoculation): no significant morbidity or blood count alterations Newborn (i.c. inoculation) showed no morbidity/mortality and adult (i.p. inoculation) failed to generate detectable viremias and showed no outward signs of disease STAT2 KO (s.c. inoculation) showed viremia and morbidity/mortality | [7,73,76] |
| rabbits | New Zealand white (s.c. inoculation): no detectable viremia or serum RNA, low seroconversion rates; (tick feed): no detectable serum RNA with seroconversion | [12,50] | ||
| raccoons | Field-collected (s.c. inoculation): no detectable viremia or serum RNA, low seroconversion | [12] | ||
| ungulates | White-tailed deer (i.d. inoculation): no detectable viremia or clinical disease with modest seroconversion; goats (s.c. inoculation): no detectable viremia or serum RNA, low seroconversion | [12,69] | Goats (s.c. inoculation): transient viremia with seroconversion without outward signs of disease Goats (field-tick exposure): transient viremia with seroconversion without outward signs of disease | [68] |
| avian | chickens (s.c. inoculation): no detectable viremia or serum RNA, no detectable seroconversion | [12] | ||
| non-human primates | Cynomolgus macaques (s.c. inoculated): no detectable viremia, no clinical signs or lesions | [70] | Rhesus macaques (i.m. inoculated): mild symptoms, viremia and seroconversion detected, thrombocytopenia, leukocytopenia, liver/kidney lesions Cynomolgus macaques (s.c. inoculated): no detectable viremia or lesions, thrombocytopenia in one animal | [70,77] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brault, A.C.; Savage, H.M.; Duggal, N.K.; Eisen, R.J.; Staples, J.E. Heartland Virus Epidemiology, Vector Association, and Disease Potential. Viruses 2018, 10, 498. https://doi.org/10.3390/v10090498
Brault AC, Savage HM, Duggal NK, Eisen RJ, Staples JE. Heartland Virus Epidemiology, Vector Association, and Disease Potential. Viruses. 2018; 10(9):498. https://doi.org/10.3390/v10090498
Chicago/Turabian StyleBrault, Aaron C., Harry M. Savage, Nisha K. Duggal, Rebecca J. Eisen, and J. Erin Staples. 2018. "Heartland Virus Epidemiology, Vector Association, and Disease Potential" Viruses 10, no. 9: 498. https://doi.org/10.3390/v10090498
APA StyleBrault, A. C., Savage, H. M., Duggal, N. K., Eisen, R. J., & Staples, J. E. (2018). Heartland Virus Epidemiology, Vector Association, and Disease Potential. Viruses, 10(9), 498. https://doi.org/10.3390/v10090498





