Zoonotic Potential of Influenza A Viruses: A Comprehensive Overview
Abstract
1. Introduction
2. Classification of Influenza A Viruses (IAVs)
2.1. Human versus Avian IAVs
2.2. Highly versus Low Pathogenic Avian Influenza Viruses (HPAIV vs. LPAIV)
3. Evolution and Epidemiology of IAV
3.1. H5N1 (HPAIV)
3.2. H5N6 (HPAIV)
3.3. H6N1 (LPAIV)
3.4. H7N2 and H7N3 (LPAIV and HPAIV)
3.5. H7N4 (HPAIV)
3.6. H7N7 (HPAIV)
3.7. H7N9 (LPAIV)
3.8. H9N2 (LPAIV)
3.9. H10N7 (LPAIV)
3.10. H10N8 (LPAIV)
4. Sources of Human Infections with Zoonotic IAV
5. Viral Determinants for Zoonotic Potential of IAV
5.1. The Viral Polymerase Complex and NP Protein
5.2. Viral Surface Glycoproteins (HA and NA)
5.3. Non-Structural Protein 1 (NS1)
6. Control and Prevention of IAV
6.1. Vaccination against IAV
6.2. Antivirals against IAV
6.2.1. Adamantanes
6.2.2. Neuraminidase Inhibitors
6.2.3. Membrane Fusion Inhibitors
6.2.4. RNA-Dependent RNA Polymerase (RdRp) Inhibitors
6.3. Anti-Influenza Drugs in Late-Phase Clinical Trials
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hause, B.M.; Collin, E.A.; Liu, R.; Huang, B.; Sheng, Z.; Lu, W.; Wang, D.; Nelson, E.A.; Li, F. Characterization of a novel influenza virus in cattle and Swine: Proposal for a new genus in the orthomyxoviridae family. mBio 2014, 5, e00031-14. [Google Scholar] [CrossRef] [PubMed]
- Chiapponi, C.; Faccini, S.; De Mattia, A.; Baioni, L.; Barbieri, I.; Rosignoli, C.; Nigrelli, A.; Foni, E. Detection of influenza d virus among swine and cattle, Italy. Emerg. Infect. Dis. 2016, 22, 352–354. [Google Scholar] [CrossRef] [PubMed]
- Shaw, M.L.; Palese, P. Orthomyxoviridae, 6th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; Volume 1, pp. 1151–1185. [Google Scholar]
- Skehel, J.J.; Wiley, D.C. Receptor binding and membrane fusion in virus entry: The influenza hemagglutinin. Annu. Rev. Biochem. 2000, 69, 531–569. [Google Scholar] [CrossRef] [PubMed]
- Wilson, I.A.; Skehel, J.J.; Wiley, D.C. Structure of the haemagglutinin membrane glycoprotein of influenza virus at 3 A resolution. Nature 1981, 289, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.H.; Holsinger, L.J.; Lamb, R.A. Influenza virus M2 protein has ion channel activity. Cell 1992, 69, 517–528. [Google Scholar] [CrossRef]
- Hutchinson, E.C.; Charles, P.D.; Hester, S.S.; Thomas, B.; Trudgian, D.; Martinez-Alonso, M.; Fodor, E. Conserved and host-specific features of influenza virion architecture. Nat. Commun. 2014, 5, 4816. [Google Scholar] [CrossRef] [PubMed]
- Pleschka, S. Overview of influenza viruses. Curr. Top. Microbiol. Immunol. 2013, 370, 1–20. [Google Scholar] [PubMed]
- Iwai, A.; Shiozaki, T.; Kawai, T.; Akira, S.; Kawaoka, Y.; Takada, A.; Kida, H.; Miyazaki, T. Influenza a virus polymerase inhibits type I interferon induction by binding to interferon beta promoter stimulator 1. J. Biol. Chem. 2010, 285, 32064–32074. [Google Scholar] [CrossRef] [PubMed]
- Yamayoshi, S.; Watanabe, M.; Goto, H.; Kawaoka, Y. Identification of a novel viral protein expressed from the pb2 segment of influenza a virus. J. Virol. 2016, 90, 444–456. [Google Scholar] [CrossRef] [PubMed]
- Schmolke, M.; Manicassamy, B.; Pena, L.; Sutton, T.; Hai, R.; Varga, Z.T.; Hale, B.G.; Steel, J.; Perez, D.R.; Garcia-Sastre, A. Differential contribution of pb1-f2 to the virulence of highly pathogenic h5n1 influenza a virus in mammalian and avian species. PLoS Pathog. 2011, 7, e1002186. [Google Scholar] [CrossRef] [PubMed]
- Varga, Z.T.; Ramos, I.; Hai, R.; Schmolke, M.; Garcia-Sastre, A.; Fernandez-Sesma, A.; Palese, P. The influenza virus protein pb1-f2 inhibits the induction of type I interferon at the level of the MAVS adaptor protein. PLoS Pathog. 2011, 7, e1002067. [Google Scholar] [CrossRef] [PubMed]
- Krumbholz, A.; Philipps, A.; Oehring, H.; Schwarzer, K.; Eitner, A.; Wutzler, P.; Zell, R. Current knowledge on pb1-f2 of influenza A viruses. Med. Microbiol. Immunol. 2011, 200, 69–75. [Google Scholar] [CrossRef] [PubMed]
- McAuley, J.L.; Hornung, F.; Boyd, K.L.; Smith, A.M.; McKeon, R.; Bennink, J.; Yewdell, J.W.; McCullers, J.A. Expression of the 1918 influenza a virus pb1-f2 enhances the pathogenesis of viral and secondary bacterial pneumonia. Cell Host Microbe 2007, 2, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Zamarin, D.; Ortigoza, M.B.; Palese, P. Influenza A virus pb1-f2 protein contributes to viral pathogenesis in mice. J. Virol. 2006, 80, 7976–7983. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, A.K.; Pasricha, G. An insight into the pb1f2 protein and its multifunctional role in enhancing the pathogenicity of the influenza A viruses. Virology 2013, 440, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Tauber, S.; Ligertwood, Y.; Quigg-Nicol, M.; Dutia, B.M.; Elliott, R.M. Behaviour of influenza A viruses differentially expressing segment 2 gene products in vitro and in vivo. J. Gen. Virol. 2012, 93, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Wise, H.M.; Foeglein, A.; Sun, J.; Dalton, R.M.; Patel, S.; Howard, W.; Anderson, E.C.; Barclay, W.S.; Digard, P. A complicated message: Identification of a novel PB1-related protein translated from influenza a virus segment 2 mRNA. J. Virol. 2009, 83, 8021–8031. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Sun, Y.; Hu, J.; Qi, L.; Wang, J.; Xiong, X.; Wang, Y.; He, Q.; Lin, Y.; Kong, W.; et al. The contribution of pa-x to the virulence of pandemic 2009 h1n1 and highly pathogenic h5n1 avian influenza viruses. Sci. Rep. 2015, 5, 8262. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Mo, Y.; Wang, X.; Gu, M.; Hu, Z.; Zhong, L.; Wu, Q.; Hao, X.; Hu, S.; Liu, W.; et al. Pa-x decreases the pathogenicity of highly pathogenic h5n1 influenza a virus in avian species by inhibiting virus replication and host response. J. Virol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Jagger, B.W.; Wise, H.M.; Digard, P.; Holmes, E.C.; Taubenberger, J.K. Evolutionary conservation of the pa-x open reading frame in segment 3 of influenza a virus. J. Virol. 2012, 86, 12411–12413. [Google Scholar] [CrossRef] [PubMed]
- Muramoto, Y.; Noda, T.; Kawakami, E.; Akkina, R.; Kawaoka, Y. Identification of novel influenza a virus proteins translated from pa mRNA. J. Virol. 2013, 87, 2455–2462. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Li, Q.; Liu, T.; Chang, G.; Sun, Z.; Gao, Z.; Wang, F.; Zhou, H.; Liu, R.; Zheng, M.; et al. Host interaction analysis of pa-n155 and pa-n182 in chicken cells reveals an essential role of uba52 for replication of h5n1 avian influenza virus. Front. Microbiol. 2018, 9, 936. [Google Scholar] [CrossRef] [PubMed]
- Bui, M.; Wills, E.G.; Helenius, A.; Whittaker, G.R. Role of the influenza virus m1 protein in nuclear export of viral ribonucleoproteins. J. Virol. 2000, 74, 1781–1786. [Google Scholar] [CrossRef] [PubMed]
- Wise, H.M.; Hutchinson, E.C.; Jagger, B.W.; Stuart, A.D.; Kang, Z.H.; Robb, N.; Schwartzman, L.M.; Kash, J.C.; Fodor, E.; Firth, A.E.; et al. Identification of a novel splice variant form of the influenza a virus m2 ion channel with an antigenically distinct ectodomain. PLoS Pathog. 2012, 8, e1002998. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.F.; Read, E.K.C.; Wise, H.M.; Amorim, M.J.; Digard, P. Influenza a virus ns1 protein promotes efficient nuclear export of unspliced viral M1 mRNA. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Ayllon, J.; Garcia-Sastre, A. The NS1 protein: A multitasking virulence factor. Curr. Top. Microbiol. Immunol. 2015, 386, 73–107. [Google Scholar] [PubMed]
- Fan, S.; Macken, C.A.; Li, C.; Ozawa, M.; Goto, H.; Iswahyudi, N.F.; Nidom, C.A.; Chen, H.; Neumann, G.; Kawaoka, Y. Synergistic effect of the pdz and p85beta-binding domains of the ns1 protein on virulence of an avian H5N1 influenza A virus. J. Virol. 2013, 87, 4861–4871. [Google Scholar] [CrossRef] [PubMed]
- Golebiewski, L.; Liu, H.; Javier, R.T.; Rice, A.P. The avian influenza virus NS1 ESEV PDZ binding motif associates with dlg1 and scribble to disrupt cellular tight junctions. J. Virol. 2011, 85, 10639–10648. [Google Scholar] [CrossRef] [PubMed]
- Hale, B.G.; Randall, R.E.; Ortin, J.; Jackson, D. The multifunctional ns1 protein of influenza A viruses. J. Gen. Virol. 2008, 89, 2359–2376. [Google Scholar] [CrossRef] [PubMed]
- Robb, N.C.; Smith, M.; Vreede, F.T.; Fodor, E. Ns2/nep protein regulates transcription and replication of the influenza virus RNA genome. J. Gen. Virol. 2009, 90, 1398–1407. [Google Scholar] [CrossRef] [PubMed]
- Neumann, G.; Hughes, M.T.; Kawaoka, Y. Influenza a virus ns2 protein mediates vrnp nuclear export through nes-independent interaction with hcrm1. EMBO J. 2000, 19, 6751–6758. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, R.E.; Talon, J.; Palese, P. The influenza virus nep (ns2 protein) mediates the nuclear export of viral ribonucleoproteins. EMBO J. 1998, 17, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, J.; Nakada, S.; Kato, A.; Toyoda, T.; Ishihama, A. Molecular assembly of influenza virus: Association of the ns2 protein with virion matrix. Virology 1993, 196, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Selman, M.; Dankar, S.K.; Forbes, N.E.; Jia, J.-J.; Brown, E.G. Adaptive mutation in influenza a virus non-structural gene is linked to host switching and induces a novel protein by alternative splicing. Emerg. Microbes Infect. 2012, 1, e42. [Google Scholar] [CrossRef] [PubMed]
- Pleschka, S.; Jaskunas, R.; Engelhardt, O.G.; Zurcher, T.; Palese, P.; Garcia-Sastre, A. A plasmid-based reverse genetics system for influenza a virus. J. Virol. 1996, 70, 4188–4192. [Google Scholar] [PubMed]
- Neumann, G.; Watanabe, T.; Ito, H.; Watanabe, S.; Goto, H.; Gao, P.; Hughes, M.; Perez, D.R.; Donis, R.; Hoffmann, E.; et al. Generation of influenza A viruses entirely from cloned cDNAs. Proc. Natl. Acad. Sci. USA 1999, 96, 9345–9350. [Google Scholar] [CrossRef] [PubMed]
- Fodor, E.; Devenish, L.; Engelhardt, O.G.; Palese, P.; Brownlee, G.G.; Garcia-Sastre, A. Rescue of influenza A virus from recombinant DNA. J. Virol. 1999, 73, 9679–9682. [Google Scholar] [PubMed]
- Tong, S.; Li, Y.; Rivailler, P.; Conrardy, C.; Castillo, D.A.; Chen, L.M.; Recuenco, S.; Ellison, J.A.; Davis, C.T.; York, I.A.; et al. A distinct lineage of influenza A virus from bats. Proc. Natl. Acad. Sci. USA 2012, 109, 4269–4274. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.; Zhu, X.; Li, Y.; Shi, M.; Zhang, J.; Bourgeois, M.; Yang, H.; Chen, X.; Recuenco, S.; Gomez, J.; et al. New world bats harbor diverse influenza A viruses. PLoS Pathog. 2013, 9, e1003657. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Tefsen, B.; Shi, Y.; Gao, G.F. Bat-derived influenza-like viruses h17n10 and h18n11. Trends Microbiol. 2014, 22, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Wright, P.F.; Neumann, G.; Kawaoka, Y. Orthomyxoviruses; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; Volume 1, pp. 1186–1241. [Google Scholar]
- Webster, R.G.; Bean, W.J.; Gorman, O.T.; Chambers, T.M.; Kawaoka, Y. Evolution and ecology of Influenza A viruses. Microbiol. Rev. 1992, 56, 152–179. [Google Scholar] [PubMed]
- Short, K.R.; Richard, M.; Verhagen, J.H.; van Riel, D.; Schrauwen, E.J.A.; van den Brand, J.M.A.; Mänz, B.; Bodewes, R.; Herfst, S. One health, multiple challenges: The inter-species transmission of Influenza A virus. One Health 2015, 1, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Gatherer, D. The 2009 h1n1 influenza outbreak in its historical context. J. Clin. Virol. 2009, 45, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Hay, A.J.; Gregory, V.; Douglas, A.R.; Lin, Y.P. The evolution of human influenza viruses. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2001, 356, 1861–1870. [Google Scholar]
- Lessler, J.; Reich, N.G.; Cummings, D.A.; Nair, H.P.; Jordan, H.T.; Thompson, N. Outbreak of 2009 pandemic influenza A (h1n1) at a New York City school. N. Engl. J. Med. 2009, 361, 2628–2636. [Google Scholar] [CrossRef] [PubMed]
- Reid, A.H.; Fanning, T.G.; Hultin, J.V.; Taubenberger, J.K. Origin and evolution of the 1918 “Spanish” influenza virus hemagglutinin gene. Proc. Natl. Acad. Sci. USA 1999, 96, 1651–1656. [Google Scholar] [CrossRef] [PubMed]
- Parry, J. H7N9 avian flu infects humans for the first time. BMJ 2013, 346, f2151. [Google Scholar] [CrossRef] [PubMed]
- To, K.K.; Tsang, A.K.; Chan, J.F.; Cheng, V.C.; Chen, H.; Yuen, K.Y. Emergence in China of human disease due to avian influenza a(h10n8)—Cause for concern? J. Infect. 2014, 68, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Yuen, K.Y.; Chan, P.K.; Peiris, M.; Tsang, D.N.; Que, T.L.; Shortridge, K.F.; Cheung, P.T.; To, W.K.; Ho, E.T.; Sung, R.; et al. Clinical features and rapid viral diagnosis of human disease associated with avian influenza a h5n1 virus. Lancet 1998, 351, 467–471. [Google Scholar] [CrossRef]
- Shi, W.; Shi, Y.; Wu, Y.; Liu, D.; Gao, G.F. Origin and molecular characterization of the human-infecting h6n1 influenza virus in Taiwan. Protein Cell 2013, 4, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.S.; Yuen, K.Y. Avian influenza virus infections in humans. Chest 2006, 129, 156–168. [Google Scholar] [CrossRef] [PubMed]
- WHO. Human Infection with Avian Influenza A(H7n4) Virus—China. Available online: http://www.Who.Int/csr/don/22-february-2018-ah7n4-china/en/ (accessed on 24 February 2018).
- Chen, H.; Yuan, H.; Gao, R.; Zhang, J.; Wang, D.; Xiong, Y.; Fan, G.; Yang, F.; Li, X.; Zhou, J.; et al. Clinical and epidemiological characteristics of a fatal case of avian influenza A H10N8 virus infection: A descriptive study. Lancet 2014, 383, 714–721. [Google Scholar] [CrossRef]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Emerging infectious diseases of wildlife—Threats to biodiversity and human health. Science 2000, 287, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Swayne, D.E.; Suarez, D.L. Highly pathogenic avian influenza. Rev. Sci. Tech. 2000, 19, 463–482. [Google Scholar] [CrossRef] [PubMed]
- Steinhauer, D.A. Role of hemagglutinin cleavage for the pathogenicity of influenza virus. Virology 1999, 258, 1–20. [Google Scholar] [CrossRef] [PubMed]
- OIE. Highly Pathogenic Avian Influenza, Ghana. Available online: http://www.oie.int/wahis_2/public/wahid.php/Reviewreport/Review?page_refer=MapFullEventReport&reportid=25915 (accessed on 24 February 2018).
- Alexander, D.J. A review of avian influenza in different bird species. Vet. Microbiol. 2000, 74, 3–13. [Google Scholar] [CrossRef]
- Chen, J.; Lee, K.H.; Steinhauer, D.A.; Stevens, D.J.; Skehel, J.J.; Wiley, D.C. Structure of the hemagglutinin precursor cleavage site, a determinant of influenza pathogenicity and the origin of the labile conformation. Cell 1998, 95, 409–417. [Google Scholar] [CrossRef]
- Pereira, H.G.; Tumova, B.; Law, V.G. Avian influenza A viruses. Bull. World Health Organ. 1965, 32, 855–860. [Google Scholar] [PubMed]
- Becker, W.B. The isolation and classification of tern virus: Influenza a-tern South Africa—1961. J. Hyg. 1966, 64, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Smith, G.J.; Zhang, S.Y.; Qin, K.; Wang, J.; Li, K.S.; Webster, R.G.; Peiris, J.S.; Guan, Y. Avian flu: H5n1 virus outbreak in migratory waterfowl. Nature 2005, 436, 191–192. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Zhang, Z.; Liu, W.; Yin, Y.; Hong, J.; Li, X.; Wang, H.; Wong, G.; Chen, J.; Li, Y.; et al. Highly pathogenic avian influenza a(h5n1) virus struck migratory birds in China in 2015. Sci. Rep. 2015, 5, 12986. [Google Scholar] [CrossRef] [PubMed]
- Selim, A.A.; Erfan, A.M.; Hagag, N.; Zanaty, A.; Samir, A.H.; Samy, M.; Abdelhalim, A.; Arafa, A.A.; Soliman, M.A.; Shaheen, M.; et al. Highly pathogenic avian influenza virus (h5n8) clade 2.3.4.4 infection in migratory birds, Egypt. Emerg. Infect. Dis. 2017, 23, 1048–1051. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, H.; Bi, Y.; Sun, J.; Wong, G.; Liu, D.; Li, L.; Liu, J.; Chen, Q.; Wang, H.; et al. Highly Pathogenic avian influenza a(h5n8) virus in wild migratory birds, Qinghai Lake, China. Emerg. Infect. Dis. 2017, 23, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Capua, I.; Alexander, D.J. Human health implications of avian influenza viruses and paramyxoviruses. Eur. J. Clin. Microbiol. Infect. Dis. 2004, 23, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Xie, Z.; Xie, Z.; Xie, L.; Huang, L.; Huang, J.; Deng, X.; Zeng, T.; Wang, S.; Zhang, Y.; et al. Surveillance of live poultry markets for low pathogenic avian influenza viruses in Guangxi Province, Southern China, from 2012–2015. Sci. Rep. 2017, 7, 17577. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Kong, W.; Qi, W.; Long, L.-P.; Cao, Z.; Huang, L.; Qi, H.; Cao, N.; Wang, W.; Zhao, F.; et al. Identification of an H6N6 swine influenza virus in southern China. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2011, 11, 1174–1177. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.I.; Holmes, E.C. The evolution of epidemic influenza. Nat. Rev. Genet. 2007, 8, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Fourment, M.; Holmes, E.C. Avian influenza virus exhibits distinct evolutionary dynamics in wild birds and poultry. BMC Evol. Biol. 2015, 15, 120. [Google Scholar] [CrossRef] [PubMed]
- Conenello, G.M.; Tisoncik, J.R.; Rosenzweig, E.; Varga, Z.T.; Palese, P.; Katze, M.G. A single N66S mutation in the PB1-F2 protein of influenza A virus increases virulence by inhibiting the early interferon response in vivo. J. Virol. 2011, 85, 652–662. [Google Scholar] [CrossRef] [PubMed]
- Urbaniak, K.; Markowska-Daniel, I. In vivo reassortment of influenza viruses. Acta Biochim. Pol. 2014, 61, 427–431. [Google Scholar] [PubMed]
- Ozawa, M.; Kawaoka, Y. Crosstalk between animal and human influenza viruses. Annu. Rev. Anim. Biosci. 2013, 1, 21–42. [Google Scholar] [CrossRef] [PubMed]
- Medina, R.A.; Garcia-Sastre, A. Influenza A viruses: New research developments. Nat. Rev. Microbiol. 2011, 9, 590–603. [Google Scholar] [CrossRef] [PubMed]
- Garten, R.J.; Davis, C.T.; Russell, C.A.; Shu, B.; Lindstrom, S.; Balish, A.; Sessions, W.M.; Xu, X.; Skepner, E.; Deyde, V.; et al. Antigenic and genetic characteristics of swine-origin 2009 a(h1n1) influenza viruses circulating in humans. Science 2009, 325, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Broberg, E.; Snacken, R.; Adlhoch, C.; Beaute, J.; Galinska, M.; Pereyaslov, D.; Brown, C.; Penttinen, P.; Collective on behalf of the WHO European Region; The European Influenza Surveillance Network. Start of the 2014/15 influenza season in Europe: Drifted influenza a(h3n2) viruses circulate as dominant subtype. Eur. Commun. Dis. Bull. 2015, 20. [Google Scholar] [CrossRef]
- Meijer, A.; Swaan, C.M.; Voerknecht, M.; Jusic, E.; van den Brink, S.; Wijsman, L.A.; Voordouw, B.C.; Donker, G.A.; Sleven, J.; Dorigo-Zetsma, W.W.; et al. Case of seasonal reassortant a(h1n2) influenza virus infection, the Netherlands, March 2018. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Claas, E.C.; Osterhaus, A.D.; van Beek, R.; De Jong, J.C.; Rimmelzwaan, G.F.; Senne, D.A.; Krauss, S.; Shortridge, K.F.; Webster, R.G. Human influenza a h5n1 virus related to a highly pathogenic avian influenza virus. Lancet 1998, 351, 472–477. [Google Scholar] [CrossRef]
- Subbarao, K.; Klimov, A.; Katz, J.; Regnery, H.; Lim, W.; Hall, H.; Perdue, M.; Swayne, D.; Bender, C.; Huang, J.; et al. Characterization of an avian influenza a (h5n1) virus isolated from a child with a fatal respiratory illness. Science 1998, 279, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Bahgat, M.M.; Kutkat, M.A.; Nasraa, M.H.; Mostafa, A.; Webby, R.; Bahgat, I.M.; Ali, M.A. Characterization of an avian influenza virus h5n1 Egyptian isolate. J. Virol. Methods 2009, 159, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Abdelwhab, E.M.; Hassan, M.K.; Abdel-Moneim, A.S.; Naguib, M.M.; Mostafa, A.; Hussein, I.T.; Arafa, A.; Erfan, A.M.; Kilany, W.H.; Agour, M.G.; et al. Introduction and enzootic of a/h5n1 in Egypt: Virus evolution, pathogenicity and vaccine efficacy ten years on. Infect. Genet. Evol. 2016, 40, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.J. Avian influenza viruses and human health. Dev. Biol. 2006, 124, 77–84. [Google Scholar]
- Tweed, S.A.; Skowronski, D.M.; David, S.T.; Larder, A.; Petric, M.; Lees, W.; Li, Y.; Katz, J.; Krajden, M.; Tellier, R.; et al. Human illness from avian influenza h7n3, British Columbia. Emerg. Infect. Dis. 2004, 10, 2196–2199. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, M.; Wilbrink, B.; Conyn, M.; Natrop, G.; van der Nat, H.; Vennema, H.; Meijer, A.; van Steenbergen, J.; Fouchier, R.; Osterhaus, A.; et al. Transmission of h7n7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet 2004, 363, 587–593. [Google Scholar] [CrossRef]
- Abdelwhab, E.M.; Abdel-Moneim, A.S. Epidemiology, ecology and gene pool of influenza A virus in Egypt: Will Egypt be the epicentre of the next influenza pandemic? Virulence 2015, 6, 6–18. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.; Su, C.; Wang, D.; Peng, Y.; Liu, M.; Hua, S.; Li, T.; Gao, G.F.; Tang, H.; Chen, J.; et al. Sequential reassortments underlie diverse influenza h7n9 genotypes in China. Cell Host Microbe 2013, 14, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Zhou, X.; Shi, W.; Huang, L.; Xia, W.; Liu, D.; Li, H.; Chen, S.; Lei, F.; Cao, L.; et al. Genesis of the novel human-infecting influenza a(h10n8) virus and potential genetic diversity of the virus in poultry, China. Eur Surveill. 2014, 19, 20841. [Google Scholar] [CrossRef]
- Lam, T.T.; Wang, J.; Shen, Y.; Zhou, B.; Duan, L.; Cheung, C.L.; Ma, C.; Lycett, S.J.; Leung, C.Y.; Chen, X.; et al. The genesis and source of the h7n9 influenza viruses causing human infections in China. Nature 2013, 502, 241–244. [Google Scholar] [CrossRef] [PubMed]
- WHO. Cumulative Number of Confirmed Human Cases of Avian Influenza A(H5n1) Reported to WHO. Available online: http://www.who.int/influenza/human_animal_interface/H5N1_cumulative_table_archives/en/ (accessed on 19 July 2018).
- EMPRES. Disease Events. Available online: http://empres-i.fao.org/eipws3g/ (accessed on 19 July 2018).
- FAO. H7N9 Situation Update. Available online: http://www.fao.org/ag/againfo/programmes/en/empres/h7n9/situation_update.html (accessed on 19 July 2018).
- Lin, Y.P.; Shaw, M.; Gregory, V.; Cameron, K.; Lim, W.; Klimov, A.; Subbarao, K.; Guan, Y.; Krauss, S.; Shortridge, K.; et al. Avian-to-human transmission of h9n2 subtype influenza A viruses: Relationship between h9n2 and H5N1 human isolates. Proc. Natl. Acad. Sci. USA 2000, 97, 9654–9658. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Shortridge, K.F.; Krauss, S.; Webster, R.G. Molecular characterization of h9n2 influenza viruses: Were they the donors of the “internal” genes of h5n1 viruses in Hong Kong? Proc. Natl. Acad. Sci. USA 1999, 96, 9363–9367. [Google Scholar] [CrossRef] [PubMed]
- Monne, I.; Yamage, M.; Dauphin, G.; Claes, F.; Ahmed, G.; Giasuddin, M.; Salviato, A.; Ormelli, S.; Bonfante, F.; Schivo, A.; et al. Reassortant avian influenza a(h5n1) viruses with h9n2-pb1 gene in poultry, Bangladesh. Emerg. Infect. Dis. 2013, 19, 1630–1634. [Google Scholar] [CrossRef] [PubMed]
- Lei, F.; Shi, W. Prospective of genomics in revealing transmission, reassortment and evolution of wildlife-borne avian influenza a (h5n1) Viruses. Curr. Genomics 2011, 12, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Gerloff, N.A.; Khan, S.U.; Balish, A.; Shanta, I.S.; Simpson, N.; Berman, L.; Haider, N.; Poh, M.K.; Islam, A.; Gurley, E.; et al. Multiple reassortment events among highly pathogenic avian influenza a(h5n1) viruses detected in Bangladesh. Virology 2014, 450–451, 297–307. [Google Scholar] [CrossRef] [PubMed]
- WHO. Continued evolution of highly pathogenic avian influenza A (h5n1): Updated nomenclature. Influenza Other Respir. Viruses 2012, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization/World Organisation for Animal Health/Food; Agriculture Organization (WHO/OIE/FAO) H5N1 Evolution Working Group. Revised and updated nomenclature for highly pathogenic avian influenza A (H5N1) viruses. Influenza Other Respir. Viruses 2014, 8, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Bertran, K.; Kwon, J.H.; Swayne, D.E. Evolution, global spread, and pathogenicity of highly pathogenic avian influenza H5Nx clade 2.3.4.4. J. Vet. Sci. 2017, 18, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Vijaykrishna, D.; Bahl, J.; Riley, S.; Duan, L.; Zhang, J.X.; Chen, H.; Peiris, J.S.M.; Smith, G.J.D.; Guan, Y. Evolutionary Dynamics and emergence of panzootic 1 influenza viruses. PLoS Pathog. 2008, 4, e1000161. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.J.; Donis, R.O.; World Health Organization/World Organisation for Animal Health/Food; Agriculture Organization (WHO/OIE/FAO) H5 Evolution Working Group. Nomenclature updates resulting from the evolution of avian influenza a(h5) virus clades 2.1.3.2a, 2.2.1, and 2.3.4 during 2013-2014. Influenza Other Respir. Viruses 2015, 9, 271–276. [Google Scholar] [PubMed]
- Arafa, A.S.; Yamada, S.; Imai, M.; Watanabe, T.; Yamayoshi, S.; Iwatsuki-Horimoto, K.; Kiso, M.; Sakai-Tagawa, Y.; Ito, M.; Imamura, T.; et al. Risk assessment of recent egyptian h5n1 influenza viruses. Sci. Rep. 2016, 6, 38388. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wang, H.; Zhao, L.; Ma, L.; Wang, R.; Lei, Y.; Li, Y.; Yang, G.; Chen, J.; Chen, G.; et al. First documented case of avian influenza (h5n1) virus infection in a lion. Emerg. Microbes Infect. 2016, 5, e125. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Shi, J.; Qi, X.; Huang, G.; Chen, H.; Lu, C. Lethal infection by a novel reassortant h5n1 avian influenza A virus in a zoo-housed tiger. Microbes Infect. 2015, 17, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Pabbaraju, K.; Tellier, R.; Wong, S.; Li, Y.; Bastien, N.; Tang, J.W.; Drews, S.J.; Jang, Y.; Davis, C.T.; Fonseca, K.; et al. Full-genome analysis of avian influenza a(h5n1) virus from a human, North America, 2013. Emerg. Infect. Dis. 2014, 20, 887–891. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Chen, J.; Zhang, Z.; Li, M.; Cai, T.; Sharshov, K.; Susloparov, I.; Shestopalov, A.; Wong, G.; He, Y.; et al. Highly pathogenic avian influenza h5n1 clade 2.3.2.1c virus in migratory birds, 2014–2015. Virol. Sin. 2016, 31, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Naguib, M.M.; Kinne, J.; Chen, H.; Chan, K.H.; Joseph, S.; Wong, P.C.; Woo, P.C.; Wernery, R.; Beer, M.; Wernery, U.; et al. Outbreaks of highly pathogenic avian influenza h5n1 clade 2.3.2.1c in hunting falcons and kept wild birds in Dubai implicate intercontinental virus spread. J. Gen. Virol. 2015, 96, 3212. [Google Scholar] [CrossRef] [PubMed]
- Dharmayanti, N.L.; Hartawan, R.; Pudjiatmoko Wibawa, H.; Hardiman Balish, A.; Donis, R.; Davis, C.T.; Samaan, G. Genetic characterization of clade 2.3.2.1 avian influenza a(h5n1) viruses, Indonesia, 2012. Emerg. Infect. Dis. 2014, 20, 671–674. [Google Scholar] [CrossRef] [PubMed]
- Marinova-Petkova, A.; Feeroz, M.M.; Rabiul Alam, S.M.; Kamrul Hasan, M.; Akhtar, S.; Jones-Engel, L.; Walker, D.; McClenaghan, L.; Rubrum, A.; Franks, J.; et al. Multiple introductions of highly pathogenic avian influenza h5n1 viruses into Bangladesh. Emerg. Microbes Infect. 2014, 3, e11. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Gao, R.; Lv, Q.; Huang, S.; Zhou, Z.; Yang, L.; Li, X.; Zhao, X.; Zou, X.; Tong, W.; et al. Human infection with a novel, highly pathogenic avian influenza a (h5n6) virus: Virological and clinical findings. J. Infect. 2016, 72, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhu, W.; Li, X.; Bo, H.; Zhang, Y.; Zou, S.; Gao, R.; Dong, J.; Zhao, X.; Chen, W.; et al. Genesis and dissemination of highly pathogenic H5N6 avian influenza viruses. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Chen, M.; Yang, J.; Jia, Y.; Han, S.; Holmes, E.C.; Cui, J. Molecular evolution and emergence of h5n6 avian influenza virus in central China. J. Virol. 2017, 91, e00143-17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, M.; Huang, Y.; Zhu, W.; Yang, L.; Gao, L.; Li, X.; Bi, F.; Huang, C.; Kang, N.; et al. Human infections with novel reassortant h5n6 avian influenza viruses in China. Emerg. Microbes Infect. 2017, 6, e50. [Google Scholar] [CrossRef] [PubMed]
- WHO. Avian Influenza Weekly Update Number 629. Available online: http://www.wpro.who.int/emerging_diseases/ai_weekly_629_wpro_20180323.pdf (accessed on 19 July 2018).
- Gillim-Ross, L.; Santos, C.; Chen, Z.; Aspelund, A.; Yang, C.F.; Ye, D.; Jin, H.; Kemble, G.; Subbarao, K. Avian influenza h6 viruses productively infect and cause illness in mice and ferrets. J. Virol. 2008, 82, 10854–10863. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.H.; Yang, J.R.; Wu, H.S.; Chang, M.C.; Lin, J.S.; Lin, C.Y.; Liu, Y.L.; Lo, Y.C.; Yang, C.H.; Chuang, J.H.; et al. Human infection with avian influenza a h6n1 virus: An epidemiological analysis. Lancet Respir. Med. 2013, 1, 771–778. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Chia, M.-Y.; Chen, P.-L.; Yeh, C.-T.; Cheng, M.-C.; Su, I.-J.; Lee, M.-S. Assessment of pathogenicity and antigenicity of American lineage influenza h5n2 viruses in Taiwan. Virology 2017, 508, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Belser, J.A.; Bridges, C.B.; Katz, J.M.; Tumpey, T.M. Past, present, and possible future human infection with influenza virus A subtype H7. Emerg. Infect. Dis. 2009, 15, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Prevention. Notes from the field: Highly pathogenic avian influenza a (h7n3) virus infection in two poultry workers—Jalisco, Mexico, July 2012. MMWR Morb. Mortal. Wkly. Rep. 2012, 61, 726–727. [Google Scholar]
- Van Kolfschooten, F. Dutch veterinarian becomes first victim of avian influenza. Lancet 2003, 361, 1444. [Google Scholar] [CrossRef]
- Puzelli, S.; Rossini, G.; Facchini, M.; Vaccari, G.; Di Trani, L.; Di Martino, A.; Gaibani, P.; Vocale, C.; Cattoli, G.; Bennett, M.; et al. Human infection with highly pathogenic a(h7n7) avian influenza virus, Italy, 2013. Emerg. Infect. Dis. 2014, 20, 1745–1749. [Google Scholar] [CrossRef] [PubMed]
- Ke, C.; Mok, C.K.P.; Zhu, W.; Zhou, H.; He, J.; Guan, W.; Wu, J.; Song, W.; Wang, D.; Liu, J.; et al. Human Infection with Highly Pathogenic Avian Influenza a(h7n9) Virus, China. Emerg. Infect. Dis. 2017, 23, 1332–1340. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhu, W.; Li, X.; Chen, M.; Wu, J.; Yu, P.; Qi, S.; Huang, Y.; Shi, W.; Dong, J.; et al. Genesis and spread of newly emerged highly pathogenic h7n9 avian viruses in mainland China. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Yamayoshi, S.; Kiso, M.; Yasuhara, A.; Ito, M.; Shu, Y.; Kawaoka, Y. Enhanced replication of highly pathogenic influenza a(h7n9) virus in humans. Emerg. Infect. Dis. 2018, 24, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Xiao, D.; Li, D.; Wang, Y.; Yan, T.; Dai, B.; Wang, X. The temporal distribution of new h7n9 avian influenza infections based on laboratory-confirmed cases in Mainland China, 2013-2017. Sci. Rep. 2018, 8, 4051. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Gu, M.; Liu, D.; Cui, J.; Gao, G.F.; Zhou, J.; Liu, X. Epidemiology, evolution, and pathogenesis of h7n9 influenza viruses in five epidemic waves since 2013 in China. Trends Microbiol. 2017, 25, 713–728. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, S.; Bing, G.; Carter, R.A.; Wang, Z.; Wang, J.; Wang, C.; Wang, L.; Wu, G.; Webster, R.G.; et al. Genetic evolution of influenza h9n2 viruses isolated from various hosts in China from 1994 to 2013. Emerg. Microbes Infect. 2017, 6, e106. [Google Scholar] [CrossRef] [PubMed]
- Smietanka, K.; Minta, Z.; Swieton, E.; Olszewska, M.; Jozwiak, M.; Domanska-Blicharz, K.; Wyrostek, K.; Tomczyk, G.; Pikula, A. Avian influenza h9n2 subtype in Poland--characterization of the isolates and evidence of concomitant infections. Avian Pathol. 2014, 43, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Aamir, U.B.; Wernery, U.; Ilyushina, N.; Webster, R.G. Characterization of avian h9n2 influenza viruses from United Arab Emirates 2000 to 2003. Virology 2007, 361, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Matrosovich, M.N.; Krauss, S.; Webster, R.G. H9n2 influenza A viruses from poultry in Asia have human virus-like receptor specificity. Virology 2001, 281, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H. Challenge for One Health: Co-Circulation of Zoonotic h5n1 and h9n2 avian influenza viruses in Egypt. Viruses 2018, 10, 121. [Google Scholar] [CrossRef] [PubMed]
- El Houadfi, M.; Fellahi, S.; Nassik, S.; Guerin, J.L.; Ducatez, M.F. First outbreaks and phylogenetic analyses of avian influenza h9n2 viruses isolated from poultry flocks in Morocco. Virol. J. 2016, 13, 140. [Google Scholar] [CrossRef] [PubMed]
- Nagy, A.; Mettenleiter, T.C.; Abdelwhab, E.M. A brief summary of the epidemiology and genetic relatedness of avian influenza h9n2 virus in birds and mammals in the Middle East and North Africa. Epidemiol. Infect. 2017, 145, 3320–3333. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Xu, L.; Wang, X.; Liu, X. Current situation of h9n2 subtype avian influenza in China. Vet. Res. 2017, 48, 49. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Tian, B.; Jianfang, Z.; Yongkun, C.; Xiaodan, L.; Wenfei, Z.; Yan, L.; Jing, T.; Junfeng, G.; Tao, C.; et al. A comprehensive retrospective study of the seroprevalence of h9n2 avian influenza viruses in occupationally exposed populations in China. PLoS ONE 2017, 12, e0178328. [Google Scholar] [CrossRef] [PubMed]
- Yuan, R.; Liang, L.; Wu, J.; Kang, Y.; Song, Y.; Zou, L.; Zhang, X.; Ni, H.; Ke, C. Human infection with an avian influenza a/h9n2 virus in Guangdong in 2016. J. Infect. 2017, 74, 422–425. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.Q.; Zhang, Y.; Zhao, N.; Yu, Z.; Pan, H.; Chan, T.C.; Zhang, Z.R.; Liu, S.L. Comparative epidemiology of human fatal infections with novel, high (h5n6 and h5n1) and low (h7n9 and h9n2) pathogenicity avian influenza A viruses. Int. J. Environ. Res. Public Health 2017, 14, 263. [Google Scholar] [CrossRef] [PubMed]
- Marinova-Petkova, A.; Franks, J.; Tenzin, S.; Dahal, N.; Dukpa, K.; Dorjee, J.; Feeroz, M.M.; Rehg, J.E.; Barman, S.; Krauss, S.; et al. Highly pathogenic reassortant avian influenza a(h5n1) virus clade 2.3.2.1a in poultry, bhutan. Emerg. Infect. Dis. 2016, 22, 2137–2141. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Peng, X.; Peng, X.; Cheng, L.; Lu, X.; Jin, C.; Xie, T.; Yao, H.; Wu, N. Genetic and molecular characterization of h9n2 and h5 avian influenza viruses from live poultry markets in Zhejiang Province, eastern China. Sci. Rep. 2015, 5, 17508. [Google Scholar] [CrossRef] [PubMed]
- Yuan, R.; Zou, L.; Kang, Y.; Wu, J.; Zeng, X.; Lu, J.; Liang, L.; Song, Y.; Zhang, X.; Ni, H.; et al. Reassortment of Avian Influenza A/H6N6 Viruses from Live Poultry Markets in Guangdong, China. Front. Microbiol. 2016, 7, 65. [Google Scholar] [CrossRef] [PubMed]
- Arzey, G.G.; Kirkland, P.D.; Arzey, K.E.; Frost, M.; Maywood, P.; Conaty, S.; Hurt, A.C.; Deng, Y.M.; Iannello, P.; Barr, I.; et al. Influenza virus A (H10N7) in chickens and poultry abattoir workers, Australia. Emerg. Infect. Dis. 2012, 18, 814–816. [Google Scholar] [CrossRef] [PubMed]
- Van den Brand, J.M.; Wohlsein, P.; Herfst, S.; Bodewes, R.; Pfankuche, V.M.; van de Bildt, M.W.; Seehusen, F.; Puff, C.; Richard, M.; Siebert, U.; et al. Influenza A (H10N7) Virus Causes Respiratory Tract Disease in Harbor Seals and Ferrets. PLoS ONE 2016, 11, e0159625. [Google Scholar]
- Bodewes, R.; Bestebroer, T.M.; van der Vries, E.; Verhagen, J.H.; Herfst, S.; Koopmans, M.P.; Fouchier, R.A.; Pfankuche, V.M.; Wohlsein, P.; Siebert, U.; et al. Avian Influenza A(H10N7) virus-associated mass deaths among harbor seals. Emerg. Infect. Dis. 2015, 21, 720–722. [Google Scholar] [CrossRef] [PubMed]
- Dittrich, A.; Scheibner, D.; Salaheldin, A.H.; Veits, J.; Gischke, M.; Mettenleiter, T.C.; Abdelwhab, E.M. Impact of Mutations in the Hemagglutinin of H10N7 Viruses Isolated from Seals on Virus Replication in Avian and Human Cells. Viruses 2018, 10, 83. [Google Scholar] [CrossRef] [PubMed]
- Lai, S.; Qin, Y.; Cowling, B.J.; Ren, X.; Wardrop, N.A.; Gilbert, M.; Tsang, T.K.; Wu, P.; Feng, L.; Jiang, H.; et al. Global epidemiology of avian influenza A H5N1 virus infection in humans, 1997-2015: A systematic review of individual case data. Lancet Infect. Dis. 2016, 16, e108–e118. [Google Scholar] [CrossRef]
- Vong, S.; Ly, S.; Van Kerkhove, M.D.; Achenbach, J.; Holl, D.; Buchy, P.; Sorn, S.; Seng, H.; Uyeki, T.M.; Sok, T.; et al. Risk factors associated with subclinical human infection with avian influenza A (H5N1) virus--Cambodia, 2006. J. Infect. Dis. 2009, 199, 1744–1752. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jiang, H.; Wu, P.; Uyeki, T.M.; Feng, L.; Lai, S.; Wang, L.; Huo, X.; Xu, K.; Chen, E.; et al. Epidemiology of avian influenza A H7N9 virus in human beings across five epidemics in mainland China, 2013–2017: An epidemiological study of laboratory-confirmed case series. Lancet Infect. Dis. 2017, 17, 822–832. [Google Scholar] [CrossRef]
- Belser, J.A.; Lash, R.R.; Garg, S.; Tumpey, T.M.; Maines, T.R. The eyes have it: Influenza virus infection beyond the respiratory tract. Lancet Infect. Dis. 2018, 18, e220–e227. [Google Scholar] [CrossRef]
- Pfeiffer, D.U.; Otte, M.J.; Roland-Holst, D.; Zilberman, D. A one health perspective on HPAI H5N1 in the Greater Mekong sub-region. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Guan, D.; Kang, M.; Wu, J.; Zeng, X.; Lu, J.; Rutherford, S.; Zou, L.; Liang, L.; Ni, H.; et al. Family clusters of avian influenza A H7N9 virus infection in Guangdong Province, China. J. Clin. Microbiol. 2015, 53, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.J.; Ungchusak, K.; Sovann, L.; Uyeki, T.M.; Dowell, S.F.; Cox, N.J.; Aldis, W.; Chunsuttiwat, S. Family clustering of avian influenza A (H5N1). Emerg. Infect. Dis. 2005, 11, 1799–1801. [Google Scholar] [CrossRef] [PubMed]
- Abdelwhab, E.M.; Hafez, H.M. An overview of the epidemic of highly pathogenic H5N1 avian influenza virus in Egypt: Epidemiology and control challenges. Epidemiol. Infect. 2011, 139, 647–657. [Google Scholar] [CrossRef] [PubMed]
- Van Kerkhove, M.D.; Mumford, E.; Mounts, A.W.; Bresee, J.; Ly, S.; Bridges, C.B.; Otte, J. Highly pathogenic avian influenza (h5n1): Pathways of exposure at the animal-human interface, a systematic review. PLoS ONE 2011, 6, e14582. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.U.; Anderson, B.D.; Heil, G.L.; Liang, S.; Gray, G.C. A Systematic Review and Meta-Analysis of the Seroprevalence of Influenza a(h9n2) Infection Among Humans. J. Infect. Dis. 2015, 212, 562–569. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ju, L.; Liu, P.; Zhou, J.; Lv, X.; Li, L.; Shen, H.; Su, H.; Jiang, L.; Jiang, Q. Serological and virological surveillance of avian influenza a virus h9n2 subtype in humans and poultry in Shanghai, China, between 2008 and 2010. Zoonoses Public Health 2015, 62, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Fragaszy, E.; Ishola, D.A.; Brown, I.H.; Enstone, J.; Nguyen-Van-Tam, J.S.; Simons, R.; Tucker, A.W.; Wieland, B.; Williamson, S.M.; Hayward, A.C.; et al. Increased risk of A(H1N1)pdm09 influenza infection in UK pig industry workers compared to a general population cohort. Influenza Other Respir. Viruses 2016, 10, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Siembieda, J.; Johnson, C.K.; Boyce, W.; Sandrock, C.; Cardona, C. Risk for avian influenza virus exposure at human-wildlife interface. Emerg. Infect. Dis. 2008, 14, 1151–1153. [Google Scholar] [CrossRef] [PubMed]
- Gray, G.C.; Ferguson, D.D.; Lowther, P.E.; Heil, G.L.; Friary, J.A. A national study of us bird banders for evidence of avian influenza virus infections. J. Clin. Virol. 2011, 51, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, E.; Krauss, S.; Perez, D.; Webby, R.; Webster, R.G. Eight-plasmid system for rapid generation of influenza virus vaccines. Vaccine 2002, 20, 3165–3170. [Google Scholar] [CrossRef]
- Mostafa, A.; Kanrai, P.; Petersen, H.; Ibrahim, S.; Rautenschlein, S.; Pleschka, S. Efficient generation of recombinant influenza A viruses employing a new approach to overcome the genetic instability of ha segments. PLoS ONE 2015, 10, e0116917. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, A.; Kanrai, P.; Ziebuhr, J.; Pleschka, S. Improved dual promotor-driven reverse genetics system for influenza viruses. J. Virol. Methods 2013, 193, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Reperant, L.A.; Kuiken, T.; Osterhaus, A.D. Adaptive pathways of zoonotic influenza viruses: From exposure to establishment in humans. Vaccine 2012, 30, 4419–4434. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Qu, N.; Guo, Y.; Cao, L.; Wu, S.; Mei, K.; Sun, H.; Lu, Y.; Qin, Z.; Jiao, P.; et al. Phylogeny, pathogenicity, and transmission of h5n1 avian influenza viruses in chickens. Front. Cell. Infect. Microbiol. 2017, 7, 328. [Google Scholar] [CrossRef] [PubMed]
- Kandeil, A.; El-Shesheny, R.; Maatouq, A.; Moatasim, Y.; Cai, Z.; McKenzie, P.; Webby, R.; Kayali, G.; Ali, M.A. Novel reassortant h9n2 viruses in pigeons and evidence for antigenic diversity of h9n2 viruses isolated from quails in Egypt. J. Gen. Virol. 2017, 98, 548–562. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.; Tian, Y.; Hou, T.; Bao, D.; Chen, H.; Teng, Q.; Yang, J.; Li, X.; Wang, G.; Li, Z.; et al. H9n2 influenza virus isolated from minks has enhanced virulence in mice. Transbound. Emerg. Dis. 2018, 65, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Cauldwell, A.V.; Long, J.S.; Moncorge, O.; Barclay, W.S. Viral determinants of influenza A virus host range. J. Gen. Virol. 2014, 95, 1193–1210. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Qiao, C.; Marjuki, H.; Bawa, B.; Ma, J.; Guillossou, S.; Webby, R.J.; Richt, J.A.; Ma, W. Combination of PB2 271A and SR polymorphism at positions 590/591 is critical for viral replication and virulence of swine influenza virus in cultured cells and in vivo. J. Virol. 2012, 86, 1233–1237. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Wang, P.; Mok, B.W.; Lau, S.Y.; Huang, X.; Wu, W.L.; Zheng, M.; Wen, X.; Yang, S.; Chen, Y.; et al. The K526R substitution in viral protein PB2 enhances the effects of E627K on influenza virus replication. Nat. Commun. 2014, 5, 5509. [Google Scholar] [CrossRef] [PubMed]
- Mok, C.K.; Yen, H.L.; Yu, M.Y.; Yuen, K.M.; Sia, S.F.; Chan, M.C.; Qin, G.; Tu, W.W.; Peiris, J.S. Amino acid residues 253 and 591 of the PB2 protein of avian influenza virus a h9n2 contribute to mammalian pathogenesis. J. Virol. 2011, 85, 9641–9645. [Google Scholar] [CrossRef] [PubMed]
- Shinya, K.; Hamm, S.; Hatta, M.; Ito, H.; Ito, T.; Kawaoka, Y. PB2 amino acid at position 627 affects replicative efficiency, but not cell tropism, of Hong Kong H5N1 influenza A viruses in mice. Virology 2004, 320, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Hatta, M.; Gao, P.; Halfmann, P.; Kawaoka, Y. Molecular basis for high virulence of Hong Kong H5N1 influenza A viruses. Science 2001, 293, 1840–1842. [Google Scholar] [CrossRef] [PubMed]
- Massin, P.; van der Werf, S.; Naffakh, N. Residue 627 of PB2 is a determinant of cold sensitivity in RNA replication of avian influenza viruses. J. Virol. 2001, 75, 5398–5404. [Google Scholar] [CrossRef] [PubMed]
- Hatta, M.; Hatta, Y.; Kim, J.H.; Watanabe, S.; Shinya, K.; Nguyen, T.; Lien, P.S.; Le, Q.M.; Kawaoka, Y. Growth of H5N1 influenza A viruses in the upper respiratory tracts of mice. PLoS Pathog. 2007, 3, 1374–1379. [Google Scholar] [CrossRef] [PubMed]
- Steel, J.; Lowen, A.C.; Mubareka, S.; Palese, P. Transmission of influenza virus in a mammalian host is increased by PB2 amino acids 627K or 627E/701N. PLoS Pathog. 2009, 5, e1000252. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Wang, J.; Wang, P.; Song, W.; Zheng, Z.; Chen, R.; Guo, K.; Zhang, T.; Peiris, J.S.; Chen, H.; et al. Substitution of lysine at 627 position in PB2 protein does not change virulence of the 2009 pandemic H1N1 virus in mice. Virology 2010, 401, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Taubenberger, J.K.; Reid, A.H.; Lourens, R.M.; Wang, R.; Jin, G.; Fanning, T.G. Characterization of the 1918 influenza virus polymerase genes. Nature 2005, 437, 889–893. [Google Scholar] [CrossRef] [PubMed]
- Kocer, Z.A.; Fan, Y.; Huether, R.; Obenauer, J.; Webby, R.J.; Zhang, J.; Webster, R.G.; Wu, G. Survival analysis of infected mice reveals pathogenic variations in the genome of avian H1N1 viruses. Sci. Rep. 2014, 4, 7455. [Google Scholar] [CrossRef] [PubMed]
- Kawaoka, Y.; Krauss, S.; Webster, R.G. Avian-to-human transmission of the PB1 gene of influenza A viruses in the 1957 and 1968 pandemics. J. Virol. 1989, 63, 4603–4608. [Google Scholar] [PubMed]
- Pappas, C.; Aguilar, P.V.; Basler, C.F.; Solorzano, A.; Zeng, H.; Perrone, L.A.; Palese, P.; Garcia-Sastre, A.; Katz, J.M.; Tumpey, T.M. Single gene reassortants identify a critical role for PB1, HA, and NA in the high virulence of the 1918 pandemic influenza virus. Proc. Natl. Acad. Sci. USA 2008, 105, 3064–3069. [Google Scholar] [CrossRef] [PubMed]
- Wendel, I.; Rubbenstroth, D.; Doedt, J.; Kochs, G.; Wilhelm, J.; Staeheli, P.; Klenk, H.D.; Matrosovich, M. The avian-origin pb1 gene segment facilitated replication and transmissibility of the h3n2/1968 pandemic influenza virus. J. Virol. 2015. [Google Scholar] [CrossRef] [PubMed]
- McAuley, J.L.; Chipuk, J.E.; Boyd, K.L.; Van De Velde, N.; Green, D.R.; McCullers, J.A. PB1-F2 proteins from H5N1 and 20 century pandemic influenza viruses cause immunopathology. PLoS Pathog. 2010, 6, e1001014. [Google Scholar] [CrossRef] [PubMed]
- Conenello, G.M.; Zamarin, D.; Perrone, L.A.; Tumpey, T.; Palese, P. A single mutation in the PB1-F2 of H5N1 (HK/97) and 1918 influenza A viruses contributes to increased virulence. PLoS Pathog. 2007, 3, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
- Song, M.S.; Pascua, P.N.; Lee, J.H.; Baek, Y.H.; Lee, O.J.; Kim, C.J.; Kim, H.; Webby, R.J.; Webster, R.G.; Choi, Y.K. The polymerase acidic protein gene of influenza a virus contributes to pathogenicity in a mouse model. J. Virol. 2009, 83, 12325–12335. [Google Scholar] [CrossRef] [PubMed]
- Bussey, K.A.; Desmet, E.A.; Mattiacio, J.L.; Hamilton, A.; Bradel-Tretheway, B.; Bussey, H.E.; Kim, B.; Dewhurst, S.; Takimoto, T. PA residues in the 2009 H1N1 pandemic influenza virus enhance avian influenza virus polymerase activity in mammalian cells. J. Virol. 2011, 85, 7020–7028. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Zhang, X.; Gao, W.; Wang, C.; Wang, J.; Sun, H.; Sun, Y.; Guo, L.; Zhang, R.; Chang, K.C.; et al. Prevailing pa mutation k356r in avian influenza h9n2 virus increases mammalian replication and pathogenicity. J. Virol. 2016, 90, 8105–8114. [Google Scholar] [CrossRef] [PubMed]
- Mehle, A.; Dugan, V.G.; Taubenberger, J.K.; Doudna, J.A. Reassortment and mutation of the avian influenza virus polymerase PA subunit overcome species barriers. J. Virol. 2012, 86, 1750–1757. [Google Scholar] [CrossRef] [PubMed]
- Jagger, B.W.; Wise, H.M.; Kash, J.C.; Walters, K.A.; Wills, N.M.; Xiao, Y.L.; Dunfee, R.L.; Schwartzman, L.M.; Ozinsky, A.; Bell, G.L.; et al. An overlapping protein-coding region in influenza A virus segment 3 modulates the host response. Science 2012, 337, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Killip, M.J.; Staeheli, P.; Randall, R.E.; Jackson, D. The human interferon-induced MxA protein inhibits early stages of influenza A virus infection by retaining the incoming viral genome in the cytoplasm. J. Virol. 2013, 87, 13053–13058. [Google Scholar] [CrossRef] [PubMed]
- Nigg, P.E.; Pavlovic, J. Oligomerization and gtp-binding requirements of mxa for viral target recognition and antiviral activity against influenza a virus. J. Biol. Chem. 2015, 290, 29893–29906. [Google Scholar] [CrossRef] [PubMed]
- Dittmann, J.; Stertz, S.; Grimm, D.; Steel, J.; Garcia-Sastre, A.; Haller, O.; Kochs, G. Influenza A virus strains differ in sensitivity to the antiviral action of Mx-GTPase. J. Virol. 2008, 82, 3624–3631. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, P.; Manz, B.; Haller, O.; Schwemmle, M.; Kochs, G. The viral nucleoprotein determines Mx sensitivity of influenza A viruses. J. Virol. 2011, 85, 8133–8140. [Google Scholar] [CrossRef] [PubMed]
- Manz, B.; Dornfeld, D.; Gotz, V.; Zell, R.; Zimmermann, P.; Haller, O.; Kochs, G.; Schwemmle, M. Pandemic influenza A viruses escape from restriction by human MxA through adaptive mutations in the nucleoprotein. PLoS Pathog. 2013, 9, e1003279. [Google Scholar] [CrossRef] [PubMed]
- Riegger, D.; Hai, R.; Dornfeld, D.; Manz, B.; Leyva-Grado, V.; Sanchez-Aparicio, M.T.; Albrecht, R.A.; Palese, P.; Haller, O.; Schwemmle, M.; et al. The nucleoprotein of newly emerged H7N9 influenza A virus harbors a unique motif conferring resistance to antiviral human MxA. J. Virol. 2015, 89, 2241–2252. [Google Scholar] [CrossRef] [PubMed]
- Gotz, V.; Magar, L.; Dornfeld, D.; Giese, S.; Pohlmann, A.; Hoper, D.; Kong, B.W.; Jans, D.A.; Beer, M.; Haller, O.; et al. Influenza A viruses escape from MxA restriction at the expense of efficient nuclear vRNP import. Sci. Rep. 2016, 6, 23138. [Google Scholar] [CrossRef] [PubMed]
- Ashenberg, O.; Padmakumar, J.; Doud, M.B.; Bloom, J.D. Deep mutational scanning identifies sites in influenza nucleoprotein that affect viral inhibition by MxA. PLoS Pathog. 2017, 13, e1006288. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, E.C.; Fodor, E. Nuclear import of the influenza A virus transcriptional machinery. Vaccine 2012, 30, 7353–7358. [Google Scholar] [CrossRef] [PubMed]
- Goldfarb, D.S.; Corbett, A.H.; Mason, D.A.; Harreman, M.T.; Adam, S.A. Importin alpha: A multipurpose nuclear-transport receptor. Trends Cell Biol. 2004, 14, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Sankhala, R.S.; Florio, T.J.; Zhou, L.; Nguyen, N.L.T.; Lokareddy, R.K.; Cingolani, G.; Pante, N. Synergy of two low-affinity NLSs determines the high avidity of influenza A virus nucleoprotein NP for human importin alpha isoforms. Sci. Rep. 2017, 7, 11381. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, R.E.; Jaskunas, R.; Blobel, G.; Palese, P.; Moroianu, J. Nuclear import of influenza virus RNA can be mediated by viral nucleoprotein and transport factors required for protein import. J. Biol. Chem. 1995, 270, 22701–22704. [Google Scholar] [CrossRef] [PubMed]
- Mukaigawa, J.; Nayak, D.P. Two signals mediate nuclear localization of influenza virus (A/WSN/33) polymerase basic protein 2. J. Virol. 1991, 65, 245–253. [Google Scholar] [PubMed]
- Nath, S.T.; Nayak, D.P. Function of two discrete regions is required for nuclear localization of polymerase basic protein 1 of A/WSN/33 influenza virus (H1 N1). Mol. Cell. Biol. 1990, 10, 4139–4145. [Google Scholar] [CrossRef] [PubMed]
- Nieto, A.; de la Luna, S.; Barcena, J.; Portela, A.; Ortin, J. Complex structure of the nuclear translocation signal of influenza virus polymerase PA subunit. J. Gen. Virol. 1994, 75, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, G.; Herwig, A.; Klenk, H.D. Interaction of polymerase subunit PB2 and NP with importin alpha1 is a determinant of host range of influenza A virus. PLoS Pathog. 2008, 4, e11. [Google Scholar] [CrossRef] [PubMed]
- Bertram, S.; Thiele, S.; Dreier, C.; Resa-Infante, P.; Preuss, A.; van Riel, D.; Mok, C.K.P.; Schwalm, F.; Peiris, J.S.M.; Klenk, H.D.; et al. H7n9 influenza a virus exhibits importin-alpha7-mediated replication in the mammalian respiratory tract. Am. J. Pathol. 2017, 187, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, G.; Klingel, K.; Otte, A.; Thiele, S.; Hudjetz, B.; Arman-Kalcek, G.; Sauter, M.; Shmidt, T.; Rother, F.; Baumgarte, S.; et al. Differential use of importin-α isoforms governs cell tropism and host adaptation of influenza virus. Nat. Commun. 2011, 2, 156. [Google Scholar] [CrossRef] [PubMed]
- Hudjetz, B.; Gabriel, G. Human-like pb2 627k influenza virus polymerase activity is regulated by importin-α1 and -α7. PLoS Pathog. 2012, 8, e1002488. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Li, R.; Du, L.; Liu, S. Roles of the hemagglutinin of influenza A virus in viral entry and development of antiviral therapeutics and vaccines. Protein Cell 2010, 1, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Kido, H.; Yokogoshi, Y.; Sakai, K.; Tashiro, M.; Kishino, Y.; Fukutomi, A.; Katunuma, N. Isolation and characterization of a novel trypsin-like protease found in rat bronchiolar epithelial Clara cells. A possible activator of the viral fusion glycoprotein. J. Biol. Chem. 1992, 267, 13573–13579. [Google Scholar] [PubMed]
- Bottcher-Friebertshauser, E.; Garten, W.; Matrosovich, M.; Klenk, H.D. The hemagglutinin: A determinant of pathogenicity. Curr. Top. Microbiol. Immunol. 2014, 385, 3–34. [Google Scholar] [PubMed]
- Franca, M.; Stallknecht, D.E.; Howerth, E.W. Expression and distribution of sialic acid influenza virus receptors in wild birds. Avian Pathol. 2013, 42, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Ito, T.; Suzuki, T.; Holland, R.E., Jr.; Chambers, T.M.; Kiso, M.; Ishida, H.; Kawaoka, Y. Sialic acid species as a determinant of the host range of influenza A viruses. J. Virol. 2000, 74, 11825–11831. [Google Scholar] [CrossRef] [PubMed]
- Kumlin, U.; Olofsson, S.; Dimock, K.; Arnberg, N. Sialic acid tissue distribution and influenza virus tropism. Influenza Other Respir. Viruses 2008, 2, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Shinya, K.; Ebina, M.; Yamada, S.; Ono, M.; Kasai, N.; Kawaoka, Y. Avian flu: Influenza virus receptors in the human airway. Nature 2006, 440, 435–436. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Couceiro, J.N.; Kelm, S.; Baum, L.G.; Krauss, S.; Castrucci, M.R.; Donatelli, I.; Kida, H.; Paulson, J.C.; Webster, R.G.; et al. Molecular basis for the generation in pigs of influenza A viruses with pandemic potential. J. Virol. 1998, 72, 7367–7373. [Google Scholar] [PubMed]
- Wan, H.; Perez, D.R. Quail carry sialic acid receptors compatible with binding of avian and human influenza viruses. Virology 2006, 346, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Mair, C.M.; Ludwig, K.; Herrmann, A.; Sieben, C. Receptor binding and pH stability—How influenza A virus hemagglutinin affects host-specific virus infection. Biochim. Biophys. Acta 2014, 1838, 1153–1168. [Google Scholar] [CrossRef] [PubMed]
- Myers, J.L.; Wetzel, K.S.; Linderman, S.L.; Li, Y.; Sullivan, C.B.; Hensley, S.E. Compensatory hemagglutinin mutations alter antigenic properties of influenza viruses. J. Virol. 2013, 87, 11168–11172. [Google Scholar] [CrossRef] [PubMed]
- Ayora-Talavera, G.; Shelton, H.; Scull, M.A.; Ren, J.; Jones, I.M.; Pickles, R.J.; Barclay, W.S. Mutations in H5N1 influenza virus hemagglutinin that confer binding to human tracheal airway epithelium. PLoS ONE 2009, 4, e7836. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.I.; Lee, I.; Park, S.; Lee, S.; Hwang, M.W.; Bae, J.Y.; Heo, J.; Kim, D.; Jang, S.I.; Song, J.W.; et al. Effects of a hemagglutinin D222G substitution on the pathogenicity of 2009 influenza A (H1N1) virus in mice. Arch. Virol. 2014, 159, 2559–2565. [Google Scholar] [CrossRef] [PubMed]
- Stieneke-Grober, A.; Vey, M.; Angliker, H.; Shaw, E.; Thomas, G.; Roberts, C.; Klenk, H.D.; Garten, W. Influenza virus hemagglutinin with multibasic cleavage site is activated by furin, a subtilisin-like endoprotease. EMBO J. 1992, 11, 2407–2414. [Google Scholar] [CrossRef] [PubMed]
- Luczo, J.M.; Stambas, J.; Durr, P.A.; Michalski, W.P.; Bingham, J. Molecular pathogenesis of H5 highly pathogenic avian influenza: The role of the haemagglutinin cleavage site motif. Rev. Med. Virol. 2015, 25, 406–430. [Google Scholar] [CrossRef] [PubMed]
- Callan, R.J.; Hartmann, F.A.; West, S.E.; Hinshaw, V.S. Cleavage of influenza A virus H1 hemagglutinin by swine respiratory bacterial proteases. J. Virol. 1997, 71, 7579–7585. [Google Scholar] [PubMed]
- Zhirnov, O.P.; Ikizler, M.R.; Wright, P.F. Cleavage of influenza a virus hemagglutinin in human respiratory epithelium is cell associated and sensitive to exogenous antiproteases. J. Virol. 2002, 76, 8682–8689. [Google Scholar] [CrossRef] [PubMed]
- Bertram, S.; Glowacka, I.; Steffen, I.; Kuhl, A.; Pohlmann, S. Novel insights into proteolytic cleavage of influenza virus hemagglutinin. Rev. Med. Virol. 2010, 20, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Chutinimitkul, S.; Herfst, S.; Steel, J.; Lowen, A.C.; Ye, J.; van Riel, D.; Schrauwen, E.J.; Bestebroer, T.M.; Koel, B.; Burke, D.F.; et al. Virulence-associated substitution D222G in the hemagglutinin of 2009 pandemic influenza A(H1N1) virus affects receptor binding. J. Virol. 2010, 84, 11802–11813. [Google Scholar] [CrossRef] [PubMed]
- Schrauwen, E.J.; Fouchier, R.A. Host adaptation and transmission of influenza A viruses in mammals. Emerg. Microbes Infect. 2014, 3, e9. [Google Scholar] [CrossRef] [PubMed]
- Imai, M.; Watanabe, T.; Hatta, M.; Das, S.C.; Ozawa, M.; Shinya, K.; Zhong, G.; Hanson, A.; Katsura, H.; Watanabe, S.; et al. Experimental adaptation of an influenza H5 HA confers respiratory droplet transmission to a reassortant H5 HA/H1N1 virus in ferrets. Nature 2012, 486, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Herfst, S.; Schrauwen, E.J.; Linster, M.; Chutinimitkul, S.; de Wit, E.; Munster, V.J.; Sorrell, E.M.; Bestebroer, T.M.; Burke, D.F.; Smith, D.J.; et al. Airborne transmission of influenza A/H5N1 virus between ferrets. Science 2012, 336, 1534–1541. [Google Scholar] [CrossRef] [PubMed]
- Sutton, T.C.; Finch, C.; Shao, H.; Angel, M.; Chen, H.; Capua, I.; Cattoli, G.; Monne, I.; Perez, D.R. Airborne transmission of highly pathogenic H7N1 influenza virus in ferrets. J. Virol. 2014, 88, 6623–6635. [Google Scholar] [CrossRef] [PubMed]
- Schmier, S.; Mostafa, A.; Haarmann, T.; Bannert, N.; Ziebuhr, J.; Veljkovic, V.; Dietrich, U.; Pleschka, S. In silico prediction and experimental confirmation of HA residues conferring enhanced human receptor specificity of H5N1 influenza A viruses. Sci. Rep. 2015, 5, 11434. [Google Scholar] [CrossRef] [PubMed]
- Mitnaul, L.J.; Matrosovich, M.N.; Castrucci, M.R.; Tuzikov, A.B.; Bovin, N.V.; Kobasa, D.; Kawaoka, Y. Balanced hemagglutinin and neuraminidase activities are critical for efficient replication of influenza A virus. J. Virol. 2000, 74, 6015–6020. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Zhu, X.; McBride, R.; Nycholat, C.M.; Yu, W.; Paulson, J.C.; Wilson, I.A. Functional balance of the hemagglutinin and neuraminidase activities accompanies the emergence of the 2009 H1N1 influenza pandemic. J. Virol. 2012, 86, 9221–9232. [Google Scholar] [CrossRef] [PubMed]
- Baum, L.G.; Paulson, J.C. The N2 neuraminidase of human influenza virus has acquired a substrate specificity complementary to the hemagglutinin receptor specificity. Virology 1991, 180, 10–15. [Google Scholar] [CrossRef]
- Kobasa, D.; Kodihalli, S.; Luo, M.; Castrucci, M.R.; Donatelli, I.; Suzuki, Y.; Suzuki, T.; Kawaoka, Y. Amino acid residues contributing to the substrate specificity of the influenza A virus neuraminidase. J. Virol. 1999, 73, 6743–6751. [Google Scholar] [PubMed]
- Zhou, H.; Yu, Z.; Hu, Y.; Tu, J.; Zou, W.; Peng, Y.; Zhu, J.; Li, Y.; Zhang, A.; Yu, Z.; et al. The special neuraminidase stalk-motif responsible for increased virulence and pathogenesis of H5N1 influenza A virus. PLoS ONE 2009, 4, e6277. [Google Scholar] [CrossRef] [PubMed]
- Sorrell, E.M.; Song, H.; Pena, L.; Perez, D.R. A 27-amino-acid deletion in the neuraminidase stalk supports replication of an avian H2N2 influenza A virus in the respiratory tract of chickens. J. Virol. 2010, 84, 11831–11840. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Tan, Y.; Wei, K.; Sun, H.; Shi, Y.; Pu, J.; Yang, H.; Gao, G.F.; Yin, Y.; Feng, W.; et al. Amino acid 316 of hemagglutinin and the neuraminidase stalk length influence virulence of h9n2 influenza virus in chickens and mice. J. Virol. 2013, 87, 2963–2968. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, T.W.; Munier, S.; Larcher, T.; Soubieux, D.; Ledevin, M.; Esnault, E.; Tourdes, A.; Croville, G.; Guerin, J.L.; Quere, P.; et al. Length variations in the NA stalk of an H7N1 influenza virus have opposite effects on viral excretion in chickens and ducks. J. Virol. 2012, 86, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Blumenkrantz, D.; Roberts, K.L.; Shelton, H.; Lycett, S.; Barclay, W.S. The short stalk length of highly pathogenic avian influenza H5N1 virus neuraminidase limits transmission of pandemic H1N1 virus in ferrets. J. Virol. 2013, 87, 10539–10551. [Google Scholar] [CrossRef] [PubMed]
- Marc, D. Influenza virus non-structural protein NS1: Interferon antagonism and beyond. J. Gen. Virol. 2014, 95, 2594–2611. [Google Scholar] [CrossRef] [PubMed]
- Rivas, H.G.; Schmaling, S.K.; Gaglia, M.M. Shutoff of host gene expression in influenza a virus and herpesviruses: Similar mechanisms and common themes. Viruses 2016, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Brenner, D.; Wang, Z.; Dauber, B.; Ehrhardt, C.; Hogner, K.; Herold, S.; Ludwig, S.; Wolff, T.; Yu, K.; et al. The NS segment of an H5N1 highly pathogenic avian influenza virus (HPAIV) is sufficient to alter replication efficiency, cell tropism, and host range of an H7N1 HPAIV. J. Virol. 2010, 84, 2122–2133. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Robb, N.C.; Lenz, E.; Wolff, T.; Fodor, E.; Pleschka, S. NS reassortment of an H7-type highly pathogenic avian influenza virus affects its propagation by altering the regulation of viral RNA production and antiviral host response. J. Virol. 2010, 84, 11323–11335. [Google Scholar] [CrossRef] [PubMed]
- Kanrai, P.; Mostafa, A.; Madhugiri, R.; Lechner, M.; Wilk, E.; Schughart, K.; Ylosmaki, L.; Saksela, K.; Ziebuhr, J.; Pleschka, S. Identification of specific residues in avian influenza A virus NS1 that enhance viral replication and pathogenicity in mammalian systems. J. Gen. Virol. 2016, 97, 2135–2148. [Google Scholar] [CrossRef] [PubMed]
- Burgui, I.; Aragon, T.; Ortin, J.; Nieto, A. PABP1 and eIF4GI associate with influenza virus NS1 protein in viral mRNA translation initiation complexes. J. Gen. Virol. 2003, 84, 3263–3274. [Google Scholar] [CrossRef] [PubMed]
- Falcon, A.M.; Marion, R.M.; Zurcher, T.; Gomez, P.; Portela, A.; Nieto, A.; Ortin, J. Defective RNA replication and late gene expression in temperature-sensitive influenza viruses expressing deleted forms of the NS1 protein. J. Virol. 2004, 78, 3880–3888. [Google Scholar] [CrossRef] [PubMed]
- Dankar, S.K.; Miranda, E.; Forbes, N.E.; Pelchat, M.; Tavassoli, A.; Selman, M.; Ping, J.; Jia, J.; Brown, E.G. Influenza A/Hong Kong/156/1997(H5N1) virus NS1 gene mutations F103L and M106I both increase IFN antagonism, virulence and cytoplasmic localization but differ in binding to RIG-I and CPSF30. Virol. J. 2013, 10, 243. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.H.; Hoffmann, E.; Webster, R.G. The NS1 gene of H5N1 influenza viruses circumvents the host anti-viral cytokine responses. Virus Res. 2004, 103, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Jin, M.; Chen, H.; Huag, Q.; Yu, Z. Genome-sequence analysis of the pathogenic H5N1 avian influenza A virus isolated in China in 2004. Virus Genes 2006, 32, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Long, J.X.; Peng, D.X.; Liu, Y.L.; Wu, Y.T.; Liu, X.F. Virulence of H5N1 avian influenza virus enhanced by a 15-nucleotide deletion in the viral nonstructural gene. Virus Genes 2008, 36, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Yang, H.; Chen, W.; Cao, W.; Zhong, G.; Jiao, P.; Deng, G.; Yu, K.; Yang, C.; Bu, Z.; et al. A naturally occurring deletion in its NS gene contributes to the attenuation of an H5N1 swine influenza virus in chickens. J. Virol. 2008, 82, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Hale, B.G.; Steel, J.; Medina, R.A.; Manicassamy, B.; Ye, J.; Hickman, D.; Hai, R.; Schmolke, M.; Lowen, A.C.; Perez, D.R.; et al. Inefficient control of host gene expression by the 2009 pandemic H1N1 influenza A virus NS1 protein. J. Virol. 2010, 84, 6909–6922. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.M.; Nogales, A.; Martinez-Sobrido, L.; Topham, D.J.; DeDiego, M.L. Functional evolution of influenza virus ns1 protein in currently circulating human 2009 pandemic H1N1 viruses. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.; Hossain, M.J.; Hickman, D.; Perez, D.R.; Lamb, R.A. A new influenza virus virulence determinant: The NS1 protein four C-terminal residues modulate pathogenicity. Proc. Natl. Acad. Sci. USA 2008, 105, 4381–4386. [Google Scholar] [CrossRef] [PubMed]
- Hale, B.G.; Steel, J.; Manicassamy, B.; Medina, R.A.; Ye, J.; Hickman, D.; Lowen, A.C.; Perez, D.R.; Garcia-Sastre, A. Mutations in the NS1 C-terminal tail do not enhance replication or virulence of the 2009 pandemic H1N1 influenza A virus. J. Gen. Virol. 2010, 91, 1737–1742. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Liu, H.; Rice, A.P. Regulation of interferon-beta by MAGI-1 and its interaction with influenza A virus NS1 protein with ESEV PBM. PLoS ONE 2012, 7, e41251. [Google Scholar] [CrossRef]
- Petersen, H.; Mostafa, A.; Tantawy, M.A.; Iqbal, A.A.; Hoffmann, D.; Tallam, A.; Selvakumar, B.; Pessler, F.; Beer, M.; Rautenschlein, S.; et al. NS segment of a 1918 influenza a virus-descendent enhances replication of h1n1pdm09 and virus-induced cellular immune response in mammalian and avian systems. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Houser, K.; Subbarao, K. Influenza Vaccines: Challenges and Solutions. Cell Host Microbe 2015, 17, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, H.; Govorkova, E.A.; Li, C.; Xiong, X.; Webster, R.G.; Webby, R.J. Generation of high-yielding influenza A viruses in African green monkey kidney (Vero) cells by reverse genetics. J. Virol. 2004, 78, 1851–1857. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.M.; Williams, M.S. History of inactivated influenza vaccines. In Textbook of Influenza; Nicholson, K.G., Webster, R.G., Hay, A.J., Eds.; Blackwell Science Ltd.: Oxford, UK, 1998; pp. 317–323. [Google Scholar]
- Subbarao, K.; Chen, H.; Swayne, D.; Mingay, L.; Fodor, E.; Brownlee, G.; Xu, X.; Lu, X.; Katz, J.; Cox, N.; et al. Evaluation of a genetically modified reassortant H5N1 influenza A virus vaccine candidate generated by plasmid-based reverse genetics. Virology 2003, 305, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Schickli, J.H.; Flandorfer, A.; Nakaya, T.; Martinez-Sobrido, L.; Garcia-Sastre, A.; Palese, P. Plasmid-only rescue of influenza A virus vaccine candidates. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2001, 356, 1965–1973. [Google Scholar]
- Hu, W.; Zhang, H.; Han, Q.; Li, L.; Chen, Y.; Xia, N.; Chen, Z.; Shu, Y.; Xu, K.; Sun, B. A Vero-cell-adapted vaccine donor strain of influenza A virus generated by serial passages. Vaccine 2015, 33, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Kandeil, A.; Sabir, J.S.M.; Abdelaal, A.; Mattar, E.H.; El-Taweel, A.N.; Sabir, M.J.; Khalil, A.A.; Webby, R.; Kayali, G.; Ali, M.A. Efficacy of commercial vaccines against newly emerging avian influenza H5N8 virus in Egypt. Sci. Rep. 2018, 8, 9697. [Google Scholar] [CrossRef] [PubMed]
- Kayali, G.; Kandeil, A.; El-Shesheny, R.; Kayed, A.S.; Gomaa, M.R.; Kutkat, M.A.; Debeauchamp, J.; McKenzie, P.P.; Webster, R.G.; Webby, R.J.; et al. Do commercial avian influenza H5 vaccines induce cross-reactive antibodies against contemporary H5N1 viruses in Egypt? Poult. Sci. 2013, 92, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Kitikoon, P.; Gauger, P.C.; Anderson, T.K.; Culhane, M.R.; Swenson, S.; Loving, C.L.; Perez, D.R.; Vincent, A.L. Swine influenza virus vaccine serologic cross-reactivity to contemporary US swine H3N2 and efficacy in pigs infected with an H3N2 similar to 2011–2012 H3N2v. Influenza Other Respir. Viruses 2013, 7, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Capua, I.; Alexander, D.J. Avian influenza vaccines and vaccination in birds. Vaccine 2008, 26, D70–D73. [Google Scholar] [CrossRef] [PubMed]
- Loving, C.L.; Lager, K.M.; Vincent, A.L.; Brockmeier, S.L.; Gauger, P.C.; Anderson, T.K.; Kitikoon, P.; Perez, D.R.; Kehrli, M.E. Efficacy in pigs of inactivated and live attenuated influenza virus vaccines against infection and transmission of an emerging h3n2 similar to the 2011–2012 h3n2v. J. Virol. 2013, 87, 9895–9903. [Google Scholar] [CrossRef] [PubMed]
- Tarigan, S.; Wibowo, M.H.; Indriani, R.; Sumarningsih, S.; Artanto, S.; Idris, S.; Durr, P.A.; Asmara, W.; Ebrahimie, E.; Stevenson, M.A.; et al. Field effectiveness of highly pathogenic avian influenza H5N1 vaccination in commercial layers in Indonesia. PLoS ONE 2018, 13, e0190947. [Google Scholar] [CrossRef] [PubMed]
- Kandeil, A.; Mostafa, A.; El-Shesheny, R.; El-Taweel, A.N.; Gomaa, M.; Galal, H.; Kayali, G.; Ali, M.A. Avian influenza H5N1 vaccination efficacy in Egyptian backyard poultry. Vaccine 2017, 35, 6195–6201. [Google Scholar] [CrossRef] [PubMed]
- Abdelwhab, E.M.; Hafez, H.M. Insight into alternative approaches for control of avian influenza in poultry, with emphasis on highly pathogenic h5n1. Viruses 2012, 4, 3179–3208. [Google Scholar] [CrossRef] [PubMed]
- CDC. Prevention and Treatment of Avian Influenza A Viruses in People. Available online: https://www.cdc.gov/flu/avianflu/prevention.htm (accessed on 20 July 2018).
- Wong, S.S.; Webby, R.J. Traditional and new influenza vaccines. Clin. Microbiol. Rev. 2013, 26, 476–492. [Google Scholar] [CrossRef] [PubMed]
- McNicholl, I.R.; McNicholl, J.J. Neuraminidase inhibitors: Zanamivir and oseltamivir. Ann. Pharmacother. 2001, 35, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Paules, C.; Subbarao, K. Influenza. Lancet 2017, 390, 697–708. [Google Scholar] [CrossRef]
- Koszalka, P.; Tilmanis, D.; Hurt, A.C. Influenza antivirals currently in late-phase clinical trial. Influenza Other Respir. Viruses 2017, 11, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Li, T.C.; Chan, M.C.; Lee, N. Clinical implications of antiviral resistance in influenza. Viruses 2015, 7, 4929–4944. [Google Scholar] [CrossRef] [PubMed]
- Belshe, R.B.; Smith, M.H.; Hall, C.B.; Betts, R.; Hay, A.J. Genetic basis of resistance to rimantadine emerging during treatment of influenza virus infection. J. Virol. 1988, 62, 1508–1512. [Google Scholar] [PubMed]
- Hu, Y.; Musharrafieh, R.; Ma, C.; Zhang, J.; Smee, D.F.; DeGrado, W.F.; Wang, J. An M2-V27A channel blocker demonstrates potent in vitro and in vivo antiviral activities against amantadine-sensitive and—Resistant influenza A viruses. Antivir Res. 2017, 140, 45–54. [Google Scholar] [CrossRef] [PubMed]
- El-Shesheny, R.; Bagato, O.; Kandeil, A.; Mostafa, A.; Mahmoud, S.H.; Hassanneen, H.M.; Webby, R.J.; Ali, M.A.; Kayali, G. Re-emergence of amantadine-resistant variants among highly pathogenic avian influenza H5N1 viruses in Egypt. Infect. Genet. Evol. 2016, 46, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Galvin, H.D.; Haw, T.Y.; Nutsford, A.N.; Husain, M. Drug resistance in influenza A virus: The epidemiology and management. Infect. Drug Resist. 2017, 10, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.; Peng, C.; Luo, J.; Wang, C.; Han, L.; Wu, B.; Ji, G.; He, H. Adamantane-resistant influenza A viruses in the world (1902–2013): Frequency and distribution of m2 gene mutations. PLoS ONE 2015, 10, e0119115. [Google Scholar] [CrossRef] [PubMed]
- CDC. Influenza Antiviral Drug Resistance. 2018. Available online: https://www.cdc.gov/flu/about/qa/antiviralresistance.htm (accessed on 3 August 2018).
- Abdelwhab, E.M.; Veits, J.; Mettenleiter, T.C. Biological fitness and natural selection of amantadine resistant variants of avian influenza H5N1 viruses. Virus Res. 2017, 228, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Beigel, J.H.; Voell, J.; Munoz, P.; Kumar, P.; Brooks, K.M.; Zhang, J.; Iversen, P.; Heald, A.; Wong, M.; Davey, R.T. Safety, tolerability, and pharmacokinetics of radavirsen (AVI-7100), an antisense oligonucleotide targeting influenza a M1/M2 translation. Br. J. Clin. Pharmacol. 2018, 84, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Colman, P.M. Influenza virus neuraminidase: Structure, antibodies, and inhibitors. Protein Sci. 1994, 3, 1687–1696. [Google Scholar] [CrossRef] [PubMed]
- Colman, P.M.; Varghese, J.N.; Laver, W.G. Structure of the catalytic and antigenic sites in influenza virus neuraminidase. Nature 1983, 303, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.S.; Jeong, J.H.; Kwon, J.J.; Ahn, S.J.; Lloren, K.K.S.; Kwon, H.I.; Chae, H.B.; Hwang, J.; Kim, M.H.; Kim, C.J.; et al. Screening for neuraminidase inhibitor resistance markers among avian influenza viruses of the N4, N5, N6, and N8 neuraminidase subtypes. J. Virol. 2018, 92. [Google Scholar] [CrossRef] [PubMed]
- Tu, V.; Abed, Y.; Fage, C.; Baz, M.; Boivin, G. Impact of R152K and R368K neuraminidase catalytic substitutions on in vitro properties and virulence of recombinant A(H1N1)pdm09 viruses. Antivir. Res. 2018, 154, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Gubareva, L.V.; Kaiser, L.; Hayden, F.G. Influenza virus neuraminidase inhibitors. Lancet 2000, 355, 827–835. [Google Scholar] [CrossRef]
- Yamashita, M.; Tomozawa, T.; Kakuta, M.; Tokumitsu, A.; Nasu, H.; Kubo, S. CS-8958, a prodrug of the new neuraminidase inhibitor R-125489, shows long-acting anti-influenza virus activity. Antimicrob. Agents Chemother. 2009, 53, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Fiore, A.E.; Fry, A.; Shay, D.; Gubareva, L.; Bresee, J.S.; Uyeki, T.M.; Centers for Disease Control and Prevention (CDC). Antiviral agents for the treatment and chemoprophylaxis of influenza—Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 2011, 60, 1–24. [Google Scholar] [PubMed]
- McKimm-Breschkin, J.L. Influenza neuraminidase inhibitors: Antiviral action and mechanisms of resistance. Influenza Other Respir Viruses 2013, 7 (Suppl. 1), 25–36. [Google Scholar] [CrossRef] [PubMed]
- McKimm-Breschkin, J.L.; Barrett, S.; Wong, F.Y.K.; Pudjiatmoko; Azhar, M.; Selleck, P.; Davies, K.R.; Hartaningsih, N.; McGrane, J. Identification of Indonesian clade 2.1 highly pathogenic influenza a(h5n1) viruses with N294S and S246N neuraminidase substitutions which further reduce oseltamivir susceptibility. Antiviral Res. 2018, 153, 95–100. [Google Scholar] [PubMed]
- McKimm-Breschkin, J.L.; Barrett, S.; Pudjiatmoko; Azhar, M.; Wong, F.Y.K.; Selleck, P.; Mohr, P.G.; McGrane, J.; Kim, M. I222 Neuraminidase mutations further reduce oseltamivir susceptibility of indonesian clade 2.1 highly pathogenic avian influenza a(h5n1) viruses. PLoS ONE 2013, 8, e66105. [Google Scholar] [CrossRef]
- Myoung-don, O.; Rabeh, E.-S.; Mohamed, A.K.; Ahmed, K.; Ahmed, M.; Mariette, F.D.; Pamela, M.; Elena, A.G.; Mohamed, H.N.; Robert, G.W.; et al. Continuing threat of influenza (H5N1) virus circulation in Egypt. Emerg. Infect. Dis. J. 2011, 17, 2306. [Google Scholar] [CrossRef]
- Yen, H.L.; McKimm-Breschkin, J.L.; Choy, K.T.; Wong, D.D.; Cheung, P.P.; Zhou, J.; Ng, I.H.; Zhu, H.; Webby, R.J.; Guan, Y.; et al. Resistance to neuraminidase inhibitors conferred by an R292K mutation in a human influenza virus H7N9 isolate can be masked by a mixed R/K viral population. mBio 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Ikematsu, H.; Kawai, N.; Iwaki, N.; Kashiwagi, S.; Ishikawa, Y.; Yamaguchi, H.; Shiosakai, K. In vitro neuraminidase inhibitory concentration (IC50) of four neuraminidase inhibitors in the Japanese 2016–2017 season: Comparison with the 2010–2011 to 2015–2016 seasons. J. Infect. Chemother. 2018. [Google Scholar] [CrossRef]
- Govorkova Elena, A. Consequences of resistance: In vitro fitness, in vivo infectivity, and transmissibility of oseltamivir-resistant influenza A viruses. Influenza Other Respir. Viruses 2012, 7, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Butler, J.; Hooper, K.A.; Petrie, S.; Lee, R.; Maurer-Stroh, S.; Reh, L.; Guarnaccia, T.; Baas, C.; Xue, L.; Vitesnik, S.; et al. Estimating the fitness advantage conferred by permissive neuraminidase mutations in recent oseltamivir-resistant A(H1N1)pdm09 influenza viruses. PLoS Pathog. 2014, 10, e1004065. [Google Scholar] [CrossRef] [PubMed]
- Marjuki, H.; Mishin, V.P.; Chesnokov, A.P.; De La Cruz, J.A.; Davis, C.T.; Villanueva, J.M.; Fry, A.M.; Gubareva, L.V. Neuraminidase mutations conferring resistance to oseltamivir in influenza a(h7n9) viruses. J. Virol. 2015, 89, 5419–5426. [Google Scholar] [CrossRef] [PubMed]
- Baek, Y.H.; Song, M.S.; Lee, E.Y.; Kim, Y.I.; Kim, E.H.; Park, S.J.; Park, K.J.; Kwon, H.I.; Pascua, P.N.; Lim, G.J.; et al. Profiling and characterization of influenza virus N1 strains potentially resistant to multiple neuraminidase inhibitors. J. Virol. 2015, 89, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Leung, K.; Lipsitch, M.; Yuen, K.Y.; Wu, J.T. Monitoring the fitness of antiviral-resistant influenza strains during an epidemic: A mathematical modeling study. Lancet Infect. Dis. 2017, 17, 339–347. [Google Scholar] [CrossRef]
- Scheuch, G.; Canisius, S.; Nocker, K.; Hofmann, T.; Naumann, R.; Pleschka, S.; Ludwig, S.; Welte, T.; Planz, O. Targeting intracellular signaling as an antiviral strategy: Aerosolized LASAG for the treatment of influenza in hospitalized patients. Emerg. Microbes. Infect. 2018, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Haasbach, E.; Muller, C.; Ehrhardt, C.; Schreiber, A.; Pleschka, S.; Ludwig, S.; Planz, O. The MEK-inhibitor CI-1040 displays a broad anti-influenza virus activity in vitro and provides a prolonged treatment window compared to standard of care in vivo. Antivir. Res. 2017, 142, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Schrader, T.; Dudek, S.E.; Schreiber, A.; Ehrhardt, C.; Planz, O.; Ludwig, S. The clinically approved MEK inhibitor Trametinib efficiently blocks influenza A virus propagation and cytokine expression. Antivir. Res. 2018, 157, 80–92. [Google Scholar] [CrossRef] [PubMed]
- WHO. Overview of Amino Acid Substitutions in Influenza Neuraminidase Associated with Resistance or Reduced Susceptibility to NAIs. Available online: http://www.who.int/influenza/gisrs_laboratory/antiviral_susceptibility/en/ (accessed on 24 July 2018).
- Kwon, J.J.; Choi, W.S.; Jeong, J.H.; Kim, E.H.; Lee, O.J.; Yoon, S.W.; Hwang, J.; Webby, R.J.; Govorkova, E.A.; Choi, Y.K.; et al. An I436N substitution confers resistance of influenza A(H1N1)pdm09 viruses to multiple neuraminidase inhibitors without affecting viral fitness. J. Gen. Virol. 2018, 99, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Cheng, Y.; Li, X.; Tan, M.; Wei, H.; Zhao, X.; Xiao, N.; Dong, J.; Wang, D. Neuraminidase inhibitor susceptibility profile of human influenza viruses during the 2016-2017 influenza season in Mainland China. J. Infect. Chemother. 2018. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, J.H.; Lexmond, P.; Vuong, O.; Schutten, M.; Guldemeester, J.; Osterhaus, A.D.M.E.; Elbers, A.R.W.; Slaterus, R.; Hornman, M.; Koch, G.; et al. Discordant detection of avian influenza virus subtypes in time and space between poultry and wild birds; Towards improvement of surveillance programs. PLoS ONE 2017, 12, e0173470. [Google Scholar] [CrossRef] [PubMed]
- Kadam, R.U.; Wilson, I.A. Structural basis of influenza virus fusion inhibition by the antiviral drug Arbidol. Proc. Natl. Acad. Sci. USA 2017, 114, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.T.; Hoopes, J.D.; Smee, D.F.; Prichard, M.N.; Driebe, E.M.; Engelthaler, D.M.; Le, M.H.; Keim, P.S.; Spence, R.P.; Went, G.T. Triple combination of oseltamivir, amantadine, and ribavirin displays synergistic activity against multiple influenza virus strains in vitro. Antimicrob. Agents Chemother. 2009, 53, 4115–4126. [Google Scholar] [CrossRef] [PubMed]
- Droebner, K.; Haasbach, E.; Dudek, S.E.; Scheuch, G.; Nocker, K.; Canisius, S.; Ehrhardt, C.; von Degenfeld, G.; Ludwig, S.; Planz, O. Pharmacodynamics, pharmacokinetics, and antiviral activity of bay 81-8781, a novel nf-kappab inhibiting anti-influenza drug. Front. Microbiol. 2017, 8, 2130. [Google Scholar] [CrossRef] [PubMed]
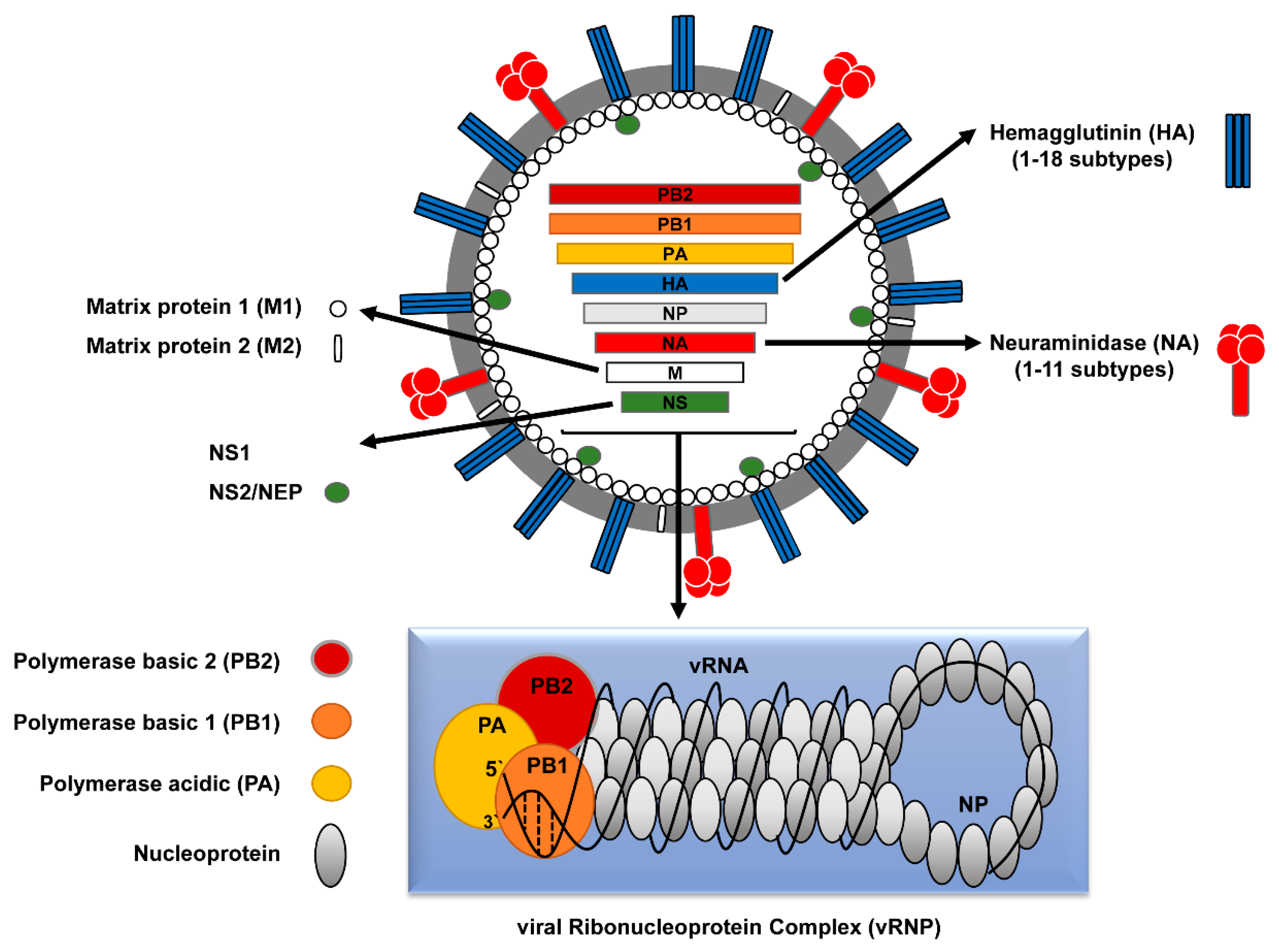
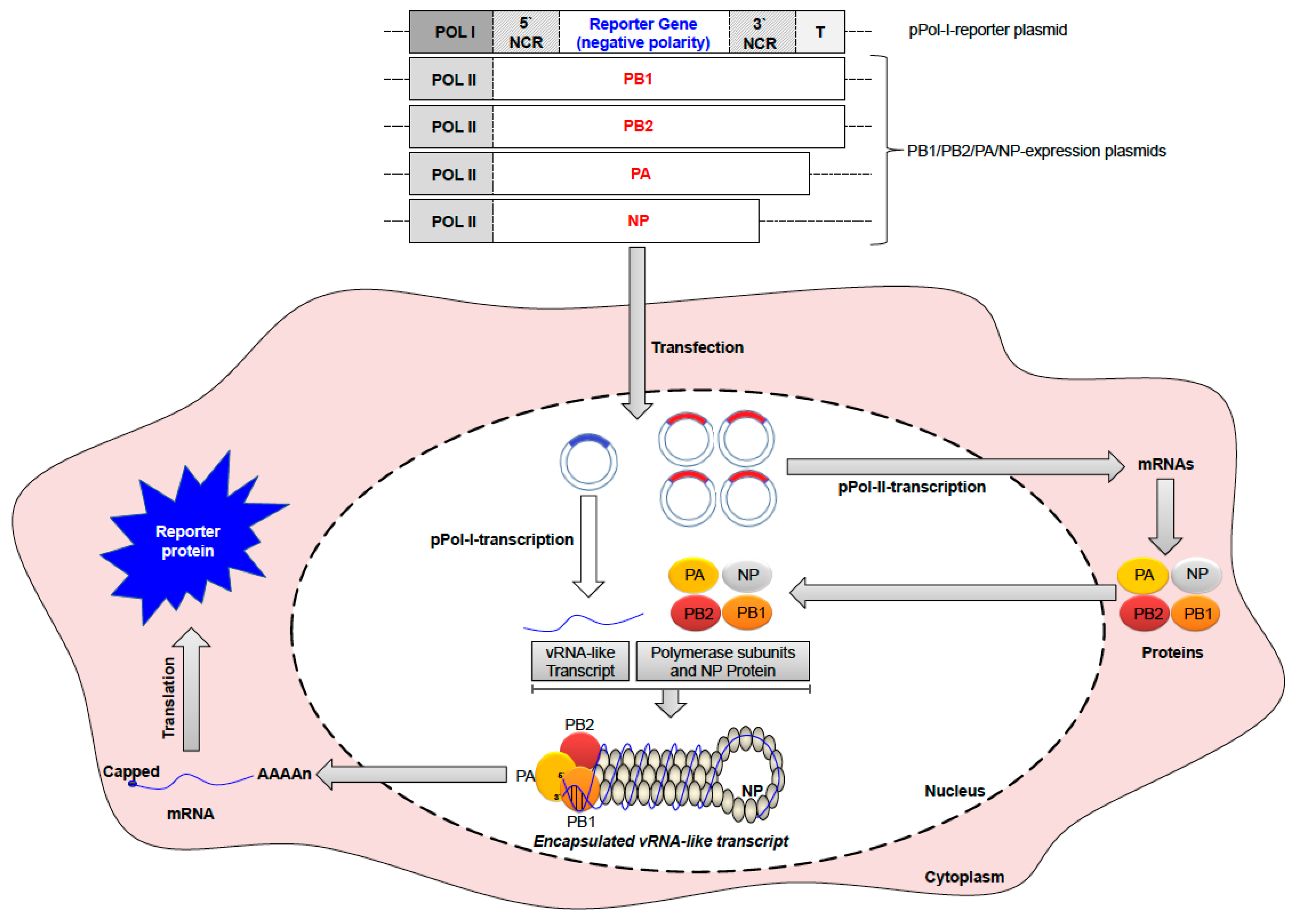
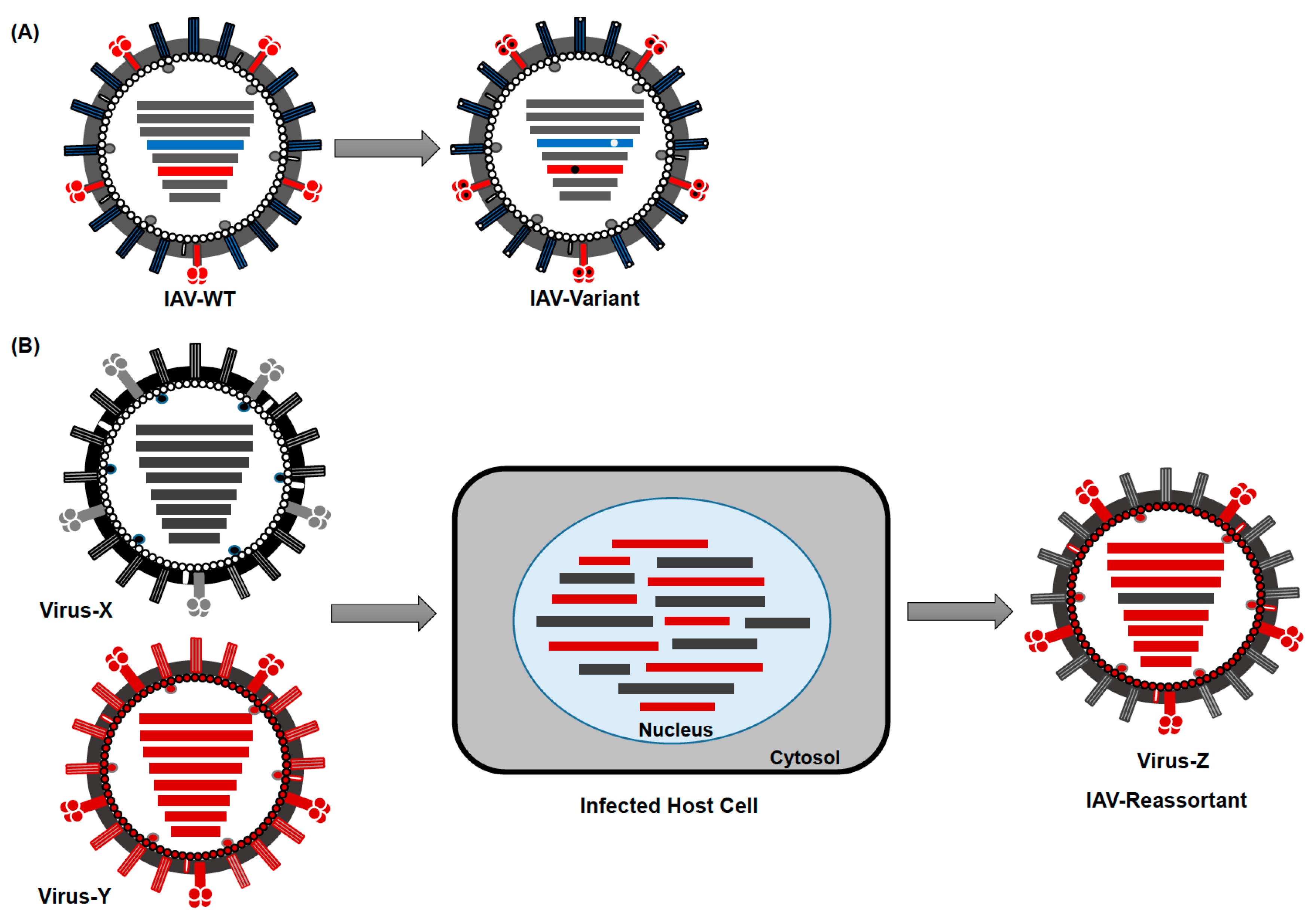
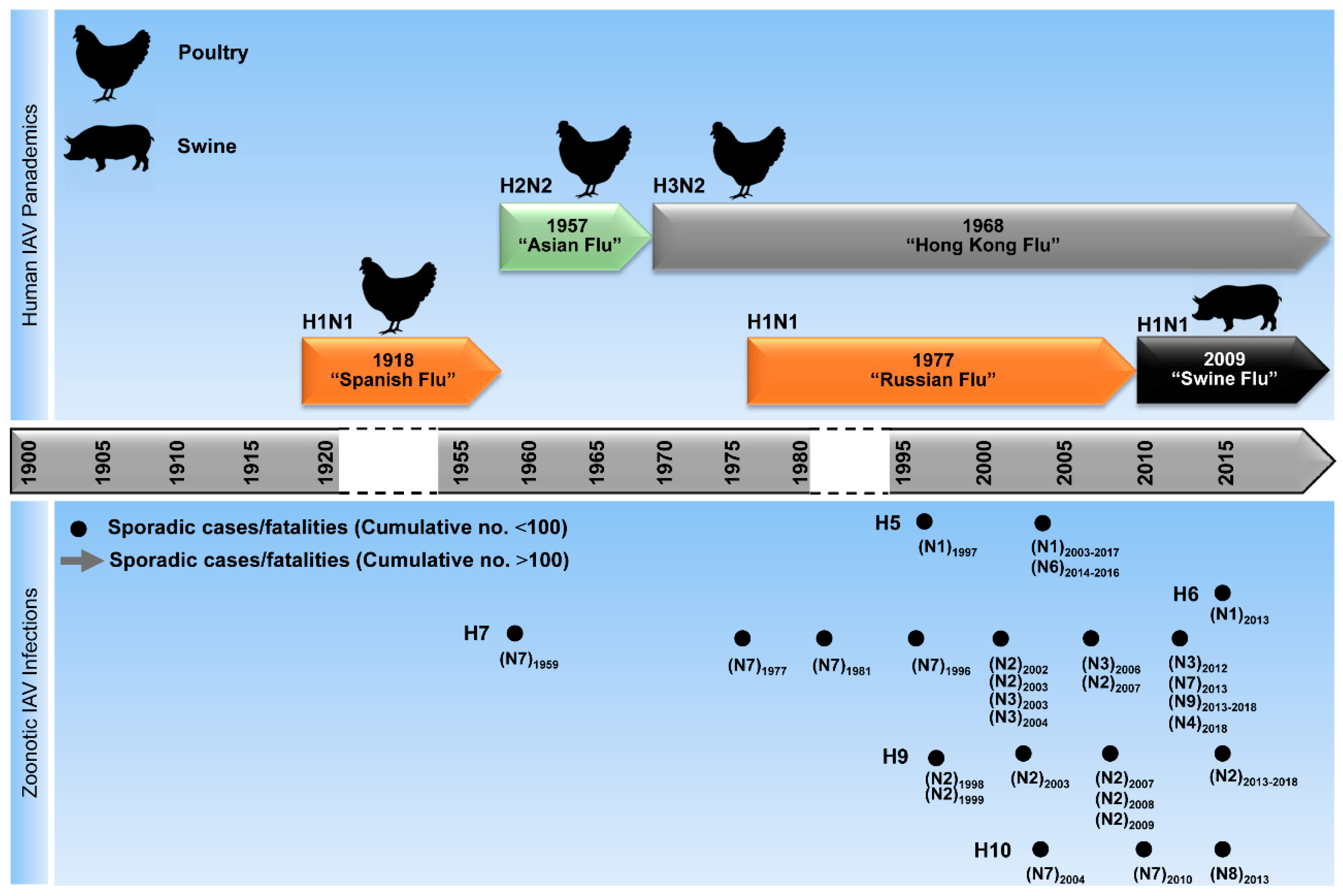
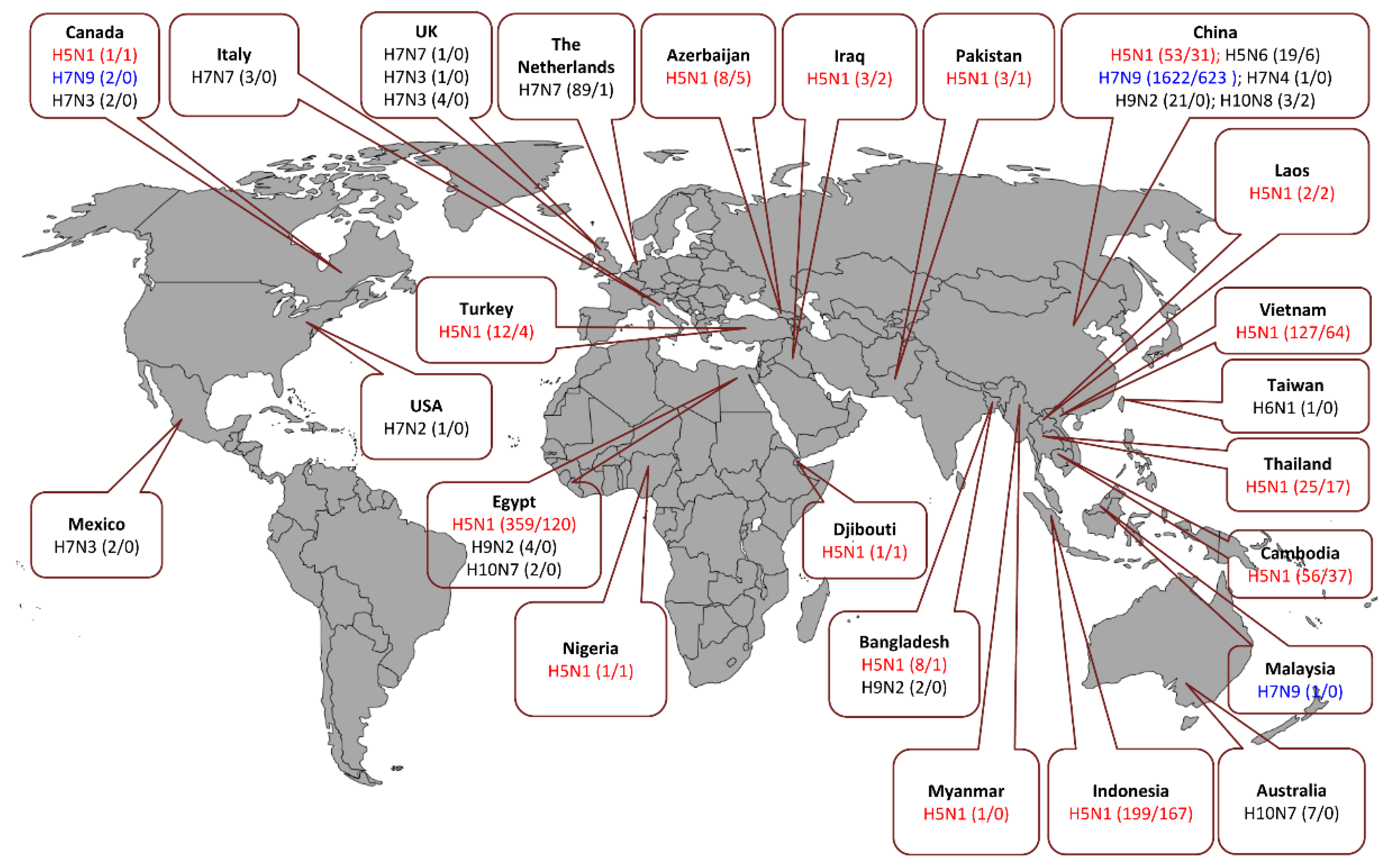
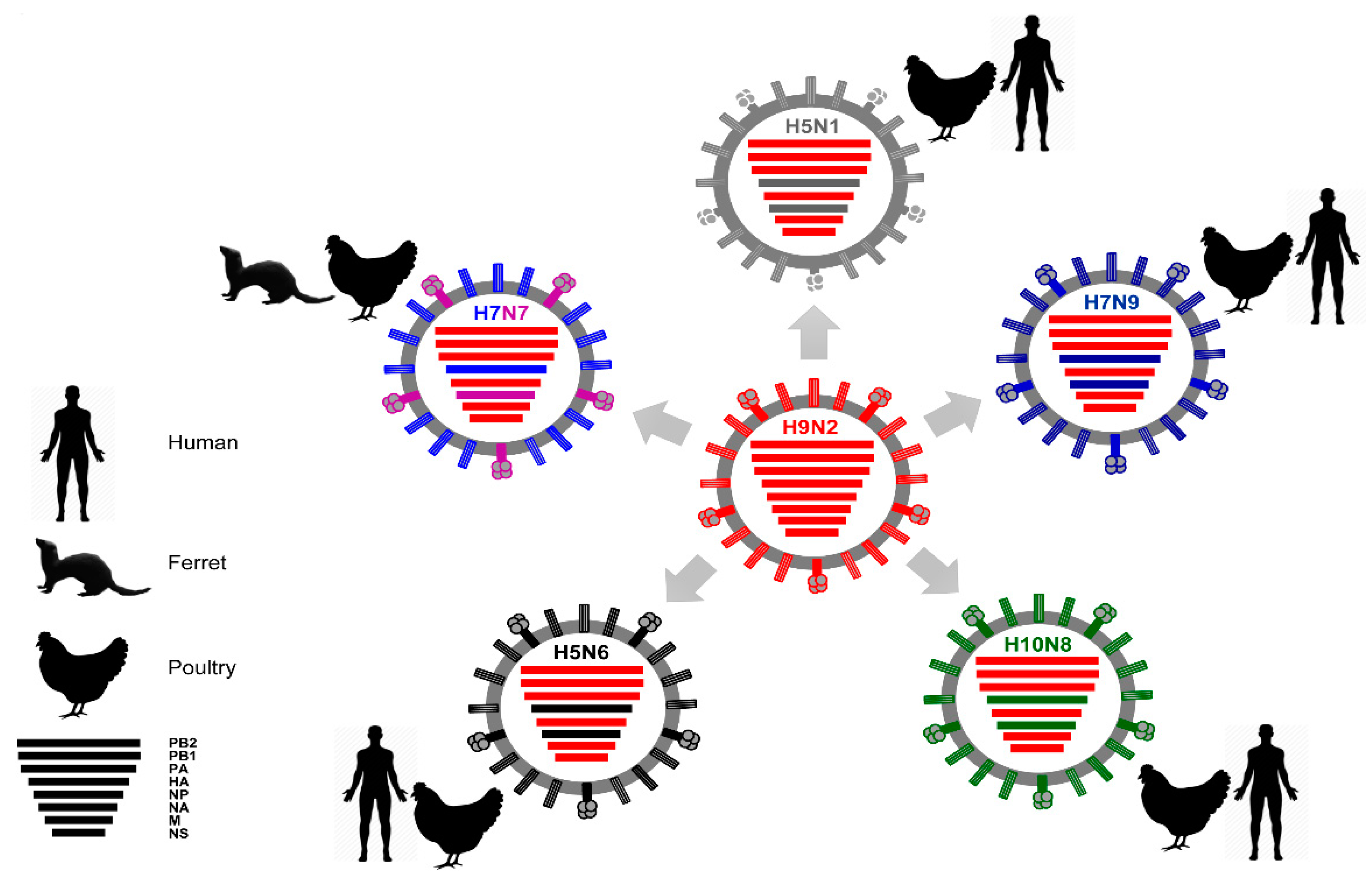
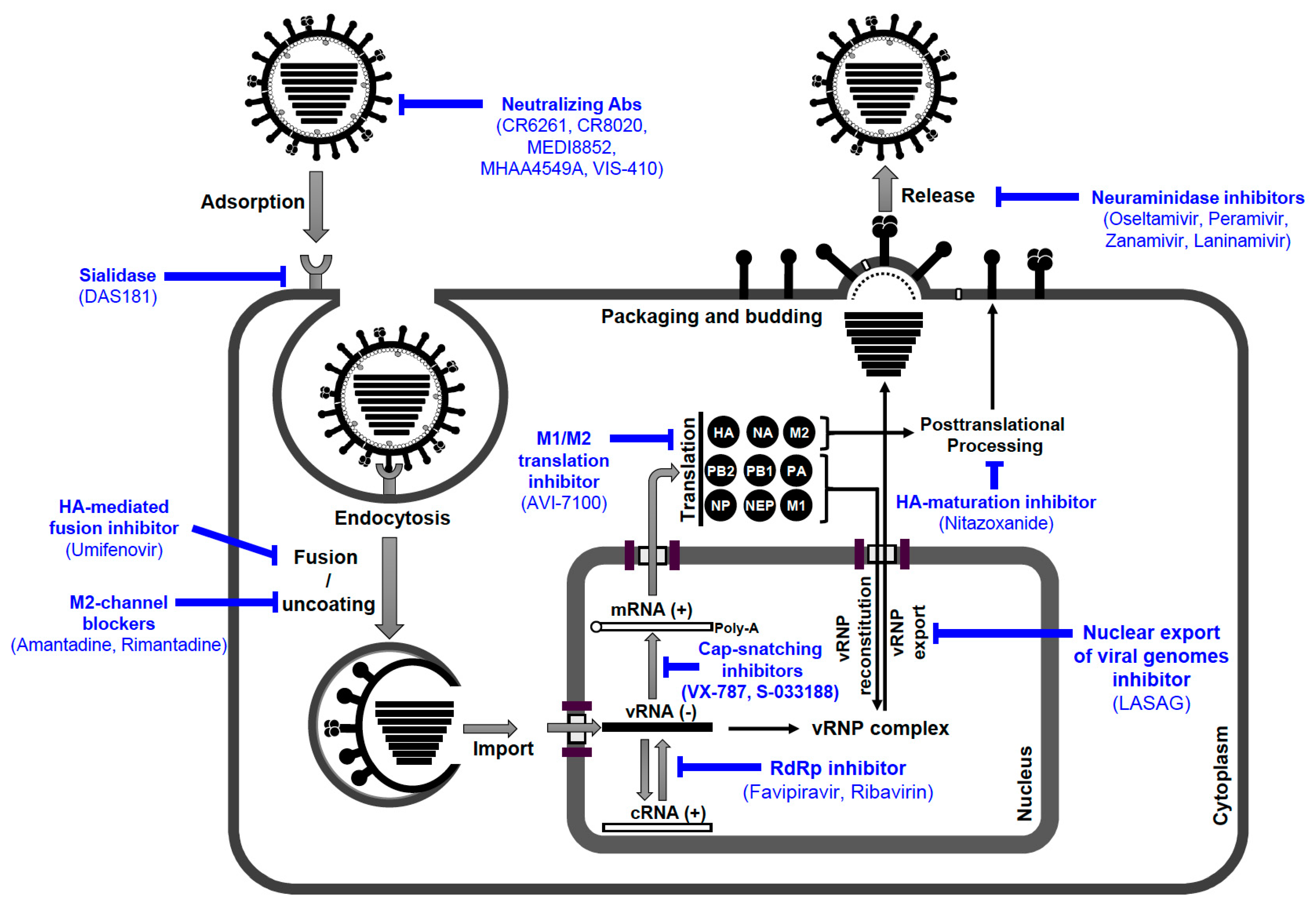
| Segment | vRNA(nt) | Viral Protein(s) | Protein | Molecules per Virion | Main Functions | Ref. | |
|---|---|---|---|---|---|---|---|
| (aa) | (kD) | ||||||
| 1 | 2341 | PB2 | 759 | 80 | 30–60 | (i) recognition and binding to the cellular mRNA cap sequence; (ii) RIG-I-mediated IFN-expression by binding to the mitochondrial antiviral signalling protein (MAVS) | [3,8,9] |
| PB2-S1 *† | 508 | 55 | N/A | (i) inhibition of RIG-I-dependent interferon signaling pathway, (ii) interference with RdRp activity via competitive binding to PB1 | [10] | ||
| 2 | 2341 | PB1 | 757 | 90 | 30–60 | (i) captures of snatched Cap-structure to prime the viral mRNA transcription, (ii) transcribes vRNA into complementary RNA (cRNA) as template for further vRNA synthesis, (iii) initiates vRNA synthesis | [8] |
| PB1-F2 † | 87–90 | 10.5 | N/A | (i) induces apoptosis, (ii) modulates host interferon response, (iii) modulates the susceptibility to secondary bacterial infection | [11,12,13,14,15,16] | ||
| PB1-N40 † | 718 | ≈80 | N/A | maintains the balance between PB1 and PB1-F2 expression | [17,18] | ||
| 3 | 2233 | PA | 716 | 83 | 30–60 | RNA endonuclease to cleave small capped RNA structures to be used for viral mRNA synthesis | [3,8] |
| PA-X † | 252 | 29 | N/A | modulation of the host response and viral virulence | [19,20,21] | ||
| PA-N155 † | 561 | 62 | N/A | Promote viral replication and pathogenicity of IAV | [22,23] | ||
| PA-N182 † | 534 | 60 | N/A | ||||
| 4 | 1778 | HA | 566 | 77 | 500 | (i) receptor binding; (ii) membrane fusion; (iii) major antigen | [3,8] |
| 5 | 1565 | NP | 498 | 55 | 1000 | (i) vRNA binding and protection; (ii) vRNA synthesis by vRNP complex; (iii) nuclear import of vRNP | [3,8] |
| 6 | 1413 | NA | 454 | 56 | 100 | (i) sialidase activity to release the virion progeny; (ii) help the viral particle to penetrate the mucus barrier of the respiratory tract to reach and infect the host cell | [3,8] |
| 7 | 1027 | M1 | 252 | 28 | 3000 | (i) nuclear import and export of vRNPs; (ii) viral assembly, budding and morphogenesis | [3,8,24] |
| M2 * | 97 | 15 | 20–60 | (i) Ion channel activity; (ii) uncoating process | [3,6,8] | ||
| M3 *† | 9 | N/A | N/A | N/A | [25] | ||
| M4 *† | 54 | N/A | N/A | N/A | |||
| M42 *† | 99 | N/A | N/A | Functionally complements M2 | |||
| 8 | 890 | NS1 | 230 | 26 | N/A | (i) vRNP entry by hijacking importin-α; (ii) antagonize cellular antiviral responses including interferons; (iii) support viral mRNA splicing, maturation and translation; (iv) inhibits cellular mRNA maturation and translation | [8,26,27,28,29,30] |
| NEP/NS2 * | 121 | 14 | 130–200 | (i) important for vRNP nuclear export; (ii) regulates vRNA transcription/replication | [31,32,33,34] | ||
| NS3 *† | 187 | 20 | 0 | N/A | [35] | ||
| HA-Subtype | NA-Subtype | Human | Swine | Equine | Domestic Poultry | Waterfowl Shorebirds | Sea Mammals (Seal/Whale) | Bat |
|---|---|---|---|---|---|---|---|---|
| H1 | N1 | H1/N1 | H1/N1 | H1/N1 | H1/N1 | H1 | ||
| H2 | N2 | H2/N2 | H2/N2 | H2/N2 | H2/N2 | N2 | ||
| H3 | N3 | H3/N3 | H3 | H3 | H3/N3 | H3/N3 | H3/N3 | |
| H4 | N4 | N4 | H4 | H4/N4 | H4/N4 | H4 | ||
| H5 | N5 | H5 | H5 | H5/N5 | H5/N5 | N5 | ||
| H6 | N6 | H6/N6 | H6/N6 | H6/N6 | H6/N6 | |||
| H7 | N7 | H7/N7 | H7/N7 | H7/N7 | H7/N7 | H7/N7 | ||
| H8 | N8 | N8 | N8 | H8/N8 | H8/N8 | |||
| H9 | H9 | H9/H9 | H9 | H9/N9 | H9/N9 | N9 | ||
| H10 | N10 | H10 | H10 | H10 | H10 | N10 | ||
| H11 | N11 | H11 | H11 | N11 | ||||
| H12 | H12 | H12 | ||||||
| H13 | H13 | H13 | H13 | |||||
| H14 | H14 | H14 | ||||||
| H15 | H15 | H15 | ||||||
| H16 | H16 | H16 | ||||||
| H17 | H17 | |||||||
| H18 | H18 |
| Wave Number | Period | Phenotype | Cases | Fatalities | Fatality Rate |
|---|---|---|---|---|---|
| 1 | February 2013–September 2013 | LPAIV | 134 | 45 | 34% |
| 2 | October 2013–September 2014 | LPAIV | 306 | 131 | 43% |
| 3 | October 2014–September 2015 | LPAIV | 219 | 102 | 47% |
| 4 | October 2015–September 2016 | LPAIV | 114 | 47 | 41% |
| 5 | October 2016–September 2017 | LPAIV/HPAIV | 848 | 295 | 35% |
| 6 | Since October 2017 | LPAIV/HPAIV | 4 | 3 | 75% |
| NA Active Site (N2 Numbering) | Influenza Virus Type/Subtype | Resistance Marker (N2 Numbering)/Location within NA Active Site | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Catalytic Residues (CR) | Framework Residues (FWR) | Oseltamivir | Zanamivir | Peramivir | Laninamivir | ||||||
| Amino Acid (aa) | Site | Amino Acid (aa) | Site | Amino Acid (aa) | Site | Amino Acid (aa) | Site | ||||
| 118R 151D 152R 224R 276E 292R 371R 406Y | 119E 156R 178W 179S 198D 222I 227E 274H 277E 294N 425E | Non-seasonal A/H5N1 † and A/H7N9 | A/H5N1 | V116A | - | V116A | - | E119D/G | FWR | ||
| E119A/D | FWR | E119A/D/G | FWR | H274Y | FWR | ||||||
| Q136L | - | Q136L | - | N294S | FWR | ||||||
| D198G | FWR | D198G | FWR | E119A/D/G + H274Y | FWR | ||||||
| I222M | FWR | N294S | FWR | I222M/V + H274Y | FWR | ||||||
| S246N | - | K432T | - | ||||||||
| H274Y | FWR | E119A/D/G + H274Y | FWR | ||||||||
| N294S | FWR | ||||||||||
| I117V + I314V | - | ||||||||||
| E119A/D/G + H274Y | FWR | ||||||||||
| I222L + S246N | FWR/- | ||||||||||
| I222M/V + H274Y | FWR | ||||||||||
| A/H7N9 | E119A/D | FWR | E119A/D/G | FWR | E119A/G | FWR | E119A | FWR | |||
| I222K/R | FWR | Q136K | - | E119D | FWR | E119D/G | FWR | ||||
| T247P | - | I222K | FWR | Q136K | - | Q136K | - | ||||
| H274Y | FWR | T247P | - | I222K | FWR | R152K | CR | ||||
| E276D | CR | E276D | CR | E276D | CR | I222K/R | FWR | ||||
| R292K | CR | R292K | CR | R292K | CR | E276D | CR | ||||
| R371K | CR | N294S | FWR | R371K | CR | R292K | CR | ||||
| E119V + E222V | FWR | R371K | CR | R371K | CR | ||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mostafa, A.; Abdelwhab, E.M.; Mettenleiter, T.C.; Pleschka, S. Zoonotic Potential of Influenza A Viruses: A Comprehensive Overview. Viruses 2018, 10, 497. https://doi.org/10.3390/v10090497
Mostafa A, Abdelwhab EM, Mettenleiter TC, Pleschka S. Zoonotic Potential of Influenza A Viruses: A Comprehensive Overview. Viruses. 2018; 10(9):497. https://doi.org/10.3390/v10090497
Chicago/Turabian StyleMostafa, Ahmed, Elsayed M. Abdelwhab, Thomas C. Mettenleiter, and Stephan Pleschka. 2018. "Zoonotic Potential of Influenza A Viruses: A Comprehensive Overview" Viruses 10, no. 9: 497. https://doi.org/10.3390/v10090497
APA StyleMostafa, A., Abdelwhab, E. M., Mettenleiter, T. C., & Pleschka, S. (2018). Zoonotic Potential of Influenza A Viruses: A Comprehensive Overview. Viruses, 10(9), 497. https://doi.org/10.3390/v10090497







