Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses
Abstract
:1. Introduction
2. Material and Methods
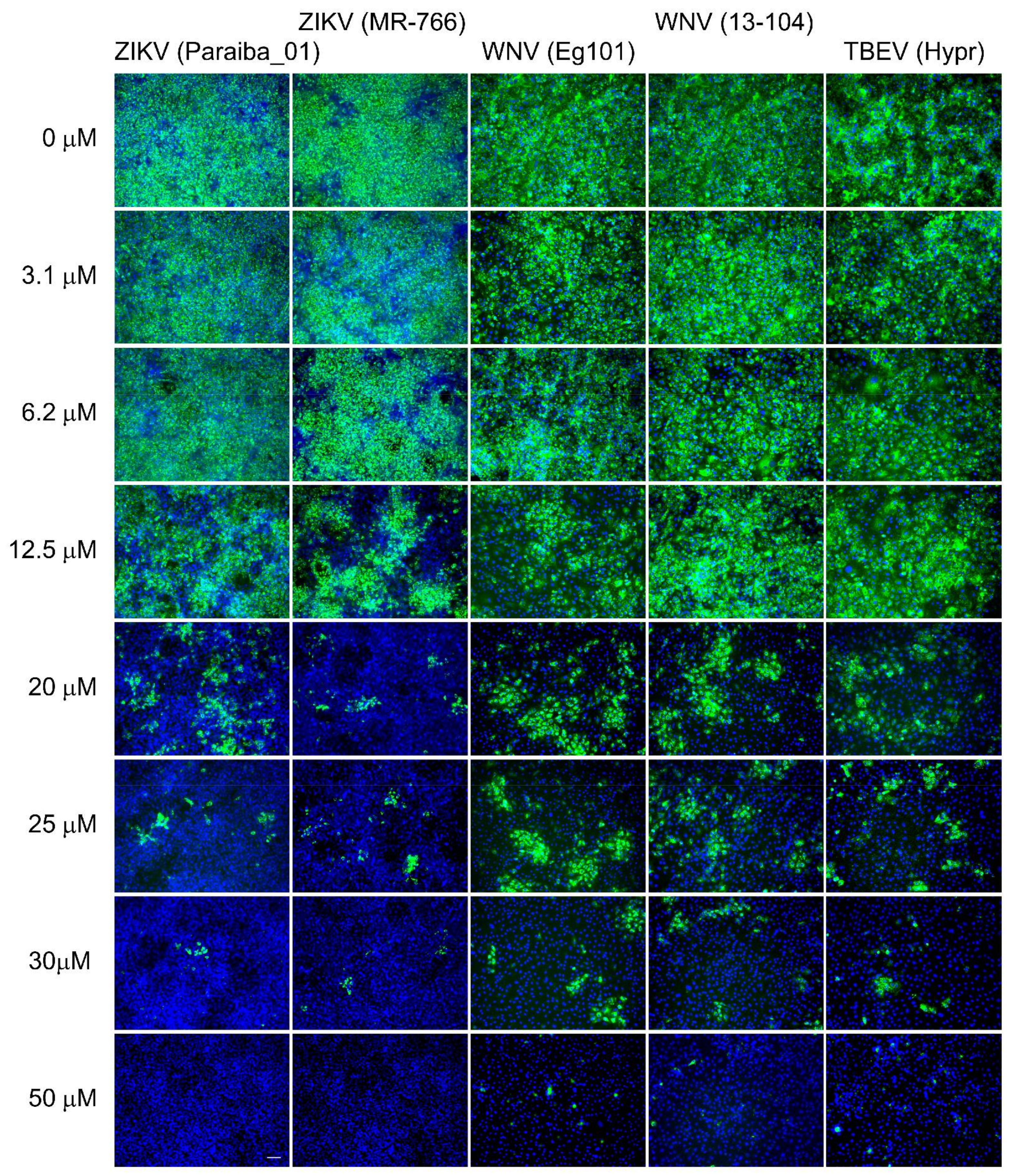
3. Results and Discussion
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Funding
References
- Baier, A. Flaviviral infections and potential targets for antiviral therapy. In Flavivirus Encephalitis, 1st ed.; Ruzek, D., Ed.; InTech: Rijeka, Croatia, 2011; pp. 89–104. ISBN 978-953-307-669-0. [Google Scholar]
- Lazear, H.M.; Stringer, E.M.; de Silva, A.M. The Emerging Zika Virus Epidemic in the Americas Research Priorities. JAMA 2016, 315, 1945–1946. [Google Scholar] [CrossRef] [PubMed]
- Hayes, E.B.; Sejvar, J.; Zaki, S.R.; Lanciotti, R.S.; Bode, A.V.; Campbell, G.L. Virology, pathology, and clinical manifestations of West Nile virus disease. Emerg. Infect. Dis. 2005, 11, 1174–1179. [Google Scholar] [CrossRef] [PubMed]
- Samuel, M.A.; Diamond, M.S. Pathogenesis of West Nile virus infection: A balance between virulence, innate and adaptive immunity, and viral evasion. J. Virol. 2006, 80, 9349–9360. [Google Scholar] [CrossRef] [PubMed]
- Malone, R.W.; Homan, J.; Callahan, M.V.; Glasspool-Malone, J.; Damodaran, L.; Schneider Ade, B.; Zimler, R.; Talton, J.; Cobb, R.R.; Ruzic, I.; et al. Zika Virus: Medical Countermeasure Development Challenges. PLoS Negl. Trop. Dis. 2016, 10, e0004530. [Google Scholar] [CrossRef] [PubMed]
- Panisheva, E.K.; Mikerova, N.I.; Nikolayeva, I.S.; Fomina, A.N.; Cherkasova, A.A.; Golovanova, E.A.; Krylova, L.Y. Synthesis and Antiviral Activity of 5-Hydroxyindole Derivatives. Pharm. Chem. 1988, 22, 1455–1458. [Google Scholar]
- Boriskin, Y.S.; Leneva, I.A.; Pecheur, E.I.; Polyak, S.J. Arbidol: A broad-spectrum antiviral compound that blocks viral fusion. Curr. Med. Chem. 2008, 15, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Pecheur, E.I.; Borisevich, V.; Halfmann, P.; Morrey, J.D.; Smee, D.F.; Prichard, M.; Mire, C.E.; Kawaoka, Y.; Geisbert, T.W.; Polyak, S.J. The Synthetic Antiviral Drug Arbidol Inhibits Globally Prevalent Pathogenic Viruses. J. Virol. 2016, 90, 3086–3092. [Google Scholar] [CrossRef] [PubMed]
- Villalain, J. Membranotropic Effects of Arbidol, a Broad Anti-Viral Molecule, on Phospholipid Model Membranes. J. Phys. Chem. B 2010, 114, 8544–8554. [Google Scholar] [CrossRef] [PubMed]
- Leneva, I.A.; Russell, R.J.; Boriskin, Y.S.; Hay, A.J. Characteristics of arbidol-resistant mutants of influenza virus: Implications for the mechanism of anti-influenza action of arbidol. Antivir. Res. 2009, 81, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Boriskin, Y.S.; Pecheur, E.I.; Polyak, S.J. Arbidol: A broad-spectrum antiviral that inhibits acute and chronic HCV infection. Virol. J. 2006, 3, 56. [Google Scholar] [CrossRef] [PubMed]
- Pecheur, E.I.; Lavillette, D.; Alcaras, F.; Molle, J.; Boriskin, Y.S.; Roberts, M.; Cosset, F.L.; Polyak, S.J. Biochemical mechanism of hepatitis C virus inhibition by the broad-spectrum antiviral arbidol. Biochemistry 2007, 46, 6050–6059. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Xiong, H.R.; Lu, L.; Liu, Y.Y.; Luo, F.; Hou, W.; Yang, Z.Q. Antiviral and anti-inflammatory activity of arbidol hydrochloride in influenza A (H1N1) virus infection. Acta Pharmacol. Sin. 2013, 34, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.N.; Fan, T.W.M. Regulation of mammalian nucleotide metabolism and biosynthesis. Nucleic Acids Res. 2015, 43, 2466–2485. [Google Scholar] [CrossRef] [PubMed]
- Peterson, L.W.; McKenna, C.E. Prodrug approaches to improving the oral absorption of antiviral nucleotide analogues. Expert Opin. Drug Deliv. 2009, 6, 405–420. [Google Scholar] [CrossRef] [PubMed]
- Kozuch, O.; Mayer, V. Pig Kidney Epithelial (Ps) Cells—Perfect Tool for Study of Flaviviruses and Some Other Arboviruses. Acta Virol. 1975, 19, 498. [Google Scholar] [PubMed]
- Ruzek, D.; Vancova, M.; Tesarova, M.; Ahantarig, A.; Kopecky, J.; Grubhoffer, L. Morphological changes in human neural cells following tick-borne encephalitis virus infection. J. Gen. Virol. 2009, 90, 1649–1658. [Google Scholar] [CrossRef] [PubMed]
- DeMadrid, A.T.; Porterfield, J.S. A Simple Micro-Culture Method for Study of Group B Arboviruses. Bull. World Health Organ. 1969, 40, 113–121. [Google Scholar]
- Eyer, L.; Valdés, J.J.; Gil, V.A.; Nencka, R.; Hřebabecký, H.; Šála, M.; Salát, J.; Černý, J.; Palus, M.; de Clercq, E.; Růžek, D. Nucleoside Inhibitors of Tick-Borne Encephalitis Virus. Antimicrob. Agents Chemother. 2015, 59, 5483–5493. [Google Scholar] [CrossRef] [PubMed]
- Chapel, C.; Zitzmann, N.; Zoulim, F.; Durantel, D. Virus morphogenesis and viral entry as alternative targets for novel hepatitis C antivirals. Future Virol. 2006, 1, 197–209. [Google Scholar] [CrossRef]
- Eyer, L.; Nencka, R.; de Clercq, E.; Seley-Radtke, K.; Ruzek, D. Nucleoside analogs as a rich source of antiviral agents active against arthropod-borne Flaviviruses. Antivir. Chem. Chemother. 2018. [Google Scholar] [CrossRef] [PubMed]
- Becher, F.; Landman, R.; Mboup, S.; Kane, C.N.; Canestri, A.; Liegeois, F.; Vray, M.; Prevot, M.H.; Leleu, G.; Benech, H. Monitoring of didanosine and stavudine intracellular trisphosphorylated anabolite concentrations in HIV-infected patients. AIDS 2004, 18, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.Y.; Shirasaka, T.; Johns, D.G.; Broder, S.; Mitsuya, H. Differential Phosphorylation of Azidothymidine, Dideoxycytidine, and Dideoxyinosine in Resting and Activated Peripheral-Blood Mononuclear-Cells. J. Clin. Investig. 1993, 91, 2326–2333. [Google Scholar] [CrossRef] [PubMed]
- Mumtaz, N.; Jimmerson, L.C.; Bushman, L.R.; Kiser, J.J.; Aron, G.; Reusken, C.B.E.M.; Koopmans, M.P.G.; van Kampen, J.J.A. Cell-line dependent antiviral activity of sofosbuvir against Zika virus. Antivir. Res. 2017, 146, 161–163. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.Q.; Zhang, N.N.; Li, C.F.; Tian, M.; Hao, J.N.; Xie, X.P.; Shi, P.Y.; Qin, C.F. Adenosine Analog NITD008 Is a Potent Inhibitor of Zika Virus. Open Forum Infect. Dis. 2016, 3, ofw175. [Google Scholar] [CrossRef] [PubMed]
- Eyer, L.; Nencka, R.; Huvarová, I.; Palus, M.; Joao Alves, M.; Gould, E.A.; de Clercq, E.; Růžek, D. Nucleoside Inhibitors of Zika Virus. J. Infect. Dis. 2016, 214, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Julander, J.G.; Siddharthan, V.; Evans, J.; Taylor, R.; Tolbert, K.; Apuli, C.; Stewart, J.; Collins, P.; Gebre, M.; Neilson, S.; et al. Efficacy of the broad-spectrum antiviral compound BCX4430 against Zika virus in cell culture and in a mouse model. Antivir. Res. 2017, 137, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Sacramento, C.Q.; de Melo, G.R.; de Freitas, C.S.; Rocha, N.; Hoelz, L.V.; Miranda, M.; Fintelman-Rodrigues, N.; Marttorelli, A.; Ferreira, A.C.; Barbosa-Lima, G.; et al. The clinically approved antiviral drug sofosbuvir inhibits Zika virus replication. Sci. Rep. 2017, 7, 40920. [Google Scholar] [CrossRef] [PubMed]
- Leneva, I.A.; Fedyakina, I.T.; Guskova, T.A.; Glushkov, R.G. Sensitivity of various influenza virus strains to arbidol. Influence of arbidol combination with different antiviral drugs on reproduction of influenza virus A. Terapevticheskii arkhiv 2005, 77, 84–88. [Google Scholar] [PubMed]
- Delogu, I.; Pastorino, B.; Baronti, C.; Nougairede, A.; Bonnet, E.; De Lamballerie, X. In vitro antiviral activity of arbidol against Chikungunya virus and characteristics of a selected resistant mutant. Antivir. Res. 2011, 90, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Oestereich, L.; Rieger, T.; Neumann, M.; Bernreuther, C.; Lehmann, M.; Krasemann, S.; Wurr, S.; Emmerich, P.; de Lamballerie, X.; Ölschläger, S.; Günther, S. Evaluation of Antiviral Efficacy of Ribavirin, Arbidol, and T-705 (Favipiravir) in a Mouse Model for Crimean-Congo Hemorrhagic Fever. PLoS Negl. Trop. Dis. 2014, 8, e2804. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.Y.; Luo, F.; Shi, L.Q.; Zhong, Q.; Liu, Y.J.; Yang, Z.Q. Efficacy of arbidol on lethal hantaan virus infections in suckling mice and in vitro. Acta Pharmacol. Sin. 2009, 30, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Xiong, H.; He, J.; Deng, H.; Li, Q.; Zhong, Q.; Hou, W.; Cheng, L.; Xiao, H.; Yang, Z. Antiviral activity of arbidol against influenza A virus, respiratory syncytial virus, rhinovirus, coxsackie virus and adenovirus in vitro and in vivo. Arch. Virol. 2007, 152, 1447–1455. [Google Scholar] [CrossRef] [PubMed]
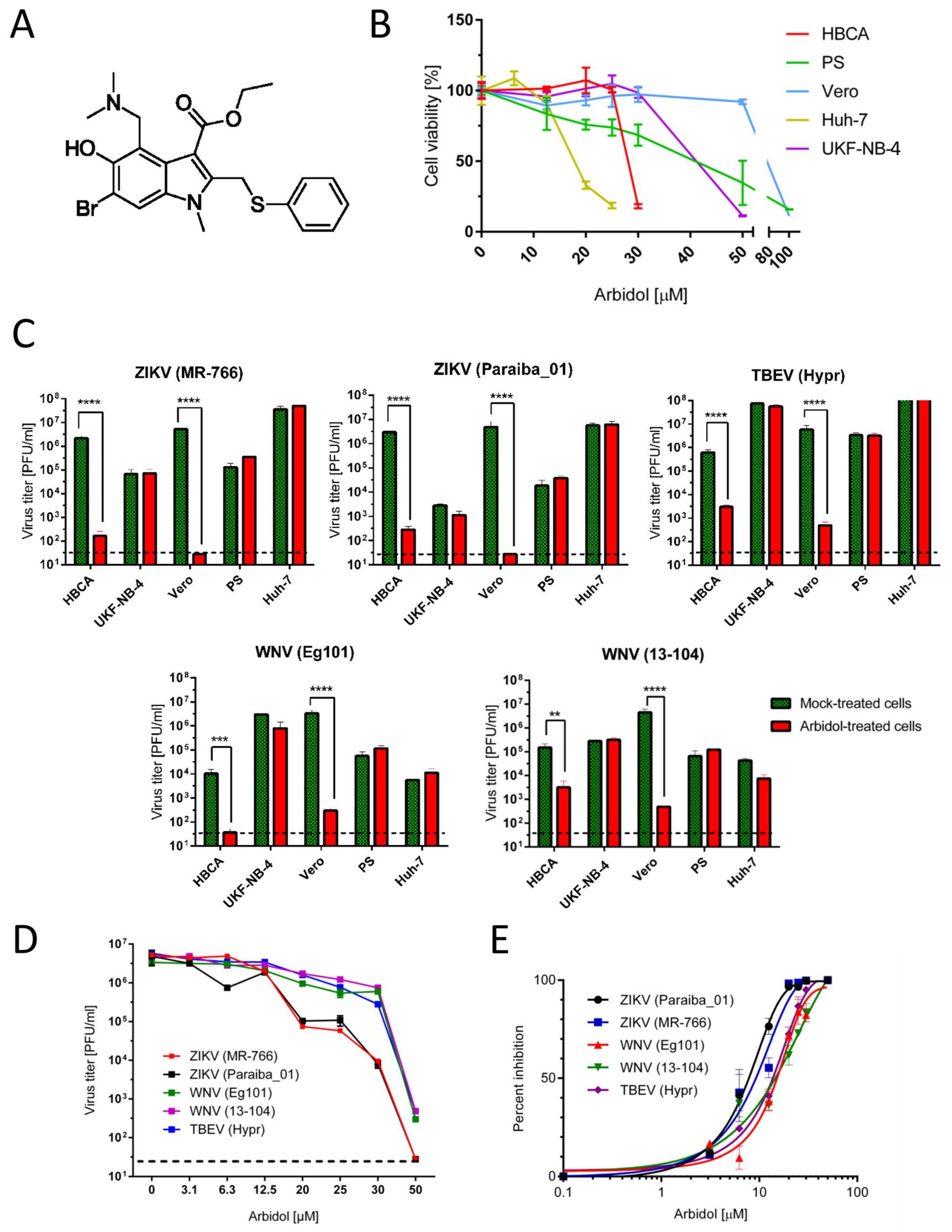
| Cell Line | CC50 (µM) a |
|---|---|
| Human brain cortical astrocytes (HBCA) | 24.78 ± 0.01 |
| Human neuroblastoma UKF-NB-4 cells | 46.99 ± 0.10 |
| Vero cells | 89.72 ± 0.19 |
| Human hepatocarcinoma cells (Huh-7) | 18.69 ± 0.10 |
| Porcine kidney stable cells (PS) | 46.81 ± 1.65 |
| Virus | Strain | EC50 (µM) a,b | CC50 (µM) a,c | SI (CC50/EC50) |
|---|---|---|---|---|
| ZIKV | MR-766 | 12.09 ± 0.77 | 89.72 ± 0.19 | 7.42 |
| Paraiba_01 | 10.57 ± 0.74 | 8.49 | ||
| WNV | Eg101 | 18.78 ± 0.21 | 4.78 | |
| 13-104 | 19.16 ± 0.29 | 4.68 | ||
| TBEV | Hypr | 18.67 ± 0.15 | 4.81 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haviernik, J.; Štefánik, M.; Fojtíková, M.; Kali, S.; Tordo, N.; Rudolf, I.; Hubálek, Z.; Eyer, L.; Ruzek, D. Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses. Viruses 2018, 10, 184. https://doi.org/10.3390/v10040184
Haviernik J, Štefánik M, Fojtíková M, Kali S, Tordo N, Rudolf I, Hubálek Z, Eyer L, Ruzek D. Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses. Viruses. 2018; 10(4):184. https://doi.org/10.3390/v10040184
Chicago/Turabian StyleHaviernik, Jan, Michal Štefánik, Martina Fojtíková, Sabrina Kali, Noël Tordo, Ivo Rudolf, Zdeněk Hubálek, Luděk Eyer, and Daniel Ruzek. 2018. "Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses" Viruses 10, no. 4: 184. https://doi.org/10.3390/v10040184
APA StyleHaviernik, J., Štefánik, M., Fojtíková, M., Kali, S., Tordo, N., Rudolf, I., Hubálek, Z., Eyer, L., & Ruzek, D. (2018). Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses. Viruses, 10(4), 184. https://doi.org/10.3390/v10040184






