DNA Vaccine-Encoded Flagellin Can Be Used as an Adjuvant Scaffold to Augment HIV-1 gp41 Membrane Proximal External Region Immunogenicity
Abstract
1. Introduction
2. Materials and Methods
2.1. Construction of FliC and HIV-1 gp41 Expression Vectors
2.2. Cell Culture
2.3. Transfection and Cell Lysate/Supernatant Preparation
2.4. SDS-PAGE, Immunoblotting and Deglycosylation Studies
2.5. Secretion ELISA
2.6. TLR5 Agonist Activity Assays
2.6.1. HEK-Blue-hTLR5
2.6.2. Transcriptional Activation of IL-1β in Monocyte/Macrophage Cell Lines
2.7. ELISA to Detect Binding by Monoclonal bnAb
2.8. Mice and Immunizations
2.9. ELISA to Detect Mouse Anti-gp41 IgG Response
3. Results
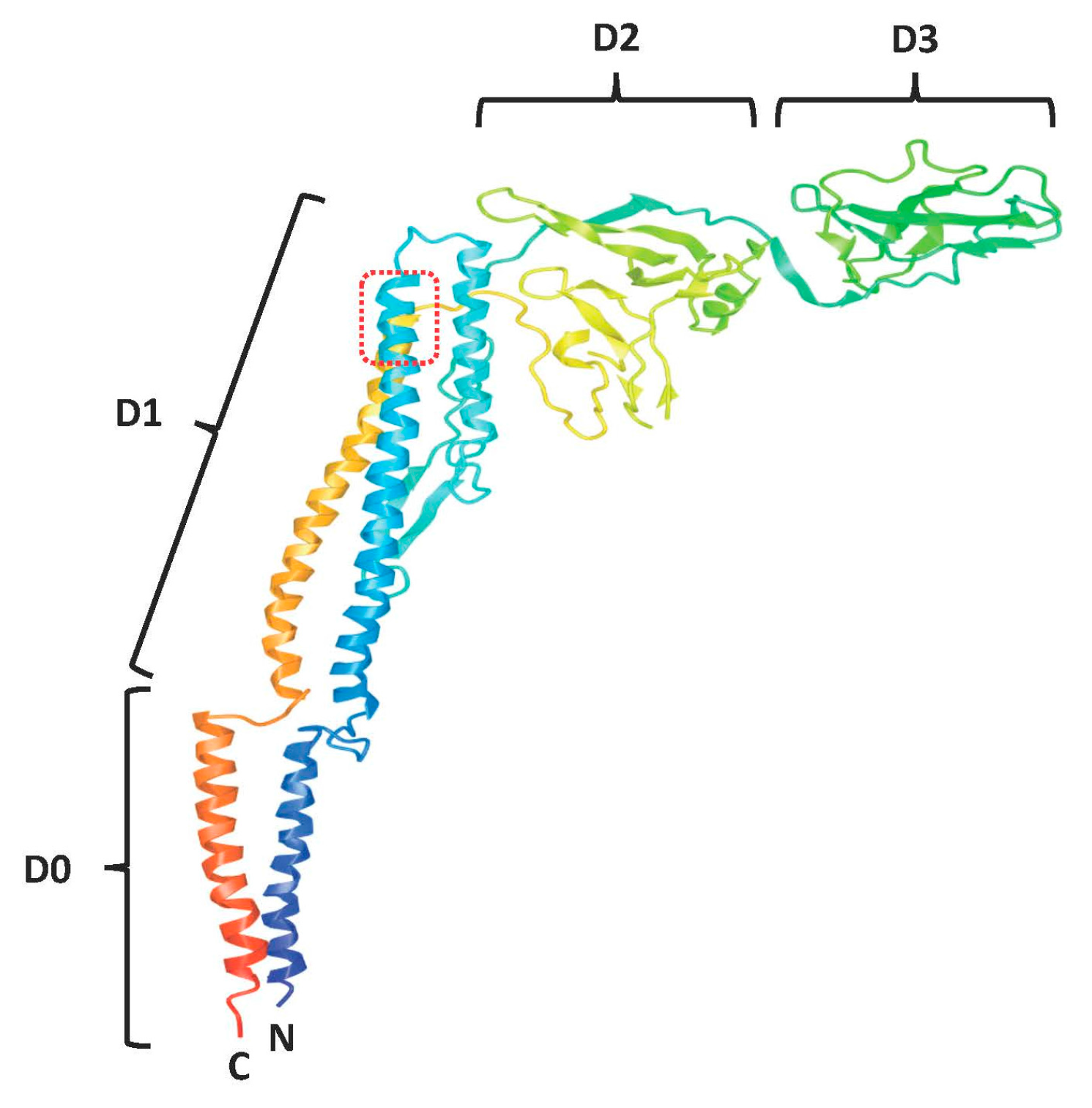
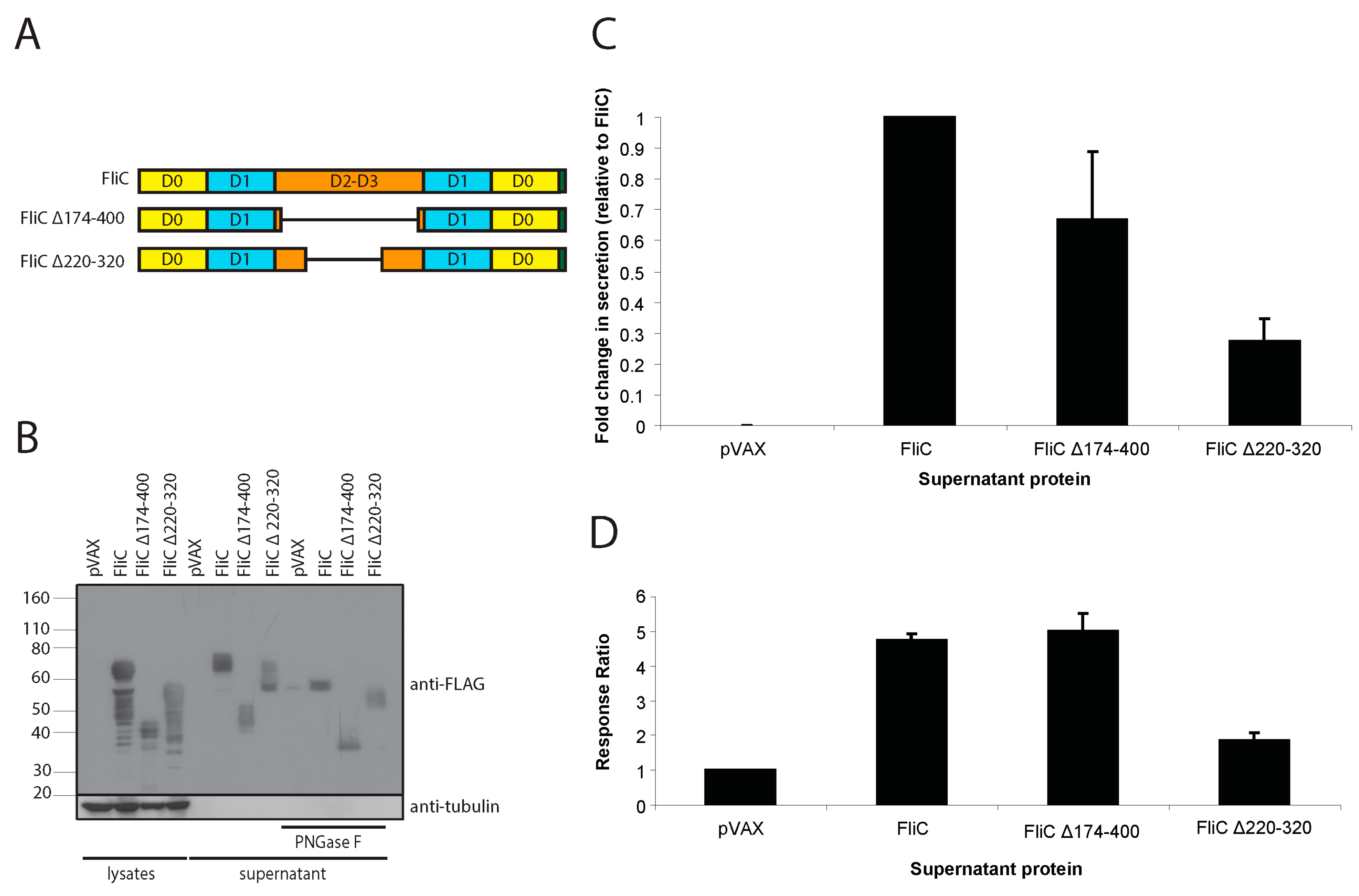
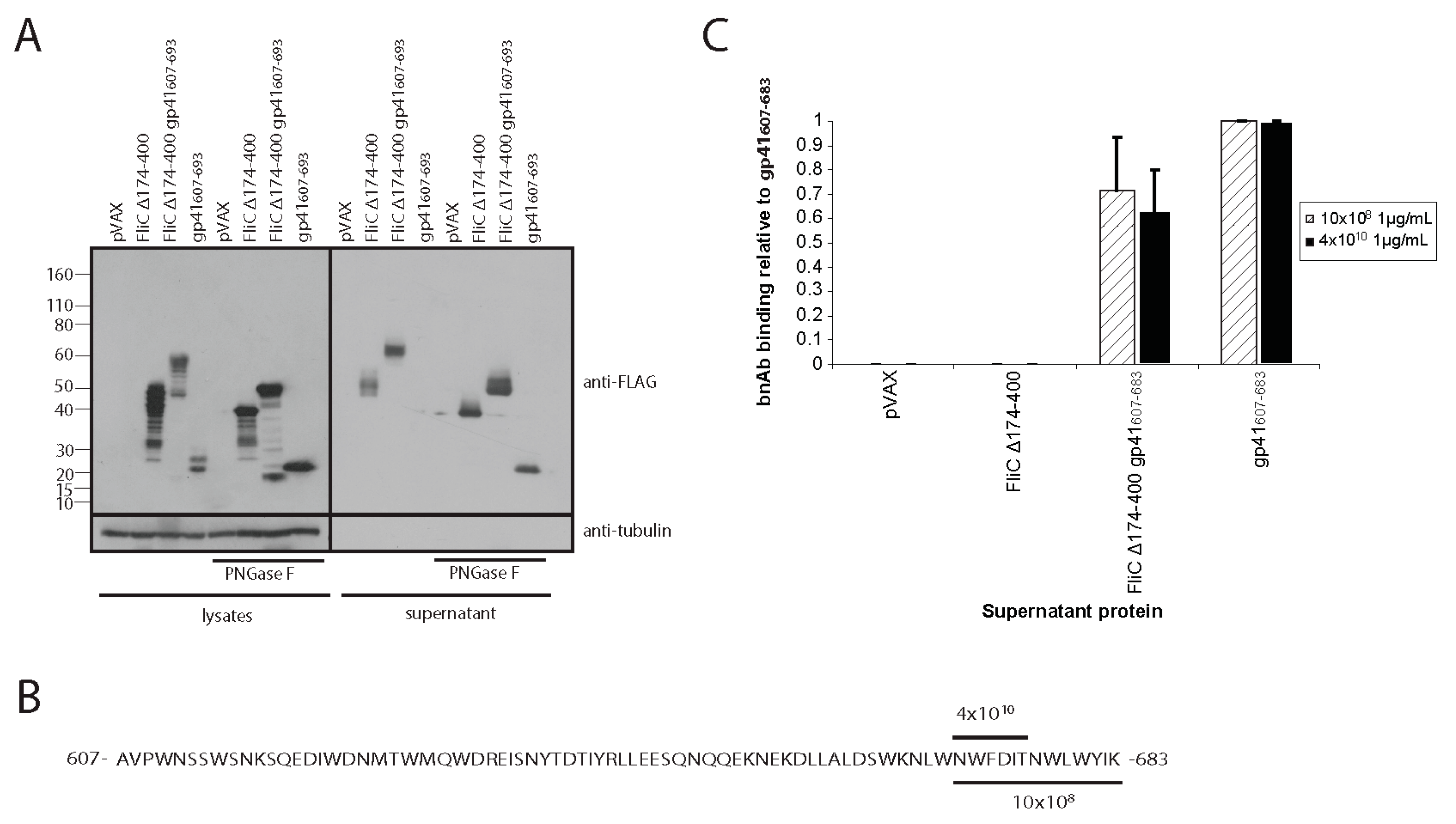
3.1. Codon-Optimized Salmonella enterica Subsp. enterica Serovar Typhi FliC Can Be Expressed in and Effectively Secreted from Mammalian Cells
3.2. Mammalian Cell-Expressed and Secreted FliC Retains TLR5 Agonist Activity
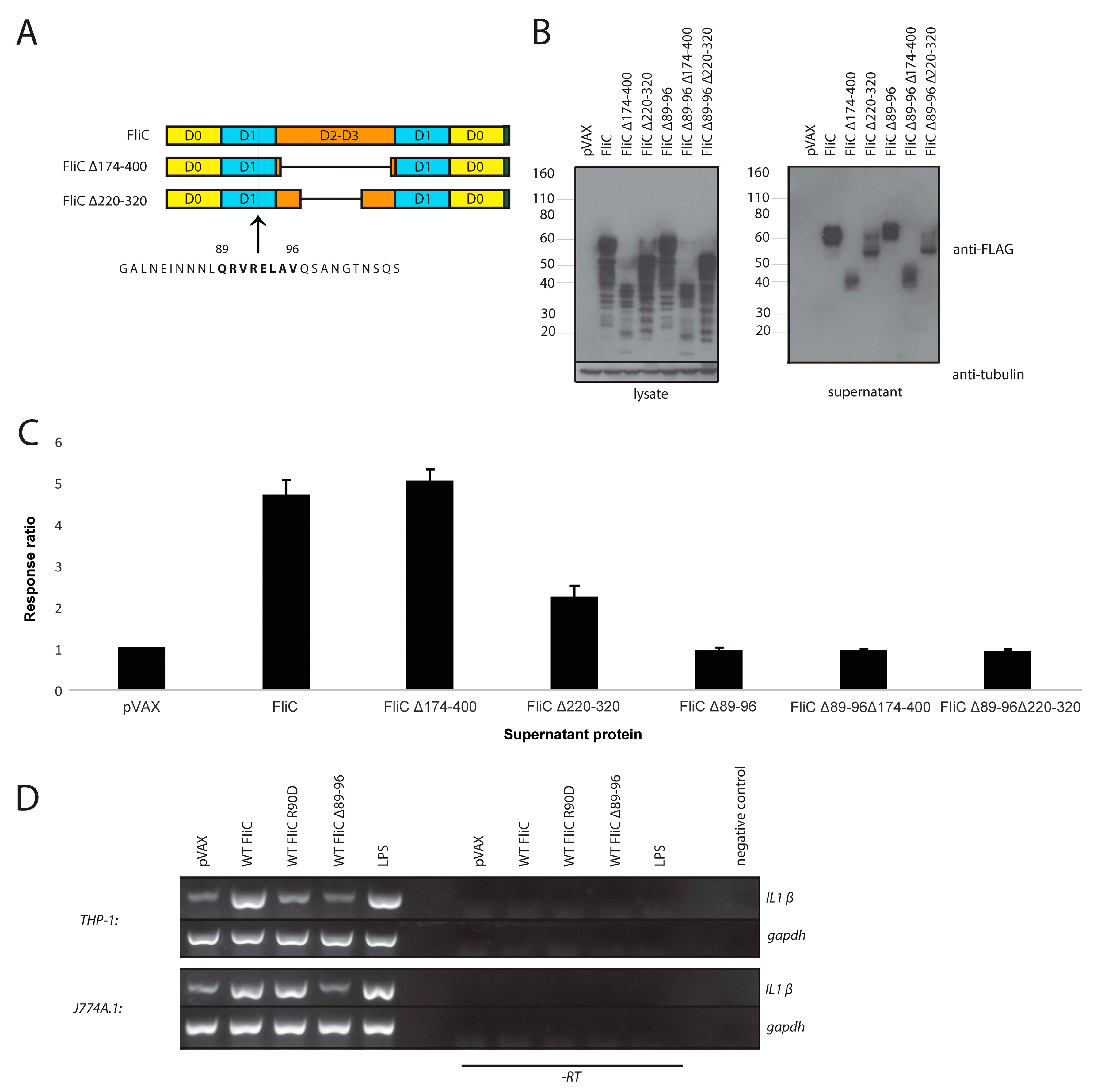
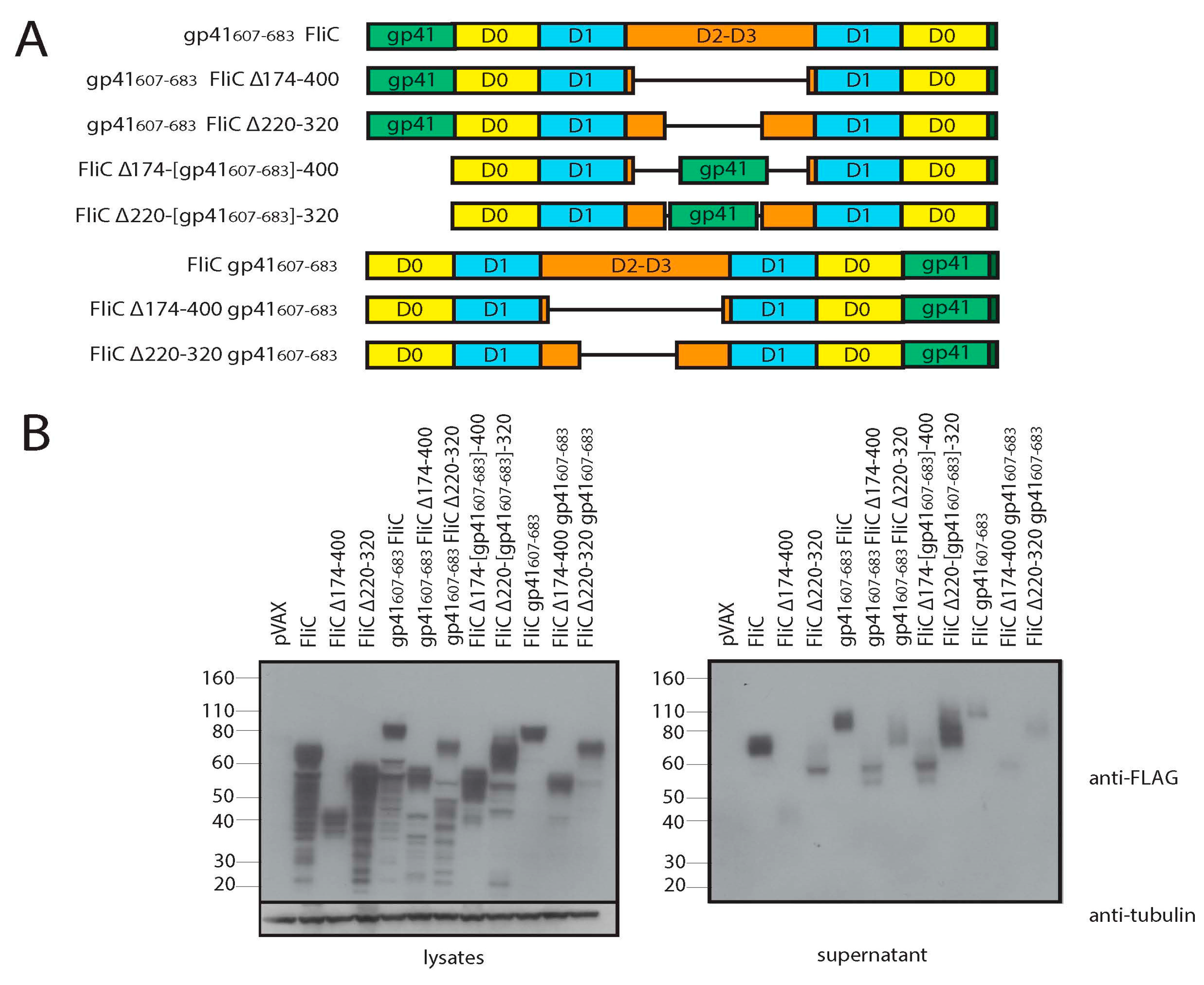
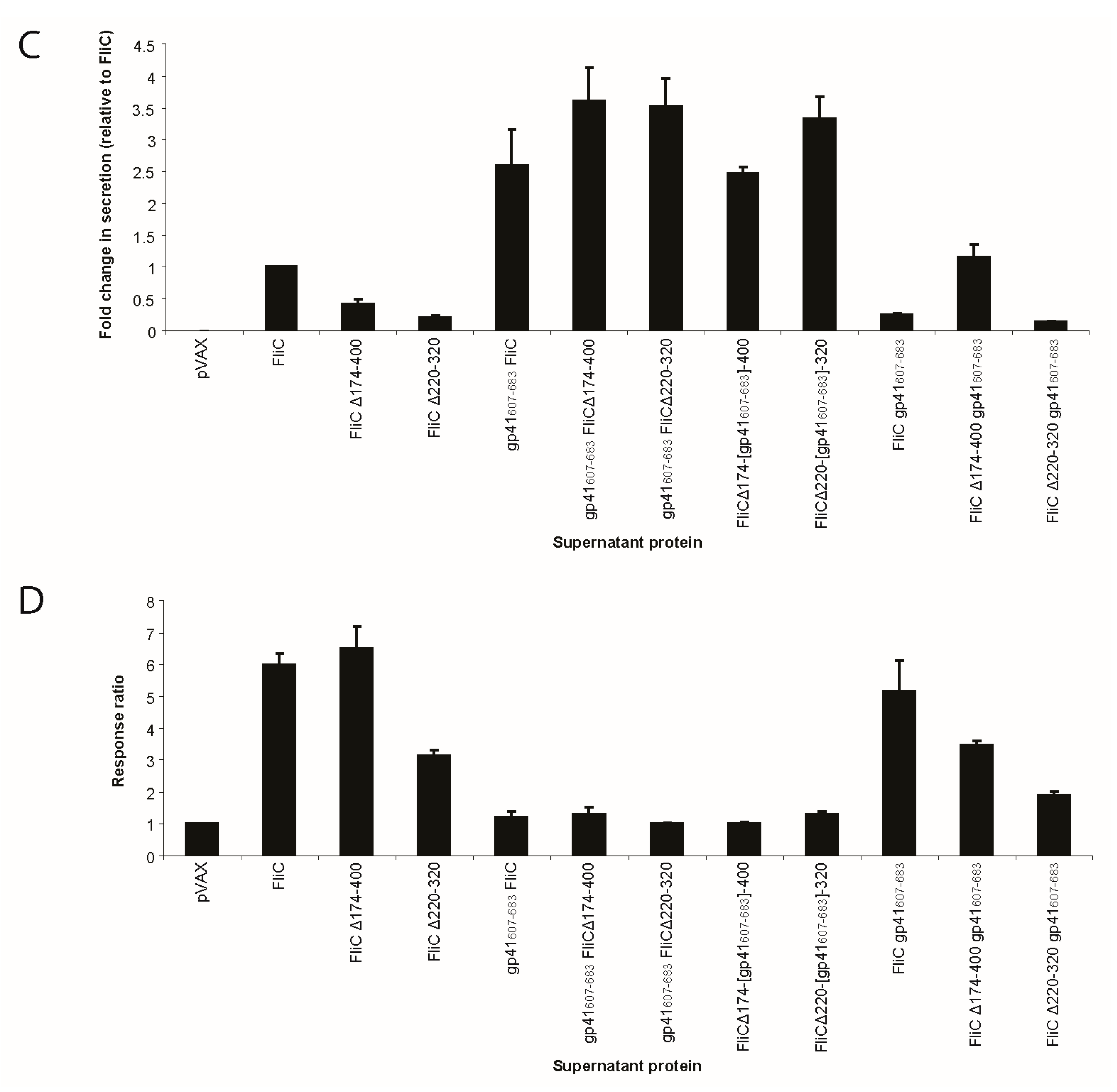
3.3. Adding gp41 MPER (gp41607–683) to the C Terminus of FliCΔ174–400 Results in a Secreted Protein that Maintains TLR5 Agonist Activity
3.4. Adding gp41 MPER (gp41607–683) to the C Terminus of FliCΔ174–400 Maintains gp41 Antigenicity
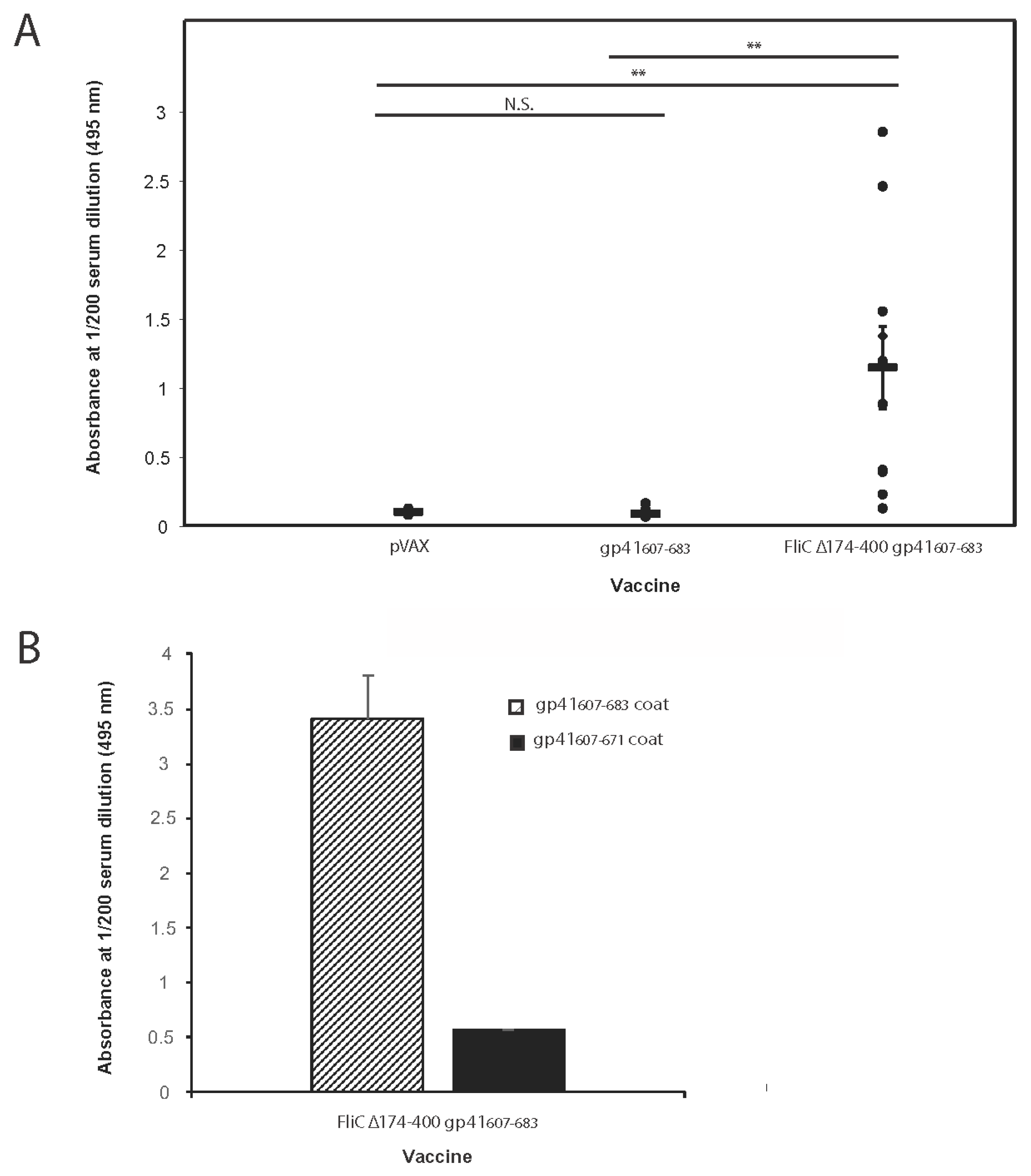
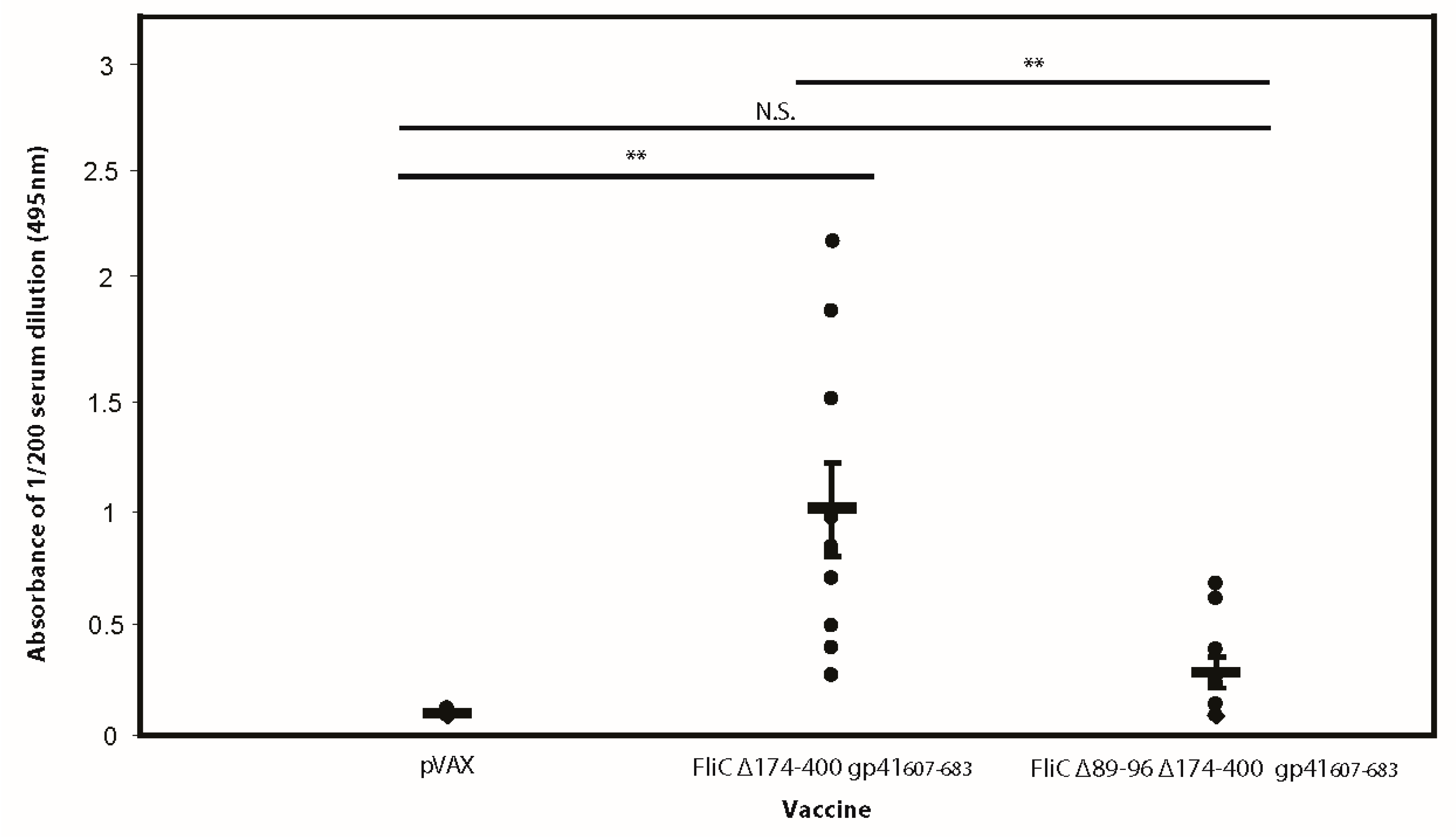
3.5. FliC Augments gp41607–683 Immunogenicity and Elicits MPER-Specific Humoral Responses
3.6. The Adjuvant Effect of FliC is TLR5 Dependent
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Plotkin, S.A. Vaccines: The fourth century. Clin. Vaccine Immunol. 2009, 16, 1709–1719. [Google Scholar] [CrossRef] [PubMed]
- Koff, W.C.; Burton, D.R.; Johnson, P.R.; Walker, B.D.; King, C.R.; Nabel, G.J.; Ahmed, R.; Bhan, M.K.; Plotkin, S.A. Accelerating Next-Generation Vaccine Development for Global Disease Prevention. Science 2013, 340, 1232910. [Google Scholar] [CrossRef] [PubMed]
- Koff, W.C. A shot at AIDS. Curr. Opin. Biotechnol. 2016, 42, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Amed Ouattara, M.B.L. Vaccines Against Malaria. Clin. Infect. Dis. 2015, 60, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Orme, I.M.; Robinson, R.T.; Cooper, A.M. The balance between protective and pathogenic immune responses in the TB-infected lung. Nat. Immunol. 2014, 16, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.C.; Orme, I.M.; Starke, J.R. 35—Tuberculosis vaccines. In Vaccines, 6th ed.; Plotkin, S.A., Orenstein, W.A., Offit, P.A., Eds.; W.B. Saunders: London, UK, 2013; pp. 789–811. [Google Scholar]
- Liang, T.J. Current progress in development of hepatitis C virus vaccines. Nat. Med. 2013, 19, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Andersen, P.; Kaufmann, S.H.E. Novel Vaccination Strategies against Tuberculosis. Cold Spring Harb. Perspect. Med. 2014, 4, a018523. [Google Scholar] [CrossRef] [PubMed]
- Parks, C.L. Replication-Competent Viral Vectors for Vaccine Delivery. In Human Vaccines; Modjarrad, K., Koff, W.C., Eds.; Academic Press: Cambridge, MA, USA, 2017; Chapter 2; pp. 25–63. [Google Scholar]
- Coffman, R.L.; Sher, A.; Seder, R.A. Vaccine Adjuvants: Putting Innate Immunity to Work. Immunity 2010, 33, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Garçon, N.; Hem, S.; Friede, M. Evolution of adjuvants across the centuries. In Vaccines, 6th ed.; Elsevier: London, UK, 2013; pp. 58–70. [Google Scholar]
- Levitz, S.M.; Golenbock, D.T. Beyond Empiricism: Informing Vaccine Development through Innate Immunity Research. Cell 2012, 148, 1284–1292. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, C.; Bertholet, S.; Philpott, D.J.; De Gregorio, E. Unleashing the potential of NOD- and Toll-like agonists as vaccine adjuvants. Proc. Natl. Acad. Sci. USA 2014, 111, 12294–12299. [Google Scholar] [CrossRef] [PubMed]
- Rossez, Y.; Wolfson, E.B.; Holmes, A.; Gally, D.L.; Holden, N.J. Bacterial Flagella: Twist and Stick, or Dodge across the Kingdoms. PLoS Pathog. 2015, 11, e1004483. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, F.; Smith, K.D.; Ozinsky, A.; Hawn, T.R.; Yi, E.C.; Goodlett, D.R.; Eng, J.K.; Akira, S.; Underhill, D.M.; Aderem, A. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5: Article: Nature. Nature 2001, 410, 1099–1103. [Google Scholar] [CrossRef] [PubMed]
- Gewirtz, A.T.; Navas, T.A.; Lyons, S.; Godowski, P.J.; Madara, J.L. Cutting edge: Bacterial flagellin activates basolaterally expressed TLR5 to induce epithelial proinflammatory gene expression. J. Immunol. 2001, 167, 1882–1885. [Google Scholar] [CrossRef] [PubMed]
- Means, T.K.; Hayashi, F.; Smith, K.D.; Aderem, A.; Luster, A.D. The Toll-like receptor 5 stimulus bacterial flagellin induces maturation and chemokine production in human dendritic cells. J. Immunol. 2003, 170, 5165–5175. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.D.; Andersen-Nissen, E.; Hayashi, F.; Strobe, K.; Bergman, M.A.; Barrett, S.L.R.; Cookson, B.T.; Aderem, A. Toll-like receptor 5 recognizes a conserved site on flagellin required for protofilament formation and bacterial motility. Nat. Immunol. 2003, 4, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Murthy, K.G.K.; Deb, A.; Goonesekera, S.; Szabó, C.; Salzman, A.L. Identification of conserved domains in Salmonella muenchen flagellin that are essential for its ability to activate TLR5 and to induce an inflammatory response in vitro. J. Biol. Chem. 2004, 279, 5667–5675. [Google Scholar] [CrossRef] [PubMed]
- Feuillet, V.; Medjane, S.; Mondor, I.; Demaria, O.; Pagni, P.P.; Galán, J.E.; Flavell, R.A.; Alexopoulou, L. Involvement of Toll-like receptor 5 in the recognition of flagellated bacteria. PNAS 2006, 103, 12487–12492. [Google Scholar] [CrossRef] [PubMed]
- Honko, A.N.; Mizel, S.B. Effects of flagellin on innate and adaptive immunity. Immunol. Res. 2005, 33, 83–101. [Google Scholar] [CrossRef]
- Vijay-Kumar, M.; Gewirtz, A.T. Flagellin: Key target of mucosal innate immunity. Mucosal Immunol. 2009, 2, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Mizel, S.B.; Bates, J.T. Flagellin as an adjuvant: Cellular mechanisms and potential. J. Immunol. 2010, 185, 5677–5682. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-I.; Kurnasov, O.; Natarajan, V.; Hong, M.; Gudkov, A.V.; Osterman, A.L.; Wilson, I.A. Structural Basis of TLR5-Flagellin Recognition and Signaling. Science 2012, 335, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Sun, P.D. The Structure of the TLR5-Flagellin Complex: A New Mode of Pathogen Detection, Conserved Receptor Dimerization for Signaling. Sci. Signal. 2012, 5, pe11. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Didierlaurent, A.; Ferrero, I.; Otten, L.A.; Dubois, B.; Reinhardt, M.; Carlsen, H.; Blomhoff, R.; Akira, S.; Kraehenbuhl, J.-P.; Sirard, J.-C. Flagellin promotes myeloid differentiation factor 88-dependent development of Th2-type response. J. Immunol. 2004, 172, 6922–6930. [Google Scholar] [CrossRef] [PubMed]
- Lightfield, K.L.; Persson, J.; Brubaker, S.W.; Witte, C.E.; von Moltke, J.; Dunipace, E.A.; Henry, T.; Sun, Y.-H.; Cado, D.; Dietrich, W.F.; et al. Critical function for Naip5 in inflammasome activation by a conserved carboxy-terminal domain of flagellin. Nat. Immunol. 2008, 9, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Kofoed, E.M.; Vance, R.E. Innate immune recognition of bacterial ligands by NAIPs determines inflammasome specificity. Nature 2011, 477, 592–595. [Google Scholar] [CrossRef] [PubMed]
- Kortmann, J.; Brubaker, S.W.; Monack, D.M. Cutting Edge: Inflammasome Activation in Primary Human Macrophages Is Dependent on Flagellin. J. Immunol. 2015, 195, 815–819. [Google Scholar] [CrossRef] [PubMed]
- Rauch, I.; Tenthorey, J.L.; Nichols, R.D.; Moussawi Al, K.; Kang, J.J.; Kang, C.; Kazmierczak, B.I.; Vance, R.E. NAIP proteins are required for cytosolic detection of specific bacterial ligands in vivo. J. Exp. Med. 2016, 213, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Shi, J.; Shi, X.; Wang, Y.; Wang, F.; Shao, F. Genetic functions of the NAIP family of inflammasome receptors for bacterial ligands in mice. J. Exp. Med. 2016, 213, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Shao, F. The NAIP–NLRC4 inflammasome in innate immune detection of bacterial flagellin and type III secretion apparatus. Immunol. Rev. 2015, 265, 85–102. [Google Scholar] [CrossRef] [PubMed]
- Vijay-Kumar, M.; Carvalho, F.A.; Aitken, J.D.; Fifadara, N.H.; Gewirtz, A.T. TLR5 or NLRC4 is necessary and sufficient for promotion of humoral immunity by flagellin. Eur. J. Immunol. 2010, 40, 3528–3534. [Google Scholar] [CrossRef] [PubMed]
- Miao, E.A.; Alpuche-Aranda, C.M.; Dors, M.; Clark, A.E.; Bader, M.W.; Miller, S.I.; Aderem, A. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1β via Ipaf. Nat. Immunol. 2006, 7, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Franchi, L.; Amer, A.; Body-Malapel, M.; Kanneganti, T.-D.; Özören, N.; Jagirdar, R.; Inohara, N.; Vandenabeele, P.; Bertin, J.; Coyle, A.; et al. Cytosolic flagellin requires Ipaf for activation of caspase-1 and interleukin 1β in salmonella-infected macrophages. Nat. Immunol. 2006, 7, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Franchi, L.; Eigenbrod, T.; Muñoz-Planillo, R.; Núñez, G. The inflammasome: A caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat. Immunol. 2009, 10, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Aachoui, Y.; Miao, E.A. Down with doublespeak: NAIP/NLRC4 inflammasomes get specific. J. Exp. Med. 2016, 213, 646. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yonekura, K.; Maki-Yonekura, S.; Namba, K. Complete atomic model of the bacterial flagellar filament by electron cryomicroscopy. Nature 2003, 424, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Beatson, S.A.; Minamino, T.; Pallen, M.J. Variation in bacterial flagellins: From sequence to structure. Trends Microbiol. 2006, 14, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Eaves-Pyles, T.D.; Wong, H.R.; Odoms, K.; Pyles, R.B. Salmonella flagellin-dependent proinflammatory responses are localized to the conserved amino and carboxyl regions of the protein. J. Immunol. 2001, 167, 7009–7016. [Google Scholar] [CrossRef] [PubMed]
- Andersen-Nissen, E.; Smith, K.D.; Strobe, K.L.; Barrett, S.L.R.; Cookson, B.T.; Logan, S.M.; Aderem, A. Evasion of Toll-like receptor 5 by flagellated bacteria. Proc. Natl. Acad. Sci. USA 2005, 102, 9247–9252. [Google Scholar] [CrossRef] [PubMed]
- Nempont, C.; Cayet, D.; Rumbo, M.; Bompard, C.; Villeret, V.; Sirard, J.-C. Deletion of flagellin’s hypervariable region abrogates antibody-mediated neutralization and systemic activation of TLR5-dependent immunity. J. Immunol. 2008, 181, 2036–2043. [Google Scholar] [CrossRef] [PubMed]
- Song, W.S.; Jeon, Y.J.; Namgung, B.; Hong, M.; Yoon, S.-I. A conserved TLR5 binding and activation hot spot on flagellin. Sci. Rep. 2017, 7, 40878. [Google Scholar] [CrossRef] [PubMed]
- Halff, E.F.; Diebolder, C.A.; Versteeg, M.; Schouten, A.; Brondijk, T.H.C.; Huizinga, E.G. Formation and structure of a NAIP5-NLRC4 inflammasome induced by direct interactions with conserved N- and C-terminal regions of flagellin. J. Biol. Chem. 2012, 287, 38460–38472. [Google Scholar] [CrossRef] [PubMed]
- Kuwajima, G. Flagellin domain that affects H antigenicity of Escherichia coli K-12. J. Bacteriol. 1988, 170, 485–488. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, K.; Aizawa, S.; Yamaguchi, S. Flagellar filament structure and cell motility of Salmonella typhimurium mutants lacking part of the outer domain of flagellin. J. Bacteriol. 1995, 177, 1090–1093. [Google Scholar] [CrossRef] [PubMed]
- He, X.S.; Rivkina, M.; Stocker, B.A.; Robinson, W.S. Hypervariable region IV of Salmonella gene fliCd encodes a dominant surface epitope and a stabilizing factor for functional flagella. J. Bacteriol. 1994, 176, 2406–2414. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hoiseth, S.K.; Stocker, B.A.D. Aromatic-dependent Salmonella typhimurium are non-virulent and effective as live vaccines. Nature 1981, 291, 238–239. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Hormaeche, C.E.; de Hormaeche, R.D.; Winther, M.; Dougan, G.; Maskell, D.J.; Stocker, B.A.D. An Attenuated aroA Salmonella typhimurium Vaccine Elicits Humoral and Cellular Immunity to Cloned β-Galactosidase in Mice. J. Infect. Dis. 1987, 155, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Sadoff, J.C.; Ballou, W.R.; Baron, L.S.; Majarian, W.R.; Brey, R.N.; Hockmeyer, W.T.; Young, J.F.; Cryz, S.J.; Ou, J.; Lowell, G.H.; et al. Oral Salmonella typhimurium vaccine expressing circumsporozoite protein protects against malaria. Science 1988, 240, 336–338. [Google Scholar] [CrossRef] [PubMed]
- Chatfield, S.; Strugnell, R.; Dougan, G. Live Salmonella as vaccines and carriers of foreign antigenic determinants. Vaccine 1989, 7, 495–498. [Google Scholar] [CrossRef]
- Newton, S.M.; Jacob, C.O.; Stocker, B.A. Immune response to cholera toxin epitope inserted in Salmonella flagellin. Science 1989, 244, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.Y.; Newton, S.; Judd, A.; Stocker, B.; Robinson, W.S. Expression of immunogenic epitopes of hepatitis B surface antigen with hybrid flagellin proteins by a vaccine strain of Salmonella. Proc. Natl. Acad. Sci. USA 1989, 86, 4726–4730. [Google Scholar] [CrossRef] [PubMed]
- Newton, S.M.; Kotb, M.; Poirier, T.P.; Stocker, B.A.; Beachey, E.H. Expression and immunogenicity of a streptococcal M protein epitope inserted in Salmonella flagellin. Infect. Immun. 1991, 59, 2158–2165. [Google Scholar] [PubMed]
- Cárdenas, L.; Clements, J.D. Oral immunization using live attenuated Salmonella spp. as carriers of foreign antigens. Clin. Microbiol. Rev. 1992, 5, 328–342. [Google Scholar] [CrossRef] [PubMed]
- McEwen, J.; Levi, R.; Horwitz, R.J.; Arnon, R. Synthetic recombinant vaccine expressing influenza haemagglutinin epitope in Salmonella flagellin leads to partial protection in mice. Vaccine 1992, 10, 405–411. [Google Scholar] [CrossRef]
- Verma, N. Induction of a cellular immune response to a defined T-cell epitope as an insert in the flagellin of a live vaccine strain of Salmonella. Vaccine 1995, 13, 235–244. [Google Scholar] [CrossRef]
- Levi, R. Synthetic recombinant influenza vaccine induces efficient long-term immunity and cross-strain protection. Vaccine 1996, 14, 85–92. [Google Scholar] [CrossRef]
- Ben-Yedidia, T.; Arnon, R. Effect of pre-existing carrier immunity on the efficacy of synthetic influenza vaccine. Immunol. Lett. 1998, 64, 9–15. [Google Scholar] [CrossRef]
- Ben-Yedidia, T.; Tarrab-Hazdai, R.; Schechtman, D.; Arnon, R. Intranasal administration of synthetic recombinant peptide-based vaccine protects mice from infection by Schistosoma mansoni. Infect. Immun. 1999, 67, 4360–4366. [Google Scholar] [PubMed]
- Stocker, B.A.D.; Newton, S.M.C. Immune Responses to Epitopes Inserted in Salmonella Flagellin. Int. Rev. Immunol. 2009, 11, 167–178. [Google Scholar] [CrossRef]
- Liu, G.; Tarbet, B.; Song, L.; Reiserova, L.; Weaver, B.; Chen, Y.; Li, H.; Hou, F.; Liu, X.; Parent, J.; et al. Immunogenicity and Efficacy of Flagellin-Fused Vaccine Candidates Targeting 2009 Pandemic H1N1 Influenza in Mice. PLoS ONE 2011, 6, e20928. [Google Scholar] [CrossRef] [PubMed]
- Ghose, C.; Verhagen, J.M.; Chen, X.; Yu, J.; Huang, Y.; Chenesseau, O.; Kelly, C.P.; Ho, D.D. Toll-like receptor 5-dependent immunogenicity and protective efficacy of a recombinant fusion protein vaccine containing the nontoxic domains of Clostridium difficile toxins A and B and Salmonella enterica serovar typhimurium flagellin in a mouse model of Clostridium difficile disease. Infect. Immun. 2013, 81, 2190–2196. [Google Scholar] [PubMed]
- Carapau, D.; Mitchell, R.; Nacer, A.; Shaw, A.; Othoro, C.; Frevert, U.; Nardin, E. Protective Humoral Immunity Elicited by a Needle-Free Malaria Vaccine Comprised of a Chimeric Plasmodium falciparum Circumsporozoite Protein and a Toll-Like Receptor 5 Agonist, Flagellin. Infect. Immun. 2013, 81, 4350–4362. [Google Scholar] [CrossRef] [PubMed]
- Leal, M.T.A.; Camacho, A.G.A.; Teixeira, L.H.; Bargieri, D.Y.; Soares, I.S.; Tararam, C.A.; Rodrigues, M.M. Immunogenicity of recombinant proteins consisting of Plasmodium vivax circumsporozoite protein allelic variant-derived epitopes fused with Salmonella enterica Serovar Typhimurium flagellin. Clin. Vaccine Immunol. 2013, 20, 1418–1425. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Song, L.; Reiserova, L.; Trivedi, U.; Li, H.; Liu, X.; Noah, D.; Hou, F.; Weaver, B.; Tussey, L. Flagellin-HA vaccines protect ferrets and mice against H5N1 highly pathogenic avian influenza virus (HPAIV) infections. Vaccine 2012, 30, 6833–6838. [Google Scholar] [CrossRef] [PubMed]
- Braga, C.J.M.; Massis, L.M.; Sbrogio-Almeida, M.E.; Alencar, B.C.G.; Bargieri, D.Y.; Boscardin, S.B.; Rodrigues, M.M.; Ferreira, L.C.S. CD8+ T cell adjuvant effects of Salmonella FliCd flagellin in live vaccine vectors or as purified protein. Vaccine 2010, 28, 1373–1382. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Zhang, Y.; Yun, N.E.; Poussard, A.L.; Smith, J.N.; Smith, J.K.; Borisevich, V.; Linde, J.J.; Zacks, M.A.; Li, H.; et al. Superior efficacy of a recombinant flagellin:H5N1 HA globular head vaccine is determined by the placement of the globular head within flagellin. Vaccine 2009, 27, 5875–5884. [Google Scholar] [CrossRef] [PubMed]
- Mizel, S.B.; Graff, A.H.; Sriranganathan, N.; Ervin, S.; Lees, C.J.; Lively, M.O.; Hantgan, R.R.; Thomas, M.J.; Wood, J.; Bell, B. Flagellin-F1-V fusion protein is an effective plague vaccine in mice and two species of nonhuman primates. Clin. Vaccine Immunol. 2009, 16, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Delaney, K.N.; Phipps, J.P.; Johnson, J.B.; Mizel, S.B. A Recombinant Flagellin-Poxvirus Fusion Protein Vaccine Elicits Complement-Dependent Protection Against Respiratory Challenge with Vaccinia Virus in Mice. Viral Immunol. 2010, 23, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Huleatt, J.W.; Nakaar, V.; Desai, P.; Huang, Y.; Hewitt, D.; Jacobs, A.; Tang, J.; McDonald, W.; Song, L.; Evans, R.K.; et al. Potent immunogenicity and efficacy of a universal influenza vaccine candidate comprising a recombinant fusion protein linking influenza M2e to the TLR5 ligand flagellin. Vaccine 2008, 26, 201–214. [Google Scholar] [CrossRef] [PubMed]
- McDonald, W.F.; Huleatt, J.W.; Foellmer, H.G.; Hewitt, D.; Tang, J.; Desai, P.; Price, A.; Jacobs, A.; Takahashi, V.N.; Huang, Y.; et al. A West Nile virus recombinant protein vaccine that coactivates innate and adaptive immunity. J. Infect. Dis. 2007, 195, 1607–1617. [Google Scholar] [CrossRef] [PubMed]
- Huleatt, J.W.; Jacobs, A.R.; Tang, J.; Desai, P.; Kopp, E.B.; Huang, Y.; Song, L.; Nakaar, V.; Powell, T.J. Vaccination with recombinant fusion proteins incorporating Toll-like receptor ligands induces rapid cellular and humoral immunity. Vaccine 2007, 25, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Xiong, D.; Hu, M.; Kang, X.; Pan, Z.; Jiao, X. Immunopotentiation of Different Adjuvants on Humoral and Cellular Immune Responses Induced by HA1-2 Subunit Vaccines of H7N9 Influenza in Mice. PLoS ONE 2016, 11, e0150678. [Google Scholar] [CrossRef] [PubMed]
- Stepanova, L.A.; Kotlyarov, R.Y.; Kovaleva, A.A.; Potapchuk, M.V.; Korotkov, A.V.; Sergeeva, M.V.; Kasianenko, M.A.; Kuprianov, V.V.; Ravin, N.V.; Tsybalova, L.M.; et al. Protection against Multiple Influenza A Virus Strains Induced by Candidate Recombinant Vaccine Based on Heterologous M2e Peptides Linked to Flagellin. PLoS ONE 2015, 10, e0119520. [Google Scholar] [CrossRef] [PubMed]
- Kalnin, K.; Tibbitts, T.; Yan, Y.; Stegalkina, S.; Shen, L.; Costa, V.; Sabharwal, R.; Anderson, S.F.; Day, P.M.; Christensen, N.; et al. Low doses of flagellin-L2 multimer vaccines protect against challenge with diverse papillomavirus genotypes. Vaccine 2014, 32, 3540–3547. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.-Z.; Gill, H.S.; Kang, S.-M.; Wang, L.; Wang, Y.-C.; Vassilieva, E.V.; Compans, R.W. Enhanced influenza virus-like particle vaccines containing the extracellular domain of matrix protein 2 and a Toll-like receptor ligand. Clin. Vaccine Immunol. 2012, 19, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.-Z.; Quan, F.-S.; Kang, S.-M.; Bozja, J.; Skountzou, I.; Compans, R.W. Incorporation of membrane-anchored flagellin into influenza virus-like particles enhances the breadth of immune responses. J. Virol. 2008, 82, 11813–11823. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Song, L.; Beasley, D.W.C.; Putnak, R.; Parent, J.; Misczak, J.; Li, H.; Reiserova, L.; Liu, X.; Tian, H.; et al. Immunogenicity and Efficacy of Flagellin-Envelope Fusion Dengue Vaccines in Mice and Monkeys. Clin. Vaccine Immunol. 2015, 22, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.-H.; Chang, L.-S.; Tian, C.-Y.; Yeh, Y.-C.; Chen, Y.-J.; Chuang, T.-H.; Liu, S.-J.; Leng, C.-H. Carboxyl-terminal fusion of E7 into Flagellin shifts TLR5 activation to NLRC4/NAIP5 activation and induces TLR5-independent anti-tumor immunity. Sci. Rep. 2016, 6, 24199. [Google Scholar] [CrossRef] [PubMed]
- Talbot, H.K.; Rock, M.T.; Johnson, C.; Tussey, L.; Kavita, U.; Shanker, A.; Shaw, A.R.; Taylor, D.N. Immunopotentiation of Trivalent Influenza Vaccine When Given with VAX102, a Recombinant Influenza M2e Vaccine Fused to the TLR5 Ligand Flagellin. PLoS ONE 2010, 5, e14442. [Google Scholar] [CrossRef] [PubMed]
- Treanor, J.J.; Taylor, D.N.; Tussey, L.; Hay, C.; Nolan, C.; Fitzgerald, T.; Liu, G.; Kavita, U.; Song, L.; Dark, I.; et al. Safety and immunogenicity of a recombinant hemagglutinin influenza–flagellin fusion vaccine (VAX125) in healthy young adults. Vaccine 2010, 28, 8268–8274. [Google Scholar] [CrossRef] [PubMed]
- Turley, C.B.; Rupp, R.E.; Johnson, C.; Taylor, D.N.; Wolfson, J.; Tussey, L.; Kavita, U.; Stanberry, L.; Shaw, A. Safety and immunogenicity of a recombinant M2e–flagellin influenza vaccine (STF2.4xM2e) in healthy adults. Vaccine 2011, 29, 5145–5152. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.N.; Treanor, J.J.; Sheldon, E.A.; Johnson, C.; Umlauf, S.; Song, L.; Kavita, U.; Liu, G.; Tussey, L.; Ozer, K.; et al. Development of VAX128, a recombinant hemagglutinin (HA) influenza-flagellin fusion vaccine with improved safety and immune response. Vaccine 2012, 30, 5761–5769. [Google Scholar] [CrossRef] [PubMed]
- Tussey, L.; Strout, C.; Davis, M.; Johnson, C.; Lucksinger, G.; Umlauf, S.; Song, L.; Liu, G.; Abraham, K.; White, C.J. Phase 1 Safety and Immunogenicity Study of a Quadrivalent Seasonal Flu Vaccine Comprising Recombinant Hemagglutinin-Flagellin Fusion Proteins. Open Forum Infect. Dis. 2016, 3, ofw015. [Google Scholar] [CrossRef] [PubMed]
- McSorley, S.J.; Ehst, B.D.; Yu, Y.; Gewirtz, A.T. Bacterial Flagellin Is an Effective Adjuvant for CD4+ T Cells In Vivo. J. Immunol. 2002, 169, 3914–3919. [Google Scholar] [CrossRef] [PubMed]
- Honko, A.N.; Sriranganathan, N.; Lees, C.J.; Mizel, S.B. Flagellin is an effective adjuvant for immunization against lethal respiratory challenge with Yersinia pestis. Infect. Immun. 2006, 74, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Skountzou, I.; Martin, M.D.P.; Wang, B.; Ye, L.; Koutsonanos, D.; Weldon, W.; Jacob, J.; Compans, R.W. Salmonella flagellins are potent adjuvants for intranasally administered whole inactivated influenza vaccine. Vaccine 2010, 28, 4103–4112. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.R.; Holbrook, B.C.; Hayward, S.L.; Blevins, L.K.; Jorgensen, M.J.; Kock, N.D.; De Paris, K.; D’Agostino, R.B., Jr.; Aycock, S.T.; Mizel, S.B.; Parks, G.D.; Alexander-Miller, M.A. Inclusion of Flagellin during Vaccination against Influenza Enhances Recall Responses in Nonhuman Primate Neonates. J. Virol. 2015, 89, 7291–7303. [Google Scholar] [CrossRef] [PubMed]
- Cuadros, C.; Lopez-Hernandez, F.J.; Dominguez, A.L.; McClelland, M.; Lustgarten, J. Flagellin fusion proteins as adjuvants or vaccines induce specific immune responses. Infect. Immun. 2004, 72, 2810–2816. [Google Scholar] [CrossRef] [PubMed]
- Bates, J.T.; Uematsu, S.; Akira, S.; Mizel, S.B. Direct stimulation of tlr5+/+ CD11c+ cells is necessary for the adjuvant activity of flagellin. J. Immunol. 2009, 182, 7539–7547. [Google Scholar] [CrossRef] [PubMed]
- Sanders, C.J.; Franchi, L.; Yarovinsky, F.; Uematsu, S.; Akira, S.; Núñez, G.; Gewirtz, A.T. Induction of adaptive immunity by flagellin does not require robust activation of innate immunity. Eur. J. Immunol. 2009, 39, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Letran, S.E.; Lee, S.J.; Atif, S.M.; Uematsu, S.; Akira, S.; McSorley, S.J. TLR5 functions as an endocytic receptor to enhance flagellin-specific adaptive immunity. Eur. J. Immunol. 2011, 41, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Bates, J.T.; Graff, A.H.; Phipps, J.P.; Grayson, J.M.; Mizel, S.B. Enhanced antigen processing of flagellin fusion proteins promotes the antigen-specific CD8+ T cell response independently of TLR5 and MyD. J. Immunol. 2011, 186, 6255–6262. [Google Scholar] [CrossRef] [PubMed]
- López-Yglesias, A.H.; Zhao, X.; Quarles, E.K.; Lai, M.A.; VandenBos, T.; Strong, R.K.; Smith, K.D. Flagellin induces antibody responses through a TLR5- and inflammasome-independent pathway. J. Immunol. 2014, 192, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, S.A. Vaccines: Correlates of vaccine-induced immunity. Clin. Infect. Dis. 2008, 47, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, S.A. Correlates of protection induced by vaccination. Clin. Vaccine Immunol. 2010, 17, 1055–1065. [Google Scholar] [CrossRef] [PubMed]
- Burton, D.R.; Hangartner, L. Broadly Neutralizing Antibodies to HIV and Their Role in Vaccine Design. Annu. Rev. Immunol. 2016, 34, 635–659. [Google Scholar] [CrossRef] [PubMed]
- Sadanand, S.; Suscovich, T.J.; Alter, G. Broadly Neutralizing Antibodies Against HIV: New Insights to Inform Vaccine Design. Annu. Rev. Med. 2016, 67, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Montero, M.; van Houten, N.E.; Wang, X.; Scott, J.K. The membrane-proximal external region of the human immunodeficiency virus type 1 envelope: Dominant site of antibody neutralization and target for vaccine design. Microbiol. Mol. Biol. Rev. 2008, 72, 54–84, table of contents. [Google Scholar] [CrossRef] [PubMed]
- Hessell, A.J.; Rakasz, E.G.; Tehrani, D.M.; Huber, M.; Weisgrau, K.L.; Landucci, G.; Forthal, D.N.; Koff, W.C.; Poignard, P.; Watkins, D.I.; et al. Broadly neutralizing monoclonal antibodies 2F5 and 4E10 directed against the human immunodeficiency virus type 1 gp41 membrane-proximal external region protect against mucosal challenge by simian-human immunodeficiency virus SHIVBa-L. J. Virol. 2010, 84, 1302–1313. [Google Scholar] [CrossRef] [PubMed]
- Pegu, A.; Yang, Z.-Y.; Boyington, J.C.; Wu, L.; Ko, S.-Y.; Schmidt, S.D.; McKee, K.; Kong, W.-P.; Shi, W.; Chen, X.; et al. Neutralizing antibodies to HIV-1 envelope protect more effectively in vivo than those to the CD4 receptor. Sci. Transl. Med. 2014, 6, 243ra88. [Google Scholar] [CrossRef] [PubMed]
- Pegu, A.; Hessell, A.J.; Mascola, J.R.; Haigwood, N.L. Use of broadly neutralizing antibodies for HIV-1 prevention. Immunol. Rev. 2017, 275, 296–312. [Google Scholar] [CrossRef] [PubMed]
- Phogat, S.; Svehla, K.; Tang, M.; Spadaccini, A.; Muller, J.; Mascola, J.; Berkower, I.; Wyatt, R. Analysis of the human immunodeficiency virus type 1 gp41 membrane proximal external region arrayed on hepatitis B surface antigen particles. Virology 2008, 373, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Venditto, V.J.; Wieczorek, L.; Molnar, S.; Teque, F.; Landucci, G.; Watson, D.S.; Forthal, D.; Polonis, V.R.; Levy, J.A.; Szoka, F.C. Chemically modified peptides based on the membrane-proximal external region of the HIV-1 envelope induce high-titer, epitope-specific nonneutralizing antibodies in rabbits. Clin. Vaccine Immunol. 2014, 21, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Dennison, S.M.; Sutherland, L.L.; Jaeger, F.H.; Anasti, K.M.; Parks, R.; Stewart, S.; Bowman, C.; Xia, S.-M.; Zhang, R.; Shen, X.; et al. Induction of Antibodies in Rhesus Macaques That Recognize a Fusion-Intermediate Conformation of HIV-1 gp. PLoS ONE 2011, 6, e27824. [Google Scholar] [CrossRef] [PubMed]
- Strasz, N.; Morozov, V.A.; Kreutzberger, J.; Keller, M.; Eschricht, M.; Denner, J. Immunization with Hybrid Proteins Containing the Membrane Proximal External Region of HIV-1. AIDS Res. Hum. Retrovir. 2014, 30, 498–508. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Wen, Z.; Dong, K.; Wang, X.; Bu, Z.; Zhang, H.; Compans, R.W.; Yang, C. Induction of HIV Neutralizing Antibodies against the MPER of the HIV Envelope Protein by HA/gp41 Chimeric Protein-Based DNA and VLP Vaccines. PLoS ONE 2011, 6, e14813. [Google Scholar] [CrossRef] [PubMed]
- Venditto, V.J.; Watson, D.S.; Motion, M.; Montefiori, D.; Szoka, F.C., Jr. Rational Design of Membrane Proximal External Region Lipopeptides Containing Chemical Modifications for HIV-1 Vaccination. Clin. Vaccine Immunol. 2013, 20, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Patrick, A.J.; Rosenthal, K.L. Multiple tandem copies of conserved gp41 epitopes incorporated in gag virus-like particles elicit systemic and mucosal antibodies in an optimized heterologous vector delivery regimen. Vaccine 2010, 28, 7070–7080. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Zhong, Z.; Zariffard, M.; Spear, G.T.; Qiao, L. Bovine papillomavirus-like particles presenting conserved epitopes from membrane-proximal external region of HIV-1 gp41 induced mucosal and systemic antibodies. Vaccine 2013, 31, 5422–5429. [Google Scholar] [CrossRef] [PubMed]
- Yi, G.; Lapelosa, M.; Bradley, R.; Mariano, T.M.; Dietz, D.E.; Hughes, S.; Wrin, T.; Petropoulos, C.; Gallicchio, E.; Levy, R.M.; et al. Chimeric Rhinoviruses Displaying MPER Epitopes Elicit Anti-HIV Neutralizing Responses. PLoS ONE 2013, 8, e72205. [Google Scholar] [CrossRef] [PubMed]
- Boehme, K.W.; Ikizler, M.; Iskarpatyoti, J.A.; Wetzel, J.D.; Willis, J.; Crowe, J.E.; LaBranche, C.C.; Montefiori, D.C.; Wilson, G.J.; Dermody, T.S.; et al. Engineering Recombinant Reoviruses To Display gp41 Membrane-Proximal External-Region Epitopes from HIV-1. mSphere 2016, 1, e00086-16. [Google Scholar] [CrossRef] [PubMed]
- Molinos-Albert, L.M.; Bilbao, E.; Agulló, L.; Marfil, S.; García, E.; de Concepción, M.L.R.L.; Izquierdo-Useros, N.; Vilaplana, C.; Nieto-Garai, J.A.; Contreras, F.X.; et al. Proteoliposomal formulations of an HIV-1 gp41-based miniprotein elicit a lipid-dependent immunodominant response overlapping the 2F5 binding motif. Sci. Rep. 2017, 7, 40800. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Song, L.; Moon, J.; Sun, Z.-Y.J.; Bershteyn, A.; Hanson, M.; Cain, D.; Goka, S.; Kelsoe, G.; Wagner, G.; et al. Immunogenicity of membrane-bound HIV-1 gp41 membrane-proximal external region (MPER) segments is dominated by residue accessibility and modulated by stereochemistry. J. Biol. Chem. 2013, 288, 31888–31901. [Google Scholar] [CrossRef] [PubMed]
- Scholz, C.; Schaarschmidt, P.; Engel, A.M.; Andres, H.; Schmitt, U.; Faatz, E.; Balbach, J.; Schmid, F.X. Functional Solubilization of Aggregation-prone HIV Envelope Proteins by Covalent Fusion with Chaperone Modules. J. Mol. Biol. 2005, 345, 1229–1241. [Google Scholar] [CrossRef] [PubMed]
- Enshell-Seijffers, D.; Smelyanski, L.; Vardinon, N.; Yust, I.; Gershoni, J.M. Dissection of the humoral immune response toward an immunodominant epitope of HIV: A model for the analysis of antibody diversity in HIV+ individuals. FASEB J. 2001, 15, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Molinos-Albert, L.M.; Carrillo, J.; Curriu, M.; de la Concepción, M.L.R.; Marfil, S.; García, E.; Clotet, B.; Blanco, J. Anti-MPER antibodies with heterogeneous neutralization capacity are detectable in most untreated HIV-1 infected individuals. Retrovirology 2014, 11, 44. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Haynes, B.F.; Fleming, J.; Clair, E.W.S.; Katinger, H.; Stiegler, G.; Kunert, R.; Robinson, J.; Scearce, R.M.; Plonk, K.; Staats, H.F.; et al. Cardiolipin Polyspecific Autoreactivity in Two Broadly Neutralizing HIV-1 Antibodies. Science 2005, 308, 1906–1908. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Frey, G.; Peng, H.; Rits-Volloch, S.; Garrity, J.; Seaman, M.S.; Chen, B. Mechanism of HIV-1 neutralization by antibodies targeting a membrane-proximal region of gp41. J. Virol. 2014, 88, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Melnychuk, L.; Ajamian, L.; Jean-Pierre, P.; Liang, J.; Gheorghe, R.; Wainberg, M.A.; Zaharatos, G.J. Development of a DNA vaccine expressing a secreted HIV-1 gp41 ectodomain that includes the membrane-proximal external region. Vaccine 2017, 35, 2736–2744. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Howard, A.; Kelley, C.; Delogu, G.; Collins, F.; Morris, S. Immunogenicity of DNA vaccines expressing tuberculosis proteins fused to tissue plasminogen activator signal sequences. Infect. Immun. 1999, 67, 4780–4786. [Google Scholar] [PubMed]
- Logan, S.M. Flagellar glycosylation—A new component of the motility repertoire? Microbiology 2006, 152, 1249–1262. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. Toll-like Receptors and Their Crosstalk with Other Innate Receptors in Infection and Immunity. Immunity 2011, 34, 637–650. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Kawai, T.; Akira, S. Microbial sensing by Toll-like receptors and intracellular nucleic acid sensors. Cold Spring Harb. Perspect. Biol. 2015, 7, a016246. [Google Scholar] [CrossRef] [PubMed]
- Andersen-Nissen, E.; Smith, K.D.; Bonneau, R.; Strong, R.K.; Aderem, A. A conserved surface on Toll-like receptor 5 recognizes bacterial flagellin. J. Exp. Med. 2007, 204, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Takeshita, F.; Matsuda, T.; Jounai, N.; Kobiyama, K.; Matsumoto, T.; Sasaki, S.; Yoshida, A.; Xin, K.-Q.; Klinman, D.M.; et al. Blocking of the TLR5 activation domain hampers protective potential of flagellin DNA vaccine. J. Immunol. 2007, 179, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Forstnerič, V.; Ivičak-Kocjan, K.; Ljubetič, A.; Jerala, R.; Benčina, M. Distinctive Recognition of Flagellin by Human and Mouse Toll-Like Receptor. PLoS ONE 2016, 11, e0158894. [Google Scholar]
- Ahmed, M.; Puckett, S.; Arimilli, S.; Braxton, C.L.; Mizel, S.B.; Lyles, D.S. Stimulation of human dendritic cells by wild-type and M protein mutant vesicular stomatitis viruses engineered to express bacterial flagellin. J. Virol. 2010, 84, 12093–12098. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Knudsen, M.L.; Johansson, D.X.; Kostic, L.; Nordström, E.K.L.; Tegerstedt, K.; Pasetto, A.; Applequist, S.E.; Ljungberg, K.; Sirard, J.-C.; Liljeström, P. The Adjuvant Activity of Alphavirus Replicons Is Enhanced by Incorporating the Microbial Molecule Flagellin into the Replicon. PLoS ONE 2013, 8, e65964. [Google Scholar] [CrossRef] [PubMed]
- Rady, H.F.; Dai, G.; Huang, W.; Shellito, J.E.; Ramsay, A.J. Flagellin Encoded in Gene-Based Vector Vaccines Is a Route-Dependent Immune Adjuvant. PLoS ONE 2016, 11, e0148701. [Google Scholar] [CrossRef] [PubMed]
- Dikic, I. Proteasomal and Autophagic Degradation Systems. Annu. Rev. Biochem. 2017, 86, 193–224. [Google Scholar] [CrossRef] [PubMed]
- Vonderviszt, F.; Kanto, S.; Aizawa, S.; Namba, K. Terminal regions of flagellin are disordered in solution. J. Mol. Biol. 1989, 209, 127–133. [Google Scholar] [CrossRef]
- Newton, S.M.C.; Joys, T.M.; Anderson, S.A.; Kennedy, R.C.; Hovi, M.E.; Stocker, B.A.D. Expression and immunogenicity of an 18-residue epitope of HIV1 gp41 inserted in the flagellar protein of a Salmonella live vaccine. Res. Microbiol. 1995, 146, 203–216. [Google Scholar] [CrossRef]
- Cattozzo, E.M.; Stocker, B.A.D.; Radaelli, A.; De Giuli Morghen, C.; Tognon, M. Expression and immunogenicity of V3 loop epitopes of HIV-1, isolates SC and WMJ2, inserted in Salmonella flagellin. J. Biotechnol. 1997, 56, 191–203. [Google Scholar] [CrossRef]
- Vassilieva, E.V.; Wang, B.-Z.; Vzorov, A.N.; Wang, L.; Wang, Y.-C.; Bozja, J.; Xu, R.; Compans, R.W. Enhanced Mucosal Immune Responses to HIV Virus-Like Particles Containing a Membrane-Anchored Adjuvant. mBio 2010, 2, e00328-10. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajamian, L.; Melnychuk, L.; Jean-Pierre, P.; Zaharatos, G.J. DNA Vaccine-Encoded Flagellin Can Be Used as an Adjuvant Scaffold to Augment HIV-1 gp41 Membrane Proximal External Region Immunogenicity. Viruses 2018, 10, 100. https://doi.org/10.3390/v10030100
Ajamian L, Melnychuk L, Jean-Pierre P, Zaharatos GJ. DNA Vaccine-Encoded Flagellin Can Be Used as an Adjuvant Scaffold to Augment HIV-1 gp41 Membrane Proximal External Region Immunogenicity. Viruses. 2018; 10(3):100. https://doi.org/10.3390/v10030100
Chicago/Turabian StyleAjamian, Lara, Luca Melnychuk, Patrick Jean-Pierre, and Gerasimos J. Zaharatos. 2018. "DNA Vaccine-Encoded Flagellin Can Be Used as an Adjuvant Scaffold to Augment HIV-1 gp41 Membrane Proximal External Region Immunogenicity" Viruses 10, no. 3: 100. https://doi.org/10.3390/v10030100
APA StyleAjamian, L., Melnychuk, L., Jean-Pierre, P., & Zaharatos, G. J. (2018). DNA Vaccine-Encoded Flagellin Can Be Used as an Adjuvant Scaffold to Augment HIV-1 gp41 Membrane Proximal External Region Immunogenicity. Viruses, 10(3), 100. https://doi.org/10.3390/v10030100




