RVFV Infection in Goats by Different Routes of Inoculation
Abstract
1. Introduction
2. Materials & Methods
2.1. Ethics Statement
2.2. Cells
2.3. Virus Production and Titration
2.4. Genome Sequencing
2.4.1. Sanger Sequencing
2.4.2. Bioinformatic Sequencing Analysis
2.4.3. Phylogenetic Analysis
2.5. Goat Inoculation
2.5.1. Subcutaneous (Nubian and LaMancha Goats)
2.5.2. Mosquito-Primed Subcutaneous (Nubian Goats Only)
2.5.3. Intranasal (Nubian and LaMancha Goats)
2.6. Clinical Scoring, Goat Sampling & Tissue Collection
2.6.1. Scoring
2.6.2. Sampling
2.6.3. Tissues
2.7. Virus Isolation by Plaque Assay
2.8. RNA Isolation and RT-PCR
2.9. In Situ Hybridization of RVFV-Probe in Tissues
2.10. Neutralizing Antibody Detection (PRNT)
3. Results
3.1. Phylogenetic Analysis of a 2006–2007 Strain of the Kenya-UAP RVFV Strain
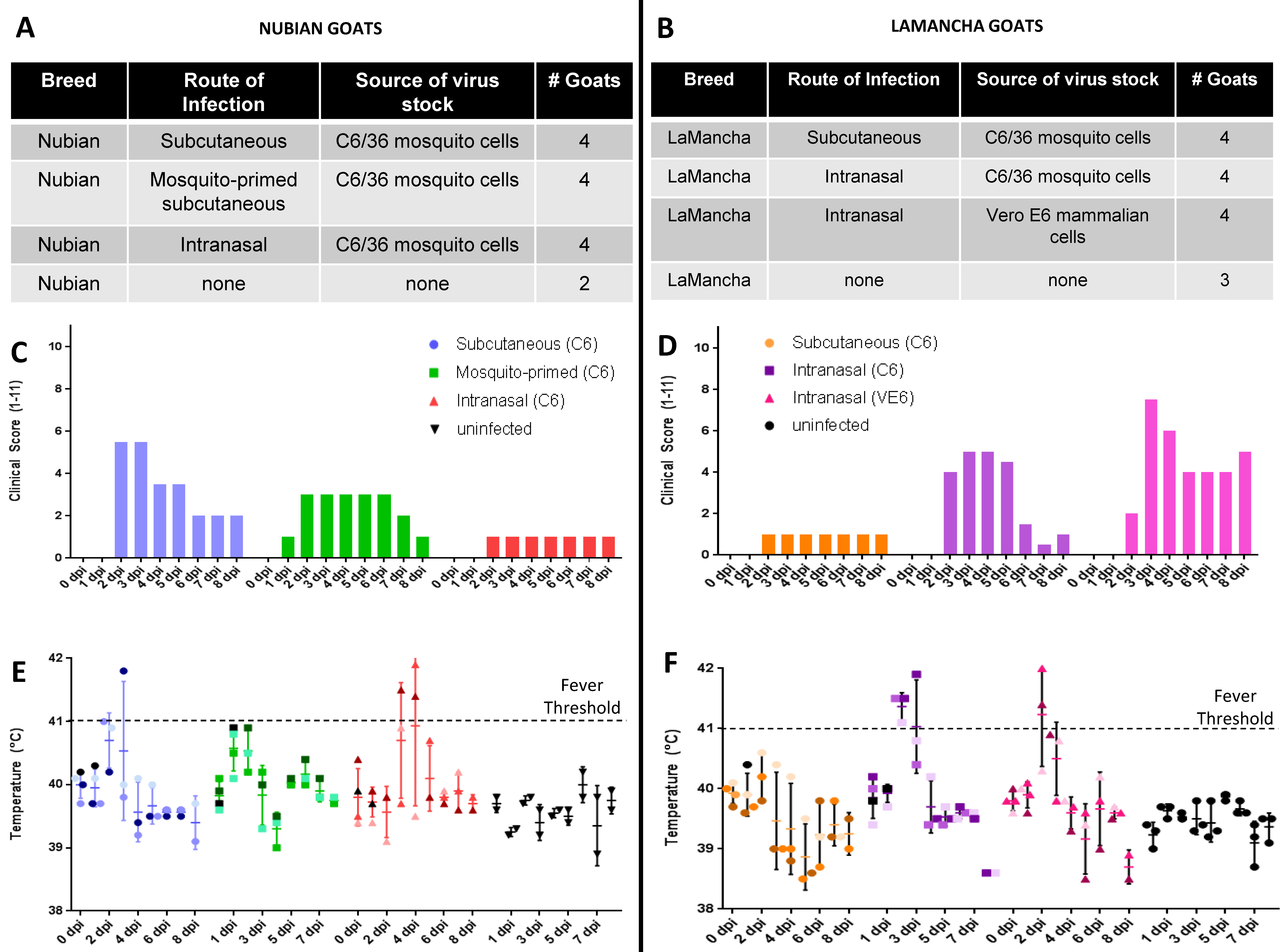
3.2. Experimental Design
3.3. Clinical Signs and Gross Pathology
3.4. Viremia, Shedding and Tissue Viral Load
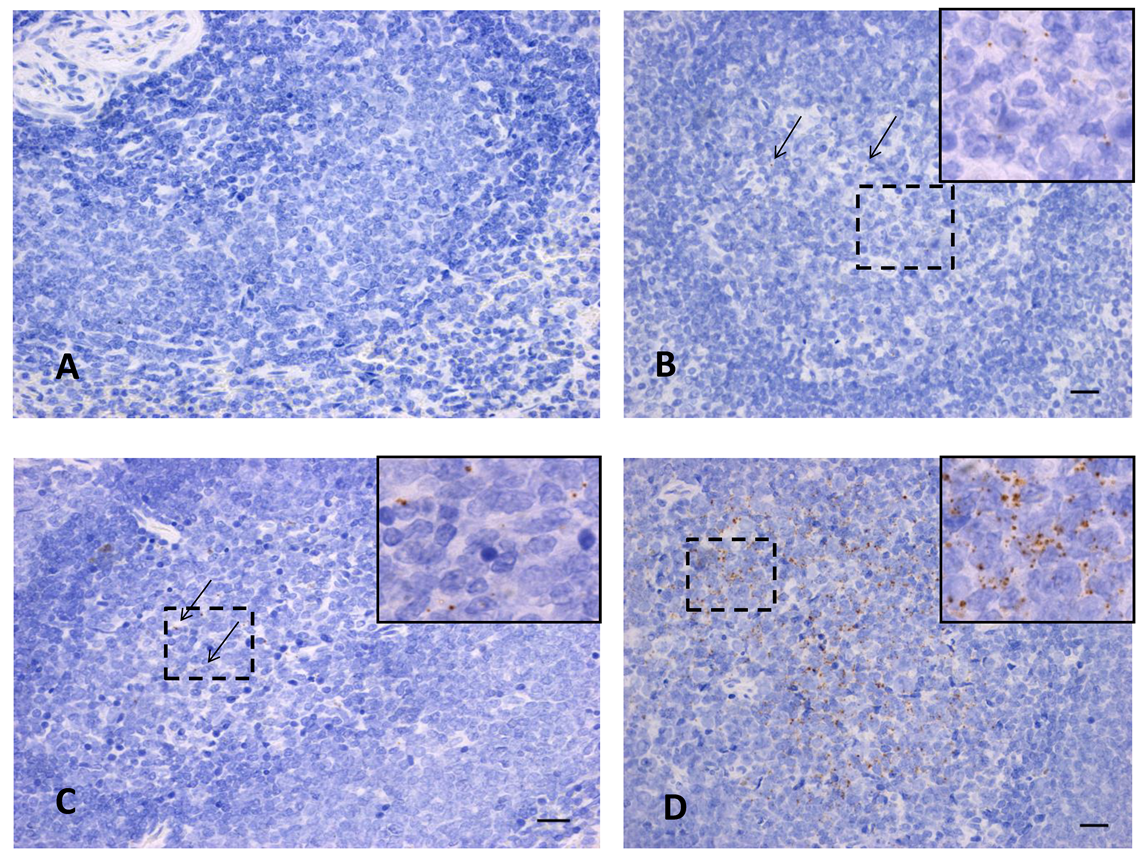
3.5. In-Situ Hybridization for RVFV
3.6. Neutralizing Antibodies
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, J.; Sun, Y.; Shi, W.; Tan, S.; Pan, Y.; Cui, S.; Zhang, Q.; Dou, X.; Lv, Y.; Li, X.; et al. The first imported case of Rift Valley fever in China reveals a genetic reassortment of different viral lineages. Emerg Microbes Infect. 2017, 6, e4. [Google Scholar] [CrossRef]
- Gür, S.; Kale, M.; Erol, N.; Yapici, O.; Mamak, N.; Yavru, S. The first serological evidence for Rift Valley fever infection in the camel, goitered gazelle and Anatolian water buffaloes in Turkey. Trop. Anim. Health Prod. 2017, 49, 1531–1535. [Google Scholar] [CrossRef]
- Bett, B.; Kiunga, P.; Gachohi, J.; Sindato, C.; Mbotha, D.; Robinson, T.; Lindahl, J.; Grace, D. Effects of climate change on the occurrence and distribution of livestock diseases. Prev. Vet. Med. 2017, 137, 119–129. [Google Scholar] [CrossRef]
- Balkhy, H.H.; Memish, Z.A. Rift Valley Fever: an uninvited zoonosis in the Arabian peninsula. Int J Antimicrob Agents. 2003, 21, 153–157. [Google Scholar] [CrossRef]
- Bird, B.H.; Bawiec, D.A.; Ksiazek, T.G.; Shoemaker, T.R.; Nichol, S.T. Highly sensitive and broadly reactive quantitative reverse transcription-PCR assay for high-throughput detection of Rift Valley fever virus. J. Clin. Microbiol. 2007, 45, 3506–3513. [Google Scholar] [CrossRef]
- Chevalier, V. Relevance of Rift Valley fever to public health in the European Union. Clin Microbiol Infect. 2013, 19, 705–708. [Google Scholar] [CrossRef]
- Chevalier, V.; Pepin, M.; Plee, L.; Lancelot, R. Rift Valley fever–a threat for Europe? Euro Surveill. 2010, 10, 19506. [Google Scholar] [CrossRef]
- Davies, F.G.; Linthicum, K.J.; James, A.D. Rainfall and epizootic Rift Valley fever. Bull. World Health Organ. 1985, 63, 941–943. [Google Scholar]
- Hassan, O.A.; Ahlm, C.; Sang, R.; Evander, M. The 2007 Rift valley fever outbreak in Sudan. PLoS Negl. Trop. Dis. 2011. [Google Scholar] [CrossRef]
- Leedale, J.; Jones, A.E.; Caminade, C.; Morse, A.P. A dynamic, climate-driven model of rift valley fever. Geospat. Health 2016, 11. [Google Scholar] [CrossRef]
- Redding, D.W.; Tiedt, S.; Lo Iacono, G.; Bett, B.; Jones, K.E. Spatial, seasonal and climatic predictive models of Rift Valley fever disease across Africa. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160165. [Google Scholar] [CrossRef]
- Napp, S.; Chevalier, V.; Busquets, N.; Calistri, P.; Casal, J.; Attia, M.; Elbassal, R.; Hosni, H.; Farrag, H.; Hassan, N.; et al. Understanding the legal trade of cattle and camels and the derived risk of Rift Valley Fever introduction into and transmission within Egypt. PLoS Negl. Trop. Dis. 2018, 12. [Google Scholar] [CrossRef]
- Brustolin, M.; Talavera, S.; NuÑez, A.; SantamarÍa, C.; Rivas, R.; Pujol, N.; Valle, M.; Verdún, M.; Brun, A.; Pagès, N.; et al. Rift Valley fever virus and European mosquitoes: Vector competence of Culex pipiens and Stegomyia albopicta (= Aedes albopictus). Med. Vet. Entomol. 2017, 31, 365–372. [Google Scholar] [CrossRef]
- Ndiaye, E.H.; Fall, G.; Gaye, A.; Bob, N.S.; Talla, C.; Diagne, C.T.; Diallo, D.; BA, Y.; Dia, I.; Kohl, A.; et al. Vector competence of Aedes vexans (Meigen), Culex poicilipes (Theobald) and Cx. quinquefasciatus Say from Senegal for West and East African lineages of Rift Valley fever virus. Parasites Vectors 2016, 9. [Google Scholar] [CrossRef]
- Turell, M.J.; Britch, S.C.; Aldridge, R.L.; Xue, R.; De Smith, M.L.; Cohnstaedt, L.W.; Linthicum, K.J. Potential for Psorophora columbiae and Psorophora ciliata Mosquitoes (Diptera: Culicidae) to Transmit Rift Valley Fever Virus. J. Med. Entomol. 2015, 52, 1111–1116. [Google Scholar] [CrossRef]
- Coetzer, J.A. The pathology of Rift Valley fever. I. Lesions occurring in natural cases in new-born lambs. Onderstepoort J. Vet. Res. 1977, 4, 205–211. [Google Scholar]
- Coetzer, J.A. The pathology of Rift Valley fever. II. Lesions occurring in field cases in adult cattle, calves and aborted foetuses. Onderstepoort J. Vet. Res. 1982, 1, 11–17. [Google Scholar]
- Archer, B.N.; Thomas, J.; Weyer, J.; Cengimbo, A.; Landoh, D.E.; Jacobs, C.; Ntuli, S.; Modise, M.; Mathonsi, M.; Blumberg, L.; et al. Epidemiologic investigations into outbreaks of Rift Valley fever in humans, South Africa, 2008–2011. Emerg. Infect. Dis. 2013, 19, 1918–1925. [Google Scholar] [CrossRef]
- Métras, R.; Porphyre, T.; Pfeiffer, D.U.; Kemp, A.; Thompson, P.N.; Collins, L.M.; White, R.G. Exploratory Space-Time Analyses of Rift Valley Fever in South Africa in 2008–2011. PLoS Negl. Trop. Dis. 2012, 6. [Google Scholar] [CrossRef]
- Monaco, F.; Pinoni, C.; Cosseddu, G.M.; Khaiseb, S.; Calistri, P.; Molini, U.; Bishi, A.; Conte, A.; Scacchia, M.; Lelli, R. Rift Valley fever in Namibia, 2010. Emerg. Infect. Dis. 2013, 19, 2025–2027. [Google Scholar] [CrossRef]
- Nicholas, D.E.; Jacobsen, K.H.; Waters, N.M. Risk factors associated with human Rift Valley fever infection: Systematic review and meta-analysis. Trop. Med. Int. Health 2014, 19. [Google Scholar] [CrossRef]
- Ikegami, T.; Makino, S. The pathogenesis of rift valley fever. Viruses 2011, 3, 493–519. [Google Scholar] [CrossRef]
- Nguku, P.M.; Sharif, S.K.; Mutonga, D.; Amwayi, S.; Omolo, J.; Mohammed, O.; Farnon, E.C.; Gould, L.H.; Lederman, E.; Rao, C.; et al. An investigation of a major outbreak of rift valley fever in Kenya: 2006–2007. Am. J. Trop. Med. Hyg. 2010, 83 (Suppl. 2), 5–13. [Google Scholar] [CrossRef]
- Sow, A.; Faye, O.; Ba, Y.; Ba, H.; Diallo, D.; Faye, O.; Loucoubar, C.; Boushab, M.; Barry, Y.; Diallo, M.; et al. Rift valley fever outbreak, Southern Mauritania, 2012. Emerg. Infect. Dis. 2014, 20, 296–299. [Google Scholar] [CrossRef]
- Dungu, B.; Louw, I.; Lubisi, A.; Hunter, P.; von Teichman, B.F.; Bouloy, M. Evaluation of the efficacy and safety of the Rift Valley Fever Clone 13 vaccine in sheep. Vaccine 2010, 28, 4581–4587. [Google Scholar] [CrossRef]
- Indran, S.V.; Ikegami, T. Novel approaches to develop Rift Valley fever vaccines. Front. Cell. Infect. Microbiol. 2012, 2. [Google Scholar] [CrossRef]
- Kortekaas, J.; Antonis, A.F.G.; Kant, J.; Vloet, R.P.M.; Vogel, A.; Oreshkova, N.; de Boer, S.M.; Bosch, B.J.; Moormann, R.J.M. Efficacy of three candidate Rift Valley fever vaccines in sheep. Vaccine 2012, 30, 3423–3429. [Google Scholar] [CrossRef]
- Mansfield, K.L.; Banyard, A.C.; McElhinney, L.; Johnson, N.; Horton, D.L.; Hernández-Triana, L.M.; Fooks, A.R. Rift Valley fever virus: A review of diagnosis and vaccination, and implications for emergence in Europe. Vaccine 2015. [Google Scholar] [CrossRef]
- Morrill, J.C.; Laughlin, R.C.; Lokugamage, N.; Wu, J.; Pugh, R.; Kanani, P.; Adams, L.G.; Makino, S.; Peters, C.J. Immunogenicity of a recombinant Rift Valley fever MP-12-NSm deletion vaccine candidate in calves. Vaccine 2013, 31, 4988–4994. [Google Scholar] [CrossRef]
- Pittman, P.R.; McClain, D.; Quinn, X.; Coonan, K.M.; Mangiafico, J.; Makuch, R.S.; Morrill, J.; Peters, C.J. Safety and immunogenicity of a mutagenized, live attenuated Rift Valley fever vaccine, MP-12, in a Phase 1 dose escalation and route comparison study in humans. Vaccine 2016, 34, 424–429. [Google Scholar] [CrossRef]
- Soi, R.K.; Rurangirwa, F.R.; McGuire, T.C.; Rwambo, P.M.; DeMartini, J.C.; Crawford, T.B. Protection of sheep against Rift Valley fever virus and sheep poxvirus with a recombinant capripoxvirus vaccine. Clin. Vaccine Immunol. 2010, 17, 1842–1849. [Google Scholar] [CrossRef]
- Warimwe, G.M.; Lorenzo, G.; Lopez-Gil, E.; Reyes-Sandoval, A.; Cottingham, M.G.; Spencer, A.J.; Collins, K.A.; Dicks, M.D.; Amar Lall, A.M.; Furze, J.; et al. Immunogenicity and efficacy of a chimpanzee adenovirus-vectored Rift Valley Fever vaccine in mice. Virol. J. 2013, 10. [Google Scholar] [CrossRef]
- Wilson, W.C.; Davis, A.S.; Gaudreault, N.N.; Faburay, B.; Trujillo, J.D.; Shivanna, V.; Sunwoo, S.Y.; Balogh, A.; Endalew, A.; Ma, W.; et al. Experimental infection of calves by two genetically-distinct strains of rift valley fever virus. Viruses 2016, 8, 145. [Google Scholar] [CrossRef]
- Faburay, B.; Gaudreault, N.N.; Liu, Q.; Davis, A.S.; Shivanna, V.; Sunwoo, S.Y.; Lang, Y.; Morozov, I.; Ruder, M.; Drolet, B.; et al. Development of a sheep challenge model for Rift Valley fever. Virology 2016, 489, 128–140. [Google Scholar] [CrossRef]
- Weingartl, H.M.; Miller, M.; Nfon, C.; Wilson, W.C. Development of a rift valley fever virus viremia challenge model in sheep and goats. Vaccine 2014, 32, 2337–2344. [Google Scholar] [CrossRef]
- Nfon, C.K.; Marszal, P.; Zhang, S.; Weingartl, H.M. Innate immune response to rift valley fever virus in goats. PLoS Negl. Trop. Dis. 2012, 6. [Google Scholar] [CrossRef]
- Le Coupanec, A.; Babin, D.; Fiette, L.; Jouvion, G.; Ave, P.; Misse, D.; Bouloy, M.; Choumet, V. Aedes Mosquito Saliva Modulates Rift Valley Fever Virus Pathogenicity. PLoS Negl. Trop. Dis. 2013, 7. [Google Scholar] [CrossRef]
- Reed, C.; Lin, K.; Wilhelmsen, C.; Friedrich, B.; Nalca, A.; Keeney, A.; Donnelly, G.; Shamblin, J.; Hensley, L.E.; Olinger, G.; et al. Aerosol Exposure to Rift Valley Fever Virus Causes Earlier and More Severe Neuropathology in the Murine Model, which Has Important Implications for Therapeutic Development. PLoS Negl. Trop. Dis. 2013, 7. [Google Scholar] [CrossRef]
- Hartman, A.L.; Powell, D.S.; Bethel, L.M.; Caroline, A.L.; Schmid, R.J.; Oury, T.; Reed, D.S. Aerosolized Rift Valley Fever Virus Causes Fatal Encephalitis in African Green Monkeys and Common Marmosets. J. Virol. 2014, 88, 2235–2245. [Google Scholar] [CrossRef]
- Smith, D.R.; Bird, B.H.; Lewis, B.; Johnston, S.C.; McCarthy, S.; Keeney, A.; Botto, M.; Donnelly, G.; Shamblin, J.; Albariño, C.G.; et al. Development of a novel nonhuman primate model for Rift Valley fever. J. Virol. 2012, 86, 2109–2120. [Google Scholar] [CrossRef]
- Yedloutschnig, R.J.; Dardiri, A.H.W.J. Rift Valley Fever Infection in sheep by contact exposure. Contr. Epidem. Biostat. 1981, 3, 53–59. [Google Scholar]
- Moutailler, S.; Roche, B.; Thiberge, J.M.; Caro, V.; Rougeon, F.; Failloux, A.B. Host alternation is necessary to maintain the genome stability of Rift Valley fever virus. PLoS Negl. Trop. Dis. 2011, 5. [Google Scholar] [CrossRef]
- Weingartl, H.M.; Zhang, S.; Marszal, P.; McGreevy, A.; Burton, L.; Wilson, W. Rift Valley Fever Virus Incorporates the 78 kDa Glycoprotein into Virions Matured in Mosquito C6/36 Cells. PLoS ONE 2014, 9, e87385. [Google Scholar] [CrossRef]
- Jones, D.T.; Taylor, W.R.; Thornton, J.M. The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci. 1992, 8, 275–282. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Espach, A.; Romito, M.; Nel, L.H.; Viljoen, G.J. Development of a diagnostic one-tube RT-PCR for the detection of Rift Valley fever virus. Onderstepoort J. Vet. Res. 2002, 69, 247–252. [Google Scholar]
- Schaeffer, E.; Flacher, V.; Papageorgiou, V.; Decossas, M.; Fauny, J.; Kramer, M.; Mueller, C. Dermal CD14+ dendritic cell and macrophage infection by dengue virus is stimulated by interleukin-4. J. Investig. Dermatol. 2015, 135, 1743–1751. [Google Scholar] [CrossRef]
- Klimstra, W.B.; Nangle, E.M.; Smith, M.S.; Yurochko, A.D.; Ryman, K.D. DC-SIGN and L-SIGN can act as attachment receptors for alphaviruses and distinguish between mosquito cell- and mammalian cell-derived viruses. J. Virol. 2003, 77, 12022–12032. [Google Scholar] [CrossRef]
- Agarwal, A.; Joshi, G.; Nagar, D.P.; Sharma, A.K.; Sukumaran, D.; Pant, S.C.; Pant, S.C.; Parida, M.M.; Dash, P.K. Mosquito saliva induced cutaneous events augment Chikungunya virus replication and disease progression. Infect. Genet. Evol. 2016, 40, 126–135. [Google Scholar] [CrossRef]
- Styer, L.M.; Lim, P.-Y.; Louie, K.L.; Albright, R.G.; Kramer, L.D.; Bernard, K.A. Mosquito Saliva Causes Enhancement of West Nile Virus Infection in Mice. J. Virol. 2011, 85, 1517–1527. [Google Scholar] [CrossRef]
- Wichit, S.; Ferraris, P.; Choumet, V.; Missé, D. The effects of mosquito saliva on dengue virus infectivity in humans. Curr. Opin. Virol. 2016. [Google Scholar] [CrossRef]
- Rippy, M.K.; Topper, M.J.; Mebus, C.A.; Morrill, J.C. Rift Valley Fever Virus-induced Encephalomyelitis and Hepatitis in Calves. Vet. Pathol. 1992, 29, 495–502. [Google Scholar] [CrossRef]
- Gaudreault, N.N.; Indran, S.V.; Bryant, P.K.; Richt, J.A.; Wilson, W.C. Comparison of Rift Valley fever virus replication in NOrth American livestock and wildlife cell lines. Front. Microbiol. 2015, 6, 664. [Google Scholar] [CrossRef]
- Busquets, N.; Xavier, F.; Martín-Folgar, R.; Lorenzo, G.; Galindo-Cardiel, I.; del Val, B.P.; Rivas, R.; Iglesias, J.; Rodríguez, F.; Solanes, D.; et al. Experimental infection of young adult European breed sheep with Rift Valley fever virus field isolates. Vector Borne Zoonotic Dis. 2010, 10, 689–696. [Google Scholar] [CrossRef]
- Easterday, B.C.; Mcgavran, M.H.; Rooney, J.R.; Murphy, L.C. The pathogenesis of Rift Valley fever in lambs. Am. J. Vet. Res. 1962, 23, 470–479. [Google Scholar]
- Harrington, D.G.; Lupton, H.W.; Crabbs, C.L.; Peters, C.J.; Reynolds, J.A.; Slone, T.W. Evaluation of a formalin-inactivated Rift Valley fever vaccine in sheep. Am. J. Vet. Res. 1980, 41, 1559–1564. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kroeker, A.L.; Smid, V.; Embury-Hyatt, C.; Moffat, E.; Collignon, B.; Lung, O.; Lindsay, R.; Weingartl, H. RVFV Infection in Goats by Different Routes of Inoculation. Viruses 2018, 10, 709. https://doi.org/10.3390/v10120709
Kroeker AL, Smid V, Embury-Hyatt C, Moffat E, Collignon B, Lung O, Lindsay R, Weingartl H. RVFV Infection in Goats by Different Routes of Inoculation. Viruses. 2018; 10(12):709. https://doi.org/10.3390/v10120709
Chicago/Turabian StyleKroeker, Andrea L., Valerie Smid, Carissa Embury-Hyatt, Estella Moffat, Brad Collignon, Oliver Lung, Robbin Lindsay, and Hana Weingartl. 2018. "RVFV Infection in Goats by Different Routes of Inoculation" Viruses 10, no. 12: 709. https://doi.org/10.3390/v10120709
APA StyleKroeker, A. L., Smid, V., Embury-Hyatt, C., Moffat, E., Collignon, B., Lung, O., Lindsay, R., & Weingartl, H. (2018). RVFV Infection in Goats by Different Routes of Inoculation. Viruses, 10(12), 709. https://doi.org/10.3390/v10120709



