Antiviral RNA Interference Activity in Cells of the Predatory Mosquito, Toxorhynchites amboinensis
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Viruses
2.3. Plasmids
2.4. In Vitro Transcription of dsRNA
2.5. Nucleic Acid Transfection
2.6. Luciferase Assay
2.7. Small RNA Sequencing and Analysis
2.8. In Vitro Dicer Cleavage Assay
2.9. Statistical Analysis
2.10. Data Availability
3. Results
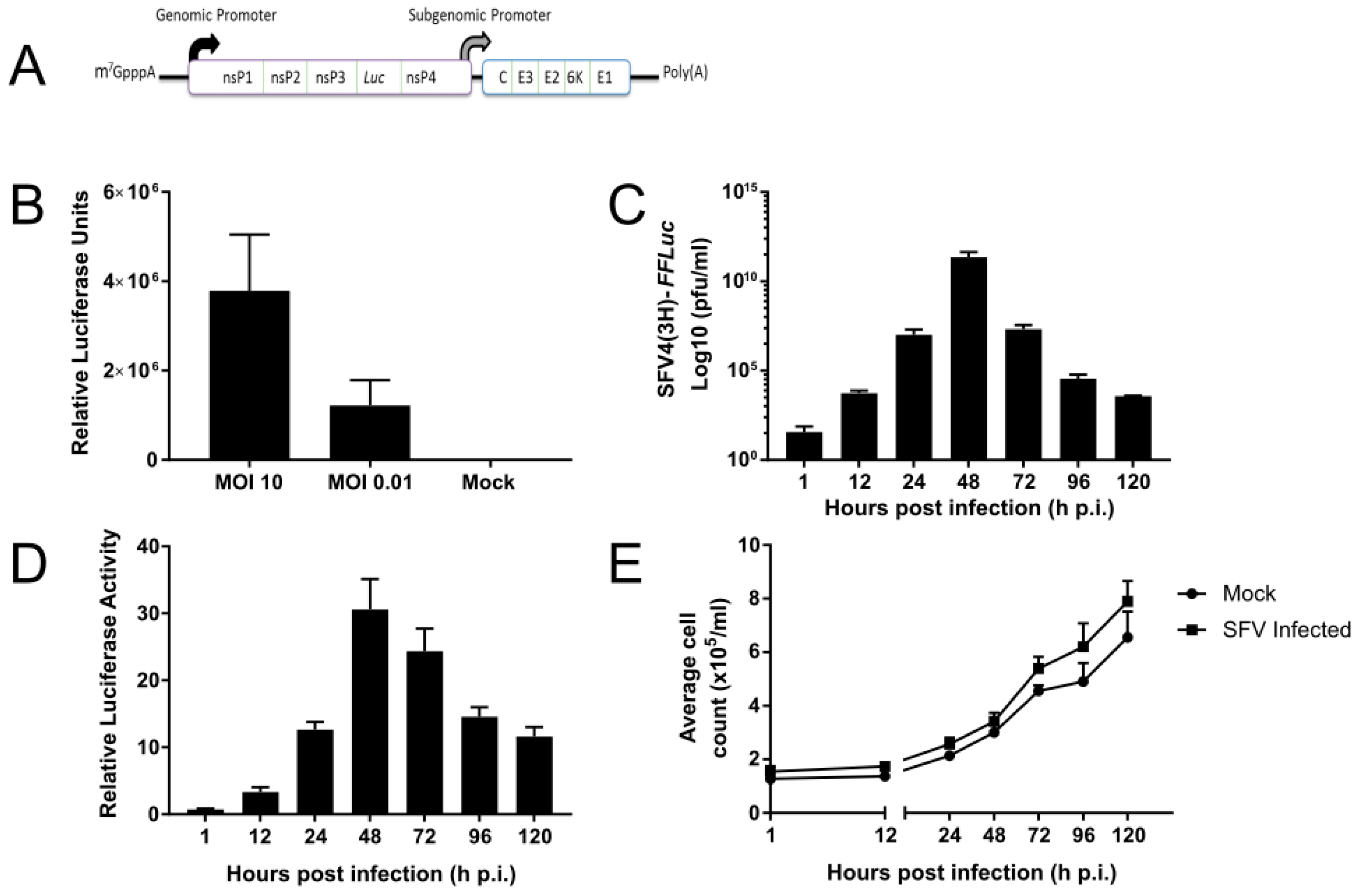
3.1. SFV Infects Tx. amboinensis-Derived TRA-171 Cells
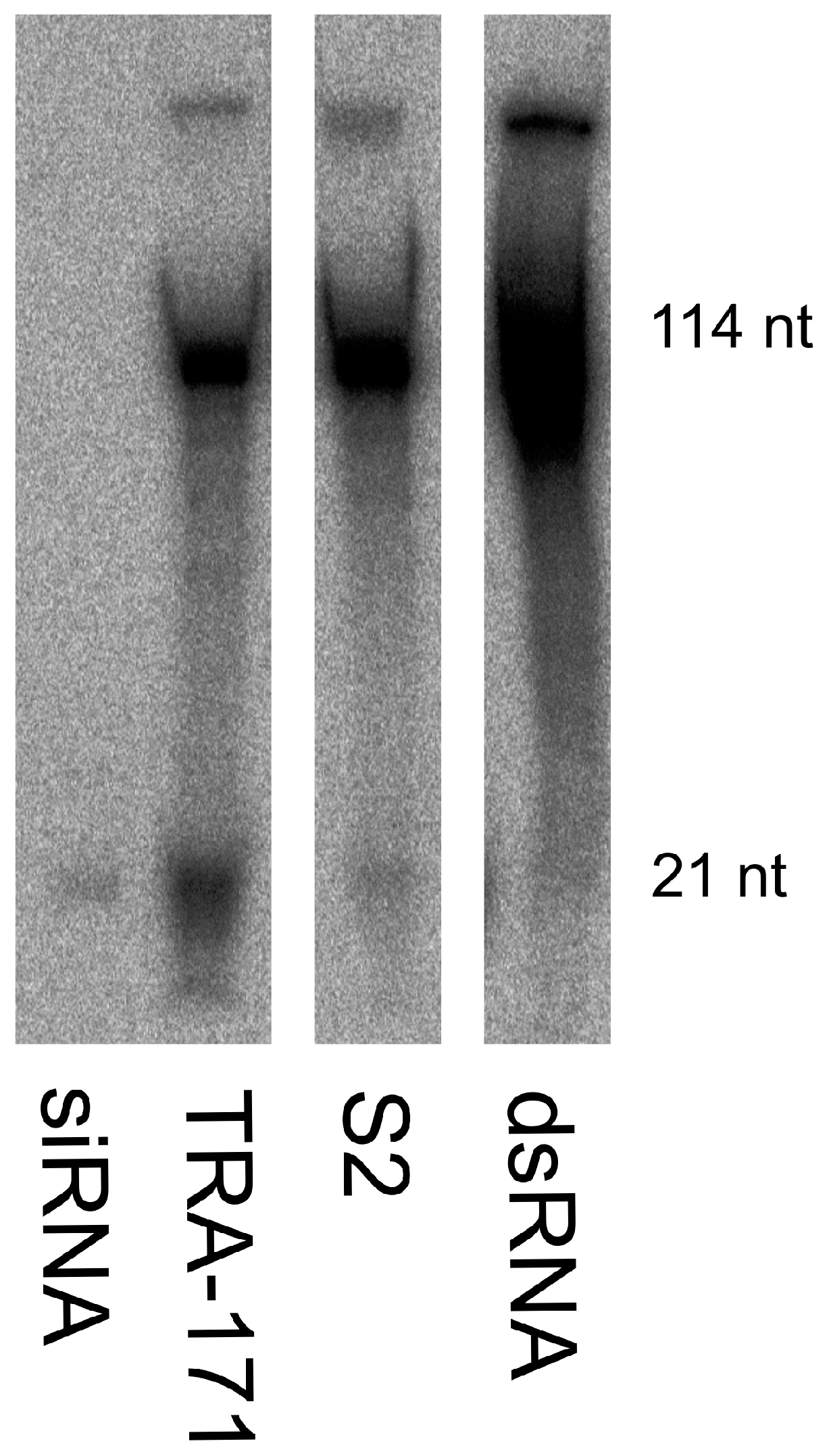
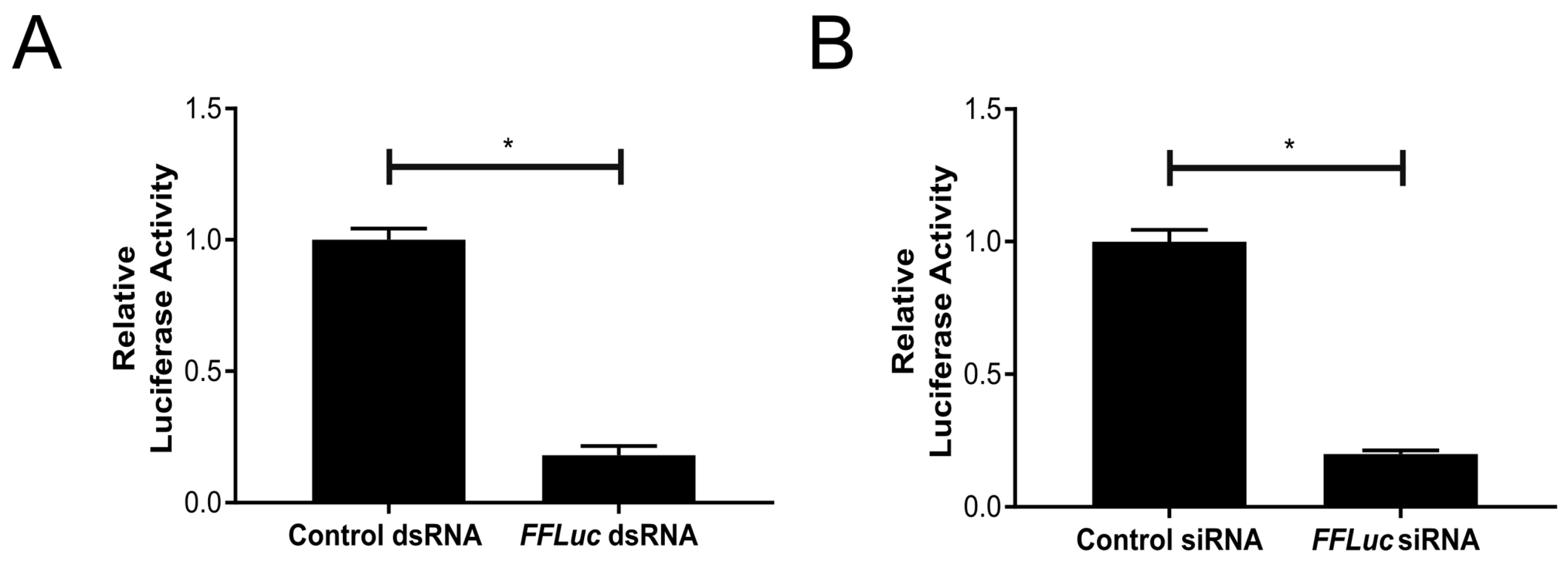
3.2. Functional RNAi Pathways are Present in TRA-171 Cells
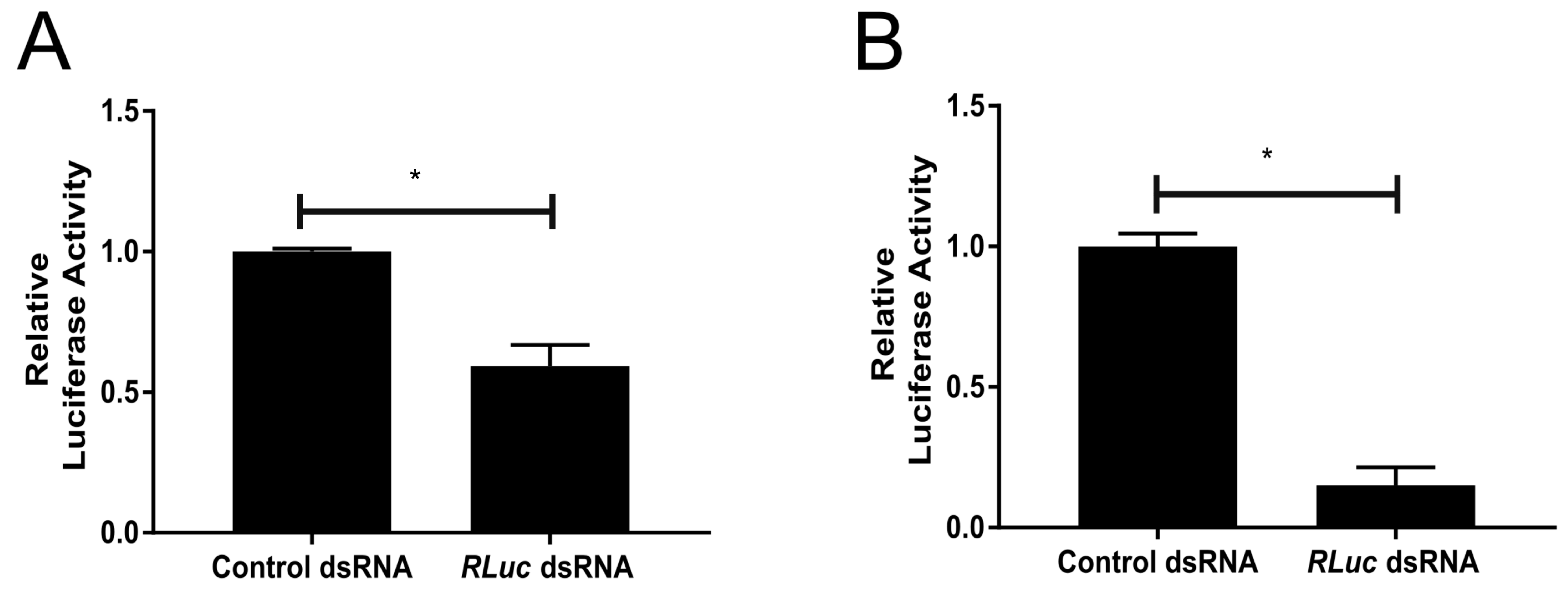
3.3. An Active dsRNA-Inducible RNAi Response has Antiviral Activity against SFV Infection in TRA-171 Cells
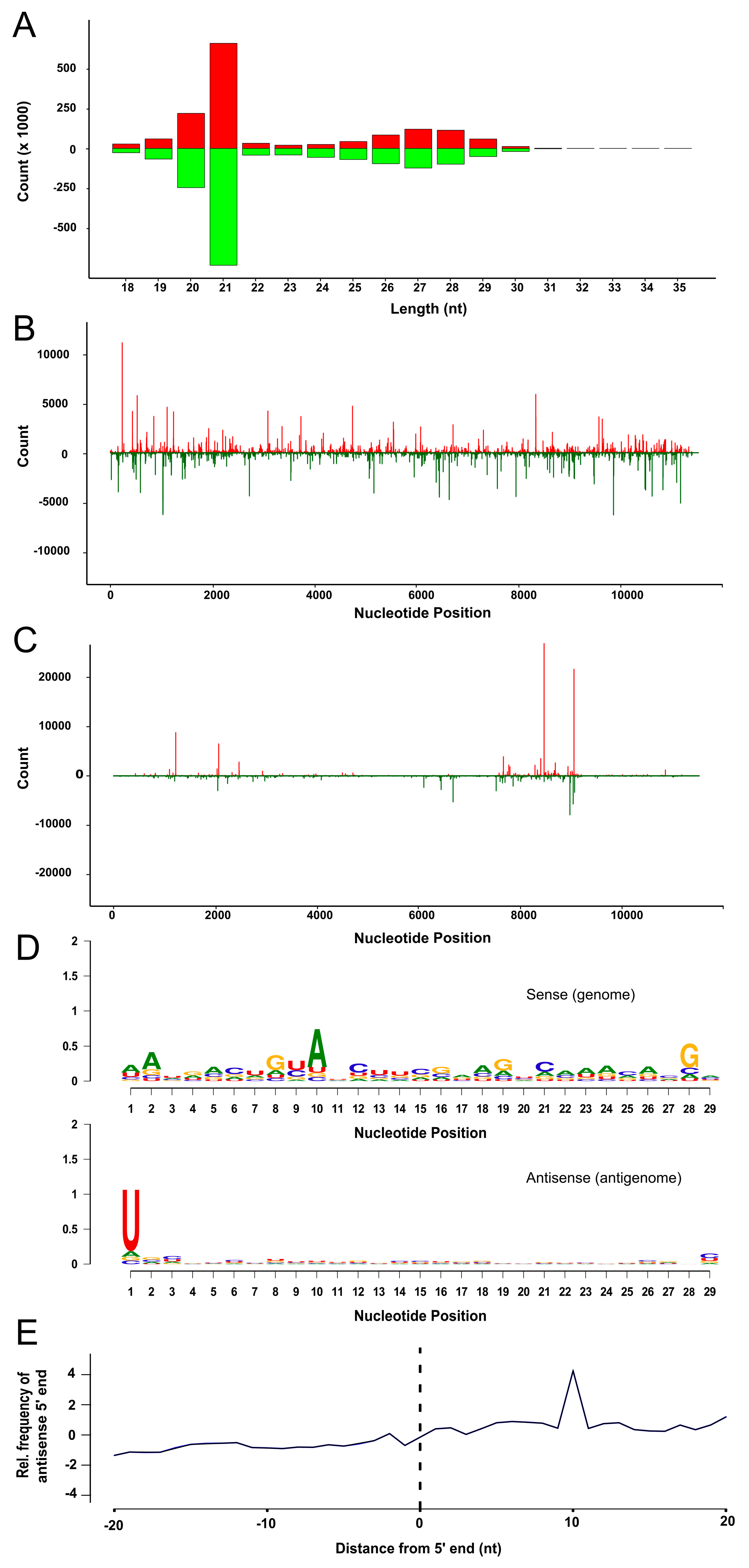
3.4. SFV Infection Induces Small RNA Production in TRA-171 Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zuharah, W.F.; Fadzly, N.; Yusof, N.A.; Dieng, H. Risky behaviors: Effects of toxorhynchites splendens (diptera: Culicidae) predator on the behavior of three mosquito species. J. Insect Sci. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.S.; Higgs, S.; Vanlandingham, D.L. Biological control strategies for mosquito vectors of arboviruses. Insects 2017, 8, 21. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, E.T. Toxorhynchites. J. Am. Mosq. Control. Assoc. 2007, 23, 129–132. [Google Scholar] [CrossRef]
- Focks, D.A. Toxorhynchites as biocontrol agents. J. Am. Mosq. Control. Assoc. 2007, 23, 118–127. [Google Scholar] [CrossRef]
- Rosen, L. The use of toxorhynchites mosquitoes to detect and propagate dengue and other arboviruses. Am. J. Trop. Med. Hyg. 1981, 30, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.; Shroyer, D.A. Comparative susceptibility of five species of toxorhynchites mosquitoes to parenteral infection with dengue and other flaviviruses. Am. J. Trop. Med. Hyg. 1985, 34, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.; Tesh, R.B.; Lien, J.C.; Cross, J.H. Transovarial transmission of Japanese encephalitis virus by mosquitoes. Science 1978, 199, 909–911. [Google Scholar] [CrossRef] [PubMed]
- Tesh, R.B. A method for the isolation and identification of dengue viruses, using mosquito cell cultures. Am. J. Trop. Med. Hyg. 1979, 28, 1053–1059. [Google Scholar] [CrossRef]
- Tesh, R.B.; McLean, R.G.; Shroyer, D.A.; Calisher, C.H.; Rosen, L. Ross river virus (togaviridae: Alphavirus) infection (epidemic polyarthritis) in American Samoa. Trans. R. Soc. Trop. Med. Hyg. 1981, 75, 426–431. [Google Scholar] [CrossRef]
- Scherer, W.F.; Chin, J. Sensitivity of toxorhynchites amboinensis mosquitoes versus chicken embryonic cell cultures for assays of Venezuelan encephalitis virus. J. Clin. Microbiol. 1981, 13, 947–950. [Google Scholar]
- Kuno, G.; Gubler, D.J.; Velez, M.; Oliver, A. Comparative sensitivity of three mosquito cell lines for isolation of dengue viruses. Bull. World Health Organ. 1985, 63, 279–286. [Google Scholar] [PubMed]
- Kuno, G. Persistent infection of a nonvector mosquito cell line (TRA-171) with dengue viruses. Intervirology 1982, 18, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Kuno, G. Replication of dengue, yellow fever, St. Louis encephalitis and vesicular stomatitis viruses in a cell line (TRA-171) derived from Toxorhynchites amboinensis. In Vitro 1981, 17, 1011–1015. [Google Scholar] [CrossRef] [PubMed]
- Sanchez Legrand, F.; Hotta, S. Susceptibility of cloned toxorhynchites amboinensis cells to dengue and chikungunya viruses. Microbiol. Immunol. 1983, 27, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Lequime, S.; Lambrechts, L. Vertical transmission of arboviruses in mosquitoes: A historical perspective. Infect. Genet. Evol. 2014, 28, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Merkling, S.H.; van Rij, R.P. Beyond RNAi: Antiviral defense strategies in drosophila and mosquito. J. Insect Physiol. 2013, 59, 159–170. [Google Scholar] [CrossRef]
- Blair, C.D. Mosquito RNAi is the major innate immune pathway controlling arbovirus infection and transmission. Future Microbiol. 2011, 6, 265–277. [Google Scholar] [CrossRef]
- Blair, C.D.; Olson, K.E. The role of RNA interference (RNAi) in arbovirus-vector interactions. Viruses 2015, 7, 820–843. [Google Scholar] [CrossRef]
- Olson, K.E.; Blair, C.D. Arbovirus-mosquito interactions: RNAi pathway. Curr. Opin. Virol. 2015, 15, 119–126. [Google Scholar] [CrossRef]
- Donald, C.L.; Kohl, A.; Schnettler, E. New insights into control of arbovirus replication and spread by insect RNA interference pathways. Insects 2012, 3, 511–531. [Google Scholar] [CrossRef]
- Samuel, G.H.; Adelman, Z.N.; Myles, K.M. Antiviral immunity and virus-mediated antagonism in disease vector mosquitoes. Trends Microbiol. 2018, 26, 447–461. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, I.; Shi, X.; McFarlane, M.; Watson, M.; Blomstrom, A.L.; Skelton, J.K.; Kohl, A.; Elliott, R.M.; Schnettler, E. The antiviral RNAi response in vector and non-vector cells against orthobunyaviruses. PLoS Negl. Trop. Dis. 2017, 11, e0005272. [Google Scholar] [CrossRef] [PubMed]
- McFarlane, M.; Arias-Goeta, C.; Martin, E.; O’Hara, Z.; Lulla, A.; Mousson, L.; Rainey, S.M.; Misbah, S.; Schnettler, E.; Donald, C.L.; et al. Characterization of Aedes aegypti innate-immune pathways that limit chikungunya virus replication. PLoS Negl. Trop. Dis. 2014, 8, e2994. [Google Scholar] [CrossRef] [PubMed]
- Schnettler, E.; Donald, C.L.; Human, S.; Watson, M.; Siu, R.W.; McFarlane, M.; Fazakerley, J.K.; Kohl, A.; Fragkoudis, R. Knockdown of piRNA pathway proteins results in enhanced semliki forest virus production in mosquito cells. J. Gen. Virol. 2013, 94, 1680–1689. [Google Scholar] [CrossRef] [PubMed]
- Varjak, M.; Donald, C.L.; Mottram, T.J.; Sreenu, V.B.; Merits, A.; Maringer, K.; Schnettler, E.; Kohl, A. Characterization of the zika virus induced small RNA response in Aedes aegypti cells. PLoS Negl. Trop. Dis. 2017, 11, e0006010. [Google Scholar] [CrossRef] [PubMed]
- Myles, K.M.; Wiley, M.R.; Morazzani, E.M.; Adelman, Z.N. Alphavirus-derived small RNAs modulate pathogenesis in disease vector mosquitoes. Proc. Natl. Acad. Sci. USA 2008, 105, 19938–19943. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vargas, I.; Scott, J.C.; Poole-Smith, B.K.; Franz, A.W.; Barbosa-Solomieu, V.; Wilusz, J.; Olson, K.E.; Blair, C.D. Dengue virus type 2 infections of Aedes aegypti are modulated by the mosquito’s RNA interference pathway. PLoS Pathog. 2009, 5, e1000299. [Google Scholar] [CrossRef] [PubMed]
- Siu, R.W.; Fragkoudis, R.; Simmonds, P.; Donald, C.L.; Chase-Topping, M.E.; Barry, G.; Attarzadeh-Yazdi, G.; Rodriguez-Andres, J.; Nash, A.A.; Merits, A.; et al. Antiviral RNA interference responses induced by semliki forest virus infection of mosquito cells: Characterization, origin, and frequency-dependent functions of virus-derived small interfering RNAs. J. Virol. 2011, 85, 2907–2917. [Google Scholar] [CrossRef]
- Scott, J.C.; Brackney, D.E.; Campbell, C.L.; Bondu-Hawkins, V.; Hjelle, B.; Ebel, G.D.; Olson, K.E.; Blair, C.D. Comparison of dengue virus type 2-specific small RNAs from RNA interference-competent and -incompetent mosquito cells. PLoS Negl. Trop. Dis. 2010, 4, e848. [Google Scholar] [CrossRef]
- Leger, P.; Lara, E.; Jagla, B.; Sismeiro, O.; Mansuroglu, Z.; Coppee, J.Y.; Bonnefoy, E.; Bouloy, M. Dicer-2- and piwi-mediated RNA interference in rift valley fever virus-infected mosquito cells. J. Virol. 2013, 87, 1631–1648. [Google Scholar] [CrossRef]
- Vodovar, N.; Bronkhorst, A.W.; van Cleef, K.W.; Miesen, P.; Blanc, H.; van Rij, R.P.; Saleh, M.C. Arbovirus-derived piRNAs exhibit a ping-pong signature in mosquito cells. PLoS ONE 2012, 7, e30861. [Google Scholar] [CrossRef] [PubMed]
- Morazzani, E.M.; Wiley, M.R.; Murreddu, M.G.; Adelman, Z.N.; Myles, K.M. Production of virus-derived ping-pong-dependent piRNA-like small RNAs in the mosquito soma. PLoS Pathog. 2012, 8, e1002470. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Keene, K.M.; Brackney, D.E.; Olson, K.E.; Blair, C.D.; Wilusz, J.; Foy, B.D. Aedes aegypti uses RNA interference in defense against sindbis virus infection. BMC Microbiol 2008, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Brackney, D.E.; Beane, J.E.; Ebel, G.D. RNAi targeting of West Nile virus in mosquito midguts promotes virus diversification. PLoS Pathog. 2009, 5, e1000502. [Google Scholar] [CrossRef] [PubMed]
- Miesen, P.; Girardi, E.; van Rij, R.P. Distinct sets of piwi proteins produce arbovirus and transposon-derived piRNAs in Aedes aegypti mosquito cells. Nucleic Acids Res. 2015, 43, 6545–6556. [Google Scholar] [CrossRef] [PubMed]
- Miesen, P.; Ivens, A.; Buck, A.H.; van Rij, R.P. Small RNA profiling in dengue virus 2-infected Aedes mosquito cells reveals viral piRNAs and novel host mirnas. PLoS Negl. Trop. Dis. 2016, 10, e0004452. [Google Scholar] [CrossRef] [PubMed]
- Varjak, M.; Maringer, K.; Watson, M.; Sreenu, V.B.; Fredericks, A.C.; Pondeville, E.; Donald, C.L.; Sterk, J.; Kean, J.; Vazeille, M.; et al. Aedes aegypti piwi4 is a noncanonical piwi protein involved in antiviral responses. mSphere 2017, 2. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, I.; Jansen, S.; Fall, G.; Lorenzen, S.; Rudolf, M.; Huber, K.; Heitmann, A.; Schicht, S.; Ndiaye, E.H.; Watson, M.; et al. RNA interference restricts rift valley fever virus in multiple insect systems. mSphere 2017, 2. [Google Scholar] [CrossRef]
- Varjak, M.; Dietrich, I.; Sreenu, V.B.; Till, B.E.; Merits, A.; Kohl, A.; Schnettler, E. Spindle-e acts antivirally against alphaviruses in mosquito cells. Viruses 2018, 10, 88. [Google Scholar] [CrossRef]
- Varjak, M.; Leggewie, M.; Schnettler, E. The antiviral piRNA response in mosquitoes? J. Gen. Virol. 2018. [Google Scholar] [CrossRef]
- Miesen, P.; Joosten, J.; van Rij, R.P. Piwis go viral: Arbovirus-derived piRNAs in vector mosquitoes. PLoS Pathog. 2016, 12, e1006017. [Google Scholar] [CrossRef] [PubMed]
- Schneider, I. Cell lines derived from late embryonic stages of drosophila melanogaster. J. Embryol. Exp. Morphol. 1972, 27, 353–365. [Google Scholar]
- Rainey, S.M.; Martinez, J.; McFarlane, M.; Juneja, P.; Sarkies, P.; Lulla, A.; Schnettler, E.; Varjak, M.; Merits, A.; Miska, E.A.; et al. Wolbachia blocks viral genome replication early in infection without a transcriptional response by the endosymbiont or host small RNA pathways. PLoS Pathog. 2016, 12, e1005536. [Google Scholar] [CrossRef] [PubMed]
- Schnettler, E.; Hemmes, H.; Goldbach, R.; Prins, M. The NS3 protein of rice hoja blanca virus suppresses RNA silencing in mammalian cells. J. Gen. Virol. 2008, 89, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Ongus, J.R.; Roode, E.C.; Pleij, C.W.; Vlak, J.M.; van Oers, M.M. The 5’ non-translated region of varroa destructor virus 1 (genus iflavirus): Structure prediction and IRES activity in lymantria dispar cells. J. Gen. Virol. 2006, 87, 3397–3407. [Google Scholar] [CrossRef]
- Ou, J.; Wolfe, S.A.; Brodsky, M.H.; Zhu, L.J. Motifstack for the analysis of transcription factor binding site evolution. Nat. Methods 2018, 15, 8–9. [Google Scholar] [CrossRef]
- Antoniewski, C. Computing siRNA and piRNA overlap signatures. Methods Mol. Biol. 2014, 1173, 135–146. [Google Scholar]
- Fragkoudis, R.; Chi, Y.; Siu, R.W.; Barry, G.; Attarzadeh-Yazdi, G.; Merits, A.; Nash, A.A.; Fazakerley, J.K.; Kohl, A. Semliki forest virus strongly reduces mosquito host defence signaling. Insect Mol. Biol. 2008, 17, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Davey, M.W.; Dalgarno, L. Semliki forest virus replication in cultured Aedes albopictus cells: Studies on the establishment of persistence. J. Gen. Virol. 1974, 24, 453–463. [Google Scholar] [CrossRef]
- Schnettler, E.; Ratinier, M.; Watson, M.; Shaw, A.E.; McFarlane, M.; Varela, M.; Elliott, R.M.; Palmarini, M.; Kohl, A. RNA interference targets arbovirus replication in culicoides cells. J. Virol. 2013, 87, 2441–2454. [Google Scholar] [CrossRef]
- Bolling, B.G.; Weaver, S.C.; Tesh, R.B.; Vasilakis, N. Insect-specific virus discovery: Significance for the arbovirus community. Viruses 2015, 7, 4911–4928. [Google Scholar] [CrossRef] [PubMed]
- Schnettler, E.; Sreenu, V.B.; Mottram, T.; McFarlane, M. Wolbachia restricts insect-specific flavivirus infection in Aedes aegypti cells. J. Gen. Virol. 2016, 97, 3024–3029. [Google Scholar] [CrossRef] [PubMed]
- Stollar, V.; Thomas, V.L. An agent in the Aedes aegypti cell line (peleg) which causes fusion of Aedes albopictus cells. Virology 1975, 64, 367–377. [Google Scholar] [CrossRef]
- Crabtree, M.B.; Sang, R.C.; Stollar, V.; Dunster, L.M.; Miller, B.R. Genetic and phenotypic characterization of the newly described insect flavivirus, kamiti river virus. Arch. Virol. 2003, 148, 1095–1118. [Google Scholar] [CrossRef] [PubMed]
- Nouri, S.; Matsumura, E.E.; Kuo, Y.W.; Falk, B.W. Insect-specific viruses: From discovery to potential translational applications. Curr. Opin. Virol. 2018, 33, 33–41. [Google Scholar] [CrossRef]
- Halbach, R.; Junglen, S.; van Rij, R.P. Mosquito-specific and mosquito-borne viruses: Evolution, infection, and host defense. Curr. Opin. Insect Sci. 2017, 22, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.A.; Bielefeldt-Ohmann, H.; McLean, B.J.; O’Brien, C.A.; Colmant, A.M.; Piyasena, T.B.; Harrison, J.J.; Newton, N.D.; Barnard, R.T.; Prow, N.A.; et al. Commensal viruses of mosquitoes: Host restriction, transmission, and interaction with arboviral pathogens. Evol. Bioinform. Online 2016, 12, 35–44. [Google Scholar] [CrossRef]
- Roundy, C.M.; Azar, S.R.; Rossi, S.L.; Weaver, S.C.; Vasilakis, N. Insect-specific viruses: A historical overview and recent developments. Adv. Virus Res. 2017, 98, 119–146. [Google Scholar]
- Bolling, B.G.; Vasilakis, N.; Guzman, H.; Widen, S.G.; Wood, T.G.; Popov, V.L.; Thangamani, S.; Tesh, R.B. Insect-specific viruses detected in laboratory mosquito colonies and their potential implications for experiments evaluating arbovirus vector competence. Am. J. Trop. Med. Hyg. 2015, 92, 422–428. [Google Scholar] [CrossRef]
- Arensburger, P.; Hice, R.H.; Wright, J.A.; Craig, N.L.; Atkinson, P.W. The mosquito Aedes aegypti has a large genome size and high transposable element load but contains a low proportion of transposon-specific pirnas. BMC Genom. 2011, 12, 606. [Google Scholar] [CrossRef]
- Aliyari, R.; Wu, Q.; Li, H.W.; Wang, X.H.; Li, F.; Green, L.D.; Han, C.S.; Li, W.X.; Ding, S.W. Mechanism of induction and suppression of antiviral immunity directed by virus-derived small RNAs in drosophila. Cell Host Microbe 2008, 4, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Chotkowski, H.L.; Ciota, A.T.; Jia, Y.; Puig-Basagoiti, F.; Kramer, L.D.; Shi, P.Y.; Glaser, R.L. West Nile virus infection of drosophila melanogaster induces a protective RNAi response. Virology 2008, 377, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Galiana-Arnoux, D.; Dostert, C.; Schneemann, A.; Hoffmann, J.A.; Imler, J.L. Essential function in vivo for dicer-2 in host defense against RNA viruses in drosophila. Nat. Immunol. 2006, 7, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Saleh, M.C.; Tassetto, M.; van Rij, R.P.; Goic, B.; Gausson, V.; Berry, B.; Jacquier, C.; Antoniewski, C.; Andino, R. Antiviral immunity in drosophila requires systemic RNA interference spread. Nature 2009, 458, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Zambon, R.A.; Vakharia, V.N.; Wu, L.P. RNAi is an antiviral immune response against a dsrna virus in drosophila melanogaster. Cell. Microbiol. 2006, 8, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Marques, J.T.; Wang, J.P.; Wang, X.; de Oliveira, K.P.; Gao, C.; Aguiar, E.R.; Jafari, N.; Carthew, R.W. Functional specialization of the small interfering RNA pathway in response to virus infection. PLoS Pathog. 2013, 9, e1003579. [Google Scholar] [CrossRef]
- van Rij, R.P.; Saleh, M.C.; Berry, B.; Foo, C.; Houk, A.; Antoniewski, C.; Andino, R. The RNA silencing endonuclease argonaute 2 mediates specific antiviral immunity in drosophila melanogaster. Genes Dev. 2006, 20, 2985–2995. [Google Scholar] [CrossRef]
- Han, Y.H.; Luo, Y.J.; Wu, Q.; Jovel, J.; Wang, X.H.; Aliyari, R.; Han, C.; Li, W.X.; Ding, S.W. RNA-based immunity terminates viral infection in adult drosophila in the absence of viral suppression of RNA interference: Characterization of viral small interfering RNA populations in wild-type and mutant flies. J. Virol 2011, 85, 13153–13163. [Google Scholar] [CrossRef]
- Wang, X.H.; Aliyari, R.; Li, W.X.; Li, H.W.; Kim, K.; Carthew, R.; Atkinson, P.; Ding, S.W. RNA interference directs innate immunity against viruses in adult drosophila. Science 2006, 312, 452–454. [Google Scholar] [CrossRef]
- Steffan, W.A.; Neal, L.E. Biology of toxorhynchites. Annu. Rev. Entomol. 1981, 26, 159–181. [Google Scholar] [CrossRef]
- Zhou, X.; Rinker, D.C.; Pitts, R.J.; Rokas, A.; Zwiebel, L.J. Divergent and conserved elements comprise the chemoreceptive repertoire of the nonblood-feeding mosquito toxorhynchites amboinensis. Genome Biol. Evol. 2014, 6, 2883–2896. [Google Scholar] [CrossRef] [PubMed]
- Myles, K.M.; Morazzani, E.M.; Adelman, Z.N. Origins of alphavirus-derived small RNAs in mosquitoes. RNA Biol. 2009, 6, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.H.; Salmela, H.; Obbard, D.J. Duplication and diversification of dipteran argonaute genes, and the evolutionary divergence of piwi and aubergine. Genome Biol. Evol. 2016, 8, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Keene, K.M.; Foy, B.D.; Sanchez-Vargas, I.; Beaty, B.J.; Blair, C.D.; Olson, K.E. RNA interference acts as a natural antiviral response to o’nyong-nyong virus (alphavirus; togaviridae) infection of anopheles gambiae. Proc. Natl. Acad. Sci. USA 2004, 101, 17240–17245. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donald, C.L.; Varjak, M.; Aguiar, E.R.G.R.; Marques, J.T.; Sreenu, V.B.; Schnettler, E.; Kohl, A. Antiviral RNA Interference Activity in Cells of the Predatory Mosquito, Toxorhynchites amboinensis. Viruses 2018, 10, 694. https://doi.org/10.3390/v10120694
Donald CL, Varjak M, Aguiar ERGR, Marques JT, Sreenu VB, Schnettler E, Kohl A. Antiviral RNA Interference Activity in Cells of the Predatory Mosquito, Toxorhynchites amboinensis. Viruses. 2018; 10(12):694. https://doi.org/10.3390/v10120694
Chicago/Turabian StyleDonald, Claire L., Margus Varjak, Eric Roberto Guimarães Rocha Aguiar, João T. Marques, Vattipally B. Sreenu, Esther Schnettler, and Alain Kohl. 2018. "Antiviral RNA Interference Activity in Cells of the Predatory Mosquito, Toxorhynchites amboinensis" Viruses 10, no. 12: 694. https://doi.org/10.3390/v10120694
APA StyleDonald, C. L., Varjak, M., Aguiar, E. R. G. R., Marques, J. T., Sreenu, V. B., Schnettler, E., & Kohl, A. (2018). Antiviral RNA Interference Activity in Cells of the Predatory Mosquito, Toxorhynchites amboinensis. Viruses, 10(12), 694. https://doi.org/10.3390/v10120694





