Resistance to HIV Integrase Inhibitors: About R263K and E157Q Mutations
Abstract
:1. Introduction
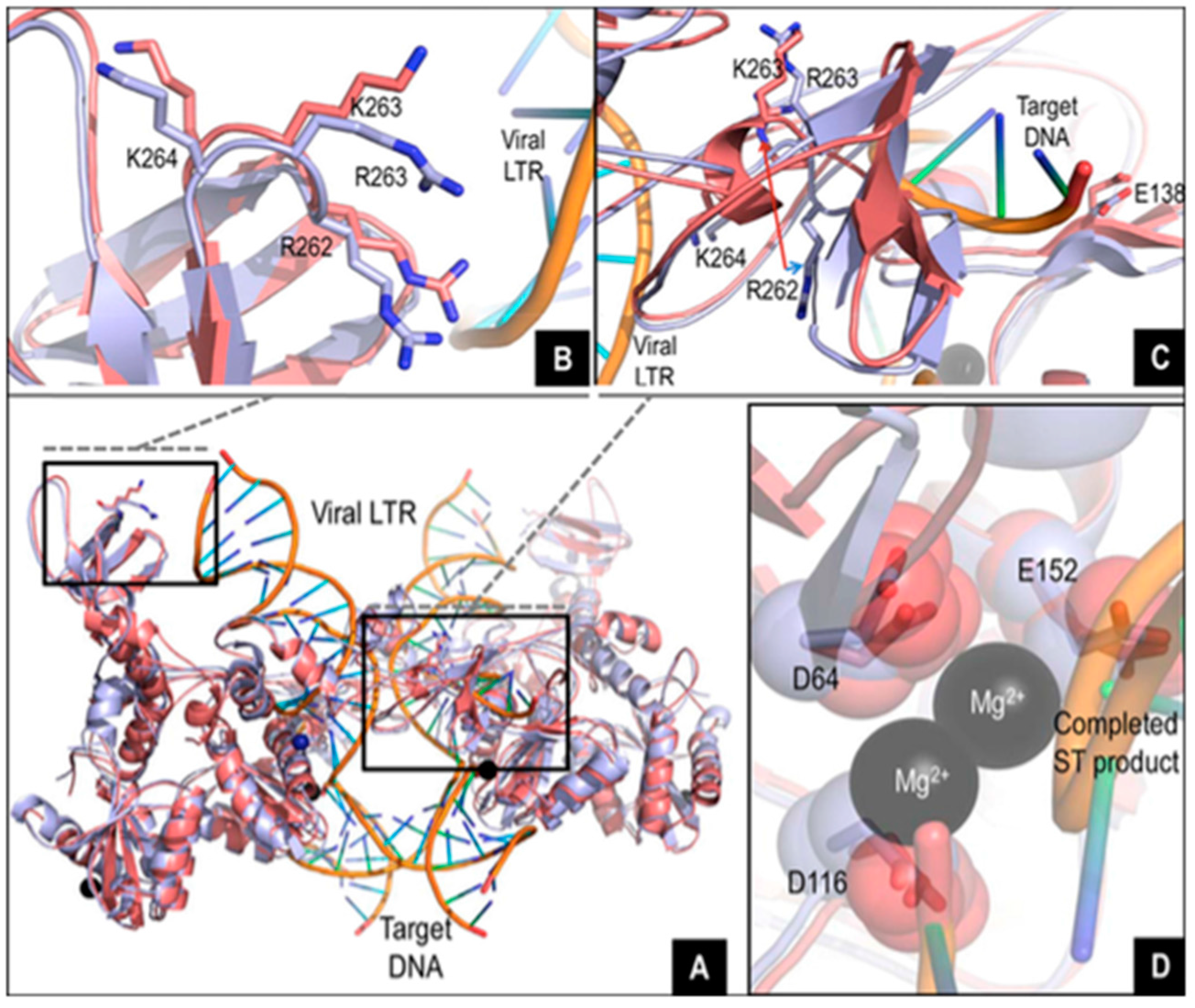
2. R263K Integrase Mutation
2.1. First In Vitro Data on R263K Mutant
2.2. First In Vivo Data on R263K Mutation Selection
2.3. Prevalence of R263K among cART-Naïve Patients
2.4. In Vitro Characterization of R263K Mutants
2.5. R263K and Potential Compensatory Mutations
2.6. R263K and HIV Viral Subtype
2.7. Selection of R263K Mutation and What Behind: In Vitro Data
3. E157Q Integrase Mutation
3.1. Prevalence of E157Q in cART-Naïve Patients
3.2. In Vivo Selection of E157Q Mutation at Virological Failure
3.3. Phenotypic Analysis of E157Q Mutants
3.4. Virological Response to an INI-Based Regimen
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Cahn, P.; Pozniak, A.L.; Mingrone, H.; Shuldyakov, A.; Brites, C.; Andrade-Villanueva, J.F.; Richmond, G.; Buendia, C.B.; Fourie, J.; Ramgopal, M.; et al. Dolutegravir versus raltegravir in antiretroviral-experienced, integrase-inhibitor-naive adults with HIV: Week 48 results from the randomised, double-blind, non-inferiority SAILING study. Lancet 2013, 382, 700–708. [Google Scholar] [CrossRef]
- Quashie, P.K.; Mesplède, T.; Han, Y.-S.; Oliveira, M.; Singhroy, D.N.; Fujiwara, T.; Underwood, M.R.; Wainberg, M.A. Characterization of the R263K mutation in HIV-1 integrase that confers low-level resistance to the second-generation integrase strand transfer inhibitor dolutegravir. J. Virol. 2012, 86, 2696–2705. [Google Scholar] [CrossRef] [PubMed]
- Saladini, F.; Giannini, A.; Boccuto, A.; Tiezzi, D.; Vicenti, I.; Zazzi, M. The HIV-1 integrase E157Q polymorphism per se does not alter susceptibility to raltegravir and dolutegravir in vitro. AIDS 2017, 31, 2307–2309. [Google Scholar] [CrossRef] [PubMed]
- Ghosn, J.; Mazet, A.-A.; Avettand-Fenoel, V.; Peytavin, G.; Wirden, M.; Delfraissy, J.-F.; Chaix, M.-L. Rapid selection and archiving of mutation E157Q in HIV-1 DNA during short-term low-level replication on a raltegravir-containing regimen. J. Antimicrob. Chemother. 2009, 64, 433–434. [Google Scholar] [CrossRef] [PubMed]
- Malet, I.; Delelis, O.; Valantin, M.-A.; Montes, B.; Soulie, C.; Wirden, M.; Tchertanov, L.; Peytavin, G.; Reynes, J.; Mouscadet, J.-F.; et al. Mutations associated with failure of raltegravir treatment affect integrase sensitivity to the inhibitor in vitro. Antimicrob. Agents Chemother. 2008, 52, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Danion, F.; Belissa, E.; Peytavin, G.; Thierry, E.; Lanternier, F.; Scemla, A.; Lortholary, O.; Delelis, O.; Avettand-Fenoel, V.; Duvivier, C. Non-virological response to a dolutegravir-containing regimen in a patient harbouring a E157Q-mutated virus in the integrase region. J. Antimicrob. Chemother. 2015, 70, 1921–1923. [Google Scholar] [CrossRef] [PubMed]
- Hassounah, S.A.; Alikhani, A.; Oliveira, M.; Bharaj, S.; Ibanescu, R.-I.; Osman, N.; Xu, H.-T.; Brenner, B.G.; Mesplède, T.; Wainberg, M.A. Antiviral Activity of Bictegravir and Cabotegravir Against Integrase Inhibitor Resistant SIVmac239 and HIV-1. Antimicrob. Agents Chemother. 2017, 61, e01695-17. [Google Scholar] [CrossRef] [PubMed]
- Malet, I.; Subra, F.; Charpentier, C.; Collin, G.; Descamps, D.; Calvez, V.; Marcelin, A.-G.; Delelis, O. Mutations Located outside the Integrase Gene Can Confer Resistance to HIV-1 Integrase Strand Transfer Inhibitors. mBio 2017, 8, e00922-17. [Google Scholar] [CrossRef] [PubMed]
- Underwood, M.; Dudas, K.; Horton, J.; Wang, R.; Deanda, F.; Griffith, S.; Dorey, D.; Hightower, K. Analysis and characterization of treatment-emergent resistance in ART-experienced, integrase inhibitor-naive subjects with dolutegravir (DTG) versus raltegravir (RAL) in SAILING (ING111762). In Proceedings of the International Workshop on HIV & Hepatitis Virus Drug Resistance and Curative Strategies, Toronto, ON, Canada, 4–8 June 2013. Abstract 21. [Google Scholar]
- Underwood, M.; Deanda, F.; Dorey, D.; Hightower, K. Resistance Post Week 48 in ART-Experienced, Integrase Inhibitor–Naive Subjects with Dolutegravir (DTG) vs. Raltegravir (RAL) in SAILING (ING111762). In Proceedings of the 13th European HIV & Hepatitis Workshop Barcelona, Barcelona, Spain, 3–5 June 2015. Abstract 6. [Google Scholar]
- Chaix, M.-L.; Assoumou, L.; Frange, P.; Orr, S.; Reigadas, S.; Beby Defaux, A.; Bettinger, D.; Bouvier, M.; Courdavault, L.; Fafi-Kremer, S.; et al. National Sentinel Surveillance of Transmitted Drug Resistance in Primary HIV-1 Infected Patients in France Over 6 Years: 2007–2012. In Proceedings of the International Workshop on HIV & Hepatitis Virus Drug Resistance and Curative Strategies, Toronto, ON, Canada, 4–8 June 2013. Abstract 67. [Google Scholar]
- Jaffré, J.; Armenia, D.; Bellocchi, M.C.; Wirden, M.; Carioti, L.; Lambert, S.; Valantin, M.-A.; Nguyen, T.; Simon, A.; Katlama, C.; et al. Ultradeep sequencing detection of the R263K integrase inhibitor drug resistance mutation. J. Antimicrob. Chemother. 2017, 72, 1537–1539. [Google Scholar] [CrossRef] [PubMed]
- Mesplède, T.; Leng, J.; Pham, H.T.; Liang, J.; Quan, Y.; Han, Y.; Wainberg, M.A. The R263K Dolutegravir Resistance-Associated Substitution Progressively Decreases HIV-1 Integration. mBio 2017, 8, e00157-17. [Google Scholar] [CrossRef]
- Kessl, J.J.; Kutluay, S.B.; Townsend, D.; Rebensburg, S.; Slaughter, A.; Larue, R.C.; Shkriabai, N.; Bakouche, N.; Fuchs, J.R.; Bieniasz, P.D.; et al. HIV-1 Integrase Binds the Viral RNA Genome and Is Essential during Virion Morphogenesis. Cell 2016, 166, 1257–1268. [Google Scholar] [CrossRef] [PubMed]
- Wares, M.; Mesplède, T.; Quashie, P.K.; Osman, N.; Han, Y.; Wainberg, M.A. The M50I polymorphic substitution in association with the R263K mutation in HIV-1 subtype B integrase increases drug resistance but does not restore viral replicative fitness. Retrovirology 2014, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Mesplède, T.; Quashie, P.K.; Osman, N.; Han, Y.; Singhroy, D.N.; Lie, Y.; Petropoulos, C.J.; Huang, W.; Wainberg, M.A. Viral fitness cost prevents HIV-1 from evading dolutegravir drug pressure. Retrovirology 2013, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Mesplède, T.; Oliveira, M.; Anstett, K.; Wainberg, M.A. The Combination of the R263K and T66I Resistance Substitutions in HIV-1 Integrase Is Incompatible with High-Level Viral Replication and the Development of High-Level Drug Resistance. J. Virol. 2015, 89, 11269–11274. [Google Scholar] [CrossRef] [PubMed]
- Anstett, K.; Cutillas, V.; Fusco, R.; Mesplède, T.; Wainberg, M.A. Polymorphic substitution E157Q in HIV-1 integrase increases R263K-mediated dolutegravir resistance and decreases DNA binding activity. J. Antimicrob. Chemother. 2016, 71, 2083–2088. [Google Scholar] [CrossRef] [PubMed]
- Mesplède, T.; Quashie, P.K.; Hassounah, S.; Osman, N.; Han, Y.; Liang, J.; Singhroy, D.N.; Wainberg, M.A. The R263K substitution in HIV-1 subtype C is more deleterious for integrase enzymatic function and viral replication than in subtype B. AIDS 2015, 29, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- Anstett, K.; Mesplede, T.; Oliveira, M.; Cutillas, V.; Wainberg, M.A. Dolutegravir resistance mutation R263K cannot coexist in combination with many classical integrase inhibitor resistance substitutions. J. Virol. 2015, 89, 4681–4684. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Mesplède, T.; Quashie, P.K.; Moïsi, D.; Wainberg, M.A. Resistance mutations against dolutegravir in HIV integrase impair the emergence of resistance against reverse transcriptase inhibitors. AIDS 2014, 28, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Anstett, K.; Fusco, R.; Cutillas, V.; Mesplède, T.; Wainberg, M.A. Dolutegravir-Selected HIV-1 Containing the N155H and R263K Resistance Substitutions Does Not Acquire Additional Compensatory Mutations under Drug Pressure That Lead to Higher-Level Resistance and Increased Replicative Capacity. J. Virol. 2015, 89, 10482–10488. [Google Scholar] [CrossRef] [PubMed]
- Singhroy, D.N.; Wainberg, M.A.; Mesplède, T. Combination of the R263K and M184I/V resistance substitutions against dolutegravir and lamivudine decreases HIV replicative capacity. Antimicrob. Agents Chemother. 2015, 59, 2882–2885. [Google Scholar] [CrossRef] [PubMed]
- Assoumou, L.; Bocket, L.; Pallier, C.; Grude, M.; Ait-Namane, R.; Izopet, J.; Charpentier, C.; Visseaux, B.; Wirden, M.; Trabaud, M.-A.; et al. Stable prevalence of transmitted drug resistance mutations and increase circulation of non-B subtype in antiretroviral-naive chronically HIV-infected patients in 2015/2016 in France: The ANRS Odyssee study. In Proceedings of the 9th IAS Conference on HIV Science, Paris, France, 23–26 July 2017. Abstract TUPEC0851. [Google Scholar]
- Charpentier, C.; Malet, I.; André-Garnier, E.; Storto, A.; Bocket, L.; Amiel, C.; Morand-Joubert, L.; Tumiotto, C.; Nguyen, T.; Maillard, A.; et al. Phenotypic analysis of HIV-1 E157Q integrase polymorphism and impact on virological outcome in patients initiating integrase inhibitor-based regimen. J. Antimicrob. Chemother. 2017, in press. [Google Scholar] [CrossRef]
- Shimura, K.; Kodama, E.; Sakagami, Y.; Matsuzaki, Y.; Watanabe, W.; Yamataka, K.; Watanabe, Y.; Ohata, Y.; Doi, S.; Sato, M.; et al. Broad antiretroviral activity and resistance profile of the novel human immunodeficiency virus integrase inhibitor elvitegravir (JTK-303/GS-9137). J. Virol. 2008, 82, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Ambrosioni, J.; Nicolás, D.; Manzardo, C.; Agüero, F.; Blanco, J.L.; Mosquera, M.M.; Peñafiel, J.; Gatell, J.M.; Marcos, M.A.; Miró, J.M. Integrase strand-transfer inhibitor polymorphic and accessory resistance substitutions in patients with acute/recent HIV infection. J. Antimicrob. Chemother. 2017, 72, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Pavkovich, D.; Richmond, D.; Veltman, F. Reviewing Clinical Outcomes of Patients with the E157Q Mutation in Detroit. In Proceedings of the IDWeek, San Diego, CA, USA, 4–8 October 2017. Abstract 1374. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Charpentier, C.; Descamps, D. Resistance to HIV Integrase Inhibitors: About R263K and E157Q Mutations. Viruses 2018, 10, 41. https://doi.org/10.3390/v10010041
Charpentier C, Descamps D. Resistance to HIV Integrase Inhibitors: About R263K and E157Q Mutations. Viruses. 2018; 10(1):41. https://doi.org/10.3390/v10010041
Chicago/Turabian StyleCharpentier, Charlotte, and Diane Descamps. 2018. "Resistance to HIV Integrase Inhibitors: About R263K and E157Q Mutations" Viruses 10, no. 1: 41. https://doi.org/10.3390/v10010041
APA StyleCharpentier, C., & Descamps, D. (2018). Resistance to HIV Integrase Inhibitors: About R263K and E157Q Mutations. Viruses, 10(1), 41. https://doi.org/10.3390/v10010041



