HBV and HCV Therapy
Abstract
1. Treatment of Chronic Hepatitis B
1.1. Treatment Indications
1.2. End-points of Therapy
1.3. Therapeutic Strategies
1.3.1. HBeAg-positive Chronic Hepatitis B
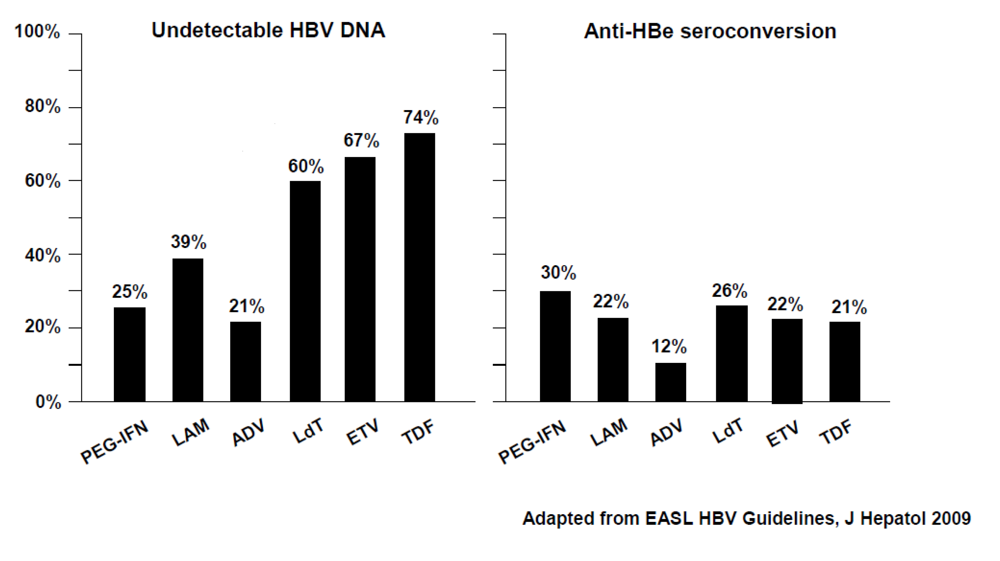
1.3.2. HBeAg-negative Chronic Hepatitis B
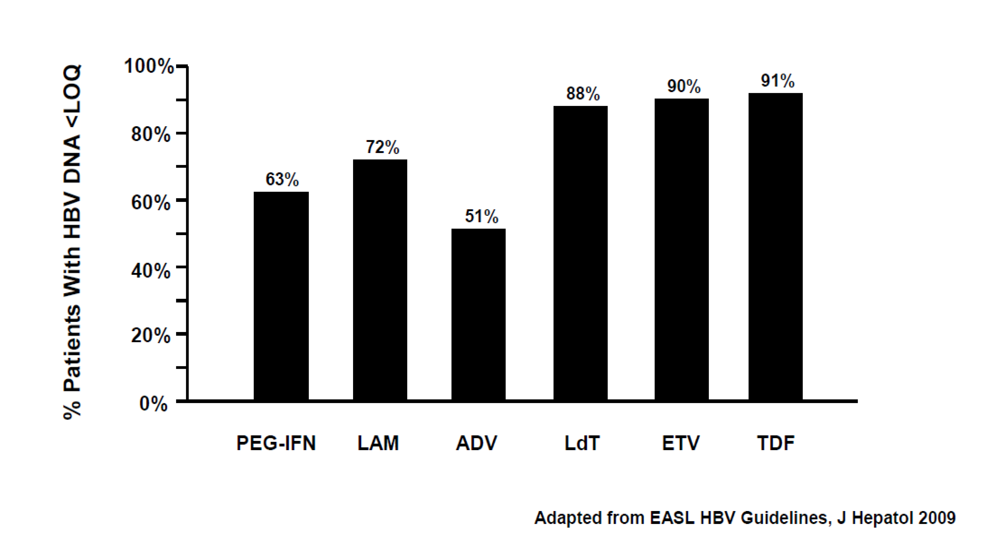
1.4. Management of Treatment Failure
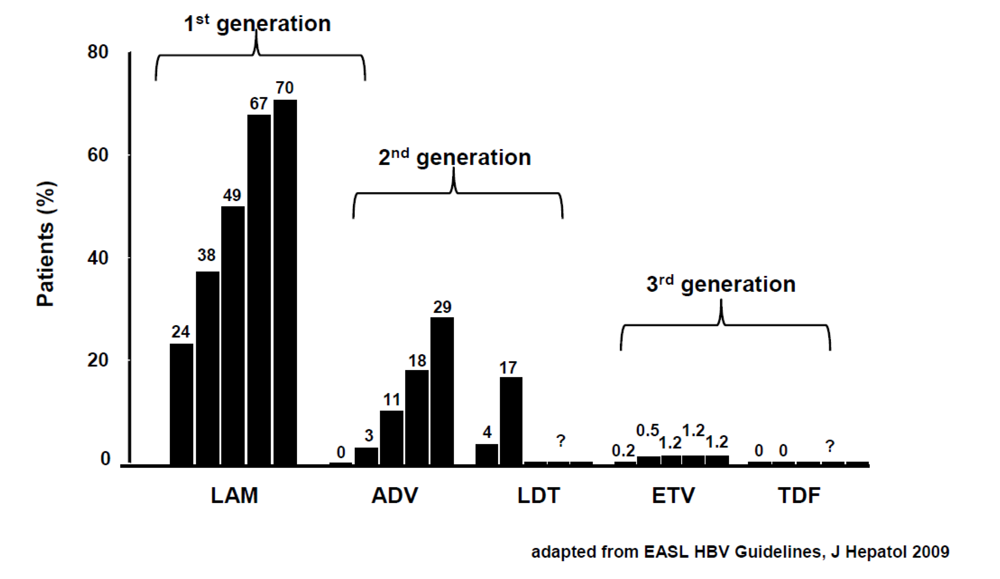
1.5. Prevention of Resistance
2. Treatment of Chronic Hepatitis C
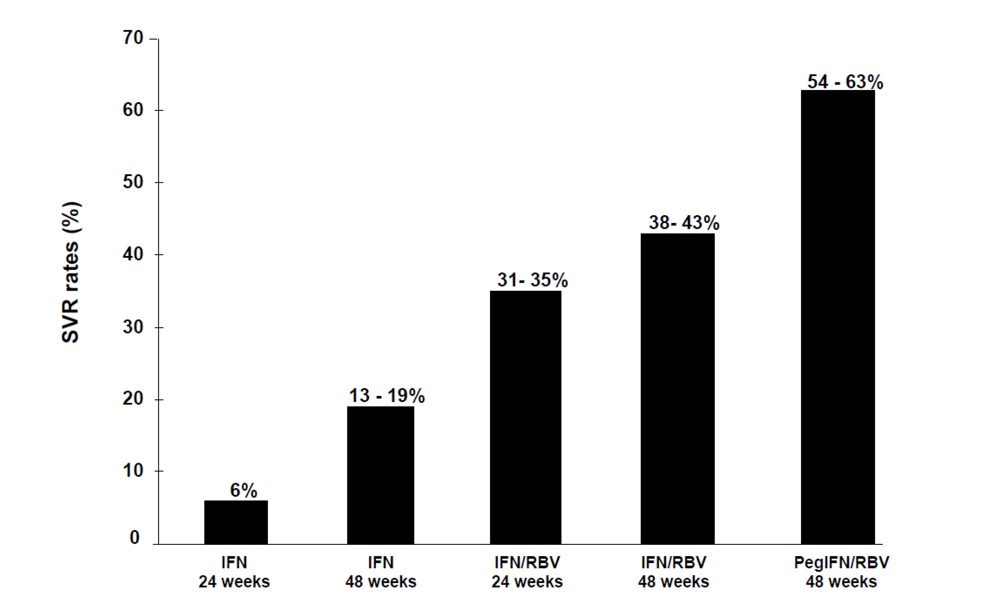
2.1. From registration trials to optimization studies
| Genotype | Peg-IFN dose | Daily Ribavirin Dose | Duration | Stopping Rule |
|---|---|---|---|---|
| HCV-1/4 | PegIFNα2a 180 μg/week | 1000 mg if < 75 kg; 1200 mg if ≥ 75 kg | 48 weeks | 12° or 24° week if NR* |
| PegIFNα2b 1.5 μg/kg/week | 800 mg if < 65 kg; 1000 mg if ≥ 65 kg and ≤ 85 kg | 48 weeks | 12° or 24° week if NR* | |
| 1200 mg if > 85 kg and ≤ 105 kg ; 1400 mg if > 105 kg | ||||
| HCV-2/3 | PegIFNα2a 180 μg/week | 800 mg | 24 weeks | - |
| PegIFNα2b 1.5 μg/kg/week | 800 mg if < 65 kg; 1000 mg if ≥ 65 kg and ≤ 85 kg | 24 weeks | - | |
| 1200 mg if > 85 kg and ≤ 105 kg; 1400 mg if > 105 kg |
2.3. Retreatment of Persons Who Failed to Respond to a Previous Treatment
3. Conclusions
References
- Lok, A.S.; McMahon, B.J. Chronic hepatitis B. Hepatology 2007, 45, 507–539. [Google Scholar] [CrossRef] [PubMed]
- Liaw, Y.F.; Leung, N.; Guan, R.; Lau, G.K.; Merican, I.; McCaughan, G.; Gane, E.; Kao, J.H.; Omata, M. Asian-Pacific consensus update working party on chronic hepatitis B. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2005 update. Liver Int. 2005, 25, 472–489. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of chronic hepatitis B. J. Hepatol. 2009, 50, 227–242. [CrossRef] [PubMed]
- Fattovich, G. Natural history and prognosis of hepatitis B. Semin. Liver Dis. 2003, 23, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Fattovich, G.; Bortolotti, F.; Donato, F. Natural history of chronic hepatitis B: special emphasis on disease progression and prognostic factors. J. Hepatol. 2008, 48, 335–352. [Google Scholar] [CrossRef] [PubMed]
- McMahon, B.J. The natural history of chronic hepatitis B virus infection. Semin. Liver Dis. 2004, 24, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.S; Chien, R.N.; Yeh, C.T.; Sheen, I.S.; Chiou, H.Y.; Chu, C.M.; Liaw, Y.F. Long-term outcome after spontaneous HBeAg seroconversion in patients with chronic hepatitis B. Hepatology 2002, 35, 1522–1527. [Google Scholar] [CrossRef] [PubMed]
- van Zonneveld M.; Honkoop P.; Hansen B.E.; Niesters H.G.; Darwish Murad S.; de Man R.A.; Schalm S.W.; Janssen H.L. Long-term follow-up of alpha-interferon treatment of patients with chronic hepatitis B. Hepatology 2004, 39, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Niederau, C.; Heintges, T.; Lange, S.; Goldmann, G.; Niederau, C.M.; Mohr, L.; Häussinger, D. Long-term follow-up of HBeAg-positive patients treated with interferon alfa for chronic hepatitis B. N. Engl. J. Med. 1996, 334, 1422–1427. [Google Scholar] [CrossRef] [PubMed]
- Papatheodoridis, G.V.; Manesis, E.; Hadziyannis, S.J. The long-term outcome of interferon-alpha treated and untreated patients with HBeAg-negative chronic hepatitis B. J. Hepatol. 2001, 34, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Brunetto, M.R.; Oliveri, F.; Coco, B.; Leandro, G.; Colombatto, P.; Gorin, J.M.; Bonino, F. Outcome of anti-HBe positive chronic hepatitis B in alpha interferon treated and untreated patients: a long term cohort study. J. Hepatol. 2002, 36, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Lampertico, P.; Del Ninno, E.; Viganò, M.; Romeo, R.; Donato, M.F.; Sablon, E.; Morabito, A.; Colombo M. Long-term suppression of hepatitis B e antigen-negative chronic hepatitis B by 24-month interferon therapy. Hepatology 2003, 37, 756–763. [Google Scholar] [CrossRef] [PubMed]
- Di Marco, V.; Lo Iacono, O.; Cammà, C.; Vaccaro, A.; Giunta, M.; Martorana, G.; Fuschi, P.; Almasio, P.L.; Craxì, A. The long term course of chronic hepatitis B. Hepatology 1999, 30, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Bonino, F.; Brunetto, M.R. Chronic hepatitis B e antigen (HBeAg) negative, anti-HBe positive hepatitis B: an overview. J. Hepatol. 2003, 39, 160–163. [Google Scholar] [CrossRef]
- Hadziyannis, S.J.; Papatheodoridis, G.V. Hepatitis B e antigen-negative chronic hepatitis B: natural history and treatment. Semin. Liver Dis. 2006, 26, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Di Marco, V.; Marzano, A.; Lampertico, P.; Andreone, P.; Santantonio, T.; Almasio, P.L.; Rizzetto, M.; Craxì, A.; Italian Association for the Study of the Liver (AISF) Lamivudine Study Group, Italy. Clinical outcome of HBeAg-negative chronic hepatitis B in relation to virological response to lamivudine. Hepatology 2004, 40, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.K.; Cheung, A.M.; O’Rourke, K.; Naylor, C.D.; Detsky, A.S.; Heathcote, J. Effect of alpha-interferon in patients with hepatitis B eantigen-positive chronic hepatitis B. A meta-analysis. Ann. Intern. Med. 1993, 119, 312–323. [Google Scholar] [PubMed]
- Lau, G.K.; Piratvisuth, T.; Luo, K.X.; Marcellin, P.; Thongsawat, S.; Cooksley, G.; Gane, E.; Fried, M.W.; Chow, W.C.; Paik, S.W.; Chang, W.Y.; Berg, T.; Flisiak, R.; McCloud, P.; Pluck, N.; Peginterferon Alfa-2a HBeAg-Positive Chronic Hepatitis B Study Group. Peginterferon Alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. N. Engl. J. Med. 2005, 352, 2682–2695. [Google Scholar] [CrossRef] [PubMed]
- Janssen, H.L.; van Zonneveld, M.; Senturk, H.; Zeuzem, S.; Akarca, U.S.; Cakaloglu, Y.; Simon, C.; So T.M.; Gerken, G.; de Man, R.A.; Niesters, H.G.; Zondervan, P.; Hansen, B.; Schalm, S.W.; HBV 99-01 Study Group; Rotterdam Foundation for Liver Research. Pegylated interferon alfa-2b alone or in combination with lamivudine for HBeAg-positive chronic hepatitis B: a randomised trial. Lancet 2005, 365, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Buster, E.H.; Flink, H.J.; Cakaloglu, Y.; Simon, K.; Trojan, J.; Tabak, F.; So, T.M.; Feinman, S.V.; Mach, T.; Akarca, U.S.; Schutten, M.; Tielemans, W.; van Vuuren, A.J.; Hansen, B.E.; Janssen, H.L. Sustained HBeAg and HBsAg loss after long-term follow-up of HBeAg-positive patients treated with peginterferon alpha-2b. Gastroenterology 2008, 135, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Fried, M.W.; Piratvisuth, T.; Lau, G.K.K.; Marcellin, P.; Chow, W.C.; Cooksley, G.; Luo, K.X.; Paik, S.W.; Liaw, Y.F.; Button, P.; Popescu, M. HBeAg and hepatitis B virus DNA as outcome predictors during therapy withpeginterferon alfa-2a for HBeAg-positive chronic hepatitis B. Hepatology 2008, 47, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Wai, C.T.; Chu, C.J.; Hussain, M.; Lok, A.S. HBV genotype B is associated with better response to interferon therapy in HBeAg(+) chronic hepatitis than genotype C. Hepatology 2002, 36, 1425–1430. [Google Scholar] [PubMed]
- Flink, H.J.; van Zonneveld, M.; Hansen, B.E.; de Man, R.A.; Schalm, S.W.; Janssen, H.L. Treatment with Peg-interferon alpha-2b forHBeAg-positive chronic hepatitis B: HBsAg loss is associated with HBV genotype. Am. J. Gastroenterol. 2006, 101, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.L.; Chien, R.N.; Leung, N.W.; Chang, T.T.; Guan, R.; Tai, D.I.; Ng, K.Y.; Wu, P.C.; Dent, J.C.; Barber, J.; Stephenson, S.L.; Gray, D.F. A one-year trial of lamivudine for chronic hepatitis B. Asia Hepatitis Lamivudine Study Group. N. Engl. J. Med. 1998, 339, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Dienstag, J.L.; Schiff, E.R.; Wright, T.L.; Perrillo, R.P.; Hann, H.W.; Goodman, Z.; Crowther, L.; Condreay, L.D.; Woessner, M.; Rubin, M.; Brown, N.A. Lamivudine as initialtreatment for chronic hepatitis B in the United States. N. Engl. J. Med. 1999, 341, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Marcellin, P.; Chang, T.T.; Lim, S.G.; Tong, M.J.; Sievert, W.; Shiffman, M.L.; Jeffers, L.; Goodman, Z.; Wulfsohn, M.S.; Xiong, S.; Fry, J.; Brosgart, C.L.; Adefovir Dipivoxil 437 Study Group. Adefovir dipivoxil for the treatment of hepatitis B e antigen-positive chronic hepatitis B. N. Engl. J. Med. 2003, 348, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.T.; Gish, R.G.; de Man, R.; Gadano, A.; Sollano, J.; Chao, Y.C.; Lok, A.S.; Han, K.H.; Goodman, Z.; Zhu, J.; Cross, A.; DeHertogh, D.; Wilber, R.; Colonno, R.; Apelian, D.; BEHoLD AI463022 Study Group. A comparison of entecavir and lamivudine for HBeAg-positive chronic hepatitis B. N. Engl. J. Med. 2006, 354, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.L.; Gane, E.; Liaw, Y.F.; Hsu, CW; Wang, Y.; Chen, Y.; Heathcote, E.J.; Rasenack, J.; Bzowej, N.; Naoumov, N.V.; Di Bisceglie, A.M.; Zeuzem, S.; Moon, Y.M.; Goodman, Z.; Chao, G.; Constance, B.F.; Brown, N.A.;Globe Study Group. Telbivudine versus lamivudine in patients with chronic hepatitis B. N. Engl. J. Med. 2007, 357, 2576–2588. [Google Scholar] [CrossRef] [PubMed]
- Marcellin, P.; Heathcote, E.J.; Buti, M.; Gane, E.; de Man, R.A.; Krastev, Z.; Germanidis, G.; Lee, S.S.; Flisiak, R.; Kaita, K.; Manns, M.; Kotzev, I.; Tchernev, K.; Buggisch, P.; Weilert, F.; Kurdas, O.O.; Shiffman, M.L.; Trinh, H.; Washington, M.K.; Sorbel, J.; Anderson, J.; Snow-Lampart, A.; Mondou, E.; Quinn, J.; Rousseau, F. Tenofovir disoproxil fumarate versus adefovir dipivoxil for chronic hepatitis B. N. Engl. J. Med. 2008, 359, 2442–2455. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.T.; Lai, C.L.; Chien, R.N.; Guan, R.; Lim, S.G.; Lee, C.M.; Ng, K.Y.; Nicholls, G.J.; Dent, J.C.; Leung, N.W. Four years oflamivudine treatment in Chinese patients with chronichepatitis B. J. Gastroenterol. Hepatol. 2004, 19, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Leung, N.W.; Lai, C.L.; Chang, T.T.; Guan, R.; Lee, C.M.; Ng, K.Y.; Lim, S.G.; Wu, P.C.; Dent, J.C.; Edmundson, S.; Condreay, L.D.; Chien, R.N.; Asia Hepatitis Lamivudine Study Group. Extended lamivudine treatment in patients with chronic hepatitis B enhances hepatitis B e antigen seroconversion rates: results after 3 years of therapy. Hepatology 2001, 33, 1527–1532. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, J.; Rodès, B.; Zoulim, F.; Bartholomeusz, A.; Soriano, V. Mutations affecting the replication capacity of the hepatitis B virus. J. Viral. Hep. 2006, 13, 427–434. [Google Scholar] [CrossRef]
- Doo, E.; Liang, J.T. Molecular anatomy and pathophysiologic implications of drug resistance in hepatitis B virus infection. Gastroenterology 2001, 120, 1000–1008. [Google Scholar] [CrossRef] [PubMed]
- Brunelle,M.N.; Jacquard,A.C.; Pichoud,C.; Durantel, D.; Carrouée-Durantel, S.; Villeneuve, J.P.; Trépo, C.; Zoulim, F. Susceptibility to antivirals of a human HBV strain with mutations conferring resistance to both lamivudine and adefovir . Hepatology 2005, 41, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Zoulim, F. Mechanism of viral persistence and resistance to nucleoside and nucleotide analogs in chronic hepatitis B virus infection. Antiviral Res. 2004, 64, 1–15. [Google Scholar] [PubMed]
- Locarnini, S.; Hatzakis, A.; Heathcote, J.; Keeffe, E.B.; Liang, T.J.; Mutimer, D.; Pawlotsky, J.M.; Zoulim, F. Management of antiviral resistance in patients with chronic hepatitis B. Antiviral Ther. 2004, 9, 679–693. [Google Scholar]
- Keeffe, E.; Dieterich, D.T.; Han, S.H.B.; Jacobson, I.M.; Martin, P.; Schiff, E.R.; Tobias, H.; Wright, T.L. A treatment algorithm for the management of chronic hepatitis B virus infection in the United States. Clin. Gastroenterol. Hepatol. 2006, 2, 87–106. [Google Scholar] [CrossRef]
- Yuen, M.F.; Sablon, E.; Hui, C.K.; Yuan, H.J.; Decraemer, H.; Lai, C.L. Factors associated with hepatitis B virus DNA breakthrough in patients receiving prolonged lamivudine therapy. Hepatology 2001, 34, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Yuen, M.F.; Fong, D.Y.; Wong, D.K.; Yuen, J.C.; Fung, J.; Lai, C.L. Hepatitis B virus DNA levels at week 4 of lamivudine treatment predict the 5-year ideal response. Hepatology 2007, 46, 1695–1703. [Google Scholar] [CrossRef] [PubMed]
- Dienstag, J.L.; Cianciara, J.; Karayalcin, S.; Kowdley, K.V.; Willems, B.; Plisek, S.; Woessner, M.; Gardner, S.; Schiff, E. Durability of serologic response after lamivudine treatment of chronic hepatitis B. Hepatology 2003, 37, 748–755. [Google Scholar] [CrossRef] [PubMed]
- Song, B.C.; Suh, D.J.; Lee, H.C.; Chung, Y.H.; Lee, Y.S. Hepatitis B e antigen seroconversion after lamivudine therapy is not durable in patients with chronic hepatitis B in Korea. Hepatology 2000, 32, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.M.; Cho, S.W.; Kim, S.W.; Kim, H.J.; Hahm, K.B.; Kim, J.H. Effect of virological response on post-treatment durability of lamivudine-induced HBeAg seroconversion. J. Viral. Hep. 2002, 9, 208–212. [Google Scholar] [CrossRef]
- Liaw, Y.F.; Sung, J.J.; Chow, W.C.; Farrell, G.; Lee, C.Z.; Yuen, H.; Tanwandee, T.; Tao, Q.M.; Shue, K.; Keene, O.N.; Dixon, J.S.; Gray, D.F.; Sabbat, J.; rrhosis Asian Lamivudine Multicentre Study Group. Lamivudine for patients with chronic hepatitis B and advanced liver disease. N. Engl. J. Med. 2004, 351, 1521–1531. [Google Scholar] [CrossRef] [PubMed]
- Marcellin, P.; Chang, T.T.; Lim, S.G.; Sievert, W.; Tong, M.; Arterburn, S.; Borroto-Esoda, K.; Frederick, D.; Rousseau, F. Long-term efficacy and safety of adefovir dipivoxil for the treatment of hepatitis B e antigen-positive chronic hepatitis B. Hepatology 2008, 48, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Gish, R.G.; Lok, A.S.; Chang, T.T.; De Man, R.A.; Gadano, A.; Sollano, J.; Han, H.H.; Chao, Y.C.; Lee, S.-D.; Harris, M.; Yang, J.; Colonno, R.; Brett–Smith, H. Entecavir Therapy for up to 96 Weeks in Patients With HBeAg-Positive Chronic Hepatitis B. Gastroenterology 2007, 133, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Colonno, R.J.; Rose, R.E.; Baldick, C.J.; Levine, S.M.; Klesczewski, K.; Tenney, D.J. High barrier to resistance results in no emergence of entecavir resistance in nucleoside-naïve subjects during the first two years of therapy. J. Hepatol. 2006, 44, 182. [Google Scholar] [CrossRef]
- Tenney, D.J.; Rose, R.E.; Baldick, C.J.; Pokornowski, K.A.; Eggers, B.J.; Fang, J.; Wichroski, M.J.; Xu, D.; Yang, J.; Wilber, R.B.; Colonno, R.J. Long-term monitoring shows hepatitis B virus resistance to entecavir in nucleoside-naïve patients is rare through 5 years of therapy. Hepatology 2009, 49, 1503–1514. [Google Scholar] [CrossRef] [PubMed]
- Heathcote, E.; Gane, E.J.; deMan, R.A.; Chan, S.; Sievert, W.; Mauss, S.; Marcellin, P.; Sorbel, J.; Anderson, J.; Mondou, E.; Quinn, J.; Rousseau, F. Two year tenofovir Disoproxil Fumarate (TDF) Treatment and Adefovir Dipivoxil (ADV) switch data in HBeAg-positive patients with chronic hepatitis B (Study 103), preliminary analysis. Hepatology 2008, 48, 376A. [Google Scholar]
- Zoulim, F.; Perrillo, R. Hepatitis B: reflections on the current approach to antiviral therapy. J. Hepatol. 2008, 48, 2–19. [Google Scholar] [CrossRef]
- Manesis, E.K.; Hadziyannis, S.J. Interferon alpha treatment and retreatment of hepatitis B e antigen-negative chronic hepatitis B. Gastroenterology 2001, 121, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Marcellin , P.; Lau , G.K.; Bonino , F.; Farci, P.; Hadziyannis, S.; Jin, R. ; Lu, Z.; Piravisuth, T.; Germanidis, G.; Yurdaydin, C.; Diago, M.; Gurel, S.; Lai, M.Y.; Button, P.; Pluck, N.; Peginterferon Alfa-2a HBeAg-Negative Chronic Hepatitis B Study Group. PeginterferonPeginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. N. Engl. J. Med. 2004, 351, 1206–1217. [Google Scholar] [CrossRef] [PubMed]
- Bonino, F.; Marcellin, P.; Lau, G.K.; Hadziyannis, S.; Jin, R.; Piratvisuth, T.; Germanidis, G.; Yurdaydin, C.; Diago, M.; Gurel, S.; Lai, M.Y.; Brunetto, M.R.; Farci, P.; Popescu, M.; McCloud, P.; Peginterferon Alfa-2a HBeAg-Negative Chronic Hepatitis B Study Group. Predicting response to peginterferon alpha-2a, lamivudine and the two combined for HBeAg-negative chronic hepatitis B. Gut 2007, 56, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Marcellin, P.; Bonino, F.; Lau, G.K.; Farci, P.; Yurdaydin, C.; Piratvisuth, T.; Luo, K.; Gurel, S.; Hadziyannis, S.; Wang, Y.; Popescu, M. Virological and biochemical response in patients with HBeAg-negative CHB treated with peginterferon alfa-2a (40KD) + lamivudine: 3-year follow-up results. J. Hepatol. 2007, 46, 25–26. [Google Scholar] [CrossRef]
- Hadziyannis, S.J.; Papatheodoridis, G.V.; Dimou, E.; Laras, A.; Papaioannou, C. Efficacy of long-term lamivudine monotherapy in patients with hepatitis B e antigen–negative chronic hepatitis B. Hepatology 2000, 32, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Tassopoulos, N.C.; Volpes, R.; Pastore, G.; Heathcote, J.; Buti, M.; Goldin, R.D.; Hawley, S.; Barber, J.; Condreay, L.; Gray, D.F. Post lamivudine treatment follow up of patients with HBeAg negative chronic hepatitis B. J. Hepatol. 1999, 30, 117. [Google Scholar]
- Santantonio, T.; Mazzola, M.; Iacovazzi, T.; Miglietta, A.; Guastadisegni, A.; Pastore, G. Long-term follow-up of patients with anti-HBe/HBV DNA-positive chronic hepatitis B treated for 12 months with lamivudine. J. Hepatol. 2000, 32, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Hadziyannis, S.J.; Tassopoulos, N.C.; Heathcote, E.J.; Chang, T.T.; Kitis, G.; Rizzetto, M.; Marcellin, P.; Lim, S.G.; Goodman, Z.; Wulfsohn, M.S.; Xiong, S.; Fry, J.; Brosgart, C.L.; Adefovir Dipivoxil 438 Study Group. Adefovir dipivoxil for the treatment of hepatitis B antigen-negative chronic hepatitis B. N. Engl. J. Med. 2003, 348, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Hadziyannis, S.J.; Tassopoulos, N.C.; Heathcote, E.J.; Chang, T.T.; Kitis, G.; Rizzetto, M.; Marcellin, P.; Lim, S.G.; Goodman, Z.; Ma, J.; Brosgart, C.L.; Borroto-Esoda, K.; Arterburn, S.; Chuck, S.L.; Adefovir Dipivoxil 438 Study Group. Long-term therapy with adefovir dipivoxil for HBeAg-negative chronic hepatitis B for up to 5 years. Gastroenterology 2006, 131, 1743–1751. [Google Scholar] [CrossRef] [PubMed]
- Hadziyannis, S.; Sevastianos, V.; Rapti, I.N.; Tassopoulos, N. Sustained biochemical and virological remission after discontinuation of 4 to 5 years of adefovir dipivoxil (ADV) treatment in HBeAg negative chronic hepatitis B. Hepatology 2006, 44, 231A. [Google Scholar]
- Lai, C.L.; Shouval, D.; Lok, A.S.; Chang, T.T.; Cheinquer, H.; Goodman, Z.; DeHertogh, D.; Wilber, R.; Zink, R.C.; Cross, A.; Colonno, R.; Fernandes, L.; BEHoLD AI463027 Study Group. Entecavir versus lamivudine for patients with HBeAg-negative chronic hepatitis B. N. Engl. J.Med. 2006, 354, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Shouval, D.; Akarca, U.S.; Hatzis, G.; Kitis, G.; Lai, C.L.; Cheinquer, H.; Chang, T.T.; Zinc, R.; Zhu, J.; Brett-Smith, H. Continued virologic and biochemical improvement through 96 weeks of entecavir treatment in HBeAg(-) chronic hepatitis B patients (Study ETV-027). J. Hepatol. 2006, 44, S21. [Google Scholar] [CrossRef]
- Lai, C.L.; Gane, E.; Hsu, C.W.; Thongsawat, S.; Wang, Y.; Chen, Y.; Heathcote, E.J.; Rasenack, J.; Bzowej, N.; Naoumov, N.; Zeuzem, S.; Di Bisceglie, A.; Chao, G.C.; Fielman, B.A.; Brown, N.A.; Study Group Globe. Two-year results from the Globe trial in patients with hepatitis B: greater clinical and antiviral efficacy for Telbivudine (LdT) vs. Lamivudine. Hepatology 2006, 44, 222A. [Google Scholar]
- Di Bisceglie, A.; Lai, C.L.; Gane, E.; Chen, Y.; Thongsawat, S.; Wang, Y.; Heathcote, E.J.; Zeuzem, S.; Rasenack, J.; Bzowej, N.; Han, S.H.; Naoumov, N.; Hwang, S.G.; Lim, S.G.; Chao, G.C.; Fielman, B.A.; Brown, N.A.; Study Group Globe The GLOBE16. 1St Telbivudine Globe trial: maximal early HBV suppression is predictive of optimal two-year efficacy in nucleoside-treated hepatitis B patients. Hepatology 2006, 44, 230A. [Google Scholar]
- Marcellin, P.; Buti, M.; Krastev, M.; Gurel, S.; Balabanska, R.I.; Dusheiko, G.; Myers, R.P.; Heathcote, E.J.; Sorbel, J.; Anderson, E.; Mondou, E.; Rousseau, F. Two Year Tenofovir Disoproxil Fumarate (TDF) Treatment and Adefovir Dipivoxil (ADV) Switch Data in HBeAg-Negative Patients with Chronic Hepatitis B (Study 102), Preliminary Analysis. Hepatology 2008, 48, 370A. [Google Scholar] [CrossRef]
- Zoulim, F. Combination of nucleoside analogues in the treatment of chronic hepatitis B infection: lesson from experimental models. J. Antimic. Chemoter. 2005, 55, 608–611. [Google Scholar] [CrossRef]
- Westland, C.E.; Yang, H.; Delaney, W.E.; Lama, N.; Gibbs, C.S.; Miller, M.D.; Fry, J.; Brosgart, C.L.; Schiff, E.R.; Xiong, S. Activity of adefovir dipivoxil against all patterns of lamivudine-resistant hepatitis B viruses in patients. J. Viral Hepat. 2005, 12, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Angus, P.; Vaughan, R.; Xiong, S.; Yang, H.; Delaney, W.; Gibbs, C.; Brosgart, C.; Colledge, D.; Edwards, R.; Ayres, A.; Bartholomeusz, A.; Locarnini, S. Resistance to adefovir dipivoxil therapy associated with the selection of a novel mutation in the HBV polymerase. Gastroenterology 2003, 125, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Villeneuve, J.P.; Durantel, D.; Durantel, S.; Westland, C.; Xiong, S.; Brosgart, C.L.; Gibbs, C.S.; Parvaz, P.; Werle, B.; Trépo, C.; Zoulim, F. Selection of a hepatitis B virus strain resistant to adefovir in a liver transplantation patient. J. Hepatol. 2003, 39, 1085–1089. [Google Scholar] [CrossRef] [PubMed]
- Lampertico, P.; Viganò, M.; Manenti, E.; Iavarone, M.; Sablon, E.; Colombo, M. Low resistance to adefovir combined with lamivudine: a 3-year study of 145 lamivudine resistant hepatitis B patients. Gastroenterology 2007, 133, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.H.; Keeffe, E.B. Chronic hepatitis B: early viral suppression and long-term outcomes of therapy with oral nucleos(t)ides. J. Viral Hepat. 2009, 16, 149–55. [Google Scholar] [CrossRef] [PubMed]
- Rapti, I.; Dimou, E.; Mitsoula, P.; Hadziyannis, S.J. Adding-on versus switching-to adefovir therapy in lamivudine-resistant HBeAg-negative chronic hepatitis B. Hepatology 2007, 45, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Lampertico, P.; Marzano, A.; Levrero, M.; Santantonio, T.; Di Marco, V.; Brunetto M.; Andreone P.; Sagnelli, E.; Fagiuoli, S.; Mazzella, G.; Raimondo, G.; Gaeta, G.; Ascione, A. ; on behalf of the Italian Association for the Study of the Liver (AISF). Adefovir and lamivudine combination therapy is superior to adefovir monotherapy for lamivudine-resistant patients with HBeAg-negative chronic hepatitis B. J. Hepatol. 2007, 46, S191. [Google Scholar] [CrossRef]
- Lampertico, P.; Viganò, M.; Manenti, E.; Iavarone, M.; Lunghi, G.; Colombo, M. Adefovir rapidly suppresses hepatitis B in HBeAg-negative patients developing genotypic resistance to lamivudine. Hepatology 2005, 42, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- Benhamou, Y.; Thibault, V.; Vig, P.; Calvez, V.; Marcelin, A.G.; Fievet, M.H.; Currie, G.; Chang, C.G.; Biao, L.; Xiong, S.; Brosgart, C.; Poynard, T. Safety and efficacy of adefovir dipivoxil in patients infected with lamivudine-resistant hepatitis B and HIV-1. J. Hepatol. 2006, 44, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Yatsuji, H.; Suzuki, F.; Sezaki, H.; Akuta, N.; Suzuki, Y.; Kawamura, Y.; Hosaka, T.; Kobayashi, M.; Saitoh, S.; Arase, Y.; Ikeda, K.; Watahiki, S.; Iwasaki, S.; Kobayashi, M.; Kumada, H. Low risk of adefovir resistance in lamivudine-resistant chronic hepatitis B patients treated with adefovir plus lamivudine combination therapy: two-year follow-up. J. Hepatol. 2008, 48, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Lampertico, P.; Viganò, M.; Facchetti, F.; Iavarone, M.; Colombo, M. Four years of Adefovir add-on in 145 lamivudine resistant patients with chronic hepatitis B: low risk of genotypic resistance to ADV and prevention of virologic breakthrough. Hepatology 2009, 48, 712A. [Google Scholar]
- Hadziyannis, S.J.; Costamena, A.; Laras, A.; Hadziyannis, E. Predictors/determinants of sustained virological response (SVR) and of HBsAg loss following long term adefovir dipivoxil (ADV) therapy in HBeAg-negative chronic hepatitis B (CHB). Hepatology 2008, 48, 896A. [Google Scholar]
- Villet, S.; Pichoud, C.; Villeneuve, J.P.; Trépo, C.; Zoulim, F. Selection of a multiple drug-resistant hepatitis B virus strain in a liver-transplanted patient. Gastroenterology 2006, 131, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Keeffe, E.B.; Zeuzem, S.; Koff, R.S.; Dieterich, D.T.; Esteban-Mur, R.; Gane, E.J.; Jacobson, I.M.; Lim, S.G.; Naoumov, N.; Marcellin, P.; Piratvisuth, T.; Zoulim, F. Report of an international workshop: Roadmap for management of patients receiving oral therapy for chronic hepatitis B. Clin. Gastroenterol. Hepatol. 2007, 5, 890–897. [Google Scholar] [CrossRef]
- Armstrong, G.L.; Wasley, A.; Simard, E.P.; McQuillan, G.M.; Kuhnert, W.L.; Alter, M.J. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann. Intern. Med. 2006, 144, 705–714. [Google Scholar] [PubMed]
- Esteban, J.I.; Sauleda, S.; Quer, J. The changing epidemiology of hepatitis C virus infection in Europe. J. Hepatol. 2008, 48, 148–162. [Google Scholar] [CrossRef] [PubMed]
- Bosetti, C.; Levi, F.; Lucchini, F.; Zatonski, W.A.; Negri, E.; La Vecchia, C. Worldwide mortality from cirrhosis: an update to 2002. J. Hepatol. 2007, 46, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Sangiovanni, A.; Prati, G.M.; Fasani, P.; Ronchi, G.; Romeo, R.; Manini, M.; Del Ninno, E.; Morabito, A.; Colombo M. The natural history of compensated cirrhosis due to hepatitis C virus: a 17-year cohort study of 214 patients. Hepatology 2006, 43, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Hoofnagle, J.H.; Mullen, K.D.; Jones, D.B.; Rustgi, V.; Di Bisceglie, A.; Peters, M.; Waggoner, J.G.; Park, Y.; Jones, E.A. Treatment of chronic non-A, non-B hepatitis with recombinant human alpha interferon. A preliminary report. N. Engl. J. Med. 1986, 315, 1575–1578. [Google Scholar] [PubMed]
- Poynard, T.; Marcellin, P.; Lee, S.S.; Niederau, C.; Minuk, G.S.; Ideo, G.; Bain, V.; Heathcote, J.; Zeuzem, S.; Trepo, C.; Albrecht, J. Randomised trial of interferon alpha2b plus ribavirin for 48 weeks or for 24 weeks versus interferon alpha2b plus placebo for 48 weeks for treatment of chronic infection with hepatitis C virus. International Hepatitis Interventional Therapy Group (IHIT). Lancet 1998, 352, 1426–1432. [Google Scholar] [CrossRef] [PubMed]
- McHutchison, J.G.; Gordon, S.C.; Schiff, E.R.; Shiffman, M.L.; Lee, W.M.; Rustgi, V.K.; Goodman, Z.D.; Ling, M.H.; Cort, S.; Albrecht, J.K. Interferon alfa-2b alone or in combination with ribavirin as initial treatment for chronic hepatitis C. Hepatitis Interventional Therapy Group. N. Engl. J. Med. 1998, 339, 1485–1492. [Google Scholar] [CrossRef] [PubMed]
- Feld, J.J.; Hoofnagle, J.H. Mechanism of action of interferon and ribavirin in treatment of hepatitis C. Nature 2005, 436, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.M.; Martin, N.E.; Modi, M. Pegylation: a novel process for modifying pharmacokinetics. Clin. Pharmacokinet. 2001, 40, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Glue, P.; Fang, J.W.; Rouzier-Panis, R.; Raffanel, C.; Sabo, R.; Gupta, S.K.; Salfi, M.; Jacobs, S. Pegylated interferon-alpha-2b : pharmacokinetics, pharmacodynamics, safety and preliminary efficacy data. Clin. Pharmacol. Ther. 2000, 68, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Bailon, P.; Palleroni, A.; Schaffer, C.A.; Spence, C.L.; Fung, W.J.; Porter, J.E.; Ehrlich, G.K.; Pan, W.; Xu, Z.X.; Modi, M.W.; Farid, A.; Berthold, W.; Graves, M. Rational design of a potent, long-lasting form of interferon: a 40 KDa branched polyethylene glycol-conjugated interferon alpha-2a for the treatment of hepatitis C. Bioconjug. Chem. 2001, 12, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Manns, M.P.; McHutchison, J.G.; Gordon, S.C.; Rustgi, V.K.; Shiffman, M.; Reindollar, R.; Goodman, Z.D.; Koury, K.; Ling, M.; Albrecht, J.K. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet 2001, 358, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Fried, M.W.; Shiffman, M.L.; Reddy, K.R.; Smith, C.; Marinos, G.; Goncales, F.L.; Haussinger, D.; Diago, M.; Carosi, G.; Dhumeaux, D.; Craxi, A.; Lin, A.; Hoffman, J.; Yu, J. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N. Engl. J. Med. 2002, 347, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Caliceti, P. Pharmacokinetics of pegylated interferons: what is misleading? Dig. Liver Dis. 2004, 36, 334–339. [Google Scholar] [CrossRef]
- Bruno, R.; Sacchi, P.; Ciappina, V.; Zocchetti, C.; Patruno, S.; Maiocchi, L.; Filice, G. Viral dynamics and pharmacokinetics of peginterferon alpha-2a and peginterferon alpha-2b in naïve patients with chronic hepatitis C: a randomized, controlled study. Antivir. Ther. 2004, 9, 491–497. [Google Scholar] [PubMed]
- Silva, M.; Poo, J.; Wagner, F.; Jackson, M.; Cutler, D.; Grace, M.; Bordens, R.; Cullen, C.; Harvey, J.; Laughlin, M. A randomised trial to compare the pharmacokinetic, pharmacodynamic, and antiviral effects of peginterferon alfa-2b and peginterferon alfa-2a in patients with chronic hepatitis C (COMPARE). J. Hepatol. 2006, 45, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Di Bisceglie, A.M.; Ghalib, R.H.; Hamzeh, F.; Rustgi, V.K. Early virologic response after peginterferon alpha-2a plus ribavirin or peginterferon alpha-2b plus ribavirin treatment in patients with chronic hepatitis C. J. Viral Hep. 2007, 14, 721–729. [Google Scholar]
- Rumi, M.G.; Aghemo, A.; Prati, G.M.; D’Ambrosio, R.; Donato, M.F.; Russo, A.; Cerami, N.; Soffredini, R.; Colombo M. Randomized study comparing peginterferon-alfa2a plus ribavirin and peginterferon-alfa2b plus ribavirin in naïve patients with chronic hepatitis C: final results of the Milan safety tolerability (MIST) study. Hepatology 2008, 48, 404A. [Google Scholar]
- Ascione, A.; De Luca, M.; Tartaglione, M.T.; Lampasi, F.; Galeota Lanza, A.; Picciotto, F.P.; Di Costanzo, G.G.; Leandro, G. Peginterferon alpha-2a plus ribavirin versus Peginterferon alpha-2b plus ribavirin in naïve patients with chronic hepatitis C virus infection: results of a prospective randomised trial. J. Hepatol. 2008, 48, 370. [Google Scholar] [CrossRef]
- Sulkowski, E.; Lawitz, M.L.; Shiffman, M.L.; Muir, A.J.; Galler, G.; McCone, J.; Nyberg, L.; Lee, W.M.; Ghalib, R.; Schiff, E.; Galati, J.; Bacon, B.; Davis, M.; Mukhopadhyay, P.; NOviello, S.; Pedicone, L.; Albrecht, J.; McHutchison, J. Final results of the ideal (individualized dosing efficacy versus flat dosing to assess optimal pegylated interferon therapy) phase IIIB study. J. Hepatol. 2008, 48, 370. [Google Scholar] [CrossRef]
- Jacobson, I.M.; Brown, R.S.; Freilich, B.; Afdhal, N.; Kwo, P.J.; Santoro, J.; Becker, S.; Wakil, A.E.; Pound, D.; Godofsky, E.; Strauss, R.; Bernstein, D.; Flamm, S.; Pauly, M.P.; Mukhopadhyay, P.; Griffel, L.H.; Brass, C.A.; WIN-R Study Group. Peginterferon alfa-2b and weight-based of flat-dose ribavirin in Chronic hepatitis C Patients: a randomized trial. Hepatology 2007, 46, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Zeuzem, S.; Hultcrantz, R.; Bourliere, M.; Goeser, T.; Marcellin, P.; Sanchez-Tapias, J.; Sarrazin, C.; Harvey, J.; Brass, C.; Albrecht, J. Peginterferon alpha-2b plus ribavirin for treatment of chronic hepatitis C in previously untreated patients infected with HCV genotypes 2 or 3. J. Hepatol. 2004, 40, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Davis, G.L.; Wong, J.B.; McHutchison, J.G.; Manns, M.P.; Harvey, J.; Albrecht, J. Early virological response to treatment with peginterferon alfa-2b plus ribavirin in patients with chronic hepatitis C. Hepatology 2003, 38, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Ghany, M.G.; Strader, D.B.; Thomas, D.L.; Seeff, L.B. Diagnosis, management, and treatment of hepatitis C: an update. Hepatology 2009, 49, 1335–1374. [Google Scholar] [CrossRef] [PubMed]
- Ferenci, P.; Fried, M.W.; Shiffman, M.L.; Smith, C.I.; Marinos, G.; Goncales Jr., F.R.; Haussinger, D.; Diago, M.; Carosi, G.; Dhumeaux, D.; Craxì, A.; Chaneac, M.; Reddy, K.R. Predicting sustained virological responses in chronic hepatitis C patients treated with peginterferon alfa-2a (40 KD)/ribavirin. J. Hepatol. 2005, 43, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Dalgard, O.; Bjøro, K.; Hellum, K.B.; Myrvang, B.; Ritland, S.; Skaug, K.; Raknerud, N.; Bell, H. Treatment with pegylated interferon and ribavirin in HCV infection with genotype 2 or 3 for 14 weeks: a pilot study. Hepatology 2004, 40, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Zeuzem, S.; Buti, M.; Ferenci, P.; Sperl, J.; Horsmans, Y.; Cianciara, J.; Ibranyi, E.; Weiland, O.; Noviello, S.; Brass, C.; Albrecht, J. Efficacy of 24 weeks treatment with peginterferon alfa-2b plus ribavirin in patients with chronic hepatitis C infected with genotype 1 and low pretreatment viremia. J. Hepatol. 2006, 44, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Jensen, D.M.; Morgan, T.R.; Marcellin, P.; Pockros, P.J.; Reddy, K.R.; Hadziyannis, S.J.; Ferenci, P.; Ackrill, A.M.; Willems, B. Early identification of HCV genotype 1 patients responding to 24 weeks peginterferon alpha-2a (40 kd)/ribavirin therapy. Hepatology 2006, 43, 954–960. [Google Scholar] [CrossRef] [PubMed]
- Mangia, A.; Santoro, R.; Minerva, N.; Ricci, G.L.; Carretta, V.; Persico, M.; Vinelli, F.; Scotto, G.; Bacca, D.; Annese, M.; Romano, M.; Zechini, F.; Sogari, F.; Spirito, F.; Andriulli, A. Peginterferon alfa-2b and ribavirin for 12 vs. 24 weeks in HCV genotype 2 or 3. N. Engl. J. Med. 2005, 352, 2609–2617. [Google Scholar] [CrossRef] [PubMed]
- von Wagner, M.; Huber, M.; Berg, T.; Hinrichsen, H.; Rasenack, J.; Heintges, T.; Bergk, A.; Bernsmeier, C.; Haussinger, D.; Herrmann, E.; Zeuzem, S. Peginterferon-alpha-2a (40KD) and ribavirin for 16 or 24 weeks in patients with genotype 2 or 3 chronic hepatitis C. Gastroenterology 2005, 129, 522–527. [Google Scholar] [PubMed]
- Shiffman, M.L.; Suter, F.; Bacon, B.R.; Nelson, D.; Harley, H.; Solà, R.; Shafran, S.D.; Barange, K.; Lin, A.; Soman, A.; Zeuzem, S.; ACCELLERATE Investigators. Peginterferon alfa-2a and ribavirin for 16 or 24 weeks in HCV genotype 2 or 3. N. Engl. J. Med. 2007, 357, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Lagging, M.; Langeland, N.; Pedersen, C.; Färkillä, M.; Buhl, M.R.; Mørch, K.; Dhillon, A.P.; Alsiø, A.; Hellstrand, K.; Westin, J.; Norkrans, G.; NORDynamiC Study Group. Randomized comparison of 12 or 24 weeks of peginterferon alpha-2a and ribavirin in chronic hepatitis C virus genotype 2/3 infection. Hepatology 2008, 47, 1837–1845. [Google Scholar] [CrossRef] [PubMed]
- Dalgard, O.; Bjøro, K.; Ring-Larsen, H.; Bjornsson, E.; Holberg-Petersen, M.; Skovlund, E.; Reichard, O.; Myrvang, B.; Sundeløf, B.; Ritland, S.; Hellum, K.; Fridén, A.; Florholmen, J.; Verbaan, H.; North-C Group. Pegylated interferon alfa and ribavirin for 14 versus 24 weeks in patients with hepatitis C virus genotype 2 or 3 and rapid virological response. Hepatology 2008, 47, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Mangia, A.; Minerva, N.; Bacca, D.; Cozzolongo, R.; Agostinacchio, E.; Sogari, F.; Scotto, G.; Vinelli, F.; Ricci, G.L.; Romano, M.; Carretta, V.; Petruzzellis, D.; Andriulli, A. Determinants of relapse after a short (12 weeks) course of antiviral therapy and re-treatment efficacy of a prolonged course in patients with chronic hepatitis C virus genotype 2 or 3 infection. Hepatology 2009, 49, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Berg, T.; von Wagner, M.; Nasser, S.; Sarrazin, C.; Heintges, T.; Gerlac, T.; Buggisch, P.; Goeser, T.; Rasenack, J.; Pape, G.R.; Schmidt, W.E.; Kallinowski, B.; Klinker, H.; Spengler, U.; Martus, P.; Aishuth, U.; Zeuzem, S. Extended treatment duration for hepatitis C virus type 1: comparing 48 versus 72 weeks of peginterferon-alfa-2a plus ribavirin. Gastroenterology 2006, 130, 1086–1097. [Google Scholar] [CrossRef] [PubMed]
- Pearlman, B.L.; Ehleben, C.; Saifee, S. Treatment extension to 72 weeks of peginterferon and ribavirin in hepatitis c genotype 1-infected slow responders. Hepatology 2007, 46, 1688–1694. [Google Scholar] [CrossRef] [PubMed]
- Buti, M.; Lurie, Y.; Zakharova, N.G.; Blokhina, N.P.; Horban, A.; Sarrazin, C.; Balciuniene, L.; Chen, R.; Yu, X.; Faruqi, R.; Esteban, R. Extended treatment duration in chronic hepatitis C genotype 1-infected slow responders: final results of the SUCCESS study. J. Hepatol. 2009, 50, 58. [Google Scholar] [CrossRef]
- McHutchison, J.G.; Bartenschlager, R.; Patel, K.; Pawlotsky, J.M. The face of future hepatitis C antiviral drug development: recent biological and virologic advances and their translation to drug development and clinical practice. J. Hepatol. 2006, 44, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Shiffman, M.L.; Di Bisceglie, A.M.; Lindsay, K.L.; Norishima, C.; Wright, E.C.; Everson, G.T.; Lok, A.S.; Morgan, T.R.; Bonkovsky, H.L.; Lee, W.M.; Dienstag, J.L.; Ghany, M.G.; Goodman, Z.D.; Everhart, J.E. Hepatitis C Antiviral Long-Term Treatment Against Cirrhosis Trial Group. Peginterferon alfa-2a and ribavirin in patients with chronic hepatitis C who have failed prior treatment. Gastroenterology 2004, 126, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Poynard, T.; Colombo, M.; Bruix, J.; Schiff, E.; Terg, R.; Flamm, S.; Moreno-Otero, R.; Carrilho, F.; Schmidt, W.; Berg, T.; McGarrity, T.; Heathcote, E.J.; Goncales, F.; Diago, M.; Craxì, A.; Silva, M.; Bedossa, P.; Mukhopadhyay, P.; Griffel, L.; Burroughs, M.; Brass, C.; Albrecht, J.; Epic Study Group. Peginterferon alfa-2b and ribavirin: effective in patients with hepatitis C who failed interferon alfa/ribavirin therapy. Gastroenterology 2009, 136, 1618–1628. [Google Scholar] [CrossRef] [PubMed]
- Jensen, D.M.; Marcellin, P.; Freilich, B.; Andreone, P.; Di Bisceglie, A.; Brandão-Mello, C.E.; Reddy, K.R.; Craxì, A.; Martin, A.O.; Teuber, G.; Messinger, D.; Thommes, J.A.; Tietz, A. Re-treatment of patients with chronic hepatitis C who do not respond to peginterferon-alpha2b: a randomized trial. Ann. Intern. Med. 2009, 150, 528–540. [Google Scholar] [PubMed]
- Hézode, C.; Forestier, N.; Dusheiko, G.; Ferenci, P.; Pol, S.; Goeser, T.; Bronowicki, J.P.; Bourlière, M.; Gharakhanian, S.; Bengtsson, L.; McNair, L.; George, S.; Kieffer, T.; Kwong, A.; Kauffman, R.S.; Alam, J.; Pawlotsky, J.M.; Zeuzem, S.; PROVE2 Study Team. Telaprevir and peginterferon with or without ribavirin for chronic HCV infection. N. Engl. J. Med. 2009, 360, 1839–1850. [Google Scholar] [CrossRef] [PubMed]
- Kwo, P.; Lawitz, E.; McCone, J.; Schiff, E.; Vierling, J.; Pound, D.; Davis, M.; Galati, J.; Gordon, S.; Ravendhran, N.; Rossaro, L.; Anderson, F.; Jacobson, I.; Rubin, R.; Koury, K.; Brass, C.; Chaudhri, E.; Albrecht, J. HCV-SPRINT 1 final results: SVR 24 from a phase II study of Boceprevir plus PegIntron (PegIFN-alfa2b)/Ribavirin in treatment-naïve subjects with genotype 1 chronic hepatitis C. J. Hepatol. 2009, 50, 4. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Share and Cite
Lampertico, P.; Aghemo, A.; Viganò, M.; Colombo, M. HBV and HCV Therapy. Viruses 2009, 1, 484-509. https://doi.org/10.3390/v1030484
Lampertico P, Aghemo A, Viganò M, Colombo M. HBV and HCV Therapy. Viruses. 2009; 1(3):484-509. https://doi.org/10.3390/v1030484
Chicago/Turabian StyleLampertico, Pietro, Alessio Aghemo, Mauro Viganò, and Massimo Colombo. 2009. "HBV and HCV Therapy" Viruses 1, no. 3: 484-509. https://doi.org/10.3390/v1030484
APA StyleLampertico, P., Aghemo, A., Viganò, M., & Colombo, M. (2009). HBV and HCV Therapy. Viruses, 1(3), 484-509. https://doi.org/10.3390/v1030484




