The Frequency of Cyclonic Wind Storms Shapes Tropical Forest Dynamism and Functional Trait Dispersion
Abstract
1. Introduction
1.1. Cyclonic Wind Disturbances Shape Forest Structure and Dynamics by Influencing Tree Community Demographics and Functional Composition
1.2. Cyclonic Wind Disturbance Frequency and Forest Resistance
1.3. Predictions for Forest Dynamism and Functional Composition with Varying Cyclonic Storm Recurrence
- (1)
- Do forests that have storm recurrence intervals that coincide with the time to reproductive maturity of tropical trees show more variable demographics than forests with more frequent or no cyclonic storms?
- (2)
- Do forests that have storm recurrence intervals that coincide with the time to reproductive maturity of tropical trees show greater changes in functional community composition (i.e., more variation in species life-history strategy per unit area over time) than forests with more frequent or no cyclonic storms?
2. Materials and Methods
2.1. Sites and Tree Census Methods
2.2. Functional Trait Data
2.3. Characterizing the Disturbance Regimes
2.4. Analyses of Forest Demographics
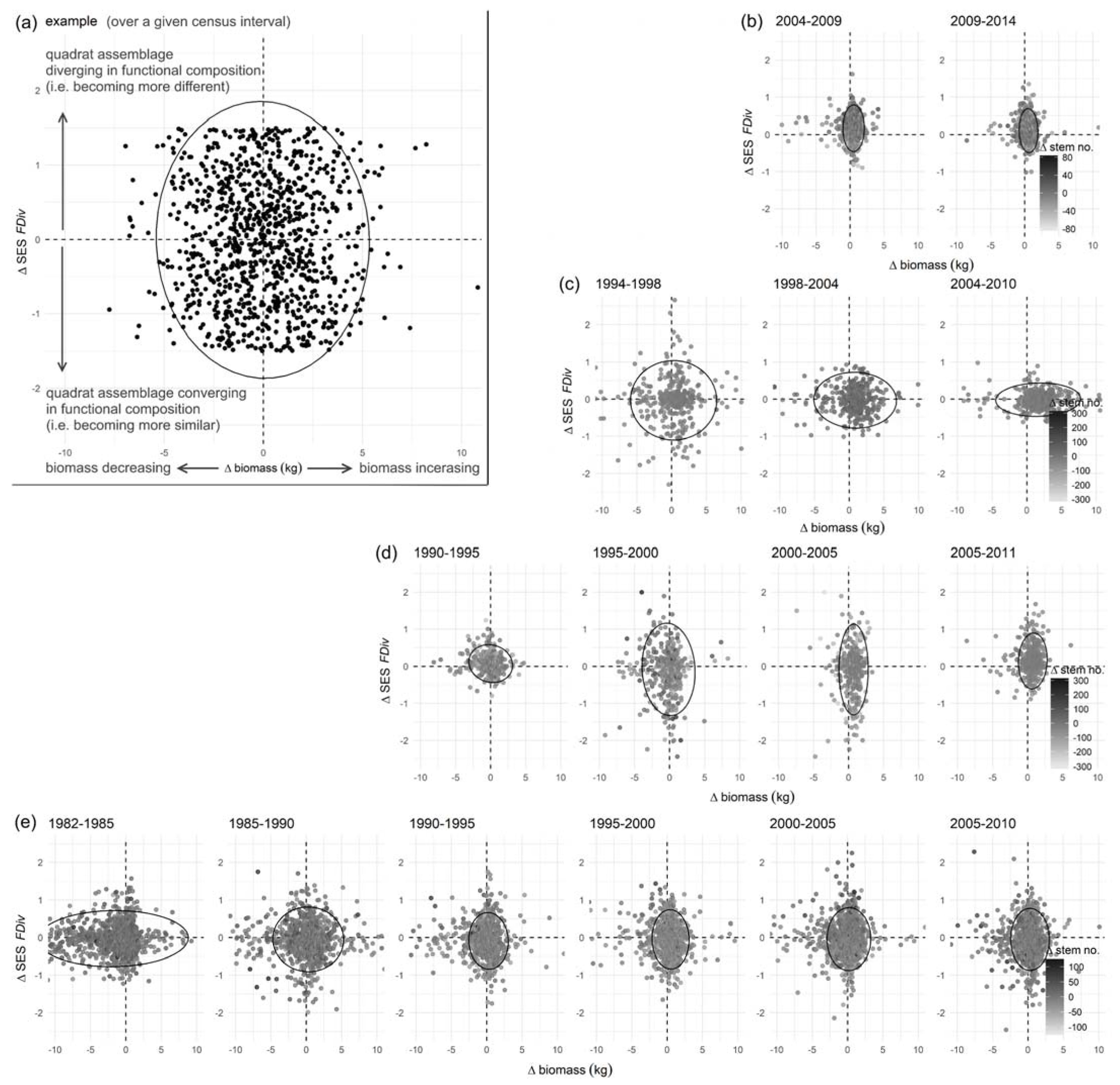
2.5. Linking Stand Demographics to Functional Traits: Changes in Biomass and Functional Divergence
3. Results
3.1. Disturbance Regimes
3.2. Forest Dynamics
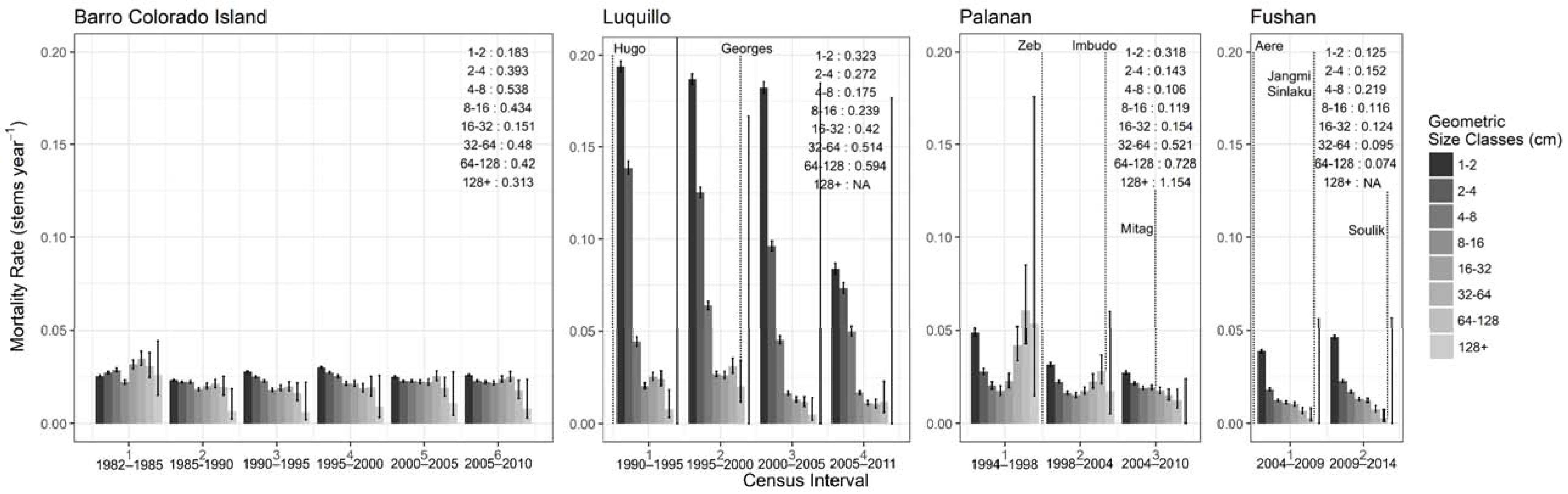
3.2.1. Stem Dynamics
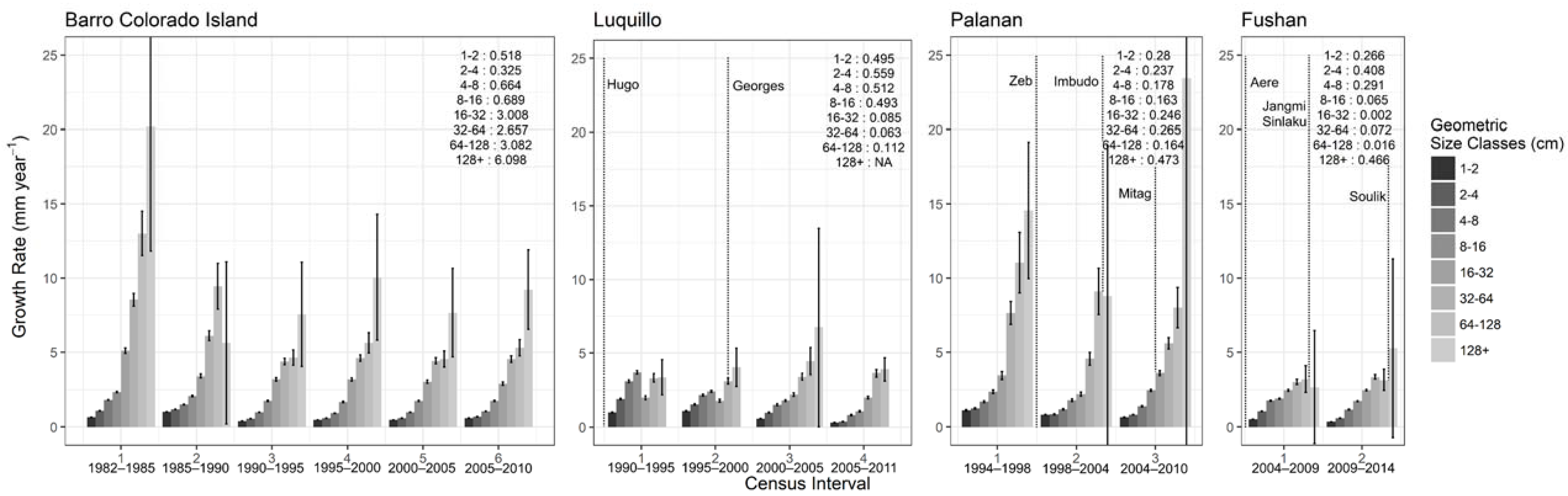
3.2.2. Stem Growth and Biomass Dynamics
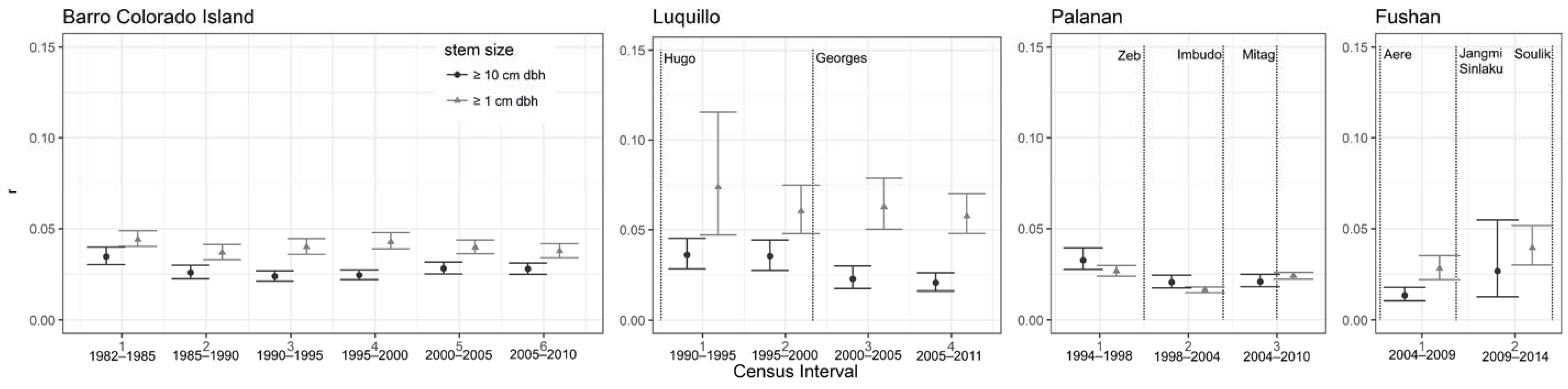
3.2.3. Population Dynamics
3.3. Functional Composition
4. Discussion
4.1. Do Forests That Have Storm Recurrence Intervals That Coincide with the Time to Reproductive Maturity of Tropical Trees Show More Variable Demographics Than Forests with More Frequent or No Cyclonic Storms?
4.2. Do Forests That Have Storm Recurrence Intervals That Coincide with the Time to Reproductive Maturity of Tropical Trees Show Greater Changes in Functional Community Composition (i.e., More Variation in Species Life-History Strategy per Unit Area over Time) Than Forests with More Frequent or No Cyclonic Storms?
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Turner, M.G.; Dale, V.H. Comparing large, infrequent disturbances: What have we learned? Ecosystems 1998, 1, 493–496. [Google Scholar] [CrossRef]
- Lugo, A.E.; Scatena, F.N. Background and catastrophic tree mortality in tropical moist, wet, and rain forests. Biotropica 1996, 28, 585–599. [Google Scholar] [CrossRef]
- Zimmerman, J.K.; Willig, M.R.; Walker, L.R.; Silver, W.L. Introduction: disturbance and Caribbean ecosystems. Biotropica 1996, 28, 414–423. [Google Scholar] [CrossRef]
- Shen, Y.; Santiago, L.S.; Ma, L.; Lin, G.-J.; Lian, J.-Y.; Cao, H.-L.; Ye, W.-H. Forest dynamics of a subtropical monsoon forest in Dinghushan, China: Recruitment, mortality and the pace of community change. J. Trop. Ecol. 2013, 29, 131–145. [Google Scholar] [CrossRef]
- Heartsill Scalley, T.; Scatena, F.N.; Lugo, A.E.; Moya, S.; Estrada Ruiz, C.R. Changes in structure, composition, and nutrients during 15 yr of hurricane-induced succession in a subtropical wet forest in Puerto Rico. Biotropica 2010, 42, 455–463. [Google Scholar] [CrossRef]
- Everham, E.M.; Brokaw, N.V. Forest damage and recovery from catastrophic wind. Bot. Rev. 1996, 62, 113–185. [Google Scholar] [CrossRef]
- Chambers, J.Q.; Fisher, J.I.; Zeng, H.; Chapman, E.L.; Baker, D.B.; Hurtt, G.C. Hurricane Katrina’s carbon footprint on U.S. Gulf Coast forests. Science 2007, 318, 1107. [Google Scholar] [CrossRef] [PubMed]
- Vandermeer, J.H.; Boucher, D.H.; de la Cerda, I.G.; Perfecto, I. Growth and development of the thinning canopy in a post-hurricane tropical rain forest in Nicaragua. For. Ecol. Manag. 2001, 148, 221–242. [Google Scholar] [CrossRef]
- Brokaw, N.; Zimmerman, J.K.; Willig, M.R.; Camilo, G.R.; Covich, A.P.; Crowl, T.A.; Fetcher, N.; Haines, B.L.; Lodge, D.J.; Lugo, A.E.; et al. Response to disturbance. In A Caribbean Forest Tapestry: The Multidimensional Nature of Disturbance and Response; Brokaw, N., Crowl, T.A., Lugo, A.E., McDowell, W.H., Scatena, F.N., Waide, R.B., Willig, M.R., Eds.; Oxford University Press: New York, NY, USA, 2012; pp. 201–271. [Google Scholar]
- Walker, L.R. Timing of post-hurricane tree mortality in Puerto Rico. J. Trop. Ecol. 1995, 11, 315–320. [Google Scholar] [CrossRef]
- Van Bloem, S.J.; Murphy, P.G.; Lugo, A.E. A link between hurricane-induced tree sprouting, high stem density and short canopy in tropical dry forest. Tree Physiol. 2007, 27, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Bellingham, P.; Tanner, E.; Healey, J. Damage and responsiveness of Jamaican montane tree species after disturbance by a hurricane. Ecology 1995, 76, 2562–2580. [Google Scholar] [CrossRef]
- Brokaw, N.V.; Grear, J.S. Forest structure before and after hurricane Hugo at three elevations in the Luquillo mountains, Puerto Rico. Biotropica 1991, 23, 386–392. [Google Scholar] [CrossRef]
- Zimmerman, J.K.; Comita, L.S.; Thompson, J.; Uriarte, M.; Brokaw, N. Patch dynamics and community metastability of a subtropical forest: Compound effects of natural disturbance and human land use. Landsc. Ecol. 2010, 25, 1099–1111. [Google Scholar] [CrossRef]
- Luke, D.; McLaren, K.; Wilson, B. Modeling hurricane exposure in a Caribbean lower montane tropical wet forest: The effects of frequent, intermediate disturbances and topography on forest structural dynamics and composition. Ecosystems 2016, 19, 1178–1195. [Google Scholar] [CrossRef]
- Lugo, A.E. Visible and invisible effects of hurricanes on forest ecosystems: An international review. Austral Ecol. 2008, 33, 368–398. [Google Scholar] [CrossRef]
- Webb, E.L.; Bult, M.; Fa’aumu, S.; Webb, R.C.; Tualaulelei, A.; Carrasco, L.R. Factors affecting tropical tree damage and survival after catastrophic wind disturbance. Biotropica 2014, 46, 32–41. [Google Scholar] [CrossRef]
- Zimmerman, J.K.; Everham, E.M., III; Waide, R.B.; Lodge, D.J.; Taylor, C.M.; Brokaw, N.V. Responses of tree species to hurricane winds in subtropical wet forest in Puerto Rico: Implications for tropical tree life histories. J. Ecol. 1994, 82, 911–922. [Google Scholar] [CrossRef]
- Uriarte, M.; Canham, C.D.; Thompson, J.; Zimmerman, J.K. A neighborhood analysis of tree growth and survival in a hurricane-driven tropical forest. Ecol. Monogr. 2004, 74, 591–614. [Google Scholar] [CrossRef]
- Tanner, E.V.J.; Rodriguez-Sanchez, F.; Healey, J.R.; Holdaway, R.J.; Bellingham, P.J. Long-term hurricane damage effects on tropical forest tree growth and mortality. Ecology 2014, 95, 2974–2983. [Google Scholar] [CrossRef]
- Monoy, C.C.; Tomlinson, K.W.; Iida, Y.; Swenson, N.G.; Slik, J.F. Temporal changes in tree species and trait composition in a cyclone-prone pacific dipterocarp forest. Ecosystems 2016, 19, 1013–1022. [Google Scholar] [CrossRef]
- Tanner, E.V.J.; Kapos, V. Leaf structure of Jamaican upper montane rain-forest trees. Biotropica 1982, 14, 16–24. [Google Scholar] [CrossRef]
- Gunderson, L.H. Ecological resilience—In theory and application. Annu. Rev. Ecol. Syst. 2000, 31, 425–439. [Google Scholar] [CrossRef]
- Scatena, F.N.; Blanco, J.F.; Beard, K.H.; Waide, R.B.; Lugo, A.E.; Brokaw, N.; Silver, W.L.; Haines, B.L.; Zimmerman, J.K. Disturbance regime. In A Caribbean Forest Tapestry: The Multidimensional Nature of Disturbance and Response; Oxford University Press: New York, NY, USA, 2012; pp. 164–200. [Google Scholar]
- Hogan, J.A.; Zimmerman, J.K.; Thompson, J.; Nytch, C.J.; Uriarte, M. The interaction of land-use legacies and hurricane disturbance in subtropical wet forest: Twenty-one years of change. Ecosphere 2016, 7. [Google Scholar] [CrossRef]
- Paine, R.T.; Tegner, M.J.; Johnson, E.A. Compounded perturbations yield ecological surprises. Ecosystems 1998, 1, 535–545. [Google Scholar] [CrossRef]
- Buma, B.; Wessman, C. Disturbance interactions can impact resilience mechanisms of forests. Ecosphere 2011, 2, 1–13. [Google Scholar] [CrossRef]
- Lugo, A.E.; Applefield, M.; Pool, D.J.; McDonald, R.B. The impact of hurricane David on the forests of Dominica. Can. J. For. Res. 1983, 13, 201–211. [Google Scholar] [CrossRef]
- Uriarte, M.; Canham, C.D.; Thompson, J.; Zimmerman, J.K.; Murphy, L.; Sabat, A.M.; Fetcher, N.; Haines, B.L. Natural disturbance and human land use as determinants of tropical forest dynamics: Results from a forest simulator. Ecol. Monogr. 2009, 79, 423–443. [Google Scholar] [CrossRef]
- Tanner, E.; Bellingham, P. Less diverse forest is more resistant to hurricane disturbance: Evidence from montane rain forests in Jamaica. J. Ecol. 2006, 94, 1003–1010. [Google Scholar] [CrossRef]
- Ostertag, R.; Silver, W.L.; Lugo, A.E. Factors affecting mortality and resistance to damage following hurricanes in a rehabilitated subtropical moist forest. Biotropica 2005, 37, 16–24. [Google Scholar] [CrossRef]
- Tanner, E.; Kapos, V.; Healey, J. Hurricane effects on forest ecosystems in the Caribbean. Biotropica 1991, 23, 513–521. [Google Scholar] [CrossRef]
- Shaw, W. Tropical cyclones: Determinants of pattern and structure in New Zealand’s indigenous forests. Pac. Sci. 1983, 37, 405–414. [Google Scholar]
- Canham, C.D.; Loucks, O.L. Catastrophic windthrow in the presettlement forests of Wisconsin. Ecology 1984, 65, 803–809. [Google Scholar] [CrossRef]
- Vandermeer, J.; Granzow De La Cerda, I.; Perfecto, I.; Boucher, D.; Ruiz, J.; Kaufmann, A. Multiple basins of attraction in a tropical forest: Evidence for nonequilibrium community structure. Ecology 2004, 85, 575–579. [Google Scholar] [CrossRef]
- Terborgh, J.; Flores, C.N.; Mueller, P.; Davenport, L. Estimating the ages of successional stand of tropical trees from growth increments. J. Trop. Ecol. 1997, 14, 833–856. [Google Scholar] [CrossRef]
- Condit, R. Tropical Forest Census FDPs: Methods and Results from Barro Colorado Island, Panama and a Comparison with Other FDPs; Springer: Berlin, Germany, 1998. [Google Scholar]
- Anderson-Teixeira, K.J.; Davies, S.J.; Bennett, A.C.; Gonzalez-Akre, E.B.; Muller-Landau, H.C.; Joseph Wright, S.; Abu Salim, K.; Almeyda Zambrano, A.M.; Alonso, A.; Baltzer, J.L. CTFS-ForestGeo: A worldwide network monitoring forests in an era of global change. Glob. Chang. Biol. 2015, 21, 528–549. [Google Scholar] [CrossRef] [PubMed]
- Condit, R.; Lao, S.; Pérez, R.; Dolins, S.B.; Foster, R.; Hubbell, S. Barro Colorado Forest Census Plot Data (Version 2012); Center for Tropical Forest Science Databases: Washington, DC, USA, 2012. [Google Scholar] [CrossRef]
- Zimmerman, J.K.; Thompson, J. Census of Species, Diameter and Location at the Luquillo Forest Dynamics Plot (LFDP); Luquillo LTER: San Juan, Puerto Rico, 2011. [Google Scholar]
- Yap, S.L.; Davies, S.J.; Condit, R. Dynamic response of a Philippine dipterocarp forest to typhoon disturbance. J. Veg. Sci. 2016, 27, 133–143. [Google Scholar] [CrossRef]
- Su, S.-H.; Chang-Yang, C.; Lu, C.; Tsui, C.; Lin, T.; Lin, C.; Chiou, W.; Kuan, L.; Chen, Z.; Hsieh, C. Fushan Subtropical Forest Dynamics Plot: Tree Species Characteristics and Distribution Patterns; Taiwan Forestry Research Institute: Taipei City, Taiwan, 2007. [Google Scholar]
- Soil Survey Staff. Order 1 Soil Survey of the Luquillo Long-Term Ecological Research Grid, Puerto Rico; USDA, Natural Resources Conservation Service Lincoln: Lincoln, NE, USA, 1995.
- Co, L.L.; Lagunzad, D.A.; LaFrankie, J.V.; Bartolome, N.A.; Molina, J.E.; Yap, S.L.; Garcia, H.G.; Bautista, J.P.; Gumpal, E.C.; Araño, R.R.; et al. Palanan forest dynamics plot, Philippines. In Tropical Forest Diversity and Dynamism: Findings from a Large-Scale Plot Network; Losos, E.C., Leigh, E.G., Eds.; University of Chicago Press: Chicago, IL, USA, 2004. [Google Scholar]
- Dietrich, W.; Windsor, D.; Dunne, T. Geology, Climate, and Hydrology of Barro Colorado Island [Tropical Forest, Panama]; Smithsonian Institutional Press: Washington, DC, USA, 1982. [Google Scholar]
- Díaz, S.; Kattge, J.; Cornelissen, J.H.; Wright, I.J.; Lavorel, S.; Dray, S.; Reu, B.; Kleyer, M.; Wirth, C.; Prentice, I.C. The global spectrum of plant form and function. Nature 2016, 529, 167. [Google Scholar] [CrossRef] [PubMed]
- Poorter, L.; Wright, S.J.; Paz, H.; Ackerly, D.D.; Condit, R.; Ibarra-Manríquez, G.; Harms, K.E.; Licona, J.; Martinez-Ramos, M.; Mazer, S. Are functional traits good predictors of demographic rates? Evidence from five neotropical forests. Ecology 2008, 89, 1908–1920. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.J.; Kitajima, K.; Kraft, N.J.; Reich, P.B.; Wright, I.J.; Bunker, D.E.; Condit, R.; Dalling, J.W.; Davies, S.J.; Díaz, S. Functional traits and the growth–mortality trade-off in tropical trees. Ecology 2010, 91, 3664–3674. [Google Scholar] [CrossRef] [PubMed]
- Swenson, N.G.; Stegen, J.C.; Davies, S.J.; Erickson, D.L.; Forero-Montaña, J.; Hurlbert, A.H.; Kress, W.J.; Thompson, J.; Uriarte, M.; Wright, S.J. Temporal turnover in the composition of tropical tree communities: Functional determinism and phylogenetic stochasticity. Ecology 2012, 93, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Lasky, J.R.; Sun, I.; Su, S.H.; Chen, Z.S.; Keitt, T.H. Trait-mediated effects of environmental filtering on tree community dynamics. J. Ecol. 2013, 101, 722–733. [Google Scholar] [CrossRef]
- Iida, Y.; Kohyama, T.S.; Swenson, N.G.; Su, S.H.; Chen, C.T.; Chiang, J.M.; Sun, I. Linking functional traits and demographic rates in a subtropical tree community: The importance of size dependency. J. Ecol. 2014, 102, 641–650. [Google Scholar] [CrossRef]
- Knapp, K.R.; Kruk, M.C.; Levinson, D.H.; Diamond, H.J.; Neumann, C.J. The international best track archive for climate stewardship (IBTrACS) unifying tropical cyclone data. Bull. Am. Meteorol. Soc. 2010, 91, 363–376. [Google Scholar] [CrossRef]
- Moyer, A.C.; Evans, J.L.; Powell, M. Comparison of observed gale radius statistics. Meterol. Atmos. Phys. 2007, 97, 41–55. [Google Scholar] [CrossRef]
- Hsu, S.; Babin, A. Estimating the radius of maximum wind via satellite during Hurricane Lili (2002) over the Gulf of Mexico. Natl. Weather Assoc. Electron. J. 2005, 6, 1–6. [Google Scholar]
- Vandermeer, J.H.; Goldberg, D.E. Population Ecology: First Principles; Princeton University Press: Princeton, NJ, USA, 2013. [Google Scholar]
- Chisholm, R.A.; Condit, R.; Rahman, K.A.; Baker, P.J.; Bunyavejchewin, S.; Chen, Y.Y.; Chuyong, G.; Dattaraja, H.; Davies, S.; Ewango, C.E. Temporal variability of forest communities: Empirical estimates of population change in 4000 tree species. Ecol. Lett. 2014, 17, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Lugo, A.E. Effects and outcomes of caribbean hurricanes in a climate change scenario. Sci. Total Environ. 2000, 262, 243–251. [Google Scholar] [CrossRef]
- Chave, J.; Réjou-Méchain, M.; Búrquez, A.; Chidumayo, E.; Colgan, M.S.; Delitti, W.B.; Duque, A.; Eid, T.; Fearnside, P.M.; Goodman, R.C. Improved allometric models to estimate the aboveground biomass of tropical trees. Glob. Chang. Biol. 2014, 20, 3177–3190. [Google Scholar] [CrossRef] [PubMed]
- Brokaw, N.V. Gap-phase regeneration in a tropical forest. Ecology 1985, 66, 682–687. [Google Scholar] [CrossRef]
- Brokaw, N.V. The definition of treefall gap and its effect on measures of forest dynamics. Biotropica 1982, 14, 158–160. [Google Scholar] [CrossRef]
- Villéger, S.; Mason, N.W.; Mouillot, D. New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 2008, 89, 2290–2301. [Google Scholar] [CrossRef] [PubMed]
- Mason, N.W.; Mouillot, D.; Lee, W.G.; Wilson, J.B. Functional richness, functional evenness and functional divergence: The primary components of functional diversity. Oikos 2005, 111, 112–118. [Google Scholar] [CrossRef]
- Laliberté, E.; Legendre, P. A distance-based framework for measuring functional diversity from multiple traits. Ecology 2010, 91, 299–305. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Swenson, N.G. Functional and Phylogenetic Ecology in R; Springer: Berlin, Germany, 2014. [Google Scholar]
- Waide, R.B.; Willig, M.R. Conceptual overview: Disturbance, gradients, and ecological response. In A Caribbean Forest Tapestry: The Multidimensional Nature of Disturbance and Response; Brokaw, N., Crowl, T.A., Lugo, A.E., McDowell, W.H., Scatena, F.N., Waide, R.B., Willig, M.R., Eds.; Oxford University Press: New York, NY, USA, 2012; pp. 42–71. [Google Scholar]
- Willig, M.R.; Walker, L.R. Disturbance in terrestrial ecosystems: Salient themes, synthesis, and future directions. Ecosyst. World 1999, 747–768. [Google Scholar]
- Foster, R.B.; Brokaw, N.V. Structure and History of the Vegetation of Barro Colorado Island [Semideciduous Forest, Panama]; Smithsonian Institutional Press: Washington, DC, USA, 1982. [Google Scholar]
- Brokaw, N.; Fraver, S.; Grear, J.S.; Thompson, J.; Zimmerman, J.K.; Waide, R.B.; Everham, E.M., III; Hubbell, S.P.; Foster, R. Disturbance and canopy structure in two tropical forests. In Tropical Forest Diversity and Dynamism: Findings from a Large-Scale Plot Network; Losos, E.C., Leigh, E.G., Eds.; University of Chicago Press: Chicago, IL, USA, 2004. [Google Scholar]
- De Gouvenain, R.C.; Silander, J.A., Jr. Do tropical storm regimes influence the structure of tropical lowland rain forests? Biotropica 2003, 35, 166–180. [Google Scholar] [CrossRef]
- Johnstone, J.F.; Allen, C.D.; Franklin, J.F.; Frelich, L.E.; Harvey, B.J.; Higuera, P.E.; Mack, M.C.; Meentemeyer, R.K.; Metz, M.R.; Perry, G.L. Changing disturbance regimes, ecological memory, and forest resilience. Front. Ecol. Environ. 2016, 14, 369–378. [Google Scholar] [CrossRef]
- Griffith, M.P.; Noblick, L.R.; Dowe, J.L.; Husby, C.E.; Calonje, M.A. Cyclone tolerance in new world arecaceae: Biogeographic variation and abiotic natural selection. Ann. Bot. 2008, 102, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Basnet, K.; Scatena, F.N.; Likens, G.E.; Lugo, A.E. Ecological consequences of root grafting in Tabonuco (Dacryides excelsa) Trees in the Luquillo Experimental Forest, Puerto Rico. Biotropica 1993, 25, 28–35. [Google Scholar] [CrossRef]
- Mabry, C.M.; Hamburg, S.P.; Lin, T.C.; Horng, F.W.; King, H.B.; Hsia, Y.J. Typhoon disturbance and stand-level damage patterns at a subtropical forest in Taiwan. Biotropica 1998, 30, 238–250. [Google Scholar] [CrossRef]
- Connell, J.H. Diversity in tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef] [PubMed]
- Sheil, D.; Burslem, D.F. Disturbing hypotheses in tropical forests. Trends Ecol. Evol. 2003, 18, 18–26. [Google Scholar] [CrossRef]
- Paine, C.; Amissah, L.; Auge, H.; Baraloto, C.; Baruffol, M.; Bourland, N.; Bruelheide, H.; Daïnou, K.; Gouvenain, R.C.; Doucet, J.-L.; et al. Globally, functional traits are weak predictors of juvenile tree growth, and we do not know why. J. Ecol. 2015, 103, 978–989. [Google Scholar] [CrossRef]
- Yang, J.; Cao, M.; Swenson, N.G. Why functional traits do not predict tree demographic rates. Trends Ecol. Evol. 2018, 33, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.; Brokaw, N.; Zimmerman, J.K.; Waide, R.B.; Everham, E.M.; Lodge, D.J.; Taylor, C.M.; García-Montiel, D.; Fluet, M. Land use history, environment, and tree composition in a tropical forest. Ecol. Appl. 2002, 12, 1344–1363. [Google Scholar] [CrossRef]
- Swenson, N.G.; Erickson, D.L.; Mi, X.; Bourg, N.A.; Forero-Montaña, J.; Ge, X.; Howe, R.; Lake, J.K.; Liu, X.; Ma, K. Phylogenetic and functional alpha and beta diversity in temperate and tropical tree communities. Ecology 2012, 93, S112–S125. [Google Scholar] [CrossRef]
- Dale, V.H.; Joyce, L.A.; McNulty, S.; Neilson, R.P.; Ayres, M.P.; Flannigan, M.D.; Hanson, P.J.; Irland, L.C.; Lugo, A.E.; Peterson, C.J. Climate change and forest disturbances: Climate change can affect forests by altering the frequency, intensity, duration, and timing of fire, drought, introduced species, insect and pathogen outbreaks, hurricanes, windstorms, ice storms, or landslides. BioScience 2001, 51, 723–734. [Google Scholar] [CrossRef]
- Niklas, K.J. Computing factors of safety against wind induced tree damage. J. Exp. Bot. 2000, 51, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Wood, C.J. Understanding wind forces on trees. In Wind and Trees; Coutts, M.P., Grace, J., Eds.; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Elsner, J.B.; Kossin, J.P.; Jagger, T.H. The increasing intensity of the strongest tropical cyclones. Nature 2008, 455, 92. [Google Scholar] [CrossRef] [PubMed]
- Velden, C.; Olander, T.; Herndon, D.; Kossin, J.P. Reprocessing the most intense historical tropical cyclones in the satellite era using the advanced Dvorak technique. Mon. Weather Rev. 2017, 145, 971–983. [Google Scholar] [CrossRef]

| Site | Location | Mean Annual Temperature (°C) | Mean Annual Precipitation (mm) | Average Number of Storms per Year (1982–2015) | Nearest Storm (km) | Average Storm Distance (km) ┼ | Highest Estimated Wind Velocity (kt) | Average Estimated Wind Velocity (kt) § |
|---|---|---|---|---|---|---|---|---|
| BCI | 9°10′ N, 79°50′ W | 27.1 | 2551 | 0.4 ± 0.2 | 102 | 168 ± 12 | 85 | 35.3 ± 5.3 |
| Luquillo | 18°20′ N, 62°49′ W | 22.8 | 3548 | 3 ± 1 | 17 | 148 ± 6 | 135 | 63.4 ± 3.1 |
| Palanan | 17°12′ N, 122°23′ E | 26.1 | 3218 | 9 ± 1 | 20 | 151 ± 3 | 125 | 64.9 ± 1.3 |
| Fushan | 24°34′ N, 121°34′ E | 18.2 | 4271 | 13 ± 2 | 6 | 139 ± 3 | 125 | 62.5 ± 0.9 |
| Site | Census Interval | Year Range | No. of Years Since Last Major Cyclonic Storm | Δ Biomass (tons ha−1) | Δ Biomass Stem−1 (tons ha−1) | Δ No. Stems ha−1 | Mortality Rate (% Yr−1) | Recruitment Rate (% Yr−1) | Stem Growth Increment (mm Yr−1) |
|---|---|---|---|---|---|---|---|---|---|
| BCI | 1 $ | 1982–1985 $ | 261 | 0.027 | 0.043 | 1.315 | |||
| 2 | 1985–1990 | −7.95 | −0.077 | 119 | 0.022 | 0.032 | 1.369 | ||
| 3 | 1990–1995 | −2.13 | 0.042 | −212 | 0.028 | 0.020 | 0.803 | ||
| 4 | 1995–2000 | 4.11 | 0.114 | −321 | 0.031 | 0.020 | 0.251 | ||
| 5 | 2000–2005 | −2.88 | −0.149 | −563 | 0.026 | 0.026 | 0.868 | ||
| 6 | 2005–2010 | −18.72 | −0.136 | −489 | 0.027 | 0.030 | 0.967 | ||
| Luquillo | 1 | 1990–1995 | 2 | −5.87 | −0.217 | −1084 | 0.119 | 0.049 | 2.011 |
| 2 | 1995–2000 | 7 ¢ | −11.31 | −0.247 | −377 | 0.105 | 0.092 | 1.868 | |
| 3 | 2000–2005 | 12 | −13.41 | −0.791 | −1242 | 0.088 | 0.017 | 1.436 | |
| 4 | 2005–2011 | 17 | −14.40 | −0.781 | −477 | 0.048 | 0.014 | 1.050 | |
| Palanan | 1 | 1994–1998 | 4.82 | 0.262 | −396 | 0.033 | 0.199 | 1.641 | |
| 2 | 1998–2004 | 0 | 12.9 | −0.388 | 978 | 0.023 | 0.064 | 1.174 | |
| 3 | 2004–2010 | 4 | 44.9 | 0.307 | −219 | 0.023 | 0.016 | 1.260 | |
| Fushan | 1 | 2004–2009 | 1 | 9.08 | 0.082 | −86 | 0.023 | 0.031 | 1.242 |
| 2 | 2009–2014 | 5,0 | 11.84 | 0.147 | −260 | 0.029 | 0.031 | 1.001 |
| Site | Census Interval | Years Since Last Major Cyclonic Storm | Intrinsic Rate of Population Growth (r) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stems ≥1 cm dbh | Stems ≥10 cm dbh | |||||||||||||
| Specific Leaf Area | Tree Height | Wood Density | Specific Leaf Area | Tree Height | Wood Density | |||||||||
| Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | |||
| BCI | 1 | 0.07 | 0.32 | −0.11 | 0.06 | 0.25 | <0.001 | −0.03 | 0.75 | −0.06 | 0.40 | 0.11 | 0.11 | |
| 2 | −0.07 | 0.36 | 0.06 | 0.32 | 0.15 | <0.01 | −0.04 | 0.61 | 0.03 | 0.63 | 0.09 | 0.19 | ||
| 3 | −0.23 | <0.01 | 0.23 | <0.001 | 0.23 | <0.001 | −0.06 | 0.45 | 0.09 | 0.16 | −0.04 | 0.54 | ||
| 4 | 0.23 | <0.01 | 0.08 | 0.19 | −0.09 | 0.16 | 0.25 | <0.01 | −0.01 | 0.86 | −0.16 | <0.05 | ||
| 5 | −0.02 | 0.81 | −0.05 | 0.42 | −0.18 | <0.01 | −0.13 | 0.09 | 0.12 | 0.07 | −0.01 | 0.27 | ||
| 6 | 0.01 | 0.87 | 0.01 | 0.89 | −0.10 | 0.10 | 0.04 | 0.64 | −0.09 | 0.19 | −0.09 | 0.19 | ||
| Luquillo | 1 | 2 | −0.33 | <0.001 | 0.17 | >0.05 | 0.23 | <0.01 | 0.19 | 0.08 | 0.17 | 0.12 | −0.25 | <0.05 |
| 2 | 7 ¢ | 0.03 | 0.72 | 0.03 | 0.75 | 0.03 | 0.78 | 0.10 | 0.39 | −0.05 | 0.64 | −0.24 | <0.05 | |
| 3 | 12 | −0.45 | <0.001 | 0.23 | 0.02 | 0.25 | <0.01 | 0.17 | 0.14 | 0.05 | 0.65 | −0.02 | 0.87 | |
| 4 | 17 | −0.53 | <0.001 | 0.13 | 0.16 | 0.2 | <0.05 | −0.20 | 0.07 | 0.15 | 0.18 | −0.06 | 0.62 | |
| Palanan | 1 | −0.10 | 0.15 | 0.15 | 0.18 | 0.31 | <0.05 | 0.09 | 0.25 | −0.03 | 0.82 | −0.17 | 0.16 | |
| 2 | 0 | 0.26 | <0.001 | −0.25 | 0.10 | −0.22 | <0.01 | 0.19 | <0.05 | 0.31 | 0.06 | −0.23 | <0.05 | |
| 3 | 4 | −0.15 | <0.05 | 0.18 | 0.22 | 0.21 | 0.07 | 0.21 | <0.01 | 0.04 | 0.72 | −0.13 | 0.41 | |
| Fushan | 1 | 1 | −0.30 | <0.01 | −0.03 | 0.79 | −0.02 | 0.88 | −0.01 | 0.93 | 0.03 | 0.85 | −0.10 | 0.45 |
| 2 | 5,0 | −0.22 | <0.05 | 0.14 | 0.21 | 0.08 | 0.44 | −0.09 | 0.47 | −0.03 | 0.81 | −0.14 | 0.30 | |
| Site | Census Interval | Years Since Last Major Cyclonic Storm | Mortality Rate | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stems ≥1 cm dbh | Stems ≥10 cm dbh | |||||||||||||
| Specific Leaf Area | Tree Height | Wood Density | Specific Leaf Area | Tree Height | Wood Density | |||||||||
| Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | Pearson’s r | p | |||
| BCI | 1 | 0.11 | 0.16 | −0.13 | <0.05 | −0.39 | <0.001 | 0.09 | 0.24 | −0.20 | <0.01 | −0.16 | <0.05 | |
| 2 | 0.35 | <0.001 | −0.34 | <0.001 | −0.29 | <0.001 | 0.21 | <0.01 | −0.25 | <0.001 | −0.23 | <0.001 | ||
| 3 | 0.31 | <0.001 | −0.34 | <0.001 | −0.34 | <0.001 | 0.21 | <0.01 | −0.29 | <0.001 | −0.09 | 0.19 | ||
| 4 | 0.31 | <0.001 | −0.33 | <0.001 | −0.28 | <0.001 | 0.02 | 0.82 | −0.22 | <0.01 | −0.16 | <0.05 | ||
| 5 | 0.37 | <0.001 | −0.24 | <0.001 | −0.19 | <0.01 | 0.27 | <0.001 | −0.22 | <0.01 | −0.08 | 0.24 | ||
| 6 | 0.43 | <0.001 | −0.23 | <0.001 | −0.25 | <0.001 | 0.23 | <0.01 | −0.21 | <0.01 | −0.18 | <0.01 | ||
| Luquillo | 1 | 2 | 0.34 | <0.001 | −0.17 | 0.06 | −0.20 | <0.05 | 0.2 | 0.07 | −0.29 | <0.01 | 0.22 | <0.05 |
| 2 | 7 ¢ | 0.55 | <0.001 | −0.20 | <0.001 | −0.23 | <0.01 | 0.11 | 0.34 | −0.19 | 0.09 | 0.22 | >0.05 | |
| 3 | 12 | 0.38 | <0.001 | −0.23 | <0.001 | −0.26 | <0.01 | 0.04 | 0.70 | −0.23 | <0.05 | 0.04 | 0.72 | |
| 4 | 17 | 0.56 | <0.001 | −0.18 | 0.06 | −0.24 | <0.01 | 0.21 | 0.06 | −0.27 | <0.01 | 0.06 | 0.59 | |
| Palanan | 1 | 0.23 | <0.01 | −0.57 | <0.01 | −0.38 | <0.01 | 0.09 | 0.25 | −0.03 | 0.82 | −0.17 | 0.16 | |
| 2 | 0 | 0.12 | 0.07 | −0.45 | <0.01 | −0.19 | 0.08 | 0.17 | 0.01 | 0.31 | 0.06 | −0.23 | <0.05 | |
| 3 | 4 | 0.26 | <0.01 | −0.48 | <0.01 | −0.32 | <0.01 | 0.21 | <0.01 | −0.04 | 0.72 | −0.13 | 0.41 | |
| Fushan | 1 | 1 | 0.38 | <0.001 | −0.28 | <0.01 | −0.24 | <0.05 | 0.04 | 0.80 | 0.05 | 0.70 | −0.01 | 0.91 |
| 2 | 5,0 | 0.37 | <0.001 | −0.46 | <0.001 | −0.24 | <0.05 | 0.09 | 0.48 | −0.10 | 0.45 | 0.02 | 0.89 | |
| Site | Census | Avg. Quadrat CWM SLA (m2 kg−1) | Avg. Quadrat CWM WD (g cm−3) | Avg. Quadrat CWM H (m) |
|---|---|---|---|---|
| BCI | 1 | 12.10 ± 0.02 | 0.594 ± 0.001 | 16.7 ± 0.1 |
| 2 | 12.10 ± 0.02 | 0.597 ± 0.001 | 16.6 ± 0.1 | |
| 3 | 12.05 ± 0.02 | 0.599 ± 0.001 | 16.8 ± 0.1 | |
| 4 | 11.95 ± 0.02 | 0.603 ± 0.001 | 17.1 ± 0.1 | |
| 5 | 11.86 ± 0.02 | 0.605 ± 0.001 | 17.2 ± 0.1 | |
| 6 | 11.78 ± 0.02 | 0.604 ± 0.001 | 17.3 ± 0.1 | |
| 7 | 11.73 ± 0.02 | 0.603 ± 0.001 | 17.4 ± 0.1 | |
| Luquillo | 1 | 19.54 ± 0.02 | 0.551 ± 0.003 | 16.1 ± 0.2 |
| 2 | 19.64 ± 0.02 | 0.543 ± 0.003 | 15.8 ± 0.2 | |
| 3 | 22.10 ± 0.03 | 0.534 ± 0.003 | 14.8 ± 0.2 | |
| 4 | 19.06 ± 0.02 | 0.538 ± 0.003 | 16.3 ± 0.2 | |
| 5 | 17.83 ± 0.02 | 0.530 ± 0.003 | 17.1 ± 0.2 | |
| Palanan | 1 | 14.07 ± 0.04 | 0.555 ± 0.002 | 16.2 ± 0.2 |
| 2 | 14.02 ± 0.04 | 0.554 ± 0.002 | 16.4 ± 0.1 | |
| 3 | 14.04 ± 0.03 | 0.548 ± 0.001 | 16.0 ± 0.1 | |
| 4 | 14.05 ± 0.03 | 0.548 ± 0.001 | 16.0 ± 0.1 | |
| Fushan | 1 | 17.45 ± 0.07 | 0.493 ± 0.001 | 15.0 ± 0.1 |
| 2 | 17.69 ± 0.07 | 0.493 ± 0.001 | 14.6 ± 0.1 | |
| 3 | 18.17 ± 0.07 | 0.493 ± 0.001 | 14.0 ± 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hogan, J.A.; Zimmerman, J.K.; Thompson, J.; Uriarte, M.; Swenson, N.G.; Condit, R.; Hubbell, S.; Johnson, D.J.; Sun, I.F.; Chang-Yang, C.-H.; et al. The Frequency of Cyclonic Wind Storms Shapes Tropical Forest Dynamism and Functional Trait Dispersion. Forests 2018, 9, 404. https://doi.org/10.3390/f9070404
Hogan JA, Zimmerman JK, Thompson J, Uriarte M, Swenson NG, Condit R, Hubbell S, Johnson DJ, Sun IF, Chang-Yang C-H, et al. The Frequency of Cyclonic Wind Storms Shapes Tropical Forest Dynamism and Functional Trait Dispersion. Forests. 2018; 9(7):404. https://doi.org/10.3390/f9070404
Chicago/Turabian StyleHogan, J. Aaron, Jess K. Zimmerman, Jill Thompson, María Uriarte, Nathan G. Swenson, Richard Condit, Stephen Hubbell, Daniel J. Johnson, I Fang Sun, Chia-Hao Chang-Yang, and et al. 2018. "The Frequency of Cyclonic Wind Storms Shapes Tropical Forest Dynamism and Functional Trait Dispersion" Forests 9, no. 7: 404. https://doi.org/10.3390/f9070404
APA StyleHogan, J. A., Zimmerman, J. K., Thompson, J., Uriarte, M., Swenson, N. G., Condit, R., Hubbell, S., Johnson, D. J., Sun, I. F., Chang-Yang, C.-H., Su, S.-H., Ong, P., Rodriguez, L., Monoy, C. C., Yap, S., & Davies, S. J. (2018). The Frequency of Cyclonic Wind Storms Shapes Tropical Forest Dynamism and Functional Trait Dispersion. Forests, 9(7), 404. https://doi.org/10.3390/f9070404








