Stem Circadian Phenology of Four Pine Species in Naturally Contrasting Climates from Sky-Island Forests of the Western USA
Abstract
1. Introduction
2. Materials and Methods
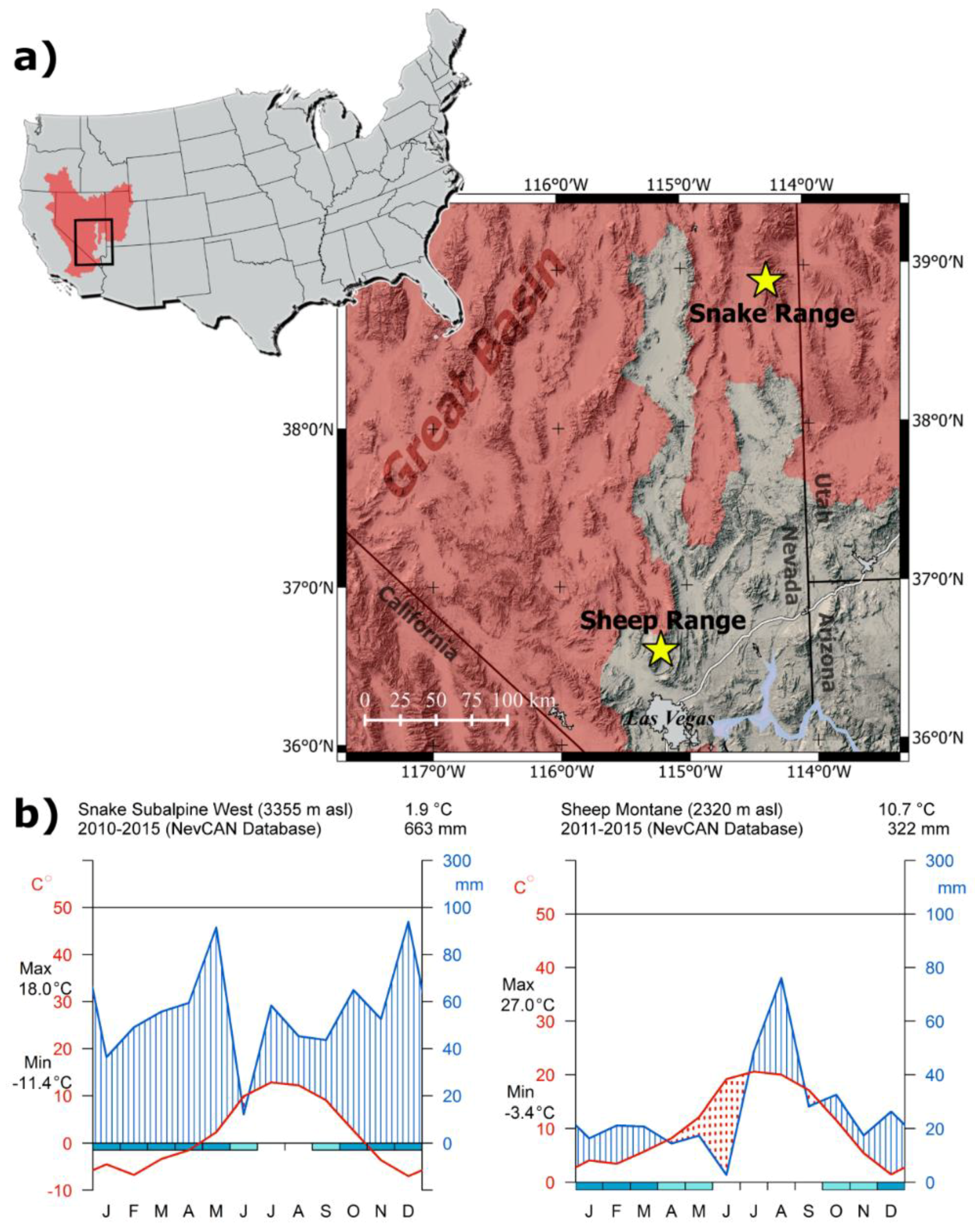
2.1. Study Area
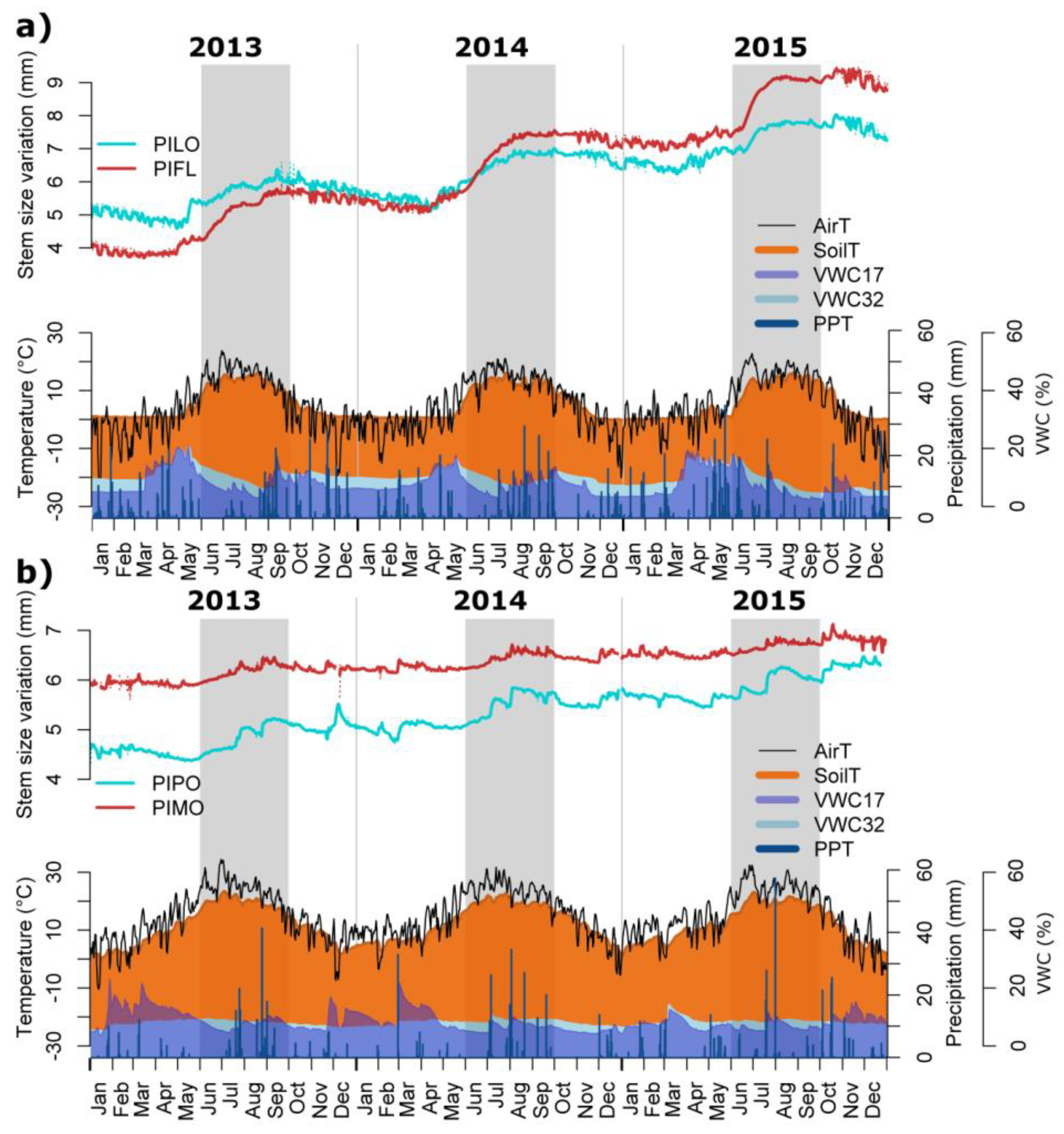
2.2. Dendrometer Data
2.3. Climatic Data
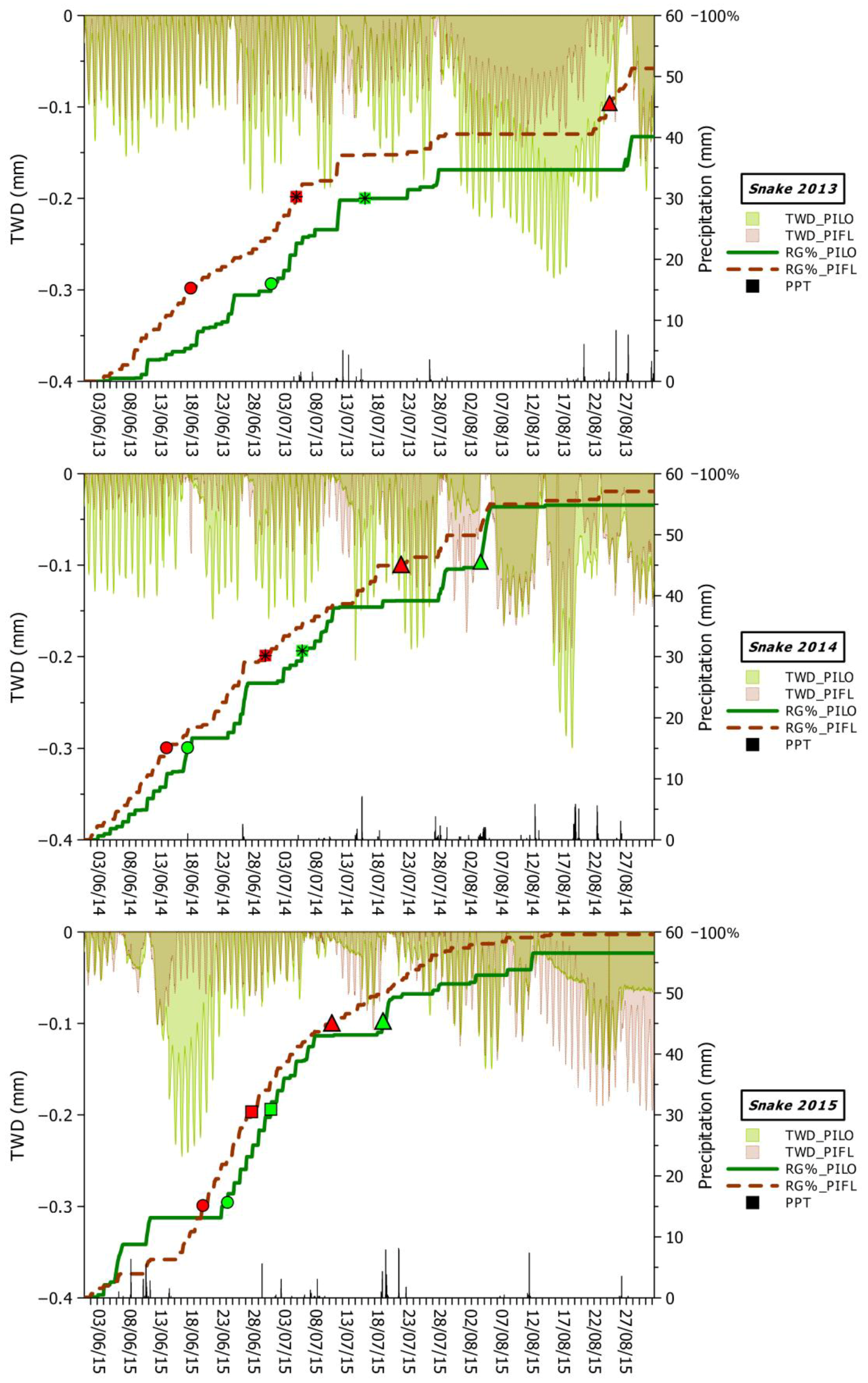
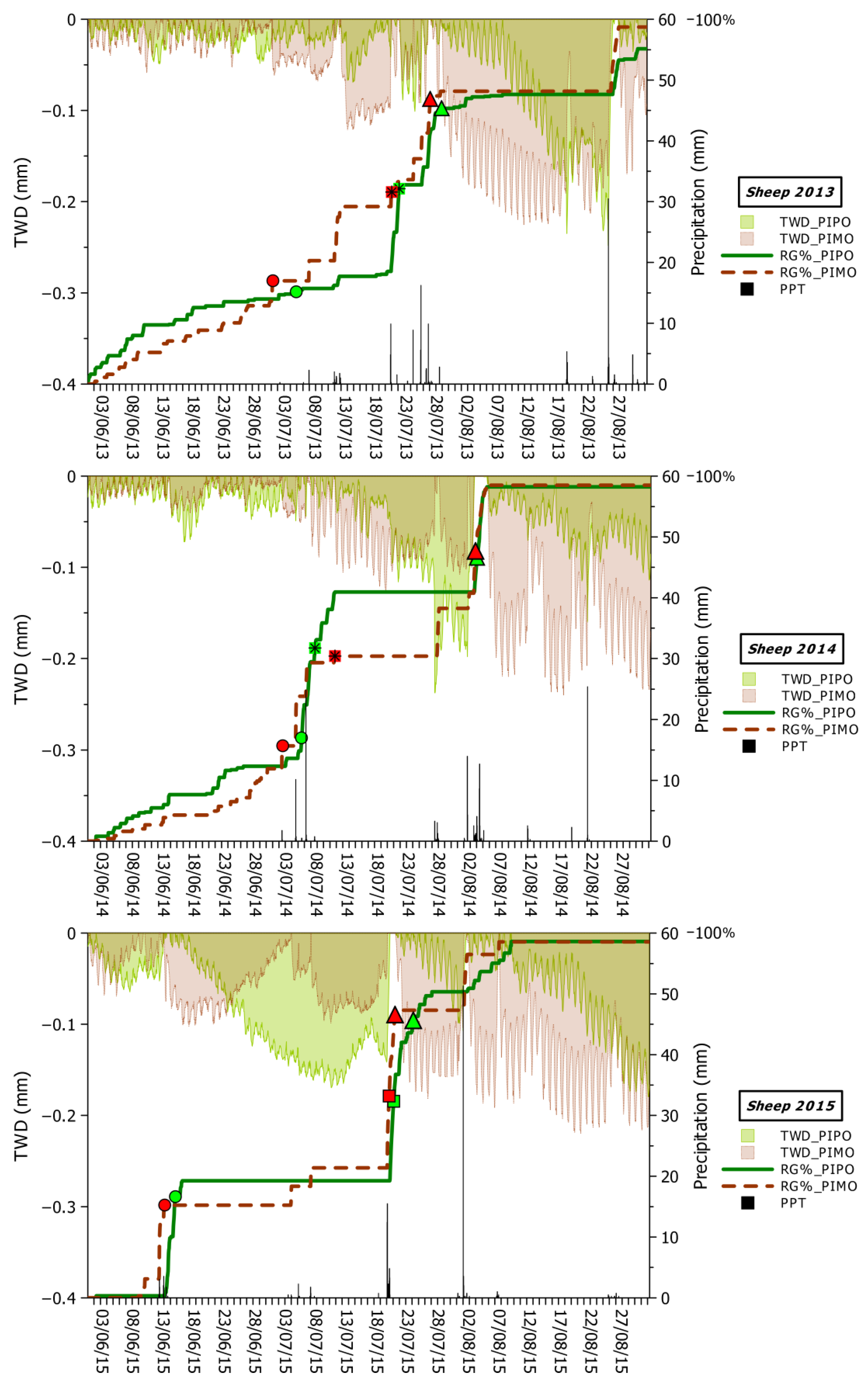
2.4. Extraction of Stem-Cycle Phases
2.5. Modeling Relationships between Climate, Tree Water Deficit (TWD), and Real Growth (RG)
3. Results
3.1. Climate
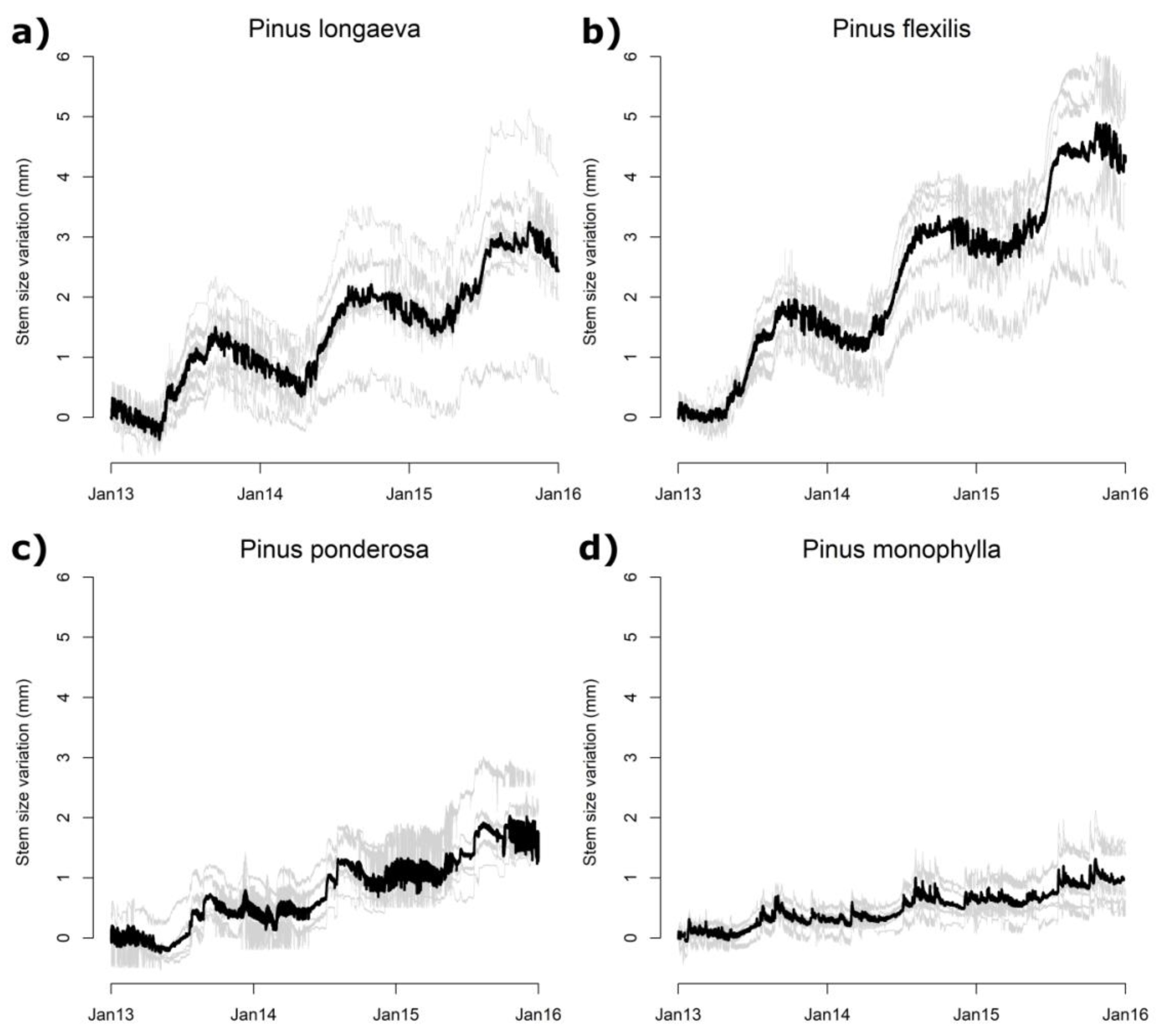
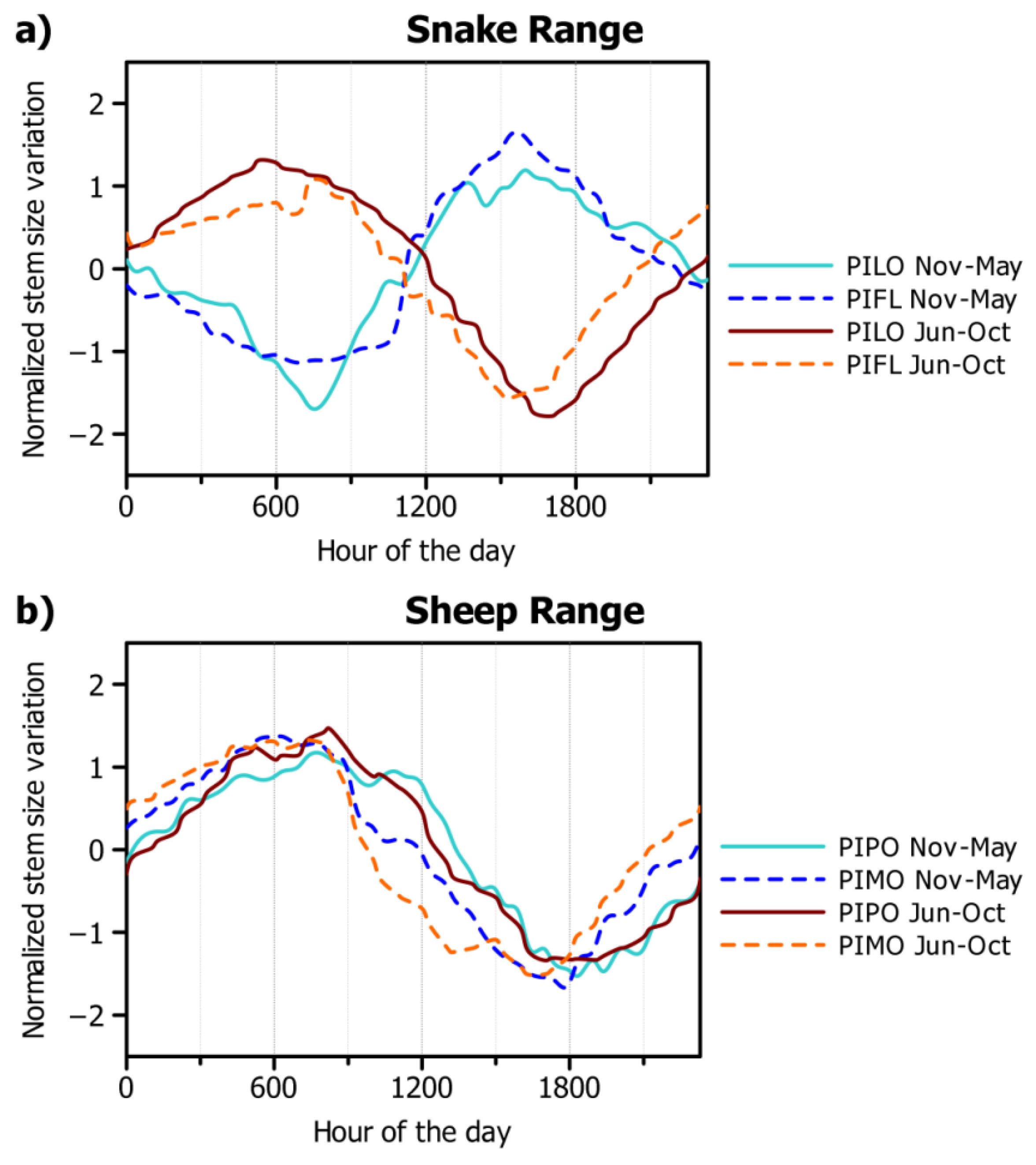
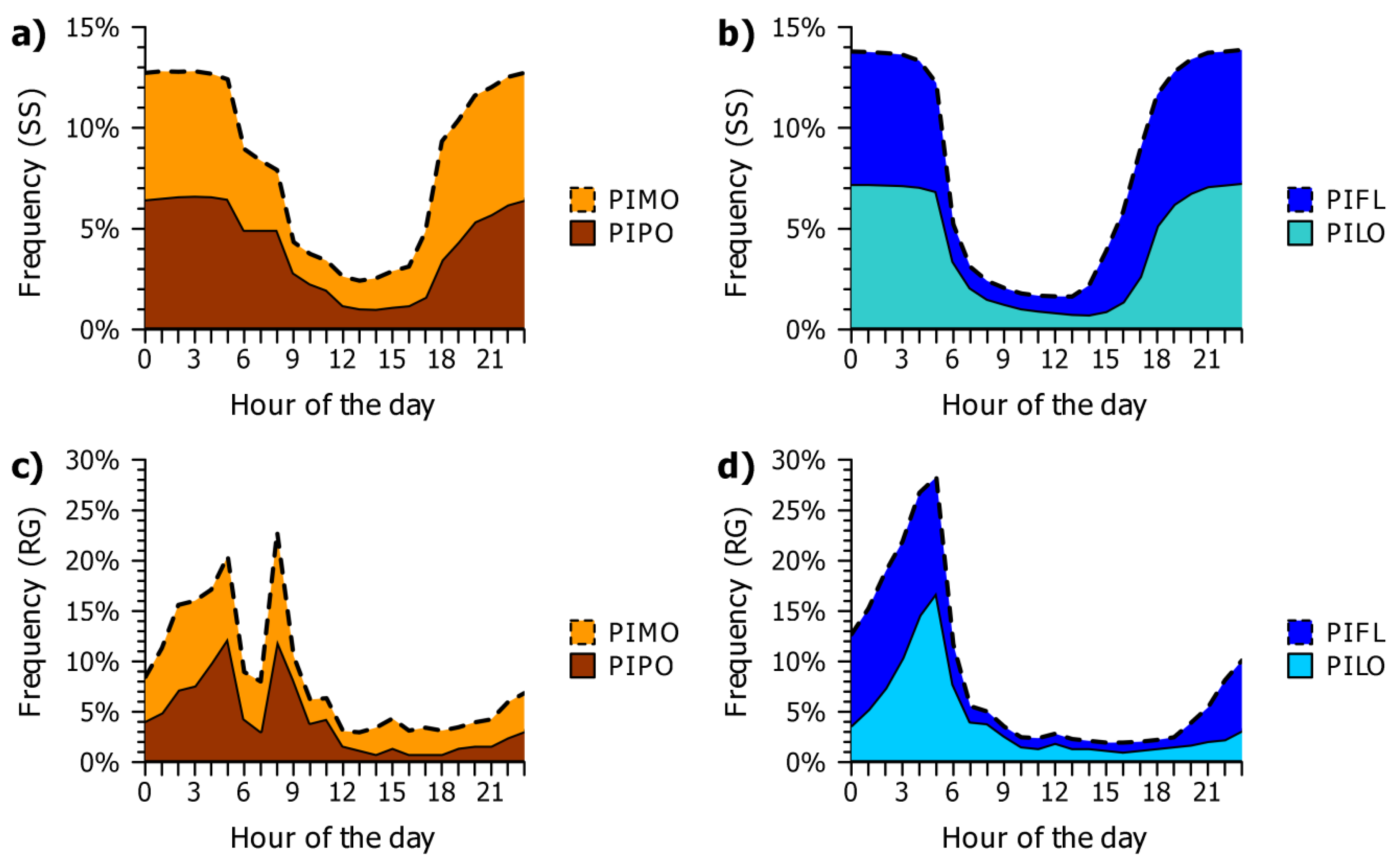
3.2. Seasonal and Daily Dynamics of Stem-Size Variation
3.3. Climatic Influences on Tree Water Deficit and Real Growth
4. Discussion
4.1. Seasonal Dynamics of Circadian Cycles
4.2. Climatic Drivers of Stem Swelling and Real Growth
4.3. Implications for Species Adaptation to Climate Change
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mendivelso, H.A.; Camarero, J.J.; Gutiérrez, E.; Zuidema, P.A. Time-dependent effects of climate and drought on tree growth in a Neotropical dry forest: Short-term tolerance vs. long-term sensitivity. Agr. Forest Meteorol. 2014, 188, 13–23. [Google Scholar] [CrossRef]
- George, S.S. An overview of tree-ring width records across the Northern Hemisphere. Quat. Sci. Rev. 2014, 95, 132–150. [Google Scholar] [CrossRef]
- Kocher, P.; Horna, V.; Leuschner, C. Environmental control of daily stem growth patterns in five temperate broad-leaved tree species. Tree Physiol. 2012, 32, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Deslauriers, A.; Anfodillo, T.; Rossi, S.; Carraro, V. Using simple causal modeling to understand how water and temperature affect daily stem radial variation in trees. Tree Physiol. 2007, 27, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Sevanto, S.; McDowell, N.G.; Dickman, L.T.; Pangle, R.; Pockman, W.T. How do trees die? A test of the hydraulic failure and carbon starvation hypotheses. Plant Cell Environ. 2014, 37, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Steppe, K.; Sterck, F.; Deslauriers, A. Diel growth dynamics in tree stems: Linking anatomy and ecophysiology. Trends Plant Sci. 2015, 20, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Coussement, J.R.; De Swaef, T.; Lootens, P.; Roldán-Ruiz, I.; Steppe, K. Introducing turgor-driven growth dynamics into functional-structural plant models. Ann. Bot. 2018, 121, 849–861. [Google Scholar] [CrossRef] [PubMed]
- van der Maaten, E.; Bouriaud, O.; van der Maaten-Theunissen, M.; Mayer, H.; Spiecker, H. Meteorological forcing of day-to-day stem radius variations of beech is highly synchronic on opposing aspects of a valley. Agr. Forest Meteorol. 2013, 181, 85–93. [Google Scholar] [CrossRef]
- Oberhuber, W. Soil water availability and evaporative demand affect seasonal growth dynamics and use of stored water in co-occurring saplings and mature conifers under drought. Trees-Struct. Funct. 2017, 31, 467–478. [Google Scholar] [CrossRef] [PubMed]
- De Swaef, T.; De Schepper, V.; Vandegehuchte, M.W.; Steppe, K. Stem diameter variations as a versatile research tool in ecophysiology. Tree Physiol. 2015, 35, 1047–1061. [Google Scholar] [CrossRef] [PubMed]
- Herzog, K.M.; Häsler, R.; Thum, R. Diurnal changes in the radius of a subalpine Norway spruce stem: their relation to the sap flow and their use to estimate transpiration. Trees 1995, 10, 94–101. [Google Scholar] [CrossRef]
- Zweifel, R.; Item, H.; Hasler, R. Link between diurnal stem radius changes and tree water relations. Tree Physiol. 2001, 21, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Braekke, F.H.; Kozlowski, T.T. Shrinkage and swelling of stems of Pinus resinosa and Betula papyrifera in northern Wisconsin. Plant Soil 1975, 43, 387–410. [Google Scholar] [CrossRef]
- Ziaco, E.; Biondi, F. Tree growth, cambial phenology, and wood anatomy of limber pine at a Great Basin (USA) mountain observatory. Trees-Struct. Funct. 2016, 30, 1507–1521. [Google Scholar] [CrossRef]
- Zweifel, R.; Haeni, M.; Buchmann, N.; Eugster, W. Are trees able to grow in periods of stem shrinkage? New Phytol. 2016, 211, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Phillips, N.G.; Ryan, M.G.; Bond, B.J.; McDowell, N.G.; Hinckley, T.M.; Čermák, J. Reliance on stored water increases with tree size in three species in the Pacific Northwest. Tree Physiol. 2003, 23, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Čermák, J.; Kučera, J.; Bauerle, W.L.; Phillips, N.; Hinckley, T.M. Tree water storage and its diurnal dynamics related to sap flow and changes in stem volume in old-growth Douglas-fir trees. Tree Physiol. 2007, 27, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Barnard, D.M.; Meinzer, F.C.; Lachenbruch, B.; McCulloh, K.A.; Johnson, D.M.; Woodruff, D.R. Climate-related trends in sapwood biophysical properties in two conifers: avoidance of hydraulic dysfunction through coordinated adjustments in xylem efficiency, safety and capacitance. Plant Cell Environ. 2011, 34, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Meinzer, F.C.; Bond, B.J.; Warren, J.M.; Woodruff, D.R. Does water transport scale universally with tree size? Funct. Ecol. 2005, 19, 558–565. [Google Scholar] [CrossRef]
- King, G.; Fonti, P.; Nievergelt, D.; Büntgen, U.; Frank, D. Climatic drivers of hourly to yearly tree radius variations along a 6 °C natural warming gradient. Agr. Forest Meteorol. 2013, 168, 36–46. [Google Scholar] [CrossRef]
- Turcotte, A.; Rossi, S.; Deslauriers, A.; Krause, C.; Morin, H. Dynamics of depletion and replenishment of water storage in stem and roots of black spruce measured by dendrometers. Front. Plant Sci. 2011, 2, 21. [Google Scholar] [CrossRef] [PubMed]
- Deslauriers, A.; Morin, H.; Urbinati, C.; Carrer, M. Daily weather response of balsam fir (Abies balsamea (L.) Mill.) stem radius increment from dendrometer analysis in the boreal forests of Québec (Canada). Trees 2003, 17, 477–484. [Google Scholar] [CrossRef]
- Biondi, F.; Rossi, S. Plant-water relationships in the Great Basin Desert of North America derived from Pinus monophylla hourly dendrometer records. Int. J. Biometeorol. 2015, 59, 939–953. [Google Scholar] [CrossRef] [PubMed]
- Drew, D.M.; Downes, G.M. The use of precision dendrometers in research on daily stem size and wood property variation: A review. Dendrochronologia 2009, 27, 159–172. [Google Scholar] [CrossRef]
- Grayson, D.K. The Great Basin: A Natural Prehistory, Revised and Expanded Edition; University of California Press: Berkeley, CA, USA, 2011. [Google Scholar]
- Osmond, C.B.; Pitelka, L.F.; Hidy, G.M. Plant Biology of the Basin and Range; Springer-Verlag: Berlin, Germany, 1990; p. 375. [Google Scholar]
- Adams, D.K.; Comrie, A.C. The North American Monsoon. Bull. Am. Meteorol. Soc. 1997, 78, 2197–2213. [Google Scholar] [CrossRef]
- Szejner, P.; Wright, W.E.; Babst, F.; Belmecheri, S.; Trouet, V.; Leavitt, S.W.; Ehleringer, J.R.; Monson, R.K. Latitudinal gradients in tree ring stable carbon and oxygen isotopes reveal differential climate influences of the North American Monsoon System. J. Geophys. Res. Biogeosci. 2016, 121, 1978–1991. [Google Scholar] [CrossRef]
- Ellison, A.M.; Bank, M.S.; Clinton, B.D.; Colburn, E.A.; Elliott, K.; Ford, C.R.; Foster, D.R.; Kloeppel, B.D.; Knoepp, J.D.; Lovett, G.M.; et al. Loss of foundation species: Consequences for the structure and dynamics of forested ecosystems. Front. Ecol. the Environ. 2005, 3, 479–486. [Google Scholar] [CrossRef]
- Mensing, S.; Strachan, S.; Arnone, J.; Fenstermaker, L.; Biondi, F.; Devitt, D.; Johnson, B.; Bird, B.; Fritzinger, E. A network for observing Great Basin climate change. EOS Trans. Amer. Geophys. Union 2013, 94, 105–106. [Google Scholar] [CrossRef]
- Johnson, B.G.; Verburg, P.S.J.; Arnone, J.A. Effects of climate and vegetation on soil nutrients and chemistry in the Great Basin studied along a latitudinal-elevational climate gradient. Plant Soil 2014, 382, 151–163. [Google Scholar] [CrossRef]
- Gochis, D.; Schemm, J.; Shi, W.; Long, L.; Higgins, W.; Douglas, A. A Forum for Evaluating Forecasts of the North American Monsoon. Eos Trans. Amer. Geophys. Union 2009, 90, 249–251. [Google Scholar] [CrossRef]
- Biondi, F.; Hartsough, P.C.; Estrada, I.G. Daily weather and tree growth at the tropical treeline of North America. Arct. Antarctic Alp. Res. 2005, 37, 16–24. [Google Scholar] [CrossRef]
- Biondi, F.; Hartsough, P. Using automated point dendrometers to analyze tropical treeline stem growth at Nevado de Colima, Mexico. Sensors 2010, 10, 5827. [Google Scholar] [CrossRef] [PubMed]
- Ziaco, E.; Biondi, F.; Rossi, S.; Deslauriers, A. Environmental drivers of cambial phenology in Great Basin bristlecone pine. Tree Physiol. 2016, 36, 818–831. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.G. The Relationship between Relative Humidity and the Dewpoint Temperature in Moist Air: A Simple Conversion and Applications. Bull. Amer. Meteorol. Soc. 2005, 86, 225–233. [Google Scholar] [CrossRef]
- Feld, S.I.; Cristea, N.C.; Lundquist, J.D. Representing atmospheric moisture content along mountain slopes: Examination using distributed sensors in the Sierra Nevada, California. Water Resour. Res. 2013, 49, 4424–4441. [Google Scholar] [CrossRef]
- McEvoy, D.J.; Mejia, J.F.; Huntington, J.L. Use of an Observation Network in the Great Basin to Evaluate Gridded Climate Data. J. Hydrometeorol. 2014, 15, 1913–1931. [Google Scholar] [CrossRef]
- Kunkel, K.E. Simple procedures for extrapolation of humidity variables in the mountainous western United States. J. Clim. 1989, 2, 656–669. [Google Scholar] [CrossRef]
- Maaten, E.V.D.; Maaten-Theunissen, M.V.D.; Smiljanić, M.; Rossi, S.; Simard, S.; Wilmking, M.; Deslauriersb, A.; Fontie, P.; Arxe, G.; Bouriaudf, O. DendrometeR: Analyzing the pulse of trees in R. Dendrochronologia 2016, 40, 12–16. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Deslauriers, A.; Rossi, S.; Turcotte, A.; Morin, H.; Krause, C. A three-step procedure in SAS to analyze the time series from automatic dendrometers. Dendrochronologia 2011, 29, 151–161. [Google Scholar] [CrossRef]
- Ziaco, E.; Truettner, C.; Biondi, F.; Bullock, S. Moisture-driven xylogenesis in Pinus ponderosa from a Mojave Desert mountain reveals high phenological plasticity. Plant Cell Environ. 2018, 41, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.; Berninger, F.; Mencuccini, M.; Nikinmaa, E. Separating water-potential induced swelling and shrinking from measured radial stem variations reveals a cambial growth and osmotic concentration signal. Plant Cell Environ. 2016, 39, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Hosmer, J.D.W.; Lemeshow, S.; Sturdivant, R.X. The Multiple Logistic Regression Model, in Applied Logistic Regression; John Wiley & Sons: Hoboken, NJ, USA, 2013; pp. 35–47. [Google Scholar]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Pearce, J.; Ferrier, S. Evaluating the predictive performance of habitat models developed using logistic regression. Ecol. Modell. 2000, 133, 225–245. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinformatics 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Oberhuber, W.; Hammerle, A.; Kofler, W. Tree water status and growth of saplings and mature Norway spruce (Picea abies) at a dry distribution limit. Front. Plant Sci. 2015, 6, 703. [Google Scholar] [CrossRef] [PubMed]
- Zweifel, R.; Häsler, R. Dynamics of water storage in mature subalpine Picea abies: Temporal and spatial patterns of change in stem radius. Tree Physiol. 2001, 21, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Maherali, H.; DeLucia, E.H. Influence of climate-driven shifts in biomass allocation on water transport and storage in ponderosa pine. Oecologia 2001, 129, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Zweifel, R.; Häsler, R. Frost-induced reversible shrinkage of bark of mature subalpine conifers. Agr. Forest Meteorol. 2000, 102, 213–222. [Google Scholar] [CrossRef]
- Vieira, J.; Rossi, S.; Campelo, F.; Freitas, H.; Nabais, C. Seasonal and daily cycles of stem radial variation of Pinus pinaster in a drought-prone environment. Agr. Forest Meteorol. 2013, 180, 173–181. [Google Scholar] [CrossRef]
- Tardif, J.; Flannigan, M.; Bergeron, Y. An Analysis of the Daily Radial Activity of 7 Boreal Tree Species, Northwestern Quebec. Environ. Monit. Assess. 2001, 67, 141–160. [Google Scholar] [CrossRef] [PubMed]
- Güney, A.; Küppers, M.; Rathgeber, C.; Şahin, M.; Zimmermann, R. Intra-annual stem growth dynamics of Lebanon Cedar along climatic gradients. Trees 2017, 31, 1–20. [Google Scholar] [CrossRef]
- Linares, J.C.; Camarero, J.J.; Carreira, J.A. Plastic responses of Abies pinsapo xylogenesis to drought and competition. Tree Physiol. 2009, 29, 1525–1536. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Morin, H.; Deslauriers, A. Multi-scale influence of snowmelt on xylogenesis of black spruce. Arct. Antarct. Alp. Res. 2011, 43, 457–464. [Google Scholar] [CrossRef]
- Gaylord, M.L.G.L.; Kolb, T.E.K.E.; Wallin, K.F.W.F.; Wagner, M.R.W.R. Seasonal dynamics of tree growth, physiology, and resin defenses in a northern Arizona ponderosa pine forest. Can. J. Forest Res. 2007, 37, 1173–1183. [Google Scholar] [CrossRef]
- Kolb, T.E.; Stone, J.E. Differences in leaf gas exchange and water relations among species and tree sizes in an Arizona pine-oak forest. Tree Physiol. 2000, 20, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kozlowski, T.T. Soil moisture and absorption of water by tree roots. J. Arboric. 1987, 13, 39–46. [Google Scholar]
- Gambetta, G.A.; Knipfer, T.; Fricke, W.; Mcelrone, A.J. Aquaporins and Root Water Uptake. In Plant Aquaporins: From Transport to Signaling; Chaumont, F., Tyerman, S.D., Eds.; Springer International Publishing: New York, NY, USA, 2017; pp. 133–153. [Google Scholar]
- Bennett, A.C.; Mcdowell, N.G.; Allen, C.D.; Andersonteixeira, K.J. Larger trees suffer most during drought in forests worldwide. Nat. Plants 2015, 1, 15139. [Google Scholar] [CrossRef] [PubMed]
- Domec, J.C.; Gartner, B.L. Relationship between growth rates and xylem hydraulic characteristics in young, mature and old-growth ponderosa pine trees. Plant Cell Environ. 2003, 26, 471–483. [Google Scholar] [CrossRef]
- Hughes, M.K.; Funkhouser, G. Frequency-Dependent Climate Signal in Upper and Lower Forest Border Tree Rings in the Mountains of the Great Basin. Clim. Change 2003, 59, 233–244. [Google Scholar] [CrossRef]
- Gazol, A.; Camarero, J.J.; Anderegg, W.R.L.; Vicente-Serrano, S.M. Impacts of droughts on the growth resilience of Northern Hemisphere forests. Global Ecol. Biogeogr. 2017, 26, 166–176. [Google Scholar] [CrossRef]
- Allen, C.D.; Breshears, D.D.; McDowell, N.G. On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 2015, 6, art129. [Google Scholar] [CrossRef]
- Grossiord, C.; Sevanto, S.; Borrego, I.; Chan, A.M.; Collins, A.D.; Dickman, L.T.; Hudson, P.J.; McBranch, N.; Michaletz, S.T.; Pockman, W.T.; et al. Tree water dynamics in a drying and warming world. Plant Cell Environ. 2017, 40, 1861–1873. [Google Scholar] [CrossRef] [PubMed]
- Peñuelas, J.; Canadell, J.G.; Ogaya, R. Increased water-use efficiency during the 20th century did not translate into enhanced tree growth. Global Ecol. Biogeogr. 2011, 20, 597–608. [Google Scholar] [CrossRef]
- Mencuccini, M. The ecological significance of long-distance water transport: short-term regulation, long-term acclimation and the hydraulic costs of stature across plant life forms. Plant Cell Environ. 2003, 26, 163–182. [Google Scholar] [CrossRef]
- Limousin, J.M.; Rambal, S.; Ourcival, J.M.; Rodríguezcalcerrada, J.; Pérezramos, I.M.; Rodríguezcortina, R.; Misson, L.; Joffre, R. Morphological and phenological shoot plasticity in a Mediterranean evergreen oak facing long-term increased drought. Oecologia 2012, 169, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.; Anchukaitis, K.J. How unusual is the 2012–2014 California drought? Geophys. Res. Lett. 2014, 41, 9017–9023. [Google Scholar] [CrossRef]
- Anderegg, W.R.; Schwalm, C.; Biondi, F.; Camarero, J.J.; Koch, G.; Litvak, M.; Ogle, K.; Shaw, J.D.; Shevliakova, E.; Williams, P.; et al. Pervasive drought legacies in forest ecosystems and their implications for carbon cycle models. Science 2015, 349, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Anderegg, W.R.L.; Kane, J.M.; Anderegg, L.D.L. Consequences of widespread tree mortality triggered by drought and temperature stress. Nat. Clim. Change 2013, 3, 30–36. [Google Scholar] [CrossRef]
- Williams, A.P.; Allen, C.D.; Macalady, A.K.; Griffin, D.; Woodhouse, C.A.; Meko, D.M.; Swetnam, T.W.; Rauscher, S.A.; Seager, R.; Grissino-Mayer, H.D.; et al. Temperature as a potent driver of regional forest drought stress and tree mortality. Nat. Clim. Change 2012, 3, 292. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Camarero, J.J.; Rozas, V.; Génova, M.; Olano, J.M.; Arzac, A.; Gazol, A.; Caminero, L.; Tejedor, E.; Luis, M.; et al. Resist, recover or both? Growth plasticity in response to drought is geographically structured and linked to intraspecific variability in Pinus pinaster. J. Biogeogr. 2018, 45, 1–14. [Google Scholar] [CrossRef]
- Martin, J.; Looker, N.; Hoylman, Z.; Jencso, K.; Hu, J. Hydrometeorology organizes intra-annual patterns of tree growth across time, space and species in a montane watershed. New Phytol. 2017, 215, 1387–1398. [Google Scholar] [CrossRef] [PubMed]
- Biondi, F.; Bradley, M.L. Long-term survivorship of single-needle pinyon (Pinus monophylla) in mixed-conifer ecosystems of the Great Basin, USA. Ecosphere 2013, 4, art120. [Google Scholar] [CrossRef]
- Lloret, F.; Keeling, E.G.; Sala, A. Components of tree resilience: effects of successive low-growth episodes in old ponderosa pine forests. Oikos 2011, 120, 1909–1920. [Google Scholar] [CrossRef]
- Daly, C.; Conklin, D.R.; Unsworth, M.H. Local atmospheric decoupling in complex topography alters climate change impacts. Int. J. Climatol. 2010, 30, 1857–1864. [Google Scholar] [CrossRef]
- Loarie, S.R.; Duffy, P.B.; Hamilton, H.; Asner, G.P.; Field, C.B.; Ackerly, D.D. The velocity of climate change. Nature 2009, 462, 1052. [Google Scholar] [CrossRef] [PubMed]
- Millar, C.I.; Westfall, R.D.; Delany, D.L. Response of high-elevation limber pine (Pinus flexilis) to multiyear droughts and 20th-century warming, Sierra Nevada, California, USA. Can. J. Forest Res. 2007, 37, 2508–2520. [Google Scholar] [CrossRef]
- Millar, C.I.; Westfall, R.D.; Delany, D.L.; King, J.C.; Graumlich, L.J. Response of Subalpine Conifers in the Sierra Nevada, California, U.S.A., to 20th-Century Warming and Decadal Climate Variability. Arc. Antarct. Alp. Res. 2004, 36, 181–200. [Google Scholar] [CrossRef]
- LaMarche, V.C., Jr.; Mooney, H.A. Recent climatic change and development of the bristlecone pine (P. longaeva Bailey) krummholz zone, Mt. Washington Nevada. Arc. Alp. Res. 1972, 4, 61–72. [Google Scholar] [CrossRef]
| Snake Range | Sheep Range | |||
|---|---|---|---|---|
| Variable | Code | Unit | Subalpine West | Montane |
| Air temperature | AirT | °C | 10.4 (−12.8/25.7) | 18.8 (−5.8/36.6) |
| Soil temperature (50 cm) | SoilT | °C | 13.7 (−0.6/21.4) | 19.0 (8.6/23.6) |
| Soil volumetric water content (17 cm) | VWC17 | % | 7.0 (2.4/22.1) | 6.9 (4.4/13.9) |
| Soil volumetric water content (32 cm) | VWC32 | % | 8.8 (4.1/18.4) | 8.3 (6.6/10.2) |
| Dew point | Dew | °C | −2.6 (−34.6/8.9) | 0.7 (−25.1/17.0) |
| Vapor Pressure Deficit | VPD | hPa | 7.7 (0.2/21.7) | 15.5 (0.2/42.2) |
| Total precipitation | PPT | mm | 179 (0/9) | 171 (0/51) |
| Snake Range | Sheep Range | |||||
|---|---|---|---|---|---|---|
| Phase | Type | Feature | Pinus longaeva | Pinus flexilis | Pinus ponderosa | Pinus monophylla |
| Contraction | Diurnal | Onset | 5:20 ± 1 h 51 m | 4:29 ± 1 h 31 m | 6:46 ± 2 h 16 m | 5:40 ± 1 h 51 m |
| Duration | 11.3 ± 2.0 | 9.9 ± 1.6 | 10.8 ± 2.5 | 9.6 ± 2.2 | ||
| Magnitude | 85 ± 40 | 69 ± 28 | 35 ± 50 | 60 ± 44 | ||
| Expansion | Diurnal | Onset | 16:16 ± 1 h 36 m | 14:11 ± 1 h 33 m | 17:05 ± 2 h 23 m | 15:14 ± 1 h 30 m |
| Duration | 10.6 ± 2.3 | 10.2 ± 3.6 | 11.6 ± 3.0 | 12.6 ± 3.2 | ||
| Magnitude | 75 ± 39 | 59 ± 27 | 28 ± 13 | 48 ± 24 | ||
| Increment | Diurnal | Onset | 4:52 ± 7 h 22 m | 13:39 ± 9 h 01 m | 5:19 ± 5 h 07 m | 7:38 ±7 h 30 m |
| Duration | 3.5 ± 2.1 | 6.0 ± 3.4 | 3.1 ± 2.4 | 4.5 ± 4.4 | ||
| Magnitude | 14 ± 14 | 21 ± 18 | 13 ± 73 | 13 ± 22 | ||
| Whole cycle | Diurnal | Onset | 5:20 ± 1 h 51 m | 4:29 ± 1 h 31 m | 6:46 ± 2 h 16 m | 5:40 ± 1 h 51 m |
| Duration | 23.7 ± 1.9 | 23.8 ± 1.6 | 23.7 ± 1.9 | 23.8 ± 1.7 | ||
| Magnitude | 95 ± 43 | 87 ± 27 | 44 ± 69 | 72 ± 61 | ||
| Contraction | Long | Onset | 6:58 ± 4 h 59 m | 5:34 ± 4 h 40 m | 7:39 ± 3 h 26 m | 7:07 ± 4 h 41 m |
| Duration | 22.6 ± 14.3 | 14.9 ± 8.3 | 21.8 ± 19.2 | 19.8 ± 21.6 | ||
| Magnitude | 73 ± 69 | 61 ± 38 | 35 ± 24 | 48 ± 34 | ||
| Expansion | Long | Onset | 15:25 ± 3 h 08 m | 14:26 ± 1 h 59 m | 17:42 ± 2 h 45 m | 15:33 ± 1 h 47 m |
| Duration | 14.2 ± 11.6 | 15.3 ± 9.9 | 21.1 ± 19.4 | 23.7 ± 16.1 | ||
| Magnitude | 45 ± 36 | 51 ± 34 | 30 ± 18 | 39 ± 29 | ||
| Increment | Long | Onset | 9:09 ± 7 h 04 m | 12:33 ± 7 h 55 m | 11:14 ± 8 h 05 m | 11:58 ± 6 h 52 m |
| Duration | 24.4 ± 17.8 | 19.5 ± 14.0 | 25.4 ± 27.6 | 15.8 ± 15.6 | ||
| Magnitude | 71 ± 54 | 53 ± 44 | 70 ± 101 | 47 ± 56 | ||
| Whole cycle | Long | Onset | 7:05 ± 4 h 57 m | 5:24 ± 4 h 33 m | 7:43 ± 3 h 26 m | 7:10 ± 4 h 43 m |
| Duration | 52.1 ± 23.5 | 46.1 ± 16.6 | 57.7 ± 32.3 | 52.3 ± 27.8 | ||
| Magnitude | 122 ± 90 | 111 ± 56 | 84 ± 94 | 85 ± 63 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ziaco, E.; Biondi, F. Stem Circadian Phenology of Four Pine Species in Naturally Contrasting Climates from Sky-Island Forests of the Western USA. Forests 2018, 9, 396. https://doi.org/10.3390/f9070396
Ziaco E, Biondi F. Stem Circadian Phenology of Four Pine Species in Naturally Contrasting Climates from Sky-Island Forests of the Western USA. Forests. 2018; 9(7):396. https://doi.org/10.3390/f9070396
Chicago/Turabian StyleZiaco, Emanuele, and Franco Biondi. 2018. "Stem Circadian Phenology of Four Pine Species in Naturally Contrasting Climates from Sky-Island Forests of the Western USA" Forests 9, no. 7: 396. https://doi.org/10.3390/f9070396
APA StyleZiaco, E., & Biondi, F. (2018). Stem Circadian Phenology of Four Pine Species in Naturally Contrasting Climates from Sky-Island Forests of the Western USA. Forests, 9(7), 396. https://doi.org/10.3390/f9070396






