1. Introduction
The recent prolonged drought in Melbourne, Australia (1997–2010) has had a deleterious effect on many trees within the urban forest. Within the City of Melbourne (a municipality of 37.7 km
2 around the Melbourne CBD), prolonged drought and resulting mandatory changes to irrigation practices have led to a premature loss of vitality in many mature trees. Climate-related tree decline within the City of Melbourne was particularly evident in exotic deciduous trees, such as
Ulmus spp., which are widely planted throughout the City of Melbourne and are recognized as an important element of the city landscape [
1].
Tree vitality can be defined in relation to the plant response to physiological stress [
2]. Very low vitality trees will not respond to treatment to ameliorate physiological stress, but will remain in a depleted state or die, possibly because of extremely low carbohydrate reserves. High vitality trees, on the other hand, will respond and recover from drought or other physiological stressors.
Chlorophyll fluorescence measurements and in particular ratios of F
v/F
m are used to examine aspects of plant photosynthetic and photochemical processes that give rise to plant vitality [
3]. However, most chlorophyll fluorescence and many other physiological measurements of plant stress utilize leaf material, which precludes the year-round assessment of deciduous trees and can confound tree vitality with the health of individual leaves [
4]. Using chlorophyll fluorescence measurements in bark tissue to assess vitality, though possible, has seldom been used in practice [
4]. The most commonly used chlorophyll fluorescence measurement is F
v/F
m, where F
v is the difference between maximum (F
m) and minimum (F
0) fluorescence [
5]. F
v/F
m is the theoretical measure of the quantum efficiency of photosystem two (PSII) during photosynthesis if all the PSII reaction centers are open. A decrease in the chlorophyll fluorescence parameter F
v/F
m indicates photoinhibitory damage. F
v/F
m values can decrease significantly with salt, heat, and herbicide damage [
3], as well as with drought stress [
6]. Leaf F
v/F
m values between 0.78 to 0.85 are typical for healthy, non-stressed trees [
3]; however, bark values are typically lower, probably due to a decreased efficiency in bark chlorenchyms and an external bark layer that may inhibit the fluorescence signal passing to the measurement instrument [
4].
Visual methods for the assessment of tree vitality have been successfully applied in natural forest stands [
7,
8], hardwood plantations [
4], and urban environments [
9,
10]. A numerical crown assessment technique was developed for living
Eucalyptus (
Corymbia)
maculata (Hook.) K.D. Hill & L.A.S. Johnson (spotted gum),
Eucalyptus fibrosa F. Muell. (ironbark) and
E. drepanophylla F. Muell ex Benth (syn.
E. cerebra F. Muell) trees by Grimes [
7]. The method used crown position in relation to other trees, crown size, crown density, the number of dead branches, and epicormic growth and developed a prediction equation for how well these factors explained incremental growth at breast height. He found that each of the five variables contributed significantly to a prediction equation for incremental growth, but that for the best results, factors should be weighted differently, for example, epicormic growth on a three-point scale and crown density on a nine-point scale. Martin et al. [
8] used the above method by Grimes [
7] to develop a visual assessment method to examine the spatial representation of eucalypt dieback across two sites in a natural forest stand. At one site, they identified a positive relationship between vitality and tree size. Aiming to assess the vitality of drought stressed and declining mature trees, Johnstone et al. [
4] adapted the method described by Martin et al. [
8] for a study in plantation eucalypts and found correlations between visual vitality and total leaf area, above ground biomass, leaf chlorophyll fluorescence (summer), and trunk bark chlorophyll fluorescence (spring, summer, and autumn).
A more detailed discussion of methods to assess long-term drought and/or vitality in trees can be found in Johnstone et al. [
2]. In this paper, the authors discuss the relative merits of measuring tree vitality using tree growth and visual parameters; leaf or needle morphology and biochemistry; canopy transparency and reflectance; electrical admittance/impedance; gaseous exchange; and chlorophyll fluorescence.
We report here on how we have used an urban visual vitality index and bark chlorophyll fluorescence measured on large branches to predict drought stress in mature urban trees. Specifically, we tested if there was a relationship between the drought stress in Ulmus procera Salisb. (English Elm) and Ulmus × hollandica (Dutch Elm) and urban visual vitality index values and/or bark chlorophyll fluorescence measurements on large branches. Our results inform management decisions for trees in urban environments for the benefit of city dwellers and users by maximizing canopy cover for the successful provision of environmental services. The results also help tree managers provide measures for the successful establishment of succession planning for tree replacement.
2. Materials and Methods
For this study, trees were examined around the central business district of the City of Melbourne, Australia (Latitude 37°47′ S, longitude 144°58′ E). Melbourne has had an average annual rainfall of 648 mm and high summer temperatures with a January mean maximum of 26 degrees Celsius from 1855 to 2015 [
11]. The experimental trees consisted of six mature
Ulmus procera located in a suburban streetscape in a contiguous row and 32 randomly selected
Ulmus ×
hollandica located within inner city parks from two avenues. All trees displayed visual drought stress symptoms and displayed variation in crown condition from very stressed to not particularly stressed, to properly evaluate the relationship between drought stress and the two assessment methods.
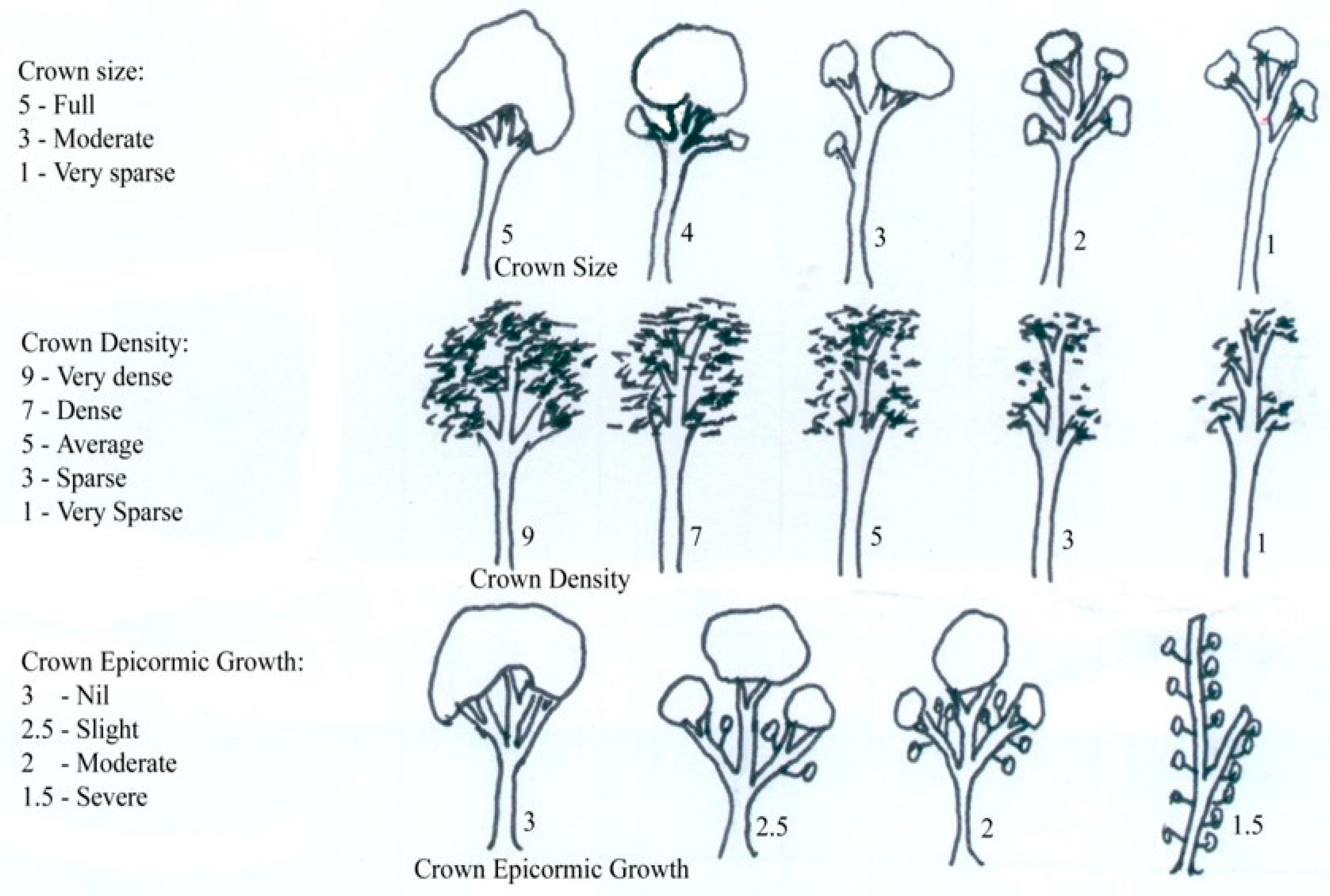
The urban visual vitality index used (
Figure 1) was adapted from a method described by Johnstone et al. [
4]. The urban visual vitality index had three criteria: crown size, crown density, and crown epicormic growth. Each of the criteria was scored numerically; 1 to 5 (crown size), 1–9 (crown density), and 1–3 (crown epicormic growth). The relative weighting of each parameter follows Grimes [
6] who, as previously stated, developed a predictive equation for how well each factor explained incremental growth at breast height. The score was totaled, with a higher score indicating greater vitality (
Figure 1). Urban visual vitality index assessments were undertaken on all trees in December 2010, January 2011, and February 2011. The lowest score in December was 3.5 and the highest was 14.5 out of a possible 17, confirming the range of tree crown conditions mentioned above.
Leaf water potential was measured in March 2011 by harvesting five fully expanded sun leaves from within the upper half of the crown of each tree. Leaves were collected using an Elevating Work Platform. For pre-dawn measurements, leaves were collected between 5:30 and 7:00 a.m., prior to direct sunlight reaching any part of the canopy on consecutive days with similar weather conditions. For midday measurements, leaves were collected between 12:00 p.m. and 1:30 p.m. also on consecutive days with similar weather conditions. Water potential readings were undertaken in a PMS Model 1000 pressure chamber (PMS instrument company, Albany, OR, USA) within 20 min of leaf harvesting. Leaf water potential was measured using the protocol first described by Scholander [
13].
Bark chlorophyll fluorescence was also measured in March 2011 with a Hansatech-Handy Plant Efficiency Analyzer (Handy PEA, Hansatech Instruments, King’s Lynn, Norfolk, UK). Bark chlorophyll fluorescence was measured by attaching a collar containing 10 apertures (modified leaf clips) as described in Johnstone et al. [
4]. The apertures were 35 mm apart and the branches were third order branches approximately 15 cm in diameter, accessed by an Elevating Work Platform. The apertures were also closed for 30 min until a steady state was achieved.
Once the darkening period was complete, a red light was then flashed (modulated) onto the bark surface with the Handy PEA and an increase in the yield of chlorophyll fluorescence occurred over 1 s (induction curve), until the PS II reaction centers progressively closed [
5]. Values for each tree were averaged for the 10 readings. Readings well outside a normal range were eliminated from the data set.
Simple linear regression analysis was undertaken with Minitab 17 software to identify relationships between assessment techniques and between species.
4. Discussion
This study aimed to test practical methods to predict tree stress, particularly long-term moisture stress. The average value for pre-dawn water potential in this study for elms was −1.0 MPa. This indicates that the elms were moderately stressed [
14,
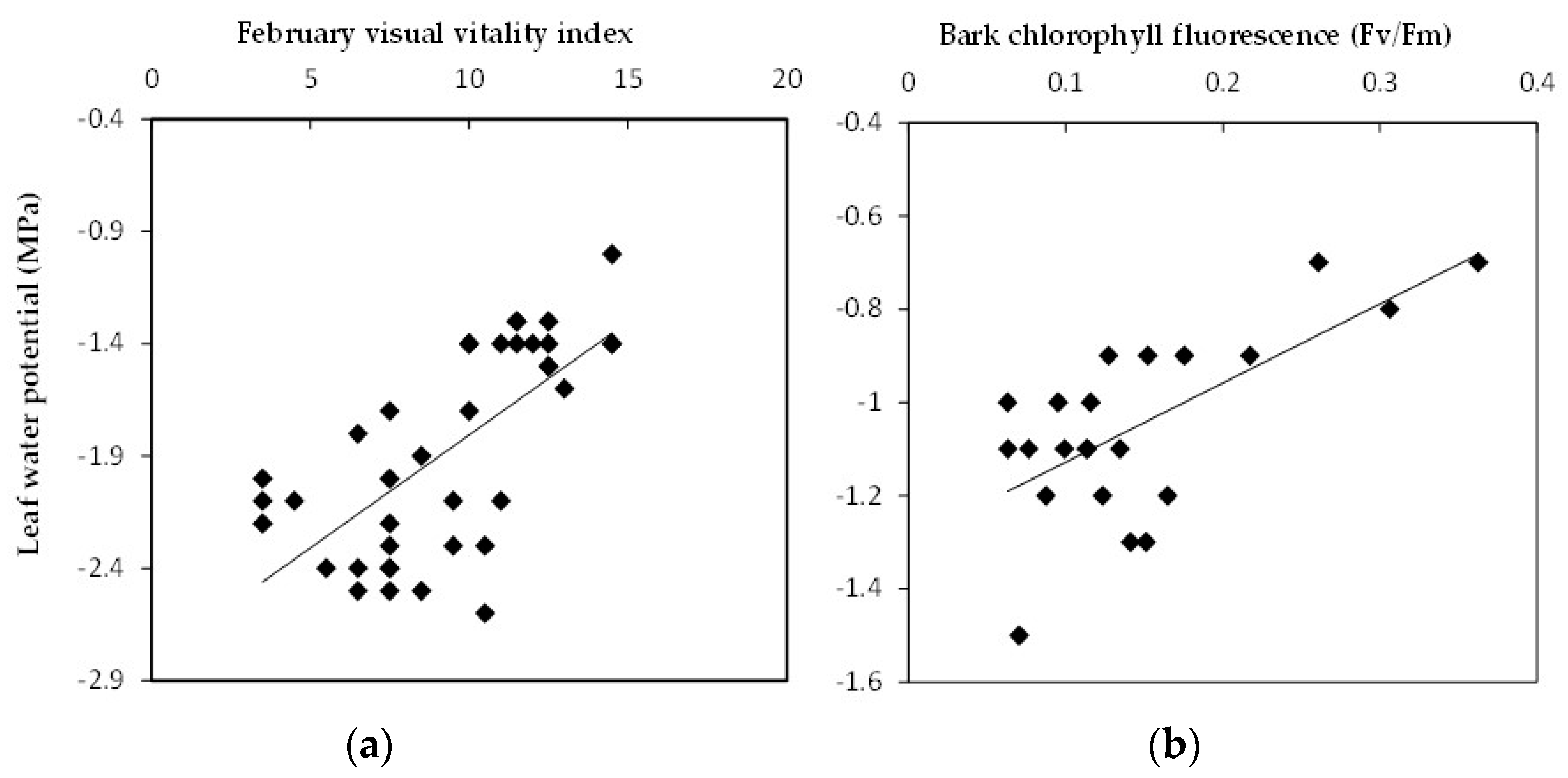
15]. However, most of the elms had been experiencing moderate to severe drought stress over a significant period, as evidenced by their low urban visual vitality indices. The average score for the elm urban visual vitality index was nine out of a possible 17. The regression analysis of the elm data indicates that the urban visual vitality index used in this study can be used to predict decreased midday leaf water potential in elm populations and in particular within the species
Ulmus × hollandica (
Figure 2a;
Table 1). There were, however, no statistical relationships observed between the February urban visual vitality index and midday leaf water potential in
Ulmus procera when the six trees of this species were analyzed separately (
Table 1). This can be explained by the very low variability in the February visual vitality and midday water potential data in
Ulmus procera (
Table 1). It is also a function of the very low number of samples for this species. The pooled Elm results suggest that the urban visual vitality index could be used as an indication of drought stress in mature elms in Summer through visual means and without the need for specialized equipment.
There were no statistical relationships observed between branch bark F
v/F
m fluorescence and pre-dawn leaf water potential in
Ulmus procera when the six trees of this species were analyzed separately (
Table 1). This can be explained by the very low variability visible in branch bark F
v/F
m fluorescence and pre-dawn leaf water potential data in
Ulmus procera (
Table 1). The lack of a relationship could again be a function of the very low number of samples for this species. But again, when all Elm results were pooled or
Ulmus × hollandica was examined separately, bark chlorophyll fluorescence testing on large branches could predict pre-dawn water status, suggesting that this technique could also be used to assess drought stress in mature elms (
Figure 2b;
Table 1). Branch bark chlorophyll fluorescence would theoretically enable the year-round testing of deciduous trees, unlike techniques that require leaf samples. Evidence from a previous study on
Eucalyptus globulus suggests that bark chlorophyll fluorescence may correlate with longer term vitality, whereas leaf chlorophyll fluorescence may be dependent on the health of individual leaves [
4].
Under existing and predicted impacts of climate change, it is likely that globally, trees within many urban environments will be subject to altered growing conditions [
16]. The ability of trees to adapt to these changing environments will vary; however, trees that have low vitality or that are nearing the end of their lifespan are likely to be less tolerant to change. Identifying suitable assessment techniques for monitoring the vitality of drought stressed mature urban trees will provide tree managers with additional resources for maintaining valuable mature tree resources.