Water Level Controls on Sap Flux of Canopy Species in Black Ash Wetlands
Abstract
:1. Introduction
2. Materials and Methods
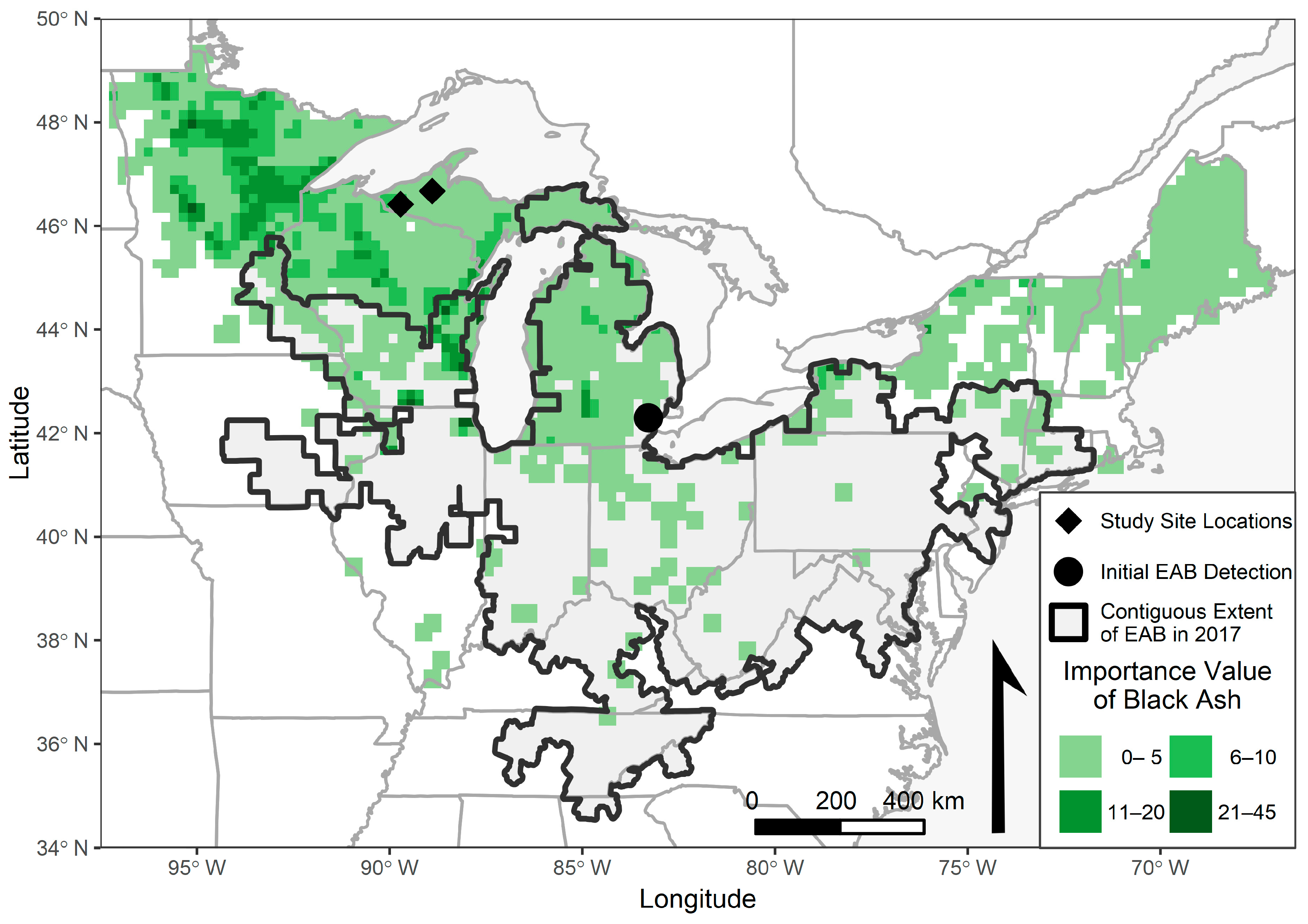
2.1. Study Area
2.2. Field Measures
2.3. Data Analysis
3. Results
3.1. Stem Elevations and Water Levels
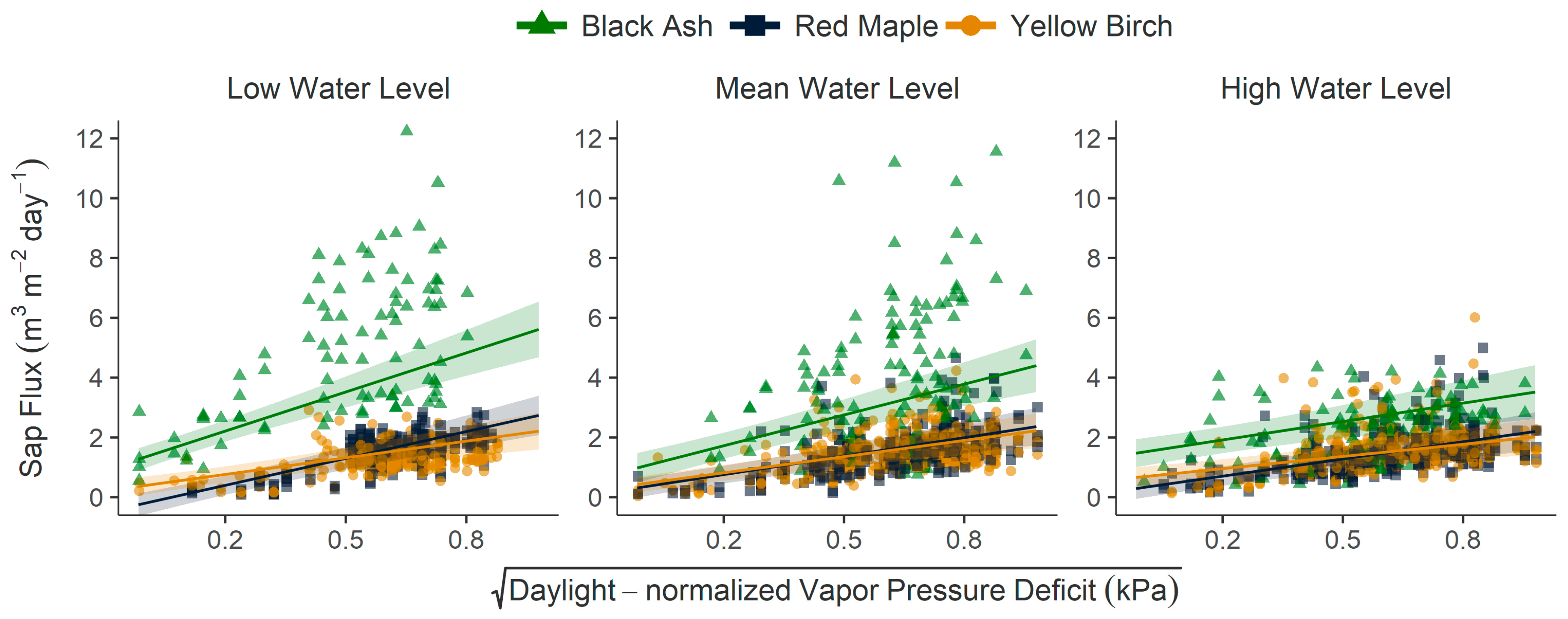
3.2. Mean Sap Flux Rates, Individual Drivers, and Interaction of Drivers
4. Discussion
4.1. Effects of Water Level on Sap Flux Rates and Response to Atmospheric Drivers
4.2. Persistence of Hydrologic Change
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Haack, R.; Jendek, E.; Liu, H.; Marchant, K. The emerald ash borer: A new exotic pest in North America. Newsl. Mich. Entomol. Soc. 2002, 47, 1–5. [Google Scholar] [CrossRef]
- Herms, D.A.; McCullough, D.G. Emerald Ash Borer Invasion of North America: History, Biology, Ecology, Impacts, and Management. Annu. Rev. Entomol. 2014, 59, 13–30. [Google Scholar] [CrossRef] [PubMed]
- Pugh, S.A.; Liebhold, A.M.; Morin, R.S. Changes in ash tree demography associated with emerald ash borer invasion, indicated by regional forest inventory data from the Great Lakes States. Can. J. For. Res. 2011, 41, 2165–2175. [Google Scholar] [CrossRef]
- Aukema, J.E.; Leung, B.; Kovacs, K.; Chivers, C.; Britton, K.O.; Englin, J.; Frankel, S.J.; Haight, R.G.; Holmes, T.P.; Liebhold, A.M.; et al. Economic impacts of Non-Native forest insects in the continental United States. PLOS ONE 2011, 6, e24587. [Google Scholar] [CrossRef] [PubMed]
- Willow, A.J. Indigenizing Invasive Species Management: Native North Americans and the Emerald Ash Borer (EAB) Beetle. Cult. Agric. Food Environ. 2011, 33, 70–82. [Google Scholar] [CrossRef]
- Gandhi, K.J.K.; Herms, D.A. Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern North America. Biol. Invasions 2010, 12, 389–405. [Google Scholar] [CrossRef]
- Prasad, A.M.; Iverson, L.R. Little’s Range and FIA Importance Value Database for 135 Eastern US Tree Species; Northeastern Research Station, USDA Forest Service: Delaware, OH, USA, 2003; Available online: http://www.fs.fed.us/ne/delaware/4153/global/littlefia/index.html (accessed on 22 April 2016).
- Burns, R.M.; Honkala, B.H. Silvics of North America; Agriculture Handbook 654; Volume 2: Hardwoods; United States Department of Agriculture (USDA), Forest Service: Washington, DC, USA, 1990.
- Kost, M.A.; Albert, D.A.; Cohen, J.G.; Slaughter, B.S.; Schillo, R.K.; Weber, C.R.; Chapman, K.A. Natural Communities of Michigan: Classification and Description; Michigan Natural Features Inventory: Lansing, MI, USA, 2007. [Google Scholar]
- Epstein, E.; Judziewicz, E.; Spencer, E. Wisconsin Natural Heritage Inventory (NHI) Recognized Natural Communities—Working Document; Wisconsin Natural Heritage Inventory: Madison, WI, USA, 2002. [Google Scholar]
- Minnesota Department of Natural Resources. Field Guide to the Native Plant Communities of Minnesota: The Prairie Parkland and Tallgrass Aspen Parklands Provinces; Ecological Land Classification Program, Minnesota County Biological Survey, Natural Heritage, Nongame Research Program: St. Paul, MN, USA, 2005.
- Davis, J.C.; Shannon, J.P.; Bolton, N.W.; Kolka, R.K.; Pypker, T.G. Vegetation responses to simulated emerald ash borer infestation in Fraxinus-nigra dominated wetlands of Upper Michigan, USA. Can. J. For. Res. 2017, 47, 319–330. [Google Scholar] [CrossRef]
- Looney, C.E.; D’Amato, A.W.; Palik, B.J.; Slesak, R.A.; Slater, M.A. The response of Fraxinus nigra forest ground-layer vegetation to emulated emerald ash borer mortality and management strategies in northern Minnesota, USA. For. Ecol. Manag. 2017, 389, 352–363. [Google Scholar] [CrossRef]
- Slesak, R.A.; Lenhart, C.F.; Brooks, K.N.; D’Amato, A.W.; Palik, B.J. Water table response to harvesting and simulated emerald ash borer mortality in black ash wetlands in Minnesota, USA. Can. J. For. Res. 2014, 44, 961–968. [Google Scholar] [CrossRef]
- Van Grinsven, M.J.; Shannon, J.P.; Davis, J.C.; Bolton, N.W.; Wagenbrenner, J.W.; Kolka, R.K.; Pypker, T.G. Source water contributions and hydrologic responses to simulated emerald ash borer infestations in depressional black ash wetlands. Ecohydrology 2017, 10, e1862. [Google Scholar] [CrossRef]
- Looney, C.E.; D’Amato, A.W.; Palik, B.J.; Slesak, R.A. Overstory treatment and planting season affect survival of replacement tree species in emerald ash borer threatened Fraxinus nigra forests in Minnesota, USA. Can. J. For. Res. 2015, 45, 1728–1738. [Google Scholar] [CrossRef]
- Looney, C.E.; Amato, A.W.D.; Palik, B.J.; Slesak, R.A. Canopy treatment influences growth of replacement tree species in Fraxinus nigra forests threatened by the emerald ash borer in Minnesota, USA. Can. J. For. Res. 2017, 192, 183–192. [Google Scholar] [CrossRef]
- Bolton, N.; Shannon, J.; Davis, J.; Van Grinsven, M.; Noh, N.J.; Schooler, S.; Kolka, R.; Pypker, T.; Wagenbrenner, J. Controls on alternative species seedlings survival and growth in black ash ecosystems threatened by emerald ash borer. Forests 2018. in review. [Google Scholar]
- Noh, N.J.; Shannon, J.P.; Bolton, N.W.; Davis, J.C.; Van Grinsven, M.J.; Pypker, T.G.; Kolka, R.K.; Wagenbrenner, J.W. Carbon dioxide fluxes from coarse dead wood in a black ash wetland. Forests 2018. in review. [Google Scholar]
- Van Grinsven, M.; Shannon, J.; Bolton, N.; Davis, J.; Wagenbrenner, J.; Kolka, R.; Pypker, T. Gaseous soil-carbon flux responses to simulated emerald ash borer infestations in depressional black ash wetlands. Forests 2018. in review. [Google Scholar]
- Brinson, M.M. A Hydrogeomorphic Classification for Wetlands; Wetlands Research Program Technical Report WRP-DE-4; East Carolina University: Greenville, NC, USA, 1993. [Google Scholar] [CrossRef]
- Sanford, W.E.; Selnick, D.L. Estimation of Evapotranspiration Across the Conterminous United States Using a Regression With Climate and Land-Cover Data. JAWRA J. Am. Water Resour. Assoc. 2013, 49, 217–230. [Google Scholar] [CrossRef]
- Oren, R.; Phillips, N.; Ewers, B.E.; Pataki, D.E.; Megonigal, J.P. Sap-flux-scaled transpiration responses to light, vapor pressure deficit, and leaf area reduction in a flooded Taxodium distichum forest. Tree Physiol. 1999, 19, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.; McNulty, S.G.; Shepard, J.P.; Amatya, D.M.; Riekerk, H.; Comerford, N.B.; Skaggs, W.; Swift, L. Effects of timber management on the hydrology of wetland forests in the southern United States. For. Ecol. Manag. 2001, 143, 227–236. [Google Scholar] [CrossRef]
- Sebestyen, S.D.; Verry, E.S.; Brooks, K.N. Hydrological responses to changes in forest cover on uplands and peatlands. In Peatland biogeochemistry and watershed hydrology at the Marcell Experimental Forest; Kolka, R.K., Sebestyen, S.D., Verry, E.S., Brooks, K.N., Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 401–432. [Google Scholar]
- EDDMapS Early Detection & Distribution Mapping System; The University of Georgia—Center for Invasive Species and Ecosystem Health: Athens, GA, USA, 2017.
- Kozlowski, T.T.; Pallardy, S.G. Acclimation and Adaptive Responses of Woody Plants to Environmental Stresses Acclimation and Adaptive Responses of Woody Plants to Environmental Stresses. Bot. Rev. 2014, 68, 270–334. [Google Scholar] [CrossRef]
- Tardif, J.; Dery, S.; Bergeron, Y. Sexual Regeneration of Black Ash (Fraxinus nigra Marsh.) in a Boreal Floodplain. Am. Midl. Nat. 1994, 132, 124–135. [Google Scholar] [CrossRef]
- Kozlowski, T.T.; Pallardy, S.G. Stomatal Responses of Fraxinus pennsylvanica Seedlings during and after Flooding. Physiol. Plant. 1979, 46, 155–158. [Google Scholar] [CrossRef]
- Gomes, A.R.S.; Kozlowski, T.T. Growth responses and adaptations of Fraxinus pennsylvanica seedlings to flooding. Plant Physiol. 1980, 66, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Will, R.E.; Seiler, J.R.; Feret, P.P.; Aust, W.M. Effects of Rhizosphere Inundation on the Growth and Physiology of Wet and Dry-Site Acer-Rubrum (Red Maple) Populations. Am. Midl. Nat. 1995, 134, 127–139. [Google Scholar] [CrossRef]
- Pezeshki, S.R.; Chambers, J.L. Stomatal and photosynthetic response of sweet gum (Liquidambar styraciflua) to flooding. Can. J. For. Res. 1985, 15, 371–375. [Google Scholar] [CrossRef]
- Reece, C.F.; Riha, S.J. Role of root systems of eastern larch and white spruce in response to flooding. Plant Cell Environ. 1991, 14, 229–234. [Google Scholar] [CrossRef]
- Angeles, G.; Evert, R.F.; Kozlowski, T.T. Development of lenticels and adventitious roots in flooded Ulmus americana seedlings. Can. J. For. Res. 1986, 16, 585–590. [Google Scholar] [CrossRef]
- Angelov, M.N.; Sung, S.-J.J.S.; Doong, R.L.; Harms, W.R.; Kormanik, P.P.; Black, C.C.; Black, C.C., Jr. Long- and short-term flooding effects on survival and sink-source relationships of swamp-adapted tree species. Tree Physiol. 1996, 16, 477–484. [Google Scholar] [CrossRef] [PubMed]
- McJannet, D. Water table and transpiration dynamics in a seasonally inundated Melaleuca quinquenervia forest, north Queensland, Australia. Hydrol. Process. 2008, 22, 3079–3090. [Google Scholar] [CrossRef]
- Arguze, A.; Durre, I.; Applequist, S.; Squires, M.; Vose, R.; Yin, X.; Bilotta, R. NOAA’s 1981–2010 U.S. Climate Normals: An Overview. Bull. Am. Meteorol. Soc. 2012, 93, 1687–1697. [Google Scholar]
- Granier, A. Une nouvelle methode pour la mesure du flux de seve brute dans le tronc des arbres. Ann. Sci. Forestieres (Fr.) 1985, 42, 193–200. [Google Scholar] [CrossRef]
- Granier, A. Evaluation of transpiration in a Douglas-fir stand by means of sap flow measurements. Tree Physiol. 1987, 3, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.W.; Bond, B.J.; Jones, J.A.; Meinzer, F.C. Thermal-dissipation sap flow sensors may not yield consistent sap-flux estimates over multiple years. Trees 2009, 24, 165–174. [Google Scholar] [CrossRef]
- Lu, P.; Urban, L.; Zhao, P. Granier’s thermal dissipation probe (TDP) method for measuring sap flow in trees: Theory and practice. Acta Bot. Sin. 2004, 46, 631–646. [Google Scholar]
- Tang, J.; Bolstad, P.V.; Ewers, B.E.; Desai, A.R.; Davis, K.J.; Carey, E.V. Sap flux-upscaled canopy transpiration, stomatal conductance, and water use efficiency in an old growth forest in the Great Lakes region of the United States. J. Geophys. Res. Biogeosci. 2006, 111, 1–12. [Google Scholar] [CrossRef]
- Bovard, B.D.; Curtis, P.S.; Vogel, C.S.; Schmid, H.P. Environmental controls on sap flow in a northern hardwood forest. Tree Physiol. 2005, 25, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Western Regional Climate Center. RAWS USA Climate Archive; Desert Research Institute: Reno, NV, USA, 2013.
- Horel, J.; Splitt, M.; Dunn, L.; Pechmann, J.; White, B.; Ciliberti, C.; Lazarus, S.; Slmmer, J.; Zaff, D.; Burks, J. Mesowest: Cooperative Mesonets in the Western United States. Bull. Am. Meteorol. Soc. 2002, 83, 211–225. [Google Scholar] [CrossRef]
- Oren, R.; Zimmermann, R.; Terbough, J. Transpiration in upper Amazonia floodplain and upland forests in response to drought-breaking rains. Ecology 1996, 77, 968–973. [Google Scholar] [CrossRef]
- Phillips, N.; Oren, R. Intra- and Inter-Annual Variations in Transpiration of a Pine Forest. Ecol. Appl. 2001, 11, 385–396. [Google Scholar] [CrossRef]
- Herbst, M.; Rosier, P.T.W.; Roberts, J.M.; Taylor, M.E.; Gowing, D.J. Edge effects and forest water use: A field study in a mixed deciduous woodland. For. Ecol. Manag. 2007, 176–186. [Google Scholar] [CrossRef]
- Lu, P. A direct method for estimating the average sap flux density using a modified Granier measuring system. Aust. J. Plant Physiol. 1997, 24, 701–705. [Google Scholar] [CrossRef]
- Clearwater, M.J.; Meinzer, F.C.; Andrade, J.L.; Goldstein, G.; Holbrook, N.M. Potential errors in measurement of nonuniform sap flow using heat dissipation probes. Tree Physiol. 1999, 681–688. [Google Scholar] [CrossRef]
- Lechowicz, M.J. Why Do Temperate Deciduous Trees Leaf Out at Different Times? Adaptation and Ecology of Forest Communities. Am. Nat. 1984, 124, 821–842. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing, Version 3.4.3; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- RStudio Team. RStudio: Integrated Development Environment for R; RStudio, Inc.: Boston, MA, USA, 2012. [Google Scholar]
- Lenth, R.V. emmeans: Estimated Marginal Means, Aka Least-Squares Means. R Package Version 1.1.2. 2018. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 22 April 2017).
- Kamil, B. MuMIn: Multi-Model Inference. R Package Version 1.40.4 2018. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 22 April 2017).
- Anella, L.B.; Whitlow, T.H. Photosynthetic Response to Flooding of Acer rubrum Seedlings from Wet and Dry Sites. Am. Midl. Nat. 2000, 143, 330–341. [Google Scholar] [CrossRef]
- Yan, X.L.; Xi, B.Y.; Jia, L.M.; Li, G.D. Response of sap flow to flooding in plantations of irrigated and non-irrigated triploid poplar. J. For. Res. 2015, 20, 375–385. [Google Scholar] [CrossRef]
- Wang, S.; Callaway, R.M.; Zhou, D.W.; Weiner, J. Experience of inundation or drought alters the responses of plants to subsequent water conditions. J. Ecol. 2017, 105, 176–187. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Rust, W.G. Tree Growth Responses to Flooding in a Bottomland in Northeastern Illinois. For. Sci. 1984, 30, 499–510. [Google Scholar]
- Duberstein, J.A.; Krauss, K.W.; Conner, W.H.; Bridges, W.C.; Shelburne, V.B. Do hummocks provide a physiological advantage to even the most flood tolerant of tidal freshwater trees? Wetlands 2013, 33, 399–408. [Google Scholar] [CrossRef]
- Burns, R.M.; Honkala, B.H. (Eds.) Silvics of North America; Agriculture Handbook 654; Volume 1: Conifers; United States Department of Agriculture (USDA), Forest Service: Washington, DC, USA, 1990.
- Heim, M. Personal communication, 2017.
- Ewers, B.E.; Mackay, D.S.; Gower, S.T.; Ahl, D.E.; Burrows, S.N.; Samanta, S.S. Tree species effects on stand transpiration in northern Wisconsin. Water Resour. Res. 2002, 38, 1–11. [Google Scholar] [CrossRef]
- Adelman, J.D.; Ewers, B.E.; MacKay, D.S. Use of temporal patterns in vapor pressure deficit to explain spatial autocorrelation dynamics in tree transpiration. Tree Physiol. 2008, 28, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Oren, R.; Pataki, D.E. Transpiration in response to variation in microclimate and soil moisture in southeastern deciduous forests. Oecologia 2001, 127, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Ford, C.R.; Goranson, C.E.; Mitchell, R.J.; Will, R.E.; Teskey, R.O. Modeling canopy transpiration using time series analysis: A case study illustrating the effect of soil moisture deficit on Pinus taeda. Agric. For. Meteorol. 2005, 130, 163–175. [Google Scholar] [CrossRef]
- McLaren, J.D.; Arain, M.A.; Khomik, M.; Peichl, M.; Brodeur, J. Water flux components and soil water-atmospheric controls in a temperate pine forest growing in a well-drained sandy soil. J. Geophys. Res. Biogeosci. 2008, 113. [Google Scholar] [CrossRef]
- Traver, E.; Ewers, B.E.; Mackay, D.S.; Loranty, M.M. Tree transpiration varies spatially in response to atmospheric but not edaphic conditions. Funct. Ecol. 2010, 24, 273–282. [Google Scholar] [CrossRef]
- Pezeshki, S.R.; Anderson, P.H. Responses of three bottomland species with different flood tolerance capabilities to various flooding regimes. Wetl. Ecol. Manag. 1996, 4, 245–256. [Google Scholar] [CrossRef]
- Telander, A.C.; Slesak, R.A.; D’Amato, A.W.; Palik, B.J.; Brooks, K.N.; Lenhart, C.F. Sap flow of black ash in wetland forests of northern Minnesota, USA: Hydrologic implications of tree mortality due to emerald ash borer. Agric. For. Meteorol. 2015, 206, 4–11. [Google Scholar] [CrossRef]
- Shannon, J.; Van Grinsven, M. Sap Flux and Water Levels for Black Ash Wetlands in western Michigan, USA from 2012 to 2014. Knowl. Netw. Biocomplex. 2018. [Google Scholar] [CrossRef]
| Species | Number of Instrumented Trees | Mean Diameter (cm) | Range of Diameters (cm) |
|---|---|---|---|
| Black Ash | 6 | 25.58 | 15.00, 40.00 |
| Red Maple | 5 | 23.12 | 18.00, 34.10 |
| Yellow Birch | 6 | 24.62 | 13.00, 39.10 |
| Species | Minimum Observed (cm) | Low/Mean Threshold (cm) | Mean Observed (cm) | Mean/High Threshold (cm) | Maximum Observed (cm) |
|---|---|---|---|---|---|
| Black Ash | −82.6 | −46.5 | −37.3 | −28.0 | 1.6 |
| Red Maple | −122.1 | −61.8 | −51.7 | −41.7 | −21.2 |
| Yellow Birch | −127.0 | −62.0 | −51.9 | −41.8 | −18.5 |
| Species | Low Water Level | Mean Water Level | High Water Level |
|---|---|---|---|
| Black Ash | |||
| Red Maple | |||
| Yellow Birch |
| Species | Low Water Level | Mean Water Level | High Water Level |
|---|---|---|---|
| Black Ash | |||
| Red Maple | |||
| Yellow Birch |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shannon, J.; Van Grinsven, M.; Davis, J.; Bolton, N.; Noh, N.J.; Pypker, T.; Kolka, R. Water Level Controls on Sap Flux of Canopy Species in Black Ash Wetlands. Forests 2018, 9, 147. https://doi.org/10.3390/f9030147
Shannon J, Van Grinsven M, Davis J, Bolton N, Noh NJ, Pypker T, Kolka R. Water Level Controls on Sap Flux of Canopy Species in Black Ash Wetlands. Forests. 2018; 9(3):147. https://doi.org/10.3390/f9030147
Chicago/Turabian StyleShannon, Joseph, Matthew Van Grinsven, Joshua Davis, Nicholas Bolton, Nam Jin Noh, Thomas Pypker, and Randall Kolka. 2018. "Water Level Controls on Sap Flux of Canopy Species in Black Ash Wetlands" Forests 9, no. 3: 147. https://doi.org/10.3390/f9030147
APA StyleShannon, J., Van Grinsven, M., Davis, J., Bolton, N., Noh, N. J., Pypker, T., & Kolka, R. (2018). Water Level Controls on Sap Flux of Canopy Species in Black Ash Wetlands. Forests, 9(3), 147. https://doi.org/10.3390/f9030147







