Recent Trends in Large Hardwoods in the Pacific Northwest, USA
Abstract
1. Introduction
1.1. Background on the Potential for Hardwood Declines
1.2. Need for Current Trend Analysis
1.3. Objectives and Scope
2. Materials and Methods
2.1. Literature Review to Select Indicators
2.1.1. Species Distribution
2.1.2. Indicators for Fruit Production
2.1.3. Indicators for Wildlife Habitat
2.1.4. Tolerance of Shade, Fire, and Phytophthora ramorum
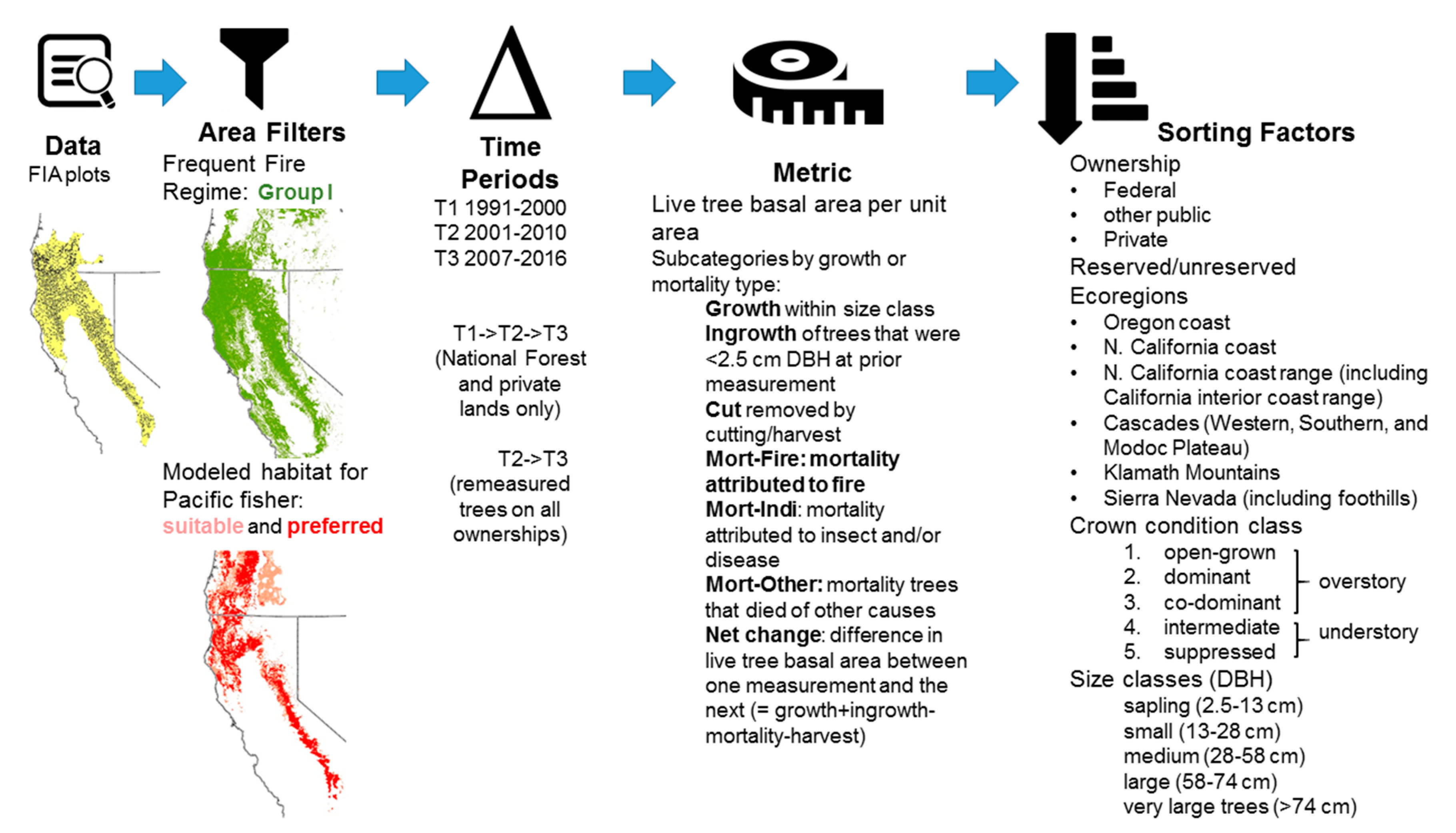
2.2. Overview of Forest Inventory Data
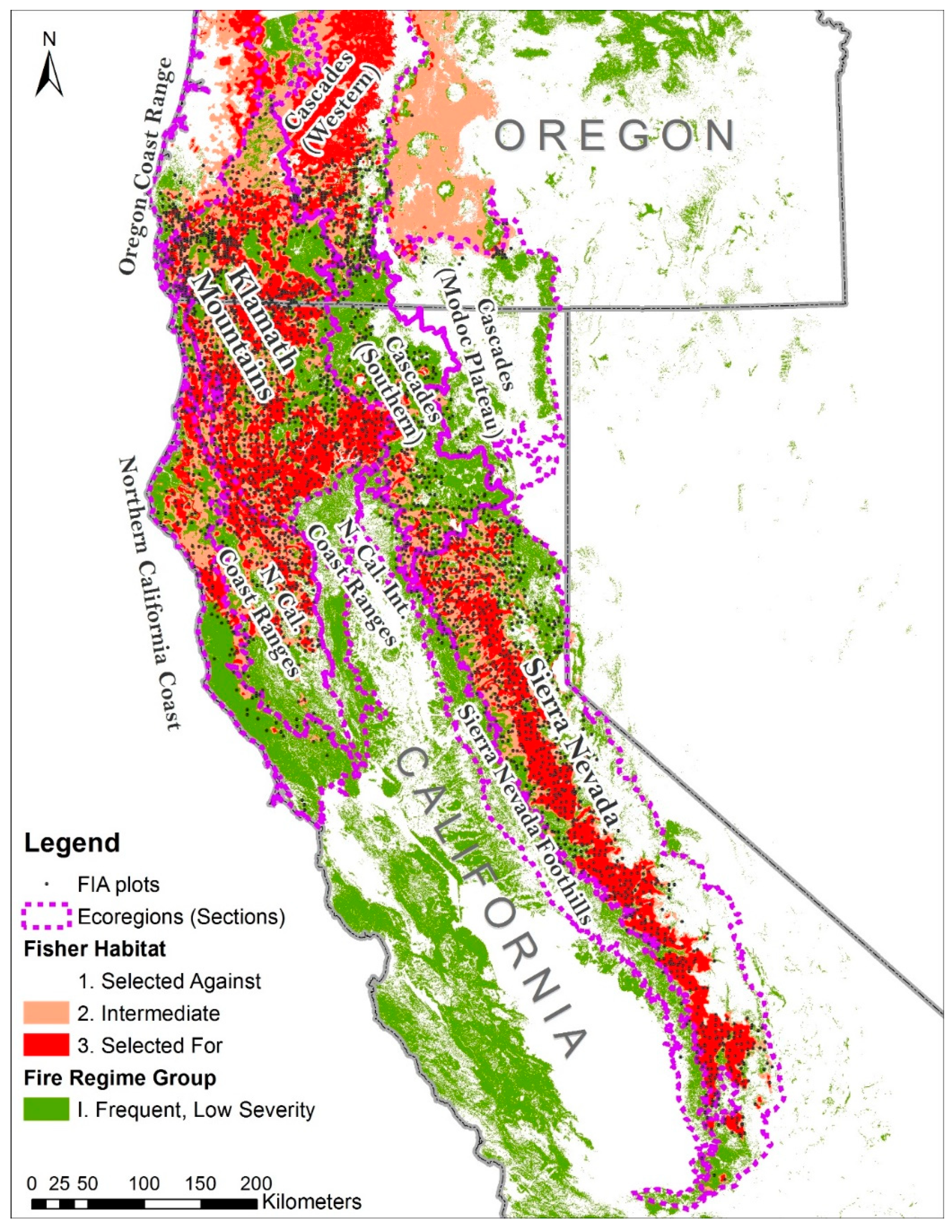
2.3. Study Area
2.4. Measurement Periods
- the FIA periodic inventory of non-National Forest System (NFS) lands in California (1991–1994; mean 1992),
- the NFS inventory in California (1992 to 2000; mean 1998),
- the FIA periodic inventory of non-NFS lands in Oregon (1995–1998; mean 1997), and,
- the NFS inventory of Oregon (1993–1997; mean 1995).
2.5. Metrics and Hypotheses
3. Results
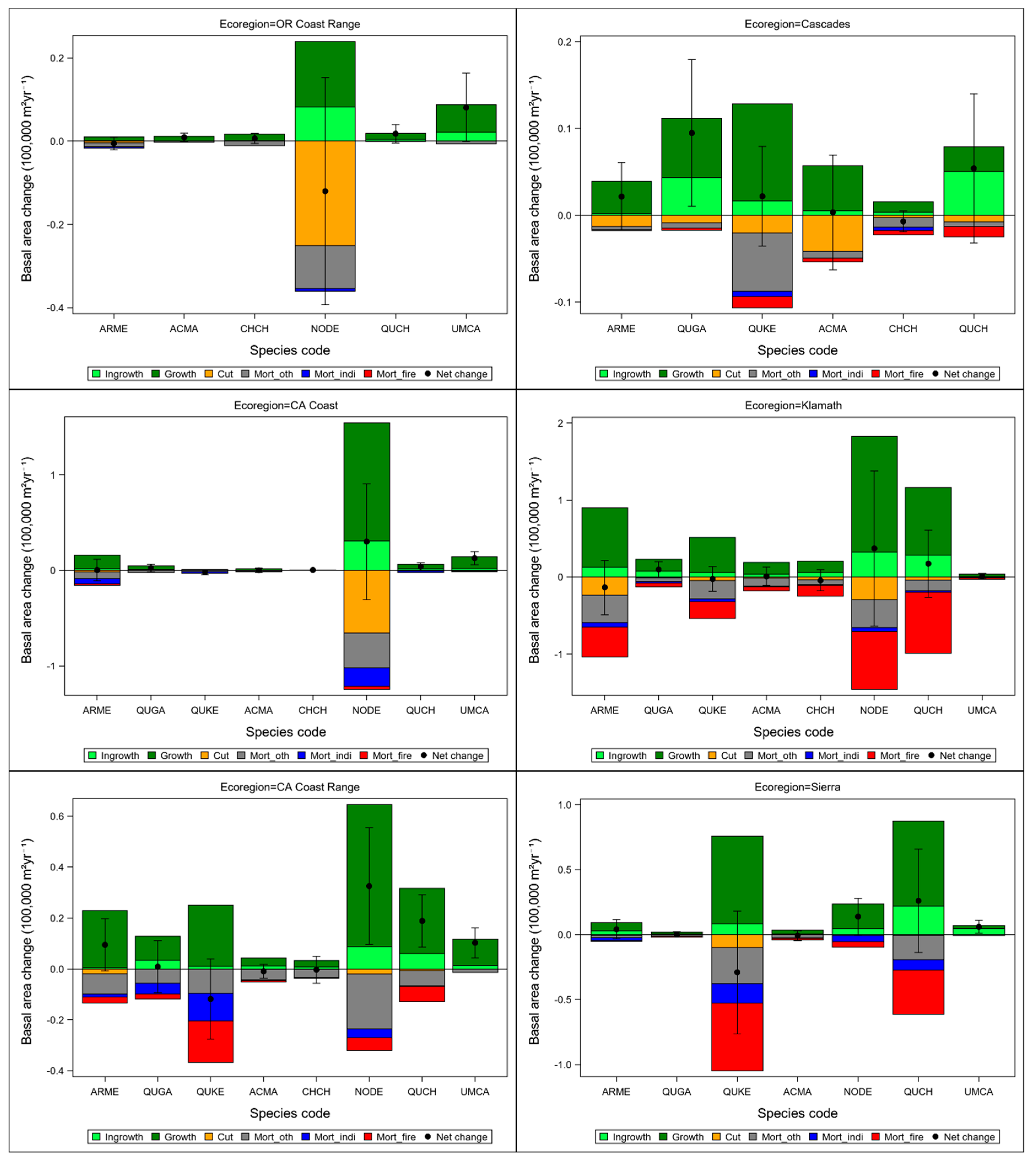
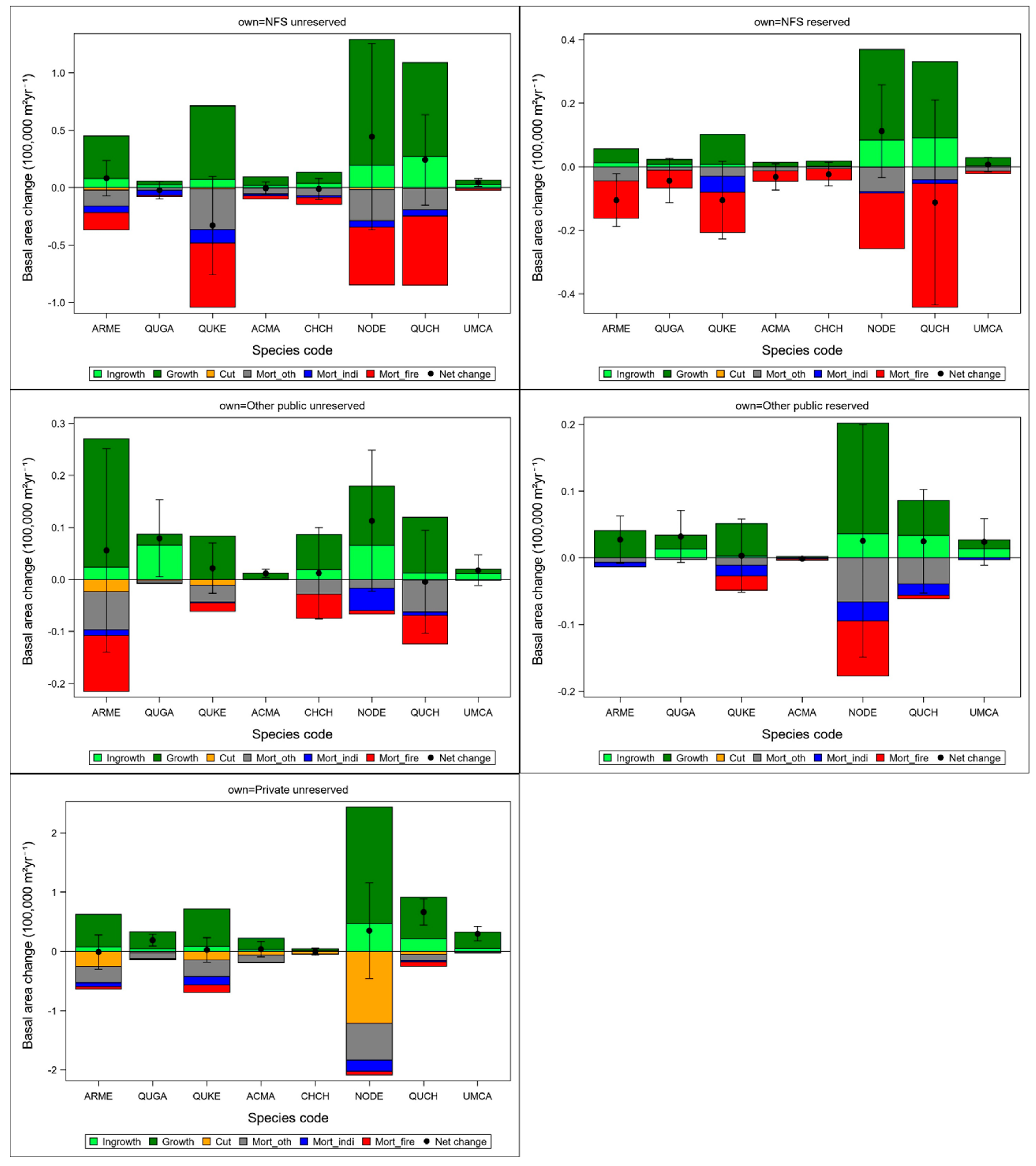
3.1. Distribution of Basal Area and Rates of Disturbances across Ecoregions and Ownership Areas
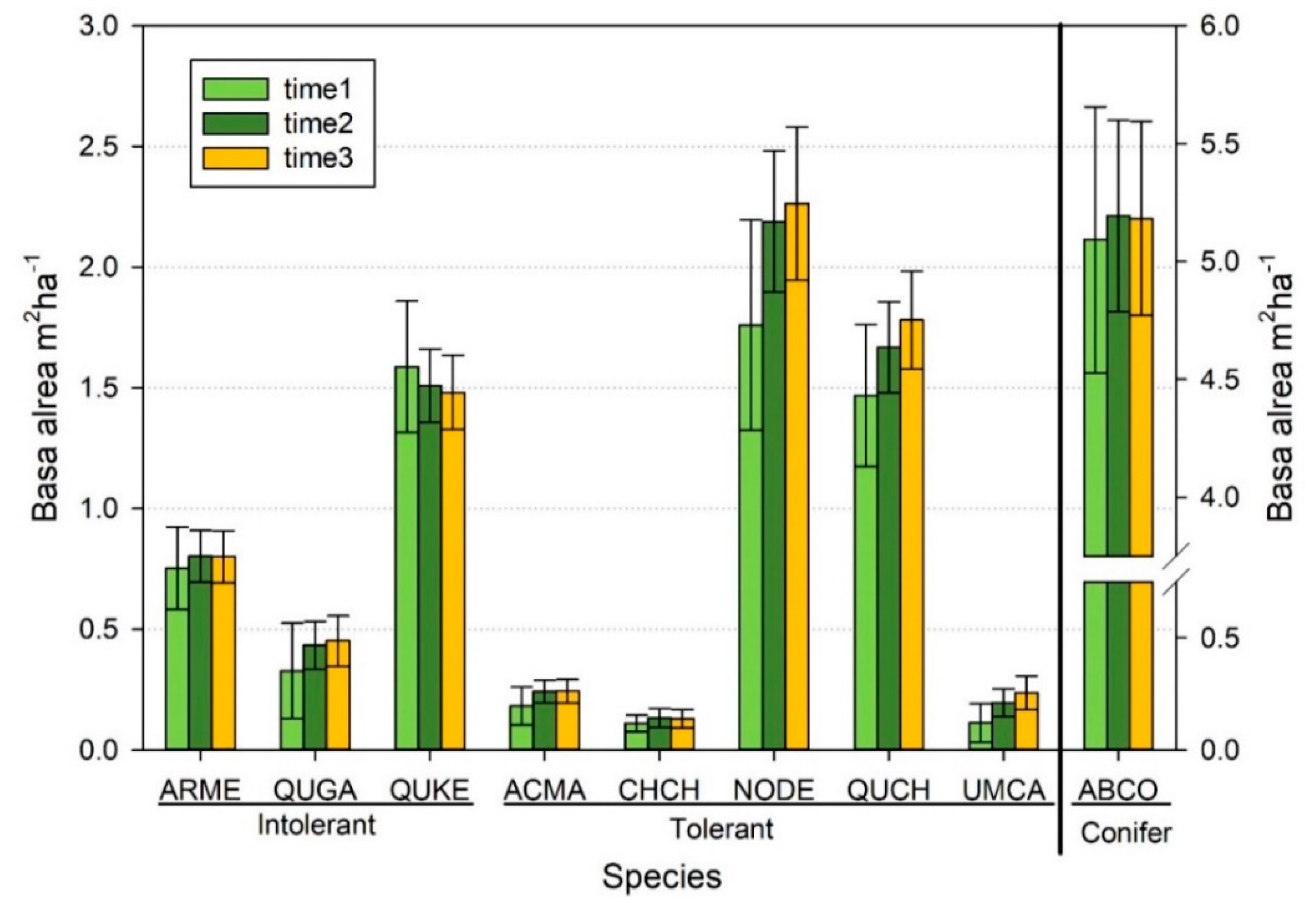
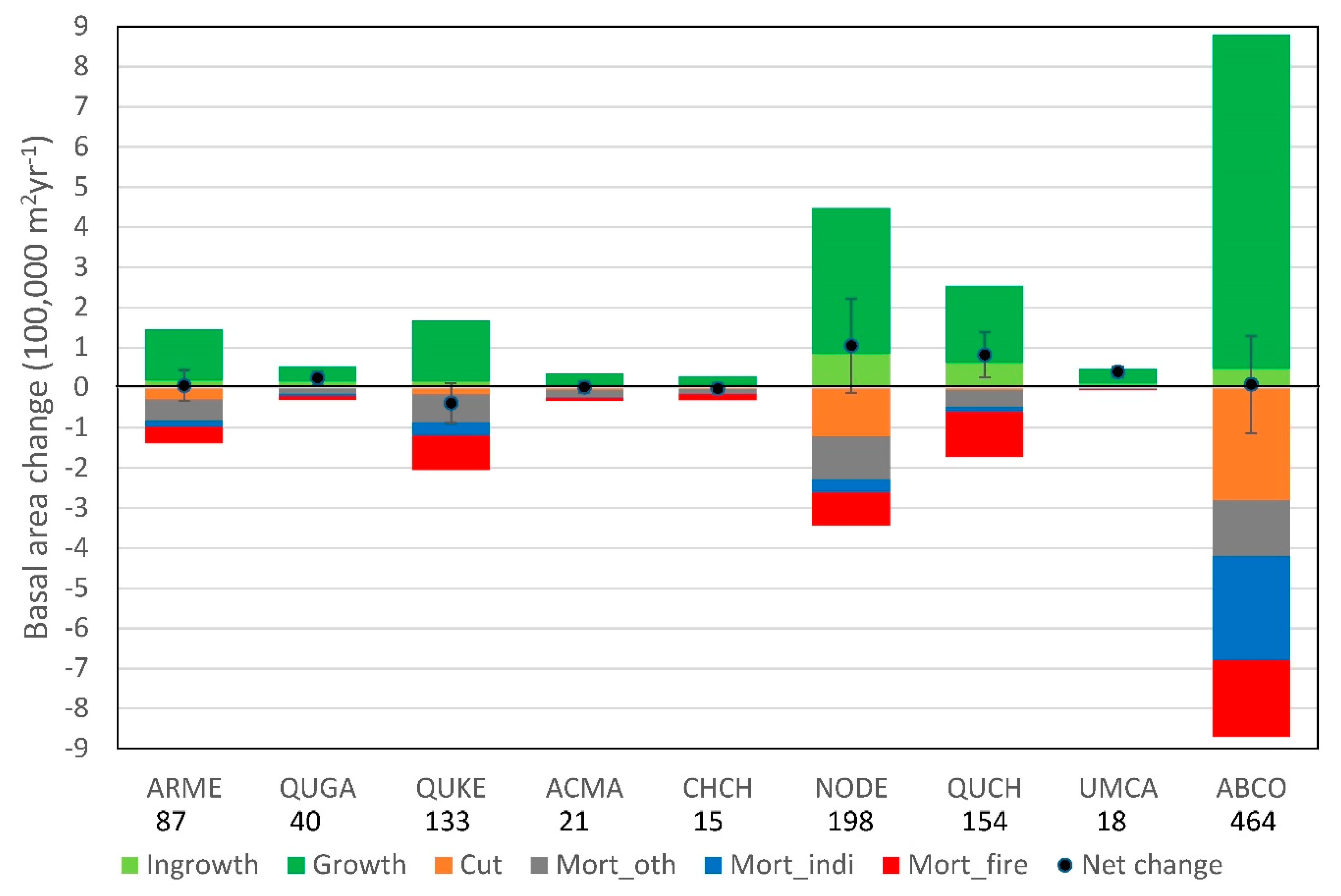
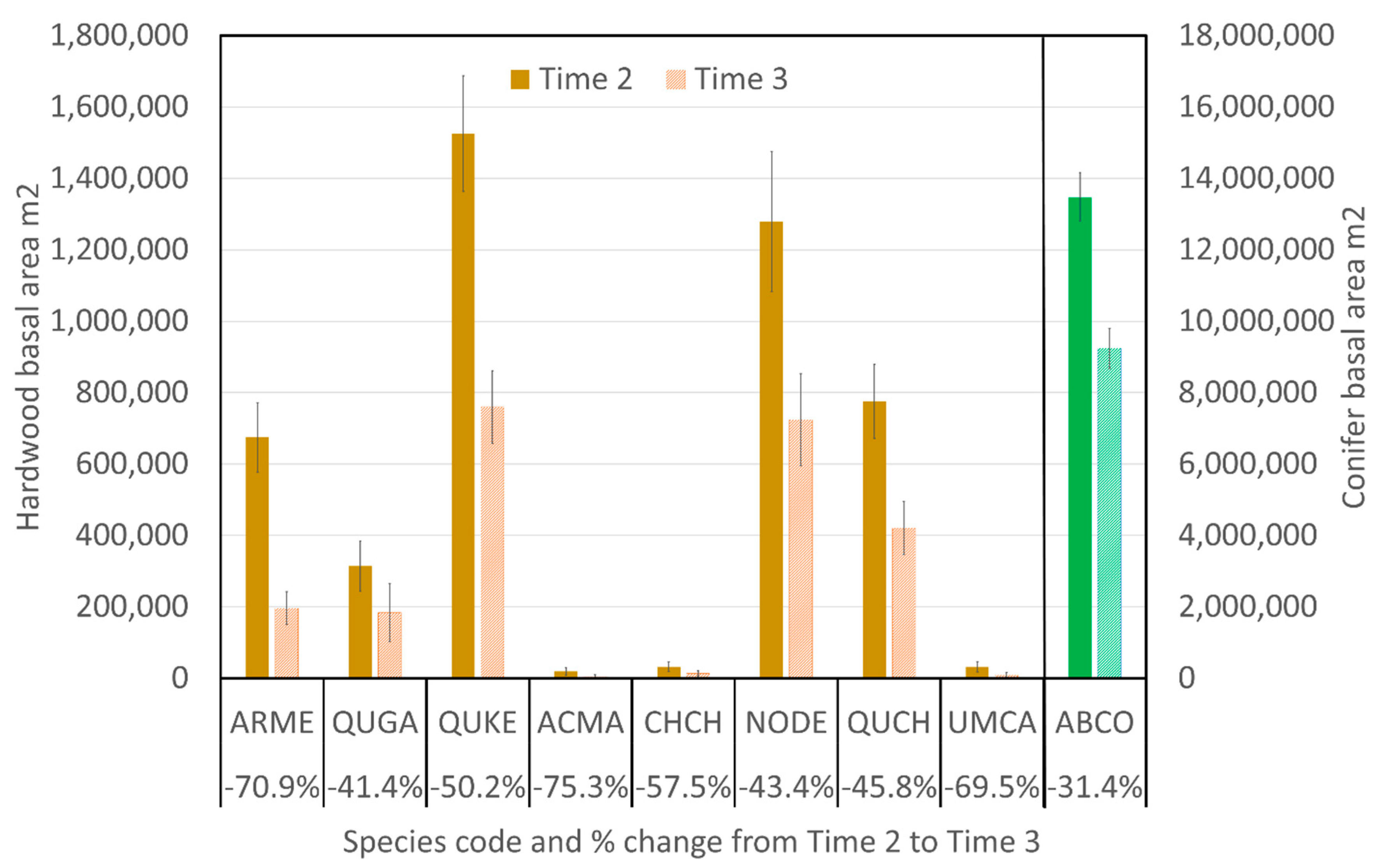
3.2. Overall Changes in Basal Area and Associated Causes of Mortality
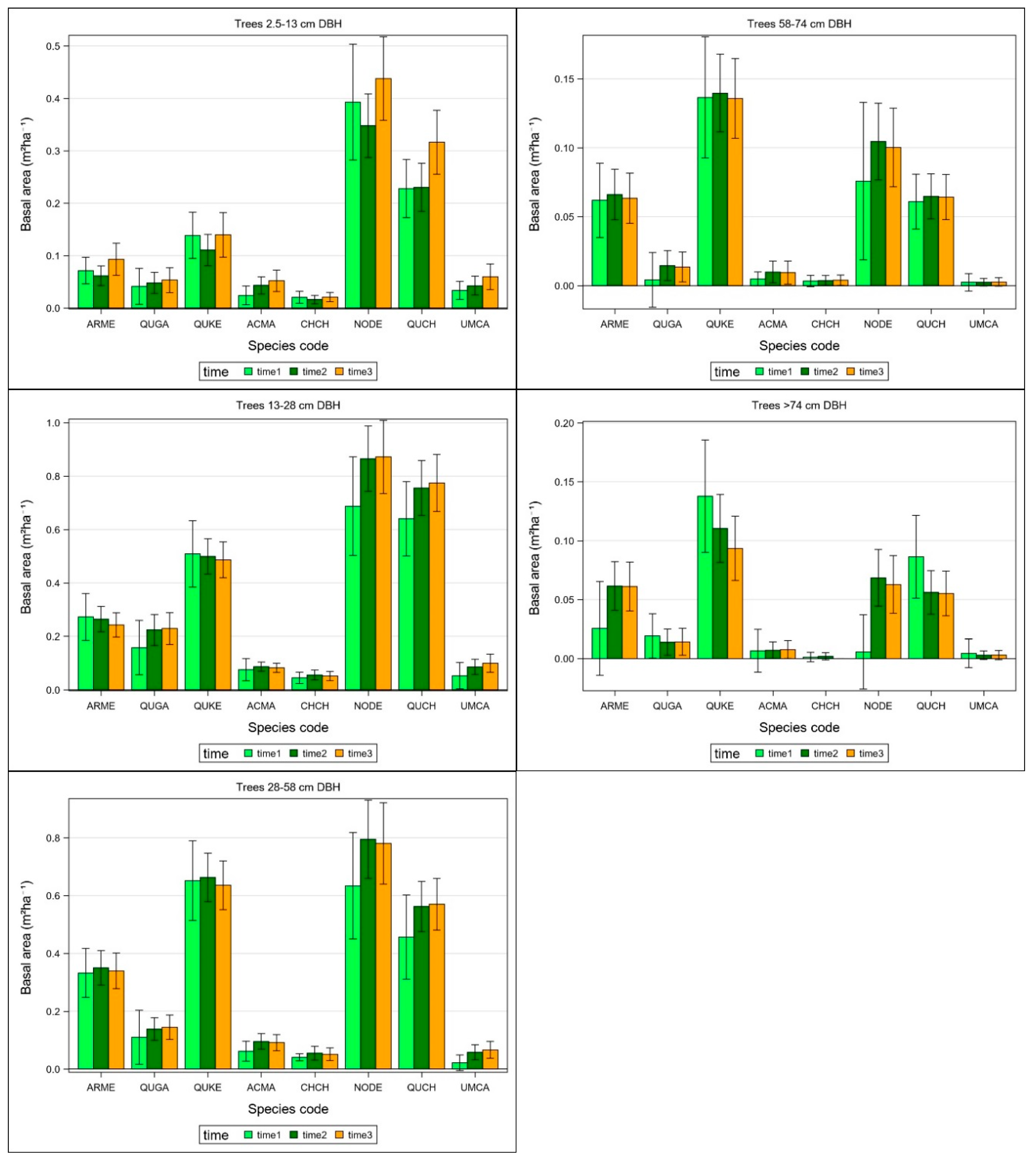
3.3. Changes in Basal Area within Size Classes
3.4. Changes in Basal Area Across Ecoregions and Ownership
3.5. Changes in Basal Area by Crown Condition Class
4. Discussion
4.1. Overall Trends
4.2. Strategies and Tactics Conserving Large Hardwood Trees
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Taylor, A.H.; Skinner, C.N. Spatial patterns and controls on historical fire regimes and forest structure in the Klamath Mountains. Ecol. Appl. 2003, 13, 704–719. [Google Scholar] [CrossRef]
- McShea, W.J.; Healy, W.M.; Devers, P.; Fearer, T.; Koch, F.H.; Stauffer, D.; Waldon, J. Forestry Matters: Decline of Oaks Will Impact Wildlife in Hardwood Forests. J. Wildl. Manag. 2007, 71, 1717–1728. [Google Scholar] [CrossRef]
- McComb, B.C.; Spies, T.A.; Olsen, K.A. Sustaining biodiversity in the Oregon Coast Range: Potential effects of forest policies in a multi-ownership province. Ecol. Soc. 2007, 12, art29. [Google Scholar] [CrossRef]
- Hagar, J.C. Wildlife species associated with non-coniferous vegetation in Pacific Northwest conifer forests: A review. For. Ecol. Manag. 2007, 246, 108–122. [Google Scholar] [CrossRef]
- Long, J.W.; Lake, F.K. Escaping a socioecological trap through tribal stewardship on national forest lands in the Pacific Northwest, USA. Ecol. Soc. 2018, 23, art10. [Google Scholar] [CrossRef]
- Bowcutt, F. Tanoak landscapes: Tending a Native American nut tree. Madrono 2013, 60, 64–86. [Google Scholar] [CrossRef]
- McGregor, R.R.; Sakalidis, M.L.; Hamelin, R.C. Neofusicoccum arbuti: A hidden threat to Arbutus menziesii characterized by widespread latent infections and a broad host range. Can. J. Plant Pathol. 2016, 38, 70–81. [Google Scholar] [CrossRef]
- Long, J.W.; Anderson, M.K.; Quinn-Davidson, L.N.; Goode, R.W.; Lake, F.K.; Skinner, C.N. Restoring California Black Oak Ecosystems to Promote Tribal Values and Wildlife; U.S. Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 2016.
- Coops, N.C.; Waring, R.H. Estimating the vulnerability of fifteen tree species under changing climate in Northwest North America. Ecol. Model. 2011, 222, 2119–2129. [Google Scholar] [CrossRef]
- Case, M.J.; Lawler, J.J. Relative vulnerability to climate change of trees in western North America. Clim. Chang. 2016, 136, 367–379. [Google Scholar] [CrossRef]
- Nemens, D.G.; Varner, J.M.; Kidd, K.R.; Wing, B. Do repeated wildfires promote restoration of oak woodlands in mixed-conifer landscapes? For. Ecol. Manag. 2018, 427, 143–151. [Google Scholar] [CrossRef]
- Cocking, M.I.; Varner, J.M.; Sherriff, R.L. California black oak responses to fire severity and native conifer encroachment in the Klamath Mountains. For. Ecol. Manag. 2012, 270, 25–34. [Google Scholar] [CrossRef]
- Hammett, E.J.; Ritchie, M.W.; Berrill, J.-P. Resilience of California black oak experiencing frequent fire: Regeneration following two large wildfires 12 years apart. Fire Ecol. 2017, 13, 91–103. [Google Scholar] [CrossRef]
- Filipe, J.A.; Cobb, R.C.; Meentemeyer, R.K.; Lee, C.A.; Valachovic, Y.S.; Cook, A.R.; Rizzo, D.M.; Gilligan, C.A. Landscape epidemiology and control of pathogens with cryptic and long-distance dispersal: Sudden oak death in northern Californian forests. PLoS Comp. Biol. 2012, 8, e1002328. [Google Scholar] [CrossRef] [PubMed]
- Lofroth, E.; Raley, C.; Higley, J.; Truex, R.; Yaeger, J.; Lewis, J.; Happe, P.; Finley, L.; Naney, R.; Hale, L. Conservation of Fishers (Martes pennanti) in South-Central British Columbia, Western Washington, Western Oregon, and California–Volume I: Conservation Assessment; USDI Bureau of Land Management: Denver, CO, USA, 2010.
- Long, J.W.; Lake, F.K.; Lynn, K.; Viles, C. Tribal Ecocultural Resources and Engagement; USDA Forest Service, Pacific Northwest Research Station: Portland, OR, USA, 2018.
- Anderson, M.K. Indigenous Uses Management, and Restoration of Oaks of the Far Western United States; NRCS National Plant Data Center: Washington, DC, USA, 2007. [Google Scholar]
- Baumhoff, M.A. California Athabascan groups. Anthr. Rec. 1958, 16, 157–238. [Google Scholar]
- Kniffen, F.B. Achomawi geography. Univ. Calif. Publ. Am. Archaeol. Ethnol. 1928, 23, 297–332. [Google Scholar]
- Kroeber, A.L.; Gifford, E.W. World renewal: A cult system of native northwest California. Anthr. Rec. 1949, 13, 1–156. [Google Scholar]
- Du Bois, C. Wintu ethnography. Univ. Calif. Publ. Am. Archaeol. Ethnol. 1935, 36, 1–148. [Google Scholar]
- Bunnell, F.L.; Kremsater, L.L.; Wind, E. Managing to sustain vertebrate richness in forests of the Pacific Northwest: Relationships within stands. Environ. Rev. 1999, 7, 97–146. [Google Scholar] [CrossRef]
- Wu, J.X.; Siegel, R.B.; Loffland, H.L.; Tingley, M.W.; Stock, S.L.; Roberts, K.N.; Keane, J.J.; Medley, J.R.; Bridgman, R.; Stermer, C. Diversity of great gray owl nest sites and nesting habitats in California. J. Wildl. Manag. 2015, 79, 937–947. [Google Scholar] [CrossRef]
- Lindenmayer, D.B. Integrating forest biodiversity conservation and restoration ecology principles to recover natural forest ecosystems. New For. 2018. [Google Scholar] [CrossRef]
- Wasyl, J. Uncle oak: The giant of Palomar Mountain. Int. Oaks 2011, 22, 100–103. [Google Scholar]
- Duckworth, J. Remembering Fallen Champion Trees; American Forests: Washington, DC, USA, 2016; Available online: http://www.americanforests.org/blog/remembering-fallen-champion-trees/ (accessed on 1 April 2018).
- Cocking, M.I.; Varner, J.M.; Knapp, E.E. Long-term effects of fire severity on oak-conifer dynamics in the southern Cascades. Ecol. Appl. 2014, 24, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Baker, W.L. Historical forest structure and fire in Sierran mixed-conifer forests reconstructed from General Land Office survey data. Ecosphere 2014, 5, 1–70. [Google Scholar] [CrossRef]
- Cocking, M.I.; Varner, J.M.; Engber, E.A. Conifer Encroachment in California Oak Woodlands; U.S. Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 2015.
- Crawford, J.N.; Mensing, S.A.; Lake, F.K.; Zimmerman, S.R. Late Holocene fire and vegetation reconstruction from the western Klamath Mountains, California, USA: A multi-disciplinary approach for examining potential human land-use impacts. Holocene 2015, 25, 1341–1357. [Google Scholar] [CrossRef]
- Taylor, A.H.; Trouet, V.; Skinner, C.N.; Stephens, S. Socioecological transitions trigger fire regime shifts and modulate fire–climate interactions in the Sierra Nevada, USA, 1600–2015 CE. Proc. Natl. Acad. Sci. USA 2016, 113, 13684–13689. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K. Tending the Wild: Native American Knowledge and the Management of California’s Natural Resources; University of California Press: Berkeley, CA, USA, 2005. [Google Scholar]
- Abrams, M.D.; Nowacki, G.J. Native Americans as active and passive promoters of mast and fruit trees in the eastern USA. Holocene 2008, 18, 1123–1137. [Google Scholar] [CrossRef]
- Chamberlain, J.L.; Emery, M.R.; Patel-Weynand, T. Assessment of Nontimber Forest Products in the United States under Changing Conditions; USDA Forest Service Southern Research Station: Asheville, NC, USA, 2018.
- Chamberlain, J.L.; Prisley, S.; McGuffin, M. Understanding the relationships between American ginseng harvest and hardwood forests inventory and timber harvest to improve co-management of the forests of eastern United States. J. Sustain. For. 2013, 32, 605–624. [Google Scholar] [CrossRef]
- Emery, M.R.; Wrobel, A.; Hansen, M.H.; Dockry, M.; Moser, W.K.; Stark, K.J.; Gilbert, J.H. Using traditional ecological knowledge as a basis for targeted forest inventories: Paper birch (Betula papyrifera) in the US Great Lakes region. J. For. 2014, 112, 207–214. [Google Scholar] [CrossRef]
- Vance, N.; Gray, A.; Haberman, B. Assessment of Western Oregon Forest Inventory for Evaluating Commercially Important Understory Plants; USDA Forest Service Pacific Northwest Research Station: Portland, OR, USA, 2002.
- Christensen, G.A.; Waddell, K.L.; Stanton, S.M.; Kuegler, O. California’s Forest Resources: Forest Inventory and Analysis, 2001-2010; USDA Forest Service, Pacific Northwest Research Station: Portland, OR, USA, 2016.
- McIntyre, P.J.; Thorne, J.H.; Dolanc, C.R.; Flint, A.L.; Flint, L.E.; Kelly, M.; Ackerly, D.D. Twentieth-century shifts in forest structure in California: Denser forests, smaller trees, and increased dominance of oaks. Proc. Natl. Acad. Sci. USA 2015, 112, 1458–1463. [Google Scholar] [CrossRef] [PubMed]
- Dolanc, C.R.; Safford, H.D.; Thorne, J.H.; Dobrowski, S.Z. Changing forest structure across the landscape of the Sierra Nevada, CA, USA, since the 1930s. Ecosphere 2014, 5, art101. [Google Scholar] [CrossRef]
- Lutz, J.A.; van Wagtendonk, J.W.; Franklin, J.F. Twentieth-century decline of large-diameter trees in Yosemite National Park, California, USA. For. Ecol. Manag. 2009, 257, 2296–2307. [Google Scholar] [CrossRef]
- Zielinski, W.J.; Gray, A.N. Using routinely collected regional forest inventory data to conclude that resting habitat for the fisher (Pekania pennanti) in California is stable over ~20 years. For. Ecol. Manag. 2018, 409, 899–908. [Google Scholar] [CrossRef]
- Zielinski, W.J.; Truex, R.L.; Dunk, J.R.; Gaman, T. Using forest inventory data to assess fisher resting habitat suitability in California. Ecol. Appl. 2006, 16, 1010–1025. [Google Scholar] [CrossRef]
- Zielinski, W.J.; Dunk, J.R.; Gray, A.N. Estimating habitat value using forest inventory data: The fisher (Martes pennanti) in northwestern California. For. Ecol. Manag. 2012, 275, 35–42. [Google Scholar] [CrossRef]
- Waddell, K.L.; Barrett, T.M. Oak Woodlands and other Hardwood Forests of California, 1990s; USDA Forest Service, Pacific Northwest Research Station: Portland, OR, USA, 2005.
- USDA Forest Service. Draft Revised Land Management Plan for the Sierra National Forest; USDA Forest Service, Pacific Southwest Region, Sierra National Forest: Clovis, CA, USA, 2016.
- McDonald, P.M.; Tappeiner, J.C. California’s Hardwood Resource: Seeds, Seedlings, and Sprouts of Three Important Forest-Zone Species; U.S. Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 2002. [Google Scholar]
- Gucker, C.L. Quercus garryana; USDA Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA. Available online: https://www.fs.fed.us/database/feis/plants/tree/quegar/all.html (accessed on 30 July 2018).
- Tollefson, J.E. Quercus chrysolepis; USDA Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA. Available online: https://www.fs.fed.us/database/feis/plants/tree/quechr/all.html (accessed on 1 May 2018).
- Meyer, R. Chrysolepis chrysophylla; USDA Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA. Available online: https://www.fs.fed.us/database/feis/plants/tree/chrchr/all.html (accessed on 30 July 2018).
- Reeves, S.L. Arbutus menziesii; USDA Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA. Available online: https://www.fs.fed.us/database/feis/plants/tree/arbmen/all.html (accessed on 30 July 2018).
- Fryer, J.L. Acer macrophyllum, bigleaf maple; USDA Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA. Available online: https://www.fs.fed.us/database/feis/plants/tree/acemac/all.html (accessed on 1 May 2018).
- Howard, J.L. Umbellularia californica; USDA Forest Service Rocky Mountain Research Station: Fort Collins, CO, USA. Available online: https://www.fs.fed.us/database/feis/plants/tree/umbcal/all.html (accessed on 1 May 2018).
- Niemiec, S.S.; Ahrens, G.R.; Willits, S.; Hibbs, D.E. Hardwoods of the Pacific Northwest; Research Contribution; Oregon State University: Corvallis, OR, USA, 1995. [Google Scholar]
- Frankel, S. (USDA Forest Service, Pacific Southwest Research Station, Albany, CA, USA). Personal communication, 2018.
- Bolsinger, C.L. The Hardwoods of California’s Timberlands, Woodlands, and Savannas; USDA Forest Service, Pacific Northwest Research Station: Portland, OR, USA, 1988. [Google Scholar]
- Little, E.L., Jr. Conifers and Important Hardwoods; Miscellaneous Publication 1186; USDA: Washington DC, USA, 1971.
- Little, E.L., Jr. Minor Western Hardwoods; Miscellaneous Publication 1314; USDA: Washington, DC, USA, 1976.
- McDonald, P.M. Chrysolepis Hjelmqvist. In Silvics of North America; Burns, R.M., Honkala, B.H., Eds.; USDA Forest Service: Washington, DC, USA, 1990; pp. 404–406. [Google Scholar]
- Peter, D.; Harrington, C. Site and tree factors in Oregon white oak acorn production in western Washington and Oregon. Northwest Sci. 2002, 76, 189–201. [Google Scholar]
- Devine, W.D.; Harrington, C.A. Restoration release of overtopped Oregon white oak increases 10-year growth and acorn production. For. Ecol. Manag. 2013, 291, 87–95. [Google Scholar] [CrossRef]
- Long, J.W.; Goode, R.W.; Gutteriez, R.J.; Lackey, J.J.; Anderson, M.K. Managing California black oak for tribal ecocultural restoration. J. For. 2017, 115, 426–434. [Google Scholar] [CrossRef]
- Yaeger, J.S. Habitat at Fisher Resting Sites in the Klamath Province of Northern California. Master’s Thesis, Humboldt State University, Arcata, CA, USA, May 2005. [Google Scholar]
- Aubry, K.B.; Raley, C.M.; Buskirk, S.W.; Zielinski, W.J.; Schwartz, M.K.; Golightly, R.T.; Purcell, K.L.; Weir, R.D.; Yaeger, J.S. Meta-analyses of habitat selection by fishers at resting sites in the Pacific coastal region. J. Wildl. Manag. 2013, 77, 965–974. [Google Scholar] [CrossRef]
- North, M.; Steger, G.; Denton, R.; Eberlein, G.; Munton, T.; Johnson, K. Association of weather and nest-site structure with reproductive success in California spotted owls. J. Wildl. Manag. 2000, 64, 797–807. [Google Scholar] [CrossRef]
- Devine, W.D.; Harrington, C.A. Changes in Oregon white oak (Quercus garryana Dougl. ex Hook.) following release from overtopping conifers. Trees 2006, 20, 747–756. [Google Scholar] [CrossRef]
- Arno, S.F. Fire in Western Forest Ecosystems; USDA Forest Service Rocky Mountain Research Station: Fort Collins, CO, USA, 2000.
- Fonda, R. Postfire response of red alder, black cottonwood, and bigleaf maple to the Whatcom Creek fire, Bellingham, Washington. Northwest Sci. 2001, 75, 25–36. [Google Scholar]
- Dale, V.H.; Hemstrom, M.; Franklin, J. Modeling the long-term effects of disturbances on forest succession, Olympic Peninsula, Washington. Can. J. For. Res. 1986, 16, 56–67. [Google Scholar] [CrossRef]
- Cobb, R.C.; Filipe, J.A.N.; Meentemeyer, R.K.; Gilligan, C.A.; Rizzo, D.M. Ecosystem transformation by emerging infectious disease: Loss of large tanoak from California forests. J. Ecol. 2012, 100, 712–722. [Google Scholar] [CrossRef]
- Bechtold, W.A.; Patterson, P.L. The Enhanced Forest Inventory and Analysis Program—National Sampling Design and Estimation Procedures; USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2005.
- Eyre, F.H. Forest Cover Types of the United States and Canada; Society of American Foresters: Washington, DC, USA, 1980. [Google Scholar]
- McNab, W.H.; Cleland, D.T.; Freeouf, J.A.; Keys Jr, J.E.; Nowacki, G.J.; Carpenter, C.A. Description of Ecological Subregions: Sections of the Conterminous United States; USDA Forest Service: Washington, DC, USA, 2007.
- LANDFIRE. Fire Regime Groups. USDI, Geological Survey. Available online: https://landfire.gov/frg.php (accessed on 31 August 2018).
- Spencer, W.D.; Sawyer, S.C.; Romsos, H.L.; Zielinski, W.J.; Sweitzer, R.A.; Thompson, C.M.; Purcell, K.L.; Clifford, D.L.; Cline, L.; Safford, H.D.; et al. Southern Sierra Nevada Fisher Conservation Assessment; Conservation Biology Institute: Portland, OR, USA, 2015. [Google Scholar]
- USDA Forest Service. Inventory Data. Available online: https://www.fs.fed.us/pnw/rma/fia-topics/inventory-data/index.php (accessed on 31 August 2018).
- Scott, C.T.; Bechtold, W.A.; Reams, G.A.; Smith, W.D.; Westfall, J.A.; Hansen, M.H.; Moisen, G.G. Sample-Based Estimators Used by the Forest Inventory and Analysis National Information Management System; USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2005. [Google Scholar]
- Tappeiner, J.; McDonald, P.M.; Roy, D.F. Lithocarpus densiflorus (Hook. & Arn.) Rehd. Tanoak. In Silvics of North America; Burns, R.M., Honkala, B.H., Eds.; USDA: Washington, DC, USA, 1990; pp. 417–425. [Google Scholar]
- Miller, J.D.; Safford, H.D. Trends in wildfire severity 1984–2010 in the Sierra Nevada, Modoc Plateau, and southern Cascades, California, USA. Fire Ecol. 2012, 8, 41–57. [Google Scholar] [CrossRef]
- Miller, J.D.; Skinner, C.N.; Safford, H.D.; Knapp, E.E.; Ramirez, C.M. Trends and causes of severity, size, and number of fires in northwestern California, USA. Ecol. Appl. 2012, 22, 184–203. [Google Scholar] [CrossRef] [PubMed]
- Reilly, M.J.; Dunn, C.J.; Meigs, G.W.; Spies, T.A.; Kennedy, R.E.; Bailey, J.D.; Briggs, K. Contemporary patterns of fire extent and severity in forests of the Pacific Northwest, USA (1985–2010). Ecosphere 2017, 8, e01695. [Google Scholar] [CrossRef]
- Paz-Kagan, T.; Brodrick, P.G.; Vaughn, N.R.; Das, A.J.; Stephenson, N.L.; Nydick, K.R.; Asner, G.P. What mediates tree mortality during drought in the southern Sierra Nevada? Ecol. Appl. 2017, 27, 2443–2457. [Google Scholar] [CrossRef] [PubMed]
- Stephens, S.L.; Collins, B.M.; Fettig, C.J.; Finney, M.A.; Hoffman, C.M.; Knapp, E.E.; North, M.P.; Safford, H.; Wayman, R.B. Drought, tree mortality, and wildfire in forests adapted to frequent fire. Bioscience 2018, 68, 77–88. [Google Scholar] [CrossRef]
- USFS. Rim Fire Restoration Environmental Impact Statement; Stanislaus National Forest: Sonora, CA, USA, 2016.
- Weatherspoon, C.P.; Almond, G.A.; Skinner, C.N. Tree-centered spot firing-a technique for prescribed burning beneath standing trees. West. J. Appl. For. 1989, 4, 29–31. [Google Scholar]

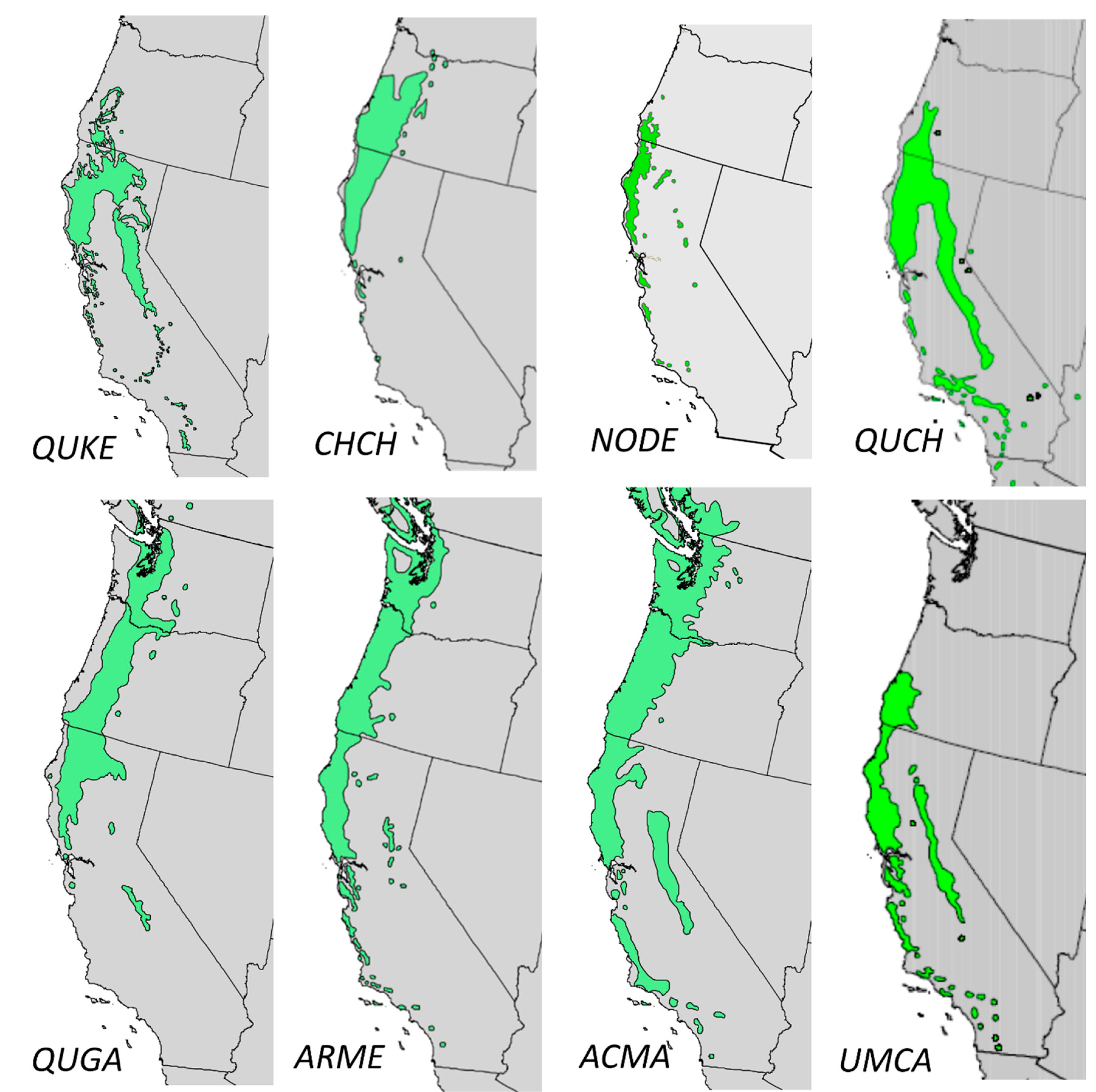

| Scientific Name | Arbutus menziesii | Quercus garryana | Quercus kelloggii | Acer macrophyllum | Chrysolepis chrysophylla | Notho-lithocarpus densiflorus | Quercus chrysolepis | Umbellularia californica |
|---|---|---|---|---|---|---|---|---|
| Species code | ARME | QUGA | QUKE | ACMA | CHCH | NODE | QUCH | UMCA |
| Common name | Pacific madrone | Oregon white oak | California black oak | bigleaf maple | giant chinquapin | tanoak | canyon live oak | California laurel |
| Upper DBH (cm) | 122 | 246 | 274 | 338 | 244 | 277 | 330 | 404 |
| Upper height (m) | 38 | 37 | 40 | 49 | 46 | 63 | 30 | 53 |
| Upper age (years) | 400 | 500 | 500 | 300 | 500 | 250+ | 300 | 200+ |
| Typical onset of fruiting (years) | 30 | 20 | 30 | 10 | <40 | 30 | 20 | 30–40 |
| Shade tolerance | intermediate to low | low | low | high | intermediate | high | high | intermediate |
| Fire tolerance | 1 | 2 | 2 | 1 | 1 | 1.5 | 0.5 | 0.5 |
| Vulner-ability to Phytophthora ramorum | mortality | not affected | mortality | no mortality; infection is limited to leaf spots | mortality | mortality | mortality | no mortality; infection is limited to leaf spots |
| Ecoregion | Total Area | Fire | Cut | Fire + Cut | Other | Total Disturbed | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ha | % | ha/year | % | ha/year | % | ha/year | ha/year | ha/year | % | |||
| Klamath | 3,037,602 | 33 | 47,582 | 1.6 | 26,982 | 0.9 | 1928 | 0.1 | 2455 | 0.1 | 78,947 | 2.6 |
| CA Coast | 743,446 | 8 | 766 | 0.1 | 10,205 | 1.4 | 484 | 0.1 | 592 | 0.1 | 12,047 | 1.6 |
| CA Coast Range | 865,360 | 9 | 11,645 | 1.3 | 3043 | 0.4 | 0.0 | 0.0 | 1090 | 0.1 | 15,779 | 1.8 |
| OR Coast Range | 178,611 | 2 | 0.0 | 2687 | 1.5 | 445 | 0.2 | 552 | 0.3 | 3684 | 2.1 | |
| Sierra | 2,766,788 | 30 | 26,437 | 1.0 | 31,994 | 1.2 | 4252 | 0.2 | 2511 | 0.1 | 65,194 | 2.4 |
| Cascades | 1,559,874 | 17 | 5276 | 0.3 | 39,948 | 2.6 | 2626 | 0.2 | 3649 | 0.2 | 51,499 | 3.3 |
| Totals | 9,151,682 | 100 | 91,706 | 1.0 | 114,859 | 1.3 | 9735 | 0.1 | 10,849 | 0.1 | 227,149 | 2.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Long, J.W.; Gray, A.; Lake, F.K. Recent Trends in Large Hardwoods in the Pacific Northwest, USA. Forests 2018, 9, 651. https://doi.org/10.3390/f9100651
Long JW, Gray A, Lake FK. Recent Trends in Large Hardwoods in the Pacific Northwest, USA. Forests. 2018; 9(10):651. https://doi.org/10.3390/f9100651
Chicago/Turabian StyleLong, Jonathan W., Andrew Gray, and Frank K. Lake. 2018. "Recent Trends in Large Hardwoods in the Pacific Northwest, USA" Forests 9, no. 10: 651. https://doi.org/10.3390/f9100651
APA StyleLong, J. W., Gray, A., & Lake, F. K. (2018). Recent Trends in Large Hardwoods in the Pacific Northwest, USA. Forests, 9(10), 651. https://doi.org/10.3390/f9100651




