Deadwood Decay in a Burnt Mediterranean Pine Reforestation
Abstract
:1. Introduction
2. Methods
2.1. Study Site
2.2. Sampling Design
2.3. Wood Decomposition
2.4. Statistical Analyses
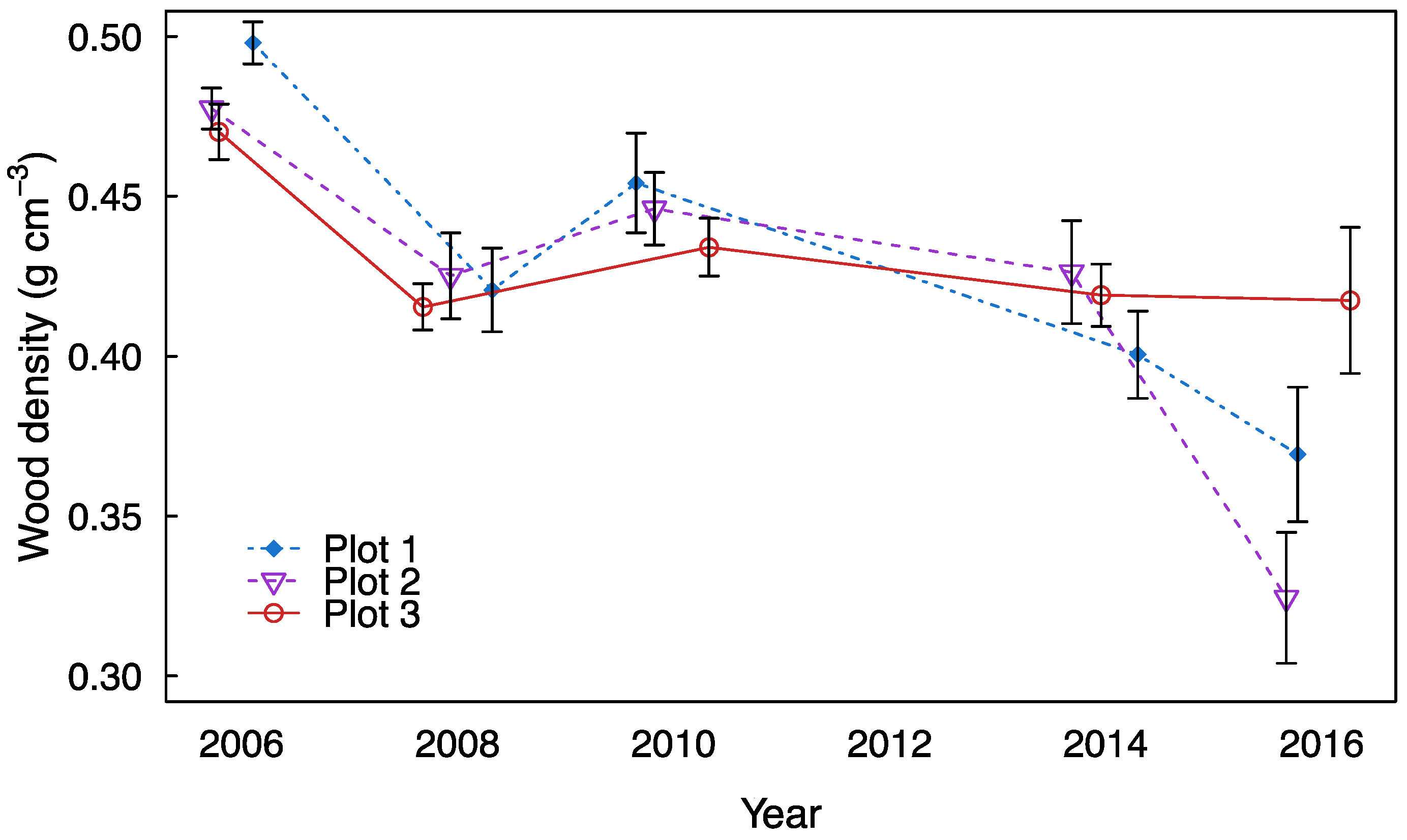
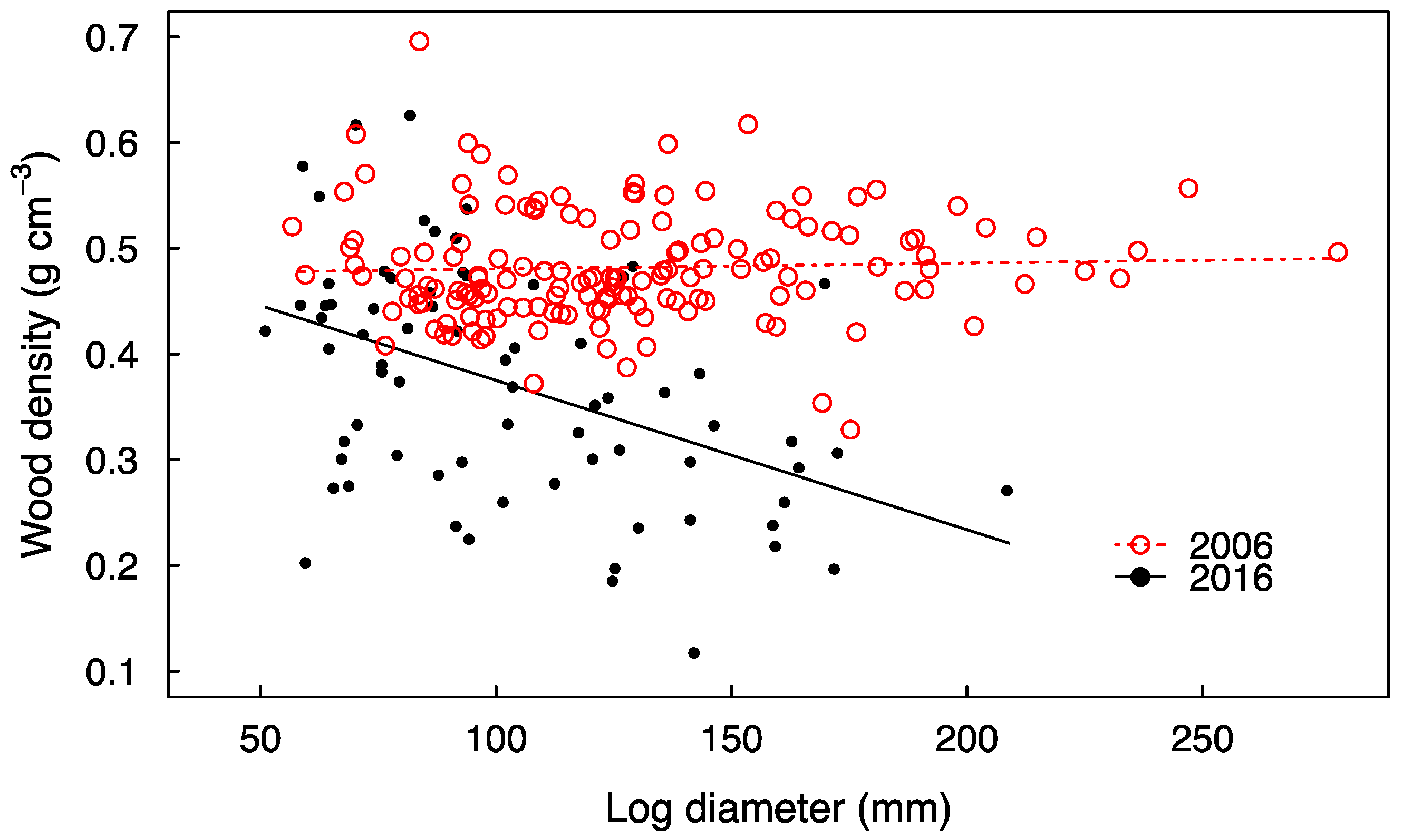
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Harmon, M.E.; Franklin, J.F.; Swanson, F.J.; Sollins, P.; Gregory, S.V.; Lattin, J.D.; Anderson, N.H.; Cline, S.P.; Aumen, N.G.; Sedell, J.R.; et al. Ecology of coarse woody debris in temperate ecosystems. Adv. Ecol. Res. 1986, 15, 133–302. [Google Scholar]
- Franklin, J.F.; Shugart, H.H.; Harmon, M.E. Tree death as an ecological process. BioScience 1987, 37, 550–556. [Google Scholar] [CrossRef]
- Chamber, C.L.; Mast, J.N. Ponderosa pine snag dynamics and cavity excavation following wildfire in northern Arizona. For. Ecol. Manag. 2005, 216, 227–240. [Google Scholar] [CrossRef]
- Stokland, J.N.; Siitonen, J.; Jonsson, B.G. Biodiversity in Dead Wood; Cambridge University Press: Cambridge, UK, 2012. [Google Scholar]
- Swift, M. The ecology of wood decompositiopn. Sci. Prog. 1977, 64, 175–199. [Google Scholar]
- Ganjegunte, G.K.; Condron, L.M.; Clinton, P.W.; Davis, M.R.; Mahieu, N. Decomposition and nutrient release from radiata pine (Pinus radiata) coarse woody debris. For. Ecol. Manag. 2004, 187, 197–211. [Google Scholar] [CrossRef]
- Palviainen, M.; Finér, L.; Kurka, A.M.; Mannerkoski, H.; Piirainen, S.; Starr, M. Decomposition and nutrient release from logging residues after clear-cutting of mixed boreal forest. Plant Soil 2004, 263, 53–67. [Google Scholar] [CrossRef]
- Russell, M.B.; Woodall, C.W.; D’Amato, A.W.; Fraver, S.; Bradford, J.B. Technical Note: Linking climate change and downed woody debris decomposition across forests of the eastern United States. Biogeosciences 2014, 11, 6417–6425. [Google Scholar] [CrossRef]
- Cornwell, W.K.; Cornelissen, J.H.C.; Allison, S.D.; Bauhus, J.; Eggleton, P.; Preston, C.M.; Scarff, F.; Weedon, J.T.; Wirth, C.; Zanne, A.E. Plant traits and wood fates across the globe: Rotted, burned, or consumed? Glob. Chang. Biol. 2009, 15, 2431–2449. [Google Scholar] [CrossRef]
- Means, J.E.; MacMillan, P.C.; Cromack, K.J. Biomas and nutrient content of douglas-fir logs and other detrital pools in a old-growth forest, Oregon, U.S.A. Can. J. For. Res. 1992, 22, 1536–1546. [Google Scholar] [CrossRef]
- Schiegg, K. Are there saproxylic beetle species characteristic of high dead wood connectivity? Ecography 2000, 23, 579–587. [Google Scholar] [CrossRef]
- Vodka, S.; Konvicka, M.; Cizek, L. Habitat preferences of oak-feeding xylophagous beetles in a temperate woodland: Implications for forest history and management. J. Insect Conserv. 2009, 13, 553–562. [Google Scholar] [CrossRef]
- Angelstam, P.K.; Bütler, R.; Lazdinis, M.; Mikusinski, G.; Roberge, J.-M. Habitat thresholds for focal species at multiple scales and forest biodiversity conservation—dead wood as an example. Ann. Zool. Fenn. 2003, 40, 473–482. [Google Scholar]
- Lassauce, A.; Paillet, Y.; Jactel, H.; Bouget, C. Deadwood as a surrogate for forest biodiversity: Meta-analysis of correlations between deadwood volume and species richness of saproxylic organisms. Ecol. Indic. 2011, 11, 1027–1039. [Google Scholar] [CrossRef]
- Rajandu, E.; Kikas, K.; Paal, J. Bryophytes and decaying wood in hepatica site-type boreo-nemoral Pinus sylvestris forests in Southern Estonia. For. Ecol. Manag. 2009, 257, 994–1003. [Google Scholar] [CrossRef]
- Marzano, R.; Garbarino, M.; Marcolin, E.; Pividori, M.; Lingua, E. Deadwood anisotropic facilitation on seedling establishment after a stand-replacing wildfire in Aosta Valley (NW Italy). Ecol. Eng. 2013, 51, 117–122. [Google Scholar] [CrossRef]
- Leverkus, A.B.; Lorite, J.; Navarro, F.B.; Sánchez-Cañete, E.P.; Castro, J. Post-fire salvage logging alters species composition and reduces cover, richness, and diversity in Mediterranean plant communities. J. Environ. Manag. 2014, 133, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Chmura, D.; Żarnowiec, J.; Staniaszek-Kik, M. Interactions between plant traits and environmental factors within and among montane forest belts: A study of vascular species colonising decaying logs. For. Ecol. Manag. 2016, 379, 216–225. [Google Scholar] [CrossRef]
- Cadieux, P.; Drapeau, P. Are old boreal forests a safe bet for the conservation of the avifauna associated with decayed wood in eastern Canada? For. Ecol. Manag. 2017, 385, 127–139. [Google Scholar] [CrossRef]
- Serrano-Ortiz, P.; Marañón-Jiménez, S.; Reverter, B.R.; Sánchez-Cañete, E.P.; Castro, J.; Zamora, R.; Kowalski, A.S. Post-fire salvage logging reduces carbon sequestration in Mediterranean coniferous forest. For. Ecol. Manag. 2011, 262, 2287–2296. [Google Scholar] [CrossRef]
- Shorohova, E.; Kapitsa, E. Influence of the substrate and ecosystem attributes on the decomposition rates of coarse woody debris in European boreal forests. For. Ecol. Manag. 2014, 315, 173–184. [Google Scholar] [CrossRef]
- Russell, M.B.; Fraver, S.; Aakala, T.; Gove, J.H.; Woodall, C.W.; D’Amato, A.W.; Ducey, M.J. Quantifying carbon stores and decomposition in dead wood: A review. For. Ecol. Manag. 2015, 350, 107–128. [Google Scholar] [CrossRef]
- Olajuyigbe, S.O.; Tobin, B.; Gardiner, P.; Nieuwenhuis, M. Stocks and decay dynamics of above- and belowground coarse woody debris in managed Sitka spruce forests in Ireland. For. Ecol. Manag. 2011, 262, 1109–1118. [Google Scholar] [CrossRef]
- Passovoy, M.D.; Fulé, P.Z. Snag and woody debris dynamics following severe wildfires in northern Arizona ponderosa pine forests. For. Ecol. Manag. 2006, 223, 237–246. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; Burton, P.J.; Franklin, J.F. Salvage Logging and Its Ecological Consequences; Island Press: Washington, DC, USA, 2008. [Google Scholar]
- Castro, J.; Moreno-Rueda, G.; Hodar, J.A. Experimental test of postfire management in pine forests: impact of salvage logging versus partial cutting and nonintervention on bird-species assemblages. Conserv. Biol. 2010, 24, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.W.; Knapp, E.E.; Skinner, C.N. Snag longevity and surface fuel accumulation following post-fire logging in a ponderosa pine dominated forest. For. Ecol. Manag. 2013, 287, 113–122. [Google Scholar] [CrossRef]
- Donato, D.C.; Fontaine, J.B.; Campbell, J.L.; Robinson, W.D.; Kauffman, J.B.; Law, B.E. Post-Wildfire logging hinders regeneration and increases fire risk. Science 2006, 311, 352. [Google Scholar] [CrossRef] [PubMed]
- Castro, J.; Allen, C.D.; Molina-Morales, M.; Marañón-Jiménez, S.; Sánchez-Miranda, Á.; Zamora, R. Salvage Logging versus the use of burnt wood as a nurse object to promote post-fire tree seedling establishment. Restor. Ecol. 2011, 19, 537–544. [Google Scholar] [CrossRef]
- Marañón-Jiménez, S.; Castro, J. Effect of decomposing post-fire coarse woody debris on soil fertility and nutrient availability in a Mediterranean ecosystem. Biogeochemistry 2013, 112, 519–535. [Google Scholar] [CrossRef]
- Marañón-Jiménez, S.; Castro, J.; Fernández-Ondoño, E.; Zamora, R. Charred wood remaining after a wildfire as a reservoir of macro- and micronutrients in a Mediterranean pine forest. Int. J. Wildland Fire 2013, 22, 681–695. [Google Scholar] [CrossRef]
- Thorn, S.; Bässler, C.; Brandl, R.; Burton, P.J.; Cahall, R.; Campbell, J.L.; Castro, J.; Choi, C.-Y.; Cobb, T.; Donato, D.C.; et al. Impacts of salvage logging on biodiversity—a meta-analysis. J. Appl. Ecol. 2017, in press. [Google Scholar]
- Sippola, A.; Siitonen, J.; Kallio, R. Amount and quality of coarse woody debris in natural and managed coniferous forests near the timberline in Finnish Lapland. Scand. J. For. Res. 1998, 13, 204–214. [Google Scholar] [CrossRef]
- Rock, J.; Badeck, F.-W.; Harmon, M.E. Estimating decomposition rate constants for European tree species from literature sources. Eur. J. For. Res. 2008, 127, 301–313. [Google Scholar] [CrossRef]
- Lombardi, F.; Lasserre, B.; Tognetti, R.; Marchetti, M. Deadwood in relation to stand management and forest type in central apennines (Molise, Italy). Ecosystems 2008, 11, 882–894. [Google Scholar] [CrossRef]
- Lombardi, F.; Cherubini, P.; Tognetti, R.; Cocozza, C.; Lasserre, B.; Marchetti, M. Investigating biochemical processes to assess deadwood decay of beech and silver fir in Mediterranean mountain forests. Ann. For. Sci. 2013, 70, 101–111. [Google Scholar] [CrossRef]
- Liu, W.; Schaefer, D.; Qiao, L.; Liu, X. What controls the variability of wood-decay rates? For. Ecol. Manag. 2013, 310, 623–631. [Google Scholar] [CrossRef]
- Herrmann, S.; Bauhus, J. Effects of moisture, temperature and decomposition stage on respirational carbon loss from coarse woody debris (CWD) of important European tree species. Scand. J. For. Res. 2013, 28, 346–357. [Google Scholar] [CrossRef]
- Fravolini, G.; Egli, M.; Derungs, C.; Cherubini, P.; Ascher-Jenull, J.; Gómez-Brandón, M.; Bardelli, T.; Tognetti, R.; Lombardi, F.; Marchetti, M. Soil attributes and microclimate are important drivers of initial deadwood decay in sub-alpine Norway spruce forests. Sci. Total Environ. 2016, 569, 1064–1076. [Google Scholar] [CrossRef] [PubMed]
- De Aza, C.H.; Turrión, M.B.; Pando, V.; Bravo, F. Carbon in heartwood, sapwood and bark along the stem profile in three Mediterranean Pinus species. Ann. For. Sci. 2011, 68, 1067–1076. [Google Scholar] [CrossRef]
- Boddy, L. Fungal community ecology and wood decomposition processes in angiosperms: From standing tree to complete decay of coarse woody debris. Ecol. Bull. 2001, 49, 43–56. [Google Scholar]
- Ulyshen, M.D. Wood decomposition as influenced by invertebrates. Biol. Rev. 2016, 91, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Hazenberg, G. Sapwood and heartwood width relationship to tree age in Pinus banksiana. Can. J. For. Res. 1991, 21, 251–525. [Google Scholar] [CrossRef]
- Leverkus, A.B.; Puerta-Piñero, C.; Guzmán-Álvarez, J.; Navarro, J.; Castro, J. Post-fire salvage logging increases restoration costs in a Mediterranean mountain ecosystem. New For. 2012, 43, 601–613. [Google Scholar] [CrossRef]
- Leverkus, A.B.; Castro, J.; Delgado-Capel, M.J.; Molinas-González, C.; Pulgar, M.; Marañón-Jiménez, S.; Delgado-Huertas, A.; Querejeta, J.I. Restoring for the present or restoring for the future: Enhanced performance of two sympatric oaks (Quercus ilex and Quercus pyrenaica) above the current forest limit. Restor. Ecol. 2015, 23, 936–946. [Google Scholar] [CrossRef]
- Molinas-González, C.R.; Leverkus, A.B.; Marañón-Jiménez, S.; Castro, J. Fall rate of burnt pines across an elevational gradient in a Mediterranean mountain. Eur. J. For. Res. 2017. [Google Scholar] [CrossRef]
- Fraver, S.; Ringvall, A.; Jonsson, B.G. Refining volume estimates of down woody debris. Can. J. For. Res. 2007, 37, 627–633. [Google Scholar] [CrossRef]
- R Core Team. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. Linear and Nonlinear Mixed Effects Models. Available online: https://CRAN.R-project.org/package=nlme (accessed on 10 December 2016).
- Crawley, M.J. The R Book, 2nd ed.; John Wiley & Sons: West Sussex, UK, 2013. [Google Scholar]
- Pinheiro, J.C.; Bates, D.M. Mixed effects models in S and S-Plus; Springer: New York, NY, USA, 2000. [Google Scholar]
- Foster, J.R.; Lang, G.E. Decomposition of red spruce and balsam fir boles in the White Mountains of New Hampshire. Can. J. For. Res. 1982, 12, 617–626. [Google Scholar] [CrossRef]
- Coleman, D.C.; Crossley, D.A. Fundamentals of Soil Ecology, 2nd ed.; Academic Press: Waltham, MA, USA, 2003. [Google Scholar]
- Mackensen, J.; Bauhus, J. Density loss and respiration rates in coarse woody debris of Pinus radiata, Eucalyptus regnans and Eucalyptus maculata. Soil Biol. Biochem. 2003, 35, 177–186. [Google Scholar] [CrossRef]
- Brown, S.; Mo, J.; McPherson, J.K.; Bell, D. Decomposition of woody debris in western Austalian forest. Can. J. For. Res. 1996, 26, 954–966. [Google Scholar] [CrossRef]
- Yang, F.-F.; Li, Y.-L.; Zhou, G.-Y.; Wenigmann, K.O.; Zhang, D.-Q.; Wenigmann, M.; Liu, S.-Z.; Zhang, Q.-M. Dynamics of coarse woody debris and decomposition rates in an old-growth forest in lower tropical China. For. Ecol. Manag. 2010, 259, 1666–1672. [Google Scholar] [CrossRef]
- Aschmann, H. Distribution and Peculiarity of Mediterranean Ecosystems. In Mediterranean Type Ecosystem Origin and Structure; Di Castri, F., Mooney, H.A., Eds.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1973; pp. 11–19. [Google Scholar]
- Laiho, R.; Prescott, C.E. The contribution of coarse woody debris to carbon, nitrogen, and phosphorus cycles in three rocky mountain coniferous forests. Can. J. For. Res. 1999, 29, 1592–1603. [Google Scholar] [CrossRef]
- Mackensen, J.; Bauhus, J.; Webber, E. Decomposition rates of coarse woody debris—A review with particular emphasis on Australian tree species. Aust. J. Bot. 2003, 51, 27–37. [Google Scholar] [CrossRef]
- Jonsell, M.; Hansson, J.; Wedmo, L. Diversity of saproxylic beetle species in logging residues in Sweden—Comparisons between tree species and diameters. Biol. Conserv. 2007, 138, 89–99. [Google Scholar] [CrossRef]
- Weedon, J.T.; Cornwell, W.K.; Cornelissen, J.H.C.; Zanne, A.E.; Wirth, C.; Coomes, D.A. Global meta-analysis of wood decomposition rates: A role for trait variation among tree species? Ecol. Lett. 2009, 12, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Kitchell, J.F.; O’Neill, R.V.; Webb, D.; Gallepp, G.W.; Bartell, S.M.; Koonce, J.F.; Aumus, B.S. Regulation of nutrient cycling. Bioscience 1979, 29, 28–34. [Google Scholar] [CrossRef]
- Harvey, A.E.; Jurgensen, M.F.; Larsen, M.J. Seasonal distribution of ectomycorrhizae in a mature douglas-fir/larch forest soil in Western Montana. For. Sci. 1978, 24, 203–208. [Google Scholar]
- Erickson, H.E.; Edmonds, R.L.; Peterson, C.E. Decomposition of logging residues in douglas-fir, western hemlock, pacific silver fir, and ponderosa pine ecosystems. Can. J. For. Res. 1985, 15, 914–921. [Google Scholar] [CrossRef]
- Kimmey, J.W.; Furnis, R.L. Deterioration of Fire-Killed Douglas-Fir; Tecnical Bulletin USDA, U.S. Department of Agriculture: Washington, DC, USA, 1943.
- Yatskov, M.; Harmon, M.E.; Krankina, O.N. A chronosequence of wood decomposition in the boreal forests of Russia. Can. J. For. Res. 2003, 33, 1211–1226. [Google Scholar] [CrossRef]
- Johnstone, J.F.; Allen, C.D.; Franklin, J.F.; Frelich, L.E.; Harvey, B.J.; Higuera, P.E.; Mack, M.C.; Meentemeyer, R.K.; Metz, M.R.; Perry, G.L.W.; et al. Changing disturbance regimes, ecological memory, and forest resilience. Front. Ecol. Environ. 2016, 14, 369–378. [Google Scholar] [CrossRef]

| Plot | |||
|---|---|---|---|
| 1 | 2 | 3 | |
| Coordinates 1 | 36°57′12.1″ N 03°29′36.3″ W | 36°58′11.9″ N 03°30′1.7″ W | 36°58′6.5″ N 03°28′49.1″ W |
| Elevation (m above sea level 1 | 1477 | 1698 | 2053 |
| Mean daily minimum temp. (°C) 2 | 6.8 ± 0.2 | 5.6 ± 0.2 | 3.4 ± 0.2 |
| Mean daily maximum temp. (°C) 2 | 17.1 ± 0.2 | 16.2 ± 0.2 | 13.4 ± 0.2 |
| Mean ann. precip. (mm) 2 | 536 ± 41 | 550 ± 40 | 630 ± 42 |
| Dominant species | Pinus pinaster | P. nigra | P. sylvestris |
| Mean log diameter (cm) 3 | 12.6 ± 0.4 | 12.8 ± 0.3 | 10.0 ± 0.2 |
| Term Removed from Model | Likelihood Ratio | p-Value |
|---|---|---|
| Year × Diameter × Plot 1 | 1.63 | 0.44 |
| Year × Diameter 2 | 8.26 | <0.01 |
| Year × Plot 2 | 6.48 | <0.05 |
| Diameter × Plot 2 | 10.78 | <0.01 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molinas-González, C.R.; Castro, J.; Leverkus, A.B. Deadwood Decay in a Burnt Mediterranean Pine Reforestation. Forests 2017, 8, 158. https://doi.org/10.3390/f8050158
Molinas-González CR, Castro J, Leverkus AB. Deadwood Decay in a Burnt Mediterranean Pine Reforestation. Forests. 2017; 8(5):158. https://doi.org/10.3390/f8050158
Chicago/Turabian StyleMolinas-González, Carlos R., Jorge Castro, and Alexandro B. Leverkus. 2017. "Deadwood Decay in a Burnt Mediterranean Pine Reforestation" Forests 8, no. 5: 158. https://doi.org/10.3390/f8050158
APA StyleMolinas-González, C. R., Castro, J., & Leverkus, A. B. (2017). Deadwood Decay in a Burnt Mediterranean Pine Reforestation. Forests, 8(5), 158. https://doi.org/10.3390/f8050158






