The Effect of Harvest on Forest Soil Carbon: A Meta-Analysis
Abstract
:1. Introduction
- (1)
- What is the overall effect of forest harvesting on soil C pools?
- (2)
- How does the effect of forest harvest on soil C change with soil depth?
- (3)
- To what extent does the effect of harvesting differ among soil orders?
- (4)
- Do site pretreatment strategies or increasing harvesting intensity (i.e., whole tree harvest) moderate or accentuate harvesting impacts on soil C?
- (5)
- How long does soil C take to recover from harvest across different soil types?
2. Materials and Methods
3. Results
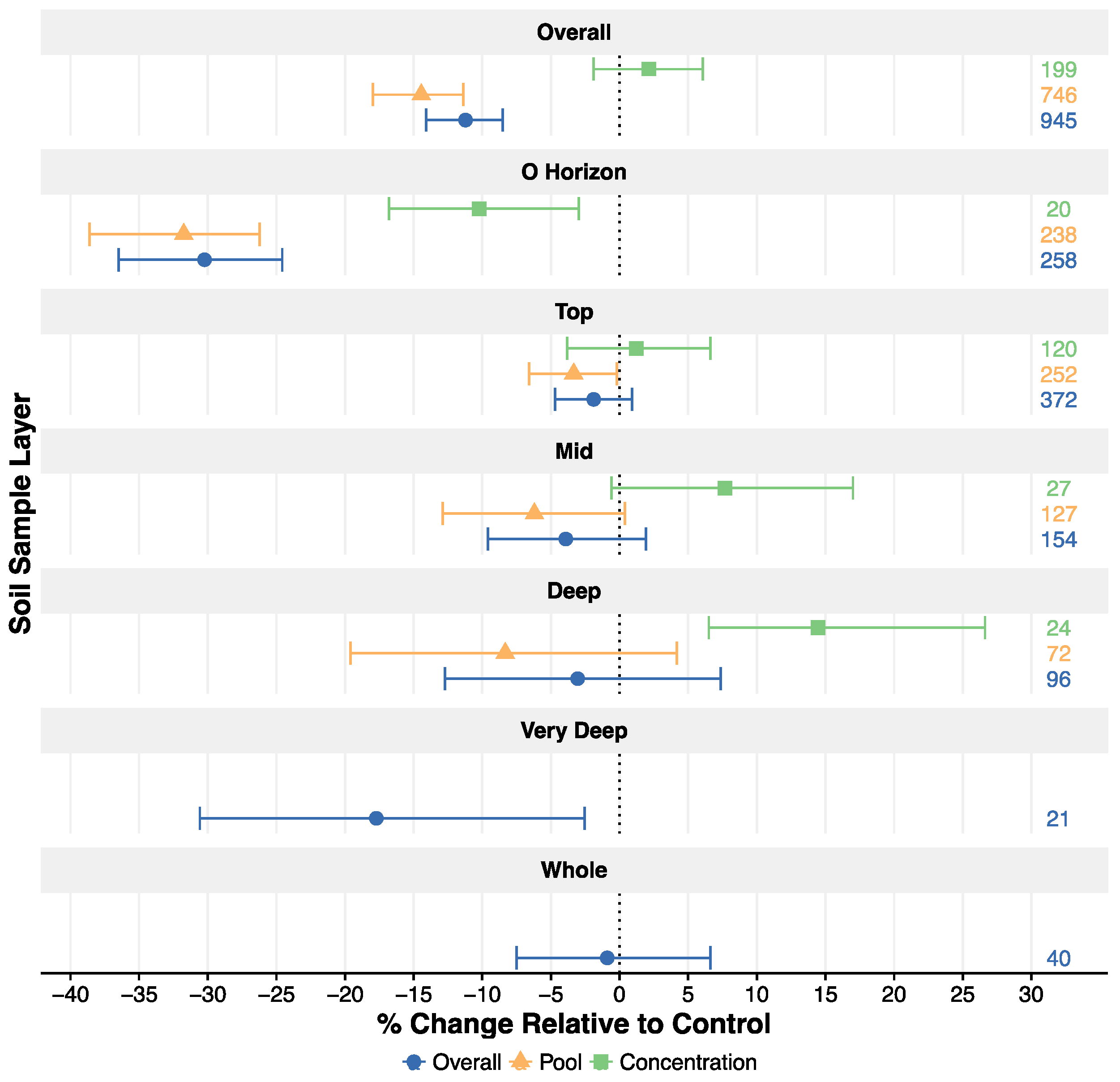
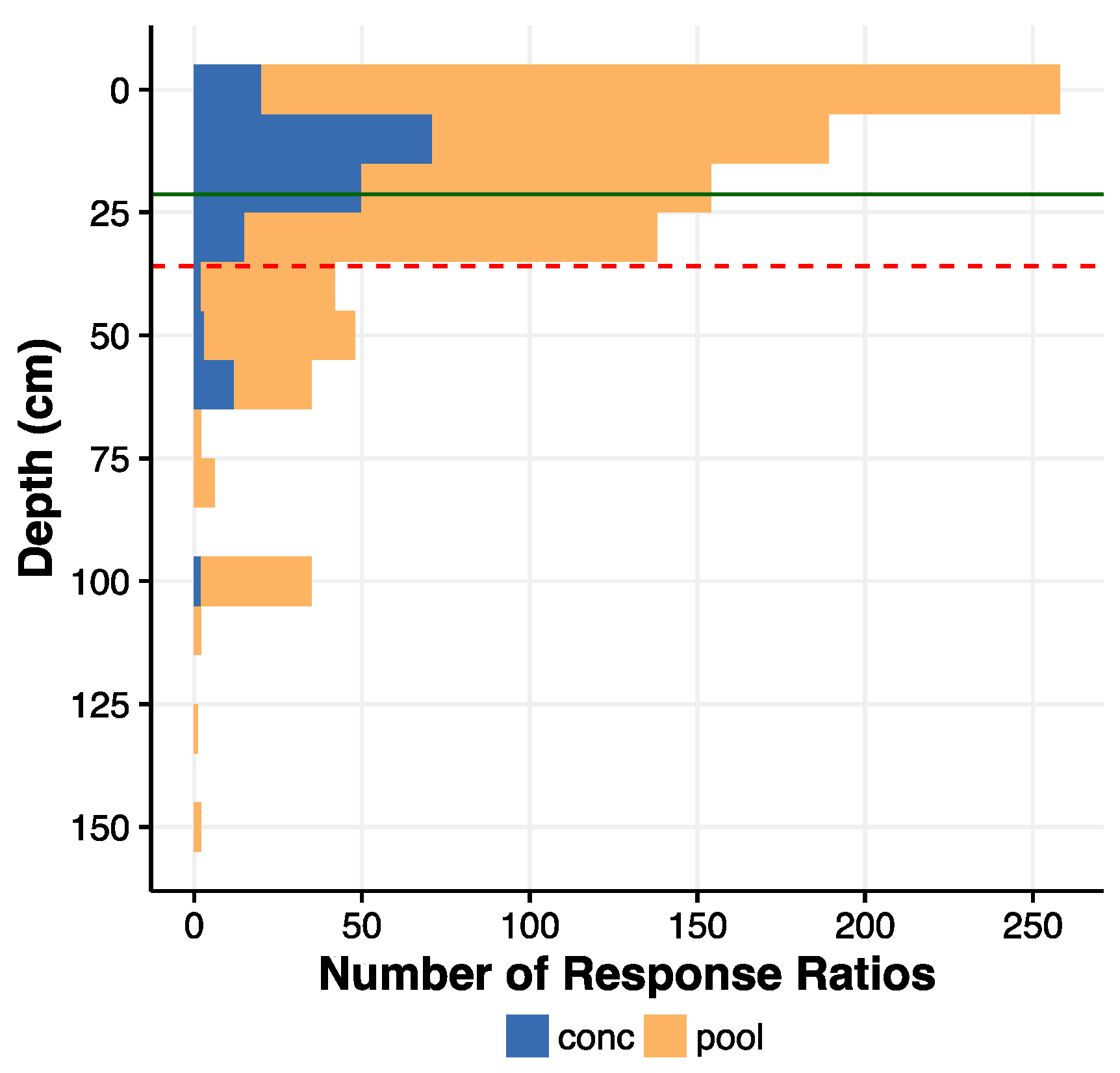
3.1. Overall Effect and Change with Depth
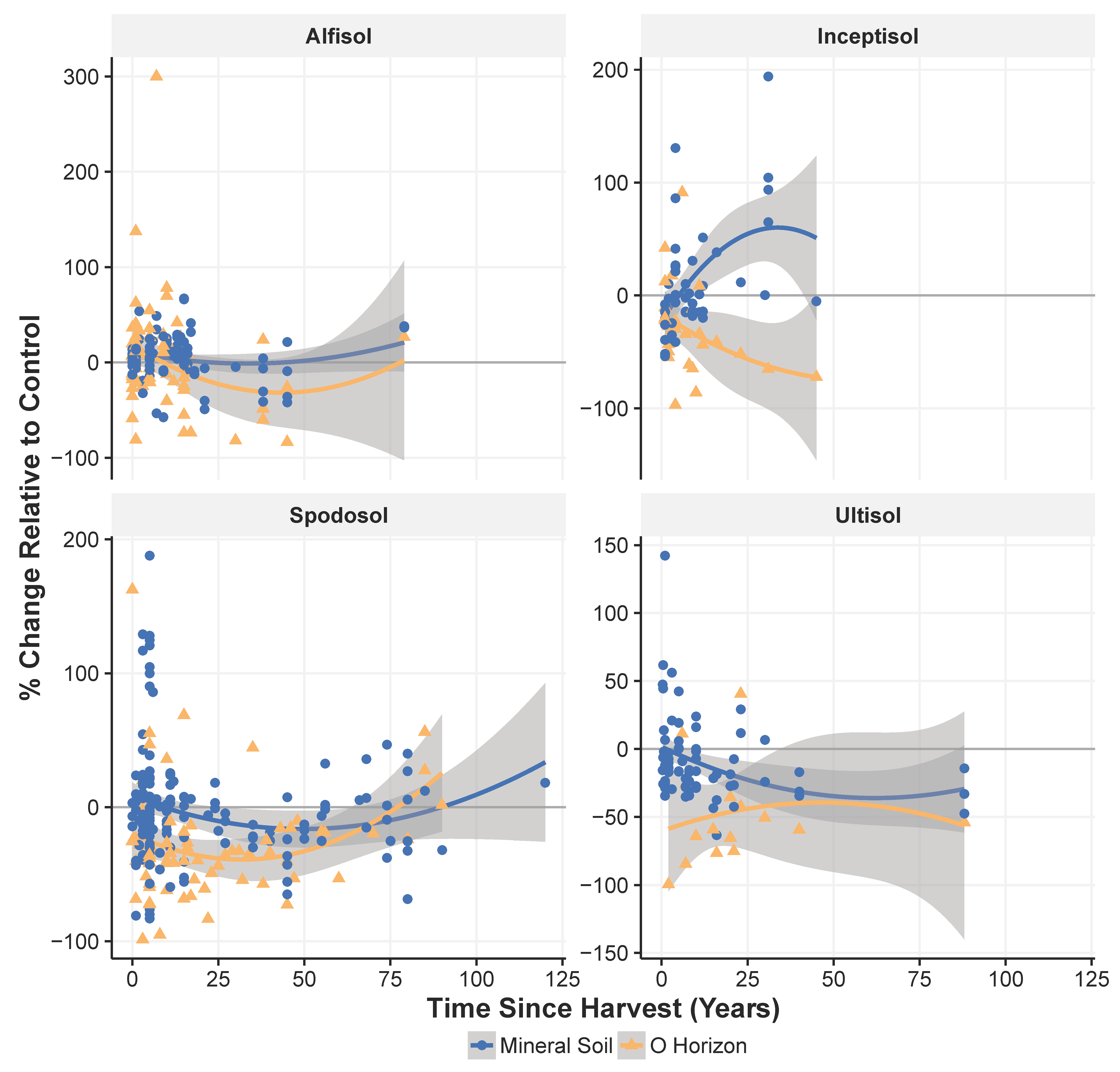
3.2. Effect of Harvesting across Soil Orders
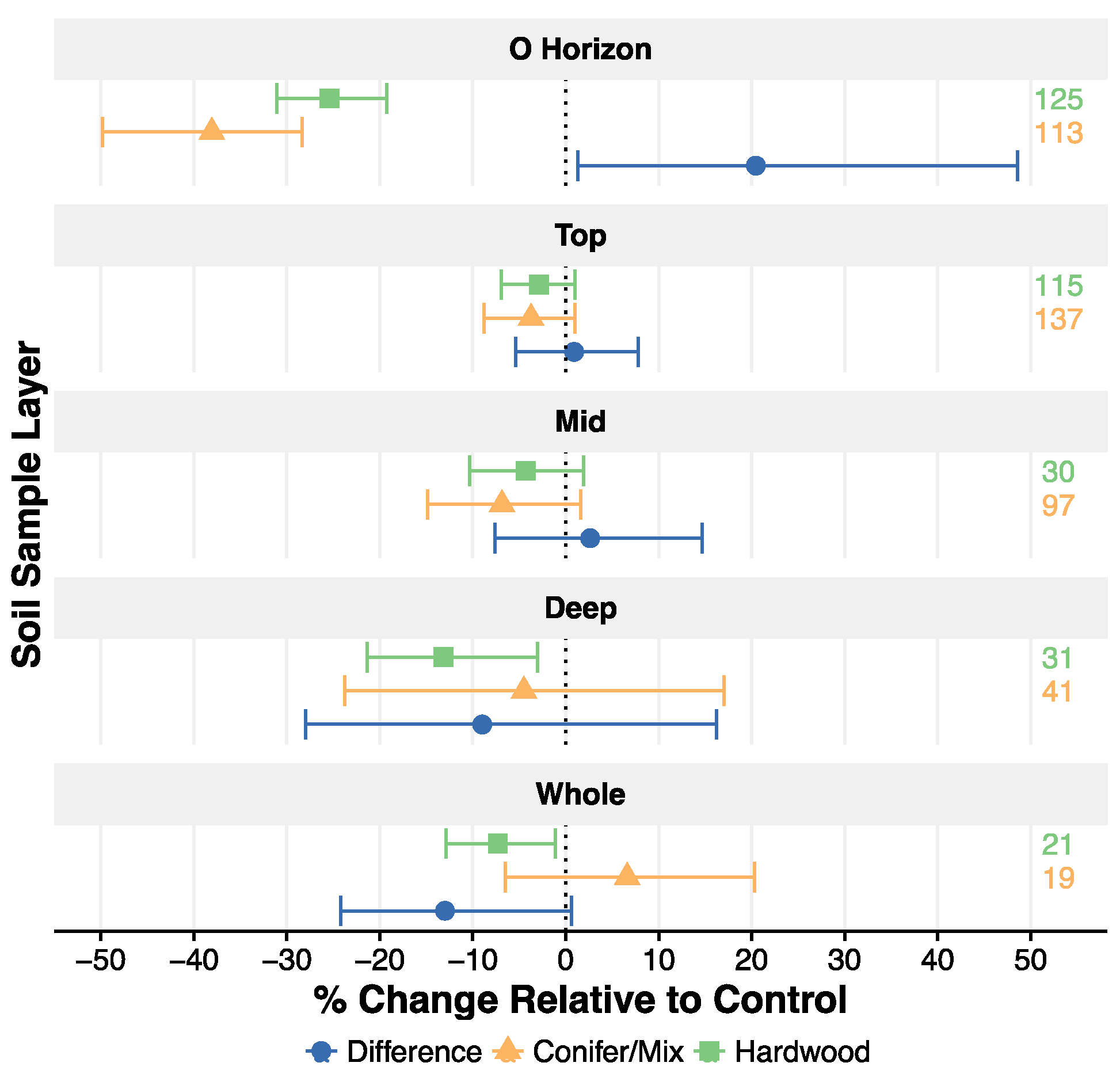
3.3. Differences in Response to Harvest between Forest Types
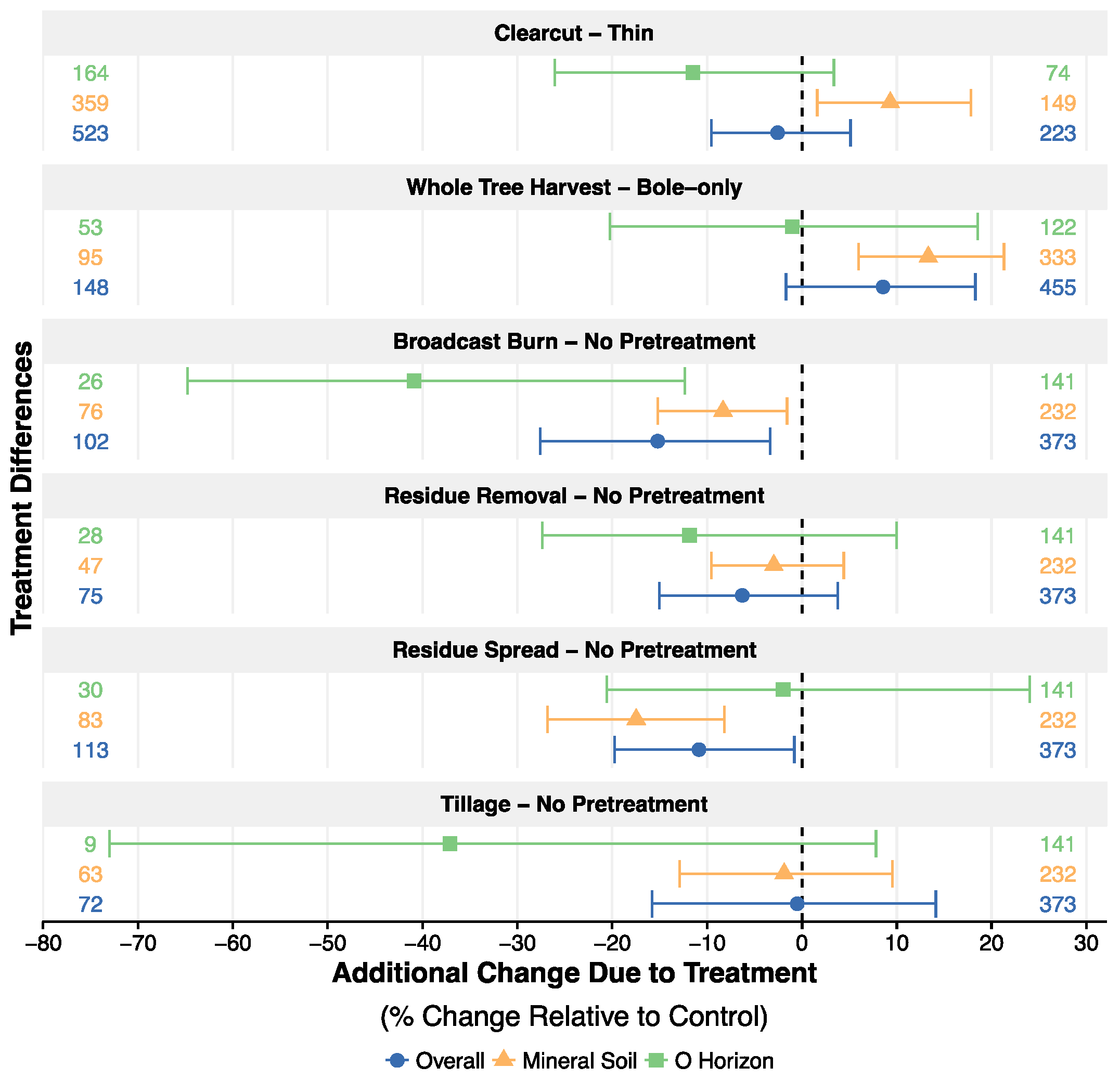
3.4. Harvest Intensity, Residue Management and Site Pretreatment
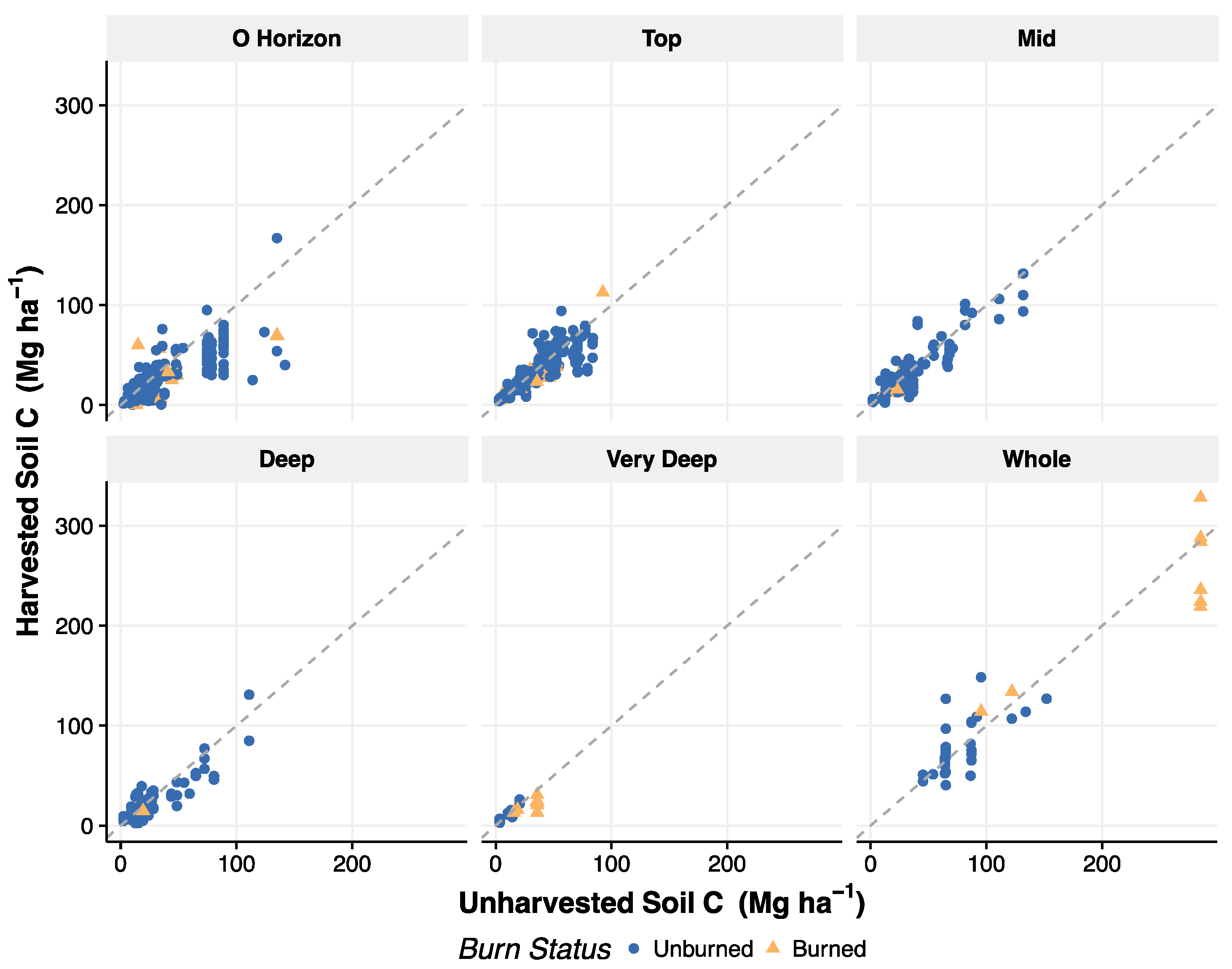
3.5. Recovery of Soil C after Harvest
4. Discussion
4.1. Overall Effect of Harvesting on Soil C
4.2. Depth Distribution of Soil C Response to Harvest
4.3. Differences in Soil C Response to Harvest among Soil Orders
4.4. Recovery of Soil C after Harvest
4.5. The Effect of Harvest Strategies on Soil C
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A. Publications Providing Response Ratios for This Analysis
| Reference | Year | k | Max Depth (cm) | Time Since Harvest a (years) | Location |
|---|---|---|---|---|---|
| Alban and Perala [53] | 1992 | 7 | 50 | 35 | MN, USA |
| Bauhus et al. [54] | 2004 | 6 | 40 | 9 | Germany |
| Bisbing et al. [55] | 2010 | 6 | 100 | 40 | MT, USA |
| Black and Harden [56] | 1995 | 15 | 20 | 23 | CA, USA |
| Boerner et al. [57] | 2006 | 4 | 10 | 2 | SC, USA |
| Borchers and Perry [58] | 1992 | 4 | 15 | 14 | OR, USA |
| Bravo-Oviedo et al. [59] | 2015 | 8 | 30 | 15 | Spain |
| Cade-Menun et al. [60] | 2000 | 12 | 26 | 5 | BC, Canada |
| Carter et al. [61] | 2002 | 8 | 15 | 2 | LA, TX, USA |
| Chatterjee et al. [62] | 2009 | 19 | 54 | 21 | WY, USA |
| Chen et al. [63] | 2013 | 24 | 100 | 29 | China |
| Chiti et al. [64] | 2016 | 24 | 100 | 24 | Ghana, Cameroon, Gabon |
| Christophel et al. [65] | 2013 | 6 | 30 | 15 | Germany |
| Christophel et al. [66] | 2015 | 18 | 30 | 33 | Germany |
| Cromack et al. [67] | 1999 | 1 | 100 | 10 | OR, USA |
| Dai et al. [68] | 2001 | 3 | 70 | 14 | NH, USA |
| DeByle et al. [69] | 1980 | 10 | 5 | 3 | WY, USA |
| Deluca and Zouhar [52] | 2000 | 6 | 8 | 5 | MT, USA |
| Diochon et al. [70] | 2009 | 28 | 50 | 35 | NS, Canada |
| Edmonds and McColl [71] | 1989 | 4 | 20 | 3 | Australia |
| Edwards and Ross-Todd [72] | 1983 | 6 | 45 | 1 | TN, USA |
| Elliott and Knoepp [73] | 2005 | 3 | 15 | 3 | NC, USA |
| Ellis et al. [73] | 1982 | 4 | 10 | 2 | Tasmania |
| Ellis and Graley [74] | 1983 | 2 | 10 | 1 | Tasmania |
| Esquilin et al. [75] | 2008 | 1 | 10 | 14 | CO, USA |
| Falsone et al. [76] | 2012 | 3 | 130 | 5 | Russia |
| Fraterrigo et al. [77] | 2005 | 1 | 15 | 30 | NC, USA |
| Frazer et al. [78] | 1990 | 4 | 14 | 12 | CA, USA |
| Gartzia-Bengoetxea et al. [79] | 2009 | 2 | 5 | 10 | Spain |
| Gillon et al. [80] | 1999 | 2 | 0 | 1 | France |
| Goh and Phillips [81] | 1991 | 4 | 60 | 2 | New Zealand |
| Goodale and Aber [82] | 2001 | 2 | 10 | 85 | NH, USA |
| Gough et al. [83] | 2007 | 15 | 80 | 41 | MI, USA |
| Grady and Hart [84] | 2006 | 2 | 15 | 12 | AZ, USA |
| Grand and Lavkulich [85] | 2012 | 6 | 80 | BC, Canada | |
| Gresham [86] | 2002 | 6 | 30 | 10 | SC, USA |
| Griffiths and Swanson [87] | 2001 | 3 | 10 | 20 | OR, USA |
| Gundale et al. [88] | 2005 | 4 | 10 | 3 | MT, USA |
| Gupta and DeLuca [89] | 2012 | 12 | 50 | 5 | Wales |
| Hart et al. [90] | 2006 | 2 | 15 | 1 | AZ, USA |
| Hendrickson and Chattarpaul [91] | 1989 | 6 | 20 | 3 | ON, Canada |
| Herman et al. [92] | 2003 | 2 | 9 | 8 | CA, USA |
| Holscher et al. [93] | 2001 | 2 | 20 | 22 | Germany |
| Hwang and Son [94] | 2006 | 2 | 30 | 2 | Korea |
| Jang and Page-Dumroese [95] | 2015 | 8 | 30 | 38 | MT, USA |
| Johnson [96] | 1991 | 3 | 20 | 3 | NH, USA |
| Johnson and Todd [97] | 1998 | 6 | 45 | 15 | TN, USA |
| Johnson [98] | 1995 | 12 | 7 | NH, USA | |
| Johnson et al. [99] | 1997 | 14 | 53 | 6 | NH, USA |
| Johnson et al. [100] | 2014 | 4 | 60 | 1 | CA, USA |
| Jones et al. [101] | 2011 | 12 | 30 | 15 | New Zealand |
| Kaye and Hart [102] | 1998 | 2 | 15 | 1 | AZ, USA |
| Keenan et al. [103] | 1994 | 1 | 20 | 4 | BC, Canada |
| Kelliher et al. [104] | 2004 | 4 | 50 | 22 | OR, USA |
| Kishchuk et al. [105] | 2014 | 4 | 7 | 6 | AB, Canada |
| Klockow et al. [106] | 2013 | 9 | 20 | 1 | MN, USA |
| Klopatek [107] | 2002 | 6 | 20 | 30 | WA, USA |
| Knoepp and Swank [108] | 1997 | 4 | 30 | 33 | NC, USA |
| Korb et al. [109] | 2004 | 1 | 10 | 1 | AZ, USA |
| Kraemer and Hermann [110] | 1979 | 2 | 10 | 26 | WA, USA |
| Kurth et al. [111] | 2014 | 72 | 30 | 8 | MI, MN, USA |
| Laiho et al. [112] | 2003 | 5 | 22 | 5 | NC, LA, USA |
| Latty et al. [113] | 2004 | 2 | 15 | 90 | NY, USA |
| Law et al. [114] | 2001 | 3 | 100 | 21 | OR, USA |
| Law et al. [115] | 2003 | 9 | 100 | 62 | OR, USA |
| Leduc and Rothstein [116] | 2007 | 1 | 10 | 5 | MI, USA |
| Maassen and Wirth [117] | 2004 | 2 | 5 | Germany | |
| Mattson and Smith [118] | 1993 | 30 | 10 | 11 | WV, USA |
| Mattson and Swank [119] | 1989 | 8 | 60 | 5 | NC, USA |
| May and Attiwill [120] | 2003 | 2 | 10 | 5 | Australia |
| McLaughlin and Phillips [121] | 2006 | 2 | 50 | 17 | ME, USA |
| McKee et al. [122] | 2013 | 8 | 60 | 24 | AL, USA |
| McLaughlin [123] | 1996 | 10 | 50 | 5 | MI, USA |
| Merino and Edeso [124] | 1999 | 6 | 15 | 1 | Spain |
| Moreno-Fernandez et al. [125] | 2015 | 54 | 50 | 60 | Spain |
| Mu et al. [126] | 2013 | 18 | 50 | 5 | China |
| Murphy et al. [127] | 2006 | 20 | 60 | 1 | CA, USA |
| Neher et al. [128] | 2003 | 3 | 20 | 2 | NC, USA |
| Norris et al. [129] | 2009 | 15 | 100 | 16 | SK, Canada |
| O’Brien et al. [130] | 2003 | 6 | 50 | 18 | Australia |
| Powers et al. [131] | 2011 | 20 | 30 | 13 | MN, WI, USA |
| Prest et al. [132] | 2014 | 5 | 50 | 35 | NS, Canada |
| Prietzel et al. [133] | 2004 | 4 | 0 | 1 | WA, USA |
| Puhlick et al. [134] | 2016 | 10 | 100 | ME, USA | |
| Rab [135] | 1996 | 8 | 10 | 1 | Australia |
| Riley and Jones [136] | 2003 | 3 | 10 | 1 | SC, USA |
| Roaldson et al. [137] | 2014 | 16 | 20 | 5 | CA, USA |
| Rothstein and Spaulding [138] | 2010 | 6 | 30 | MI, USA | |
| Sanchez et al. [139] | 2007 | 6 | 105 | 2 | SC, USA |
| Sanscrainte et al. [140] | 2003 | 4 | 70 | WA, USA | |
| Saynes et al. [141] | 2012 | 8 | 5 | 11 | Mexico |
| Selig et al. [142] | 2008 | 3 | 30 | 14 | VA, USA |
| Shelburne et al. [143] | 2004 | 4 | 10 | 1 | SC, USA |
| Sheng et al. [144] | 2015 | 5 | 100 | 8 | China |
| Skovsgaard et al. [145] | 2006 | 12 | 30 | 0 | Denmark |
| Slesak et al. [146] | 2012 | 12 | 60 | 5 | OR, WA, USA |
| Small and McCarthy [147] | 2005 | 3 | 10 | 7 | OH, USA |
| Stone et al. [148] | 1999 | 1 | 15 | 1 | AZ, USA |
| Stone and Elioff [149] | 1998 | 4 | 30 | 5 | MN, USA |
| Strong [150] | 1997 | 8 | 40 | 18 | MN, USA |
| Strukelj et al. [151] | 2015 | 12 | 10 | 5 | QC, Canada |
| Tang et al. [152] | 2009 | 12 | 60 | 29 | MI, WI, USA |
| Trettin et al. [153] | 2011 | 6 | 150 | 11 | MI, USA |
| Ussiri and Johnson [154] | 2007 | 15 | 60 | 8 | NH, USA |
| Vario et al. [155] | 2014 | 6 | 60 | 49 | NH, USA |
| Vesterdal et al. [156] | 1995 | 9 | 0 | Denmark | |
| Waldrop et al. [157] | 2003 | 3 | 0 | 1 | CA, USA |
| Wu et al. [158] | 2010 | 1 | 20 | 10 | China |
| Xiang et al. [159] | 2009 | 8 | 30 | 0 | China |
| Yanai et al. [160] | 2000 | 35 | 0 | 29 | NH, USA |
| Zabowski et al. [161] | 2008 | 2 | 20 | 25 | OR, WA, USA |
| Zhong and Makeshin [162] | 2003 | 2 | 10 | 16 | Germany |
| Zummo and Friedland [163] | 2011 | 15 | 60 | 3 | NH, USA |
References
- Dixon, R.K.; Brown, S.; Houghton, R.A.; Solomon, A.M.; Trexler, M.C.; Wisniewski, J. Carbon Pools and Flux of Global Forest Ecosystems. Science 1994, 263, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Prentice, I.C.; Farquhar, G.; Fasham, M.J.R.; Goulden, M.L.; Heimann, M.; Jarmillo, V.J.; Kheshgi, H.S.; Le Quere, C.; Scholes, R.J.; Wallace, D.W.R. The Carbon Cycle and Atmospheric Carbon Dioxide; Cambridge University Press: Cambridge, UK, 2001; pp. 183–237. [Google Scholar]
- Batjes, N.H. Total carbon and nitrogen in the soils of the world. Eur. J. Soil Sci. 1996, 47, 151–163. [Google Scholar] [CrossRef]
- Jobbagy, E.G.; Jackson, R.B. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol. Appl. 2000, 10, 423–436. [Google Scholar] [CrossRef]
- Six, J.; Conant, R.T.; Paul, E.A.; Paustian, K. Stabilization mechanisms of soil organic matter: Implications for C-saturation of soils. Plant Soil 2002, 241, 155–176. [Google Scholar] [CrossRef]
- Lal, R. Forest soils and carbon sequestration. For. Ecol. Manag. 2005, 220, 242–258. [Google Scholar] [CrossRef]
- Schimel, D.S. Terrestrial ecosystems and the carbon cycle. Glob. Chang. Biol. 1995, 1, 77–91. [Google Scholar] [CrossRef]
- Jandl, R.; Lindner, M.; Vesterdal, L.; Bauwens, B.; Baritz, R.; Hagedorn, F.; Johnson, D.W.; Minkkinen, K.; Byrne, K.A. How strongly can forest management influence soil carbon sequestration? Geoderma 2007, 137, 253–268. [Google Scholar] [CrossRef]
- Nave, L.E.; Vance, E.D.; Swanston, C.W.; Curtis, P.S. Harvest impacts on soil carbon storage in temperate forests. For. Ecol. Manag. 2010, 259, 857–866. [Google Scholar] [CrossRef]
- Schlesinger, W.H.; Bernhardt, E.S. Biogeochemistry: An Analysis of Global Change; Academic Press: Oxford, UK, 2013. [Google Scholar]
- Brady, N.C.; Weil, R.R. The Nature and Properties of Soils; Macmillan Publ. Co.: New York, NY, USA, 2002; Volume 13, p. 960. [Google Scholar]
- Krull, E.S.; Skjemstad, J.O.; Baldock, J.A. Functions of Soil Organic Matter and the Effect on Soil Properties; Final Rep. CSO00029; Grains Research and Development Corporation: Glen Osmond, South Australia, 2009; p. 128. [Google Scholar]
- Six, J.; Paustian, K. Aggregate-associated soil organic matter as an ecosystem property and a measurement tool. Soil Biol. Biochem. 2014, 68, A4–A9. [Google Scholar] [CrossRef]
- Buchholz, T.; Friedland, A.J.; Hornig, C.E.; Keeton, W.S.; Zanchi, G.; Nunery, J. Mineral soil carbon fluxes in forests and implications for carbon balance assessments. Glob. Chang. Biol. Bioenergy 2014, 6, 305–311. [Google Scholar] [CrossRef]
- Ryan, M.G.; Harmon, M.E.; Birdsey, R.A.; Giardina, C.P.; Heath, L.S.; Houghton, R.A.; Jackson, R.B.; McKinley, D.C.; Morrison, J.F.; Murray, B.C.; et al. A synthesis of the science on forests and carbon for U.S. Forests. Issues Ecol. 2010, 13, 1–16. [Google Scholar]
- McKinley, D.C.; Ryan, M.G.; Birdsey, R.A.; Giardina, C.P.; Harmon, M.E.; Heath, L.S.; Houghton, R.A.; Jackson, R.B.; Morrison, J.F.; Murray, B.C.; et al. A synthesis of current knowledge on forests and carbon storage in the United States. Ecol. Appl. 2011, 21, 1902–1924. [Google Scholar] [CrossRef] [PubMed]
- Fahey, T.J.; Woodbury, P.B.; Battles, J.J.; Goodale, C.L.; Hamburg, S.P.; Ollinger, S.V.; Woodall, C.W. Forest carbon storage: Ecology, management, and policy. Front. Ecol. Environ. 2010, 8, 245–252. [Google Scholar] [CrossRef]
- American Carbon Registry. The ACR Forest Carbon Project Standard v 2.1; Winrock International: Arlington, VA, USA, 2011. [Google Scholar]
- Verified Carbon Standard. Agriculture, Forestry and other Land Use (AFOLU) Requirements; VCS Version 3 Requirements Doc., v3.4 2013; Verified Carbon Standard: Washington, DC, USA, 2013. [Google Scholar]
- IPCC. 2006 IPCC Guidelines for national greenhouse gas inventories. In Agriculture, Forestry and Other Land Use; Institute for Global Environmental Strategies: Hayama, Japan, 2006; Volume 4, pp. 1–29. [Google Scholar]
- Bechtold, W.A.; Patterson, P.L. The Enhanced Forest Inventory and Analysis Program—National Sampling Design and Estimation Procedures. In USDA General Technical Report SRS-80; U.S. Department of Agriculture, Forest Service, Southern Research Station: Asheville, NC, USA, 2005; p. 85. [Google Scholar]
- Zanchi, G.; Pena, N.; Bird, N. Is woody bioenergy carbon neutral? A comparative assessment of emissions from consumption of woody bioenergy and fossil fuel. GCB Bioenergy 2012, 4, 761–772. [Google Scholar] [CrossRef]
- Homann, P.S.; Bormann, B.T.; Boyle, J.R. Detecting treatment differences in soil carbon and nitrogen resulting from forest manipulations. Soil Sci. Soc. Am. J. 2001, 65, 463–469. [Google Scholar] [CrossRef]
- Hedges, L.V.; Gurevitch, J.; Curtis, P.S. The meta-analysis of response ratios in experimental ecology. Ecology 1999, 80, 1150–1156. [Google Scholar] [CrossRef]
- Johnson, D.W.; Curtis, P.S. Effects of forest management on soil C and N storage: Meta analysis. For. Ecol. Manag. 2001, 140, 227–238. [Google Scholar] [CrossRef]
- Harrison, R.B.; Footen, P.W.; Strahm, B.D. Deep soil horizons: Contribution and importance to soil carbon pools and in assessing whole-ecosystem response to management and global change. For. Sci. 2011, 57, 67–76. [Google Scholar]
- Schmidt, M.W.I.; Torn, M.S.; Abiven, S.; Dittmar, T.; Guggenberger, G.; Janssens, I.A.; Kleber, M.; Koegel-Knabner, I.; Lehmann, J.; Manning, D.A.C.; et al. Persistence of soil organic matter as an ecosystem property. Nature 2011, 478, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Deb Richter, D.; Yaalon, D.H. “The Changing Model of Soil” Revisited. Soil Sci. Soc. Am. J. 2012, 76, 76–778. [Google Scholar] [CrossRef]
- Marin-Spiotta, E.; Gruley, K.E.; Crawford, J.; Atkinson, E.E.; Miesel, J.R.; Greene, S.; Cardona-Correa, C.; Spencer, R.G.M. Paradigm shifts in soil organic matter research affect interpretations of aquatic carbon cycling: Transcending disciplinary and ecosystem boundaries. Biogeochemistry 2014, 117, 279–297. [Google Scholar] [CrossRef]
- Lehmann, J.; Kleber, M. The contentious nature of soil organic matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, S.; Barot, S.; Barre, P.; Bdioui, N.; Mary, B.; Rumpel, C. Stability of organic carbon in deep soil layers controlled by fresh carbon supply. Nature 2007, 450, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.C.; Gurevitch, J.; Rosenberg, M.S. Resampling tests for meta-analysis of ecological data. Ecology 1997, 78, 1277–1283. [Google Scholar] [CrossRef]
- Gerlanc, D.; Kirby, K. bootES: Bootstrap Effect Sizes, R Package, Version 1.2; R Foundation for Statistical Computing: Vienna, Austria, 2015.
- R Development Core Team. A Language and Environment for Statistical Computing, R Foundation for Statistical Computing: Vienna, Austria, 2016.
- Currie, W.S. Responsive C and N biogeochemistryof the temperate forest floor. Trends Ecol. Evol. 1999, 14, 316–320. [Google Scholar] [CrossRef]
- Silver, W.; Miya, R. Global patterns in root decomposition: Comparisons of climate and litter quality effects. Oecologia 2001, 129, 407–419. [Google Scholar] [CrossRef]
- James, J.; Devine, W.; Harrison, R.; Terry, T. Deep soil carbon: quantification and modeling in subsurface layers. Soil Sci. Soc. Am. J. 2014, 78, S1–S9. [Google Scholar] [CrossRef]
- Richter, D.D.; Markewitz, D. How Deep Is Soil? Bioscience 1995, 45, 600–609. [Google Scholar] [CrossRef]
- Canadell, J.; Jackson, R.B.; Ehleringer, J.R.; Mooney, H.A.; Sala, O.E.; Schulze, E.D. Maximum rooting depth of vegetation types at the global scale. Oecologia 1996, 108, 583–595. [Google Scholar] [CrossRef]
- Strahm, B.D.; Harrison, R.B.; Terry, T.A.; Flaming, B.L.; Licata, C.W.; Petersen, K.S. Soil solution nitrogen concentrations and leaching rates as influenced by organic matter retention on a highly productive Douglas-fir site. For. Ecol. Manag. 2005, 218, 74–88. [Google Scholar] [CrossRef]
- Likens, G.E.; Bormann, F.H.; Johnson, N.M. Nitrification—Importance to nutrient losses from a cutover forested ecosystem. Science 1969, 163, 1205–1206. [Google Scholar] [CrossRef] [PubMed]
- Kuzyakov, Y. Priming effects: Interactions between living and dead organic matter. Soil Biol. Biochem. 2010, 42, 1363–1371. [Google Scholar] [CrossRef]
- Blagodatskaya, E.; Kuzyakov, Y. Mechanisms of real and apparent priming effects and their dependence on soil microbial biomass and community structure: Critical review. Biol. Fertil. Soils 2008, 45, 115–131. [Google Scholar] [CrossRef]
- Mobley, M.L.; Lajtha, K.; Kramer, M.G.; Bacon, A.R.; Heine, P.R.; Richter, D.D. Surficial gains and subsoil losses of soil carbon and nitrogen during secondary forest development. Glob. Chang. Biol. 2015, 21, 986–996. [Google Scholar] [CrossRef] [PubMed]
- Soil Survey Staff. Keys to Soil Taxonomy; Department of Agriculture, Natural Resources Conservation Service: Washington, DC, USA, 2014.
- Wada, K. Minerals formed and mineral formation from volcanic ash by weathering. Chem. Geol. 1987, 60, 17–28. [Google Scholar] [CrossRef]
- Nanzyo, M.; Shoji, S.; Dahlgren, R. Volcanic Ash Soils—Genesis, Properties and Utilization; Elsevier: Amsterdam, The Netherlands, 1993; Volume 21. [Google Scholar]
- Blagodatskaya, E.V.; Blagodatsky, S.A.; Anderson, T.H.; Kuzyakov, Y. Priming effects in Chernozem induced by glucose and N in relation to microbial growth strategies. Appl. Soil Ecol. 2007, 37, 95–105. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Friedel, J.K.; Stahr, K. Review of mechanisms and quantification of priming effects. Soil Biol. Biochem. 2000, 32, 1485–1498. [Google Scholar] [CrossRef]
- Valette, J.; Gomendy, V.; Marechal, J.; Houssard, C.; Gillon, D. Heat Transfer in the Soil During Very Low-intensity Experimental Fires: The Role of Duff and Soil Moisture Content. Int. J. Wildland Fire 1994, 4, 225–237. [Google Scholar] [CrossRef]
- Certini, G. Effects of fire on properties of forest soils: A review. Oecologia 2005, 143, 1–10. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, T.H.; Zouhar, K.L. Effects of selection harvest and prescribed fire on the soil nitrogen status of ponderosa pine forests. For. Ecol. Manag. 2000, 138, 263–271. [Google Scholar] [CrossRef]
- Alban, D.H.; Perala, D.A. Carbon storage in lake states aspen ecosystems. Can. J. For. Res. 1992, 22, 1107–1110. [Google Scholar] [CrossRef]
- Bauhus, J.; Vor, T.; Bartsch, N.; Cowling, A. The effects of gaps and liming on forest floor decomposition and soil C and N dynamics in a Fagus sylvatica forest. Can. J. For. Res. 2004, 34, 509–518. [Google Scholar] [CrossRef]
- Bisbing, S.M.; Alaback, P.B.; DeLuca, T.H. Carbon storage in old-growth and second growth fire-dependent western larch (Larix occidentalis Nutt.) forests of the Inland Northwest, USA. For. Ecol. Manag. 2010, 259, 1041–1049. [Google Scholar] [CrossRef]
- Black, T.A.; Harden, J.W. Effect of timber harvest on soil carbon storage at Blodgett Experimental Forest, California. Can. J. For. Res. 1995, 25, 1385–1396. [Google Scholar] [CrossRef]
- Boerner, R.E.J.; Waldrop, T.A.; Shelburne, V.B. Wildfire mitigation strategies affect soil enzyme activity and soil organic carbon in loblolly pine (Pinus taeda) forests. Can. J. For. Res. 2006, 36, 3148–3154. [Google Scholar] [CrossRef]
- Borchers, J.G.; Perry, D.A. The influence of soil texture and aggregation on carbon and nitrogen dynamics in southwest Oregon forest and clearcuts. Can. J. For. Res. 1992, 22, 298–305. [Google Scholar] [CrossRef]
- Bravo-Oviedo, A.; Ruiz-Peinado, R.; Modrego, P.; Alonso, R.; Montero, G. Forest thinning impact on carbon stock and soil condition in Southern European populations of P. sylvestris L. For. Ecol. Manag. 2015, 357, 259–267. [Google Scholar] [CrossRef]
- Cade-Menun, B.J.; Berch, S.M.; Preston, C.M.; Lavkulich, L.M. Phosphorus forms and related soil chemistry of Podzolic soils on northern Vancouver Island. II. The effects of clear-cutting and burning. Can. J. For. Res. 2000, 30, 1726–1741. [Google Scholar] [CrossRef]
- Carter, M.C.; Dean, T.J.; Zhou, M.; Messina, M.G.; Wang, Z. Short-term changes in soil C, N, and biota following harvesting and regeneration of loblolly pine (Pinus taeda L.). For. Ecol. Manag. 2002, 164, 67–88. [Google Scholar] [CrossRef]
- Chatterjee, A.; Vance, G.F.; Tinker, D.B. Carbon pools of managed and unmanaged stands of ponderosa and lodgepole pine forests in Wyoming. Can. J. For. Res. 2009, 39, 1893–1900. [Google Scholar] [CrossRef]
- Chen, L.-C.; Liang, M.-J.; Wang, S.-L. Carbon stock density in planted versus natural Pinus massoniana forests in sub-tropical China. Ann. For. Sci. 2016, 73, 461–472. [Google Scholar] [CrossRef]
- Chiti, T.; Perugini, L.; Vespertino, D.; Valentini, R. Effect of selective logging on soil organic carbon dynamics in tropical forests in central and western Africa. Plant Soil 2015, 399, 283–294. [Google Scholar] [CrossRef]
- Christophel, D.; Spengler, S.; Schmidt, B.; Ewald, J.; Prietzel, J. Customary selective harvesting has considerably decreased organic carbon and nitrogen stocks in forest soils of the Bavarian Limestone Alps. For. Ecol. Manag. 2013, 305, 167–176. [Google Scholar] [CrossRef]
- Christophel, D.; Höllerl, S.; Prietzel, J.; Steffens, M. Long-term development of soil organic carbon and nitrogen stocks after shelterwood- and clear-cutting in a mountain forest in the Bavarian Limestone Alps. Eur. J. For. Res. 2015, 134, 623–640. [Google Scholar] [CrossRef]
- Cromack, K.; Miller, R.E.; Anderson, H.W.; Helgerson, O.T.; Smith, R.B. Soil Carbon and Nutrients in a Coastal Oregon Douglas-Fir Plantation with Red Alder. Soil Sci. Soc. Am. J. 1999, 63, 232–239. [Google Scholar] [CrossRef]
- Dai, K.H.; Johnson, C.E.; Driscoll, C.T. Organic matter chemistry and dynamics in clear-cut and unmanaged hardwood forest ecosystems. Biogeochemistry 2001, 54, 51–83. [Google Scholar] [CrossRef]
- DeByle, N.V. Harvesting and Site Treatment Influences on the Nutrient Status of Lodgepole Pine Forests in Western Wyoming. In General Technical Report INT-90; USDA Forest Service Intermountain Forest And Range Experimental Station: Ogden, UT, USA, 1979; pp. 137–156. [Google Scholar]
- Diochon, A.; Kellman, L.; Beltrami, H. Looking deeper: An investigation of soil carbon losses following harvesting from a managed northeastern red spruce (Picea rubens Sarg.) forest chronosequence. For. Ecol. Manag. 2009, 257, 413–420. [Google Scholar] [CrossRef]
- Edmonds, R.L.; McColl, G.J. Effects of forest management on soil nitrogen in Pinus radiata stands in the Australian Capital Territory. For. Ecol. Manag. 1989, 29, 199–212. [Google Scholar] [CrossRef]
- Edwards, N.T.; Ross-Todd, B.M. Soil carbon dynamics in a mixed deciduous forest following clear-cutting with and without residue removal. Soil Sci. Soc. Am. J. 1983, 47, 1014–1021. [Google Scholar] [CrossRef]
- Elliott, K.J.; Knoepp, J.D. The effects of three regeneration harvest methods on plant diversity and soil characteristics in the southern Appalachians. For. Ecol. Manag. 2005, 211, 296–317. [Google Scholar] [CrossRef]
- Ellis, R.; Graley, A.M. Gains and losses in soil nutrients associated with harvesting and burning eucalypt rainforest. Plant Soil 1983, 74, 437–450. [Google Scholar] [CrossRef]
- Esquilín, J.; Aida, E.; Stromberger, M.E.; Shepperd, W.D. Soil scarification and wildfire interactions and effects on microbial communities and carbon. Soil Sci. Soc. Am. J. 2008, 72, 111–118. [Google Scholar] [CrossRef]
- Falsone, G.; Celi, L.; Caimi, A.; Simonov, G.; Bonifacio, E. The effect of clear cutting on podzolisation and soil carbon dynamics in boreal forests (Middle Taiga zone, Russia). Geoderma 2012, 177–178, 27–38. [Google Scholar] [CrossRef]
- Fraterrigo, J.; Turner, M.; Pearson, S.M.; Dixon, P. Effects of past land use on spatial heterogeneity of soil nutrients in southern Appalachian forests. Ecol. Monogr. 2005, 75, 215–230. [Google Scholar] [CrossRef]
- Frazer, D.W.; McColl, J.G.; Powers, R.F. Soil Nitrogen Mineralization in a Clearcutting Chronosequence in a Northern California Conifer Forest. Soil Sci. Soc. Am. J. 1990, 54, 1145. [Google Scholar] [CrossRef]
- Gartzia-Bengoetxea, N.; González-Arias, A.; Merino, A.; De Arano, I.M. Soil organic matter in soil physical fractions in adjacent semi-natural and cultivated stands in temperate Atlantic forests. Soil Biol. Biochem. 2009, 41, 1674–1683. [Google Scholar] [CrossRef]
- Gillon, D.; Houssard, C.; Valette, J.C.; Rigolot, E. Nitrogen and phosphorus cycling following prescribed burning in natural and managed Aleppo pine forests. Can. J. For. Res. 1999, 29, 1237–1247. [Google Scholar] [CrossRef]
- Goh, K.M.; Phillips, M.J. Effects of clearfell logging and clearfell logging and burning of a Nothofagus forest on soil nutrient dynamics in South Island, New Zealand—Changes in forest floor organic matter and nutrient status. N. Z. J. Bot. 1991, 29, 367–384. [Google Scholar] [CrossRef]
- Goodale, C.L.; Aber, J.D. The Long-term effects of land-use history on nitrogen cycling in northern hardwood forests. Ecol. Appl. 2001, 11, 253–267. [Google Scholar] [CrossRef]
- Gough, C.M.; Vogel, C.S.; Harrold, K.H.; George, K.; Curtis, P.S. The legacy of harvest and fire on ecosystem carbon storage in a north temperate forest. Glob. Chang. Biol. 2007, 13, 1935–1949. [Google Scholar] [CrossRef]
- Grady, K.C.; Hart, S.C. Influences of thinning, prescribed burning, and wildfire on soil processes and properties in southwestern ponderosa pine forests: A retrospective study. For. Ecol. Manag. 2006, 234, 123–135. [Google Scholar] [CrossRef]
- Grand, S.; Lavkulich, L.M. Effects of forest harvest on soil carbon and related variables in Canadian spodosols. Soil Sci. Soc. Am. J. 2012, 76, 1816–1827. [Google Scholar] [CrossRef]
- Gresham, C.A. Sustainability of intensive loblolly pine plantation management in the South Carolina Coastal Plain, USA. For. Ecol. Manag. 2002, 155, 69–80. [Google Scholar] [CrossRef]
- Griffiths, R.P.; Swanson, A.K. Forest soil characteristics in a chronosequence of harvested Douglas-fir forests. Can. J. For. Res. 2001, 31, 1871–1879. [Google Scholar] [CrossRef]
- Gundale, M.J.; DeLuca, T.H.; Fiedler, C.E.; Ramsey, P.W.; Harrington, M.G.; Gannon, J.E. Restoration treatments in a Montana ponderosa pine forest: Effects on soil physical, chemical and biological properties. For. Ecol. Manag. 2005, 213, 25–38. [Google Scholar] [CrossRef]
- Das Gupta, S.; DeLuca, T.H. Short-term changes in belowground C, N stocks in recently clear felled Sitka spruce plantations on podzolic soils of North Wales. For. Ecol. Manag. 2012, 281, 48–58. [Google Scholar] [CrossRef]
- Hart, S.C.; Selmants, P.C.; Boyle, S.I.; Overby, S.T. Carbon and nitrogen cycling in southwestern ponderosa pine forests. For. Sci. 2006, 52, 683–693. [Google Scholar]
- Hendrickson, O.; Chatarpaul, L. Nutrient cycling following whole-tree and conventional harvest in a northern mixed forest. Can. J. For. Res. 2016, 19, 725–735. [Google Scholar] [CrossRef]
- Herman, D.J.; Halverson, L.J.; Firestone, M.K. Nitrogen dynamics in an annual grassland: Oak canopy, climate, and microbial population effects. Ecol. Appl. 2003, 13, 593–604. [Google Scholar] [CrossRef]
- Hölscher, D.; Schade, E.; Leuschner, C. Effects of coppicing in temperate deciduous forests on ecosystem nutrient pools and soil fertility. Basic Appl. Ecol. 2001, 2, 155–164. [Google Scholar] [CrossRef]
- Hwang, J.; Son, Y. Short-term effects of thinning and liming on forest soils of pitch pine and Japanese larch plantations in central Korea. Ecol. Res. 2006, 21, 671–680. [Google Scholar] [CrossRef]
- Jang, W.; Page-Dumroese, D.S.; Keyes, C.R. Long-term soil changes from forest harvesting and residue management in the Northern Rocky Mountains. Soil Sci. Soc. Am. J. 2016. [Google Scholar] [CrossRef]
- Johnson, C.E. Whole-tree clear-cutting effects on soil horizons and organic matter pools. Soil Sci. Soc. Am. J. 1991, 55, 497–502. [Google Scholar] [CrossRef]
- Johnson, D.; Todd, D.E. Harvesting effects on long-term changes in nutrient pools of mixed oak forest. Soil Sci. Soc. Am. J. 1998, 62, 1725–1735. [Google Scholar] [CrossRef]
- Johnson, C.E. Soil nitrogen status 8 years after whole-tree clear-cutting. Can. J. For. Res. 1995, 1346–1355. [Google Scholar] [CrossRef]
- Johnson, C.E.; Romanowicz, R.B.; Siccama, T.G. Conservation of exchangeable cations after clear-cutting of a northern hardwood forest. Can. J. For. Res. 1997, 27, 859–868. [Google Scholar] [CrossRef]
- Johnson, D.W.; Walker, R.F.; Glass, D.W.; Stein, C.M.; Murphy, J.B.; Blank, R.R.; Miller, W.W. Effects of thinning, residue mastication, and prescribed fire on soil and nutrient budgets in a Sierra Nevada mixed-conifer forest. For. Sci. 2014, 60, 170–179. [Google Scholar] [CrossRef]
- Jones, H.S.; Beets, P.N.; Kimberley, M.O.; Garrett, L.G. Harvest residue management and fertilisation effects on soil carbon and nitrogen in a 15-year-old Pinus radiata plantation forest. For. Ecol. Manag. 2011, 262, 339–347. [Google Scholar] [CrossRef]
- Kaye, J.P.; Hart, S.C. Restoration and canopy-type effects on soil respiration in a ponderosa pine-bunchgrass ecosystem. Soil Sci. Soc. Am. J. 1998, 62, 1062–1072. [Google Scholar] [CrossRef]
- Keenan, R.J.; Messier, C.; Kimmins, J.P. (Hamish) Effects of clearcutting and soil mixing on soil properties and understorey biomass in western red cedar and western hemlock forests on northern Vancouver Island, Canada. For. Ecol. Manag. 1994, 68, 251–261. [Google Scholar] [CrossRef]
- Kelliher, F.M.; Ross, D.J.; Law, B.E.; Baldocchi, D.D.; Rodda, N.J. Limitations to carbon mineralization in litter and mineral soil of young and old ponderosa pine forests. For. Ecol. Manag. 2004, 191, 201–213. [Google Scholar] [CrossRef]
- Kishchuk, B.E.; Thiffault, E.; Lorente, M.; Quideau, S.; Keddy, T.; Sidders, D. Decadal soil and stand response to fire, harvest, and salvage-logging disturbances in the western boreal mixedwood forest of Alberta, Canada. Can. J. For. Res. 2015, 45, 141–152. [Google Scholar] [CrossRef]
- Klockow, P.A.; D’Amato, A.W.; Bradford, J.B. Impacts of post-harvest slash and live-tree retention on biomass and nutrient stocks in Populus tremuloides Michx.-dominated forests, northern Minnesota, USA. For. Ecol. Manag. 2013, 291, 278–288. [Google Scholar] [CrossRef]
- Klopatek, J.M. Belowground carbon pools and processes in different age stands of Douglas-fir. Tree Physiol. 2002, 22, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Knoepp, J.D.; Swank, W.T. Forest management effects on surface soil carbon and nitrogen. Soil Sci. Soc. Am. J. 1997, 61, 928–935. [Google Scholar] [CrossRef]
- Korb, J.E.; Johnson, N.C.; Covington, W.W. Slash pile burning effects on soil biotic and chemical properties and plant establishment: Recommendations for amelioration. Restor. Ecol. 2004, 12, 52–62. [Google Scholar] [CrossRef]
- Kraemer, J.F.; Hermann, R.K. Broadcast burning: 25-year effects on forest soils in the western flanks of the Cascade Mountains. For. Sci. 1979, 25, 427–439. [Google Scholar]
- Kurth, V.J.; D’Amato, A.W.; Palik, B.J.; Bradford, J.B. Fifteen-year patterns of soil carbon and nitrogen following biomass harvesting. Soil Sci. Soc. Am. J. 2014, 78, 624–633. [Google Scholar] [CrossRef]
- Laiho, R.; Sanchez, F.; Tiarks, A.; Dougherty, P.M.; Trettin, C.C. Impacts of intensive forestry on early rotation trends in site carbon pools in the southeastern US. For. Ecol. Manag. 2003, 174, 177–189. [Google Scholar] [CrossRef]
- Latty, E.F.; Canham, C.D.; Marks, P.L. The effects of land-use history on soil properties and nutrient dynamics in northern hardwood forests of the Adirondack Mountains. Ecosystems 2004, 7, 193–207. [Google Scholar] [CrossRef]
- Law, B.E.; Thornton, P.E.; Irvine, J.; Anthoni, P.M.; Van Tuyl, S. Carbon storage and fluxes in Ponderosa pine forests at different developmental stages. Glob. Chang. Biol. 2001, 7, 755–777. [Google Scholar] [CrossRef]
- Law, B.E.; Sun, O.J.; Campbell, J.; Van Tuyl, S.; Thornton, P.E. Changes in carbon storage and fluxes in a chronoseuence of Ponderosa pine. Glob. Chang. Biol. 2003, 4, 510–524. [Google Scholar] [CrossRef]
- LeDuc, S.D.; Rothstein, D.E. Initial recovery of soil carbon and nitrogen pools and dynamics following disturbance in jack pine forests: A comparison of wildfire and clearcut harvesting. Soil Biol. Biochem. 2007, 39, 2865–2876. [Google Scholar] [CrossRef]
- Maassen, S.; Wirth, S. Soil microbiological monitoring of a pine forest after partial thinning for stand regeneration with beech seedlings. Soil Sci. Plant Nutr. 2004, 50, 815–819. [Google Scholar] [CrossRef]
- Mattson, K.G.; Smith, H.C. Detrital organic matter and soil CO2 efflux in forests regenerating from cutting in West Virginia. Soil Biol. Biochem. 1993, 25, 1241–1248. [Google Scholar] [CrossRef]
- Mattson, K.G.; Swank, W.T. Soil and detrital carbon dynamics following forest cutting in the Southern Appalachians. Biol. Fertil. Soils 1989, 7, 247–253. [Google Scholar] [CrossRef]
- May, B.M.; Attiwill, P.M. Nitrogen-fixation by Acacia dealbata and changes in soil properties 5 years after mechanical disturbance or slash-burning following timber harvest. For. Ecol. Manag. 2003, 181, 339–355. [Google Scholar] [CrossRef]
- McLaughlin, J.W.; Phillips, S.A. Soil carbon, nitrogen, and base cation cycling 17 years after whole-tree harvesting in a low-elevation red spruce (Picea rubens)-balsam fir (Abies balsamea) forested watershed in central Maine, USA. For. Ecol. Manag. 2006, 222, 234–253. [Google Scholar] [CrossRef]
- McKee, S.E.; Seiler, J.R.; Aust, W.M.; Strahm, B.D.; Schilling, E.B.; Brooks, S. Carbon pools and fluxes in a tupelo (Nyssa aquatica)-baldcypress (Taxodium distichum) swamp24-years after harvest disturbances. Biomass Bioenergy 2013, 55, 130–140. [Google Scholar] [CrossRef]
- McLaughlin, J.W.; Liu, G.; Jurgensen, M.F.; Gale, M.R. Organic carbon characteristics in a spruce swamp five years after harvesting. Soil Sci. Soc. Am. J. 1996, 60, 1228–1236. [Google Scholar] [CrossRef]
- Merino, A.; Edeso, J.M. Soil fertility rehabilitation in young Pinus radiata D. Don. plantations from northern Spain after intensive site preparation. For. Ecol. Manag. 1999, 116, 83–91. [Google Scholar] [CrossRef]
- Moreno-Fernandez, D.; Diaz-Pines, E.; Barbeito, I.; Sanchez-Gonzalez, M.; Montes, F.; Rubio, A.; Canellas, I. Temporal carbon dynamics over the rotation period of two alternative management systems in Mediterranean mountain Scots pine forests. For. Ecol. Manag. 2015, 348, 186–195. [Google Scholar] [CrossRef]
- Mu, C.C.; Lu, H.C.; Bao, X.; Wang, B.; Cui, W. Effects of selective cutting on vegetation carbon storage of boreal Larix gmelinii-Carex schmidtii forested wetlands in Daxing’anling, China. Shengtai Xuebao/Acta Ecol. Sin. 2013, 33, 5286–5298. [Google Scholar]
- Murphy, J.D.; Johnson, D.W.; Miller, W.W.; Walker, R.F.; Blank, R.R. Prescribed fire effects on forest floor and soil nutrients in a Sierra Nevada Forest. Soil Sci. 2006, 171, 181–199. [Google Scholar] [CrossRef]
- Neher, D.A.; Barbercheck, M.E.; El-Allaf, S.M.; Anas, O. Effects of disturbance and ecosystem on decomposition. Appl. Soil Ecol. 2003, 23, 165–179. [Google Scholar] [CrossRef]
- Norris, C.E.; Quideau, S.A.; Bhatti, J.S.; Wasylishen, R.E.; MacKenzie, M.D. Influence of fire and harvest on soil organic carbon in jack pine sites. Can. J. For. Res. 2009, 39, 642–654. [Google Scholar] [CrossRef]
- O’Brien, N.D.; Attiwill, P.M.; Weston, C.J. Stability of soil organic matter in Eucalyptus regnans forests and Pinus radiata plantations in south eastern Australia. For. Ecol. Manag. 2003, 185, 249–261. [Google Scholar] [CrossRef]
- Powers, M.; Kolka, R.; Palik, B.; McDonald, R.; Jurgensen, M. Long-term management impacts on carbon storage in Lake States forests. For. Ecol. Manag. 2011, 262, 424–431. [Google Scholar] [CrossRef]
- Prest, D.; Kellman, L.; Lavigne, M.B. Mineral soil carbon and nitrogen still low three decades following clearcut harvesting in a typical Acadian Forest stand. Geoderma 2014, 214–215, 62–69. [Google Scholar] [CrossRef]
- Prietzel, J.; Wagoner, G.L.; Harrison, R.B. Long-term effects of repeated urea fertilization in Douglas-fir stands on forest floor nitrogen pools and nitrogen mineralization. For. Ecol. Manag. 2004, 193, 413–426. [Google Scholar] [CrossRef]
- Puhlick, J.J.; Fernandez, I.J.; Weiskittel, A.R. Evaluation of forest management effects on the mineral soil carbon pool of a lowland, mixed-species forest in Maine, USA. Can. J. Soil Sci. 2016, 96, 207–218. [Google Scholar] [CrossRef]
- Rab, M.A. Soil physical and hydrological properties following logging and slash burning in the Eucalyptus regnans forest of southeastern Australia. For. Ecol. Manag. 1996, 84, 159–176. [Google Scholar] [CrossRef]
- Riley, J.M.; Jones, R.H. Factors limiting regeneration of Quercus alba and Cornus florida in formerly cultivated coastal plain sites, South Carolina. For. Ecol. Manag. 2003, 177, 571–586. [Google Scholar] [CrossRef]
- Roaldson, L.M.; Johnson, D.W.; Miller, W.W.; Murphy, J.D.; Walker, R.F.; Stein, C.M.; Glass, D.W.; Blank, R.R. Prescribed Fire and Timber Harvesting Effects on Soil Carbon and Nitrogen in a Pine Forest. Soil Sci. Soc. Am. J. 2014, 78, S48. [Google Scholar] [CrossRef]
- Rothstein, D.E.; Spaulding, S.E. Replacement of wildfire by whole-tree harvesting in jack pine forests: Effects on soil fertility and tree nutrition. For. Ecol. Manag. 2010, 260, 1164–1174. [Google Scholar] [CrossRef]
- Sanchez, F.G.; Scott, D.A.; Ludovici, K.H. Negligible effects of severe organic matter removal and soil compaction on loblolly pine growth over 10 years. For. Ecol. Manag. 2006, 227, 145–154. [Google Scholar] [CrossRef]
- Sanscrainte, C.L.; Peterson, D.L.; McKay, S. Carbon storage and soil properties in late-successional and second-growth subalpine forests in the North Cascade Range, Washington. Northwest Sci. 2003, 77, 297–307. [Google Scholar]
- Saynes, V.; Etchevers, J.D.; Galicia, L.; Hidalgo, C.; Campo, J. Soil carbon dynamics in high-elevation temperate forests of Oaxaca (Mexico): Thinning and rainfall effects. Bosque (Valdivia) 2012, 33, 3–11. [Google Scholar] [CrossRef]
- Selig, M.F.; Seiler, J.R.; Tyree, M.C. Soil carbon and CO2 efflux as influenced by the thinning of loblolly pine (Pinus taeda L.) plantations on the Piedmont of Virginia. For. Sci. 2008, 54, 58–66. [Google Scholar]
- Shelburne, V.B.; Boyle, M.F.; Lione, D.J.; Waldrop, T.A. Preliminary effects of prescribed burning and thinning as fuel reduction treatments on the piedmont soils of the clemson experimental forest. In General Technical Report SRS-71; U.S. Department of Agriculture, Forest Service, Southern Research Station: Asheville, NC, USA, 2004; pp. 35–38. [Google Scholar]
- Sheng, H.; Zhou, P.; Zhang, Y.; Kuzyakov, Y.; Zhou, Q.; Ge, T.; Wang, C. Loss of labile organic carbon from subsoil due to land-use changes insubtropical China. Soil Biol. Biochem. 2015, 88, 148–157. [Google Scholar] [CrossRef]
- Skovsgaard, J.P.; Stupak, I.; Vesterdal, L. Distribution of biomass and carbon in even-aged stands of Norway spruce (Picea abies (L.) Karst.): A case study on spacing and thinning effects in northern Denmark. Scand. J. For. Res. 2006, 21, 470–488. [Google Scholar] [CrossRef]
- Slesak, R.A.; Schoenholtz, S.H.; Harrington, T.B. Soil carbon and nutrient pools in Douglas-fir plantations 5 years after manipulating biomass and competing vegetation in the Pacific Northwest. For. Ecol. Manag. 2011, 262, 1722–1728. [Google Scholar] [CrossRef]
- Small, C.J.; McCarthy, B.C. Relationship of understory diversity to soil nitrogen, topographic variation, and stand age in an eastern oak forest, USA. For. Ecol. Manag. 2005, 217, 229–243. [Google Scholar] [CrossRef]
- Stone, J.E.; Kolb, T.E.; Covington, W.W. Effects of restoration thinning on presettlement Pinus ponderosa in northern Arizona. Restor. Ecol. 1999, 7, 172–182. [Google Scholar] [CrossRef]
- Stone, D.; Elioff, J. Soil properties and aspen development five years after compaction and forest floor removal. Can. J. Soil Sci. 1998, 78, 51–58. [Google Scholar] [CrossRef]
- Strong, T.F. Harvesting intensity influences the carbon distribution in a northern hardwood ecosystem. In Research Paper NC-329; Department of Agriculture, Forest Service, North Central Research Station: St. Paul, MN, USA, 1997. [Google Scholar]
- Strukelj, M.; Brais, S.; Paré, D. Nine-year changes in carbon dynamics following different intensities of harvesting in boreal aspen stands. Eur. J. For. Res. 2015, 134, 737–754. [Google Scholar] [CrossRef]
- Tang, J.; Bolstad, P.V.; Martin, J.G. Soil carbon fluxes and stocks in a Great Lakes forest chronosequence. Glob. Chang. Biol. 2009, 15, 145–155. [Google Scholar] [CrossRef]
- Trettin, C.C.; Jurgensen, M.F.; Gale, M.R.; McLaughlin, J.W. Recovery of carbon and nutrient pools in a northern forested wetland 11 years after harvesting and site preparation. For. Ecol. Manag. 2011, 262, 1826–1833. [Google Scholar] [CrossRef]
- Ussiri, D.A.N.; Johnson, C.E. Organic matter composition and dynamics in a northern hardwood forest ecosystem 15 years after clear-cutting. For. Ecol. Manag. 2007, 240, 131–142. [Google Scholar] [CrossRef]
- Vario, C.L.; Neurath, R.A.; Friedland, A.J. Response of mineral soil carbon to clear-cutting in a northern hardwood forest. Soil Sci. Soc. Am. J. 2014, 78, 309–318. [Google Scholar] [CrossRef]
- Vesterdal, L.; Dalsgaard, M.; Felby, C.; Raulund-Rasmussen, K.; Jørgensen, B.B. Effects of thinning and soil properties on accumulation of carbon, nitrogen and phosphorus in the forest floor of Norway spruce stands. For. Ecol. Manag. 1995, 77, 1–10. [Google Scholar] [CrossRef]
- Waldrop, M.P.; McColl, J.G.; Powers, R.F. Effects of forest postharvest management practices on enzyme activities in decomposing litter. Soil Sci. Soc. Am. J. 2003, 67, 1250–1256. [Google Scholar] [CrossRef]
- Wu, J.-S.; Jiang, P.-K.; Chang, S.X.; Xu, Q.-F.; Lin, Y. Dissolved soil organic carbon and nitrogen were affected by conversion of native forests to plantations in subtropical China. Can. J. Soil Sci. 2010, 90, 27–36. [Google Scholar] [CrossRef]
- Xiang, W.; Chai, H.; Tian, D.; Peng, C. Marginal effects of silvicultural treatments on soil nutrients following harvest in a Chinese fir plantation. Soil Sci. Plant Nutr. 2009, 55, 523–531. [Google Scholar] [CrossRef]
- Yanai, R.D.; Arthur, M.A.; Siccama, T.G.; Federer, C.A. Challenges of measuring forest floor organic matter dynamics: Repeated measures from a chronosequence. For. Ecol. Manag. 2000, 138, 273–283. [Google Scholar] [CrossRef]
- Zabowski, D.; Chambreau, D.; Rotramel, N.; Thies, W.G. Long-term effects of stump removal to control root rot on forest soil bulk density, soil carbon and nitrogen content. For. Ecol. Manag. 2008, 255, 720–727. [Google Scholar] [CrossRef]
- Zhong, Z.; Makeschin, F. Soil biochemical and chemical changes in relation to mature spruce (Picea abies) forest conversion and regeneration. J. Plant Nutr. Soil Sci. 2003, 166, 291–299. [Google Scholar] [CrossRef]
- Zummo, L.M.; Friedland, A.J. Soil carbon release along a gradient of physical disturbance in a harvested northern hardwood forest. For. Ecol. Manag. 2011, 261, 1016–1026. [Google Scholar] [CrossRef]

| Factor | Levels | |
|---|---|---|
| Reporting units | Pool (Mg·ha−1), concentration (% or mg·g−1) | |
| Soil Depth | O horizon | Forest Floor |
| Top Soil | 0–15 cm | |
| Mid Soil | 15–30 cm | |
| Deep Soil | 30–60 cm | |
| Very Deep | 60–100+ cm | |
| Overstory species | Hardwood, conifer/mixed | |
| Soil order | Alfisol, Andisol, Entisol, Inceptisol, Mollisol, Spodosol, Oxisol, Ultisol | |
| Geographic group | NE North America, NW North America, SE North America, SW North America, Europe, Asia, Pacific (Australia, New Zealand) | |
| Harvest type | Clearcut, thin | |
| Harvest intensity | Stem only, whole tree | |
| Residue management | Removed, spread | |
| Site preparation | Broadcast burn, tillage/scarification | |
| Soil texture | Fine (mostly silt or clay), coarse (mostly sand), organic | |
| Time since harvest | Continuous | |
| Mean Annual Temperature | 0–5, 5–7.5, 7.6–10, 10.1–15, 15.1–20, >20 (°C) | |
| Mean Annual Precipitation | <500, 500−750, 751−1000, 1001−1400, 1401−1800, >1800 (mm) | |
| Coefficient | Estimate | SE | t-Value | p-Value |
|---|---|---|---|---|
| Intercept (Alfisol, mineral soil) | 12.702 | 3.587 | 3.541 | 0.0004 |
| O horizon | −21.475 | 3.766 | −5.703 | <0.0001 |
| Inceptisol | −10.876 | 5.717 | −1.902 | 0.0577 |
| Spodosol | −14.717 | 4.320 | −3.407 | 0.0007 |
| Ultisol | −24.776 | 5.391 | −4.596 | <0.0001 |
| Time | −67.834 | 41.56 | −1.632 | 0.10325 |
| Time2 | 120.412 | 40.361 | 2.983 | 0.0030 |
| Residual SE: 40.24 on 533 DF | ||||
| F-Statistic: 10.74 on 6 and 533 df, p < 0.0001 | R2 = 0.108 | Adj. R2 = 0.098 | ||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
James, J.; Harrison, R. The Effect of Harvest on Forest Soil Carbon: A Meta-Analysis. Forests 2016, 7, 308. https://doi.org/10.3390/f7120308
James J, Harrison R. The Effect of Harvest on Forest Soil Carbon: A Meta-Analysis. Forests. 2016; 7(12):308. https://doi.org/10.3390/f7120308
Chicago/Turabian StyleJames, Jason, and Rob Harrison. 2016. "The Effect of Harvest on Forest Soil Carbon: A Meta-Analysis" Forests 7, no. 12: 308. https://doi.org/10.3390/f7120308
APA StyleJames, J., & Harrison, R. (2016). The Effect of Harvest on Forest Soil Carbon: A Meta-Analysis. Forests, 7(12), 308. https://doi.org/10.3390/f7120308






