1. Introduction
In order to mitigate fossil carbon dioxide emissions for energy generation, renewable energies such as bioenergy are being promoted as an alternative to fossil fuels. In Germany in 2013, renewable sources accounted for 12.3% of total energy consumption, of which biomass accounted for 7.6% [
1]. Within the biomass sector, forest residues have been identified as a large underused source for potentially increasing the raw material supply. Already, about 11 million·m
3 of wood has been used directly for energy purposes, which accounts for about one fifth of the total annual harvest from forests. The potential for increasing forest biomass for energy (at a sustainable rate) is estimated to be between 12 and 19 million·m
3·year
−1, which could be achieved by increasing the utilization of forest residues and currently underused hardwood stands [
2].
The process of extracting harvesting residues from forests, or whole tree harvesting, has a number of technological and ecological constraints. On the technological side, there is a high expenditure for the logistics due to the scattered location of the biomass and its low density, and thus transportability [
3,
4]. Advanced technologies and management practices could help to improve both feedstock quality and cost efficiency in the future [
3,
5]. The biomass market situation, and thus the actual price paid by biomass combustion facilities per unit of feedstock, will eventually determine economic feasibility of the utilization of forest residues for bioenergy.
On the ecological side, there is a threat of potentially high nutrient exports when extracting the nutrient-rich crown material, and thus a loss of productivity [
6,
7,
8,
9,
10]. Also, if large parts of the harvesting residues are exported from the site, there will be fewer habitats for decomposer fauna and flora and less input material to refill the site-specific humus stock [
11,
12]. The nutrient issue could be attenuated by returning nutrients into the forests from sources such as wood ash from biomass combustion facilities [
13,
14,
15,
16]. Implementing wood ash recycling systems into practice would require thorough knowledge regarding the actual amounts of nutrients removed in management scenarios of various intensity [
14]. Additionally needed is an estimation of the available soil nutrient pool, which plays a major role in assessing the impact of harvest-induced nutrient losses on site productivity, and thus on management sustainability in terms of maintaining nutrient reserves. Finally, site specific nutrient balances are needed to evaluate the impact of different harvesting intensities and to draw conclusions for adapted forest management.
Estimating nutrient fluxes associated with the extraction of aboveground biomass compartments under intensive management scenarios requires knowledge and estimation methods about the distribution of biomass and nutrients of all tree compartments. Yet yield tables and forest growth models typically used in forest management and planning in Germany concentrate on just the volume of the marketable round wood, which is typically the tree trunk up to a certain diameter (e.g., 7 cm over bark), depending on the current market situation. Information about the distribution of the biomass to above-ground tree compartments, such as the stem with a diameter smaller than 7 cm, branches, foliage, and dead branches is needed in order to estimate the profitability and the impact on the nutrient budget of going from classic stem-only (SO) harvesting to more intensive scenarios, such as whole tree without foliage (WT excl. needles) or whole tree (WT) harvesting [
17,
18]. Some studies on the impact of nutrient removals in intensive scenarios focus on the final felling and harvesting at the end of a stand’s rotation time and its impact on the growth of the next stand generation [
7,
10]. The rationale for this approach is that the biomass and nutrient fluxes in final fellings, such as clear cuts or shelterwood cuttings, are especially large. Alternately, other studies have concentrated on the impact of intensive biomass exports in thinning operations of medium aged stands [
10,
19,
20,
21,
22], which happen at a stage of stand development when the remaining trees exhibit high productivity, and thus require large amounts of nutrients for the buildup of aboveground biomass.
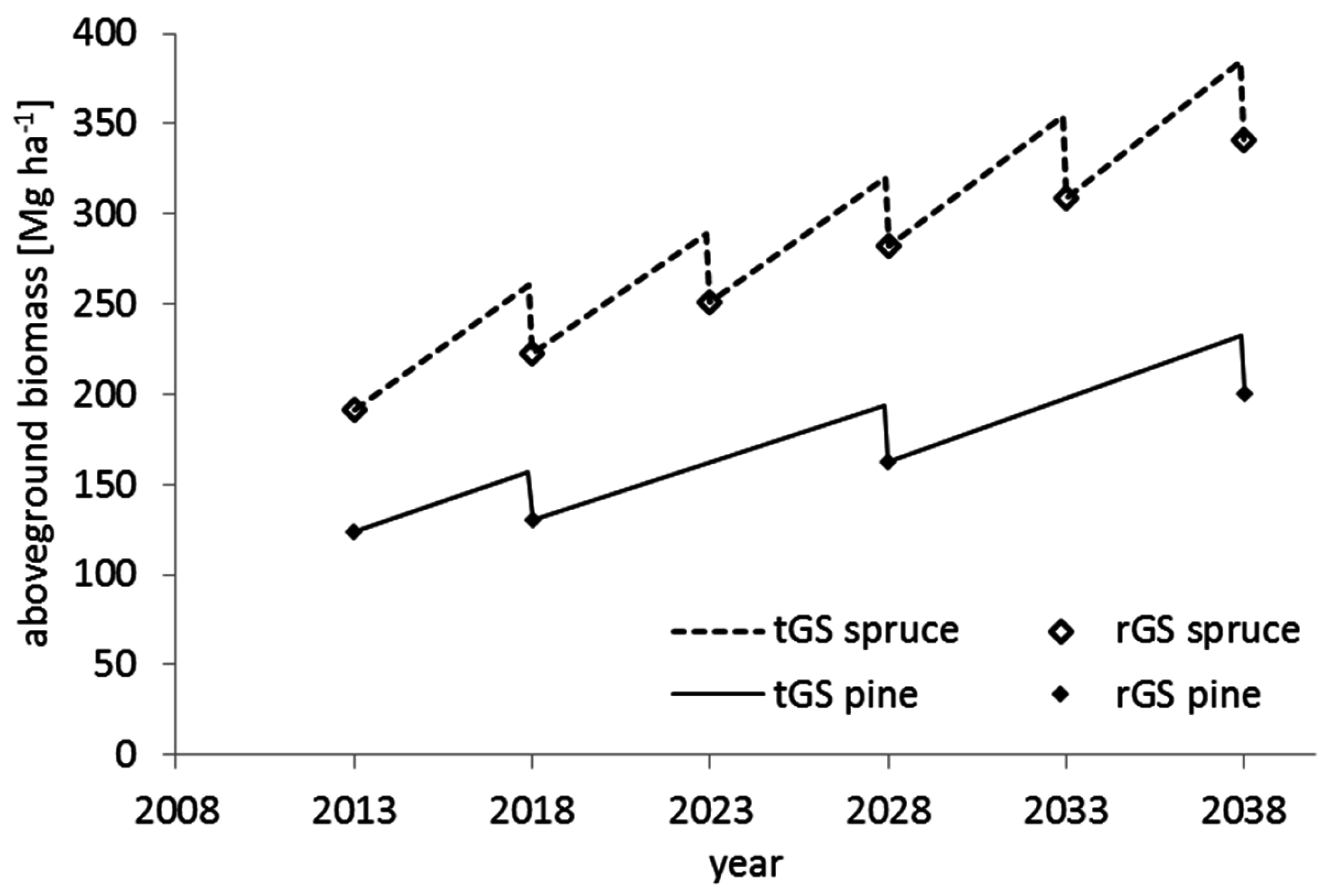
In our study, we aimed at quantifying the nutrient exports of thinning operations with variable intensities of biomass removal in two medium-aged coniferous stands representing typical site and stand conditions in the region of Saxony in northeast (NE) Germany and evaluating the impact of such treatments on the nutrient budget of the stands. We hypothesize that intensive harvesting scenarios (1) impose a strong negative effect on the nutrient balance of forest stands, even in the thinning stage; and (2) will lead to a more negative nutrient balance in the highly productive stands than in less productive stands, due to larger absolute amounts of biomass and thus nutrients removed. Therefore, we developed single-tree biomass equations to predict the dry mass of all aboveground tree compartments based on the tree’s diameter at breast height (DBH) and analyzed nutrient contents of these compartments. Based on this knowledge, we estimated the biomass and nutrient extraction by thinning with three different management intensities: conventional SO, WT (excl. needles), and WT harvesting, which refers to aboveground biomass only. In all scenarios, stumps and roots are left in the stand. Also, we calculated the nutrients required to build up aboveground biomass within the examined time span. Finally, we set the amount of extracted and stored nutrients in relation to the site-specific nutrient stocks and fluxes in order to estimate the sustainability of intensive management scenarios.
4. Discussion
In this study we simulated relatively heavy thinnings in order to sustain stand stability to adapt the stands to climate change in the long term, and make use of the high forest productivity [
28,
29], which is accelerated by high atmospheric N inputs of about 19.3 and 36.0 kg·ha·year
−1 at the pine and spruce site, respectively. The large difference in productivity between pine and spruce is both a species and a site effect, because the less demanding pine stands are usually grown on the least productive forest sites [
19].
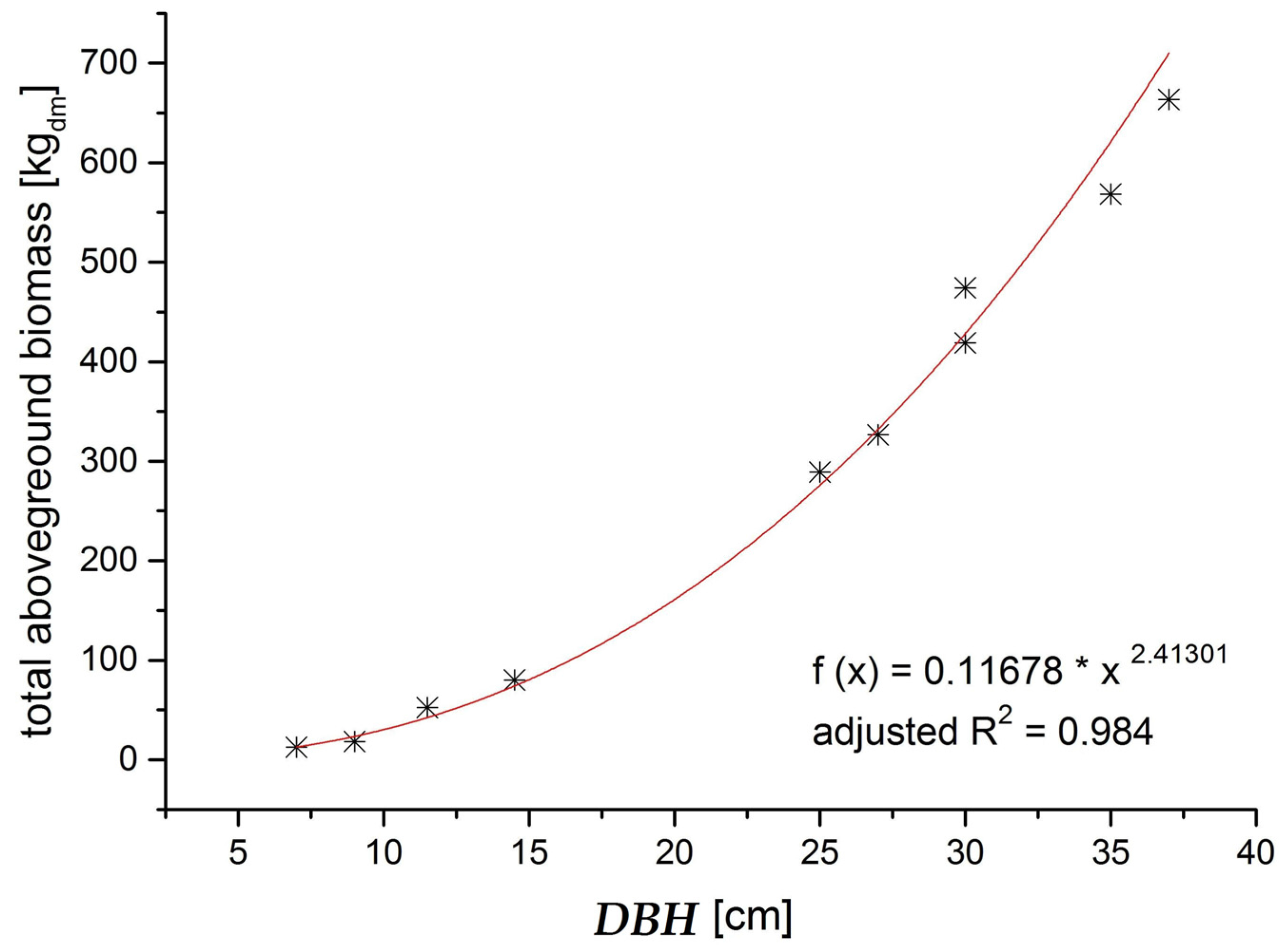
The biomass functions we established in order to model the mass of each aboveground compartment were easy to apply and fit our data very well. Only for the tree top wood of spruce and the dead branches, the R² was slightly lower than for the other compartments, probably because the tree top wood is a somewhat artificially defined compartment and the retention of dead branches on a tree depends on factors other than the tree’s DBH, such as the actual stand density in the area surrounding the respective tree. Nevertheless, since these two compartments play a minor role for addressing the questions raised in this paper, and the R
2 was still sufficient, we retained the equations for use. The drawback of the biomass functions we established is that they are stand specific, and can therefore only be applied for the DBH range for which they were calibrated. As a result, we concentrated our study on just the thinning phase. In order to create more widely applicable biomass functions, more trees with a wider range of DBH, and from stands representing a wider range of productivity and stand density, would need to be measured. The biomass equations would then need to account for this greater variability by incorporating further explanatory variables such as height and stand density, in addition to the DBH [
30].
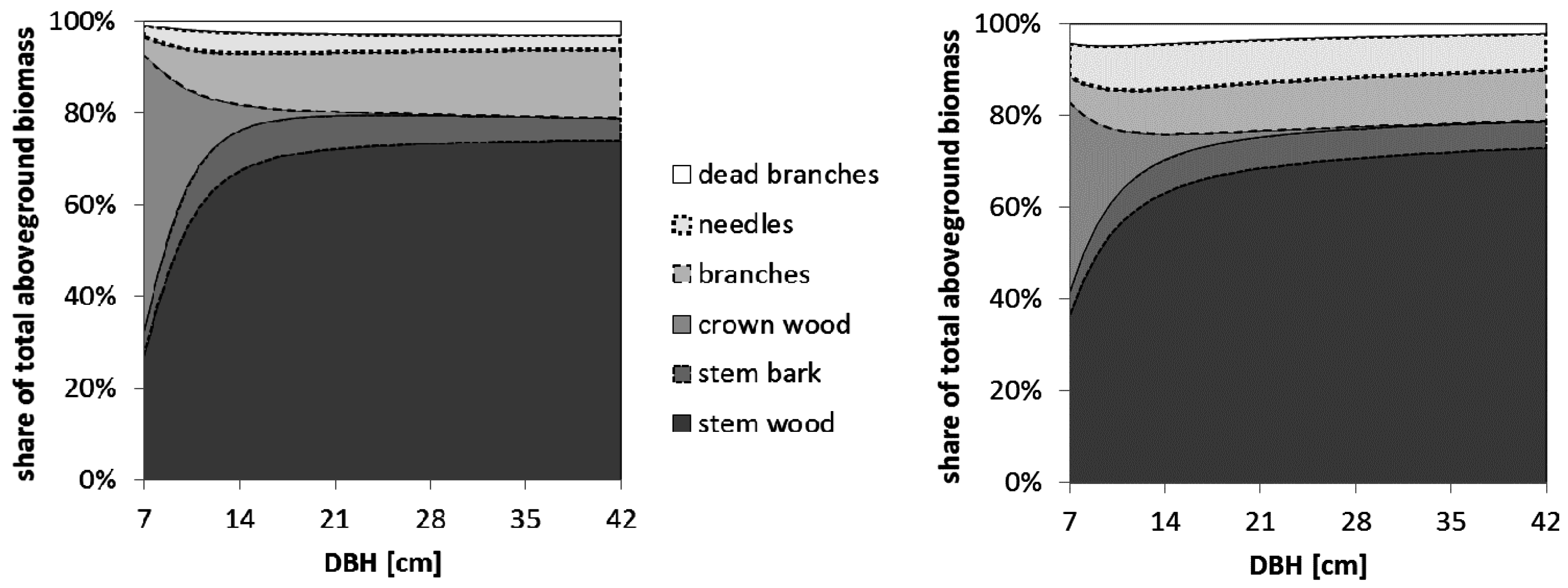
One option proposed to increase the harvested biomass, and keep nutrient exports relatively low at the same time, is to leave tree tops in the forest for one year to allow the needles to fall off before collecting the tree tops (WT excl. needles scenario) [
7,
20]. The different pattern of crown biomass allocation between pine and spruce, with a greater share of branches in pine trees versus needles in spruce trees, shows that this method would lead to a greater biomass loss in spruce than in pine trees, when compared to the WT scenario. With respect to biomass feedstock quantities, this procedure would be more viable in pine than in spruce stands. If biomass feedstock quality is also considered, it would be preferable in any case to exclude the needles, as wood is a higher quality feedstock in terms of particle size distribution when chipping at the forest industries [
3], as well as the behavior in the burning process.
The nutrient contents in the aboveground biomass compartments match with the findings of previous studies, as far as the compartments are comparable. Jacobsen et al. [
31] performed nutrient analyses that are partly comparable, and found similar nutrient contents in the stem wood, bark, needles, and branches of pine and spruce trees. The nutrient contents reported by Weis and Göttlein [
9] in the needles, bark, and wood of spruce trees are also within the range of our findings.
Due to the rough estimation method of weathering and the utilization of deposition and seepage values from similar and close-by sites (but not exactly the study sites), the nutrient balance should be interpreted with some caution. Nevertheless, it is still useful for evaluating the impact of biomass, and thus nutrient extraction, in the different thinning scenarios.
Our findings confirm our first hypothesis, that intensive harvesting scenarios impose a strong negative effect on the nutrient balance of forest stands even in the thinning stage. We demonstrated that intensive WT and WT (exc. needles) harvesting causes fairly high nutrient exports in the thinning phase of the trial sites. The biomass gain is far lower than the nutrient expenses, as the biomass gain is only 1.18 to 1.30 times compared to SO, but the ratio of nutrient losses is 1.4 to 3.6 compared to SO. The increase of nutrient losses from SO to WT harvesting are greater for N and P compared to the base cations, reflecting the over proportional abundance of N and P in the crown material, especially in the physiologically active needles. Yet the impact on the site-specific nutrient sustainability is far greater for the base cations than for N, because of excess atmospheric N inputs. We unfortunately have no data for the nutrient balance of P, but as P inputs into the forest ecosystem by weathering and deposition are usually low, a rather negative P balance similar to that of the base cations can be assumed. High seepage losses contribute strongly to the negative nutrient balance of the base cations. The seepage losses of base cations result from sulfur depositions during the 1970s to 1990s, which were greatly reduced by the end of the last century [
32]. However, SO
42− from accumulated S pools still gets washed out of the soil profile, coupled with base cations to neutralize the charge [
33]. This leads to negative nutrient balances for Ca
2+ and Mg
2+ already at the SO scenario, which decreases further in the WT (excl. needles) and the WT scenario, when K
+ also becomes negative.
According to our findings, we reject our second hypothesis, according to which the negative impact of intensive harvesting is greater in highly productive stands than in less productive stands, due to larger absolute amounts of removed biomass, and thus nutrients. In our study, the higher stand productivity of the spruce stand corresponded with a higher nutrient demand for uptake and storage, as well as higher nutrient losses due to biomass extraction, compared to the less productive pine stand. At the same time, these differences in productivity, and thus biomass-related nutrient fluxes, are balanced out by higher nutrient stocks and inputs through deposition (except for Ca), while seepage losses are equal to or even lower than the pine site. Thus, the relative impact of nutrient exports through intensive biomass extraction is almost equally severe in the spruce and pine stand, despite different absolute levels of biomass extraction.
Our findings should, however, be interpreted with some caution, as they are based on static assumptions regarding the development of forest growth, the nutrient concentrations in the biomass, and the weathering rate. In reality, increased nutrient scarcity due to intensive biomass extraction may cause reduced growth of the remaining stand, as has been shown in studies conducted in North American and Scandinavian forests [
7,
8,
10], while other studies found no clear effect on tree growth [
34,
35]. A meta-analysis by Achat et al. [
36] indicates an average growth reduction of 3%–7% after intensive biomass extraction. Declining pools of available nutrients may also cause reduced nutrient concentrations in the foliage of the remaining trees, which would also alter the nutrient balance. The nutrient balance is also known to be strongly influenced by the rate of mineral weathering [
37], which is in practice very difficult to determine and could only be roughly estimated in our study. Furthermore, mineral weathering rates are not static, but mycorrhizal weathering may be accelerated if there are nutrient deficiencies [
38]. Despite these uncertainties, our findings underline the possible nutrient balance risk of intensive biomass extraction from thinnings, which brings into question the nutrient balance sustainability of these intensive management practices.
Our study highlights that intensive harvesting scenarios like WT (excl. needles) and WT harvesting can have severe consequences for the nutrient balance of forest sites. Furthermore, intensive biomass removal is known to negatively affect the soil as the basis for biomass production. Reduced residue inputs reduce the organic carbon content [
39] and mineralization rates and cause soil compaction and thus a reduced water holding capacity [
36]. Based on the analysis of detrimental forest management practices (i.e., litter raking, fuelwood collection), the influence of biomass extraction on soil acidification was highlighted, because the uptake and storage of cations exceeds that of anions during tree growth and biomass buildup [
40,
41]. In order to mitigate the negative effects of intensive biomass extraction on the nutrient budget, the redistribution of wood ash is frequently discussed and has proven to be suitable to compensate for nutrient losses in forests, with the exception of N [
13,
42,
43,
44,
45], and was previously found to be applicable in the form of bark-ash-pellets at the study sites [
14].