Adaptation of Leaf Water Relations to Climatic and Habitat Water Availability
Abstract
:1. Introduction
2. Experimental Section
2.1. Leaf Water Relations Database
2.2. Species Climatic Envelope Analysis
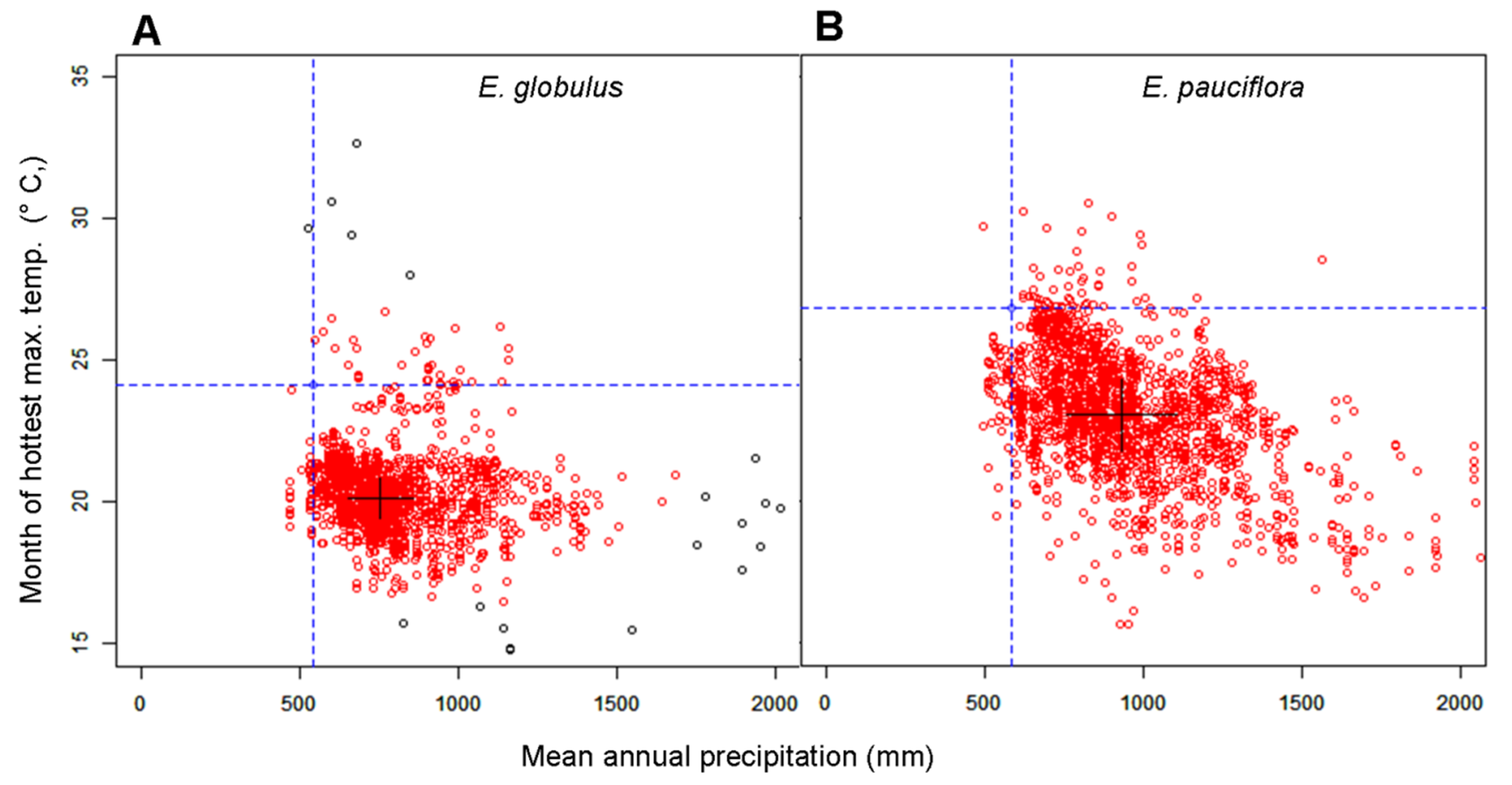
2.3. Statistical Analysis
3. Results
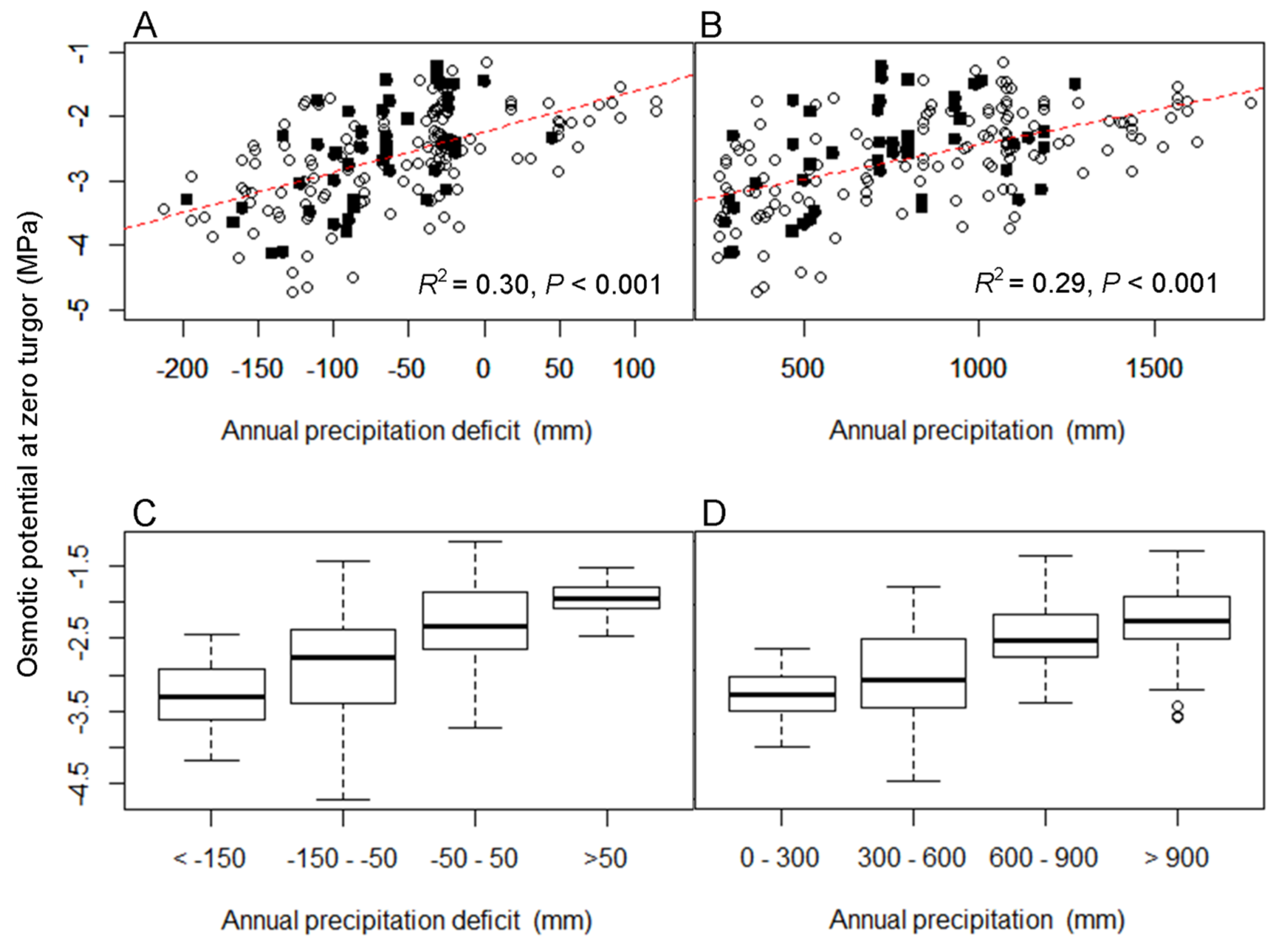
3.1. The Relationship between Turgor Loss and Climate
| Percentile of Distribution | Variable | Parameter Estimate | SE | p Value | Adjusted R2 |
|---|---|---|---|---|---|
| 0.50 | Aridity index | 0.33 | 0.05 | <0.001 | 0.16 |
| Mean annual precipitation | 0.001 | 0.0001 | <0.001 | 0.29 | |
| Precipitation deficit | 0.006 | 0.0007 | <0.001 | 0.30 | |
| Mean annual temperature | −0.05 | 0.01 | <0.001 | 0.06 | |
| Max. temperature of hottest month | −0.07 | 0.01 | <0.001 | 0.21 | |
| 0.02 | Aridity index | 0.78 | 0.11 | <0.001 | 0.21 |
| Mean annual precipitation | 0.001 | 0.0001 | <0.001 | 0.25 | |
| Precipitation deficit | 0.005 | 0.0006 | <0.001 | 0.27 | |
| 0.98 | Mean annual temperature | −0.04 | 0.01 | <0.001 | 0.05 |
| Max. temperature of hottest month | −0.06 | 0.01 | <0.001 | 0.20 |
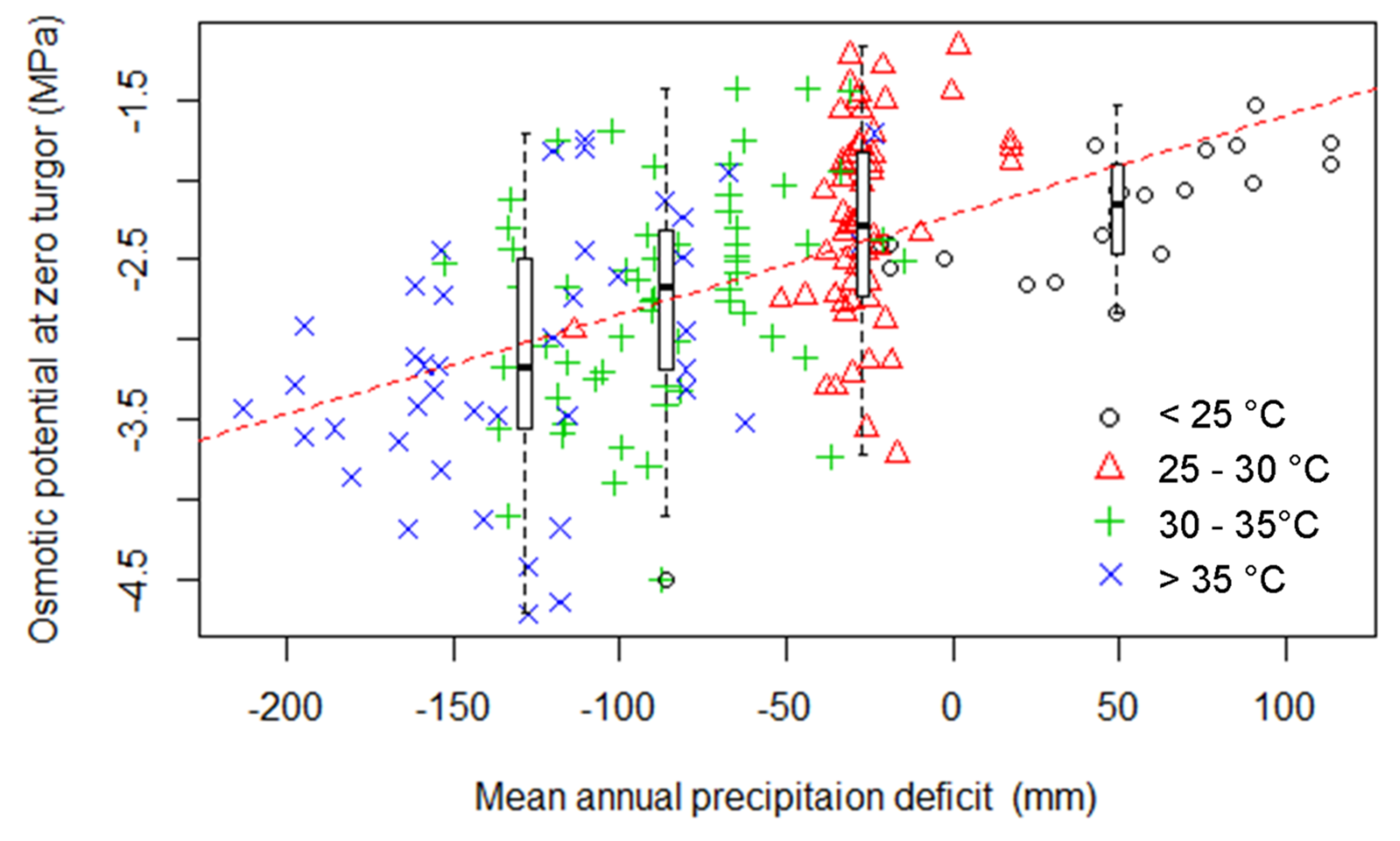
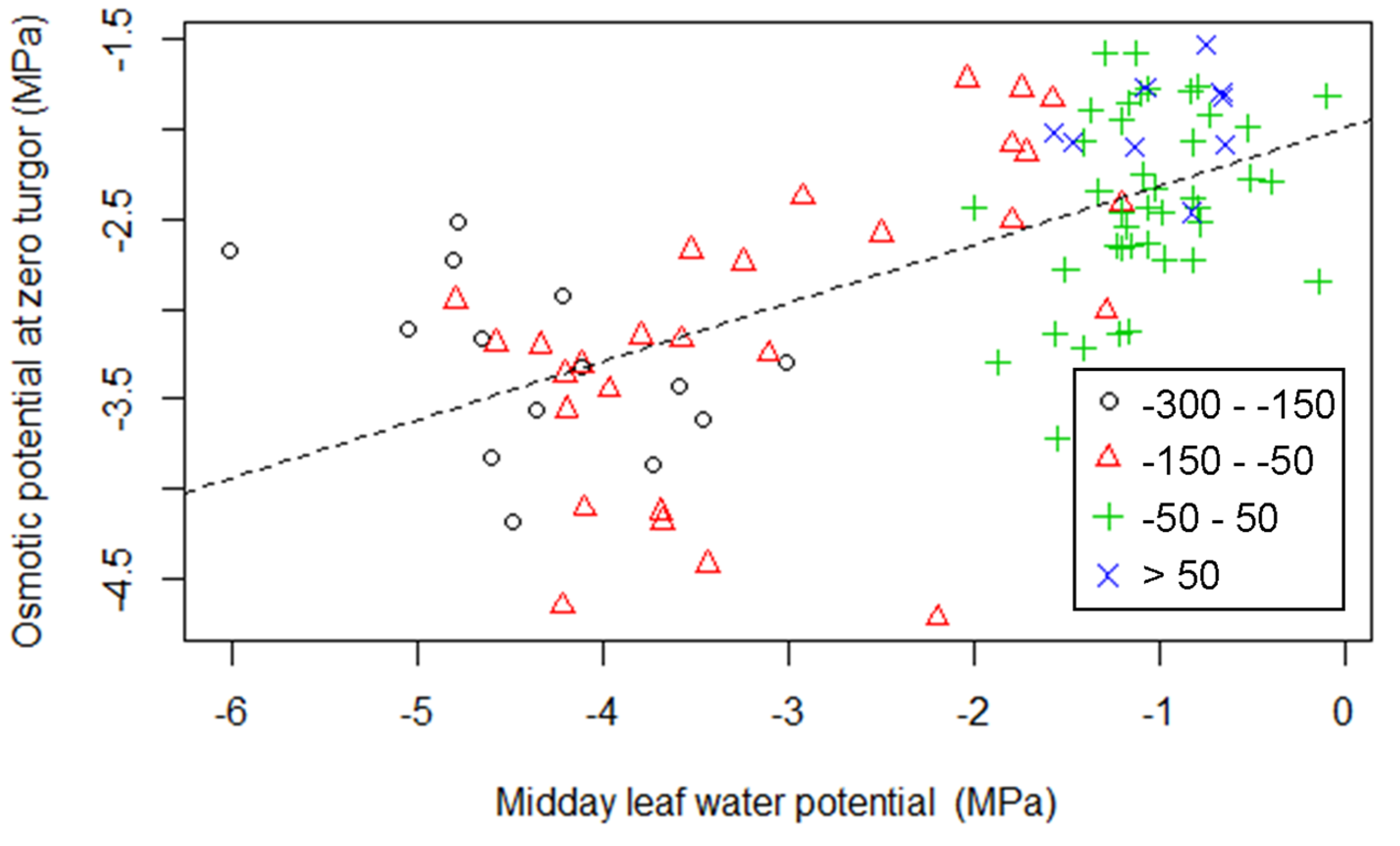
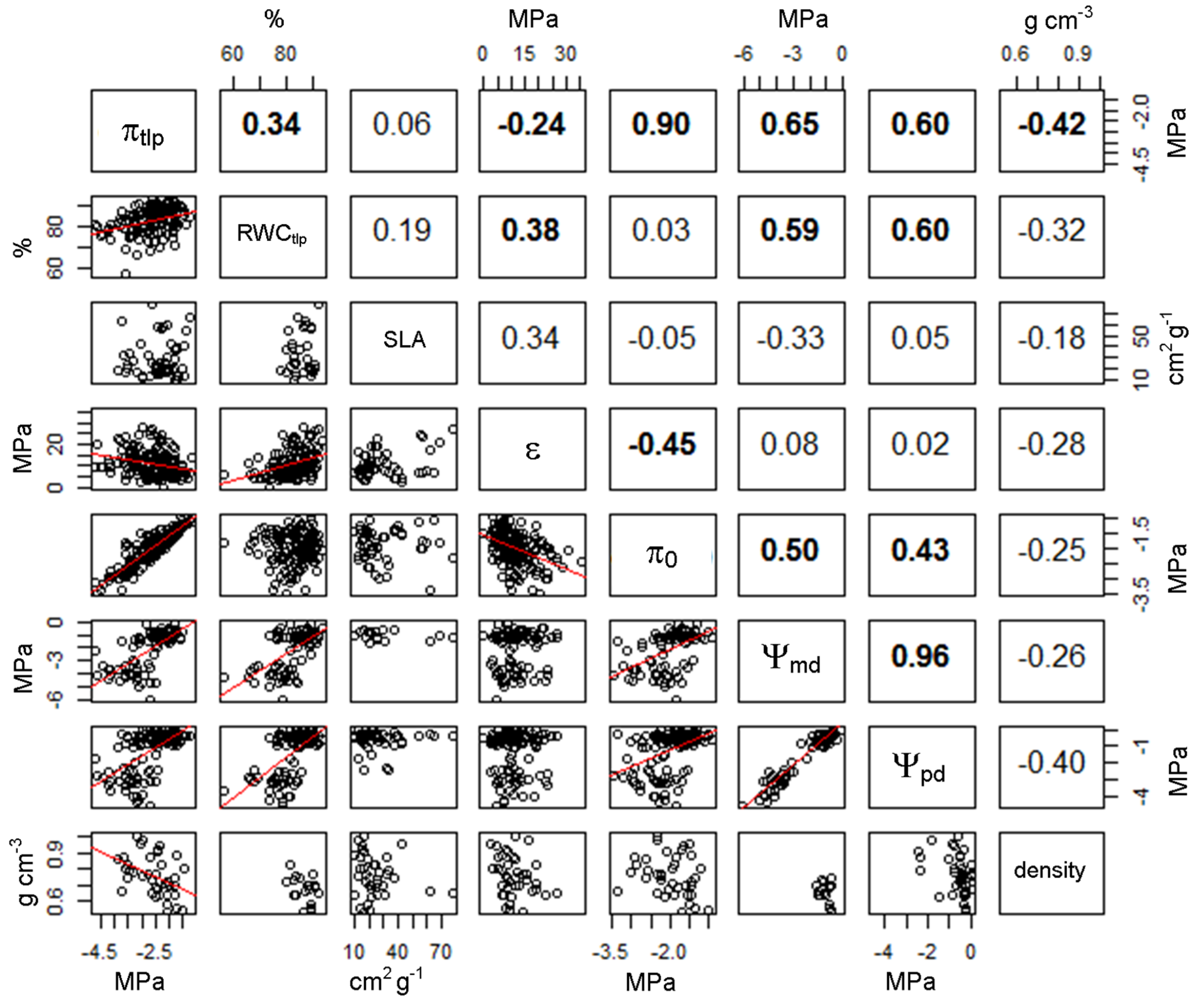
3.2. Relationship between Turgor Loss and other Leaf Traits

4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lambers, H.; Chapin, F.S.; Pons, T.L. Plant water relations. In Plant Physiological Ecology; Springer: New York, NY, USA, 2008; pp. 154–204. [Google Scholar]
- Cleland, R.E. Cell wall extension. Annu. Rev. Plant Physiol. 1971, 22, 197–222. [Google Scholar] [CrossRef]
- Franks, P.J.; Cowan, I.R.; Tyerman, S.D.; Cleary, A.I.; Lloyd, J.; Farquhar, G.D. Guard-cell pressure aperture characteristics measured with the pressure probe. Plant Cell Environ. 1995, 18, 795–800. [Google Scholar] [CrossRef]
- Frensch, J.; Hsiao, T.C. Transient responses of cell turgor and growth of maize roots as affected by changes in water potential. Plant Physiol. 1994, 104, 247–254. [Google Scholar] [PubMed]
- Fan, S.H.; Blake, T.J.; Blumwald, E. The relative contribution of elastic and osmotic adjustments to turgor maintenance of woody species. Physiol. Plant 1994, 90, 408–413. [Google Scholar] [CrossRef]
- Sanders, G.; Arndt, S. Osmotic Adjustment Under Drought Conditions. In Plant Responses to Drought Stress; Aroca, R., Ed.; Springer: Berlin, Germany, 2012; pp. 199–229. [Google Scholar]
- Tyree, M.; Hammel, H. The measurement of the turgor pressure and the water relations of plants by the pressure-bomb technique. J. Exp. Bot. 1972, 23, 267–282. [Google Scholar] [CrossRef]
- Merchant, A.; Callister, A.; Arndt, S.K.; Tausz, M.; Adams, M.A. Contrasting physiological responses of six Eucalyptus species to water deficit. Ann. Bot. 2007, 100, 1507–1515. [Google Scholar] [CrossRef] [PubMed]
- Davidson, N.J.; Reid, J.B. Response of eucalypt species to drought. Aust. J. Ecol. 1989, 14, 139–156. [Google Scholar] [CrossRef]
- Bartlett, M.K.; Scoffoni, C.; Sack, L. The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: A global meta-analysis. Ecol. Lett. 2012, 15, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Brodribb, T.J.; Holbrook, N.M. Stomatal Closure during Leaf Dehydration, Correlation with Other Leaf Physiological Traits. Plant Physiol. 2003, 132, 2166–2173. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.J.; O’Grady, A.P.; Tissue, D.T.; White, D.A.; Ottenschlaeger, M.L.; Pinkard, E.A. Drought response strategies define the relative contributions of hydraulic dysfunction and carbohydrate depletion during tree mortality. New Phytol. 2013, 197, 862–872. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.J.; Veneklaas, E.J.; Lambers, H.; Burgess, S.S.O. Using multiple trait associations to define hydraulic functional types in plant communities of south-western Australia. Oecologia 2008, 158, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.J.; Veneklaas, E.J.; Lambers, H.; Burgess, S.S.O. Leaf water relations during summer water deficit: Differential responses in turgor maintenance and variation in leaf structure among different plant communities in south-western Australia. Plant Cell Environ. 2008, 31, 1791–1802. [Google Scholar] [CrossRef] [PubMed]
- Niinemets, U. Global-scale climatic controls of leaf dry mass per area, density and thickness in trees and shrubs. Ecology 2001, 82, 453–469. [Google Scholar] [CrossRef]
- Pearce, J.L.; Boyce, M.S. Modelling distribution and abundance with presence-only data. J. Appl. Ecol. 2006, 43, 405–412. [Google Scholar] [CrossRef]
- Adams, M.A. Distribution of eucalypts in Australian landscapes: Landforms, soils, fire and nutrition. In Nutrition of the Eucalypts; Attiwill, P.M., Ed.; CSIRO: Melbourne, Australia, 1995; pp. 61–76. [Google Scholar]
- Specht, R.L.; Specht, A. Canopy structure in Eucalyptus-dominated communities in Australia along climatic gradients. Acta Ecol. 1989, 10, 191–213. [Google Scholar]
- Booth, T.H.; Williams, K.J.; Belbin, L. Developing biodiverse plantings suitable for changing climatic conditions 2: Using the Atlas of Living Australia. Ecol. Manag. Restor. 2012, 13, 274–281. [Google Scholar] [CrossRef]
- Hutchinson, M.F.; Kesteven, J.L. Monthly Mean Climate Surfaces for Australia; Centre for Resource and Environmental Studies, Australian National University: Canberra, Australia, 1998. [Google Scholar]
- Mitchell, P.J.; O’Grady, A.P.; Hayes, K.R.; Pinkard, E.A. Exposure of trees to drought-induced die-off is defined by a common climatic threshold across different vegetation types. Ecol. Evol. 2014, 4, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- Barton, K. Multi-model inference. In R Package Version 1.7.2.; R Foundation for Statistical Computing: Vienna, Austria, 2012. [Google Scholar]
- Bartlett, M.K.; Zhang, Y.; Kreidler, N.; Sun, S.; Ardy, R.; Cao, K.; Sack, L. Global analysis of plasticity in turgor loss point, a key drought tolerance trait. Ecol. Lett. 2014, 17, 1580–1590. [Google Scholar] [CrossRef] [PubMed]
- Forseth, I. Terrestrial Biomes. Nat. Educ. Knowl. 2012, 10, 11. [Google Scholar]
- Lenz, T.I.; Wright, I.J.; Westoby, M. Interrelations among pressure-volume curve traits across species and water availability gradients. Physiol. Plant 2006, 127, 423–433. [Google Scholar] [CrossRef]
- White, D.A.; Battaglia, M.; Macfarlane, C.; Mummery, D.; McGrath, J.F.; Beadle, C.L. Selecting species for recharge management in Mediterranean south Western Australia—Some ecophysiological considerations. Plant Soil 2003, 257, 283–293. [Google Scholar] [CrossRef]
- Valladares, F.; Pearcy, R.W. Interactions between water stress, sun-shade acclimation, heat tolerance and photoinhibition in the sclerophyll Heteromeles arbutifolia. Plant Cell Environ. 1997, 20, 25–36. [Google Scholar] [CrossRef]
- Niu, S.; Li, Z.; Xia, J.; Han, Y.; Wu, M.; Wan, S. Climatic warming changes plant photosynthesis and its temperature dependence in a temperate steppe of northern China. Environ. Exp. Bot. 2008, 63, 91–101. [Google Scholar] [CrossRef]
- Groom, P.K.; Lamont, B.A.; Leighton, S.; Leighton, P.; Burrows, C. Heat damage in sclerophylls is influenced by their leaf properties and plant environment. Ecoscience 2004, 11, 94–101. [Google Scholar]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Araya, Y.N.; Silvertown, J.; Gowing, D.J.; McConway, K.J.; Peter Linder, H.; Midgley, G. A fundamental, eco-hydrological basis for niche segregation in plant communities. New Phytol. 2011, 189, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Burgess, S.S.O. Measuring transpiration responses to summer precipitation in a Mediterranean climate: A simple screening tool for identifying plant water-use strategies. Physiol. Plant 2006, 127, 404–412. [Google Scholar] [CrossRef]
- Choat, B.; Jansen, S.; Brodribb, T.J.; Cochard, H.; Delzon, S.; Bhaskar, R.; Bucci, S.J.; Feild, T.S.; Gleason, S.M.; Hacke, U.G.; et al. Global convergence in the vulnerability of forests to drought. Nature 2012, 491, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Joly, J.J.; Zaerr, J.B. Alteration of cell-wall water content and elasticity in Douglas-Fir during periods of Water Deficit. Plant Physiol. 1987, 83, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Clifford, S.C.; Arndt, S.K.; Corlett, J.E.; Joshi, S.; Sankhla, M.; Jones, H.G. The role of solute accumulation, osmotic adjustment and changes in cell wall elasticity in drought tolerance Ziziphus mauritiana (Lamk). J. Exp. Bot. 1998, 49, 967–977. [Google Scholar] [CrossRef]
- White, D.A.; Turner, N.C.; Galbraith, J.H. Leaf water realtions and stomatal behaviour of four allopatric Eucalyptus species planted in Mediterranean southwestern Australia. Tree Physiol. 2000, 20, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Dalmaris, E. Eucalyptus Wandoo: Tolerance to Drought and Salinity in Relation to Provenance and Evolutionary History in Southwestern Australia. Ph.D. Thesis, the University of Western Australia, Perth, Australia, July 2012. [Google Scholar]
- Pita, P.; Pardos, J. Growth, leaf morphology, water use and tissue water relations of Eucalyptus globulus clones in response to water deficit. Tree Physiol. 2001, 21, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Tuomela, K. Leaf water relations in six provenances of Eucalyptus microtheca: A greenhouse experiment. For. Ecol. Manag. 1997, 92, 1–10. [Google Scholar] [CrossRef]
- Blake, J.; Hill, R. An Examination of the Drought and Frost Tolerance of Banksia marginata (Proteaceae) as an Explanation of Its Current Widespread Occurrence in Tasmania. Aust. J. Bot. 1996, 44, 265–281. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitchell, P.J.; O'Grady, A.P. Adaptation of Leaf Water Relations to Climatic and Habitat Water Availability. Forests 2015, 6, 2281-2295. https://doi.org/10.3390/f6072281
Mitchell PJ, O'Grady AP. Adaptation of Leaf Water Relations to Climatic and Habitat Water Availability. Forests. 2015; 6(7):2281-2295. https://doi.org/10.3390/f6072281
Chicago/Turabian StyleMitchell, Patrick J., and Anthony P. O'Grady. 2015. "Adaptation of Leaf Water Relations to Climatic and Habitat Water Availability" Forests 6, no. 7: 2281-2295. https://doi.org/10.3390/f6072281
APA StyleMitchell, P. J., & O'Grady, A. P. (2015). Adaptation of Leaf Water Relations to Climatic and Habitat Water Availability. Forests, 6(7), 2281-2295. https://doi.org/10.3390/f6072281





