Species Distribution Model for Management of an Invasive Vine in Forestlands of Eastern Texas
Abstract
:1. Introduction
2. Methods
2.1. Study Area and Data Sources
2.2. Quantification of the Range Expansion
2.3. Identification of Potential Factors Influencing Likelihood of Invasion
| Variable | Value or Unit of Measure | Mean (Range) for Continuous Data/Count for Categorical Data |
|---|---|---|
| Landscape conditions | ||
| Slope | Degree | 2.07 (0.00–32.50) |
| Adjacency to water bodies within 300 m | No | 1830 |
| Yes | 471 | |
| Climate conditions | ||
| Mean annual precipitation | cm | 127.06 (99.06–154.94) |
| Mean annual minimum temperature | °C | 12.43 (10.56–16.67) |
| Mean annual maximum temperature | °C | 24.89 (22.78–26.11) |
| Forest features | ||
| Stand age | Year | 35.76 (1–104) |
| Site productivity | L1: 0–1.39 m3·ha−1·year−1 | 9 |
| L2: 1.40–3.49 | 154 | |
| L3: 3.50–5.94 | 660 | |
| L4: 5.95–8.39 | 931 | |
| L5: 8.40–11.54 | 541 | |
| L6: 11.55–15.74 | 95 | |
| L7: >15.74 | 4 | |
| Basal area | m2 | 1.26 (0.00–7.10) |
| Species diversity | Shannon’s species diversity | 1.02 (0.00–2.53) |
| Natural regeneration a | No | 2348 |
| Yes | 46 | |
| Disturbance factors | ||
| Distance to the nearest road | D1: <30 m | 169 |
| D2: 30–91 | 382 | |
| D3: 92–152 | 273 | |
| D4: 153–305 | 492 | |
| D5: 306–805 | 674 | |
| D6: 806–1609 | 239 | |
| D7: 1610–4828 | 62 | |
| D8: 4829–8047 | 3 | |
| D9: >8047 | 2 | |
| Distance to the nearest known propagule source | m | 14,343 (0–75,529) |
| Forest disturbance a,b | No | 2294 |
| Yes | 100 | |
| Forest management activities | ||
| Harvesting a | No | 1969 |
| Yes | 425 | |
| Site preparation a | No | 2309 |
| Yes | 85 | |
| Artificial regeneration a | No | 1802 |
| Yes | 592 |
2.4. Data Analysis
2.5. Likelihood of Invasion
3. Results
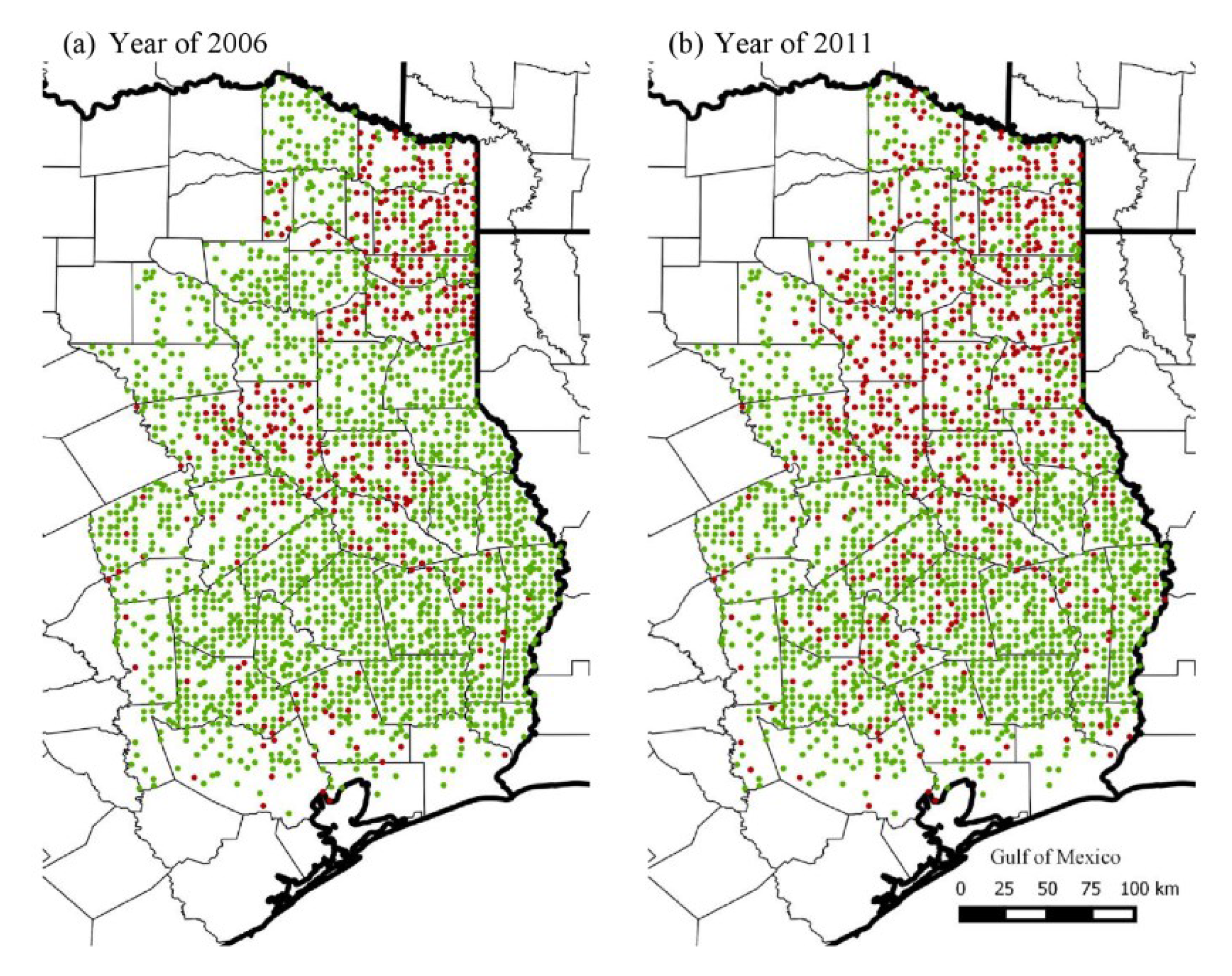
3.1. Historical Trends in Range Expansion
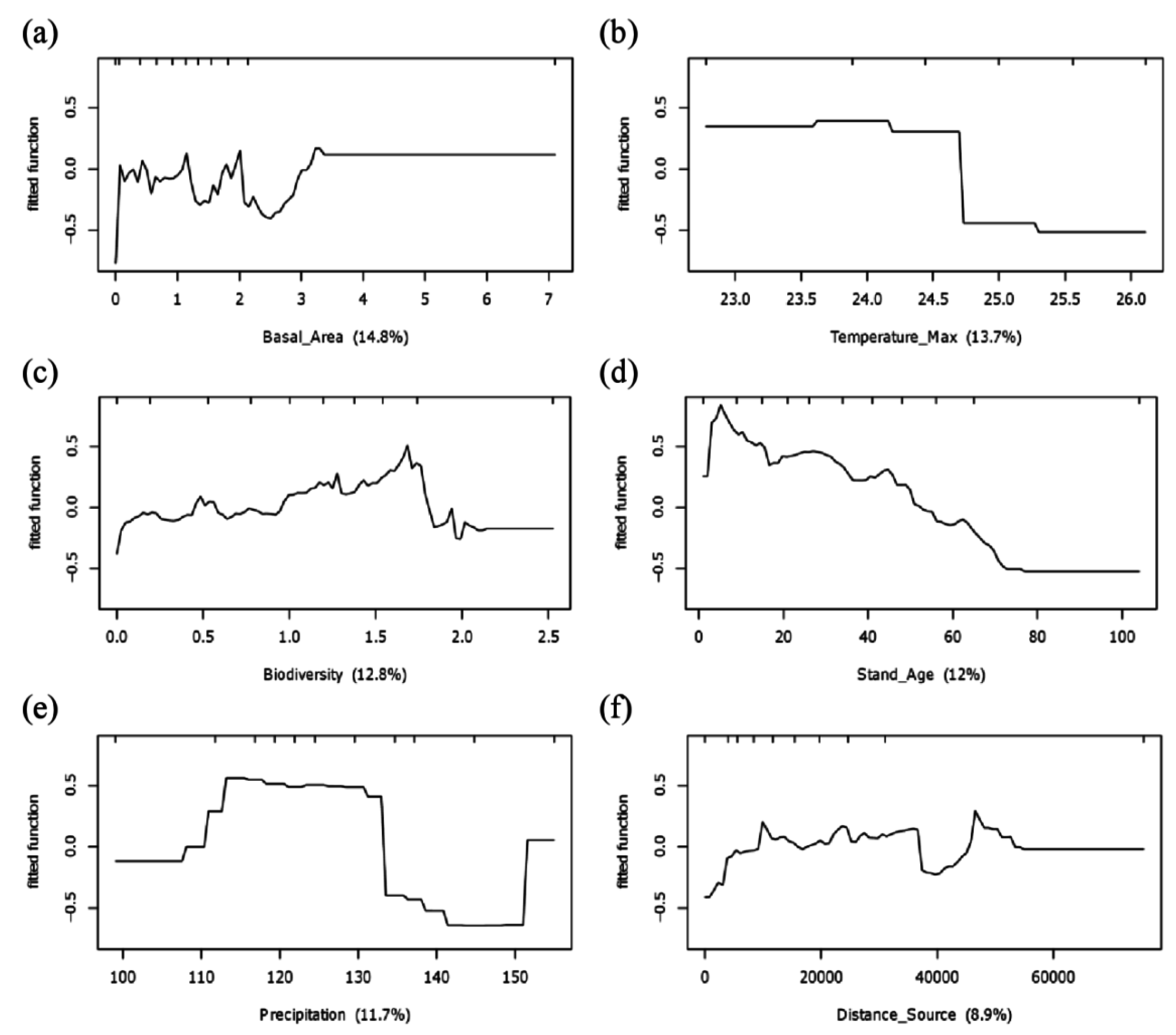
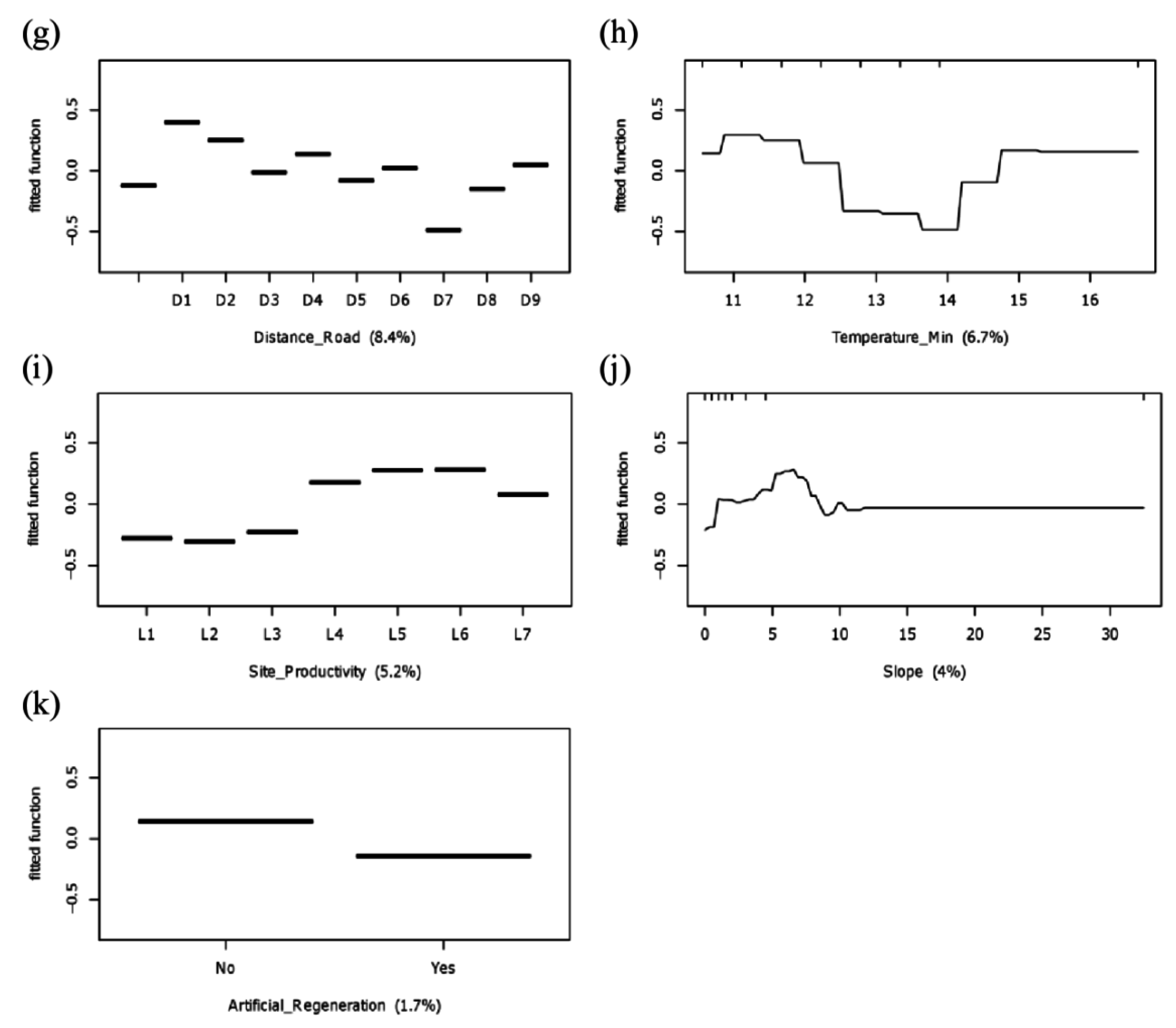
3.2. Potential Determinants of Invasion
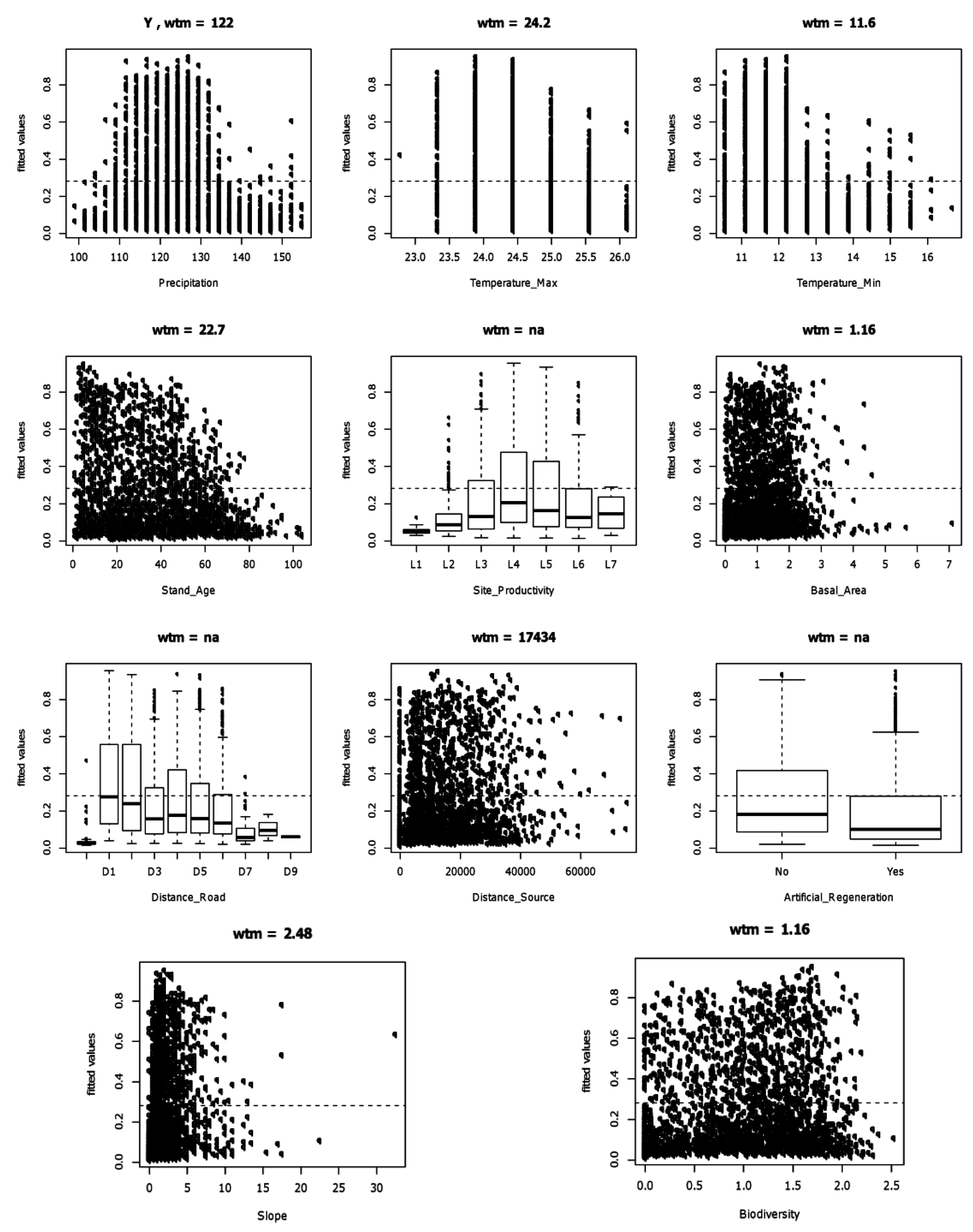
3.3. Likelihood of Invasion under Current Conditions and Different Levels of Management
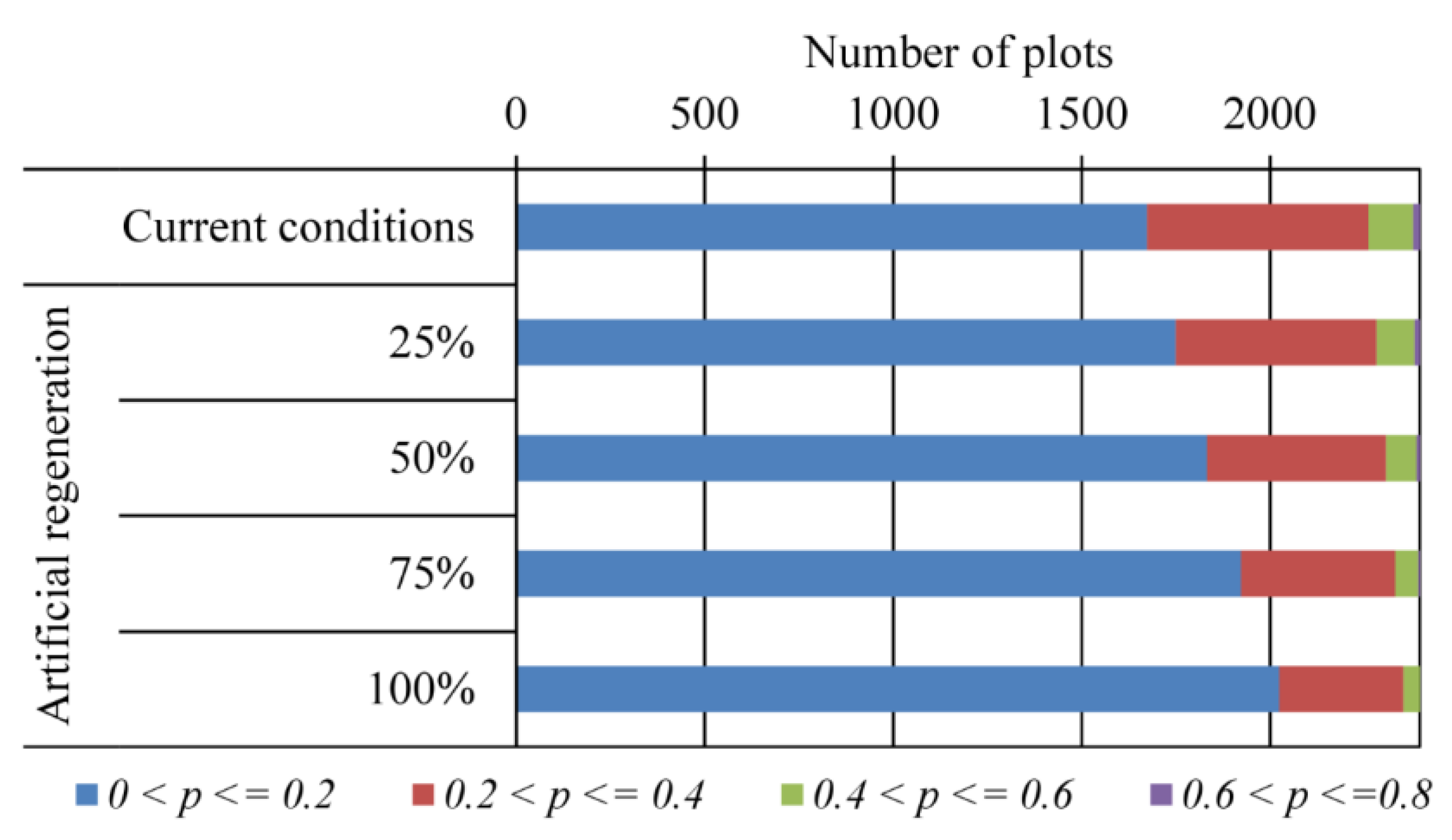
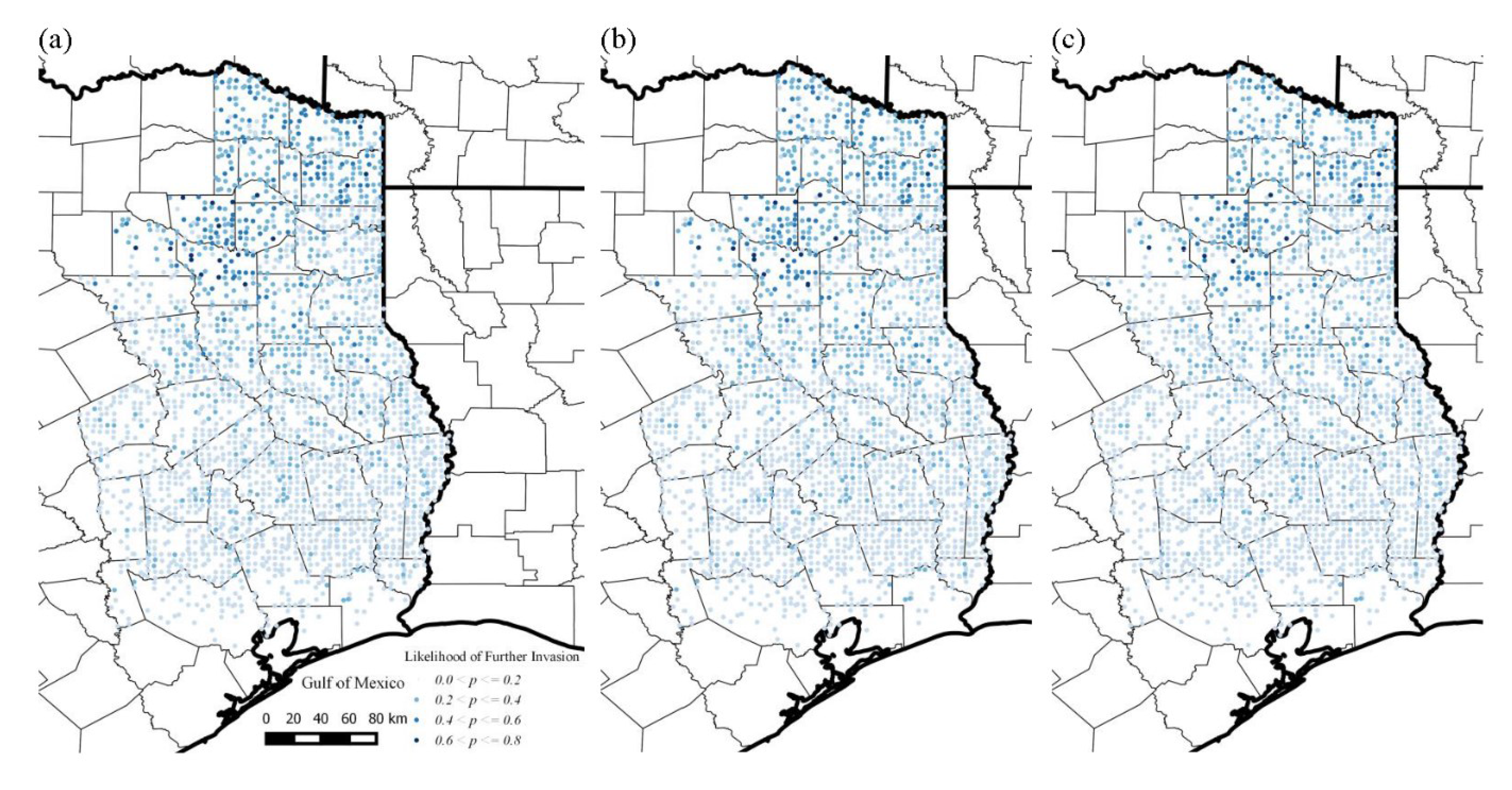
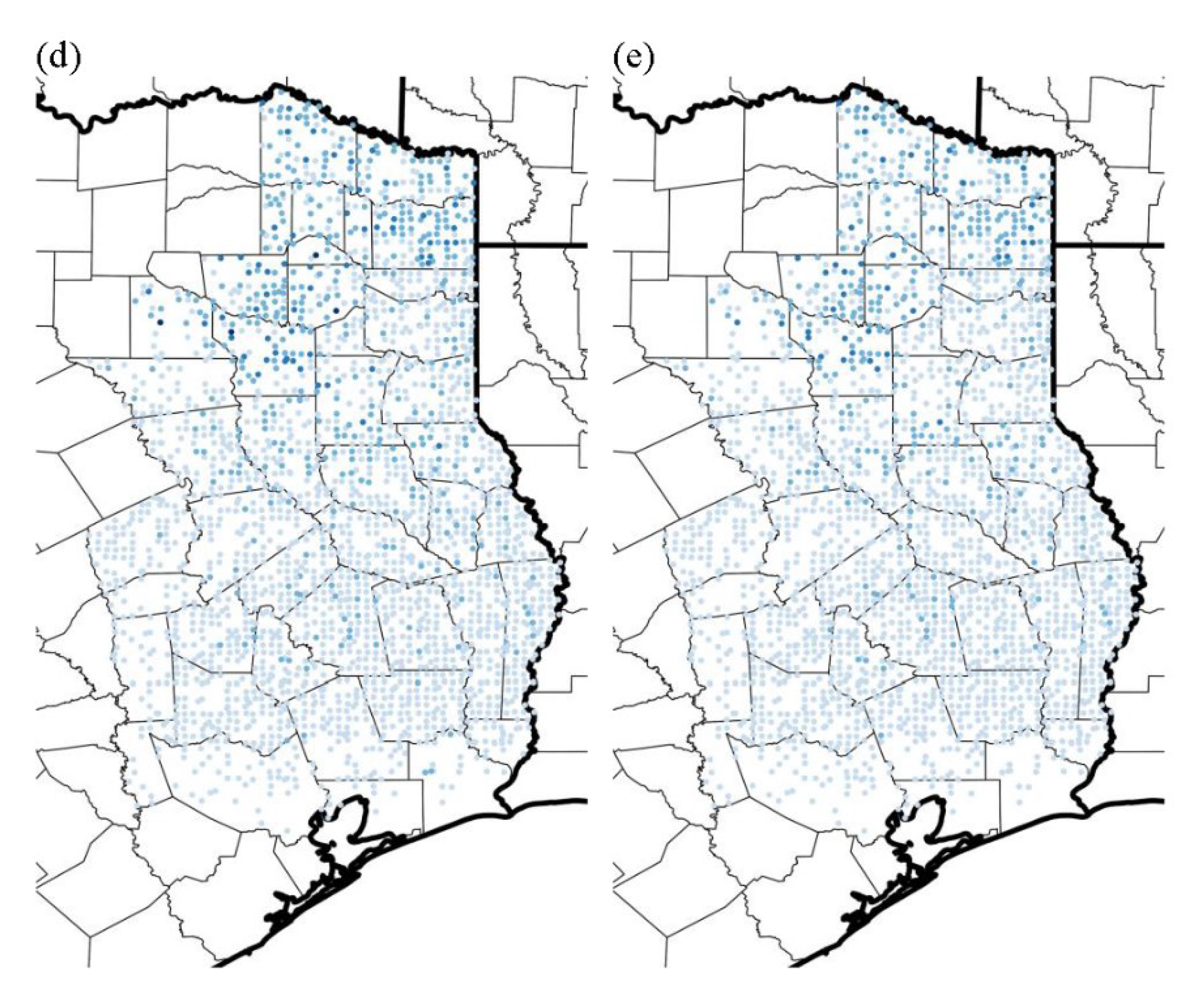
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Moser, W.K.; Barnard, E.L.; Billings, R.F.; Crocker, S.J.; Dix, M.E.; Gray, A.N.; Ice, G.G.; Kim, M.-S.; Reid, R.; Rodman, S.U.; et al. Impacts of nonnative invasive species on US forests and recommendations for policy and management. J. For. 2009, 107, 320–327. [Google Scholar]
- Pejchar, L.; Mooney, H.A. Invasive species, ecosystem services and human well-being. Trends Ecol. Evol. 2009, 24, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, D.; Zuniga, R.; Morrison, D. Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol. Econ. 2005, 52, 273–288. [Google Scholar] [CrossRef]
- Wilcove, D.S.; Rothstein, D.; Jason, D.; Phillips, A.; Losos, E. Quantifying threats to imperiled species in the United States. BioScience 1998, 48, 607–615. [Google Scholar] [CrossRef]
- Gurevitch, J.; Padilla, D.K. Are invasive species a major cause of extinctions? Trends Ecol. Evol. 2004, 19, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-H.; Grant, W.E.; Gan, J.; Rogers, W.E.; Swannack, T.M.; Koralewski, T.E.; Miller, J.H.; Taylor, J.W. Integrating spread dynamics and economics of timber production to manage Chinese tallow invasions in southern US forestlands. PLoS ONE 2012, 7, e33877. [Google Scholar] [CrossRef] [PubMed]
- McNulty, S.G.; Moore, J.A.; Iverson, L.; Prasad, A.; Abt, R.; Smith, B.; Sun, G.; Gavazzi, M.; Bartlett, J.; Murray, B.; et al. Application of linked regional scale growth, biogeography, and economic models for southeastern United States pine forests. World Resour. Rev. 2000, 12, 298–320. [Google Scholar]
- Linder, E.T. Biodiversity and Southern Forests; General Technical Report SRS 75; US Department of Agriculture, Forest Service, Southern Research Station: Asheville, NC, USA, 2004; pp. 303–306.
- Hayes, D. Earth day 1990: Threshold of the green decade. World Policy J. 1990, 7, 289–304. [Google Scholar]
- Reubens, B.; Poesen, J.; Danjon, F.; Geudens, G.; Muys, B. The role of fine and coarse roots in shallow slope stability and soil erosion control with a focus on root system architecture: A review. Trees 2007, 21, 385–402. [Google Scholar] [CrossRef]
- Schierenbeck, K.A. Japanese honeysuckle (Lonicera japonica) as an invasive species; history, ecology, and context. Crit. Rev. Plant Sci. 2004, 23, 391–400. [Google Scholar] [CrossRef]
- Natural Resources Conservation Service (NRCS). The PLANTS Database. Available online: http://plants.usda.gov (accessed on 1 July 2015).
- Hardt, R.A. Japanese honeysuckle: From “one of the best” to ruthless pest. Arnoldia 1986, 46, 27–34. [Google Scholar]
- Andrews, E.F. The Japanese honeysuckle in the eastern United States. Torreya 1919, 19, 37–43. [Google Scholar]
- Leatherman, A.D. Ecological Life-History of Lonicera japonica Thunb. Ph.D. Thesis, University of Tennessee, Knoxville, TN, USA, 1955. [Google Scholar]
- Schierenbeck, K.A. Comparative ecological and genetic studies between a native (Lonicera sempervirens L.) and an introduced congener (L. japonica Thunb.). Ph.D. Thesis, Washington State University, Pullman, WA, USA, 1992. [Google Scholar]
- West, N.M.; Gibson, D.J.; Minchin, P.R. Microhabitat analysis of the invasive exotic liana Lonicera japonica Thunb. J. Torrey Bot. Soc. 2010, 137, 380–390. [Google Scholar] [CrossRef]
- Garrison, G.A.; Bjugstad, A.J.; Duncan, D.A.; Lewis, M.E.; Smith, D.R. Vegetation and Environmental Features of Forest and Range Ecosystems; Forest Service, Department of Agriculture: Washington, DC, USA, 1977; p. 68.
- Nyboer, R. Vegetation management guideline—Japanese honeysuckle (Lonicera japonica Thunb.). Nat. Areas J. 1992, 12, 217–218. [Google Scholar]
- Oosting, H.J.; Livingston, R.B. A resurvey of a loblolly pine community twenty-nine years after ground and crown fire. Bull. Torrey Bot. Club 1964, 91, 387–395. [Google Scholar] [CrossRef]
- Yurkonis, K.A.; Meiners, S.J. Invasion impacts local species turnover in a successional system. Ecol. Lett. 2004, 7, 764–769. [Google Scholar] [CrossRef]
- McLemore, B.F. Comparison of Three Methods for Regenerating Honeysuckle-Infested Openings in Uneven-Aged Loblolly Pine Stands; Southern Forest Experiment Station, Forest Service, US Department of Agriculture: Atlanta, GA, USA, 1985.
- Wear, D.N.; Greis, J.G. Southern forest resource assessment: Summary of findings. J. For. 2002, 100, 6–14. [Google Scholar]
- McKeand, S.; Mullin, T.; Byram, T.; White, T. Deployment of genetically improved loblolly and slash pines in the south. J. For. 2003, 101, 32–37. [Google Scholar]
- United States Department of Agriculture (USDA). Southern Nonnative Invasive Plant data Extraction Tool (SNIPET). Available online: http://srsfia2.fs.fed.us/data_center/index.shtml (accessed on 1 July 2015).
- Wang, H.-H.; Wonkka, C.L.; Grant, W.E.; Rogers, W.E. Potential range expansion of Japanese honeysuckle (Lonicera japonica Thunb.) in southern US forestlands. Forests 2012, 3, 573–590. [Google Scholar] [CrossRef]
- Gan, J.; Smith, C.T. Co-benefits of utilizing logging residues for bioenergy production: The case for East Texas, USA. Biomass Bioenergy 2007, 31, 623–630. [Google Scholar] [CrossRef]
- Rudis, V.A.; Gray, A.; McWilliams, W.; O’Brien, R.; Olson, C.; Oswalt, S.; Schulz, B. Regional Monitoring of Nonnative Plant Invasions with the Forest Inventory and Analysis program, Proceedings of the Sixth Annual FIA Symposium, Denver, CO, USA, 21–24 September 2006; McRoberts, R.E., Reams, G.A., Eds.; General Technical Report WO-70. USDA Forest Service: Denver, CO, USA, 2006; pp. 49–64.
- Bechtold, W.A.; Patterson, P.L. The Enhanced Forest Inventory and Analysis Program—National Sampling Design and Estimation Procedures; General Technical Report SRS-80; Southern Research Station, Forest Service, US Department of Agriculture: Asheville, NC, USA, 2005; p. 14.
- United States Department of Agriculture (USDA). The Forest Inventory and Analysis Database: Database Description and Users Manual Version 5.1; US Department of Agriculture Forest Service: Arlington, VA, USA, 2011; pp. 12–15.
- Lemke, D.; Hulme, P.E.; Brown, J.A.; Tadesse, W. Distribution modelling of Japanese honeysuckle (Lonicera japonica) invasion in the Cumberland Plateau and Mountain Region, USA. For. Ecol. Manag. 2011, 262, 139–149. [Google Scholar] [CrossRef]
- Lemke, D.; Schweitzer, C.J.; Tadesse, W.; Wang, Y.; Brown, J.A. Geospatial assessment of invasive plants on reclaimed mines in Alabama. Invasive Plant Sci. Manag. 2013, 6, 401–410. [Google Scholar] [CrossRef]
- Daly, C.; Halbleib, M.; Smith, J.I.; Gibson, W.P.; Doggett, M.K.; Taylor, G.H.; Curtis, J.; Pasteris, P.P. Physiographically sensitive mapping of climatological temperature and precipitation across the conterminous United States. Int. J. Climatol. 2008, 28, 2031–2064. [Google Scholar] [CrossRef]
- Belda, E.J.; Sánchez, A. Seabird mortality on longline fisheries in the western Mediterranean: Factors affecting bycatch and proposed mitigating measures. Biol. Conserv. 2001, 98, 357–363. [Google Scholar] [CrossRef]
- Wills, C.; Condit, R.; Foster, R.B.; Hubbell, S.P. Strong density- and diversity-related effects help to maintain tree species diversity in a neotropical forest. Proc. Natl. Acad. Sci. USA 1997, 94, 1252–1257. [Google Scholar] [CrossRef] [PubMed]
- Filipescu, C.N.; Comeau, P.G. Competitive interactions between aspen and white spruce vary with stand age in boreal mixedwoods. For. Ecol. Manag. 2007, 247, 175–184. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R.; Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol. 2008, 77, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Ridgeway, G. Generalized Boosted Regression Models. Documentation on the R Package ‘gbm’, Version 1-5-7. Available online: http://www.i-pensieri.com/gregr/gbm.shtml (accessed on 4 April 2012).
- Friedman, J.H. Stochastic gradient boosting. Comput. Stat. Data Anal. 2002, 38, 367–378. [Google Scholar] [CrossRef]
- Hosmer, D.W.; Lemeshow, S. Applied Logistic Regression; John Wiley and Sons, Inc.: New York, NY, USA, 2000. [Google Scholar]
- Morales, J.M.; Carlo, T.A. The effects of plant distribution and frugivore density on the scale and shape of dispersal kernels. Ecology 2006, 87, 1489–1496. [Google Scholar] [CrossRef]
- Clark, J.S.; Lewis, M.; McLachlan, J.S.; HilleRisLambers, J. Estimating population spread: What can we forecast and how well? Ecology 2003, 84, 1979–1988. [Google Scholar] [CrossRef]
- Davies, K.F.; Harrison, S.; Safford, H.D.; Viers, J.H. Productivity alters the scale dependence of the diversity-invasibility relationship. Ecology 2007, 88, 1940–1947. [Google Scholar] [CrossRef] [PubMed]
- Baars, R.; Kelly, D. Survival and growth responses of native and introduced vines in New Zealand to light availability. N. Z. J. Bot. 1996, 34, 389–400. [Google Scholar] [CrossRef]
- Carter, G.A.; Teramura, A.H. Vine photosynthesis and relationships to climbing mechanics in a forest understory. Am. J. Bot. 1988, 75, 1011–1018. [Google Scholar] [CrossRef]
- Halls, L.K. Flowering and Fruiting of Southern Browse Species; US Department of Agriculture, Forest Service, Southern Forest Experiment Station: New Orleans, LA, USA, 1973; p. 10.
- Blair, R.M. Growth and nonstructural carbohydrate content of southern browse species as influenced by light intensity. J. Range Manag. 1982, 35, 756–760. [Google Scholar] [CrossRef]
- Elton, C.S. The Ecology of Invasions by Plants and Anmials; University of Chicago Press: Chicago, IL, USA, 1958. [Google Scholar]
- Hector, A.; Dobson, K.; Minns, A.; Bazeley-White, E.; Hartley Lawton, J. Community diversity and invasion resistance: An experimental test in a grassland ecosystem and a review of comparable studies. Ecol. Res. 2001, 16, 819–831. [Google Scholar] [CrossRef]
- Levine, J.M. Species diversity and biological invasions: Relating local process to community pattern. Science 2000, 288, 852–854. [Google Scholar] [CrossRef] [PubMed]
- Lyons, K.G.; Schwartz, M.W. Rare species loss alters ecosystem function—Invasion resistance. Ecol. Lett. 2001, 4, 358–365. [Google Scholar] [CrossRef]
- Levine, J.M.; D’Antonio, C.M. Elton revisited: A review of evidence linking diversity and invasibility. Oikos 1999, 87, 15–26. [Google Scholar] [CrossRef]
- Wang, H.-H.; Grant, W.E. Determinants of Chinese and European privet (Ligustrum sinense and Ligustrum vulgare) invasion and likelihood of further invasion in southern US forestlands. Invasive Plant Sci. Manag. 2012, 5, 454–463. [Google Scholar] [CrossRef]
- Richardson, D.M.; Pyšek, P.; Rejmánek, M.; Barbour, M.G.; Panetta, F.D.; West, C.J. Naturalization and invasion of alien plants: Concepts and definitions. Divers. Distrib. 2000, 6, 93–107. [Google Scholar] [CrossRef]
- Mitchell, C.E.; Agrawal, A.A.; Bever, J.D.; Gilbert, G.S.; Hufbauer, R.A.; Klironomos, J.N.; Maron, J.L.; Morris, W.F.; Parker, I.M.; Power, A.G.; et al. Biotic interactions and plant invasions. Ecol. Lett. 2006, 9, 726–740. [Google Scholar] [CrossRef] [PubMed]
- Pelczar, R. When vigor is not a virtue: Beware the beautiful but invasive Japanese honeysuckle. Horticulture 1995, 73, 64–66. [Google Scholar]
- Sasek, T.W.; Strain, B.R. Implications of atmospheric CO2 enrichment and climatic change for the geographical distribution of two introduced vines in the USA. Clim. Chang. 1990, 16, 31–51. [Google Scholar] [CrossRef]
- Holle, B.V.; Simberloff, D. Ecological resistance to biological invasion overwhelmed by propagule pressure. Ecology 2005, 86, 3212–3218. [Google Scholar] [CrossRef]
- Merritt, D.M.; Nilsson, C.; Jansson, R. Consequences of propagule dispersal and river fragmentation for riparian plant community diversity and turnover. Ecol. Monogr. 2010, 80, 609–626. [Google Scholar] [CrossRef]
- Primack, R.B.; Miao, S.L. Dispersal can limit local plant distribution. Conserv. Biol. 1992, 6, 513–519. [Google Scholar] [CrossRef]
- Tilman, D. Community invasibility, recruitment limitation, and grassland biodiversity. Ecology 1997, 78, 81–92. [Google Scholar] [CrossRef]
- Moody, M.E.; Mack, R.N. Controlling the spread of plant invasions: The importance of nascent foci. J. Appl. Ecol. 1988, 25, 1009–1021. [Google Scholar] [CrossRef]
- Sheley, R.L.; Mangold, J.M.; Anderson, J.L. Potential for successional theory to guide restoration of invasive plant-dominated rangeland. Ecol. Monogr. 2006, 76, 365–379. [Google Scholar] [CrossRef]
- Miller, J.H.; Schelhas, J. Adaptive collaborative restoration: A key concept for invasive plant management. In Invasive Plants and Forest Ecosystems; Kohli, R.K., Singh, J.P., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 251–265. [Google Scholar]
- Lodge, D.M.; Williams, S.; MacIsaac, H.J.; Hayes, K.R.; Leung, B.; Reichard, S.; Mack, R.N.; Moyle, P.B.; Smith, M.; Andow, D.A.; et al. Biological invasions: Recommendations for US policy and management. Ecol. Appl. 2006, 16, 2035–2054. [Google Scholar] [CrossRef]
- Wang, H.-H.; Swannack, T.M.; Grant, W.E.; Gan, J.; Rogers, W.E.; Koralewski, T.E.; Miller, J.H.; Taylor, J.W. Predicted range expansion of Chinese tallow tree (Triadica sebifera) in forestlands of the southern United States. Divers. Distrib. 2011, 17, 552–565. [Google Scholar] [CrossRef]
- Gelbard, J.L.; Belnap, J. Roads as conduits for exotic plant invasions in a semiarid landscape. Conserv. Biol. 2003, 17, 420–432. [Google Scholar] [CrossRef]
- Parendes, L.A.; Jones, J.A. Role of light availability and sispersal in exotic plant invasion along roads and streams in the H.J. Andrews Experimental Forest, Oregon. Conserv. Biol. 2000, 14, 64–75. [Google Scholar] [CrossRef]
- Schweitzer, J.A.; Larson, K.C. Greater morphological plasticity of exotic honeysuckle species may make them better invaders than native species. J. Torrey Bot. Soc. 1999, 126, 15–23. [Google Scholar] [CrossRef]
- Pitts, T.D. Foods of eastern bluebirds during exceptionally cold weather in Tennessee. J. Wildl. Manag. 1979, 43, 752–754. [Google Scholar] [CrossRef]
- Suthers, H.B.; Bickal, J.M.; Rodewald, P.G. Use of successional habitat and fruit resources by songbirds during autumn migration in central New Jersey. Wilson Bull. 2000, 112, 249–260. [Google Scholar] [CrossRef]
- Halls, L.K. Managing deer habitat in loblolly-shortleaf pine forest. J. For. 1973, 71, 752–757. [Google Scholar]
- Nixon, C.M.; McClain, M.W.; Russell, K.R. Deer food habits and range characteristics in Ohio. J. Wildl. Manag. 1970, 34, 870–886. [Google Scholar] [CrossRef]
- Sotala, D.J.; Kirkpatrick, C.M. Foods of white-tailed deer, Odocoileus virginianus, in Martin County, Indiana. Am. Midl. Nat. 1973, 89, 281–286. [Google Scholar] [CrossRef]
- Sheldon, J.J.; Causey, K. Deer habitat management—Availability use of Japanese honeysuckle by white-tailed deer. J. For. 1974, 72, 286–287. [Google Scholar]
- Michael, E.D. Movements of white-tailed deer on the Welder Wildlife Refuge. J. Wildl. Manag. 1965, 29, 44–52. [Google Scholar] [CrossRef]
- Inglis, J.M.; Hood, R.E.; Brown, B.A.; DeYoung, C.A. Home range of white-tailed deer in Texas coastal prairie brushland. J. Mammal. 1979, 60, 377–389. [Google Scholar] [CrossRef]
- Thomas, J.W.; Teer, J.G.; Walker, E.A. Mobility and home range of white-tailed deer on the Edwards Plateau in Texas. J. Wildl. Manag. 1964, 28, 463–472. [Google Scholar] [CrossRef]
- Sage, R.W., Jr.; Tierson, W.C.; Mattfeld, G.F.; Behrend, D.F. White-tailed deer visibility and behavior along forest roads. J. Wildl. Manag. 1983, 47, 940–953. [Google Scholar] [CrossRef]
- Kilgo, J.C.; Labisky, R.F.; Fritzen, D.E. Influences of hunting on the behavior of white-tailed deer: Implications for conservation of the Florida panther. Conserv. Biol. 1998, 12, 1359–1364. [Google Scholar] [CrossRef]
- Conner, W.; Stanturf, J.A.; Gardiner, E.S.; Schweitzer, C.J.; Ezell, A.W. Recognizing and overcoming difficult site conditions for afforestation of bottomland hardwoods. Ecol. Restor. 2004, 22, 183–193. [Google Scholar]
- Jackson, L.W. Honeysuckles. In Shrubs and Vines for Northeastern Wildlife; Gill, J.D., Healy, W.M., Eds.; US Department of Agriculture, Forest Service, Northeastern Forest Experiment Station: Upper Darby, PA, USA, 1974; pp. 71–82. [Google Scholar]
- Elith, J.; Graham, C.H. Do they? How do they? WHY do they differ? On finding reasons for differing performances of species distribution models. Ecography 2009, 32, 66–77. [Google Scholar] [CrossRef]
- Agresti, A. An Introduction to Categorical Data Analysis; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2007. [Google Scholar]
- Walters, C.J. Adaptive Management of Renewable Resources; Macmillan Publishing Company: New York, NY, USA, 1986. [Google Scholar]
- Barry, S.; Elith, J. Error and uncertainty in habitat models. J. Appl. Ecol. 2006, 43, 413–423. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R. Species distribution models: Ecological explanation and prediction across space and time. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 677–697. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.-H.; Koralewski, T.E.; McGrew, E.K.; Grant, W.E.; Byram, T.D. Species Distribution Model for Management of an Invasive Vine in Forestlands of Eastern Texas. Forests 2015, 6, 4374-4390. https://doi.org/10.3390/f6124374
Wang H-H, Koralewski TE, McGrew EK, Grant WE, Byram TD. Species Distribution Model for Management of an Invasive Vine in Forestlands of Eastern Texas. Forests. 2015; 6(12):4374-4390. https://doi.org/10.3390/f6124374
Chicago/Turabian StyleWang, Hsiao-Hsuan, Tomasz E. Koralewski, Erin K. McGrew, William E. Grant, and Thomas D. Byram. 2015. "Species Distribution Model for Management of an Invasive Vine in Forestlands of Eastern Texas" Forests 6, no. 12: 4374-4390. https://doi.org/10.3390/f6124374
APA StyleWang, H.-H., Koralewski, T. E., McGrew, E. K., Grant, W. E., & Byram, T. D. (2015). Species Distribution Model for Management of an Invasive Vine in Forestlands of Eastern Texas. Forests, 6(12), 4374-4390. https://doi.org/10.3390/f6124374







