1. Introduction
Insect outbreaks are increasing in size and severity on a global scale [
1]. In North America alone, three massive insect outbreaks occurred within the last two decades, all involving native bark beetles in conifers [
2]. Of these, the mountain pine beetle (
Dendroctonus ponderosae) outbreak is an order of magnitude larger than any previously recorded. A variety of factors, natural and anthropogenic, converged to result in these dramatic events [
2]. Each outbreak has not only had severe ecological effects, but each has also triggered human responses that, for better or for worse, have resulted in additional impacts along with massive expense [
3]. Predictions are that outbreaks of bark beetles will become more frequent and severe in the future [
4,
5] indicating an imperative need to critically assess the efficacy and impacts of our approaches to their management.
Outbreaks of bark beetles are not new. They have been occurring for millennia and have played a major role in shaping coniferous forest ecosystems of the world. While considerable research has been conducted on controlling bark beetles, massive gaps in knowledge remain. In particular, there is a disturbing dearth of rigorous replicated empirical studies assessing the effects of various management strategies, particularly timber harvest treatments, for bark beetle outbreak suppression. Even fewer studies have focused on how such treatments meet explicit goals or affect forest structure, function and future outbreak dynamics [
6]. Particularly pertinent at this time, there is a lack of information to address forest adaptation to climate change in light of increasingly “out of historic norm” behavior of bark beetles. Despite this, there is a widespread belief in the policy arena that timber harvesting is an effective and necessary tool to address beetle infestations. That belief has led to proposals for, and enactment of, significant changes in federal environmental laws to encourage more timber harvests. Our question is, does that belief have a sound grounding in current science?
In this review, we focus on mountain pine beetle as an exemplar to critically evaluate the state of science behind the use of timber harvest treatments for bark beetle suppression during outbreaks. The mountain pine beetle was chosen because it is the most studied, most intensively managed, and most aggressive of the irruptive bark beetles. It has also responded strongly to climate change, resulting in a recent massive outbreak of unprecedented size that, in turn, has initiated numerous human responses, mostly involving implementation of timber harvests. It has also initiated many policy changes with many more currently in the pipeline.
We begin with an overview of the current policy situation. We then briefly review the biology of mountain pine beetle to form a foundation for understanding the factors that initiate and maintain outbreaks and how anthropogenic factors are contributing to current problems. We then describe the primary timber harvest treatments used to suppress bark beetle outbreaks and examine how well relevant science and ecological principles support their use. We conclude with a discussion on how well policy reflects the actual state of current science and identify where significant gaps between science and practice occur particularly in light of climate change. We also discuss the need to use advanced tools, including genetics and remote sensing, to adapt old practices to new situations-particularly in the realm of climate change adaptation. It is our hope that this review will stimulate research to fill important gaps and to help guide the development of policy and management firmly based in science, and thus, more likely to aid in forest conservation, reduce financial waste, and bolster public trust in public agency decision-making and practice.
2. The Current Policy Situation
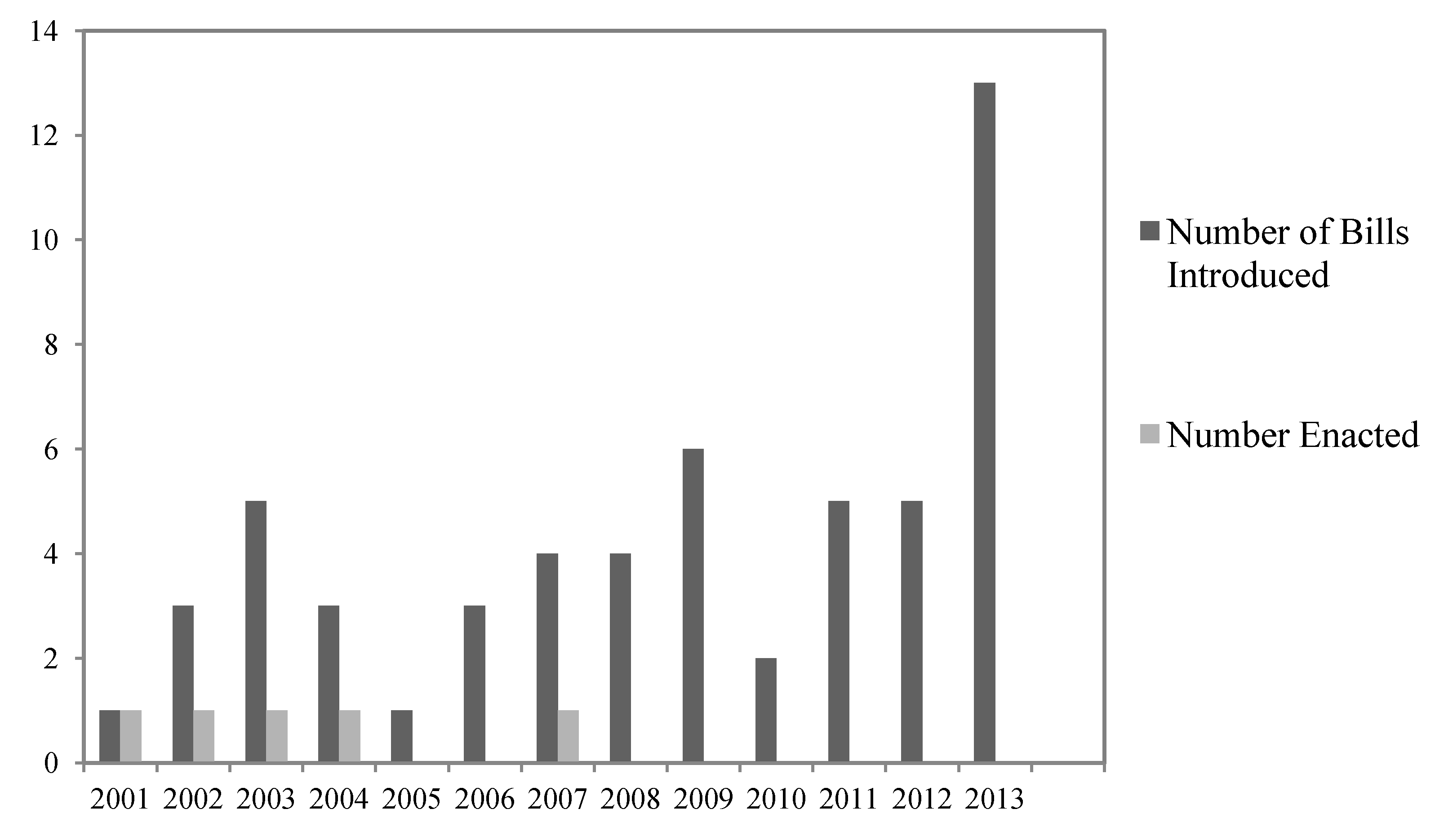
There have been many recent proposals to streamline, reduce, or eliminate perceived legal obstacles to implementing timber harvests to address beetle epidemics on federal public lands (
Figure 1). Between the 107th Congress (January 2001) and the 113th Congress (present), we found 55 bills that were introduced where at least one goal of the legislation was to increase timber harvests in order to respond to beetle infestations (
Figure 1). Most of these proposals focused on the US Forest Service, which manages the majority of forests on federal public lands.
Figure 1.
Number of bills involving timber sales that included bark beetle control that were introduced and/or enacted from 2001 to 10 July 2013.
Figure 1.
Number of bills involving timber sales that included bark beetle control that were introduced and/or enacted from 2001 to 10 July 2013.
Some of these proposals have been enacted. By far, the most important legal change has been the Healthy Forest Restoration Act of 2003 (HFRA). HFRA reduced the level of environmental analysis required for certain timber projects under the National Environmental Policy Act (NEPA), specifically by limiting the number of alternatives that the Forest Service was required to analyze. It also significantly restricted the ability of members of the public to challenge certain timber projects in court (by making participation in the agency’s administrative process a precondition for filing suit). Further, it sought to streamline the Forest Service’s internal administrative process for considering citizen challenges to certain timber projects. HFRA applies nationally to all National Forest System and Bureau of Land Management lands, and has resulted in forest treatment projects on an average of 220,000 acres of federal land per year since its enactment [
7]
HFRA authorizes this streamlined process for timber projects on “Federal land on which…the existence of an epidemic of disease or insects, or the presence of such an epidemic on immediately adjacent land and the imminent risk it will spread, poses a significant threat to an ecosystem component, or forest or rangeland resource, on the Federal land or adjacent non-Federal land” [
8,
9]. Moreover, while other types of HFRA projects in old growth forests are subject to limitations intended to protect old growth structure and large trees, timber projects to address insect epidemics can occur in old growth forests without those limitations [
10,
11].
HFRA also sets up a special experimental management process to develop better management methods for beetle infestations. After a long list of findings by Congress about the risks of beetle infestations in US forests, Congress authorized up to 250,000 acres of “applied silvicultural assessment and research treatments” on National Forests that would be categorically excluded from NEPA; these treatments could include timber harvesting [
12,
13]. HFRA section 401(b)(3) [
14] requires that these applied silvicultural assessments and treatments must be peer reviewed by non-agency scientists.
HFRA is not alone. Another enacted bill created exemptions from environmental laws to allow timber harvest projects in a geographically limited area. As part of a massive supplemental appropriations act to address recovery from the September 11, 2001 terrorist attacks, Congress exempted a series of timber harvest projects in the Black Hills of South Dakota from any and all environmental laws; the law specifically stated that the projects were intended to reduce both fire risk and beetle infestations [
15].
Other recent enactments create additional incentives for timber harvests intended to address beetle infestations. Congress permitted state forestry agencies to perform beetle control timber harvest projects on federal lands in Colorado and Utah under what is called “Good Neighbor Authority” [
16]. These state forestry agencies must also implement “similar and complementary” services on state land adjacent to federal land in order to use the authority. Additionally, in the 2008 Farm Bill, Congress expanded subsidies for the production of “renewable biomass” energy to include timber produced from projects intended to reduce or contain disease or insect infestation [
17].
There have been many more recent proposals for additional changes. Congress has considered multiple bills to expand the scope of HFRA. One proposal would require the Forest Service to implement at least one insect and disease control pilot project in at least one subwatershed in every national forest in a state that is “subject” to an insect or disease epidemic [
18,
19,
20,
21,
22,
23,
24]. Congress has also considered many other changes to encourage timber harvesting to control beetle infestations besides expanding HFRA. Some proposals would expand the exemptions to the Forest Service’s Roadless Rule (which prohibits commercial timber projects and road construction in unroaded areas of National Forests) in order to allow more timber projects that are intended to address beetle infestations; some of these projects would be exempt from judicial review [
25,
26,
27].
Congress has considered giving additional benefits under the Clean Air Act for “renewable biomass” produced from timber projects on federal lands, including projects intended to control beetle infestations [
28,
29], giving grants and other subsidies for beetle control timber projects [
30], extending the Good Neighbor Authority to more states [
31,
32,
33], and reducing or eliminating the fee that private timber contractors pay for timber contracts in exchange for agreements to implement restoration work, such as culvert removals, road improvements, or invasive weed removal, if the project provides insect control and other forest management benefits [
26]. Finally, two bills have proposed that designation of additional federal lands as protected wilderness be paired with exemptions of beetle-related timber projects from environmental laws [
34,
35].
Throughout this policy debate, members of Congress and major stakeholders have regularly stated that timber harvest on federal lands is a necessary component of efforts to fight beetle infestations and control outbreaks and that additional flexibility under environmental laws is necessary for agencies to pursue these timber harvest projects [
36,
37,
38,
39,
40,
41].
Likewise, the U.S. Forest Service and other U.S. federal land management agencies have prescribed timber harvests as a necessary component of beetle control. For example, the Forest Service’s Western Bark Beetle Strategy calls for the agency to “reduce the number of trees per acre and create more diverse stand structures to minimize extensive epidemic bark beetle areas” by using thinning and other harvest treatments [
42]. While the Forest Service has applauded HFRA as “very helpful” in addressing beetle outbreaks (U.S. Forest Service, Review of the Forest Service Response: The Bark Beetle Outbreak in Northern Colorado and Southern Wyoming, September 2011), available at [
43], agency leaders do not look favorably upon all legislative proposals to weaken environmental laws to facilitate timber harvest for beetle control. For example, Tom Tidwell, Chief of the Forest Service, criticized recent bipartisan legislation [
25] because it would “shortchange the environmental review process, cut out public engagement and collaboration…and override roadless protections.” (Testimony from House Subcommittee on Public Lands and Environmental Regulation Legislative Hearing on H.R. __, H.R. 1294, H.R. 818, H.R. 1345, H.R. __, and H.R. 1442 available at [
44].
Given the geographic concentration of federal public lands in the West, most of the bills have a specific focus on western states, and were introduced or supported by westerners (
Figure 2). But that is not universally the case. Two of the proposals to expand the scope of HFRA were sponsored by Representative Markey, a Democrat from Massachusetts [
19,
23]. Moreover, support for these bills is bipartisan, showing that the belief that timber harvest can address beetle infestations crosses the political spectrum. Of the 55 total bills, 17 were sponsored by Democrats alone, 21 sponsored by Republicans alone, and 17 had bipartisan sponsors. Markey himself has received very high ratings from the League of Conservation Voters, with a 94% lifetime score from the group.
Figure 2.
Bill sponsorship, co-sponsorship, and applicability by region. (Pacific = CA, OR, W, AK, HI; mountain states = MT, ID, NV, WY, UT, CO, AZ, NM; Midwest = ND, SD, NE, KS, MN, IA, MO, WI, IL, IN, MI, OH; SOUTH = TX, OK, AR, LA, KY, TN, MS, AL, GA, FL, SC, NC, VA, WV; east = ME, NH, VT, MA, NY, RI, CT, NJ, DE, MD, PA).
Figure 2.
Bill sponsorship, co-sponsorship, and applicability by region. (Pacific = CA, OR, W, AK, HI; mountain states = MT, ID, NV, WY, UT, CO, AZ, NM; Midwest = ND, SD, NE, KS, MN, IA, MO, WI, IL, IN, MI, OH; SOUTH = TX, OK, AR, LA, KY, TN, MS, AL, GA, FL, SC, NC, VA, WV; east = ME, NH, VT, MA, NY, RI, CT, NJ, DE, MD, PA).
The 55 bills introduced since 2001 show that many legislators, particularly those from western states, believe that timber harvests are a necessary tool to address beetle infestations. This belief has led to the enactment of laws that reduce compliance burdens under NEPA and other federal environmental laws. There are many more proposals for additional significant changes to federal environmental laws to encourage more timber harvests for beetle control. While “there is certainly a tremendous amount of social and political pressure to ‘do something’ about beetles,” there is also growing concern by many that timber harvests for beetle control are expensive and ineffective and that long-term impacts on forests are unknown [42 citing Ann Merwin, director of policy and government affairs for the Wilderness Society]. The policy debate demonstrates the need to critically examine how well these treatments work and place policy in the context of the best available science.
3. A Mountain Pine Beetle Primer
The mountain pine beetle is native to pine forests in western North America [
45]. During outbreaks, it can kill millions of trees across extensive areas. The ability to cause such widespread mortality has led it to be described as the most destructive forest pest on the continent [
46]. Indeed, economic and aesthetic impacts of outbreaks can be severe. From a manager’s perspective, outbreaks are often perceived as a symptom of poor “forest health”, while ecologists more often view outbreaks as natural ecological processes integral to the maintenance and resilience of the forest. These differing human perceptions have led to conflicting and ambiguous management goals as well as scientific, social, and political conflict.
The mountain pine beetle is polyphagous on pines (
Pinus) [
45]. It attacks not only native pines but also exotic pines used in ornamental landscaping. Within the natural range of the beetle, only
P.
jeffreyi appears to be avoided, likely due to its unusual chemistry [
45]. Pines are well defended and are not easy targets for the beetle. They produce constitutive defenses consisting of resin that can flush the tiny beetles from trees, often drowning them [
47,
48,
49]. Pines also produce induced defenses in the phloem comprised of resin containing elevated concentrations of toxic monoterpenes [
49,
50]. Induced defenses develop in response to attack, and thus, involve a lag time of one or more days to develop and can last for a month or more even when trees are killed [
51].
To contend with a defensive host, the mountain pine beetle has evolved a complex chemical communication system it uses to coordinate a mass attack on a tree [
52]. A female beetle will land, begin to tunnel, and release an aggregation pheromone that attracts conspecifics of both sexes to the tree. Subsequent arrivals release additional pheromone increasing attraction to the tree [
53]. If enough beetles respond, the tree can be overwhelmed in just a few days. As defenses are depleted, the beetles release an anti-aggregation pheromone which repels late arriving beetles and acts to reduce intra-specific competition among brood [
53]. At this point, the tree has reached “a point of no return” [
54]. It will not recover and will slowly die, although it may remain green for nine months or more due to translocation of water to needles by capillary action in the xylem.
The number of beetles needed to kill a tree varies and depends, in part, on the strength of its defenses [
55]. In general, as the strength of defenses increase so does the number of beetles needed. Several factors influence the strength of tree defenses. Trees weakened by drought, disease or damage can be overwhelmed by only a few hundred beetles while very vigorous trees may require many hundreds or even thousands [
56]. Genetics of the host tree also play an important role. Within a tree species, different genotypes result in differing levels of resistance and susceptibility [
57,
58]. Genetic differences are even more pronounced when considering differences in defenses among
Pinus species [
59,
60].
The ability of tree defenses to affect mountain pine beetle success varies by whether the beetle is in endemic (non-outbreak), incipient (building) and eruptive (outbreak) phases. During the endemic phase, when beetle populations are low, host tree defenses are the major constraint in the ability of beetles to kill trees. However, tree defenses become inconsequential once the threshold to the incipient stage has been surpassed [
61]. When numbers are low, beetles attack smaller diameter trees with low defenses. However, once populations rise to the incipient stage, beetles choose larger, healthier, resource-rich trees, despite their superior defenses [
61]. Because larger trees have thicker phloem resources to support larval development, they support greater beetle productivity which results in positive feedback that helps fuel the expansion of the outbreak. Thus, host tree traits (primarily host defenses and diameter class) that determine which trees are killed when populations are low, may be unimportant or even have an opposing effect on beetle success when populations are high [
61].
It is often reported in the press that mountain pine beetle populations are cyclical. This is not the case. The population dynamics of insects that develop cyclical outbreaks are typically dominated by
delayed negative density dependent feedback involving regulation by natural enemies and induced resistance mechanisms [
62]. This type of feedback results in predictable intervals (cycles) between outbreaks although the amplitude of population peaks can vary due to spatiotemporal variation in abiotic conditions. Bark beetle dynamics, instead, are driven by alternations of negative density dependent and positive density dependent feedbacks resulting in sporadic unpredictable population eruptions primarily driven by threshold effects and typically triggered by abiotic factors, particularly climate [
61,
62,
63]. It is critical to distinguish between cyclical and eruptive population dynamics as insects exhibiting these two types of dynamics demand different management and monitoring approaches. In particular, eruptive dynamics are triggered by abiotic factors typically outside the realm of human manipulation.
Mountain pine beetle can remain in non-outbreak phase for very long periods of time, even when forests are composed of suitable age classes of host trees and in a condition often considered to be highly susceptible and “unhealthy”. Outbreaks occur
only when multiple thresholds involving temperature, tree defenses, and brood productivity are surpassed that allow positive feedbacks to amplify across several scales [
2,
64]. While outbreak development is complex, the primary elements that must exist are an abundance of suitable hosts
and a trigger [
63]. Triggers for mountain pine beetle that allow population amplification and subsequent widespread outbreak initiation are warm temperatures and drought, conditions that often co-occur [
65]. There can also be a substantial lag period, even several years, from the initiation of the abiotic factors that trigger an outbreak to when populations actually amplify [
65,
66]. However, once a threshold number of beetles is surpassed, the outbreak becomes self-perpetuating.
While forest conditions alone do not cause outbreaks, certain forest conditions can support larger and more severe outbreaks once they are initiated. Mountain pine beetle attacks only pines (except in rare instances where it “bleeds over” into spruce) [
67], and typically only those larger than ca. 15 cm in diameter [
68]. Therefore, forests comprised mainly of large diameter pine can be at higher risk of widespread mortality when a trigger occurs than are forests comprised of young, small diameter pine or composed of a mix of tree species including non-pines [
68]. Processes that homogenize forest structure and composition such as abnormally widespread stand replacement events (e.g., fires of 1910, Yellowstone 1988) or particular types of forest management (e.g., some timber harvest practices, fire suppression) that alter forest composition and structure over large areas, can contribute substantially to the extent and severity of an outbreak once it is initiated. Processes that result in heterogeneity, such as “normative” wildfires and bark beetle outbreaks, and some land management practices (e.g., restoration treatments focused on restoring a mosaic structure of forest stands of different age classes) tend to reduce outbreak severity and extent by reducing the amount of contiguous susceptible hosts [
68].
Climate acts as a trigger for mountain pine beetle outbreaks for a very good reason. Like all insects, mountain pine beetle is poikilothermic-it cannot regulate its body temperature, and thus, all its metabolic rates and vital functions are dependent upon the temperature of its environment [
69]. As temperatures rise, feeding, activity, development and reproductive rates increase. Importantly, this also means that the length of the mountain pine beetle life cycle is determined by temperature [
69]. Under optimal thermal conditions, development is univoltine (one year). A univoltine cycle allows synchronized emergence of brood adults in mid-late summer, supporting not only mass attacks, but also attacks at a time that allows subsequent offspring to enter winter as cold-hardened larvae [
70,
71]. Cold hardening is a gradual process that occurs as temperatures fall in autumn. Once larvae are cold hardy it can take temperatures as low as −40 °C to kill significant numbers [
72]. However, cold air incursions in fall when beetles are not yet cold hardened or in spring when larvae have lost cold hardening in preparation for transitioning to the adult stage can result in widespread mortality. This can halt an outbreak if subsequent conditions are no longer favorable for the beetle. However, if favorable conditions return, beetle populations rebuild. Importantly, outbreaks require a univoltine life cycle combined with moderate winter temperatures [
73].
In areas where temperatures are too cool to support a univoltine life cycle, a semivoltine (longer than one year) life cycle occurs [
73]. A semivoltine life cycle is maladaptive for the beetle in several ways. First, adaptive seasonality is disrupted, increasing the percentage of brood that enter winter in stages vulnerable to freezing (eggs, pupae and adults). Additionally, mortality increases when beetles must pass through two winters and feed on a food source increasingly depleted in moisture, nutrients, and symbiotic fungi [
74]. Warm periods support not only greater brood production and survival in areas typically suitable for the beetle, but also allow a transition from a semivoltine to a univoltine life cycle in areas otherwise too cool. This increases the spatial extent of suitable habitat and tree mortality. Thus, abnormally warm periods can vastly increase the total area suitable for the beetle and play a major contribution to the synchronicity and coalescence of outbreaks across regions [
2,
65].
Drought can also play an important role in outbreak initiation. Host tree defense mechanisms are compromised during drought allowing beetles to more easily attack trees [
2,
75]. Tree defenses are major constraints when beetles are in non-outbreak phase. However, drought-weakened trees can support population amplification until a point where stand level densities surpass a critical threshold. Once this threshold is passed, tree defenses lose their importance in regulating beetle populations [
61]. Very importantly, drought stresses large numbers of trees at a regional scale. This results in large numbers of trees that are easier for the beetles to kill, further supporting outbreak intensification [
65,
76].
Recent studies have found that drought occurring years or even decades before the outbreak can influence outbreak initiation. Furthermore, prolonged drought stress appears to pre-condition trees to be more susceptible, an effect that can continue for years after normal precipitation has returned [
58,
65,
77]. There also appears to be a genetic component to tree sensitivity to drought, and subsequently, susceptibility to beetles. In two studies, one conducted in whitebark pine and the other in ponderosa pine, differences in growth of surviving trees and trees killed by beetles over the last century suggest that adaptive differences to changes in climate exist. In the whitebark pine study, the trees studied were co-dominants and not significantly different in diameter age or mean growth over their lifetimes [
58]. However, trees that were killed exhibited faster rates of growth in the first half of the century suggesting they were better adapted to the cooler wetter conditions of that period. The surviving trees had greater growth in the latter half of the century when conditions were warmer and drier. Millar
et al. [
58]) suggested that the beetle-caused tree mortality in the stands they studied resulted in a strong natural selection event that removed trees less fit under our current climate while leaving those more well-suited.
Likewise, Knapp
et al. [
77] found genotypes of ponderosa pine that were slow-growing in the two to three decades prior to the outbreak were much more vulnerable to beetle infestation than those that were fast-growing, again suggesting the beetle may act as a selective agent shifting genetic structures in stands over time to those most suited to prevailing climatic conditions. In lodgepole pine, trees of similar age and diameter growing intermixed in the same stand and under the same conditions exhibited different levels of sapwood moisture that were highly correlated with susceptibility to beetle attack [
74] hinting at genetic differences in water efficiency. Those with lower sapwood moisture were attacked and killed by the beetle while those with higher sapwood moisture were not [
74].
While mountain pine beetle has developed outbreaks for millennia, the current outbreak is far outside the historic norm [
2,
78]. The unprecedented size and severity of this outbreak is due to a combination of increasingly favorable climate for the beetle and forest conditions. Warming trends have supported the development of a univoltine cycle in many areas that previously were too cool and have resulted in greater beetle productivity and survival [
79]. This has led to massive tree mortality, not only in areas previously favorable for the beetle, but also in areas previously suboptimal or unusable. Warmer temperatures and high population levels have also supported expansions of the beetle’s range hundreds of kilometers further north in British Columbia and eastward across Alberta [
80,
81,
82]. In these new locations, the beetle is infesting naïve hosts including (in the eastern expansion) a novel species, jack pine [
80,
82]. These naïve hosts exhibit lower defenses to beetle attack [
83] as well as similar chemical compositions to natural hosts [
84] promoting establishment. Predictions are that the beetle will continue to move across the continent through the boreal forest and finally into eastern pine forests [
78].
Warming has also allowed the beetle to move higher in elevation where it is devastating whitebark pine, a tree that is foundational to the western North American subalpine ecosystem and that was previously protected from the beetle by cold [
73,
85]. Movement into the subalpine has been supported by overall warmer temperatures and milder winters allowing the beetle to switch from a semivoltine to a univoltine life cycle while simultaneously reducing winter mortality [
85,
86,
87]. The resulting mortality to whitebark pine in many areas, particularly the greater Yellowstone Ecosystem, has been so severe the tree is now proposed for listing as an endangered species [
88]. The tree is already listed as an endangered species in Canada due to the combined effects of mountain pine beetle and white pine blister rust [
89].
4. Mountain Pine Beetle Outbreak Suppression
Treatments used to mitigate the effects of mountain pine beetle are grouped into three broad categories. Treatments that strive to reduce or eliminate beetle populations are termed direct controls [
90]. Treatments aimed at increasing tree vigor and altering stand conditions to be less favorable for beetles are called indirect controls [
90,
91]. Prophylactic treatments aim to protect high value individual trees or stands of trees from infestation. Salvage, while often included in beetle management programs does not actually reduce or impact beetle populations-it is the removal of dead trees for economic or other reasons and often involves removal of trees that are already ‘empty’ of beetles and thus has no impact on beetle population size. Because our focus is on how well science supports the use of timber harvests (including tree felling and destruction of trees in place) to reduce or suppress bark beetle outbreaks, we will focus primarily on direct and indirect controls concentrating on these treatments.
Direct control includes sanitation treatments such as removing single trees or small patches of trees that are infested with the insect, clearcutting (also called block harvesting) and prescribed burning of infested trees, as well as fell and burn, trap trees, debarking, and application of insecticides or toxins such as MSMA (monosodium methanearsonate). Sanitation cuts attempt to remove most or all beetles in an area by removing infested trees before the beetles developing within them can emerge and disperse [
90,
92]. Prescribed burns, fell and burn, debarking, and toxin applications attempt to destroy beetles in infested trees on-site. Trap trees are trees that are baited with attractant pheromone baits in an attempt to draw beetles into specific areas where they are concentrated into the baited trees which are subsequently taken to the mill or destroyed. Each of these methods relies on killing as many beetles as possible in order to lower beetle population thresholds below which they can maintain outbreak dynamics.
Indirect controls are primarily silvicultural in nature. The main treatment used for mountain pine beetle is thinning. Thinning is thought to act by reducing inter-tree competition for water, nutrients, and light, enhancing greater tree vigor, and thus defenses against the beetle [
93]. Thinning treatments are also thought to reduce successful beetle attacks by altering microsite conditions by increasing temperatures on bark surfaces on bark in summer and decreasing them in winter, as well as disrupting beetle communication by increasing wind flow [
94,
95]. A new treatment recommended for reducing bark beetle infestation is “daylighting” which involves removing trees and shrubs from around trees that are to be protected to increase light on the tree’s stems to disrupt beetle colonization. Other silvicultural treatments include removal of beetle-suitable hosts (mature trees and old growth) and conversion of stands from species preferred by beetles (pines) to species that are not hosts or converting stands that are primarily pine to a mixed species composition [
91,
92]. Most of these approaches involve, completely or partially, the use of timber harvests.
4.1. Efficacy of Direct Controls
Direct control treatments are extremely expensive in time, effort and resources. They address only one aspect of an outbreak which is the amount of beetles present in a stand or area. Because they do not address the underlying conditions that support an outbreak (climate, tree condition/stress) their effects are considered a holding action until conditions shift to being less favorable for the beetle [
92]. Direct control efforts must be maintained at a high level on an annual basis until the outbreak ceases [
3,
90,
96]. It is highly controversial whether direct controls are effective in reducing tree mortality in the short-term, and if they can be effective in halting or suppressing outbreaks in the long-term.
One of the biggest problems in assessing the utility of direct controls is a general lack of monitoring or
post hoc assessments of the outcomes of implementing these practices. Despite decades of direct control and large-scale implementation of these practices, few rigorous studies on its efficacy have been done and there remains no agreement among scientists or foresters regarding its ability to reduce beetle populations or losses of trees. Studies conducted prior to the current outbreak have variously concluded that direct treatments may merely act to delay infestation of susceptible stands [
97], or that if used correctly, can be effective [
98,
99]. Many studies found that while some treatments slowed the rate of infestation, overall, they had little to no impact on mountain pine beetle populations [
97,
100,
101,
102,
103,
104].
The US and Canadian governments have spent hundreds of millions of dollars in direct control efforts to address the current outbreak. However, assessments of the efficacy of these efforts are nearly non-existent and only a few studies on assessments have been published. The few that have been published are reviewed here. Although much of our review addresses how well science supports US policy, we use primarily studies conducted in Canada as few studies have been published on direct control measures during the current outbreak in the US.
Nelson
et al. [
3] evaluated the efficacy of five direct control treatments in British Columbia roughly midpoint in the portion of the current outbreak as it progressed in that province. The assessment was extremely short-term and looked only at the response of beetles in the year immediately post-treatment. However, it provides one of the very few broadscale assessments ever conducted of the efficacy of direct controls during an outbreak. The treatments assessed were applications of MSMA, trap trees, fell and burn, and clearcutting. The study was split into three geographic regions to account for potential sources of variability due to location and different background levels of beetles. The northern-most region was at the margin of the beetles range (expansion zone) and possessed relatively low beetle populations, while the central and southern regions had higher beetle populations and were known to have supported high beetle populations historically. The study found that, overall, sites receiving MSMA treatments exhibited higher infestation intensities (a metric based on kernel density estimators) than randomly selected untreated sites with similar characteristics. This was particularly pronounced in the southern region. Results for trap tree treatments showed substantial variability within and among regions. A reduced infestation rate in response to treatment was observed more often than not in the northern area where beetle pressure was low. However, in the central and southern regions where beetle pressure was higher, the range of infestation intensities was similar for treated and untreated sites although a larger number of comparisons found higher infestation intensities in the treated sites. The overall conclusion was that MSMA and trap tree treatments may be effective, but not reliably, and only when beetle pressure is low and environmental conditions are not highly favorable for the beetle.
Results for fell and burn were also variable. In the northern region, intensities were lower overall in treated vs. untreated sites. However, in the central area, treated areas tended to have greater infestation intensities. In the southern area, no discernible effect of treatment was seen. Therefore, like with trap trees, fell and burn appeared to sometimes be effective, but only when populations of beetles were low, and became increasingly unreliable as beetle pressure increased and the infestation moved into outbreak phase.
Removal of trees in patches was studied only in the central region. No significant effect of treatment was detected. Clearcuts were assessed in the central and southern areas and were found to lead to a significant reduction in infestation intensity. In almost all cases, infestation intensities were lower in treated vs. untreated areas. However, this was likely due to the removal of all living trees (potential subsequent hosts) that survived the beetle as well as the infested trees. The overall conclusion of the study was that mitigation treatments are effective when populations are low to moderate and if infested trees can be kept to 2.5 or fewer per hectare. Efficacy was also recognized to be contingent upon a high level of accuracy in detecting infested trees and wide-scale and continuous implementation of treatments. However, with only one year of data, the authors could not predict how long treatments would need to be sustained to remain effective, nor what effect beetle pressure from surrounding areas might have on the subsequent fate of treated stands. No follow up study has been published to report how these treatments fared as the outbreak progressed.
Fell and burn has been a stalwart component of the direct control efforts against mountain pine beetle in Canada during the current outbreak, particularly on the advancing front as the beetle expands its range eastward. Coggins
et al. [
105] examined the efficacy of fell and burn treatments to “stabilize” such infestations (
i.
e., prevent expansion) using field plot data from sites at the expanding edge of the mountain pine beetle infestation in 2008 in eastern British Columbia and western Alberta. The authors used multiple modeling scenarios along with ground data to demonstrate how infestations may develop with and without mitigation, and to predict how long mitigation may need to be maintained to be effective given different levels of infestation and detection accuracy. They found non-mitigated plots experienced more tree mortality due to the beetle and that infestations in these plots expanded more rapidly. The higher the expansion factor (means rate of increase, e.g., 2 would indicate a doubling of the population each year) the greater the detection accuracy that was required to maintain a static population. When a beetle population had an expansion factor of 5.1 (high), an 80% detection rate was required, whereas with a population with an expansion factor of 1.1 (very low), the minimum detection rate could be as low as 10% and still be effective. The authors also modeled how long it would take to achieve population stability given different levels of infestation. On average, across their stands, with a 70% detection accuracy rate, mitigation would take 11 years, at 80% 6 years, and at 90% 3 years. The actual mean mitigation efficiency at their sites was found to be 43%, a level at which no control could occur. They concluded that the stabilization of mountain pine beetle populations is possible, but only with a much higher detection accuracy than commonly occurs coupled with an intense level of mitigation maintained potentially over a very long timeframe.
Wulder
et al. [
96] looked at the effectiveness of sustained mitigation on slowing the beetle’s expansion in western Canada. The results were difficult to assess because of the unevenness of application of mitigation treatments (for example, in one year only 68% of sites slated for mitigation were treated) and differences in background beetle populations. However, such a situation is typical and thus may represent the reality of many on-the-ground direct control efforts. One site where little mitigation was conducted early on, did exhibit a strong increase in tree mortality due to the beetle that declined once extensive mitigation efforts were implemented. However, overall, the conclusion was that mitigation must be extensive and continuous to work and may only be effective when populations are low to moderate.
Trzcinski and Reid [
104] studied the trajectory of beetle populations in treated and untreated zones in Banff National Park from 1997–2004. The Park used a combination of pheromone-baited trees and fell and burn to remove as many beetles as possible from treatment zones—they also conducted prescribed burns to reduce beetle numbers and lodgepole pine hosts. The area colonized by the beetle increased rapidly over this time period in both the untreated and treated zones. After four years of treatment, control measures did not reduce the area affected by beetles and infestations continued to expand at a similar rate in both zones. The authors estimated that between 45% and 79% of infested trees had failed to be detected in the treated areas. This equated to
only 0.7–3.7 infested trees remaining per thousand ha yet still was sufficient to support subsequent rapid beetle population growth.
A general consensus of these studies is that suppression of a beetle outbreak would require massive sustained efforts with extremely high detection rates to succeed. It has been estimated that 97.5% of beetles in an area must be killed to merely stabilize a mountain pine beetle population [
90]. Even a small increase in survival above this value can allow a substantial increase in population size. For example, if mortality drops to 95%, this would allow a population to
double in size annually. If the goal is not just to stabilize a population, but to reduce it, mortality of beetles would need to be higher than 97.5%, a goal that is highly unlikely given the vast areas that would need to be treated on a continual basis when conditions are favorable for outbreak development. Even if 100% removal of infested trees from an area was feasible, the migration of beetles into treated stands from surrounding areas allows reestablishment and subsequent tree mortality further decreasing the potential for effective direct control.
The on-the-ground reality is that direct control efforts typically fall far below the levels needed to stabilize, let alone control, mountain pine beetle populations. In the above cited studies, rates of detection in mitigated stands ranged from 45%–79%. These situations are not unusual. Direct control treatments are laborious, extremely costly and time consuming, and require high levels of training. Logistical difficulties, including proper seasonal timing, access, inclement weather, and lack of trained personnel, increase the odds that they will not be effective. The high financial cost of such efforts coupled with a volatile market for sawtimber, pulp and pellets further complicates the use of direct controls. Importantly, outbreak development is extremely swift and the amount of mitigation required can rapidly outstrip the ability of managers to respond.
During an outbreak the number of trees killed annually is often in the millions and infestations may cover hundreds of thousands of hectares [
90]. Carroll
et al. [
90] presents an example of the degree of mitigation that would be required for an outbreak that covers 300,000 hectares with a rate of increase of 2 (the population doubles in one year-a conservative rate for an outbreak). In this case, 150,000 ha of infested trees would need to be removed each year just to maintain a
static beetle population–this would still allow tree mortality to occur for many years, potentially until most or all mature trees were killed. In reality, such a high level of detection and mitigation is impossible. Given that the goal of direct management is to reduce populations and protect trees, the effort that would be needed to actually reduce such a high beetle population would require an even more unlikely effort.
Studies in other bark beetle systems also have found that a high degree of detection accuracy and intensity of mitigation is required to reduce beetle numbers. Fahse and Heurich [
106] found that control of
Ips typographus, a less aggressive European bark beetle, requires a detection and removal level of around 80% to be effective. They concluded that direct control efforts are useless and should be dropped if survival probabilities of the beetle after treatment are above 20%–30%. This estimate is in line with those developed in studies on mountain pine beetle in North America and highlights the challenge the high reproductive capacity of bark beetles poses when conditions are favorable for outbreak development.
It is not just the difficulty of dealing with the extreme spatial extent of outbreaks and the challenge of detection and treatment that makes the efficacy of direct control measures unlikely, but also the time frame over which direct controls must be maintained. Carroll
et al. [
90] estimated that to control a population involving 10,000 infested trees with expansion factor of 2 (conservative) and with a detection and removal rate of 80% (difficult), it would take at least 10 years of annual treatment to reduce the population to a single tree. If the population was tripling or quadrupling, a more likely scenario during an outbreak, it would take 18 or 41 years, respectively. A costly, intensive detection and treatment program lasting that long, assuming sufficient trees even remained to be infested, would be unlikely [
90].
Carroll
et al. [
90] emphasized three requirements for direct controls to be effective in treating
individual infestations: infestations must be detected early, efforts must be applied quickly and intensively, and control programs must be maintained continuously until the desired population level is achieved. Because of the cost and intensity of treating individual infestations, the US Forest Service recommends that direct control measures only be applied to higher value stands [
92]. However, treating individual infestations or stands during outbreaks can fail because of the regional nature of outbreaks. Outbreaks are driven by abiotic factors that affect entire regions (warm temperatures and drought). Thus, they consist of many infestations that occur synchronously across a very large area. These infestations often coalesce to form vast expanses where beetle populations are extremely high. These characteristics mean that many stand level efforts are prone to failure due to high beetle pressure and migration into treated areas by beetles from surrounding areas. Given that treating entire regions is impossible, and that many treatments are not in line with other land use objectives, direct control efforts may in some cases, not be worth their costs. The consensus of studies and retrospectives over the course of several outbreaks is that even after millions of dollars and massive efforts, suppression using direct controls has never been effectively achieved, and at best, the rate of mortality to trees was reduced only marginally [
90,
101,
102,
105]
4.2. Efficacy of Indirect Controls
Thinning is the primary indirect control measure used to manage the mountain pine beetle. It is generally considered a preemptive measure to be implemented prior to the initiation of a mountain pine beetle outbreak, although it is increasingly employed to reduce damage by the insect during outbreaks. It is often touted as a global panacea for problems with pest bark beetles. One type of thinning is even termed “beetle-proofing” [
107], further reinforcing the view among managers, the public, and policy makers, that this approach is failsafe. While overall, evidence suggests that thinning can reduce mortality of trees due to mountain pine beetle, the outcome is frequently more variable than is often recognized or reported. This is particularly true when outbreak populations are involved.
So how exactly does thinning work, and how well does thinning hold up under outbreak conditions? Surprisingly, the mechanism(s) by which thinning affects beetle activity in forest stands is still not well understood. Two, non-mutually exclusive, lines of thought exist. One hypothesis is that thinning increases tree vigor, and thus tree defenses, by reducing competition among trees for light, nutrients and water [
93,
108]. Intuitively, this makes sense, and indeed, immediate impacts of thinning on reducing water stress have been seen [
109]. Likewise, increases in growth and photosynthetic rates also have been observed post-thinning, albeit after a lag period of one or more years [
107,
109,
110]. Increases in growth and vigor are predicted to increase the amount of energy that trees allocate to defense, leading to greater resistance to beetle attack through increased resin and monoterpene production. In fact, the initial impetus for the use of thinning to manage mountain pine beetle came from an early study that found that ponderosa pines in thinned stands produced more defensive resin [
93]. However, subsequent studies have reported a variety of responses in resin production as well as growth in response to thinning. For example, Zausen
et al. [
111] found that ponderosa pines in the thinned stands exhibited lower water stress but also produced less resin. This, along with the thicker phloem (greater food resources) found in trees in thinned stands, indicates they might be not only more susceptible to attack but also a more productive resource for beetles. In contrast, McDowell
et al. [
112] found greater resin flow in thinned stands. Both studies were conducted in southwestern US ponderosa pine forests indicating that the variable responses observed were not due to major regional differences in hosts. Six and Skov [
113], in a study conducted in ponderosa pine in the northern Rocky Mountains looking at effects of thinning and burning treatments, found that resin flow was highest in trees in burn treatments, intermediate in controls, and lowest in thinned treatments. Raffa and Berryman [
114] tracked the fate of trees over time during an outbreak and found no significant difference between resin flow for lodgepole pines that survived attack vs those killed by the beetle.
A number of studies have noted a reduction in beetle caused-mortality of trees immediately after thinning treatments were applied and before trees had time to respond physiologically to lower stocking densities. This timing suggests that the effects of thinning may have more to do with microsite conditions than to changes in tree vigor or defense. These observations led to the second line of reasoning that thinning affects beetle activity through changes in microsite conditions.
Thinning alters temperature, light intensity and wind speed within a forest stand; factors that can have major effects on insect behavior and success. A number of studies have tried to describe how shifts in microsite conditions due to thinning may influence mountain pine beetle activity. Bartos and Amman [
94] investigated how incident solar radiation, wind speed, wind direction and temperature were altered by thinning and whether changes affected beetle responses to stands. They did not conduct statistical analyses on their data; however, there was a trend for south sides of trees in thinned stands to be warmer, and ambient temperatures in thinned stands to be overall warmer during parts of the day. Incident solar radiation was higher in the thinned stand. It is not known if bark temperature affects beetle attack behavior, although higher temperatures on south sides of trees in thinned stands have been suggested to be deleterious to beetle development [
94]. However, this speculation does not account for differences in local environmental conditions. For example, at cool sites, increased temperatures and insolation could ostensibly support better beetle development by increasing thermal units sufficiently to support a univoltine life cycle.
Light intensity affects the flight behavior of mountain pine beetles [
115]. However, if and how different levels of light in treated and untreated stands affect beetle attack behavior is unclear. It has been hypothesized that a reduced propensity for flight in darker stands might concentrate beetles for mass attack, while beetles may be more likely to disperse in open stands [
116].
The hypothesis that light has a strong effect on mountain pine beetle behavior, particularly in reducing attacks, has led to a new treatment called daylighting. This approach is currently being implemented on a broad scale by federal and western state agencies. Daylighting involves removing trees and vegetation from around trees that are targeted for retention and is believed to work by repelling beetles from the boles of trees by increasing light and solar radiation [
117]. While widely recommended, the efficacy of this treatment is unknown; there are no published studies on its effects on bark beetles.
Changes in wind speed and direction due to thinning have also been suggested to alter beetle behavior by disrupting beetle communication via disruption of pheromone communication. Schmid
et al. [
118] found no statistically significant differences in horizontal and vertical wind patterns in thinned and unthinned stands. However, disruption of pheromone plumes by greater wind speeds may affect communication and thus the potential for successful attacks [
95]. Ultimately, we need to look at actual population dynamics of beetles in treated and untreated stands to understand if microsite effects hold under epidemic conditions. MacQuarrie and Cooke [
119] found that, under outbreak conditions, mountain pine beetle populations exhibited density-dependent dynamics and that thinning did not change the epidemic equilibrium. In this study, population growth curves did not exhibit responses that would be expected if microsite conditions played a role in beetle behavior. It is evident that more research is needed to understand how these effects ultimately influence tree mortality due to beetle attack.
While we may not have a complete understanding of how thinning works, it is clear that this practice can have a significant effect on mountain pine beetle infestations. Several studies have reported striking differences in mortality to trees caused by beetles in thinned
vs. un-thinned forests (reviewed in [
120,
121]). In contrast, only a small number of studies have reported failures. However, the disparity in numbers of successes and failures must be placed within a broader context. Many studies assessing the efficacy of thinning have been conducted under non-outbreak conditions. Their results do not reflect how stands perform during an outbreak. Additionally, failures are often not reported, dismissed as a result of poor management ‘next door’ or targeted for management without evaluation. This is unfortunate because thinned stands that fail may have particular characteristics that could inform a better understanding and application of this approach.
Studies conducted during outbreaks indicate that thinning can fail to protect stands. In Colorado, thinning treatments in lodgepole pine implemented in response to the outbreak that began in the 90s often only slowed the spread. Klenner and Arsenault [
122] reported high levels of mortality due to the mountain pine beetle across a wide range of stands densities in lodgepole pine in British Columbia during the same outbreak. They noted that silvicultural treatments were largely ineffective in reducing damage to the beetle. Preisler and Mitchell [
123] found that once beetles invaded a thinned stand the probability of trees being killed there can be greater than in unthinned stands and that larger spacings between trees in thinned stands did not reduce the likelihood of more trees being attacked. Whitehead and Russo [
107] reported on the performance of ‘beetle-proofed’ (stands thinned to an even spacing of about 4–5 m between mature trees) and un-thinned stands in five areas in western Canada during approximately the same time period. These treatments were successful in protecting stands when they were combined with intensive direct control measures (removal of infested trees) in the areas surrounding the thinned units, but failed if units were exposed to beetle pressure from the neighboring area—a situation most thinned stands experience during an outbreak.
Unfortunately, long-term replicated studies monitoring beetle responses to thinned forests from non-outbreak to outbreak to post-outbreak phase are virtually non-existent. One large fully-replicated long-term study was initiated in 1999 under non-outbreak conditions and continues to track beetle activity [
113]. In this study, mountain pine beetle was low in all treatments in the period leading up to the outbreak, but increased in some controls and burn treatment replicates as the outbreak developed. Although more trees were killed overall in control units during the outbreak, all controls still retained a greater number of residual mature trees than did thinned stands as they entered the post-outbreak phase [
124].
Two factors contribute substantially to our inability to assess how well thinning performs under outbreak conditions. One, very few thinning treatments are monitored after implementation over either the short- or the long-term. Thus, for the vast majority of stands that have been treated, we have no data on how well they perform once an outbreak of the insect initiates (or for that matter, even under non-outbreak conditions). Second, stands that become infested, thinned or otherwise, are often targeted for intensive suppressive management and are cut without assessment or data collection. This even includes studies and sites that are intended to inform management. For example, at the sites studied by Whitehead and Russo [
107], infested trees were being removed from the study sites even before data collection for their study could be completed. The long-term study discussed previously [
113,
124] is under continual pressure to be logged to remove beetle kill even though the site lies within an experimental forest designated specifically for studies assessing the outcomes of forest management.
5. What are the Goals?
When we manage forests, we do so in an attempt to achieve one or more outcomes, preferably with minimal negative effects on non-target resources. To be effective, management must have explicit and appropriate goals as well as clear metrics for success. Ideally, management is monitored to assess how well it meets its goals, where it falls short, and whether and how it can be improved. This approach is called adaptive management and implies an iterative process through time whereby we learn from the outcomes of our actions and base future actions on improving performance [
125].
Not only outcomes, but the costs of management must be factored into decision making. These include direct financial costs as well as the less tangible (at least in dollar values) effects on ecosystem services and functions. By considering the full cost of management along with benefits as verified through monitoring and evaluation, we lessen the risk of failure, financial waste, and unnecessary negative environmental impacts.
In assessing how well we meet goals when managing for mountain pine beetle, we must ask several questions. Do our management practices actually control the beetle during outbreaks? Do the outcomes justify the financial and ecological costs? And, what long-term impacts do these treatments have on forests and their ability to adapt to climate change? These questions are difficult to answer. Only limited data are available on the short-term efficacy of direct and indirect controls, and information on long-term effects is virtually nonexistent. The results of short-term assessments can be difficult to interpret. For example, often only the proportion or numbers of trees killed by beetles post-treatment are reported. This does not allow a complete evaluation of outcomes. A study may report that 75% of trees in controls are killed by the beetle, whereas only 10% are killed in thinned stands. At first glance, this appears to be a resounding success in saving trees. However, if we approach this situation from a pretreatment perspective, our interpretation of success may change. In this example, 400 mature trees existed in each plot prior to treatment. After treatment, 100 mature trees remain in the thinned plots (300 trees have been removed by thinning). Doing the math, we find that once the beetles have run their course, more residual living trees (100) actually remain in the control plot than in the thinned plot (90) and, in fact, humans have contributed more to tree mortality than have the beetles. In the case of silvicultural intervention, humans typically must expend considerable effort and expense. They also choose the trees that remain, and thus the structure and composition of the remaining forest. This may result in very different trajectories for residual forests as discussed below.
When we include pre-treatment conditions as well as post-treatment responses we can assess the management efficacy from a more informed position. For instance, in a retrospective study investigating the effects of management on spruce beetle, researchers found that post-infestation, untreated stands had more live spruce trees and greater basal areas. When comparing only residual large spruce, final densities in both stand types were similar [
126]. Six [
124] found higher numbers of mature living trees remained in control stands of ponderosa pine than in thinned stands post-mountain pine beetle outbreak. In a study in Canada focusing on stocking density of living lodgepole pine post-outbreak, the authors found that, even in hard hit stands, stocking density in post-outbreak unmanaged stands was sufficient to maintain desired levels of productivity [
127]. Klutsch
et al. [
128] in a study conducted in lodgepole pine forests in Colorado, found greater mortality of trees due to the beetle in more densely stocked stands. However, while the density and basal area of lodgepole pine in infested plots declined 62% and 71%, respectively, the number of trees that remained and their size distribution post-outbreak indicated that lodgepole pine would remain the dominant overstory tree. In another study in Colorado, the beetle killed 60%–92% of overstory lodgepole pine. However, these stands retained residual overstory trees as well as advance regeneration. Furthermore, untreated stands were predicted to return to pre-outbreak stocking levels approximately 25 years sooner than treated stands [
129]. Other studies have found similar results for both lodgepole and ponderosa pine [
130,
131,
132,
133,
134]. These studies highlight a seldom considered impact of mountain pine beetle- that it can act as a natural thinning agent and seldom removes all mature trees during outbreaks. These effects are an important part of the ecological role that the beetle plays in western pine forests [
135].
It is also important to recognize there can be significant differences in long-term forest trajectories for stands thinned by beetles
vs. those thinned by humans. When humans thin, they select for particular size classes, often favoring the retention of larger, older trees, selecting toward one desired tree species, and often ‘thinning from below’ which removes advanced regeneration (small trees) [
123,
136]. Thinning prescriptions also typically call for relatively even spacing between residual trees [
92,
107,
121]. Mountain pine beetle, on the other hand, often selects the largest trees during outbreaks (with exceptions; [
121,
123,
131]) which can lower the mean diameter of the stand [
128]. However, beetles often leave sufficient numbers of large diameter trees to maintain a dominant overstory of pine. Beetles also leave substantial amounts of advanced regeneration to replace the mature trees that arekilled [
121,
129]. Spacing among trees after an outbreak is uneven, resulting in a clumpy network of living trees [
129]. Patches where all trees are killed are seldom extensive and add to a mosaic structure as forests recover post-outbreak. Heterogeneous stand and mosaic forest structures are more typical of natural conditions and can support greater biodiversity and resilience against fire and subsequent beetle outbreaks [
137,
138,
139]. In contrast, intensive thinning treatments by humans typically favors the retention of mature pines. Over time, these pine-dominated stands grow, they are predicted to have increased susceptibility and potential for tree mortality from future mountain pine beetle outbreaks [
123,
136].
Very importantly, the beetle exercises selectivity in the trees it kills. While extremely high numbers may override this selectivity, evidence is accumulating that, even under outbreak conditions, beetles choose trees that have particular qualities. Beetles commonly select trees for attack that exhibit lower growth rates, defenses, and higher water stress [
58,
74,
77]. While these factors can be influenced both locally and regionally by site conditions and climate, much of the variation in these properties within individual stands that affect bark beetle choice likely has a genetic basis. Outbreaks can result in strong natural selection against trees with phenotypes (and likely genotypes) favorable for the beetle and for those that possess unfavorable qualities [
58,
77]. However, when humans thin forests, trees are removed according to size, species, and density, without consideration of genetics. Thus, trees best adapted to surviving beetle outbreaks are as likely to be removed as those that are not.
When humans thin forests, they typically manage for resistance and resilience, rather than adaptation which involves genetic change. It is very important to distinguish between resistance, resilience, and adaptation, as each have different goals and operate on different temporal scales [
140]. Resistance is a short-term holding action where we try to maintain an existing state. Approaches focusing on resistance often require massive interventions and increasing physical and financial investments over time. Such approaches may set forests up for future outbreaks [
136] and even catastrophic failure as they surpass thresholds in a warming climate [
140]. In contrast, practices that promote resilience attempt to allow forests the ability to adjust to gradual changes related to climate change and to recover after disturbance. However, like resistance, resilience is not a long-term solution. In the long term, forests must be able to adapt to change. Adaptation involves genetic change driven by natural selection. Currently, much of forest management, including bark beetle management, focuses on resistance and resilience, mainly through direct and indirect management, respectively. However, neither approach allows for true adaptation. For long term continuity of our forests, it will be imperative to begin to incorporate this aspect of management into our approaches.
We also need to reassess the ecological role of bark beetles, including the mountain pine beetle, in our forest ecosystems. As has been well demonstrated by a century of fire suppression, the dampening or suppression of natural disturbance can alter forest trajectories in undesirable ways, many of which can be irreversible. Although beetle outbreaks, like fire, can have negative impacts on timber values and aesthetics, their natural role in many forest ecosystems is seldom considered and beetle suppression is often perceived as something that must be conducted at all costs. However, as with fire, suppression of beetles over the long term may alter forests in ways that are not desirable or sustainable. While intensive management for bark beetle suppression is called for in some situations such as in the wildland urban interface, it may not be appropriate in many other areas where natural processes including natural selection are needed to maintain a dynamic and functional forest.
6. What are the Needs in Research and Monitoring?
There is clearly a need to better understand how well management programs aimed at reducing mountain pine beetle work, particularly under outbreak conditions, and what impacts these treatments have on forests in both the short and long term.
Perhaps the biggest area of need is in monitoring. Monitoring is essential to understanding whether mountain pine beetle treatments work, and in which contexts, but as noted above there has been all too little long-term monitoring of the effectiveness of various treatment efforts. This is a failing among both agencies and researchers. Agencies often do not have strong incentives to conduct long-term monitoring: Monitoring is costly; external and internal political pressures focus on short time frames; and monitoring may produce information that conflicts with agency goals or missions. It is also difficult to get strong public pressure to force agencies to conduct the necessary monitoring, particularly when the public has been led to believe that outbreaks are strictly the result of a lack of management. Even for scientists, long-term monitoring projects are not encouraged by short-term funding time frames and professional incentives or norms; monitoring is often not viewed as “real” science, and the long-time frames required for monitoring to result in significant gains in information are often longer than the time frames used for professional advancement (e.g., completion of a dissertation, tenure review) [
141].
Addressing the shortage of monitoring for beetle treatments may, therefore, require far more than simply trying to provide additional funds (even assuming additional funding is politically feasible). Scientists can help by encouraging and rewarding projects that involve long-term monitoring. Agencies might try to establish units that are focused specifically on monitoring forest health, insulating monitoring projects from adverse political or bureaucratic pressure [
141]. Finally, tools that might reduce the cost of monitoring significantly, such as retrospective studies and remote sensing, should be used to complement traditional monitoring and decrease its costs.
Monitoring is all the more essential if forest health management in general, and beetle treatments in particular, are truly to be guided by adaptive management. The high levels of uncertainty and dynamism associated with beetle infestations and the effectiveness of beetle treatments make adaptive management a very appealing tool to reduce uncertainty and allow us to respond to changes in global climate and forest ecosystems. But adaptive management requires monitoring to be successful [
141], monitoring that is currently not occurring even as agencies conduct massive beetle treatments and propose to pursue even more.
There is also a real need to increase research on management efficacy and, in particular, how our approaches affect forest adaptation including genetic responses of trees to climate and the role in bark beetle selectivity and fitness. With a changing climate we will need to develop new approaches rather than trying to force old methods of questionable efficacy onto new conditions.
Unfortunately, most funding for research on bark beetles is very short-term, sometimes even as short as on an annual cycle, and thus cannot hope to address the complexities of beetle responses to treatments. Funding cuts to research personnel, particularly in agencies like the US Forest Service, have exacerbated this problem exactly at the time when the need for rigorous research is increasing at a rapid pace. The US Forest Service has recognized that long-term planning must include explicit goals to increase forest resilience and adaptation to disturbance, including outbreaks of the mountain pine beetle. However, with extreme cuts to budgets and personnel, they are highly constrained to meet these needs at this time. Likewise, cuts in federal funding to agencies such as United States Department of Agriculture and the National Science Foundation concurrently reduce the ability of academic researchers to address these problems.